Abstract
In a 5-year retrospective study of dogs presenting to the Veterinary Medical Teaching Hospital at the University of California, Davis, there were 31 histologic diagnoses of valvular endocarditis. By polymerase chain reaction (PCR) amplification of embedded valvular tissue, Bartonella organisms were exclusively associated with 6 out of 31 cases (19%). Confirmed Bartonella cases involved the aortic valve alone (five out of six) or in combination with the mitral valve (one of six). Microscopic features of Bartonella endocarditis were compared with valves from non-Bartonella endocarditis and with valvular change unrelated to infectious agents (endocardiosis). Features of Bartonella endocarditis included a combination of fibrosis, mineralization, endothelial proliferation, and neovascularization with variable inflammation. None of these features is specific; however, the combination is distinct both from endocarditis caused by culturable bacteria and from endocardiosis. Ultrastructural analyses revealed both extracellular and intraendothelial bacteria. Clinical history, serology, and PCR are currently necessary to establish an etiologic diagnosis of Bartonella endocarditis.
Keywords
Bartonella species are intra- and extracellular, pleomorphic, gram-negative alpha proteobacteria that are the causative agents of Orroya fever, Trench fever, and Cat Scratch disease. Currently, the family contains at least 19 members; many of which are emerging human and canine pathogens. 3, 4, 7, 8 Several members of the Bartonella family have become important causes of blood culture–negative infective endocarditis (“sterile”) in humans. 13, 16 There was no disease associated with Bartonella infection in dogs until 1995, when the first case of canine heart valve infection was associated with the novel species B. vinsonii berkhoffi. 2 Subsequently, in a study of 12 dogs seroreactive to B. vinsonii berkhoffi, valvular endocarditis was found in eight dogs and the 16S ribosomal subunit of B. vinsonii berkhoffi was amplified from blood or valvular tissue in three dogs. 1 B. clarridgeiae and B. washoensis have also been associated with cases of endocarditis in dogs. 3, 4 Bartonella organisms in dogs have subsequently been associated with granulomatous lymphadenitis, rhinitis, and hepatitis. 8, 15 Despite the widespread recognition that these bacteria can cause disease in animals, associated lesions are poorly characterized. This study is a retrospective report of the gross, histologic, and ultrastructural features of a series of valvular endocarditis cases associated with Bartonella.
Cases selected for inclusion in the study were obtained from the necropsy and surgical pathology services at the University of California School of Veterinary Medicine spanning the 5-year period from 1997 to 2001. Dogs were diagnosed with valvular endocarditis at the time of necropsy. With the exception of one case, necropsies were performed at the Veterinary Medical Teaching Hospital of the University of California–Davis. Of the 31 gross diagnoses of endocarditis, tissue from at least one of the affected valves was embedded in all but one case. Histologic sections were examined, and clinical data reviewed in the remaining 30 cases. In all 30 cases, serial, paraffin-embedded sections were stained with hematoxylin and eosin, with Brown and Brenn's, and with Warthin-Starry or a Steiner's silver (or both) stains. DNA was extracted from paraffin-embedded or frozen valvular tissue in all 30 endocarditis cases, and Bartonella DNA was identified by polymerase chain reaction (PCR)–restriction fragment length polymorphism of an approximately 380-bp fragment of the glt-A gene. 17 The amplified fragment was verified by both gel electrophoresis, after HhaI, AciI, MseI (New England Biolabs, Beverley, MA), and TaqI (Promega, Madison, WI) restriction endo-nuclease digestions, and by sequence analysis. Banding patterns were compared with a domestic dog isolate (B. vinsonii subsp. berkhoffii, ATCC 51672), B. henselae (strain U-4, University of California, Davis), and B. clarridgeiae (ATCC 51734). Associated inflammation was characterized by immunohistochemistry on all Bartonella-associated cases. Primary