Abstract
Canine distemper virus (CDV) caused epizootics in lions (Panthera leo) in Tanzania's Serengeti National Park in 1994 and in captive lions and other Panthera spp. in the USA in 1991–1992. In this study, immunohistochemistry was used to compare viral distribution in tissues collected from ferrets (Mustela putorius furo) inoculated with one of the two lion-derived CDV isolates, either from Serengeti (A94-11/13) or from California (A92-27/20). The California isolate resulted in severe morbidity in all nine ferrets, whereas the Serengeti isolate resulted in severe morbidity in five of the nine ferrets. A slightly higher proportion of infected cells was found in many tissues in the Serengeti isolate-inoculated ferrets. These findings indicate that the pathogenicity of the California isolate is not directly related to the number of infected cells.
Canine distemper virus (CDV) is a member of the genus Morbillivirus, family Paramyxoviridae, which includes negative-sense, single-stranded RNA viruses. CDV is a highly contagious pathogen with a worldwide distribution and has been listed as the single greatest threat to populations of susceptible carnivores. 7 This virus has a wide host range, which includes members from all families of terrestrial carnivores—including felids—and several species of phocids. 2 , 4 , 10 Infection with CDV in susceptible hosts results in epithelial, immunosuppressive, and neurological syndromes. The severity of these syndromes is dependent on the viral strain, as well as on host factors such as immuncompetency. 1 , 6
Epizootics of distemper occurred in captive lions (Panthera leo) and other Old World felids in 1991–1992 and in lions of the Serengeti National Park in 1994. 3 , 8 CDV was isolated from infected lions in both instances, and these isolates were inoculated into ferrets (Mustela putorius furo); clinical and pathologic findings from that study have been reported. 5 In this study, immunohistochemistry was used to compare quantity and distribution of viral antigen with clinical disease caused by these two CDV isolates, using tissues collected from the previous experiment. 5
The initial experimental infection protocol, as previously described, 5 entailed inoculating nine unvaccinated and nine vaccinated (Distemink®, United Vaccines, Madison, WI) ferrets with the California isolate (A92-27/20) and nine unvaccinated and nine vaccinated ferrets with the Serengeti isolate (A94-11/13). As previously reported, all the ferrets infected with the California isolate became moribund because of severe epithelial disease, whereas four ferrets inoculated with the Serengeti isolate displayed only mild clinical signs before euthanasia 4 weeks postinfection (wpi), the end of the experimental period; only one of these animals was necropsied. 5 Formalin-fixed, paraffin-embedded tissues from this experiment included trachea, lung, kidney, urinary bladder, liver, gall bladder, spleen, thymus, lymph node, external nares, external pinna, stomach, small intestine, pancreas, salivary gland, heart, adrenal gland, and brain (cerebrum at rostral thalamus, cerebrum at hippocampus, and cerebellum and brain stem at midcerebellum).
Immunohistochemistry was performed as follows. Three-micrometer sections of formalin-fixed, paraffin-embedded blocks were cut and placed on positively charged slides (Probe-On Plus, Fisher Scientific, Springfield, NJ). Antigen retrieval was performed by exposing slides immersed in citrate buffer (Unmasking solution, Vector Laboratories Inc., Burlingame, CA) to microwaves. Nonspecific binding sites were blocked by the application of 2% goat serum. The slides were then incubated with a commercially available mouse monoclonal anti-CDV nucleoprotein antibody (VMRD Inc., Pullman, WA) at a dilution of 1:3,000. A secondary biotinylated goat anti-mouse IgG antibody (DAKO Corp., Carpinteria, CA) was applied to samples at a dilution of 1:250, followed by signal amplification with avidin–biotin–conjugated peroxidase (Elite Peroxidase, Vector Laboratories). Finally, the antigen-antibody complex was observed by its reaction with 3,3′-diaminobenzidine (DAB). Slides were counterstained lightly with hematoxylin and coverslipped for permanent record.
