Abstract
The proliferation of footpad keratinocytes of canine distemper virus (CDV)—infected dogs was investigated. Footpads of 19 dogs inoculated experimentally with a virulent distemper strain (A75/17) and of two noninoculated control dogs were collected at necropsy. Dogs were divided into four groups according to results of the postmortem examination: dogs with severe distemper (group 1), dogs with mild distemper (group 2), inoculated dogs without distemper (group 3) and noninoculated dogs (group 4). There was no distinct difference of epidermal thickness among the four groups. Infection of the footpad epidermis with CDV was demonstrated using immunohistochemistry for viral nucleoprotein and in situ hybridization for nucleoprotein messenger ribonucleic acid (mRNA). Only group 1 dogs had viral antigen and mRNA in the footpad epidermis with the same distribution. Footpad epidermis of group 1 dogs had more mitotic figures in the basal layer, and significantly more basal keratinocytes were positive for the proliferation markers Ki-67 and proliferating cell nuclear antigen. Double-staining for Ki-67 and viral nucleoprotein identified rare double-labeled basal keratinocytes. These findings suggest that the presence of CDV particles in the footpad epidermis is associated with keratinocyte proliferation.
Canine distemper virus (CDV) is a single-stranded ribonucleic acid (RNA) virus of the genus Morbillivirus. Infection of carnivores usually results in systemic disease with involvement of the respiratory, gastrointestinal, and central nervous systems (CNS). 1 CDV, like other morbilliviruses, can infect epidermal cells of a variety of different species such as mink, ferret, and racoon dogs. 4,9,20 Footpad keratinocytes are infected so frequently that the demonstration of viral antigen in biopsies of the footpad was suggested as a means of intravitam diagnosis of CDV. 12 Massive CDV-induced thickening of the footpad, macroscopically quite similar to changes observed in dogs with zinc-responsive dermatosis, pemphigus foliaceus, or idiopathic nasodigital hyperkeratosis, is often called “hard pad disease”. 24 Histologically, orthokeratotic hyperkeratosis, diffuse keratinocyte vacuolization, and presence of eosinophilic viral inclusion bodies are described, whereas epithelial hyperplasia is rarely mentioned. 12,21 However, the portrayal in one textbook refers to hyperplasia. 26 In the present study, proliferation of footpad keratinocytes from dogs with experimentally induced distemper was assessed histologically using various methods. Mitotic figures were counted, and the expression of Ki-67 and proliferating cell nuclear antigen (PCNA), two well-established proliferation markers, were analyzed immunohistologically. 11 The spatial association between cells positive for the proliferation marker Ki-67 and cells containing viral nucleoprotein was studied using immunofluorescent double-labeling.
Footpads were collected from 21 juvenile, specific pathogen-free Beagle dogs, which were maintained according to the guidelines of the institution's animal care and use committee. Nineteen dogs (Nos. 1–19) were inoculated with the A75/17 CDV strain; dogs nos. 20 and 21 were not inoculated and served as controls. Splenic extract from dogs in which the virus had been propagated earlier was used for intravenous inoculation. 6 Animals were euthanatized 20–35 days later, when they showed clinical signs of distemper including fever, rhinitis, diarrhea, and central nervous disturbances. Dogs that did not show any clinical signs were euthanatized subsequently. Footpads from the metacarpus and metatarsus and body organs were fixed in 4% paraformaldehyde for 24 hours, paraffin-embedded, and cut. Sections were stained with hematoxylin and eosin (HE) for histopathology to assess the presence and severity of disease in the dog at postmortem examination. Systemic distemper was defined by the presence and frequency of viral eosinophilic inclusion bodies in multiple organs such as stomach, renal pelvic epithelium, and urinary bladder and presence of distemper lesions in the brain. Dogs were divided into the following groups: group 1, inoculated dogs with severe distemper and having typical distemper lesions (inclusion bodies, astrocytosis, microgliosis) in the CNS with demonstration of virus antigen; group 2, inoculated dogs with mild distemper and no distemper lesions in the CNS; and group 3, inoculated dogs without distemper. The noninoculated control dogs constituted group 4 (Table 1).
Dog identification number, days between inoculation and euthanasia, severity of distemper, presence of CNS lesions, and presence of CDV in footpad epidermis in 21 Beagle Dogs after experimental inoculation with virulent A75–17 distemper virus. ∗
∗ Dogs with severe distemper were classified as group 1, dogs with mild distemper as group 2, inoculated dogs but without distemper as group 3, and noninoculated controls as group 4.
