Abstract
The objective of this study was to determine whether vaccination with bacterins commonly used in the USA, when administered at a time typical of US protocol, enhances porcine circovirus type 2 (PCV2) replication and the incidence and severity of clinical signs and lesions characteristic of postweaning multisystemic wasting syndrome (PMWS) in conventional pigs. Sixty-one pigs free of PCV2 were randomly assigned to four groups. Groups 1 (n = 15) and 2 (n = 15) pigs served as sham-inoculated negative controls. Groups 3 (n = 14) and 4 (n = 17) pigs were inoculated intralymphoid with PCV2 field isolate ISU-40895. Pigs in groups 2 and 4 were vaccinated with Actinobacillus pleuropneumoniae (APP) and Mycoplasma hyopneumoniae (M. hyopneumoniae) bacterins 21 days before and again 1 day before inoculation with PCV2. Mild transient respiratory disease and diarrhea were observed from 13 to 34 days postinoculation (DPI) in pigs in groups 3 and 4. Half the pigs from each group were necropsied at 22 and 34 DPI, respectively. Moderately enlarged, tan-colored lymph nodes were observed in the majority of pigs in groups 3 and 4. There was a significantly (P < 0.05) longer length of viremia (2.14 ± 0.26 versus 4.44 ± 0.23 weeks), a higher copy number of the PCV2 genome in serum, a wider range of tissue distribution of PCV2 antigen, and an increased severity of lymphoid depletion in pigs vaccinated with commercial APP and M. hyopneumoniae vaccines and inoculated with PCV2 compared with PCV2-inoculated unvaccinated pigs. Swine producers and veterinarians may need to consider changes in vaccination protocols in herds with recurrent PCV2-associated PMWS.
Two types of porcine circovirus (PCV), referred to as type 1 PCV (PCV1) and type 2 PCV (PCV2), have been characterized in pigs. 5,24 PCV is a very small, nonenveloped, single-stranded DNA virus with a circular genome 33 and belongs to the Circoviridae family. 23 PCV1 was first recognized in 1974 as a cell culture contaminant. 35 Experimental PCV1 infections have demonstrated no effect in pigs; thus, PCV1 is considered to be nonpathogenic. 3,34 PCV2 was first recognized to be associated with postweaning multisystemic wasting syndrome (PMWS) in 1996. 12 PMWS is characterized clinically by enlargement of the lymph nodes, chronic pneumonia, progressive weight loss, and less commonly by icterus, pallor, or diarrhea. Characteristic microscopic lesions include lymphoid depletion and histiocytic replacement of follicles in lymphoid tissues. Mild-to-severe granulomatous inflammation in lymphoid and other tissues also is commonly observed. Macrophages in affected lymphoid tissues may contain basophilic inclusion bodies. 1,31
Serologic studies demonstrate that PCV2 is ubiquitous in the swine population, and essentially all herds are infected. 22,36 In contrast, PMWS is manifested in a relatively low percentage of pigs in PCV2-infected herds. 1 It has been demonstrated that other coinfections such as porcine reproductive and respiratory disease virus (PRRSV) 4,14,30 or porcine parvovirus (PPV) 2,16,18 may potentiate the development of PMWS in PCV2-infected pigs.
Recent studies have demonstrated that immune stimulation may trigger progression of PCV2 infection to disease and lesions characteristic of PMWS. Krakowka et al. 17 reproduced PMWS in gnotobiotic pigs stimulated with keyhole limpet hemocyanin in Freund's incomplete adjuvant (KLH–ICFA) and infected with PCV2. Allan et al. 6 observed PMWS in 21% of PCV2-infected and vaccinated (Mycoplasma hyopneumoniae [M. hyopneumoniae] at 13 and 31 days of age, and Actinobacillus pleuropneumoniae [APP] at 47 days of age) colostrum-fed pigs. In contrast, another recent study using conventional pigs failed to demonstrate an immune-stimulation–dependent difference in pigs inoculated with PCV2 and stimulated with KLH–ICFA. 20 These experimental models suggest that immune stimulation may in some cases increase the incidence and severity of PMWS in PCV2-infected pigs.
Field trials further supported this hypothesis. Vaccination of piglets with a M. hyopneumoniae bacterin at 1 and 4 weeks of age resulted in a significant increase in losses associated with of PMWS in the vaccinated pigs in two of five batches. 7 Kyriakis et al. 19 vaccinated a portion of the pigs in a herd experiencing a PMWS outbreak with a M. hyopneumoniae vaccine and observed PMWS in 43% of the vaccinated pigs compared with 11% of the nonvaccinated pigs.
