Abstract
Mammary tumors that spontaneously occurred in domestic Djungarian hamsters (Phodopus sungorus) were histologically examined. Forty-five mammary tumors included 14 adenomas, 18 adenocarcinomas, 1 lipid-rich carcinoma, 2 adenoacanthomas, 2 malignant adenomyoepitheliomas, 1 benign mixed tumor, and 7 “balloon cell” carcinosarcomas. The latter 4 types were newly recognized neoplasms in Djungarian hamsters. The relatively high incidence of spontaneous mammary carcinosarcomas in domestic Djungarian hamsters is intriguing. Carcinosarcomas exhibited anomalous histological features made up of a mixture of glandular cells, polygonal cells (including “balloon cells”), and sarcomatous spindle cells in varying proportions. Transitional features from glandular cells to polygonal cells and subsequently to sarcomatous spindle cells were observed. Using immunohistochemistry, we observed that glandular cells exhibited an epithelial phenotype (cytokeratin(+)/vimentin(–)), spindle cells exhibited a mesenchymal phenotype (cytokeratin(–)/vimentin(+)), and polygonal cells exhibited an intermediate phenotype (cytokeratin(+)/vimentin(+)). Reduction or loss of β-catenin expression and gain of S100A4 expression were observed in polygonal and spindle cells. The polygonal cell population included a varying number of characteristic cells that were expanded by large intracytoplasmic vacuoles. Electron microscopy revealed that these “balloon cells” had large cytoplasmic lumens lined by microvilli. These observations suggest that epithelial-mesenchymal transition may account for the pathogenesis of mammary carcinosarcomas in Djungarian hamsters.
Djungarian (Siberian or Russian) hamsters (Phodopus sungorus) are popular pets in Japan and belong to a genus unique from that of the Syrian hamster (Mesocricetus auratus). 11 There is a great deal of literature regarding tumors in Syrian hamsters, since this species has long been used in laboratory testing, 7 whereas there few reports regarding tumors in Djungarian hamsters. In Syrian hamsters, tumors of the internal organs such as adrenocortical, gastrointestinal, and hematopoietic tumors are common. 11,17 On the other hand, the prevalence of integumental tumors in Djungarian hamsters is high in comparison with that in Syrian hamsters. 11 Cutaneous squamous cell carcinoma, papilloma, and atypical fibroma that perhaps originate from skin androgen–dependent ganglion cell–like cells are known to be common spontaneous neoplasms in Djungarian hamsters. 1,11
Mammary tumors are also common in domestic and laboratory Djungarian hamsters. 12,17 According to Kondo et al, 12 12/21 (57.1%) tumors from female Djungarian hamsters kept as pets in Japan were mammary tumors. In a Djungarian hamster colony of a Russian laboratory, 72 (55.8%) of the 129 tumors occurring in females originated from mammary glands, and 52 (12.7%) of 409 female Djungarian hamsters had mammary tumors. 17 On the other hand, in some large-scale studies of spontaneous tumors in the Syrian hamster, mammary carcinomas have not been recorded, although there are a few sporadic case reports of mammary tumors of this species. 7 Mammary tumors are one of the most important diseases among females of all species, including humans. The morphology and biological behavior of mammary tumors vary between each species, and interspecies comparative studies may contribute significantly to the understanding of human breast cancer. However, an adequate histological characterization of mammary tumors in Djungarian hamsters has not yet been conducted. Therefore, we reexamined the biopsy specimens of mammary tumors in domestic Djungarian hamsters and discovered several histological types that have not been reported in this species. In particular, attention was given to several carcinosarcomas containing characteristic vacuolated cells that expanded like a balloon (“balloon cells”), with careful evaluation of their histological and immunohistochemical characteristics.