† p.i. = post inoculation; CNS = central nervous system; CDV = canine distemper virus; NA = not applicable; + = present; — = absent.
The presence of virus in the footpad epidermis was evaluated using immunohistochemistry and in situ hybridization. CDV nucleoprotein antigen in footpads was assessed using monoclonal antibody D110 in a dilution 1 : 5. 25 CDV nucleoprotein messenger ribonucleic acid (mRNA) was evaluated with in situ hybridization using a digoxigenin (DIG)–labeled anti-sense RNA probe that binds to viral mRNA. 10,27 Proliferation was studied by counting mitotic figures in the stratum basale of the footpad on HE sections. For further evaluation of keratinocyte proliferation, sections were stained immunohistochemically with antibodies against the proliferation markers Ki-67 (Zymed, San Francisco, CA, 1 : 40) and PCNA (DAKO, Zug, Switzerland, 1 : 2,000). 11 Ki-67 was demonstrated after pretreatment with citrate (pH 6.0, 2 × 5 minutes microwave at 750 W). PCNA was observed after the sections were heated at 92 C for 5 minutes in 364 pM sodium dodecyl sulfate. 19 Positive reaction was observed with 3-amino-9-ethylcarbazole (AEC, DAKO). The colocalization of Ki-67 and CDV nucleoprotein was evaluated using immunofluorescence. Ki-67 was labeled using the same antibody as above and a Texas Red–conjugated streptavidin (Vector Laboratories, Inc., Burlingame, CA) as indicator. This was followed by an antiserum specific for CDV nucleoprotein conjugated to fluorescein isothiocyanate (FITC) (VMRD, Inc., Pullman, WA). Slides were mounted with fluorescent mounting medium (DAKO).
For statistical analysis the percentage of mitotic figures and epidermal cells positive for the proliferation markers Ki-67 and PCNA within the basal layer was assessed by direct cell count. A total of 5 × 100 basal keratinocytes were counted per footpad and noted as positive or negative. Results were compared among footpads from dogs of the four groups. Results were tested for significance with a nonparametric Kruskal-Wallis test followed by Dunn's multiple comparisons test (InStat, version 3, GraphPad Com, San Diego, CA).
Distemper was diagnosed at postmortem examination in 11 dogs (Table 1). In seven dogs viral inclusions were abundant, and a severe form of distemper was diagnosed (Nos. 1–7, group 1). These were the only dogs with CDV lesions in the brain as well. In the remaining four dogs (Nos. 8–11) inclusion bodies were very rare, and no changes indicative of a CDV infection were observed in the brain; these findings were interpreted as a milder form of distemper (group 2). The other 10 dogs, including the noninoculated controls, had no evidence of distemper on the basis of anatomical change (groups 3 and 4).
The footpad epidermis of seven dogs (Nos. 1–7) contained abundant nucleoprotein and nucleoprotein mRNA in the same locations and in comparable intensity. Nucleoprotein as a red precipitate and mRNA as a dark blue stain were located in the cytoplasm of most keratinocytes in the stratum spinosum and granulosum. Staining intensity for both stains was stronger directly below the corneal layer. In addition, in three of these seven dogs basal keratinocytes contained both nucleoprotein and mRNA. All dogs with CDV in the footpad epidermis belonged to group 1. None of the remaining dogs had CDV in the footpad epidermis.