The staining pattern was evaluated visually and ranked as follows: 0, negative; 1, rare, individual positive cells; 2, rare foci with few cells per foci; or 3, multiple foci with numerous cells per foci. To compensate for the variable times of tissue collection, tissues were grouped into three time intervals: 2 wpi (12 and 15 days pi [dpi]), 3 wpi (20, 21, 22, and 23 dpi), and 4 wpi (29 dpi). The scores were then averaged for all animals of an infection group per week postinfection and rounded to the nearest whole number.
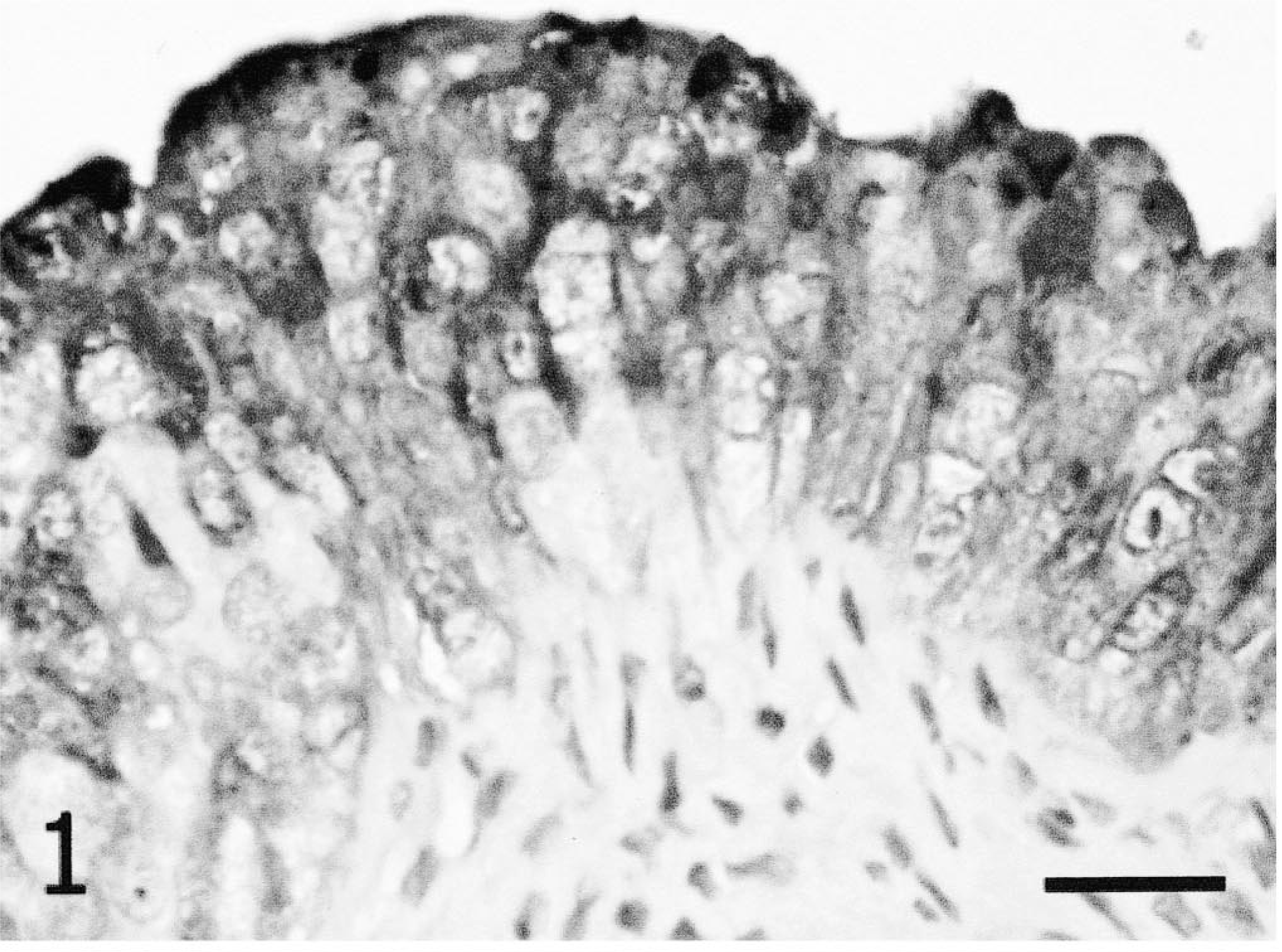
Viral antigen was not detected in the vaccinated ferret tissues or in cardiomyocytes, adrenal tissue, hepatocytes, or renal tubular epithelium of unvaccinated ferrets. Positive results are summarized in Table 1. At 2 wpi, there was extensive positivity in a number of tissues with both isolates. The ferrets infected with Serengeti isolate had same or slightly greater amounts of CDV antigen in all tissues that contained antigen. This was most evident in the urothelium (urinary bladder and renal pelves) (Figs. 1, 2), gastric parietal cells, salivary gland epithelium, biliary epithelium, and lymphoid tissues (spleen, lymph node, and small intestinal gut-associated lymphoid tissue). At 3 wpi, the staining pattern was similar in the Serengeti and California isolates in all tissues. By 4 wpi, only four Serengeti isolate–infected animals remained. All four were euthanatized before they showed any clinical signs, and one of these animals was necropsied. The brain and salivary gland of this animal stained intensely for CDV antigen, whereas other epithelial organs did not show any staining pattern.
