Abstract
Characteristic ganglion-like cell proliferation observed in the skin of Djungarian hamsters was investigated using 24 male and 24 female hamsters, 1–6 months of age, to examine the anatomic location of these ganglion-like cells and their morphologic features. One abdominal skin tumor composed of these cells and resembling proliferative fasciitis in humans was also examined. Skin ganglion-like cells were rarely observed in young animals but increased in number and extent with age, especially in males. These cells were frequently seen in the ventral and medial regions of the trunk and legs rather than in the dorsal and lateral regions. Light microscopic examination of these ganglion-like cells revealed abundant vesicular basophilic cytoplasm with delicate intracytoplasmic silver stain-positive fibrils. Ultrastructurally, these cells contained abundant rough endoplasmic reticulum and Golgi complexes with dilated cisternae; intracellular collagen fibrils were present within these cisternae. Heat shock protein 47, β-tubulin, and androgen receptor were expressed in these cells. The morphologic features of cells of one tumor resembling human proliferative fasciitis were identical to those observed in ganglion-like cells. The results of the present study suggest that these ganglion-like cells are derived from intrinsic undifferentiated mesenchymal cells in the dermis or subcutaneous adipose tissue and that any tumor-like lesion they form should be regarded as an abnormal proliferative lesion of skin ganglion-like cells rather than as proliferative fasciitis or fibroma.
Djungarian hamsters (Siberian hamster, Phodopus sungorus) are small cricetid rodents native to the steppes of eastern Kazakhstan and southwestern Siberia. They have recently become popular as pets. They develop peculiar subcutaneous neoplastic lesions, found predominantly in the abdominal skin of adult males. 7,10 The lesion is composed of proliferations of large spindle to polygonal cells that have abundant basophilic cytoplasm and a ganglion cell-like appearance accompanied by abundant interstitial collagen. Despite the relatively frequent spontaneous occurrence of this lesion in the Djungarian hamster, the identity of this lesion and its cell of origin have been controversial. Although this lesion has been tentatively diagnosed as a fibroma on the basis of ultrastructural features of proliferating cells and production of collagen fibers, 10 Fujiwara used the term proliferative fasciitis in the Djungarian hamster, based on the presence of proliferating cells that resemble the ganglion-like cells observed in proliferative fasciitis of humans. 7 Proliferative fasciitis in humans is defined as a unique pseudosarcomatous fibroblastic proliferative lesion of subcutaneous adipose tissue, occasionally associated with trauma. 12 In our retrospective observation of the skin from Djungarian hamsters with or without pathologic changes in the skin, these cells are often found in the subcutaneous tissue even in the absence of tumors composed of ganglion-like cells. There have been no reports describing these skin ganglion-like cells (SG cells) in Djungarian hamsters. Accordingly, the anatomic location of SG cells in intact male and female Djungarian hamsters was examined in the present study. We also investigated the morphologic characteristics of both SG cells and the proliferating cells in the lesions of proliferative fasciitis in Djungarian hamsters.
Materials and Methods
SG cells from intact Djungarian hamsters
Twenty-four male and 24 female Djungarian hamsters were used in this study. The animals were purchased from Saitama Jikkendobutsu Kyokyusyo (Saitama, Japan) or bred and reared in the air-conditioned animal room in our facility. They were allowed free access to commercially available diet (MF, Oriental Yeast, Tokyo, Japan) and tap water.
One to four hamsters of each sex were examined at 4, 5, 6, and 7 weeks of age and at 2, 3, 4, 5, and 6 months of age. The animals were euthanatized by exsanguination under ether anesthesia, and skin samples were removed from the dorsal and lateral regions of the head, thorax, hip, forelegs, and hind legs and the ventral and medial regions of the abdomen, forelegs, and hind legs. Tissues were fixed in 10% neutral buffered formalin, embedded in paraffin, sectioned at 3 µm, and stained with hematoxylin and eosin (HE). The tissues were examined and classified with respect to the distribution and number of SG cells as follows: −, no SG cells; +, scattered solitary or clustered SG cells; ++, thin layer of SG cells occupying over half of the section examined; +++, thick layer of SG cells occupying the section examined. Representative sections were stained with silver stain, Masson's trichrome, and periodic acid–Schiff (PAS). Frozen sections were also prepared from formalin-fixed tissue and stained with oil red O.
For immunohistochemistry, skin samples from the ventral abdomen of all animals were fixed in Carnoy's solution for 3 hours, embedded in paraffin, and sectioned representative sections were processed for immunohistochemistry using primary antibodies against androgen receptor, collagen type III, estrogen receptor, 47-kd heat shock protein (HSP47), lysozyme, 68-kd neurofilament (NF68), proliferating cell nuclear antigen (PCNA), S-100 protein, α-smooth muscle actin (α-SMA), β-tubulin (class II), and vimentin. Immunolocalization of antigens was visualized by a standard peroxidase anti-phosphatase complex (PAP) method using secondary antibodies against mouse or rabbit immunoglobulin and an anti-mouse or rabbit PAP complex (Dako Japan, Kyoto, Japan) or the avidin-biotin-peroxidase complex (ABC) method (Vector ABC Elite kit, Vector Laboratories, Peterborough, UK) using goat secondary antibodies with diaminobenzidine (Wako Pure Chemical Industries, Osaka, Japan) as chromagen. The primary antibodies and techniques employed are summarized in Table 1. The results were categorized based on the percentage of positively stained cells as follows: − = 0%; + = <30%; ++ = 30–70%; +++ = 70–100%.
The antibodies used in the present study.

For electron microscopy, selected skin samples of the ventral abdomen were fixed in Trump's fixative for <1 month, postfixed with osmium tetroxide, and embedded in Epoxy resin (Quetol-812, Nisshin EM, Tokyo, Japan). Ultrathin sections cut for transmission electron microscope were double stained with uranyl acetate and lead citrate and examined using an H-7100 electron microscope (Hitachi, Tokyo, Japan).
Pathologic examination of proliferative fasciitis in a Djungarian hamster
One subcutaneous mass in the abdomen was surgically removed from a male 11-month-old Djungarian hamster. The freshly removed mass with the adjacent skin was cut into three small pieces and fixed in 10% buffered formalin, Carnoy's solution, or Trump's fixative solution for histology, immunohistochemistry, or electron microscopic examination, respectively.
Results
Occurrence and distribution of SG cells
The anatomic locations of SG cells are listed in Table 2. SG cells were spindle-shaped oval to polygonal, with abundant, often vacuolated basophilic cytoplasm and one or two large round nuclei (Fig. 1). No mitotic figures were observed. In 4-week-old male hamsters, only one male had no SG cells in any region examined (Fig. 2). Clusters of SG cells in the adipose tissue just beneath the dermis were restricted to the abdomen and the medial region of the forelegs in two 4-week-old males. The cells were spindle shaped and were accompanied by delicate intercellular collagen fibers (Fig. 3). These cell clusters had a unique appearance and differed from normal dermal fibroblasts and adipocytes; no transition from these cell types to SG cells was observed. SG cells were observed in all male hamsters ≥5 weeks of age, and the number and extent of SG cells increased with age (Figs. 4, 5). In animals ≥5 months of age, SG cells were often distributed diffusely throughout the skin, replacing subcutaneous adipose tissue, especially in the abdomen. SG cells were observed most often in the ventral abdomen, frequently in the medial portion of the legs, seldom in the lateral regions of the legs and back, and never in the head of males. However, 4-, 5-, and 6-week-old female hamsters did not have SG cells in any portions of the skin examined; older female hamsters had a few SG cell clusters on the abdomen and in the medial regions of the legs.