Materials and Methods
For this study, 45 cases of mammary tumors (47 biopsy specimens; 2 were obtained from recurrent tumors) in domestic Djungarian hamsters were reexamined from the archive of the Department of Veterinary Pathology, Nippon Veterinary and Life Science University. All animals were kept as pets in various regions in Japan, and tumor masses had been surgically resected in animal hospitals and submitted to our laboratory from 1998 to 2011. The samples of tumors were processed by routine methods, embedded in paraffin, sectioned at 4 μm, and stained with hematoxylin and eosin (HE). Replicate sections were stained with Alcian blue (AB), and frozen sections were stained with oil red O. Additional serial sections of paraffin tissues were immunostained using a labeled streptavidin-biotin method with mouse monoclonal antibodies against cytokeratin (CK) AE1/AE3, vimentin, α–smooth muscle actin (SMA), p63, β-catenin, and proliferating cell nuclear antigen and rabbit polyclonal antibodies against CK8 and S100A4. Details of the primary antibodies for immunohistochemical analyses are described in Supplemental Table S1. Immunohistochemical statuses of sex hormone receptors were not added in this study, because there were no reliable antibodies against the tissue of Djungarian hamster.
Double-labeled fluorescent immunohistochemistry was used to detect both vimentin and CK. After being subjected to autoclave pretreatment, tumor sections were incubated overnight at 4°C in a mixture of mouse monoclonal antibody against vimentin and rabbit polyclonal antibody against CK (wide-spectrum screening). After washing, the slides were incubated in a mixture of Alexa Fluor 488 goat anti–mouse IgG and Alexa Fluor 568 goat anti–rabbit IgG (both 1:500; Molecular Probes, Eugene, OR) for 30 minutes. The slides were mounted using a mounting medium containing DAPI (Vector Laboratories, Burlingame, CA) and analyzed using a Zeiss Axiovert 200 M fluorescence microscope with an apotome system (Carl Zeiss Japan, Tokyo, Japan).
For electron microscopy, small pieces of the formalin-fixed neoplastic tissue of 1 case (hamster No. 1 of carcinosarcoma) were refixed using 1% osmium tetraoxide in 0.2 M phosphate buffer and embedded in Epon 812. After staining with uranyl acetate and lead citrate, ultrathin sections were examined using a JEM-1011 electron microscope (JEOL, Tokyo, Japan).
Results
Mammary tumors were solitary in all 45 Djungarian hamsters. They were classified into 7 histological types: adenoma (14 cases), adenocarcinoma (18 cases), lipid-rich carcinoma (1 case), adenoacanthoma (2 cases), malignant adenomyoepithelioma (2 cases), benign mixed tumor (1 case), and balloon cell carcinosarcoma (7 cases). The details of cases used in this study are described in Supplemental Table S2.
Adenomas/Adenocarcinomas
Fourteen cases were classified as adenoma and 18 cases were classified as adenocarcinoma. Three cases of adenocarcinoma occurred in male hamsters and the rest in female hamsters. The mean age of Djungarian hamsters with adenoma and adenocarcinoma was 12.0 (range, 5–36) and 13.6 (range, 7–22) months, respectively. The cut surface of the tumor masses was multicystic or solid.
Histologically, the adenomas were well-circumscribed lesions that were composed of tubular, cystic, and papillary epithelial structures (Fig. 1). Epithelial structures were lined with a single layer or pseudostratified epithelium of cuboidal or columnar luminal cells, which exhibited moderate amounts of eosinophilic or amphophilic cytoplasm. In each tumor cell, there was a single round or ovoid nucleus, with an inconspicuous or absent nucleolus. There was very little nuclear and cellular atypia, as well as low mitotic activity. Most luminal epithelial cells had apical cytoplasmic snouts projecting into the luminal spaces (apocrine secretion). The myoepithelium existed as a single continuous layer of cuboidal cells surrounding the luminal epithelium, with weakly eosinophilic cytoplasm and small, hyperchromatic nuclei.