Immunohistochemistry results from selected tissues of ferrets infected with canine distemper viruses isolated from lions. Tissues collected 12 and 15 days postinfection (dpi) were grouped as 2 weeks postinfection (wpi), 20–23 dpi were grouped as 3 wpi, and 29 dpi were considered as 4 wpi. The staining pattern was evaluated visually and ranked as follows: 0, negative; 1, rare, individual positive cells; 2, rare foci, multiple cells per foci; or 3, multiple foci, numerous cells per foci. The scores for all tissues within that category for all ferrets within that collection period were then averaged and rounded to the nearest whole number. All ferrets inoculated with the California isolate were euthanized either 2 wpi or 3 wpi.
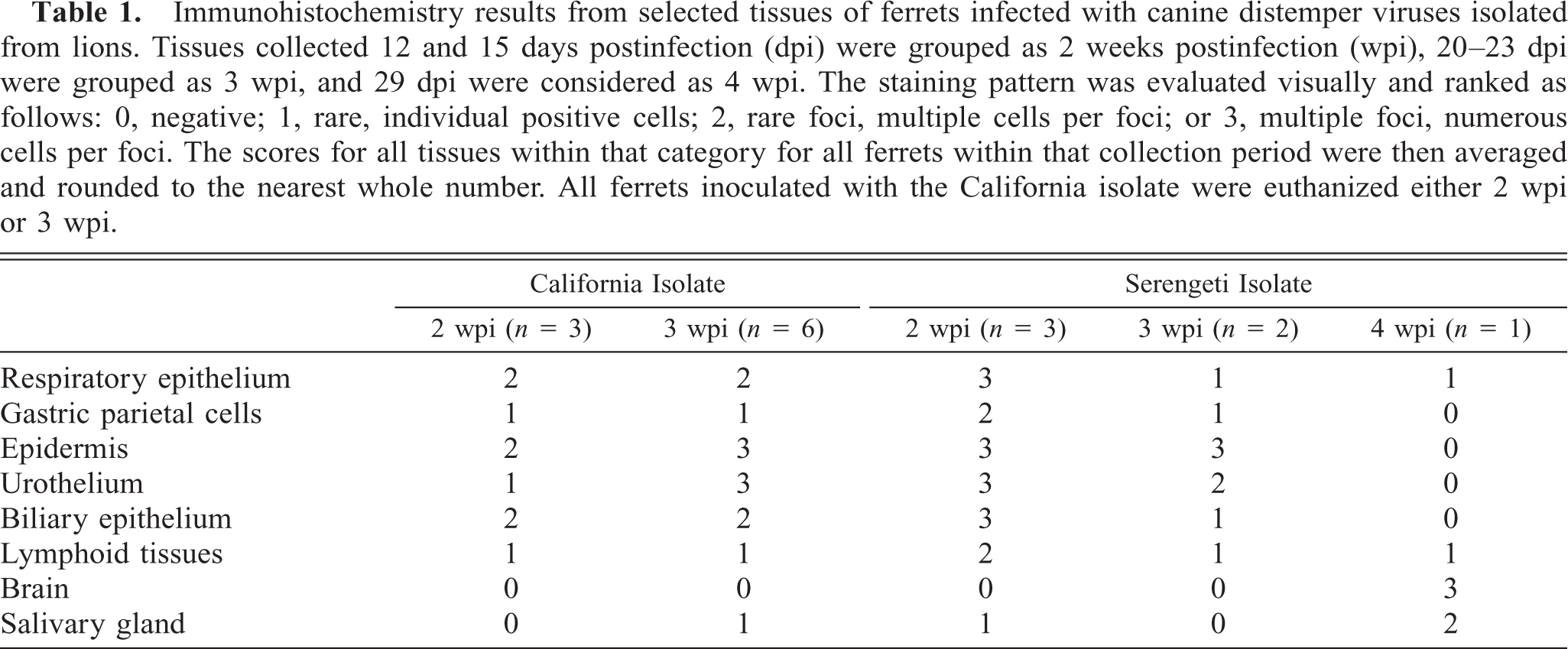
Urinary bladder; Serengeti isolate infected, 2 wpi. Canine distemper virus antigen is detected in nearly 100% of the urinary bladder epithelial cells of ferrets infected with the Serengeti isolate. Immunohistochemistry; DAB, hematoxylin counterstain. Bar = 25 µm.
Urinary bladder; California isolate infected, 2 wpi. Canine distemper virus antigen is detected in only scattered foci of urinary bladder epithelial cells of ferrets infected with the Serengeti isolate. Immunohistochemistry; DAB, hematoxylin counterstain. Bar = 25 µm.
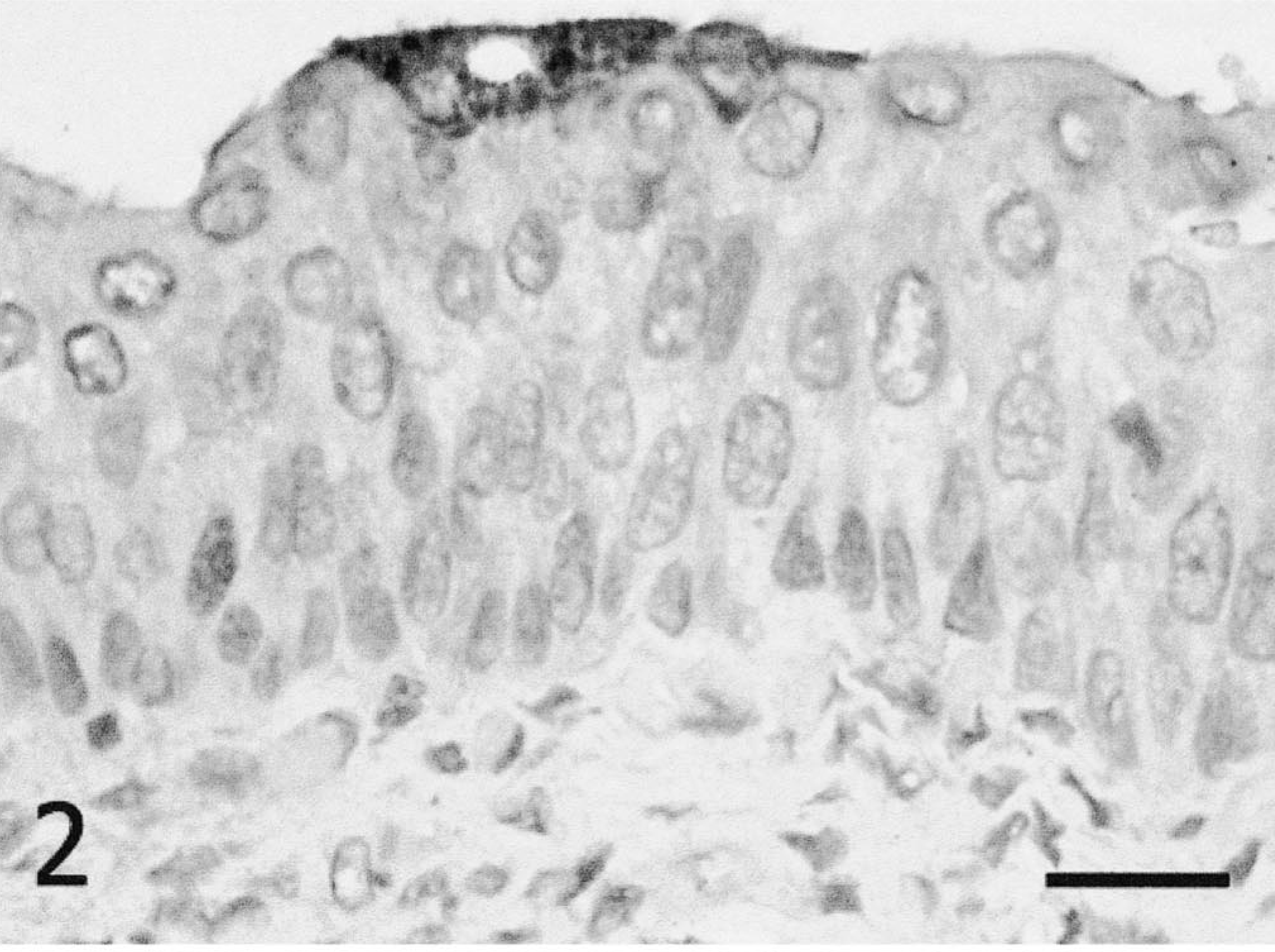
With both isolates, the staining pattern within organs was consistent with what has been described for CDV infection. 