Occurrence of SG cells in male and female Djungarian hamsters.

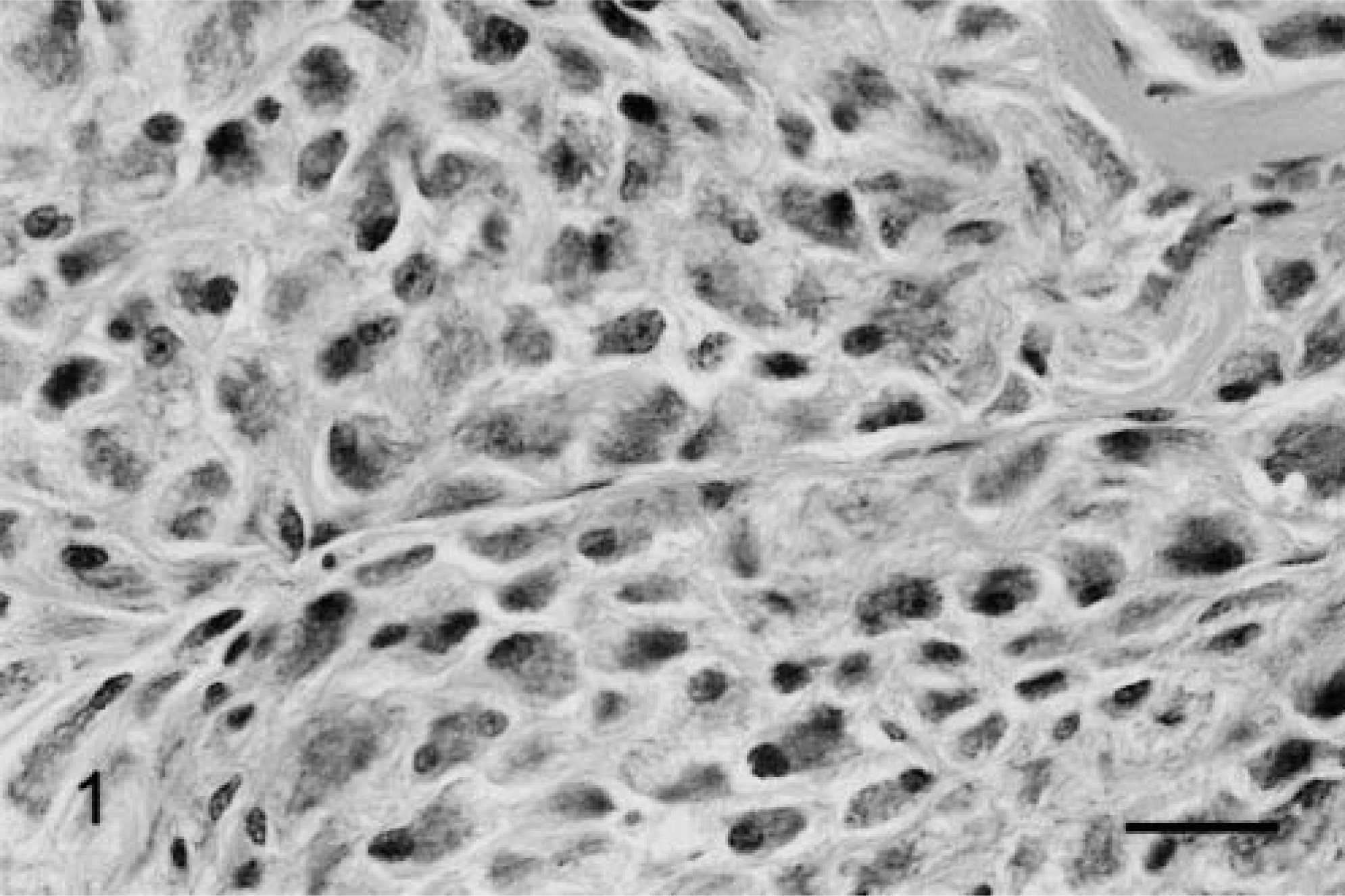
Abdominal skin; 6-month-old male hamster (animal No. 24). Large polygonal SG cells have proliferated and interstitial collagen is present. SG cells possess basophilic and vesicular cytoplasm and one or two large nuclei. HE. Bar = 25 µm.
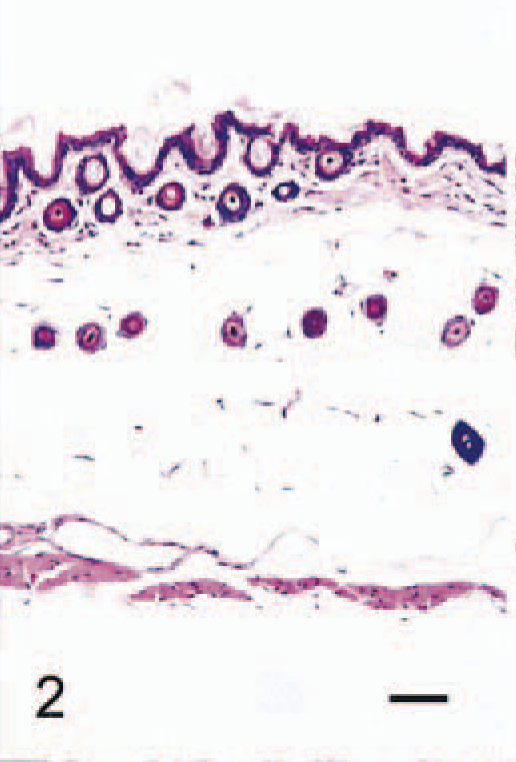
Abdominal skin; 4-week-old male hamster (animal No. 1). The skin consists of thin epidermis, dermis with spindled fibroblasts, collagen and hair roots, subcutaneous adipose tissue with hair roots, vessels and nerves, and muscle layer. HE. Bar = 40 µm.
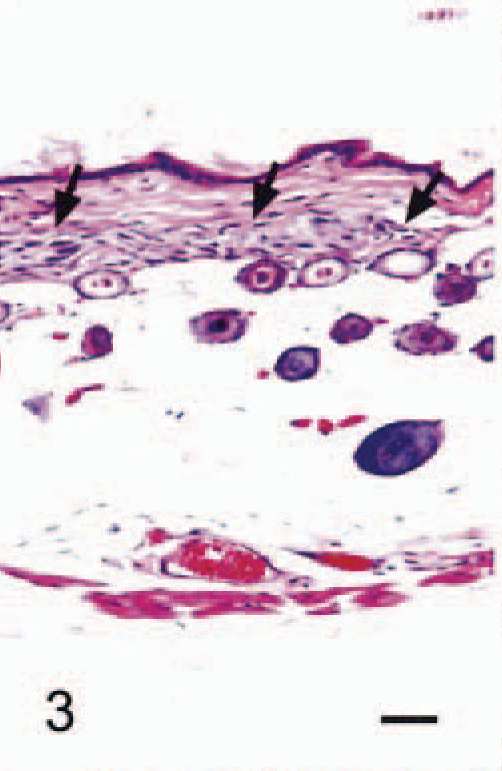
Abdominal skin; 4-week-old male hamster (animal No. 2). The cluster of spindle-shaped SG cells (arrows) is seen just beneath the dermis. HE. Bar = 40 µm.