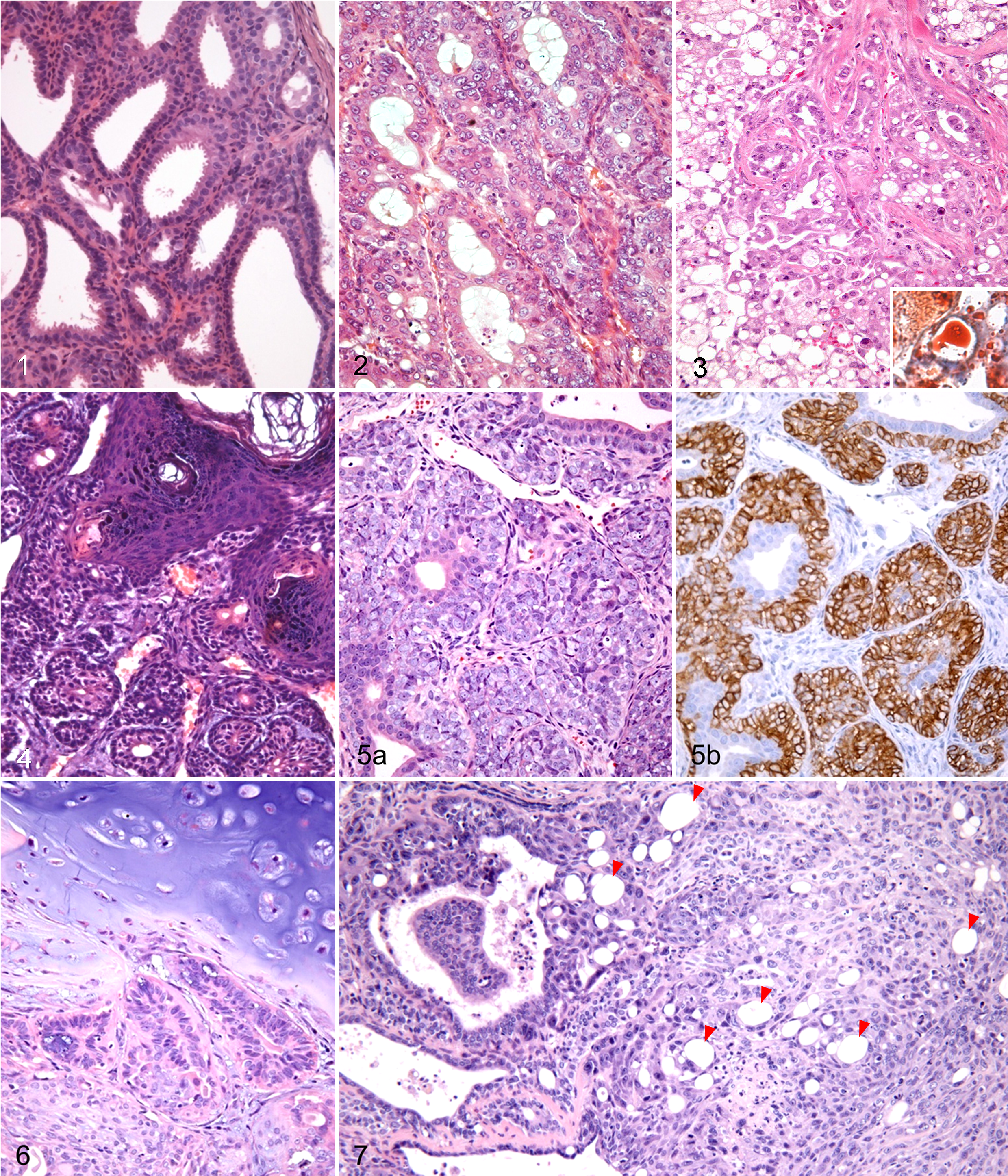
Neoplasms of the mammary gland, Djungarian hamsters.
Most adenocarcinomas were well differentiated, and their cells were morphologically similar to those of adenomas. However, at least focally, adenocarcinomas exhibited increased mitotic activity, nuclear atypia, multilayered epithelium, cribriform growth pattern, and infiltrative growth into the surrounding adipose tissue (Fig. 2). In the specimen from a recurrent tumor, there was a focal solid growth pattern and tumor emboli in the lumen of a blood vessel. Apocrine secretion was evident in the luminal epithelial cells, although to a lesser extent than in adenomas. Focal squamous metaplasia was noted in 4 adenocarcinomas.
Immunohistochemically, luminal epithelial cells of both adenomas and adenocarcinomas were positive for CK8 and negative for p63 and SMA. Myoepithelial cells were positive for p63 and SMA and negative for CK8.