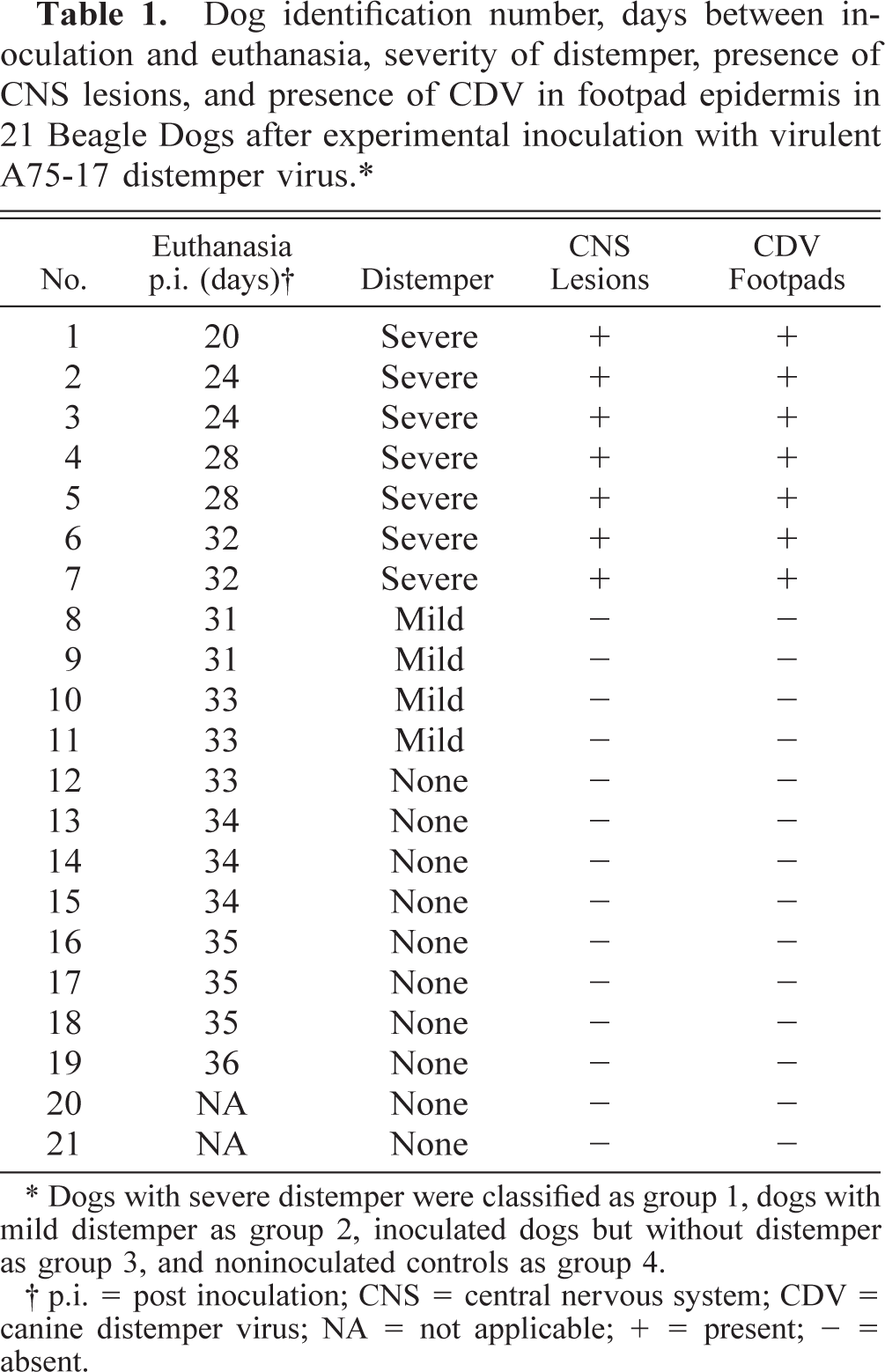
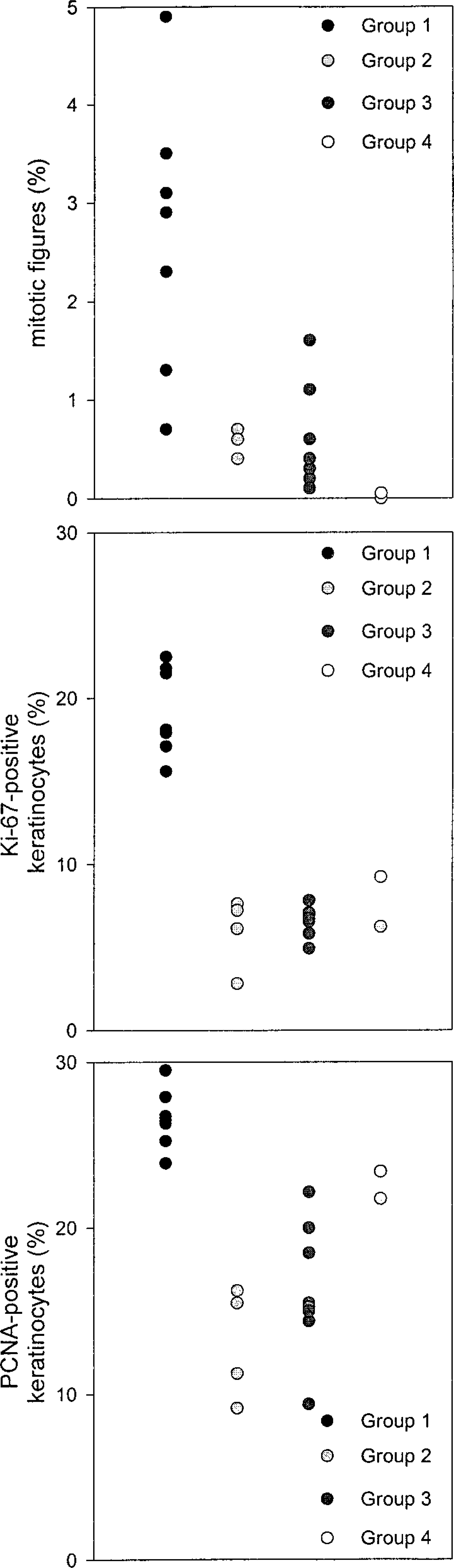
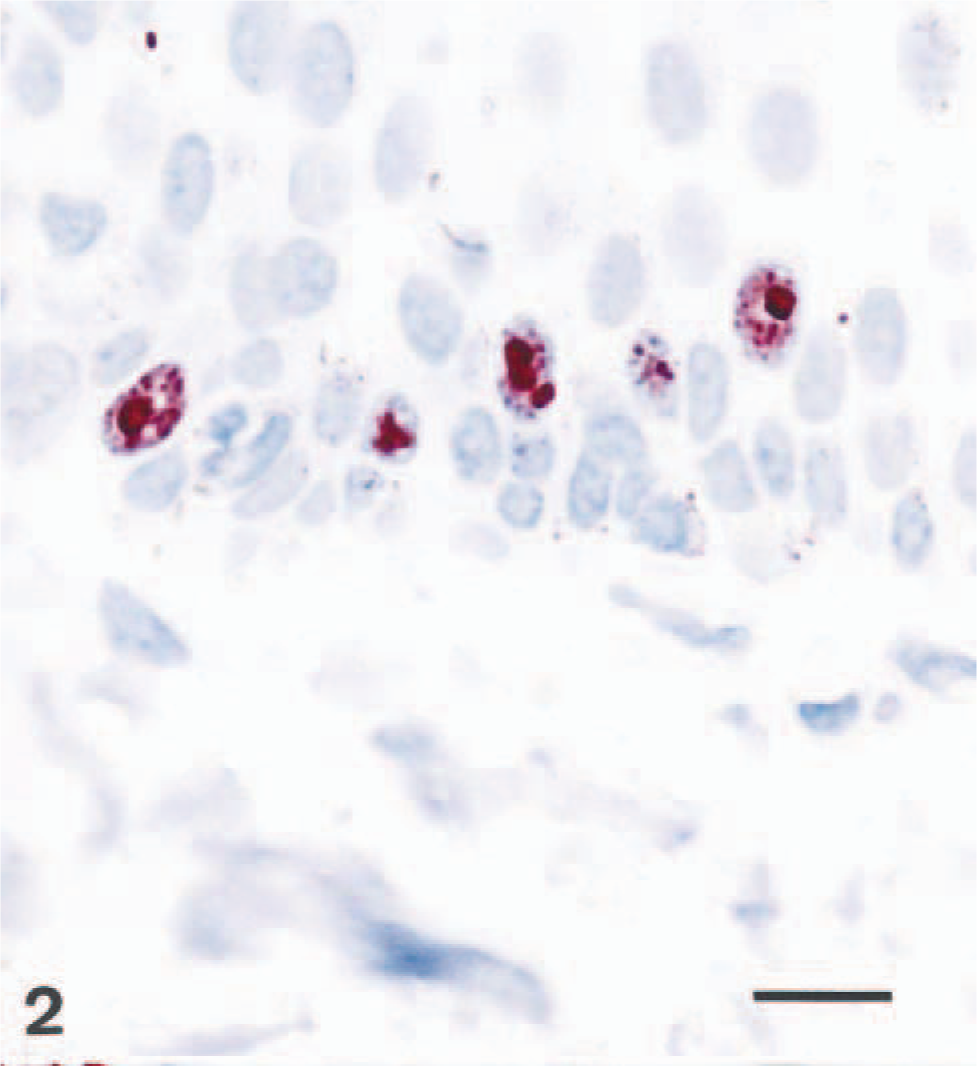
Vacuolar degeneration and presence of eosinophilic inclusion bodies was observed in the epidermis of one dog (No. 2). No distinct differences in epidermal thickness were observed among the four groups. The footpad epidermis of group 1 dogs had significantly more mitotic figures compared with groups 3 and 4 (P < 0.05 and 0.01, respectively, Fig. 1). Immunopositivity for the proliferation markers was present in keratinocyte nuclei either as a solid staining of the whole nucleus or as a granular deposit. Ki-67 was usually present in the nuclei of basal keratinocytes with only rare positive cells amongst suprabasal keratinocytes (Fig. 2). In contrast, PCNA was quite commonly observed in nuclei of the suprabasal keratinocytes in addition to basal cells (Fig. 3). The upper spinous layer did not contain any cells positive for either proliferation marker. There was a significant increase in the numbers of basal keratinocytes that stained positive for proliferation marker Ki-67 in the footpads of group 1 dogs compared with groups 2 and 3 (P < 0.01 and 0.05, respectively, Fig. 1). Similarly, footpads of group 1 dogs contained more PCNA-labeled keratinocytes compared with groups 2 and 3 (P < 0.01, Fig. 1).