9 Epithelial cells of the integumentary, urinary, and respiratory systems had extensive amounts of CDV antigen. Lymphoid tissues were also affected, with staining evident in splenic periarteriolar sheath- and GALT-associated lymphoid cells and in scattered macrophages throughout the splenic red pulp and the subcapsular sinus of the lymph nodes. Within the central nervous system, neurons and glial cells stained positive for CDV-associated antigen (predominately intracytoplasmic). Staining was located in multifocal areas of the brain stem (primarily white matter) subjacent to the meninges, with lesser numbers of foci deeper in the neuropil (Fig. 3). Staining was also present in the white and gray matters of the mesencephalon and cerebral cortex.
Brain stem; Serengeti isolate infected, 4 wpi. Canine distemper virus antigen is detected in multiple foci of glial cells in an area with prominent perivascular cuffing. Immunohistochemistry; DAB, hematoxylin counterstain. Bar = 250 µm
The current study showed that ferrets inoculated with the California isolate had slightly fewer infected cells in the majority of the tissues 2 wpi, as compared with the ferrets inoculated with the Serengeti isolate. This difference occurred when the California isolate–infected animals were uniformly and severely moribund, while only five of the nine Serengeti isolate–infected animals were moribund. These results indicate that the degree of disease severity is not necessarily proportional to the numbers of infected cells.
