Abstract
Canine distemper virus (CDV) was previously considered to have a host range restricted to the canid family. In 1994, the virus was associated with sporadic outbreaks of distemper in captive felids. However, after severe mortality occurred in the Serengeti lions (Panthera leo), attention became focused on the pathogenesis of the virus and a concerted effort was made to identify the virus as CDV or a closely related feline morbillivirus. The present study was designed to explore the susceptibility of ferrets to challenge with two morbilliviruses isolated from lions and the protective effects of a modified-live mink distemper vaccine. Because mortality in ferrets infected with pathogenic CDV approaches 100%, the ferret was selected as a test animal. Two strains of lion morbillivirus were used as a challenge, A92-27/20 (California lion isolate) and A94-11/13 (Serengeti lion isolate). The two strains of lion morbillivirus were antigenically related to CDV (Rockborn strain), and ferrets were susceptible to both of the viruses when inoculated intraperitoneally. The inoculated ferrets were anorectic at 5–6 days postinoculation (PI), exhibited oculonasal discharge at 9–12 days PI, and became moribund at 12–22 days PI. Severe bilateral conjunctivitis was the typical clinical sign. Inclusion bodies characteristic of morbillivirus (eosinophilic, intranuclear, and intracytoplasmic) were distributed in many epithelial cells, including those of the skin, conjunctiva, gallbladder, liver, pancreas, stomach, trachea, lung, urinary bladder, and kidney. Virus was reisolated from selected lung tissues collected at necropsy and identified by CDV-specific immunofluorescence. Ferrets vaccinated with the mink distemper vaccine (Onderstepoort strain) were protected from challenge with the two lion strains, adding further support to the premise that the viruses are closely related to CDV.
The host range of canine distemper virus (CDV) has come under intense scrutiny over the past few years in part because of observations of antigenically related morbilliviruses associated with or causing disease in a variety of terrestrial and marine mammals. 4,9,11,12,20,30 Sources of confusion regarding the host range of CDV in felids have arisen from disparate reports of the lack of CDV disease in domestic cats and the occurrence of fatal CDV-like disease in captive and free-roaming large felids. 4,8–10,26–28,31,34,37,45 The debate regarding CDV interspecies transmission (canid to felid) versus the existence of a unique feline morbillivirus strain antigenically related to CDV has not been fully resolved. 35 It has been postulated, based upon antigenic profiles, that the morbillivirus associated with the epizootic in free-roaming lions in the Serengeti National Park in Tanzania was a variant of CDV. 16,26,27,37 More recent studies reporting on the genetic similarity between the lion morbilliviruses and other CDV isolates from carnivores in the same region support this hypothesis. 15 The present study was initiated with three aims related to the pathogenesis of the morbilliviruses isolated from a diseased captive lion and from a diseased free-roaming lion. The first aim was to determine if the lion morbilliviruses were pathogenic when inoculated into susceptible ferrets (Mustela putorius furo). The ferret is highly susceptible to CDV and was deemed to have the greatest potential for an in vivo surrogate biologic assay for determining the pathogenesis of the lion morbilliviruses. 14,18,23,38,39,43,44 The second aim was to determine how the pathogenesis of the two different lion morbilliviruses compared with that of a classical strain of CDV, Green's ferret-adapted CDV, 22,23 and the third aim was to determine whether ferrets vaccinated with a modified-live mink distemper vaccine would be protected from the lion morbilliviruses.
Materials and Methods
Animals and husbandry
Forty male ferrets, 9 months of age, were housed in individual cages and maintained indoors at 22 ± 2 C. All ferrets received water and food ad libitum. Thirty-six ferrets served as principals, and four ferrets served as controls. Group A consisted of nine ferrets (Nos. 1–9) that were inoculated intraperitoneally (IP) with 0.5 ml of lion morbillivirus A92-27/20 (California lion isolate). Group B consisted of nine ferrets (Nos. 10–18) that were inoculated IP with 0.5 ml of lion morbillivirus A94-1l/13 (Serengeti lion isolate). Groups C (Nos. 19–27) and D (Nos. 28–36) consisted of nine ferrets each that were vaccinated with modified-live distemper vaccine (Distemink®, United Vaccines, Madison, WI) 3 weeks prior to challenge with either 0.5 ml of California lion morbillivirus or 0.5 ml of Serengeti lion morbillivirus, respectively. Group E (Nos. 37–40) consisted of four uninoculated ferrets. At predetermined times, the ferrets in groups A, B, and E were euthanatized, and selected tissues were collected for histopathologic and virologic examination. Ferrets in groups C and D were observed for the duration of the study (29 days).