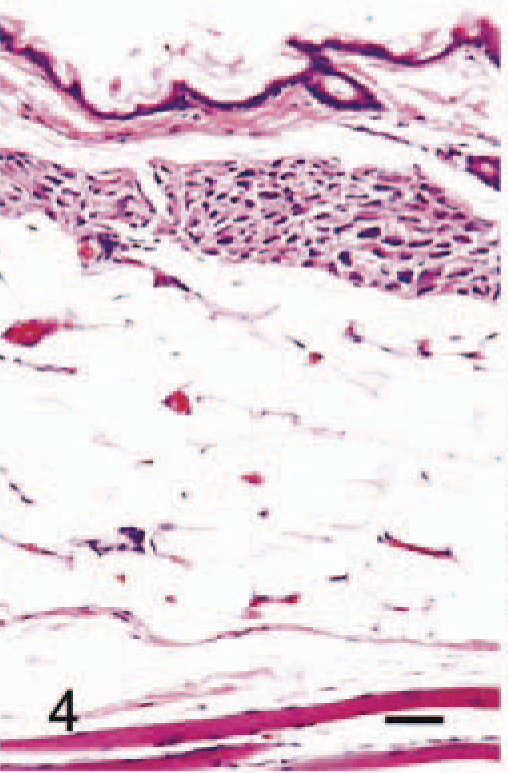
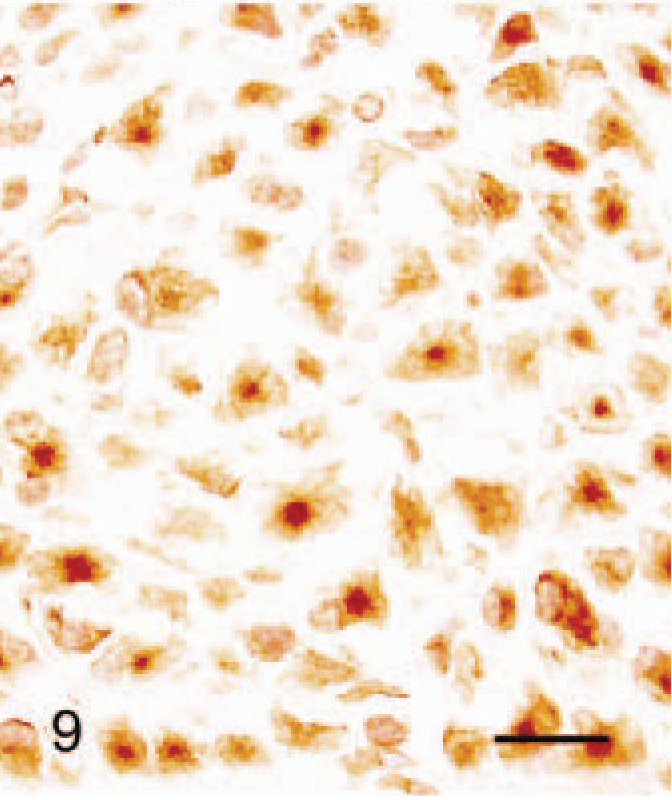
Abdominal skin; 2-month-old male hamster (animal No. 9). The cluster of SG cells is more prominent and is distinguishable from the dermis and subcutaneous adipose tissue. HE. Bar = 40 µm.
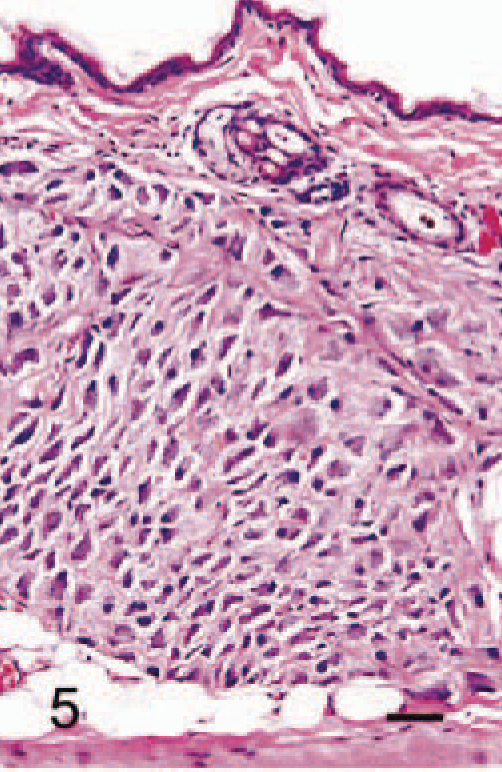
Abdominal skin; 5-month-old male hamster (animal No. 20). SG cells have proliferated extensively, as if replacing the subcutaneous adipose tissue. HE. Bar = 40 µm.
Histologic, immunohistochemical, and ultrastructural characterization of SG cells
Silver stain revealed delicate fibrils in the cytoplasm of SG cells (Fig. 6). Masson's trichrome stain revealed thick bundles of collagen in the interstitium (Fig. 7). PAS-positive granules were seen in the cytoplasm of a few SG cells. Oil red O staining was negative in all SG cells.
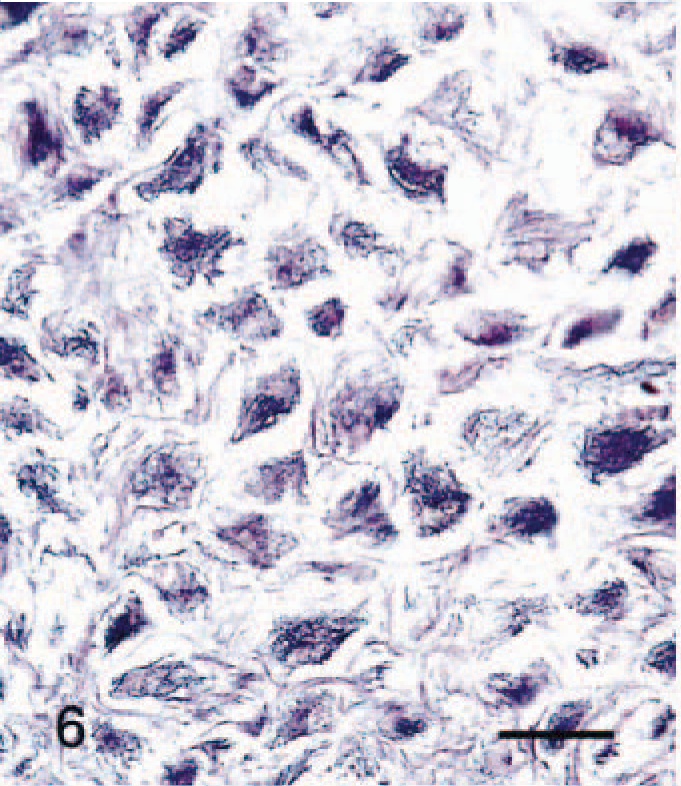
Abdominal skin; 6-month-old male hamster (animal No. 24). Delicate black fibrils are seen in SG cells. Silver stain. Bar = 27 µm.