monoclonal antibodies were anti-CD3 for T lymphocytes (clone CD3–12, Vector Laboratories, Burlingame, CA), anti-CD79a (clone HM57, Dako Corp., Carpinteria, CA) for B lymphocytes, and anti-CD18 (clone Fe3.9F2, courtesy of P. F. Moore, University of California, Davis). Tissue sections were immunostained, using primary antibody, biotinylated secondary antibody, and streptavidin–peroxidase as third reagent with 3-amino-9-ethycarbazol as a chromagen. Hematoxylin was used as a counterstain. For analyses of vascular proliferations and endocardium, tissue sections were stained using polyclonal rabbit anti-human factor VIII–related antigen or anti-CD31 (platelet endothelial cell adhesion molecule [PCAM]) (or both) (DAKO Corp., Santa Barbara, CA) as the primary antibody, followed by biotinylated, anti-rabbit secondary antibody, and alkaline phosphatase–labeled streptavidin as a third reagent (Str-AviGen Super Sensitive Kit, BioGenex, San Ramon, CA). For electron microscopy studies, aortic valve was transferred from 10% formalin to half-strength modified Karnovsky's 10 before two washes with 0.2 M sodium cacodylate and after fixation in 2% osmium tetroxide reduced with 2.5% potassium ferrocyanide. 18 After fixation, tissue was processed by routine methods for examination in a Zeiss 10C transmission electron microscope at 60 V accelerating voltage.
Bacteria other than Bartonella were either cultured directly from the valve or visible by direct histologic and special staining methods in 11 out of 30 cases. Identified bacteria included Escherichia coli, Pseudomonas, beta-hemolytic Streptococcus, and Staphylococcus spp. The mitral valve alone was involved in 5 out of 11 of these cases, whereas the aortic valve alone was involved in two cases, and both valves in three cases. One gross report did not specify the valve(s) involved. In the remaining 20 cases, bacteria were not identified by culture, histologic examination, or by Gram's staining method. Of these sterile cases, Bartonella was amplified in six cases by PCR from valve tissue, and no other bacteria or infectious agents were identified. Of these six cases, five were described as solely aortic lesions and one involved both the aortic and mitral valves. PCR amplification of valvular tissue from cases associated with culturable bacteria was negative for Bartonella with one exception. The histologic and descriptive features of this case of dual infection (Staphylococcus and B. vinsonii berkhoffii) were consistent and included, in this study, with cases of culturable bacteria. The age range of the six dogs diagnosed with Bartonella infection was 4–10 years (mean 7.3 years). This was similar to the overall age range for dogs with non-Bartonella endocarditis. Bartonella-associated lesions occurred in large breed, male or male-castrate dogs in all six cases. Of the 31 total cases, 18 dogs (58%) had clinical signs or physical exam findings (or both) referable to heart failure. Among those diagnosed with Bartonella infection, all had clinical signs referable to heart failure. Among other presenting clinical signs for the Bartonella-associated dogs, historic or ongoing joint problems were present in 67% (four out of six) of the dogs. No joint problems or lameness was reported in the non-Bartonella cases. Clinical data was available in three out of six of the Bartonella-associated cases, and the only consistent effect was on platelets, with mild thrombocytopenia (117,000–143,000/μl) present in two cases and severe thrombocytopenia (reported by the referring veterinarian) in one case. Serology for three Bartonella species (B. vinsonii subsp. berkhoffii, B. henselae, and B. clarridgeiae) was performed on four out of six of the dogs, and the results were reported recently along with descriptions of their cardiac evaluations. 14 A high (>1 : 512) antibody titer to multiple Bartonella spp. was present in all four cases. By PCR and sequence analyses, three species of Bartonella were associated with valvular lesions. B. vinsonii berkhoffi was present in four dogs and B. clarridgeiae in one dog. 3 A B. species slightly different from B. clarriigeiae and identified as B. clarridgeiae–like species was present in one dog.