Lipid-Rich Carcinomas
Two specimens that were the primary and recurrent tumors from the same animal were diagnosed as lipid-rich carcinoma. Lipid-rich carcinoma is a rare variant of invasive mammary carcinoma that is composed of cells containing an abundant amount of cytoplasmic neutral lipids (Fig. 3). We have reported this case previously. 28
Adenoacanthomas (Adenosquamous Carcinomas)
Tumors that developed in the mammary gland of a 17-month-old female and a 24-month-old female were diagnosed as adenoacanthoma (adenosquamous carcinoma). Both tumors had ductular and epidermoid structures (Fig. 4), and the ductular structures were lined with a double layer of luminal epithelial and basaloid cells. Approximately half of the tumors’ area displayed well-differentiated squamous components with laminated keratin pearls. The tumor cells exhibited a mild degree of atypia and rare mitotic figures. However, the tumors were considered malignant based on the existence of extensive necrosis and focal infiltration into the surrounding tissue.
Luminal epithelial cells were immunopositive for CK8 and negative for p63 and SMA. Basaloid cells were positive for p63 and negative for SMA and CK8. The squamous epithelium expressed p63 weakly but did not express SMA or CK8.
Malignant Adenomyoepitheliomas
Two specimens from a 15-month-old female and a 24-month-old female were diagnosed as malignant adenomyoepithelioma. Histologically, both tumors were characterized by a proliferation of layers or sheaths of polygonal tumor cells around tubules lined with cuboidal tumor cells (Fig. 5a). Both the polygonal and cuboidal cells exhibited moderate to severe atypia and frequent mitoses. The polygonal cells were immunopositive for p63 and SMA (Fig. 5b) and negative for CK8, indicating a myoepithelial nature. The cuboidal cells were positive for CK8 but negative for p63 and SMA, indicating luminal epithelial differentiation.
Benign Mixed Tumor
A benign mixed tumor of the mammary gland was diagnosed in a female, 12-month-old Djungarian hamster. The well-circumscribed and encapsulated tumor was composed of luminal epithelial and myoepithelial cells with abundant chondroid tissue (approximately 35% of the tumor’s area; Fig. 6). The luminal epithelial component was identical to that of adenomas. The spindle or stellate-shaped myoepithelial cells were arranged in nodular structures, with a pale basophilic, mucopolysaccharide ground substance among the cells.
The immunophenotypes of the luminal epithelial and myoepithelial components of this neoplasm were similar to those of adenoma. However, SMA and p63 immunoreactivity was decreased in spindle- or stellate-shaped myoepithelial cells producing the mucoid substance.
Balloon Cell Carcinosarcomas
Seven cases were diagnosed as balloon cell carcinosarcoma, which we propose as a new category of mammary tumors in Djungarian hamsters. Clinical information for the animals that developed carcinosarcomas is reported in Supplemental Table S3. All hamsters were female, and their mean age was 16.7 months (range, 12–23 months). In 5 cases, the subcutaneous mass had been noted by the owner several months prior and then had either gradually or rapidly grown. In all cases, the cut surface of the resected tumor masses was a doughnut shape, and the central necrotic cavity was filled with purulent-like fluid. This anomalous characteristic resulted in clinicians incorrectly assuming that these masses were bacterial abscesses.