Washington State University operates under an approved Animal Welfare Assurance (A 3347-01) and is accredited by the Association for Assessment and Accreditation of Laboratory Animal Care, International. This project was approved by the Animal Care and Use Committee of Washington State University.
Viruses
Two lion morbilliviruses were used in the study. Virus A92-27/20 was isolated from the white blood cell fraction of a lion in the Wildlife Waystation, San Fernando, California. 10 This virus was at passage 2 and had a titer of 107.0 TCID50/ml. Virus A94-11/13 was isolated from the cerebrospinal fluid of a free-ranging lion in the Serengeti National Park in Tanzania, Africa. 37 The specimen was originally imported to the United States by Dr. S. J. O'Brien (National Institutes of Health, Frederick, MD). The isolate was made in phytohemagglutinin-stimulated canine lymphocytes. The virus was at passage 2 and had a titer of 105.5 TCID50/ml. The viruses were stored at −80 C until the day of inoculation, at which time the vials were rapidly thawed in a 37 C water bath and stored on ice until the ferrets were inoculated IP.
Histopathology
Selected tissues from the ferrets were fixed in 10% buffered formalin and routinely processed for histopathology. Paraffin-embedded blocks of skin, conjunctiva, gallbladder, liver, pancreas, stomach, trachea, lung, urinary bladder, brain, kidney, epididymis, and salivary glands were sectioned and stained with hematoxylin and eosin (HE). Sections were evaluated by light microscopy and photographed at 100× and 160×.
Virus isolation
Lung tissues from two ferrets (Nos. 1, 6) inoculated with the California lion isolate and two ferrets (Nos. 11, 17) inoculated with the Serengeti lion isolate were homogenized in cell culture medium and inoculated into primary cultures of canine peripheral blood lymphocytes (PBL) as previously described. 6 Cultures demonstrating cytopathic effect were passaged a second time and were frozen at −80 C until identified by CDV-specific immunofluorescence in PBL cell cultures. 6,10
Virus serology
Blood was collected via cardiac puncture from anesthetized (ketamine and acepromazine) ferrets at the onset of the study and again at the time of euthanasia (sodium pentobarbital). The serum was stored at −20 C until tested by serum neutralization (SN). 7 The SN test was performed on heat-inactivated serum using the Rockborn strain of CDV as the challenge virus and Vero cells as the indicator cell line, as previously described. 24
Clinical assessment
Ferrets were observed daily for lethargy and amount of food consumed. Each ferret was graded based upon clinical signs: 1+ = off feed; 2+ = off feed and conjunctivitis (eyes matted or closed); 3+ = moribund.
Results
Experimental pathogenesis/clinical signs
The California lion isolate produced a disease course that was consistent with both dog and mink CDV field isolates, i.e., a longer course than recorded with Green's ferret-adapted CDV, but all nonvaccinated ferrets developed clinical distemper. Disease onset occurred at the fifth and sixth day postinoculation (PI), when anorexia was first observed. By the 12th day, all nine ferrets (Nos. 1–9) had nasal and ocular mucopurulent exudate and a progressively deteriorating course. Ferrets were euthanatized on days 12, 15, 20, and 21. On days 15, 20, and 21, the ferrets were moribund.
The Serengeti lion isolate was not as virulent in ferrets as was the California lion isolate. The ferrets in this group had depressed appetites on day 6 PI. Mild clinical signs consisting of serous nasal and conjunctival exudation were recorded on days 10 and 11. Five ferrets (Nos. 11, 12, 15–17) were euthanatized, showing depression, anorexia, and mucopurulent nasal and ocular exudation on days 12, 15, 19, and 22. Four ferrets (Nos. 10, 13, 14, 18) that had shown clinical signs of distemper on days 5–10 and had apparently recovered were euthanatized on day 29.
Pathology
Ferrets demonstrating severe clinical signs were euthanatized, refrigerated, and necropsied within 3 hours. Gross findings consistent with CDV-induced lesions included dark red mottling throughout the lung lobes, with variable edema. Histologically, the ferrets had marked lymphoid depletion in lymph nodes and spleen and mild to severe diffuse interstitial pneumonia. Eosinophilic intranuclear and intracytoplasmic inclusion bodies were observed in epithelial cells in skin, conjunctiva, gallbladder, liver, pancreas, stomach, trachea, lung, urinary bladder, brain, kidney, epididymis (Fig. 1), and salivary glands. The four ferrets (Nos. 10, 13, 14, 18) that were inoculated with the Serengeti lion isolate and that apparently recovered were euthanatized and necropsied on day 29. These ferrets appeared normal, with no gross lesions. Histologically, these ferrets showed residual syncytical cells in the lung, but other organs appeared normal.