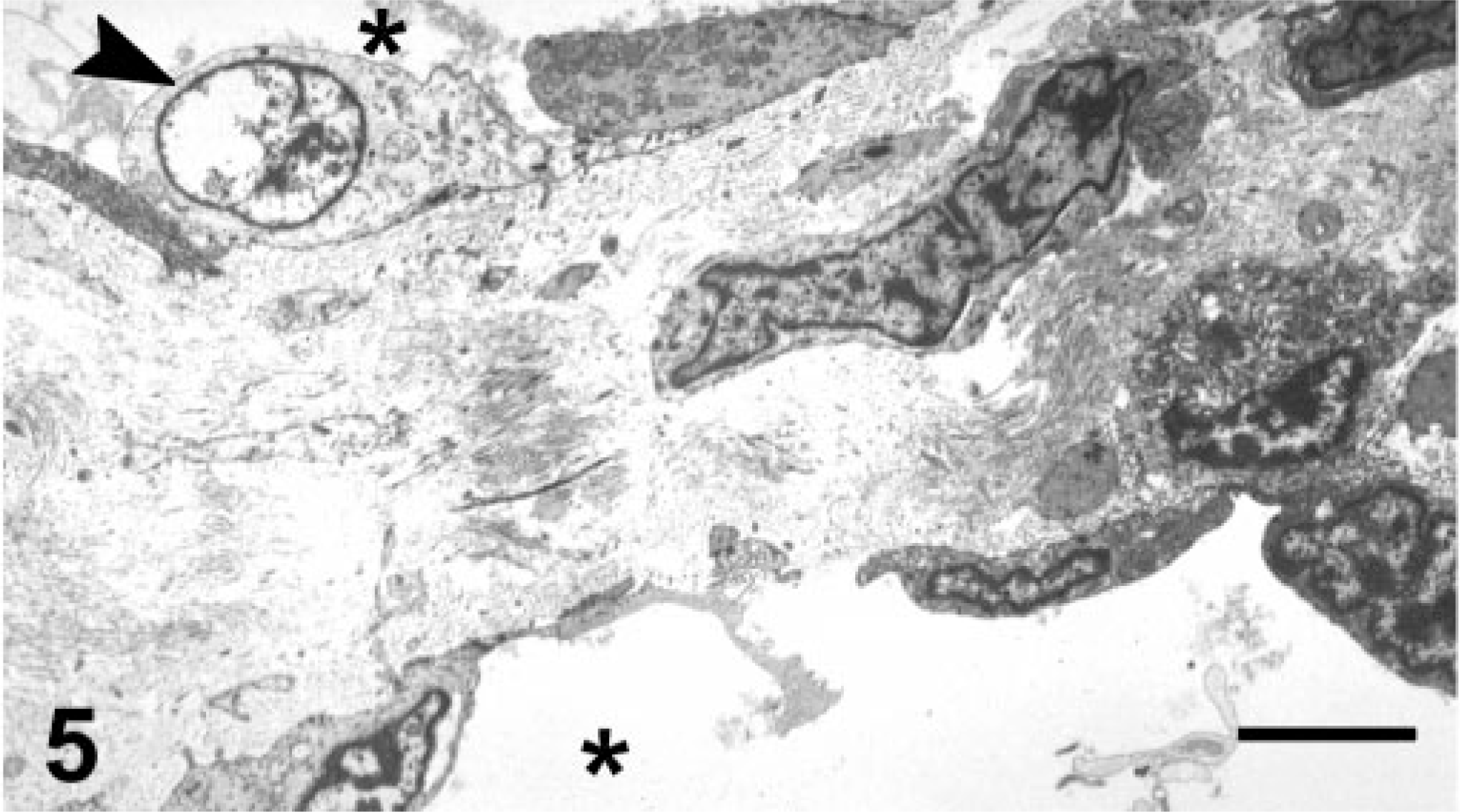
By gross analysis, valves associated with all three species of Bartonella were similar and most notably affected on their periphery, where single or multiple, variably sized nodules expanded the valve leaflet (Fig. 1). The surface texture was variable, with smaller nodules smooth and firm and larger nodules with granular and friable regions. In most cases, valves were thickened and gathered in the region of the heart base. Slightly raised, dull white plaques were present on the surface of the adjacent aorta and ventricular endocardium. By histologic analysis, the distal leaflets of affected valves were expanded markedly by fibrous tissue and mineral deposits (Fig. 2). In all cases, there was a segmental to focally extensive expansion of the subvalvular endothelium by loosely woven fibrous tissue and scattered individual or clustered cells (or both) that by immunohistochemical analysis with anti-CD31 antibody were identified as endothelial cells (Fig. 3b). In four out of six cases, the valve leaflet (most notably at the annulus and extending to the region of valve insertion overlying the ventricular septum) was disrupted by