Histologically, tumors infiltrated the normal surrounding tissue and developed extensive central necrosis. In hamster No. 6, tumor emboli were present in the blood vessels. Tumors were composed of glandular cells, polygonal cells (including balloon cells), and sarcomatous spindle cells, and the 3 cell populations were present in varying proportions for each case. Transitional features from glandular cells to polygonal cells and from polygonal cells to sarcomatous spindle cells were observed (Fig. 7). Glandular tumor cells had a histomorphology similar to that of adenocarcinoma. In hamster Nos. 3 and 7, foci of squamous metaplasia of glandular tumor cells were noted. The polygonal cell population had a varying number of characteristic vacuolated cells that had expanded like a balloon, which we called “balloon cells” (Suppl. Figs. S1, S2). Polygonal tumor cells varied in shape, ranging from short-spindle to polygonal and stellate (Fig. 8). Their nuclei were round, oval, or irregular shapes, with prominent nucleoli, and mitotic figures were often observed in these cells. Balloon cells were characterized by 1 to 3 large intracytoplasmic vacuoles, which forced the nuclei to the periphery of the cell (Fig. 9). Special stains and transmission electron microscopy analysis were performed to determine whether the vacuoles of balloon cells were due to fat droplets, intracytoplasmic mucus accumulation, intracellular edema, dilation of subcellular organelles, or intracytoplasmic lumina formation. The contents of the vacuoles stained with AB but not with oil red O. Transmission electron microscopy revealed microvilli lining the vacuoles (Fig. 10), indicating that the vacuoles were intracytoplasmic lumen. Sarcomatous spindle cells were arranged in a fascicular to storiform, herringbone pattern, with dispersed collagen bundles (Fig. 11 and Suppl. Fig. S3). In hamster No. 4, cartilaginous and osseous metaplasia was observed within the area where the sarcomatous spindle cells proliferated (Suppl. Fig. S4). Sarcomatous spindle cells varied in size and had a moderate amount of cytoplasm and round, oval, or polyhedral nuclei with prominent mitotic figures (Fig. 12).
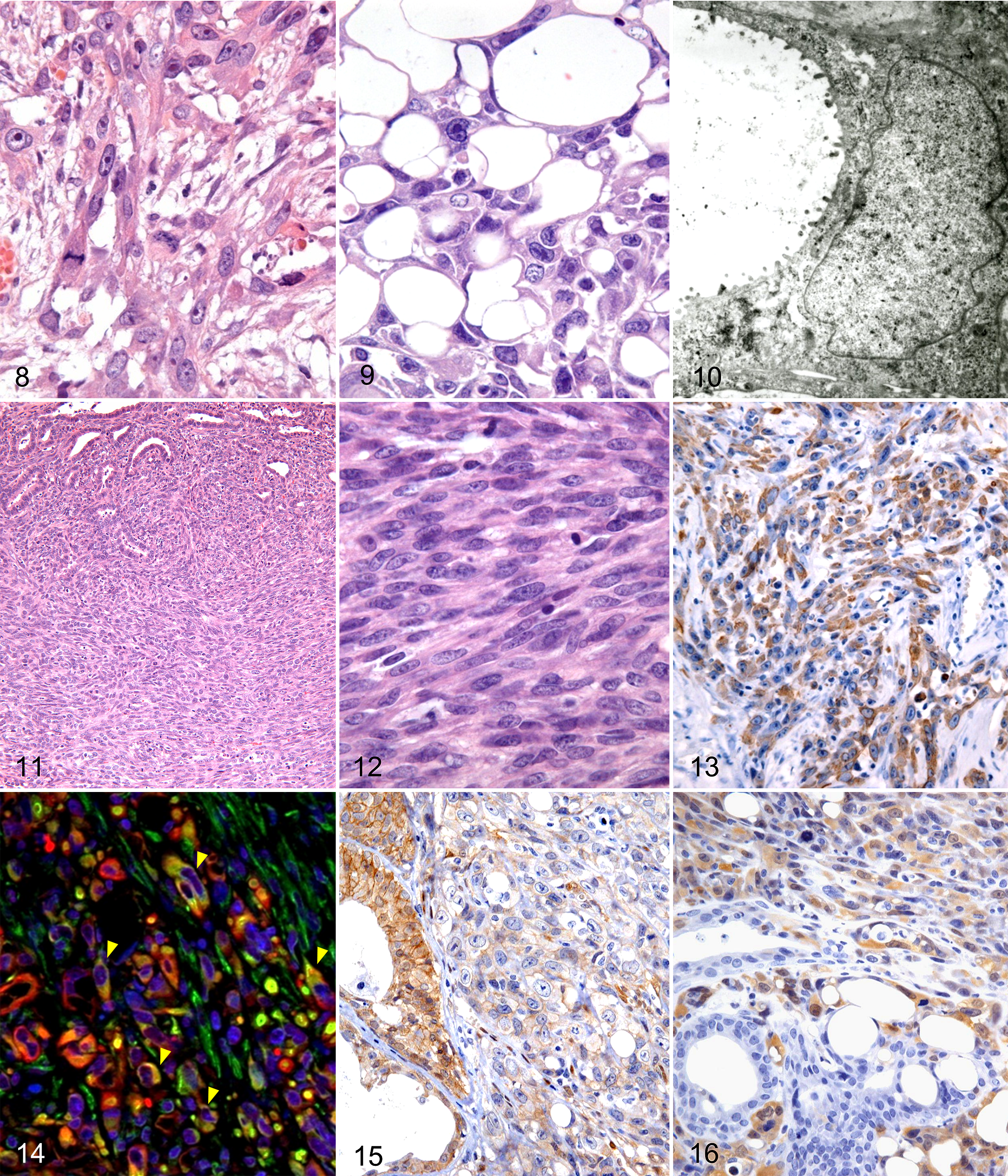
Carcinosarcoma, mammary gland, Djungarian hamsters.