The objective of this study was to determine whether vaccination with bacterins (APP and M. hyopneumoniae) commonly used in the USA at a time typically administered in USA enhances the incidence and severity of PCV2-associated PMWS lesions in conventional pigs.
Materials and Methods
Animal source
Sixty-one pigs were weaned at 12–14 days of age and brought to the livestock infectious disease isolation facility at Iowa State University. All pigs were tested by enzyme-linked immunosorbent assay (ELISA) and confirmed to be negative for antibodies to PCV2. 28 The pigs were assigned randomly to rooms and pens and were housed in two rooms, with four pens in each room.
Experimental design
The experimental design is shown in Table 1. Pigs were 7–10 weeks of age when inoculated with PCV2. For the sham-inoculation, the pigs in groups 1 and 2 received a similar volume of a 0.9% sodium chloride solution (Lot 65-181-DK; Abbot Laboratories, North Chicago, Illinois). Half the pigs were necropsied at 22 days postinoculation (DPI) and the remaining pigs at 34 DPI.
Experimental design.

∗Vaccination with Mycoplasma hyopneumoniae and Actinobacillus pleuropneumoniae bacterins.
† Days postinoculation.
Vaccines
A Mycoplasma hyopneumoniae (M. hyopneumoniae) bacterin (M+PAC®, Schering-Plough Animal Health, Omaha, Nebraska; Lot EX-0061-1108-01) and an APP bacterin (Serotypes 1, 5, and 7, PNEU PAC®, Schering-Plough Animal Health, Lot NDC-0061-0260-01) were used for immune stimulation. The APP vaccine was adjuvanted with emulsified paraffin and the M. hyopneumoniae bacterin contained Emunade®, a combination of aluminum hydroxide with oil-in-water dual-action adjuvant. The first dose of each vaccine was given 21 days before and the second dose was given 1 day before PCV2 inoculation. Each pig was vaccinated intramuscularly in the right neck with 2 ml of the APP and in the left neck with 1 ml of the M. hyopneumoniae bacterins according to the manufacturer's directions.
Virus inoculum
The PCV2 isolate used in this study, field isolate ISU-40895, was originally obtained from a pig with PMWS in a herd in western Iowa with mild-to-moderate losses associated with PMWS. 9 The virus preparation was done on PK-15 cells free of PCV1 and PCV2, as described previously. 10 Briefly, PK-15 cells were cultivated in T-25 culture flasks and transfected with the PCV2-infectious DNA clone. 10 The transfected cells were harvested at 3 days posttransfection by freezing and thawing at −80 C three times. The infectious titer of the inoculum was determined by immunofluorescence assay as described previously. 10 The virus inoculum was stored at −80 C until use. Each pig in groups 3 and 4 received 8 ml of isolate ISU-40895 with a titer of about 103. 5 tissue culture infective dose50 intralymphoid. One half of the inoculum was injected in the right and the other half in the left superficial inguinal lymph node. 10
Clinical evaluation
The pigs were evaluated daily for clinical signs, including respiratory disease (score ranging from 0 [normal breathing] to 6 [severe dyspnea and abdominal breathing]). 11 Nasal discharges and coughing were recorded by observers along with other changes such as lethargy. Rectal temperatures were recorded daily. The pigs were weighed at 20 and 3 days before inoculation and at 22 and 34 DPI.
Serology
Serum samples were taken before inoculation and at 7, 13, 21, 28, and 33 DPI. A PCV2-ELISA based on the recombinant open reading frame 2–capsid protein of PCV2 was performed on serum samples of all pigs as described previously. 28 Samples with a sample-to-positive (S : P) ratio ≥ 0.2 were considered to be positive for antibodies to PCV2.
In addition, serum of all pigs was tested for the presence of specific antibodies to porcine parvovirus by hemagglutination inhibition (HI), 25 and for the presence of PRRSV-specific antibodies by PRRSV ELISA (IDEXX Laboratories, Inc. Westbrook, MA) on 33 DPI.
Gross pathology and histopathology
At necropsy, macroscopic lung lesions (0–100% of the lung affected) and size of lymph nodes (score ranging from 0 [normal] to 3 [three times the normal size]) were determined. Bronchoalveolar lavage (BAL) for bacterial and virologic examinations was collected as described previously. 26
Tissue samples from lymph nodes (superficial inguinal, mediastinal, tracheobronchial, and mesenteric), lung, tonsil, thymus, ileum, kidney, colon, skin, spleen, liver, stomach, bone marrow, and ribs were collected during necropsy and fixed in 10% formalin and routinely processed for histologic examination. Microscopic lesions were evaluated blindly by a veterinary pathologist.