small vessels and clefts (Fig. 4). By immunohistochemical analysis, the endothelium lining these spaces was reactive to fragmented, and individual or rafts of endothelial cells were often present within vascular lumen (Fig. 4b). Scattered, individual, and clusters of histiocytes, T lymphocytes, and fewer B lymphocytes were present throughout the affected valve and rarely extended into the myocardial tissue at the base of the valve (immunohistochemistry results with CD3, CD79a, and CD18 not shown). In two cases, scattered neutrophils were present in the affected valvular stroma. Histologic features of the 11 culturable bacteria–associated endocarditis cases were consistent with acute bacterial infection and included thick, superficial, and intravalvular fibrin deposits, dense aggregates of viable and degenerate neutrophils, scattered valvular necrosis, and, infrequently, mineralization. Although small vessels were rarely present extending beyond the base of the valve, these were neither as reactive nor as prominent a feature as those associated with the Bartonella-infected valves. The characteristic histologic features of endocardiosis were similar to those described previously. 9 Clinically, these cases were distinctly present in small breed dogs and affected the mitral valve. Histologically, marked fibroblast proliferation and myxomatous change were distinct features not consistently present in Bartonella-associated valves. Proliferation of vessels was present in all cases of endocardiosis; however, in general, the vessels in these cases were more organized, less reactive, and there was no detectable endothelial necrosis.
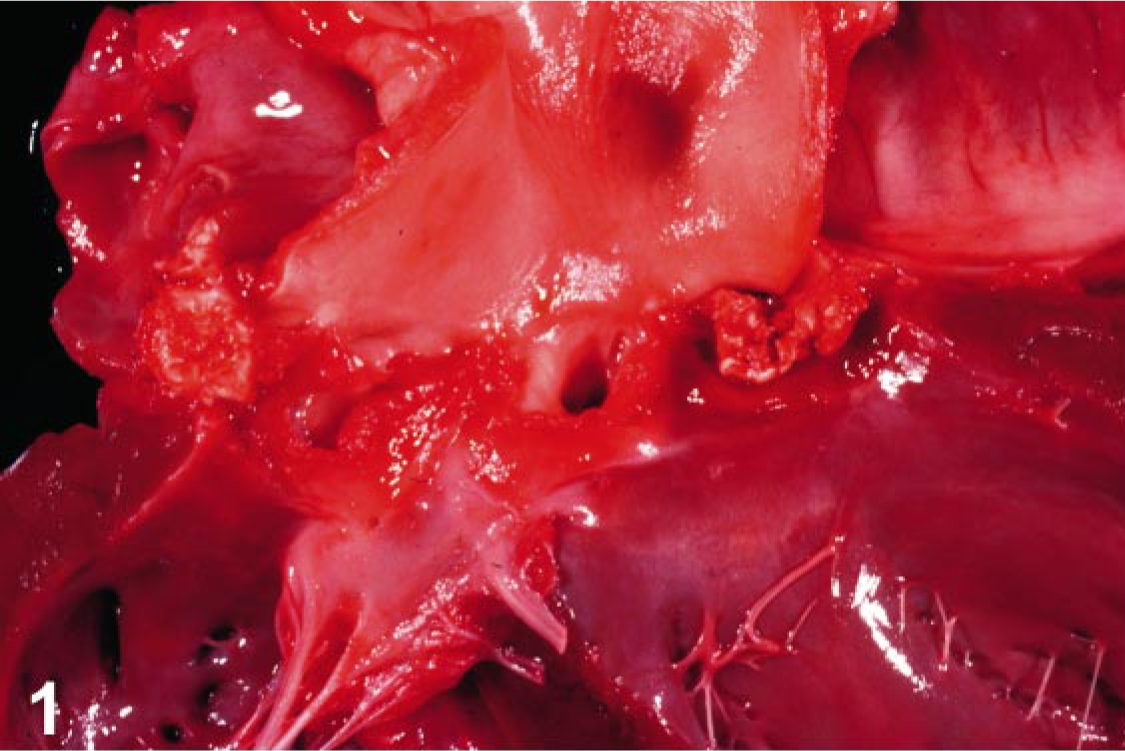
Aortic valve; canine, dog No. 2. Chronic endocarditis. The periphery of the valves is eroded. Variably sized, mineralized nodules expand the valve leaflet. There are multifocal, slightly raised, dull white plaques on the surface of the adjacent aorta and ventricular endocardium.
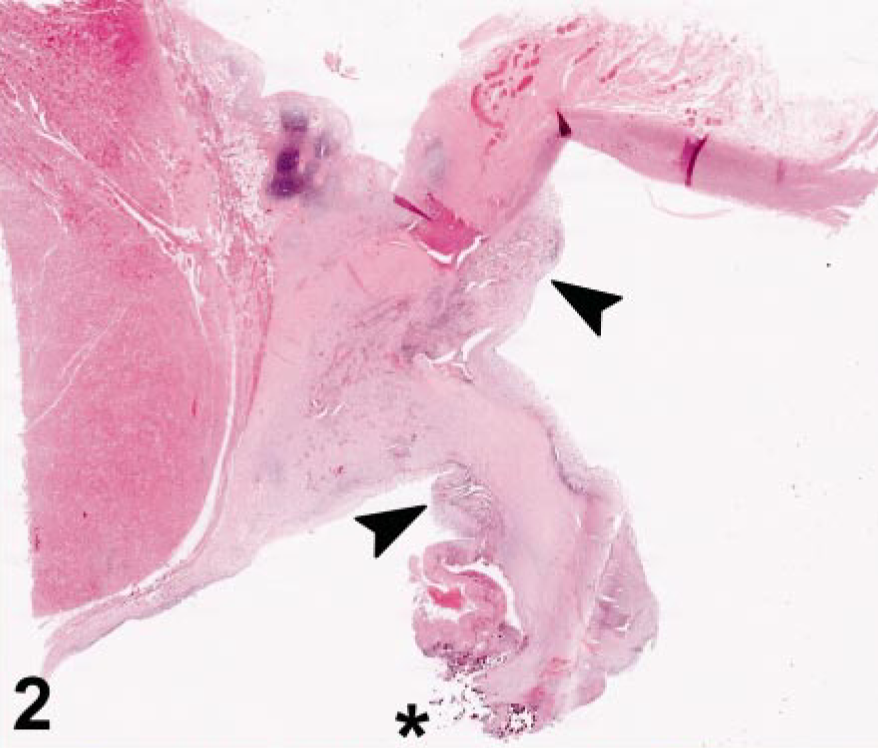
Aortic valve, aorta, and ventricular septum; canine, dog No. 4. The distal leaflet is gathered, fragmented, and contains mineral deposits (∗) and hemorrhage. At the base of the valve, the endothelial surface is lifted and there are broad bands of loose connective tissue, small vessels, and endothelial-lined clefts (arrowheads). HE. Bar = 180 μm.