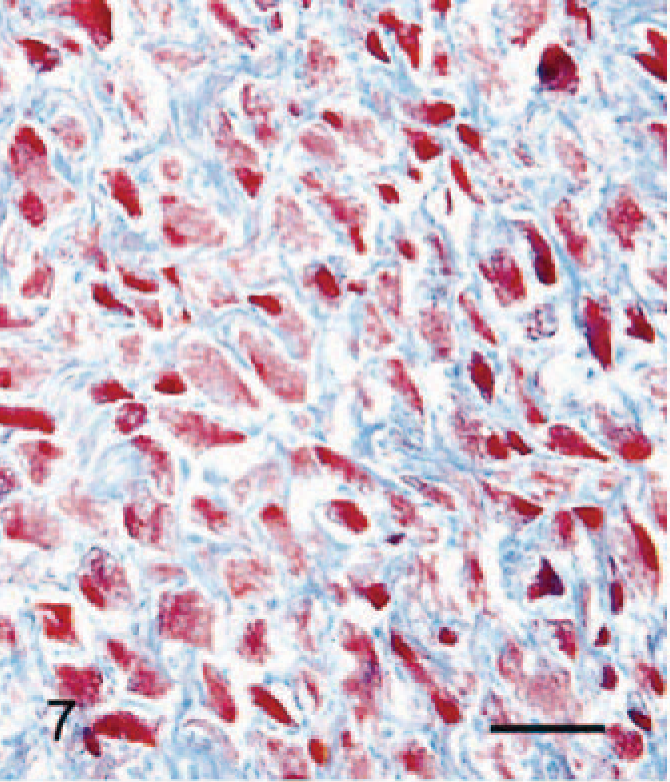
Abdominal skin; 6-month-old male hamster (animal No. 24). Red-stained cytoplasm of SG cells and blue-stained abundant collagen in the interstitium are present. Masson's trichrome stain. Bar = 27 µm.
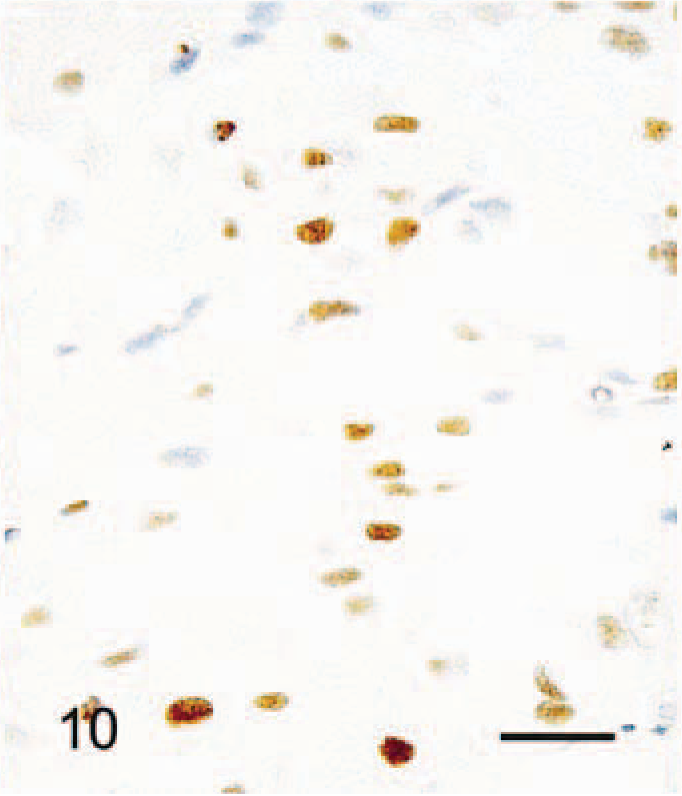
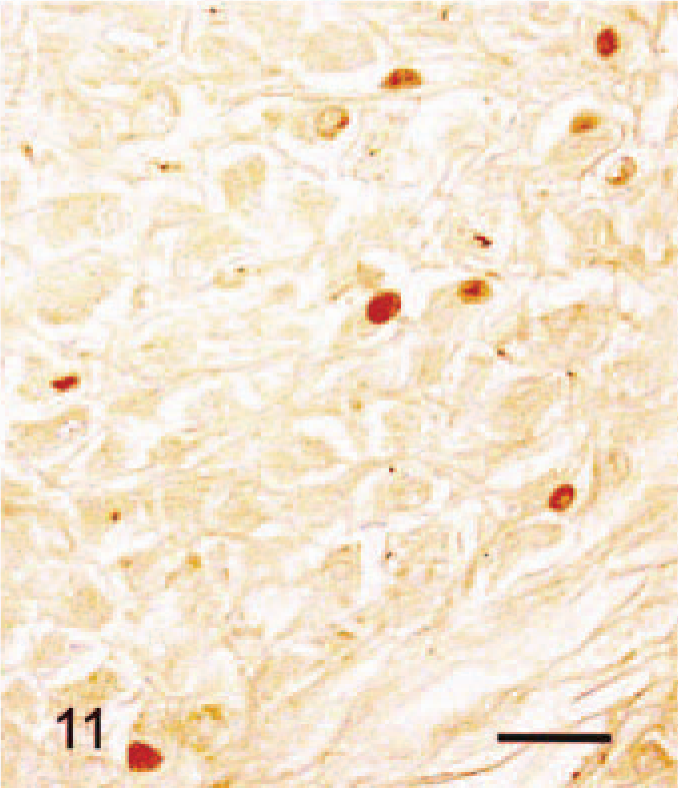
Immunohistochimistry results are given in Table 3. The cytoplasm of all SG cells stained intensely for HSP47 (Fig. 8). Collagen type III, lysozyme, β-tubulin, and vimentin staining were also positive in most SG cells. Punctate β-tubulin immunoreaction was present in some cells (Fig. 9). There was weak focal staining for S-100 protein in the cytoplasm of SG cells. Staining for PCNA and androgen receptor was positive in the nuclei of 70% and 30% of SG cells, respectively (Figs. 10, 11). Staining for estrogen receptor, NF68, and α-SMA was negative in all SG cells.
Results of immunohistochemistry in the SG cells from normal male and female hamsters and tumor cells of proliferative fasciitis from 11-month-old hamsters. ∗

∗ — = 0%; + = <30%; ++ = 30–70%; + + + = 70–100% of SG cells or tumor cells exhibiting positive staining.