Immunohistochemistry
Immunohistochemistry (IHC) for detection of PCV2-specific antigen was performed on sections of paraffin blocks of selected tissue samples (lymph nodes, including superficial inguinal, mediastinal, tracheobronchial, and mesenteric, tonsil, and spleen) using a rabbit polyclonal antiserum as described previously. 32 The amount and distribution of PCV2 antigen was compared by evaluation of IHC signals of each tissue section ranging from 0 (= no signal) to 3 (= strong positive signals) in a blinded fashion. The mean group score was determined for each lymphoid tissue and compared between groups.
Polymerase chain reaction
To confirm the presence of PCV2 infection and for the assessment of the length of PCV2 viremia, polymerase chain reaction (PCR) was performed on serum samples collected at different DPIs. Deoxyribonucleic acid (DNA) from weekly serum samples was extracted using a commercial DNA isolation kit (QIAamp® DNA Blood Mini Kit, Qiagen, Valencia, California). PCR was performed as described previously. 9 As a positive control for DNA extraction and PCR, PCV2 isolate ISU-98-15237 was used.
Quantitative PCR
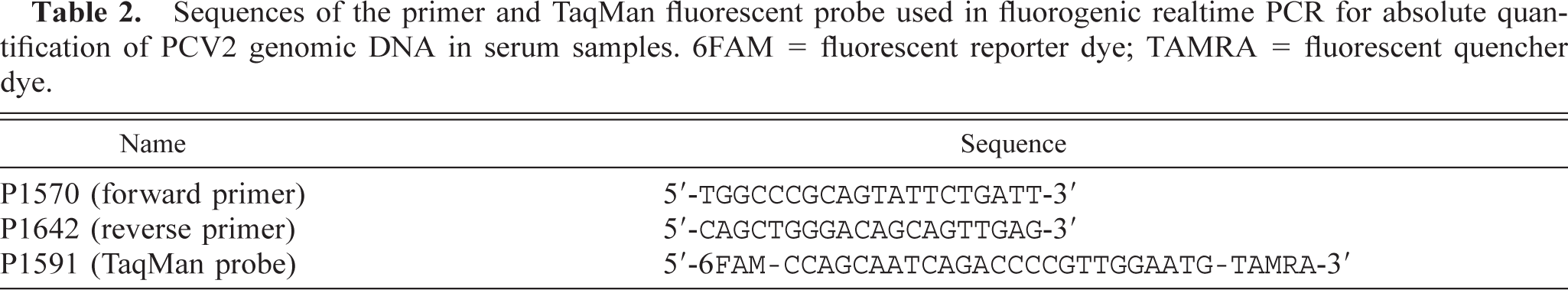
Sequence-specific oligonucleotide primers and a fluorescent probe for detection and relative quantification of PCV2 genomic DNA were designed using computer software (ABI Prism Primer Express, Version 1.5, PE Applied Biosystems, Foster City, California) according to the manufacturer's suggestions and were engineered to be within a highly conserved region of the viral genome (Table 2). The PCR reaction consisted of 50 µl PCR mixtures that contained 25 µl of the commercially available master mix (TaqMan Universal PCR Master Mix, PE Applied Biosystems), 5 µl DNA extract from each pig serum, forward and reverse primers, and detection probe with concentrations of 400, 400, and 200 nM, respectively. All reactions were carried out in triplicate. In addition, each reaction included three replicates for each of the five progressive 1 : 10 dilutions of a known copy number of PCV2 genomic DNA excised from a purified PCV2 DNA clone that served to generate a standard curve. Each plate was run in the sequence detection system (GeneAmp 5700 Sequence Detection System, Applied Biosystems) under company-specified conditions that were optimal (2 minutes at 50 C, 10 minutes at 95 C, followed by 45 cycles of 15 seconds at 95 C and 1 minute at 60 C) for the Taq Master Mix used.
Sequences of the primer and TaqMan fluorescent probe used in fluorogenic realtime PCR for absolute quantification of PCV2 genomic DNA in serum samples. 6FAM = fluorescent reporter dye; TAMRA = fluorescent quencher dye.
Bone marrow analysis
On both necropsy days, 22 and 34 DPI, bone marrow specimens were collected from the distal femur and were placed immediately into an ethylenediaminetetraacetic acid tube containing 0.5 ml sterile normal saline and 0.1 ml albumin and mixed to disperse cells. Two to three slides per animal were then prepared by cytospin preparation, and three 500 cell-count differentials were performed on each animal.