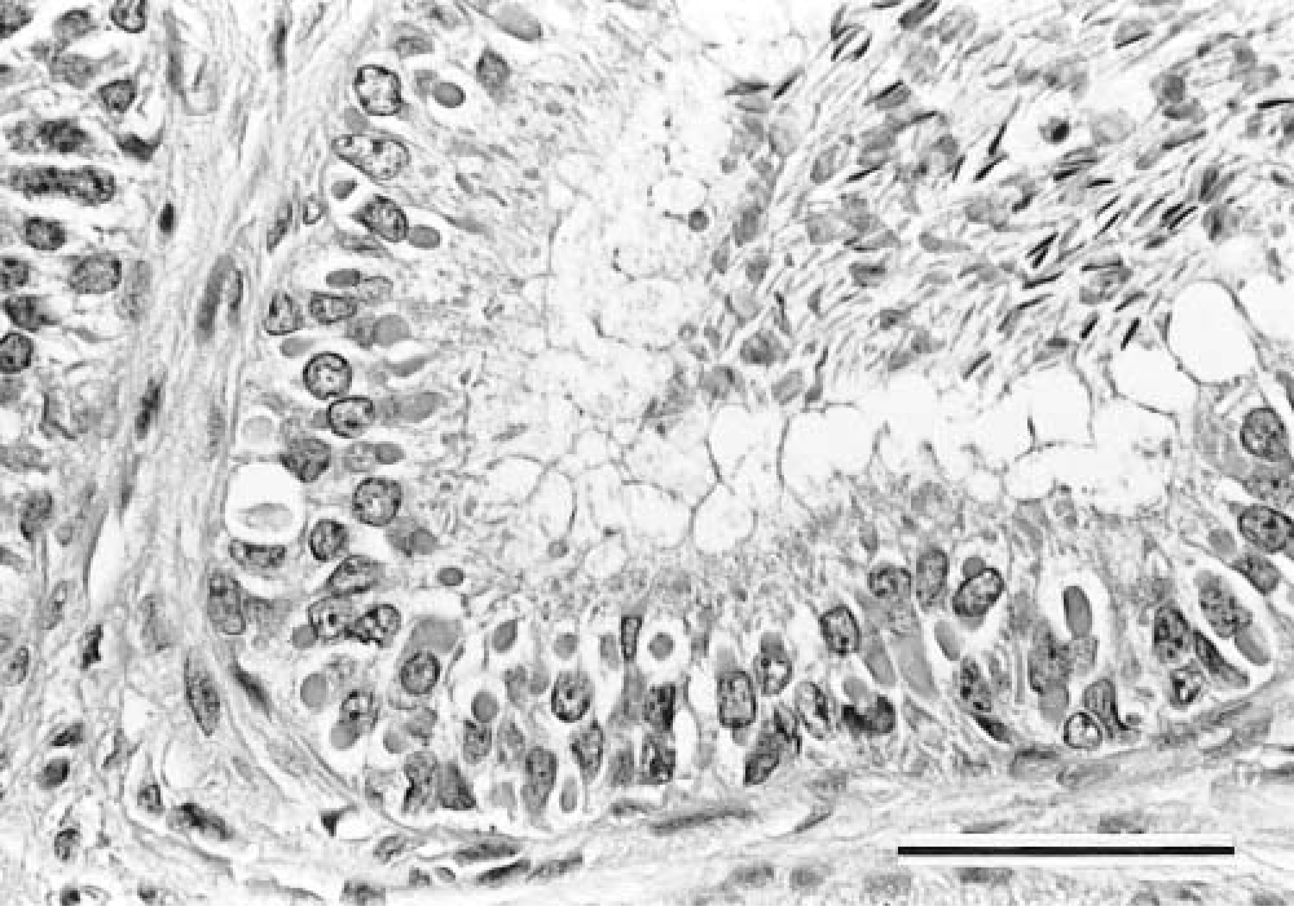
Epididymis; ferret inoculated with lion morbillivirus, California isolate A92-27/20. Note multiple intracytoplasmic inclusion bodies in the epididymal epithelium. HE. Bar = 50 µm.