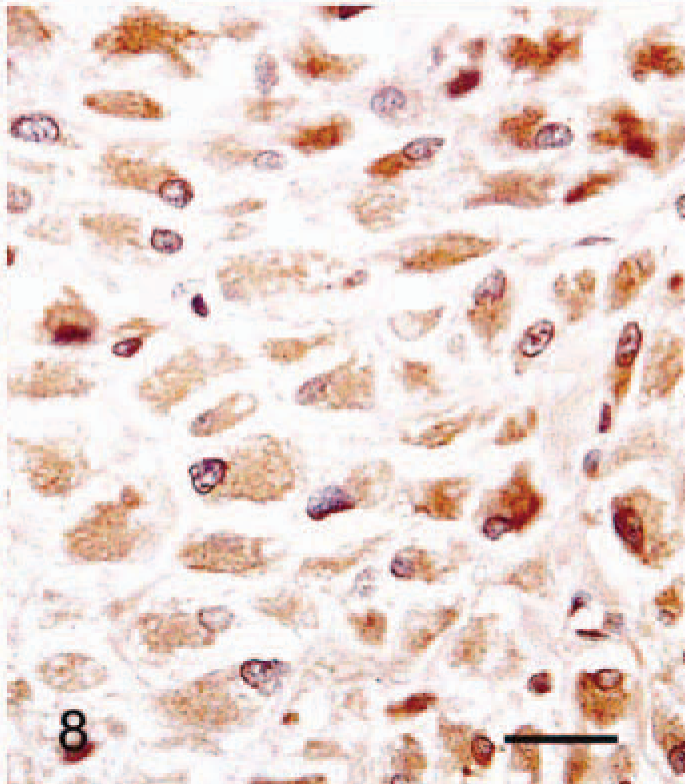
Abdominal skin; 6-month-old male hamster (animal No. 24). All SG cells are positive for HSP47, which has a delicate, fibril-like distribution. ABC method counterstained with Mayer's hematoxylin. Bar = 27 µm.
Abdominal skin; 6-month-old male hamster (animal No. 24). All SG cells are positively stained for β-tubulin as perinuclear spots. PAP method counterstained with Mayer's hematoxylin. Bar = 27 µm.
Abdominal skin; 6-month-old male hamster (animal No. 24). PCNA-positive SG cells are scattered. PAP method counterstained with Mayer's hematoxylin. Bar = 27 µm.
Abdominal skin; 6-month-old male hamster (animal No. 24). Some nuclei of SG cells are positively stained with androgen receptor. No counterstaining. Bar = 27 µm.
Electron microscopic examination revealed that SG cells had abundant rough endoplasmic reticulum and Golgi cisternae (Fig. 12). Cell cytoplasm contained vacuoles of various sizes, which were derived from dilated cisternae or sacs of rough endoplasmic reticulum and Golgi apparatus and contained moderately electron-dense fibrillar material. Prominent fibrils scattered in the cytoplasm of SG cells (Fig. 13) were identified as collagen fibrils based on their diameter of 50–80 nm with 60–70-nm striations. These fibrils were found in nearly all SG cells.
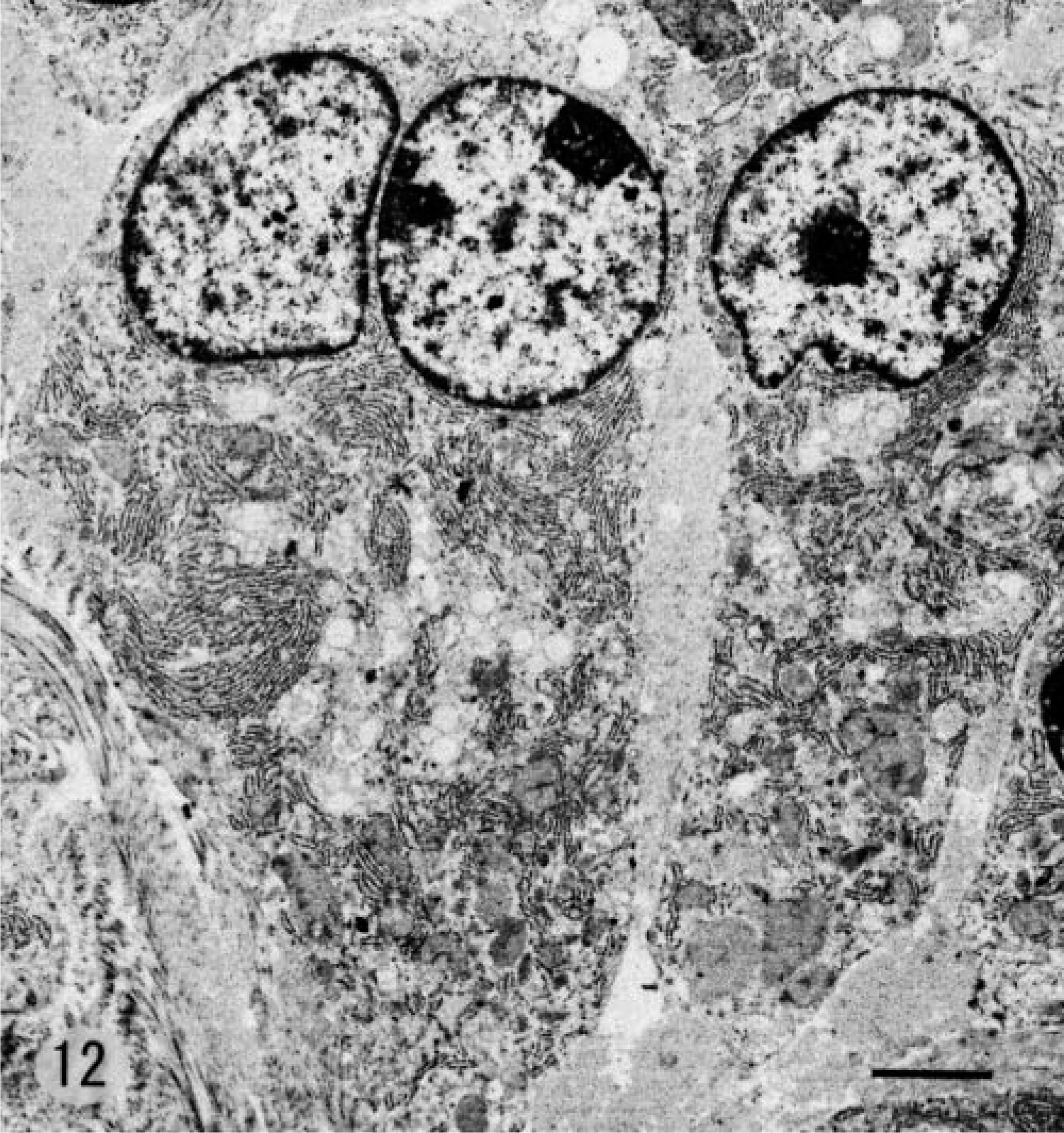
Electron photomicrograph. SG cell; 5-month-old male hamster (animal No. 18). Abundant rough endoplasmic reticulum, Golgi apparatus, and some vacuoles are seen in SG cells. Stained with uranyl acetate and lead citrate. Bar = 2.3 µm.