Immunohistochemical staining patterns for the Serengeti isolate suggest that it may have a greater chance of successful transmission than the California isolate. At 2 wpi, there was abundant viral protein being produced in many tissues, including the urinary epithelium, which constitutes a likely source of environmental contamination with this virus. Also, viral protein was detected in the salivary and central nervous tissues of the only ferret that was necropsied after apparently recovering from the infection. Therefore, although these animals were not showing clinical signs, they were producing virus that could then possibly be shed into the environment.
The initial study indicated that the mortality rate in ferrets for the California isolate was higher than that for the Serengeti isolate. However, immunohistochemistry demonstrated viral antigen in the brain of a ferret inoculated with the Serengeti isolate. The infection in the brain indicates that the clinical course of the infection may not have been complete, even though there was a lapse in the clinical signs. It is possible that if these animals were allowed to live longer, some would have developed neurological signs and succumbed to CDV-induced encephalitis. Therefore, if the study period had been longer, it is possible that the mortality rate for the Serengeti isolate would have been closer to that for the California isolate. Although immunohistochemistry cannot directly measure shedding and viral loads, it is possible that in a free-ranging population, the Serengeti isolate could cause more environmental contamination and thus more clinical cases of distemper because of the longer course of infection.
Footnotes
Acknowledgements
We are grateful to Drs. Leathers, Gorham, McKeirnan, and Appel for use of the tissue blocks.