Proliferation of footpad keratinocytes. Percentage of mitotic figures (top graph), keratinocytes positive for Ki-67 (center graph), and PCNA (bottom graph). Group 1 = dogs with severe distemper; group 2 = dogs with mild distemper; group 3 = inoculated dogs without distemper; group 4 = not inoculated. Only footpad epidermis of group 1 dogs contained CDV antigen and mRNA.
Dog No. 2. Ki-67-positive keratinocytes in the footpad epidermis. ABC-complex immunohistochemistry. Bar = 30 µm.
Dog No. 2. PCNA-positive keratinocytes in the footpad epidermis. ABC-complex immunohistochemistry. Bar = 25 µm.
Double-positivity of keratinocytes for Ki-67 and viral nucleoprotein was observed only occasionally in basal keratinocytes of 3 dogs (Nos. 2, 5, and 6, Fig. 4). Far fewer keratinocytes were double-labeled for Ki-67 and CDV nucleoprotein than for Ki-67 alone. The proliferation indices for group 1 dogs were the same regardless of whether these dogs had CDV in basal footpad keratinocytes (3/7) or not (4/7).
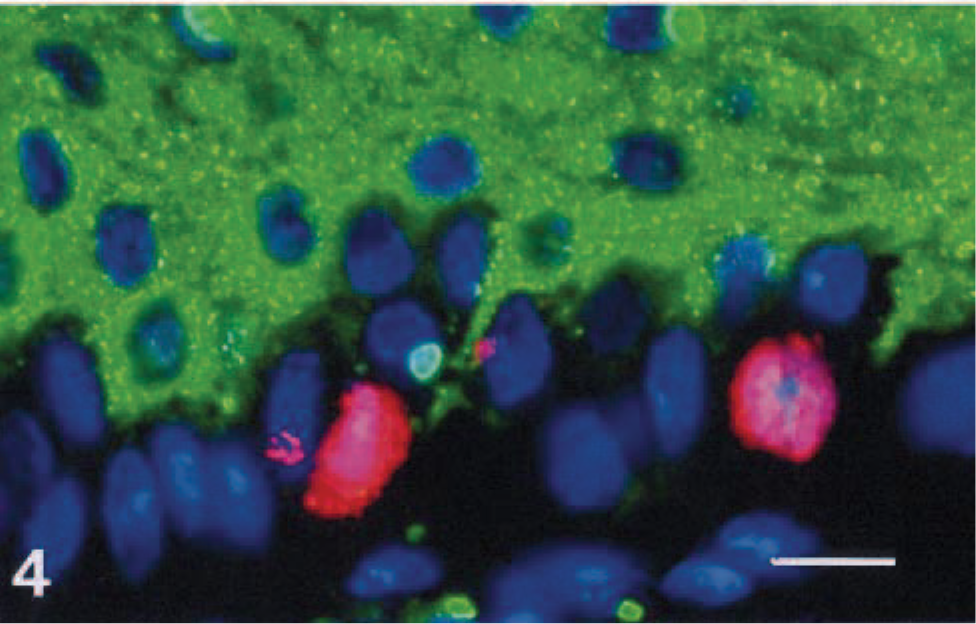
Dog No. 2. CDV-positive and Ki-67–positive keratinocytes in footpad epidermis. Cellular CDV nucleoprotein is demonstrated using an FITC-labeled antibody (green), nuclear Ki-67 is demonstrated using Texas Red (red), and nuclei are counterstained with DAPI (blue). Bar = 15 µm.