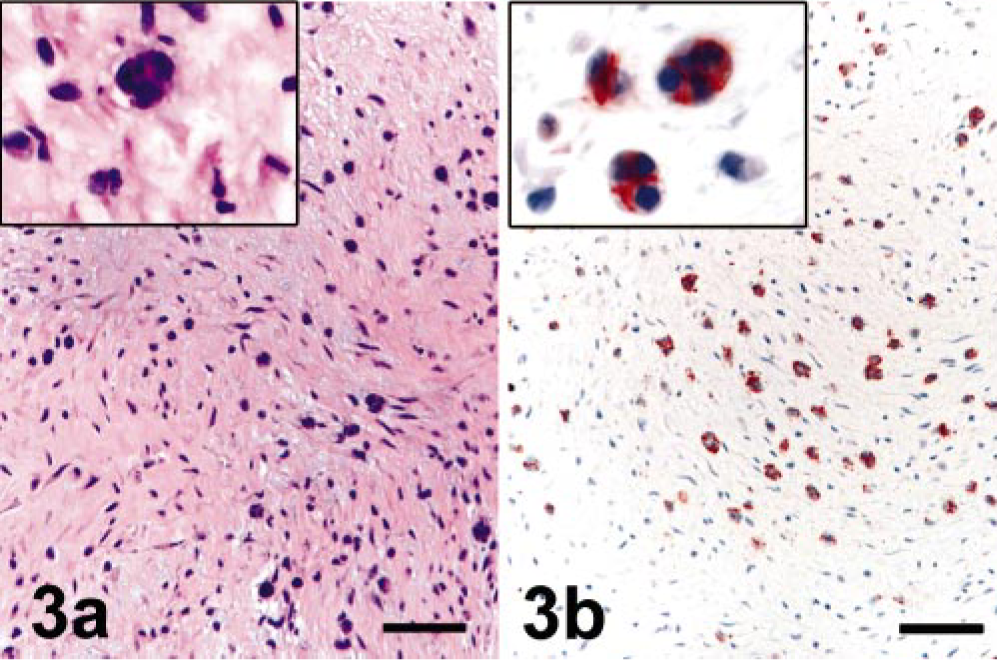
Valve surface; canine, dog No. 6.
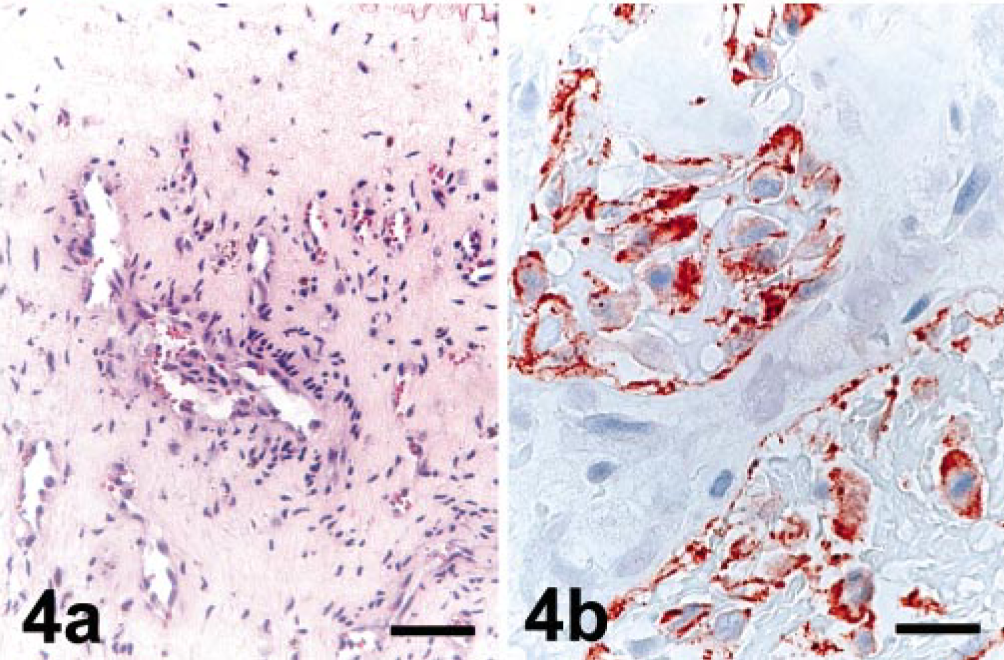
Valve leaflet with vascular proliferation; canine, dog No. 2.