Statistical analyses
Chi-square test was used to analyze the clinical scores. Daily rectal temperatures were analyzed with multiple analysis of covariance (MANCOVA), which accounts for the repeated measures on each pig and for baseline temperature. For the pigs that were necropsied at 22 DPI, all the data were analyzed from 0 to 21 DPI. For the remaining pigs, the data were analyzed from 0 to 31 DPI. If the time-by-group interaction was not significant, then the group effect was assessed. Otherwise, the data were analyzed cross-sectionally to determine at which time points the group means are different. MANCOVA also was chosen for analyzing the weight data, which accounted for the repeated measurements of each pig and for baseline weight at 20 days before inoculation. Analysis of ELISA results was done by using cross-sectional analysis (analysis of variance [ANOVA]), and differences between groups were evaluated with Tukey's test. Nonrepeated measures of necropsy and histopathology data were assessed using analysis of covariance (ANCOVA) or nonparametric ANOVA. If a nonparametric ANCOVA test was significant (P < 0.05), then Wilcoxon tests (with the Bonferroni correction) were used to assess the differences of pairs of groups. Fisher's exact test was used to compare the incidence between groups.
Statistical analysis for real-time PCR was performed using the means of three replicate wells for each sample as compared with a standard curve generated with a purified known genome copy number of PCV2 genomic DNA ranging from 10 3 to 10 7 genomic copies per PCR plate well. Serial dilutions (1 : 10) between each standard resulted in a 5-point standard curve with a correlation of >0.992. The threshold cycle (threshold cycle CT, is the cycle number at which a statistically significant increase in amplification above the threshold line [0.1 in all cases] is first detected) of triplicate unknowns were compared with CTs of triplicate standards on a standard curve to determine the absolute number of viral genomes per milliliter of pig serum.
Results
Clinical signs
Pigs in the sham-inoculated control groups 1 and 2 showed no clinical signs of disease throughout the study. Pigs in PCV2-infected groups 3 and 4 exhibited intermittent diarrhea and mild respiratory disease from 13 DPI until the end of the study at 34 DPI. The respiratory disease was characterized by sneezing, clear nasal discharge, and occasional coughing. Statistical analysis of the clinical scores (chi-square test) revealed that the clinical respiratory disease scores in pigs of groups 3 and 4 were statistically more severe than those of groups 1 and 2 pigs (P < 0.0001). There was no difference in clinical scores between pigs of groups 3 and 4 (P = 0.6). Statistical analysis of the daily rectal temperatures (0 to 33 DPI) showed neither a significant group-by-time interaction (P = 0.14) nor a significant group effect (P = 0.08). Statistical analysis of the weight data showed no statistically significant difference in time-by-group effect (P = 0.8) or group effect (P = 0.6). At 34 DPI, the average weights appeared to differ (49.1 kg [± 5.4] in group 1 pigs, 47.9 kg [± 4.2] in group 2 pigs, 48.6 kg [± 7.5] in group 3 pigs, and 45.6 kg [± 4.7] in group 4 pigs], but there was substantial variability within each group. The average daily weight gain was 0.70 kg (± 0.05) for group 1 pigs, 0.67 kg (± 0.05) for group 2 pigs, 0.70 kg (± 0.08) for group 3 pigs, and 0.63 kg (± 0.09) for group 4 pigs.