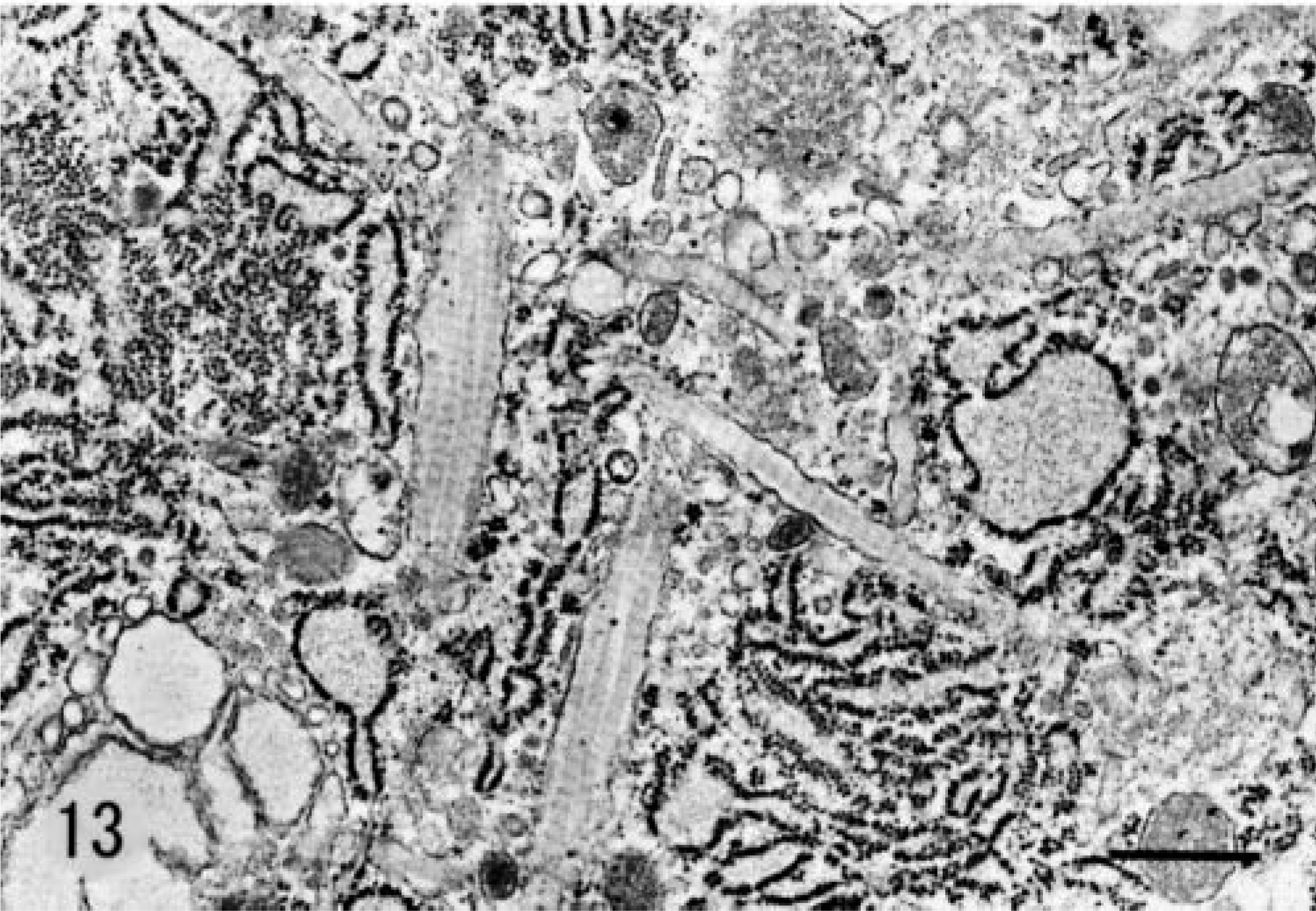
Electron photomicrograph. SG cells; 5-month-old male hamster (animal No. 18). Membrane-bound collagen fibrils are seen in the cytoplasm. Stained with uranyl acetate and lead citrate. Bar = 600 nm.
Lesion of proliferative fasciitis in the Djungarian hamster
Grossly, the 7-mm-diameter mass was oval, milk white, and soft, with a hard core. It was located in the dermis adjacent to subcutaneous adipose tissue. It was circumscribed with a thin collagenous capsule. Histologically, the mass was composed of neoplastic spindle-shaped to polygonal cells with vacuolated basophilic cytoplasm (Fig. 14). Tumor cells resembled subcutaneous SG cells but were arranged more irregularly. Tumor stroma contained abundant hyaline collagen and many capillaries. Staining characteristics of tumor cells were identical to those of normal Djungarian hamster SG cells (Table 3). The ultrastructure of tumor cells was also similar to that of SG cells; the endoplasmic reticulum and Golgi apparatus had dilated cisternae and intracytoplasmic collagen fibrils were present (Figs. 15, 16). The cytoplasm often contained vacuoles of various sizes.