The immunohistochemical results are summarized in Table 1. For CK AE1/AE3 and CK8, glandular cells were strongly positive, most polygonal cells (including balloon cells) were also positive (Fig. 13 and Suppl. Fig. S5), and sarcomatous spindle cells were negative (Suppl. Fig. S6). For vimentin, glandular cells were negative, while some polygonal cells (Suppl. Fig. S7) and all sarcomatous spindle cells were positive (Suppl. Fig. S8). A double immunofluorescence technique for simultaneous detection indicated that some polygonal cells coexpressed CK and vimentin (Fig. 14). For SMA, all glandular cells and polygonal cells were negative, and some sarcomatous spindle cells (hamster Nos. 1–3) were positive. For p63, only a few of the polygonal cells in hamster No. 4 were positive. Expression of β-catenin in the membrane was evident in glandular cells. However, polygonal cells and sarcomatous spindle cells exhibited reduced (or total loss of) membrane expression of β-catenin and/or low cytoplasmic and nuclear expression (Fig. 15). Glandular cells were S100A4 negative, whereas polygonal cells and sarcomatous spindle cells often exhibited cytoplasmic and/or nuclear immunoreactivity to S100A4 (Fig. 16). All 3 cell populations (glandular, polygonal, and spindle) exhibited high proliferative activity, as labeled by proliferating cell nuclear antigen.
Immunohistochemical Properties of Each Cell Population in Balloon Cell Carcinosarcoma.
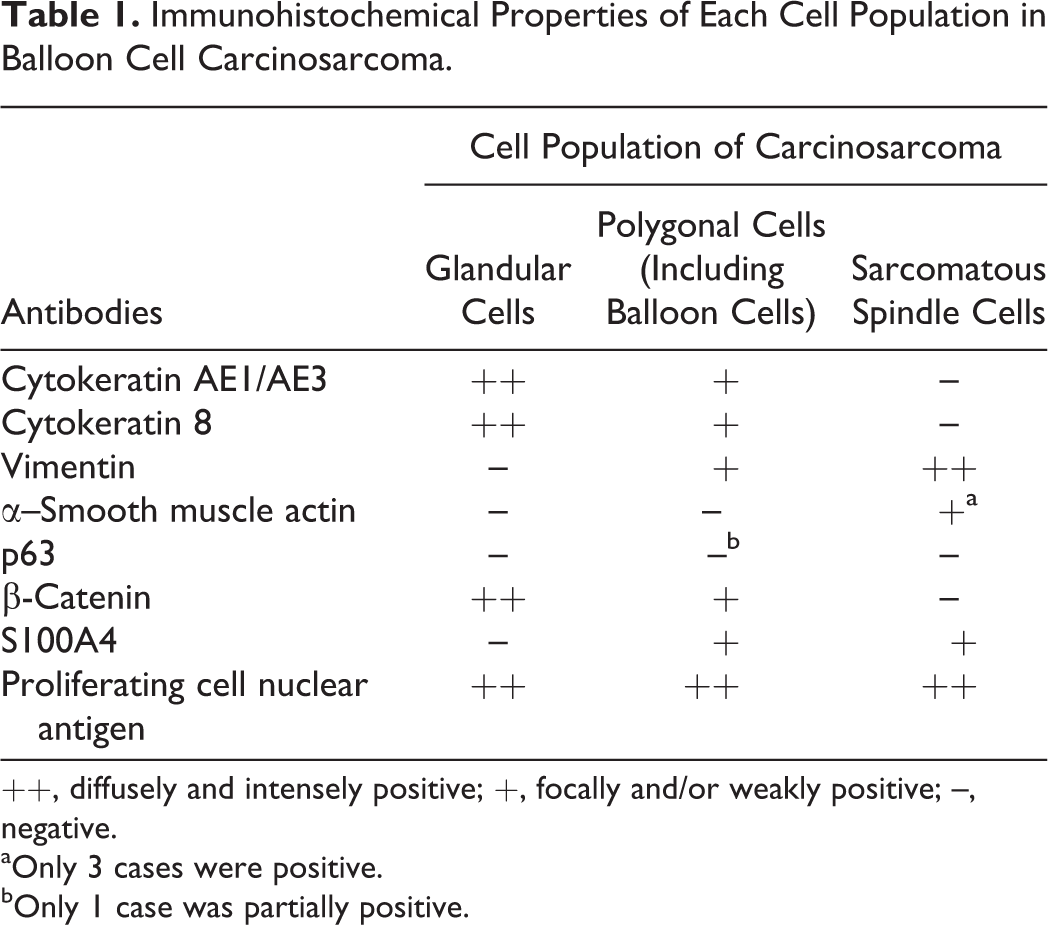
++, diffusely and intensely positive; +, focally and/or weakly positive; –, negative.
aOnly 3 cases were positive.
bOnly 1 case was partially positive.
Discussion