Gross lesions
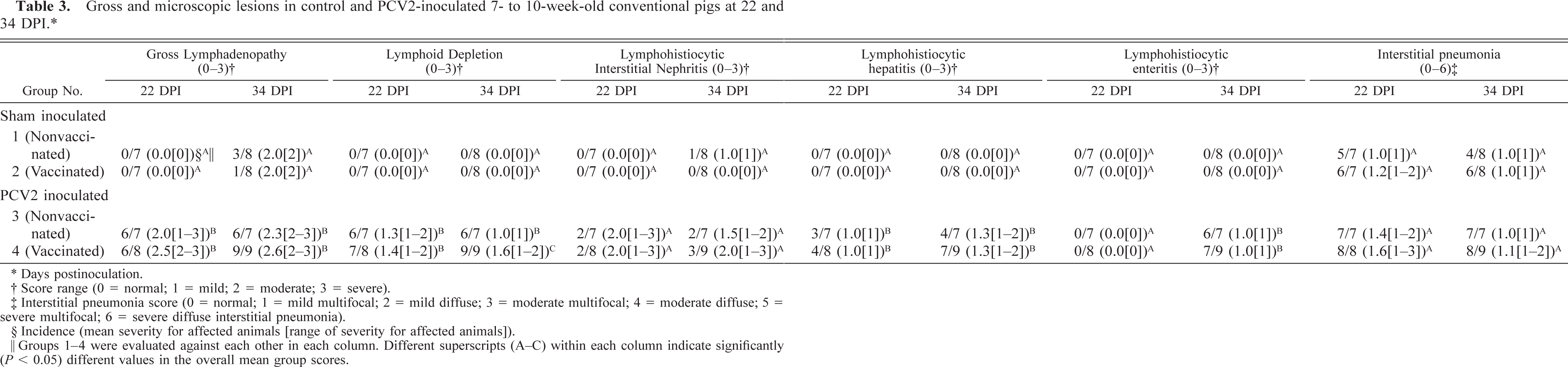
In sham-inoculated control groups 1 and 2, only 4 of the 30 pigs had visible gross lesions. The four pigs with lesions were necropsied at 34 DPI and had lymph node enlargement of approximately two times the normal size. There were significantly more pigs in groups 3 and 4 (P < 0.05) with enlarged lymph nodes (Table 3) compared with groups 1 and 2. In group 3, 12 of 14 pigs had enlargement of all lymph nodes from two to three times the normal size. In group 4, 15 of 17 pigs had lymph node enlargement of two to three times the normal size. The macroscopic evaluation of the lymph node sizes showed no statistical difference between groups 3 and 4 at the first (P = 0.33) or second (P = 0.3) necropsy.
Gross and microscopic lesions in control and PCV2-inoculated 7- to 10-week-old conventional pigs at 22 and 34 DPI. ∗
∗Days postinoculation.
† Score range (0 = normal; 1 = mild; 2 = moderate; 3 = severe).
‡Interstitial pneumonia score (0 = normal; 1 = mild multifocal; 2 = mild diffuse; 3 = moderate multifocal; 4 = moderate diffuse; 5 = severe multifocal; 6 = severe diffuse interstitial pneumonia).
§Incidence (mean severity for affected animals [range of severity for affected animals]).
‖Groups 1–4 were evaluated against each other in each column. Different superscripts (A-C) within each column indicate significantly (P < 0.05) different values in the overall mean group scores.
Microscopic lesions and IHC
There were no microscopic lesions in the lymph nodes of pigs in groups 1 and 2. In groups 3 and 4, microscopic lesions characterized by lymphoid depletion of follicles and granulomatous lymphadenitis were observed in nearly all pigs: 12 of 14 in group 3 pigs, and 16 of 17 in group 4 pigs. At 34 DPI, there was a significant difference (P = 0.02) in severity of lymphoid depletion between groups 3 (mean = 0.86) and 4 (mean = 1.56). Mild to moderate lymphoplasmacytic interstitial nephritis was observed in 1 of 15 group 1 pigs, in 4 of 14 group 3 pigs, and in 5 of 17 group 4 pigs. Mild lymphohistiocytic hepatitis was observed in 7 of 14 pigs in group 3 and in 11 of 17 pigs in group 4. Mild to moderate lymphohistiocytic enteritis was observed in 6 of 14 pigs in group 3 and in 7 of 17 pigs in group 4 (Table 3).
PCV2 antigen was detected by IHC in a variety of tissues (lymph nodes, spleen, and tonsil) in groups 3 and 4. At 22 DPI, PCV2 antigen was detected in all pigs examined. There was no difference between the amount and distribution of PCV2 antigen between groups 3 and 4 at 22 DPI. At 34 DPI, PCV2 antigen was only detected in the lymph nodes in 4 of 7 group 3 pigs. In contrast, at DPI 34, PCV2 antigen was still detectable in the lymph nodes of all group 4 pigs (9 of 9), and the amount of PCV2 antigen in the lymph nodes was significantly (P < 0.05) higher in group 4 than in group 3 pigs at 34 DPI (Table 4).
Comparison of estimated amount of PCV2 antigen (ranging from 0 = not detectable to 3 = strong signal) in lymphoid tissues between groups 3 (PCV2 infected) and 4 pigs (PCV2 infected and vaccinated).

∗Days postinoculation.
† Incidence (mean estimated amount for affected animals [estimated range]).
§Different superscripts within each column indicate significantly (P < 0.05) different values in incidence (A and B), or significantly (P < 0.05) different values in estimated overall mean group amount of PCV2 antigen (C and D).