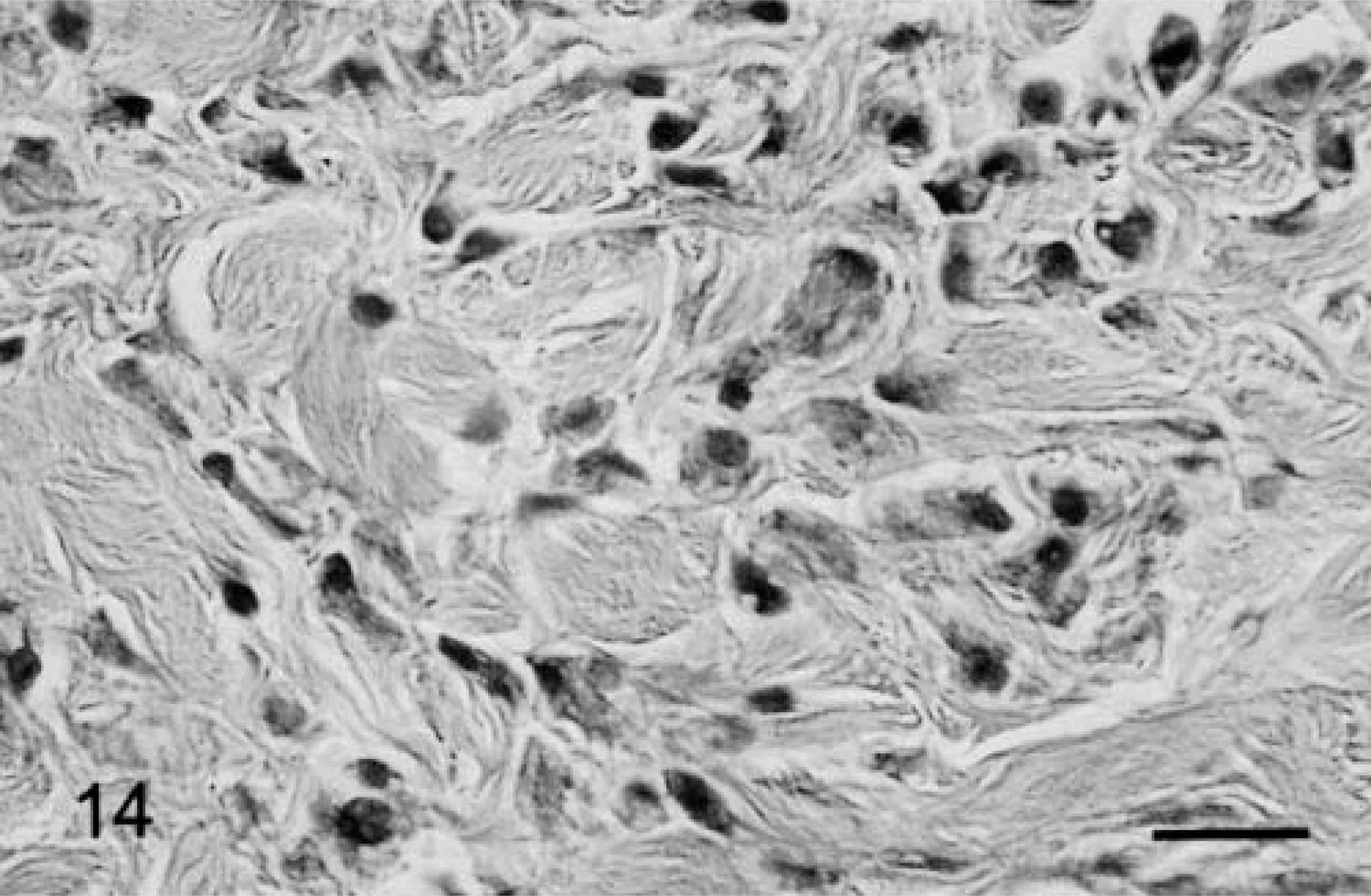
Subcutaneous mass diagnosed as proliferative fasciitis; 11-month-old male hamster. Spindled or polygonal cells have proliferated irregularly with collagen in the interstitium. Bar = 25 µm.
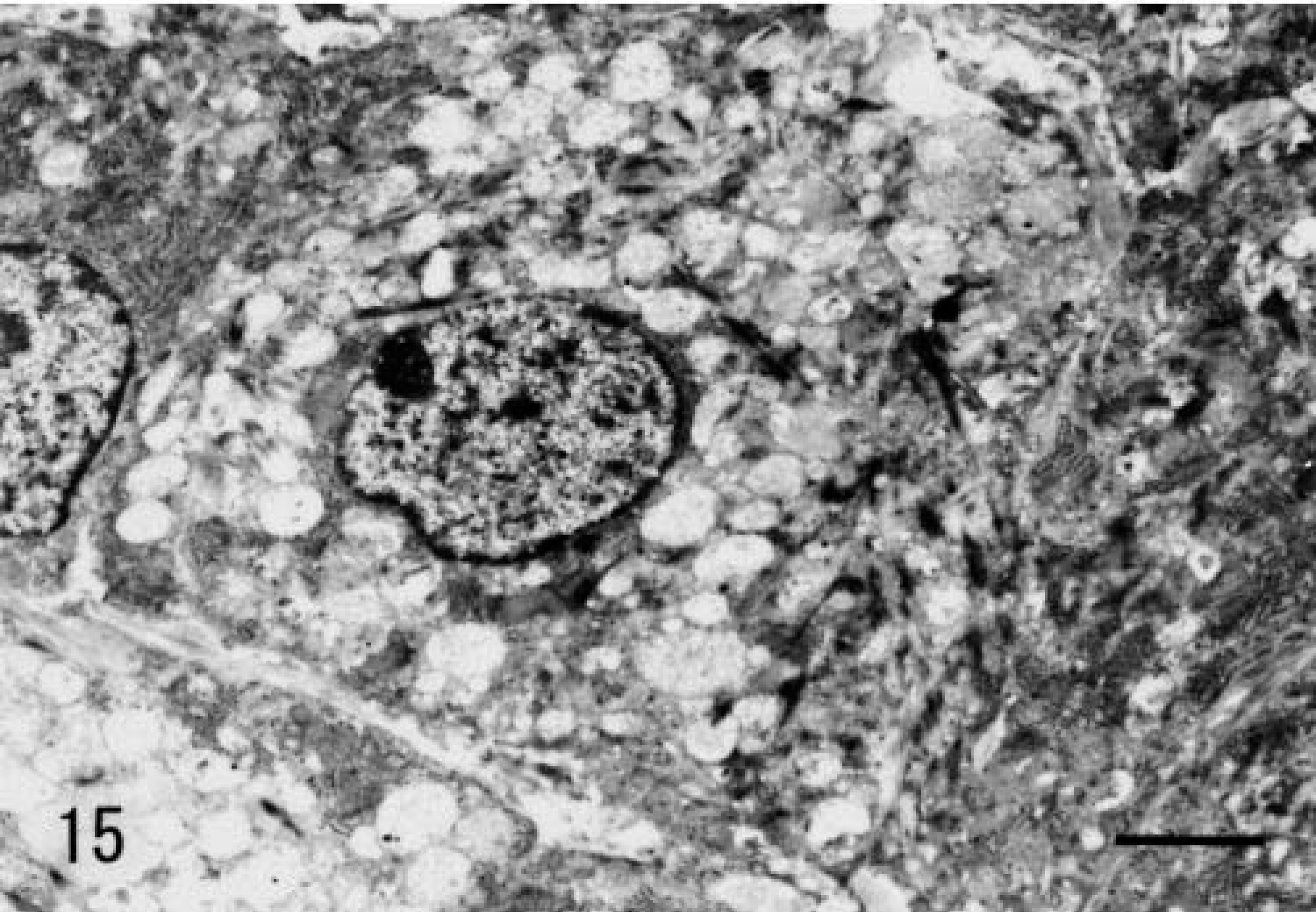
Electron photomicrograph. Proliferating cells in proliferative fasciitis; 11-month-old male hamster. Abundant, various-sized vacuoles; rough endoplasmic reticulum and collagen fibrils are seen in the cytoplasm. Stained with uranyl acetate and lead citrate. Bar = 1.6 µm.