In this study, we histologically classified 45 mammary tumors from Djungarian hamsters into 7 histological types: adenoma, adenocarcinoma, lipid-rich carcinoma, adenoacanthoma, malignant adenomyoepithelioma, benign mixed tumor, and balloon cell carcinosarcoma. In Djungarian hamsters, it is known that mammary tumors tend to develop at a younger age compared with other tumor types. 11 We observed that the mean ages of Djungarian hamsters with benign types of mammary tumors (adenoma and benign mixed tumor) were lower than those of malignant types (adenocarcinoma, lipid-rich carcinoma, adenoacanthoma, malignant adenomyoepithelioma, and balloon cell carcinosarcoma). In addition, the size of tumor masses among benign histological types also tended to be smaller than those of malignant histological types.
Apocrine secretion was noted in most glandular components of the mammary tumors, including adenomas and adenocarcinomas, which is consistent with previous reports of prominent apocrine secretion in mammary tumors of rodents. 7,20,24 Apocrine secretion was also reported in all 12 cases in a series of Djungarian hamster mammary tumors, and a relationship with the expression of androgen receptor was suggested. 12
Adenoacanthoma is an unusual histological type in mouse mammary tumors, which exhibits not only glandular growth but also extensive areas of epidermoid structure. 19,22 In the present series of mammary tumors in Djungarian hamsters, there were 2 cases that histologically resembled mouse adenoacanthoma. In mouse adenoacanthoma, the squamous epithelial area accounts for more than 25% of the whole tumor, and if the area accounts for less than 25%, it is referred to as regional squamous metaplasia. 6 Two cases in the present study had squamous epithelial areas of approximately 50% and therefore satisfy the criterion for a diagnosis of adenoacanthoma.