Antibody response
Pigs in groups 1 and 2 remained seronegative for PCV2 antibody throughout the study. Pigs in groups 3 and 4 seroconverted to PCV2 between 21 and 28 DPI. Cross-sectional analysis (using ANOVA) indicated that there were statistically different group means (P < 0.0001) at 21, 28, and 33 DPI. A Tukey's test showed that for each day neither were groups 1 and 2 statistically different from each other nor were groups 3 and 4 different from each other. All serum samples tested were negative for presence of PRRSV- and PPV-specific antibodies at 33 DPI.
PCV2 viremia
In groups 1 and 2, all serum samples collected at different DPIs were negative for PCV2 DNA by PCR. Viremia in groups 3 and 4 pigs was detected as early as 7 DPI in 24 of 31 pigs. All pigs in groups 3 and 4 were viremic at 13 DPI. By 21 DPI, 11 of 14 pigs in group 3 and 16 of 17 pigs in group 4 remained positive with PCV2 DNA. By 28 DPI, 1 of 7 pigs in group 3 was positive for PCV2 DNA in sera, whereas 7 of 9 pigs in group 4 were positive. At 33 DPI, the day before necropsy, 7 of 9 pigs in group 4 were still positive for PCV2 DNA, whereas none of the pigs (0 of 7) in group 3 were positive. The PCR data were analyzed using nonparametric ANOVA as an omnibus test and Wilcoxon pairwise post hoc tests. The overall test was significant (P < 0.001), and post hoc tests indicated that groups 3 and 4 were different (P = 0.03) from each other and from groups 1 and 2 (P < 0.0001). To determine the length of viremia, only the animals that were alive at the end of the study were analyzed. Pigs necropsied at 22 DPI were not included. The mean viremia length observed in group 3 pigs (n = 7) was 2.14 (±0.26) weeks, compared with 4.44 (±0.23) weeks in group 4 pigs (n = 9). The length of viremia in group 4 pigs was determined by the end of the study at 34 DPI. At this time point, 7 of 9 group 4 pigs were still viremic, whereas none of the nonvaccinated group 3 pigs were viremic. The difference in viremia length between groups 3 and 4 was highly significant (P = 0.0003).
PCV2 genome titer in sera
Analysis of the amount of PCV2 genomic DNA per milliliter of serum revealed major differences between groups 3 and 4 pigs. The mean copy number of PCV2 genomic DNA measured in nonvaccinated group 3 pigs was 1.3 × 105 genomic copies per milliliter serum at 7 DPI. On the following testing dates (13–33 DPI), the PCV2 genomic titer decreased in all pigs to 6.1 × 103 PCV2-copies per milliliter serum at 33 DPI. In contrast, the mean copy number of PCV2 genome in sera measured in vaccinated group 4 pigs was 2.7 × 107 copies per milliliter serum at 7 DPI. Furthermore, in the majority of group 4 pigs (7 of 9), the PCV2 genomic titer remained at a relatively high level (mean copy number per milliliter serum between 2.3 × 106 at 14 DPI and 1.7 × 106 at 33 DPI) compared with nonvaccinated group 3 pigs during the entire experiment. Instead of decreasing, PCV2 genomic titer continued to increase over time in some pigs. Figure 1 illustrates the comparison of group mean PCV2 genome titer. Differences in the amount of PCV2 genome copy number between both groups were significant (P < 0.05) on 13, 21, 28, and 32 DPI.