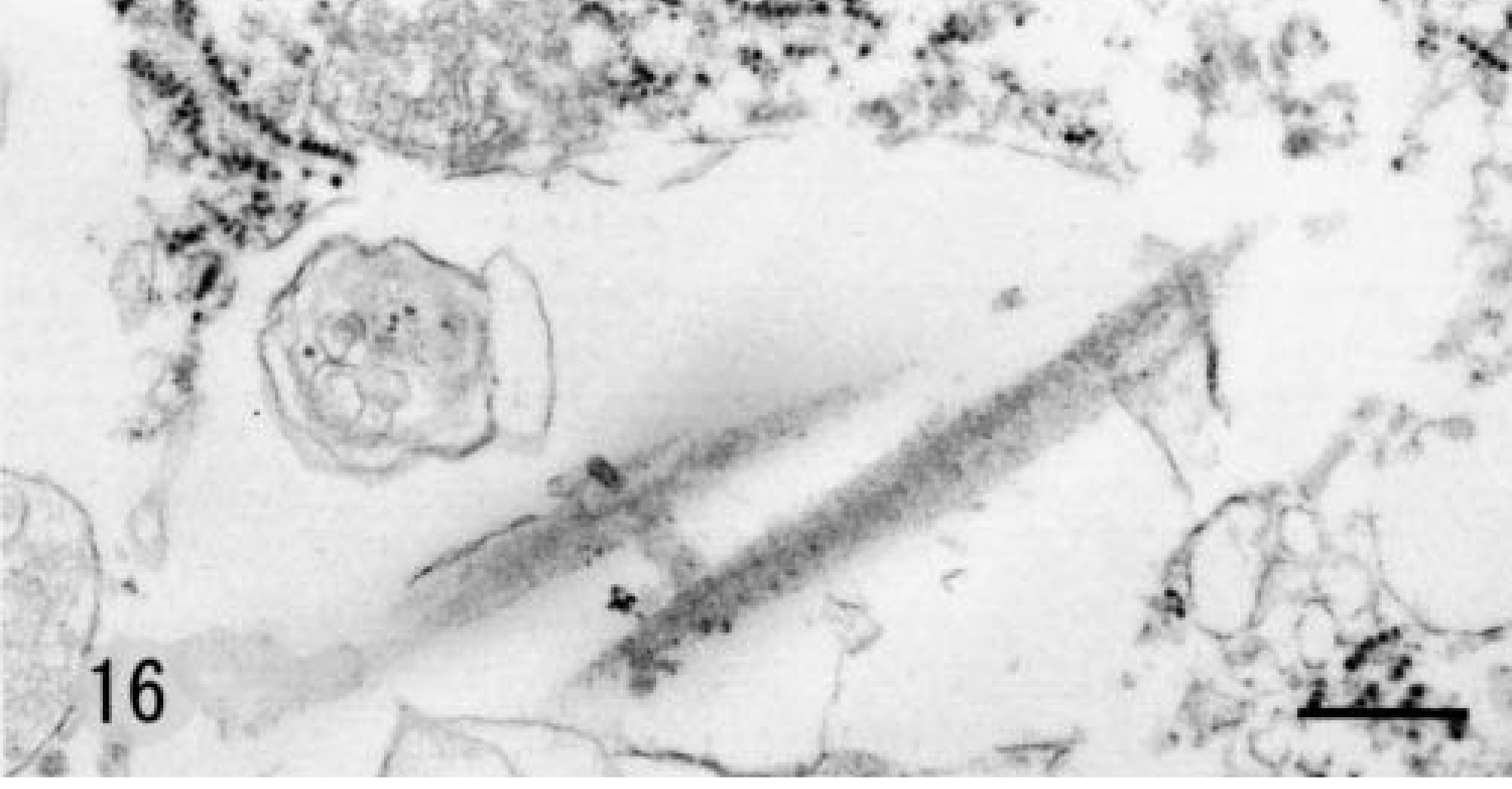
Electron photomicrograph. Proliferating cells in proliferative fasciitis; 11-month-old male hamster. Vacuole contains scattered, fibril-like structures. Stained with uranyl acetate and lead citrate. Bar = 376 nm.
Discussion
In the present study, we unexpectedly observed intracellular collagen fibrils in all SG cells examined. In a normal situation, collagen molecules assemble with collagen-specific periodic striations in the extracellular space after secretion from collagen-producing cells and elimination of collagen propeptide by specific extacellular proteinases. 4 However, intracellular collagen fibrils may be observed in several pathologic conditions, including keloids and some peculiar fibloblastic tumors in humans. 1,8,23 Interestingly, ganglion-like cells in proliferative fasciitis of humans also have intracellular collagen fibrils, and the origin of these cells is considered to be the myofibroblast, based on the presence of actin filaments. 12,16 The origin of SG cells of the Djungarian hamster is different from that of ganglion-like cells in proliferative fasciitis of humans. The ganglion-like appearance reflects active production and retention of collagen in the cytoplasm. Further investigation of the underlying mechanism of intracellular collagen fibril accumulation in SG cells is required to clarify the conditions leading to collagen retention.
Staining for HSP47 and β-tubulin class II in SG cells was prominent. HSP47 is an endoplasmic reticulum–resident chaperone protein specific for collagen. It is involved in collagen processing and secretion under normal conditions 14,18 and acts as a stabilizer for the correctly folded collagen triple helix under conditions of stress, including excessive heat. 15,20 The presence of collagen fibrils within dilated endoplasmic reticulum and Golgi cisternae of SG cells and the pronounced expression of HSP47 in our study strongly suggest excess production and retention of procollagen and enhanced polymerization of collagen. β-Tubulin class II is one isotype of the tubulin family. It is present in undifferentiated mesenchymal cells of fetal tissues and in nerve cells. 2 Based on immunohistological and ultrastructural findings, we determined that the origin of the SG cell is an undifferentiated mesenchymal cell that is distinct from the dermal fibroblast. Tumor cells and SG cells in normal tissue were positive for PCNA, suggesting active proliferation of SG cells. Accordingly, the lesion proliferative fasciitis of Djungarian hamsters is considered a benign neoplastic proliferation of SG cells. Further investigation is needed to clarify the origin of SG cells and the trigger for excessive proliferation of these cells, leading to tumor formation.
The nuclei of SG cells were positive for androgen receptors but not estrogen receptor. In males, dramatic SG cells were numerous and displayed age-related changes. Male Djungarian hamsters reach sexual maturity within 4 weeks after birth. At that time, a spermatogenic wave occurs. 3,21,24 In the present study, we observed only rare SG cells in 4-week-old male and female animals. SG cells increased in number in males after 5 weeks of age, the period of sexual maturation. A variety of skin cells, including fibroblasts and hair follicles, are known to respond to androgen directly or indirectly via insulin-like growth factor or other growth factors. 5,6,17 SG cells appear to be present in normal skin in both males and females, influenced by androgen concentration, resulting in the observed differences between the sexes.
In this study, SG cells were frequently observed in the ventral and medial regions of the trunk and limbs. These areas have the thinnest skin in the body, 13 and the fur in these areas is soft and pale in color. 11 The medial portion of the trunk has the warmest skin surface of the whole body. The normal body temperature of the Djungarian hamster is lower than that of the other rodents. 9 Because they are extremely small and come from a cold and dry environment, they have developed strategies for thermoregulation, including hibernation and shivering. 19,22 Heat stress may result in expression of HSP47 and production of collagen in restricted areas of the body.
The results of the present study suggest that SG cells of Djungarian hamsters are derived from undifferentiated mesenchymal cells, which are found in the dermis or subcutaneous adipose tissue. The function and development of SG cells and the pathogenesis of tumorlike lesions developing from these cells remain unclear. Because the morphologic features of proliferative fascitis observed in humans and the morphology of fibromas are totally different from the morphology of the lesions in the Djungarian hamster, this tumor-like lesion should be tentatively diagnosed as an abnormal proliferative lesion of SG cells.