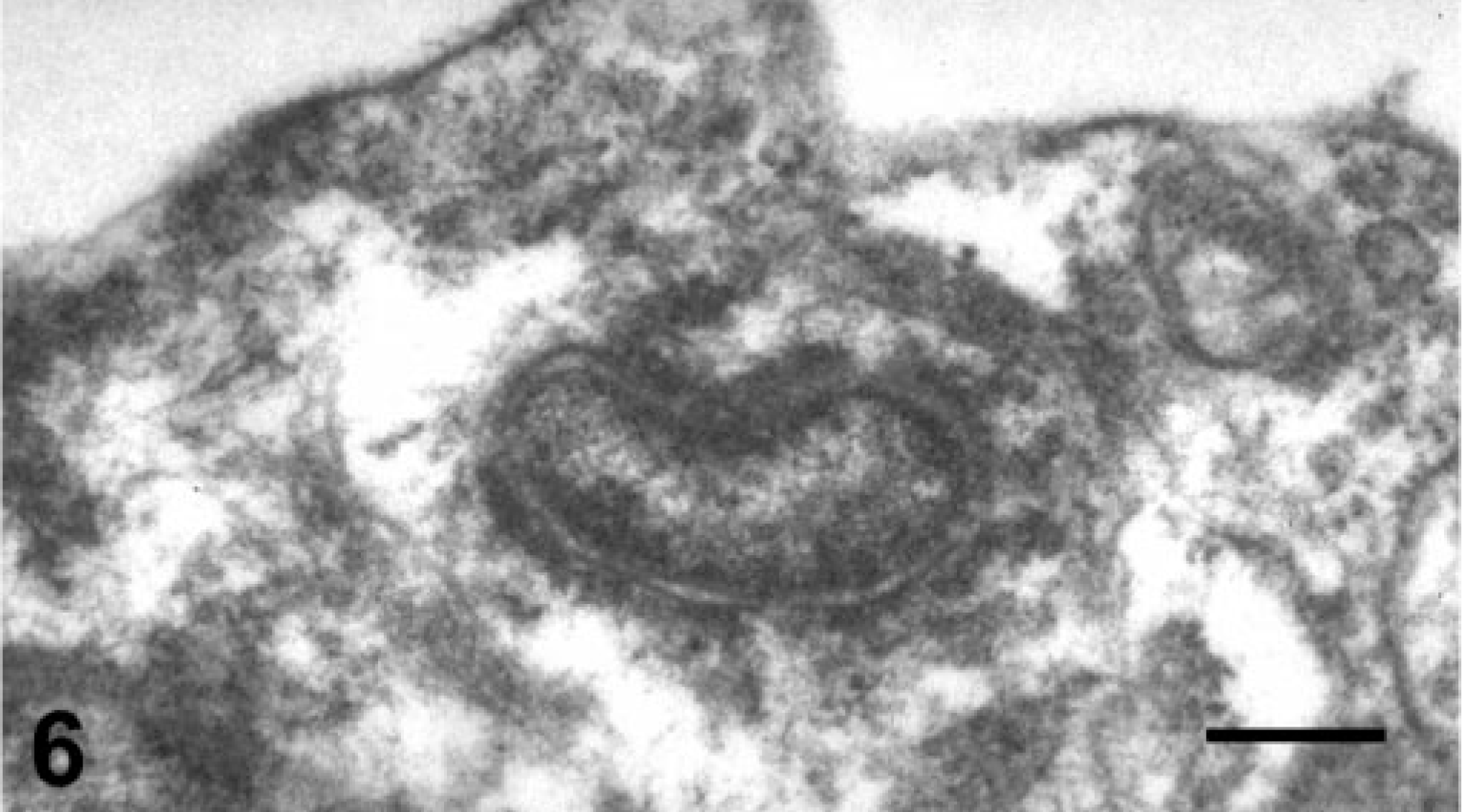
Warthin-Starry and Steiner's silver stains were of limited value in interpreting the etiology of the valvular lesions because there was extensive background staining and because, when present, the size and shape of proteobacteria is variable. In dog No. 2, there were intracytoplasmic, intraendothelial ∼5-μm-long bacteria that were confirmed by transmission electron microscopy. In the other five cases, the presence of Bartonella could not be confirmed confidently by silver or Gram's stains.