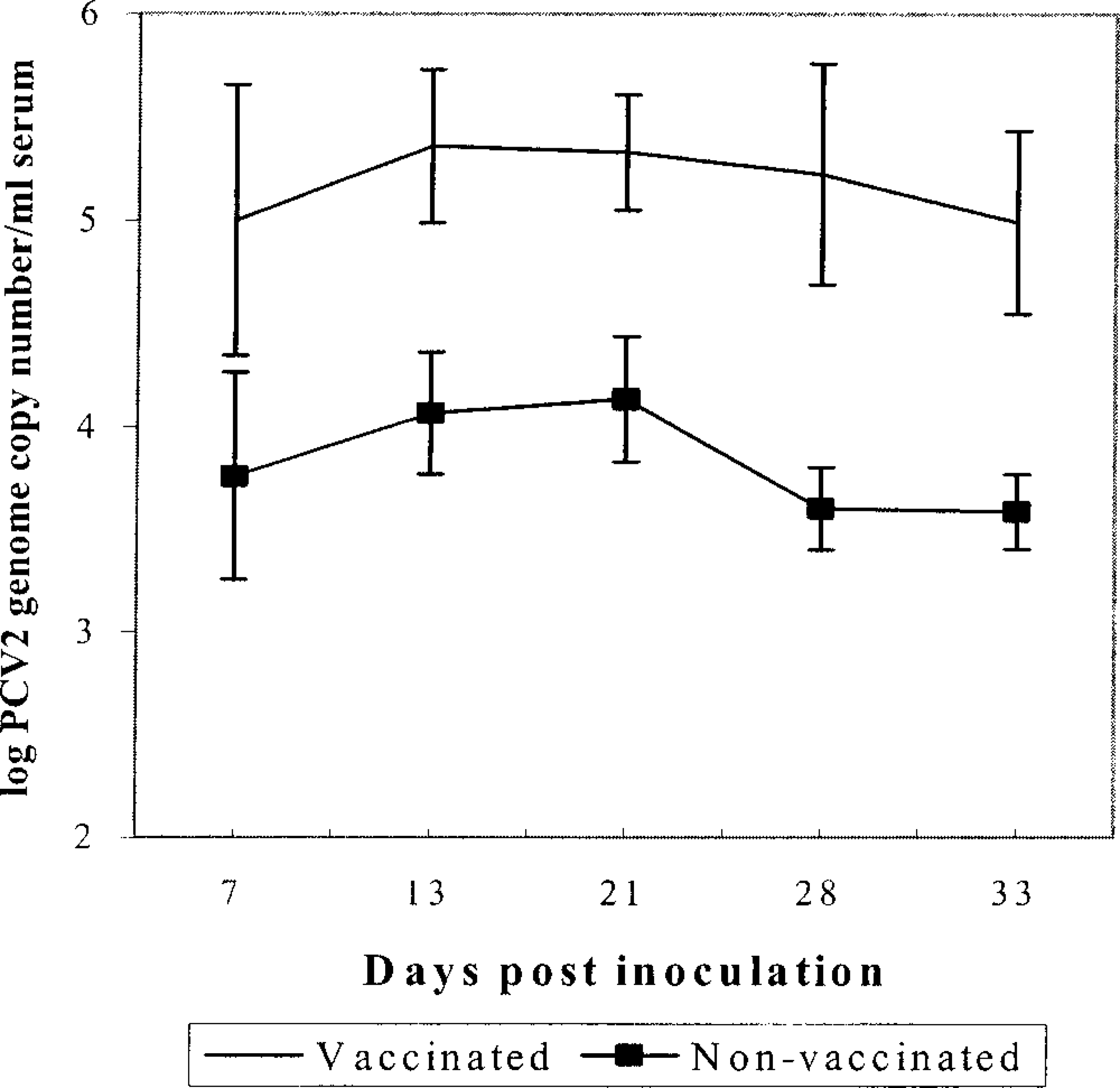
PCV2 genomic titers in sera measured by quantitative real-time PCR. The values are the mean group results for group 3 (nonvaccinated, n = 7) and group 4 (vaccinated, n = 9) pigs. Pigs that were necropsied at 22 DPI are not included. Error bars represent standard errors.
Bacteriology
There was no growth on cultures of the BAL-fluid in any of the pigs from groups 1 to 4.
Bone marrow analysis
All the variables were analyzed using nonparametric ANOVA. There were no significant differences between the groups. Erythroid and myeloid cell series were present and followed a normal maturation sequence. M : E ratios were within reference intervals for this species. Megakaryocytes were observed in all specimens. One interesting feature was the presence of many eosinophils on marrow cytospin preparations; eosinophils are rarely observed on peripheral complete blood count differentials.
Discussion
The objective of this study was to determine whether M. hyopneumoniae and APP vaccination enhanced the incidence and severity of PMWS disease and lesions associated with PCV2 infection of conventional pigs. At least three European studies, one using an experimental model 6 and two recent field studies, 7,19 provide evidence that vaccine-induced immune stimulation enhances PCV2 replication and increases the incidence of clinical PMWS. The results of these European studies are not ideally suited for application to the US field situation, where M. hyopneumoniae vaccines are typically used on older pigs. In the USA, PCV2-associated PMWS is most commonly observed in pigs aged 8–20 weeks, 13,15 whereas in Europe it is observed in pigs aged 3–10 weeks. 1 M. hyopneumoniae vaccination in the USA is typically done in nursery-age pigs from 3 to 10 weeks, whereas in Europe it is more commonly done on suckling pigs or on pigs in the early nursery phase at 1–4 weeks of age. We attempted to mimic the immune stimulation approach of the European studies by vaccinating pigs with M. hyopneumoniae and APP; however, we vaccinated at 5 and 7 weeks of age using segregated early-weaned (12–14 days) pigs, consistent with current US production systems.