Virus isolation
Virus was reisolated from two ferrets (Nos. 1, 6) inoculated with the California lion isolate and two ferrets (Nos. 11, 17) inoculated with the Serengeti lion isolate. The viruses were identified by CDV-specific immunofluorescence. 10
Serology
Sera collected before inoculation of the ferrets (Nos. 1–40) were tested by the CDV SN test. All animals were CDV seronegative (<1:5). Over the course of the experiment, serum samples were collected and banked and then tested at the conclusion of the study (day 29 PI). The four uninoculated ferrets (Nos. 37–40) remained CDV seronegative throughout the experiment. All nine ferrets (Nos. 1–9) from group A (California lion isolate) seroconverted to CDV. The SN titers ranged from 1:5 (n = 2) to 1:80 (n = 3). Four ferrets had CDV SN titers of 1:40. There were no survivors in this group. All nine ferrets (Nos. 10–18) from group B (Serengeti lion isolate) seroconverted to CDV. The SN titers ranged from 1:10 (n = 2) to 1:640 (n = 4). The remaining three ferrets had SN titers of 1:160. The four ferrets (Nos. 10, 13, 14, 18) with the elevated SN titers had mild CDV clinical signs and apparertly recovered. These animals were tested at day 29 PI prior to euthanasia.
All the ferrets (Nos. 19–36) in groups C and D (vaccinated) seroconverted to CDV (Rockborn strain) prior to challenge with the respective lion morbillivirus. The CDV SN titers ranged from 1:80 to 1:640, with a geometric mean of 1:160. There were no clinical signs noted in the vaccinated ferrets. These ferrets were subsequently used in another experiment; therefore, no gross or histologic data were available for comparison.
Discussion
The results of this study indicated that both lion morbilliviruses were pathogenic for ferrets. The California lion isolate, A92-27/20, appeared to be more virulent than the Serengeti lion isolate, A94-11/13, in susceptible ferrets. Comparison of the course of the disease of the two lion morbilliviruses with that caused by Green's ferret-adapted CDV indicated that both lion isolates were less virulent in ferrets (Fig. 2).
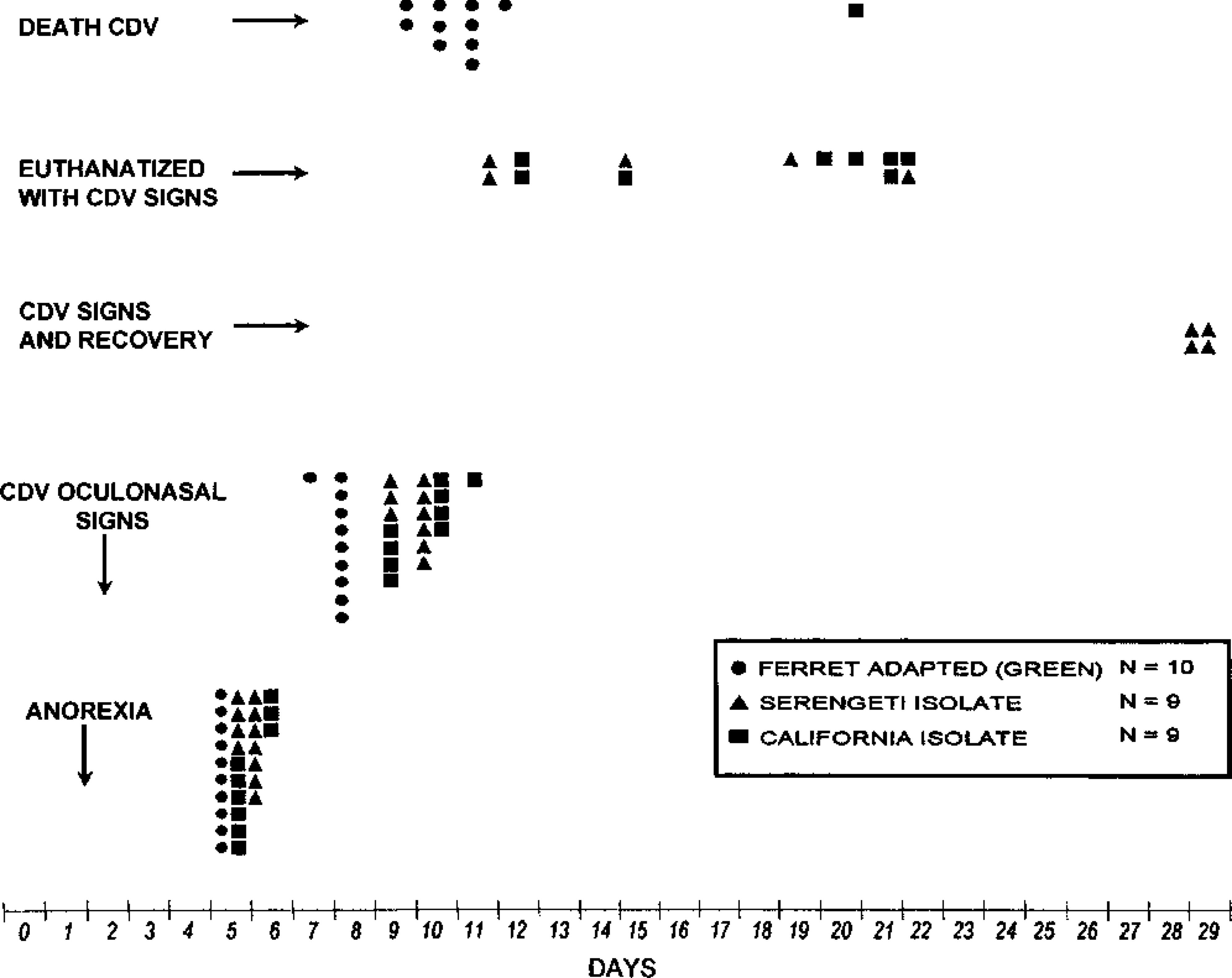
Comparison of clinical signs of infection between lion morbilliviruses and Green's ferret-adapted CDV strain.