Two additional tumors were characterized by the biphasic proliferation of luminal epithelial and myoepithelial components. Biphasic luminal epithelial-myoepithelial mammary tumors are common in dogs (complex adenoma/carcinoma as well as carcinoma and malignant myoepithelioma) 8,16 but are rare in other animals, such as humans (pleomorphic adenoma and adenomyoepithelioma) 13 and rodents (adenomyoepithelioma). 3,22 The current 2 cases had polygonal myoepithelial tumor cells with round nuclei and did not exhibit myxomatous change. Their histology resembled human malignant adenomyoepithelioma as well as canine carcinoma and malignant myoepithelioma, rather than the typical pattern of canine complex carcinoma. Therefore, we diagnosed these cases as malignant adenomyoepithelioma. One previous report has described a mammary tumor consistent with the typical histology of canine complex carcinoma in a Djungarian hamster. 12
One case was diagnosed as a benign mixed tumor that was morphologically similar to the canine counterpart. 16 The proliferation of spindle-shaped myoepithelial cells that produced the myxoid substance was recognized in the area surrounding cartilaginous tissues. The myoepithelial cells may be related to cartilaginous metaplasia as has been suggested in dogs. 16
In the present series, there were 7 cases of tumors composed of an admixture of malignant glandular and polygonal-spindle cell components, representing the histology of carcinosarcoma. The term carcinosarcoma was first used by Virchow in 1864, 25 and several theories have been proposed to explain the coexistence of carcinomatous and sarcomatous components in carcinosarcoma. In 1920, Meyer 15 proposed the collision theory, combination theory, and composition theory to explain the histogenesis of carcinosarcoma. Collision tumors are tumors formed by the merging of 2 malignant cell populations of distinct origins. Combination tumors are lesions composed of 2 cell populations that arise from a common cell (stem cell). Composition tumors are malignant tumors from the epithelial component of the parenchyma, with a subsequent pseudosarcomatous change in the stroma. Currently, most researchers agree that most carcinosarcomas are monoclonal neoplasms that derive from a single epithelial cell (or stem cell), as these tumors exhibit a transition from carcinomatous to sarcomatous components, which is supported by morphological and immunohistochemical evidence. 26,27,29 Therefore, most classic carcinosarcomas that arise from various human organs are currently renamed as sarcomatoid carcinoma, undifferentiated (anaplastic) carcinoma, pleomorphic carcinoma, metaplastic carcinoma, or spindle cell carcinoma. 4,13,27,29 Similar terminological changes have also been suggested for mouse mammary carcinosarcomas, 2,5,18 as genetically engineered mice develop mammary carcinosarcomas with areas of epithelial to spindle cell transition. The morphology is reminiscent of the epithelial-mesenchymal transition (EMT), which occurs during embryonic development. Therefore, some researchers refer to these murine mammary carcinosarcomas as EMT-phenotype tumors. 2,5,18
Similarly, in these 7 cases of Djungarian hamsters, we recognized features indicating a transition from glandular cells to polygonal cells and subsequently to sarcomatous spindle cells. In addition, polygonal cells exhibited an intermediate immunophenotype (CK(+)/vimentin(+)) between that of the epithelial and mesenchymal immunophenotypes. Moreover, reduction or loss of β-catenin, an adhesion molecule in epithelial cells, was evident in the polygonal cells, which also occurs during EMT. In addition, polygonal cells expressed S100A4, a key player in EMT. 21 These findings suggest that a glandular tumor developed initially, and a portion subsequently transitioned to polygonal cells and subsequently to sarcomatous spindle cells, as observed in human and mouse carcinosarcomas.
Mammary carcinosarcomas in Djungarian hamsters were characterized by balloon cells admixed with the polygonal cell population. The balloon cells were often positive for CK8, which is a marker for glandular epithelial cells, and their vacuoles were lined by microvilli. These findings suggested that these cells were in an early stage of EMT and had lost the ability to construct glandular structures but barely retained the ability to form a lumen within the individual cells. It has been reported that the vacuoles of some signet ring cell carcinomas of the human breast, prostate, and urinary tract are also intracytoplasmic lumens. 9,10,14,23
As mentioned above, we demonstrated the existence of various histological types of mammary tumors in Djungarian hamsters. However, it is surprising that carcinosarcomas accounted for 15.6% (7 of 45 cases) of all spontaneous mammary tumors, as their occurrence is very rare in humans, dogs, and cats. 13,16 Since mammary carcinosarcoma in Djungarian hamsters has histological and immunohistochemical features suggestive of EMT, the term EMT-phenotype tumor may be a more appropriate diagnostic term, as it is also used for mouse mammary carcinosarcomas.
Footnotes
Acknowledgements
We thank Mr. Akira Tanaka (Department of Veterinary Pathology, Nippon Veterinary and Life Science University) for his excellent technical assistance.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported, in part, by a Grant-in-Aid from Japan Society for the Promotion of Science to Hisashi Yoshimura (No. 26870622).
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