Transmission electron microscopy of three Bartonella cases revealed regions of reactive valvular and annular endothelium with evidence of endothelial proliferation and necrosis. Evidence of endothelial cell damage included swelling and distortion of mitochondria, vacuolation, and chromatin clumping (Fig. 5). Bacillary organisms in this study were relatively rare compared with a previous report, 1 but similar to this report, bacilli were slightly curved, 0.2–0.4 μm in diameter and 1–3 μm in length (Fig. 6). Bacteria were present within endocardial endothelial cells, within small vessel lumen, and scattered within the valvular interstitium.
Vascular clefts in aortic valve; canine, dog No. 4. Vascular lumina are denoted with an asterisk (∗). Vascular endothelium is hyperplastic with signs of endothelial necrosis (arrow), repair, and inflammation. Transmission electron microscopy, uranyl acetate and lead citrate. Bar = 25 μm.
Valvular surface endothelial cell; canine, Bartonella organism present within the cytoplasm of a valvular surface endothelial cell. Transmission electron microscopy, uranyl acetate and lead citrate. Bar = 0.4 μm.
Bartonella were associated with endocarditis in a significant proportion of the endocarditis cases in dogs presenting at the Veterinary Medical Teaching Hospital in Davis. Histologic alterations of Bartonella infection include chronic histiocytic and lymphocytic endocarditis, mineralization, and endothelial cell and vascular proliferation. None of these features, alone or in combination, are pathognomonic for infection with Bartonella. However, the histology is distinguished from both acute, non-Bartonella bacterial endocarditis and endocardiosis. This series of cases provides characteristic histologic features that are supplementary criteria for diagnosis. Confirmation of Bartonella-induced endocarditis relies on a combination of diagnostic techniques. Serologic results, histology, and confinement to or inclusion of the aortic valve should strongly suggest a Bartonella-induced lesion. There is no polyclonal or monoclonal antibody currently available to directly detect Bartonella spp. in formalin-fixed tissues. In the cases presented in this report, factors supporting causation included failure to isolate or observe other bacteria, PCR amplification of DNA from heart valve tissue taken at the time of biopsy/necropsy, observation of the organism in the aortic valve (transmission electron microscopy), and serology.
For culturable bacteria that cause endocarditis, the proposed pathophysiologic mechanism of valvular endocarditis is postbacteremic colonization of a damaged region of the valve or of damage-associated fibrin, followed by inflammation. 19 Very little is known regarding the proposed mechanism of invasion by intracellular, fastidious organisms that are associated with valvular endocarditis. The presence of Bartonella within endothelial and endocardial cells leaves open the question of whether these bacteria directly invade the overlying endocardial and valvular endothelium or whether they arrive through small vessels at the base of the valve (or both). Several studies indicate that the principal cellular target of Bartonella infection is the endothelial cell. 5, 6, 11, 12 B. henselae, B. bacilliformis, and B. quintana cause a proliferation of endothelial cells within the dermis (bacillary angiomatosis) and within the liver (bacillary peliosis). 12 Although it is possible that angiogenesis in these cases could be the sequelae of a chronically inflamed valve, endothelial proliferation might be caused by direct stimulation by Bartonella.