Overall, many more cells were positive for PCNA compared with Ki-67, regardless of presence or absence of CDV nucleoprotein. Higher proliferation indices for PCNA compared with Ki-67 have also been observed in other studies and were considered to be partly because of a longer half-life of this protein. 11,19 Potentially, PCNA immunohistochemistry could label cells that are no longer actively cycling. This would explain labeling of keratinocytes in the suprabasal layers. In contrast, Ki-67 appears to be a more specific marker for proliferation, and consequently, the double-labeling studies were conducted using Ki-67.
Usually, thickening of the footpad epidermis in CDV infection is described to be because of hyperkeratosis, and hyperplastic lesions are seldom mentioned. However, in footpad epidermis containing CDV particles, more keratinocytes were labeled for proliferation markers compared with footpad epidermis without evidence of CDV infection. These findings suggest that keratinocyte proliferation may take part in the thickening of footpads, as can be observed after CDV infection, although no such thickening was observed in this study. The duration of infection may have been too short to allow development of an obvious hyperplasia. Alternatively, the proliferation may represent a compensatory reaction to maintain normal epidermal thickness. However, keratinocyte infection by CDV was not cytocidal.
Hyperkeratosis of the footpad has been reported to occur around 1 to 10 weeks after onset of clinical signs or exposure. 7,16,17 The absence of metacarpal hyperkeratosis in this study was not expected but is most likely a function of time. It appears probable that dogs would have developed footpad hyperkeratosis had they lived longer. The intricate regulation of the switch of proliferation toward differentiation that occurs in keratinocyte development and maturation is at the center of many studies and, although much is known, not completely understood. 8 In many skin diseases both phenomena occur simultaneously; however, dermatoses with atrophic epidermis and hyperkeratosis as seen in conjunction with endocrine disorders exemplify that this is not always the case. The correlation between proliferation of basal keratinocytes and disturbance of the keratinization process in CDV-infected footpad epidermis is unclear at the moment. Both processes may have been disrupted in a similar manner; however, a sequence of events is also conceivable. In such a scenario, an increase in proliferation would occur first, followed by disordered keratinization.
Keratinocyte proliferation occurs after infection with various viruses, of which papilloma and pox virus are probably the best-studied examples. 2,5 The interaction between viral and cellular proteins has been at the focus of many studies. 3,14 Keratinocytes proliferation was also described as a consequence of infection with other morbilliviruses. In human AIDS patients with measles virus infection, hyperplasia of the epidermis was observed. 23 Similar findings were reported from a patient without HIV infection. 22 Hyperplasia was also a feature in seal morbillivirus dermatitis. 18 These reports demonstrate that noncytocidal infection of epithelial cells by morbilliviruses is not uncommon.
A proliferative response to infection by CDV as seen in these footpad keratinocytes is contrary to observations in lymphocytes, in which proliferation is inhibited. 13,15 One possible reason for this distinct response may be the direct infection of lymphocytes by the virus, whereas most proliferating footpad keratinocytes were negative for CDV antigen. Additionally, this distinction may reflect an intrinsic difference in the cellular response by various CDV-infected cells.
The mechanism by which CDV infection induces keratinocyte proliferation is not understood at the moment. It may be because of a direct viral effect; however, indirect mechanisms including release of cytokines or growth factors are also possible. The results of the double-labeling point strongly to a role for such a bystander effect and indicate the presence of an indirect mechanism. However, systemic CDV infection alone, as observed in mild form in group 2 dogs, was not associated with an increase in proliferation indices compared with group 1 dogs. This suggests the induction of a paracrine acting mechanism, which requires close proximity between virus-infected cells and cells capable to enter the cell cycle and proliferate. In vitro infection experiments should help to better understand the mechanism of persistent, noncytocidal CDV infection of keratinocytes.
Footnotes
Acknowledgements
We thank the Berna Biotech AG, Berne, Switzerland, for financial support and Ines Schmid and Marianne Wyder for excellent technical assistance. This work was supported by the Swiss National Science Foundation (grants #31-58657.99 and #32-61961.00).