Diagnostic criteria of PMWS include clinical evidence of wasting, histologic evidence of lymphoid depletion or granulomatous lymphadenitis, and the presence of PCV2 antigen or nucleic acids associated with the characteristic lymphoid lesions. 31 PCV2-associated microscopic lesions consistent with PMWS were observed in both PCV2-inoculated groups in this study; however, we failed to reproduce clinical manifestation of PMWS. This might be due to removal of other key stressors not present in the clean experimental environment. Unlike most field situations, our pigs had no known concurrent viral or bacterial coinfections, which have been shown to increase the incidence and severity of PMWS in PCV2-infected pigs. 2,4,14,16,18,30 The PCV2 inoculum was a pure and homogeneous infectious stock derived from an infectious DNA clone of PCV2 isolate ISU-40895, which was originally isolated from a pig with PMWS. 9,10 Therefore, the disease and lesions reproduced in this study perhaps are more definitive than those using tissue homogenate or cell culture–propagated PCV2 inoculum, which might contain other common swine agents. The differences in environment and coinfection may at least partly explain the differences in results of different experiment models and differences in severity of PCV2-associated disease from farm to farm.
Pigs in the vaccinated and nonvaccinated PCV2-infected groups developed microscopic lesions consistent with PMWS. This observation in the nonvaccinated group 3 is in contrast to many PCV2 experimental models, where no or mild microscopic lesions typical of PCV2-associated disease were observed. 4,17,18,29,30 There are only a few reports to date which describe reproduction of severe microscopic lesions characteristic of PMWS with singular PCV2 inoculation. 8,20,21 Differences in experimental results could be explained by PCV2 strain variation, individual pig susceptibility differences, differences in age of inoculation, or type of animal housing. Typically, more severe lesions were observed in young gnotobiotic pigs, 17,18 cesarean-derived-colostrum–deprived pigs, 8,14 and colostrum-deprived pigs, 2,4,16 compared with “conventional pigs.” 30 The definition of conventional pigs also may vary. In our case, these are genetics typical of the current US swine industry; pigs free of several major pathogens (PRRSV, SIV, PCV2, APP, M. hyopneumoniae) and early weaned to a distant site where they are reared in all-in-all-out fashion.
The length of PCV2 viremia in the unvaccinated group 3 pigs (2.14 ± 0.26 weeks) was consistent with observations of other research groups. 10,29 PCV2-associated lymphoid lesions peaked around 22 DPI and were minimal or absent in the nonvaccinated pigs at the end of the study at 34 DPI. In contrast, pigs in the vaccinated group 4 had significantly longer length of PCV2 viremia (4.44 ± 0.23 weeks) and a longer duration of PCV2 antigen detection in various lymphoid tissues. Quantitative PCR results further confirmed the significantly (P < 0.05) higher amount of PCV2 genome titers in sera of vaccinated pigs compared with that of nonvaccinated pigs. However, this did not influence the clinical appearance of the pigs in this study. Only mild respiratory disease and mild diarrhea were observed. Nevertheless, the increased length of viremia may increase the window of opportunity for coinfecting agents or other cofactors to trigger progression of PCV2 infection to PMWS.
Most modern pig production systems use some type of vaccine, particularly in 2- to 12-week–old pigs. It has been speculated that both the use of vaccines and the type of vaccine may play an important role in the manifestation of clinical PMWS. 6,19 In addition to the type of vaccine used, the timing of vaccination also may be important. This difference might be reflected in this current study, where none of the pigs vaccinated between 5 and 10 weeks of age developed clinical PMWS, whereas in a previous study 6 with an earlier onset of vaccination (2 weeks of age), PMWS was developed in 21% of the pigs. Early vaccination, high virus prevalence in the environment, and low maternal antibody status may lead to increased incidence and severity of PMWS typical of what is occurring in the UK, where the postweaning mortalities have increased by 5–40% in many affected herds. 27
PMWS is a disease complex influenced by many factors. One of these factors may be commonly used vaccines. However, on a herd basis, the risk of elimination of effective vaccines may be greater than the risk of inducing PMWS in a low percentage of the population. Further work is warranted to determine whether there is an optimal time for vaccination and if certain types of adjuvants or antigens play a role in vaccine-induced enhancement of PMWS. Swine producers may need to consider changing vaccine products or regimens to minimize PCV2 replication and PCV2-associated diseases in affected herds.
Footnotes
Acknowledgements
We thank the Iowa State University Livestock Infectious Disease Isolation Facility staff for animal care. This study was funded in part by Pork Check of Dollars from the National Pork Board and a grant from the Iowa Livestock Health Advisory Council.