The course of CDV disease in ferrets inoculated with the California isolate is consistent with the course observed when most dog, mink, or fox isolates are inoculated into ferrets. 5,14,18,22,23,38,41,43 The onset of signs (anorexia, lassitude) usually occurs at 5–6 days PI and the time to death varies from 13 to 30 days PI. Generally, on subsequent passages in ferrets, the time to death is reduced to 12–13 days PI. Ferrets showing moderate to severe clinical signs of distemper invariably succumb. 38,39,43
Although the fatality for ferret distemper in this study was expected to approach 100%, four of nine ferrets (Nos. 10, 13, 14, 18) that received the Serengeti lion isolate showed early, mild clinical signs but were normal at 29 days PI. There are at least three possibilities for this reduction in virulence for these ferrets: 1) passage through the lion population reduced the CDV virulence for ferrets; 2) the original source of CDV for the lions might have been attenuated by passage in domestic or wild dogs or possibly other species susceptible to CDV; or 3) the challenge inoculum was below the 100% lethal dose.
Although neurologic signs were not observed in the present trial, ferrets inoculated with different strains of CDV by the IP and/or subcutaneous routes have distinct differences in the degree of involvement of the nervous system (Gorham, unpublished data). Nervous signs were consistently observed in ferrets inoculated IP with the Synder-Hill strain, 40 infrequently observed with the Laidlaw-Dunkin strain, and not observed at all with Green's ferret-adapted CDV (Gorham, unpublished data).
The ferrets (Nos. 19–36) vaccinated with the modified-live mink distemper vaccine developed high antibody titers to CDV and were resistant to challenge with the two lion morbilliviruses. These results provide further support for the conclusion that the lion morbilliviruses are strains of CDV that have crossed host species.
This study was pivotal in determining the virulence of the lion morbilliviruses. In an earlier study, another feline morbillivirus isolate, A92-27/4 (isolated from a fatally diseased captive Chinese leopard [Patheria pardus japonensis] in California), was avirulent in specific-pathogen-free domestic cats. 27 Because a high percentage of lions in the Serengeti outbreak seroconverted to CDV without succumbing to disease, questions were posed concerning whether the virus was the direct cause of the population decline, whether there were multiple strains of lion morbillivirus as has been reported for CDV, and whether other factors were involved in the epizootic, such as nutrition and/or other coinfections. 13,19,29,31,32,36,37 Coinfections with known domestic cat pathogens, such as feline herpesvirus and feline immunodeficiency virus, although endemic in the lion populations, did not appear to contribute to the severity of the morbillivirus outbreak in Africa. 36 The present study provides direct evidence that lion morbilliviruses retain their virulence and that the ferret is susceptible to infection with these viruses and subsequent disease. The lesions observed were comparable to the lesions caused by CDV in domestic dogs and domestic ferrets. 5,12,14,18,21–23,33,39,43,44
The lion morbillivirus may have originated from domestic dogs living on the periphery of the lion habitats. 16,37,42 Hyenas (Crocuta crocuta), African wild dogs (Lycaon pictus), jackals (Canis mesomelas), and bat-eared foxes (Otocyon megalotis) 1–3,17,25 have all been reported to be infected with CDV and may serve as transition hosts between the domestic dog and lion populations. Phylogenetic analyses show a close relationship between morbilliviruses infecting hyenas and lions. 25 Additional genetic analyses of the morbilliviruses isolated from hyenas, bat-eared foxes, and domestic dogs in the Serengeti region revealed that these viruses were virtually identical to the morbilliviruses isolated from the lions in the same region. 15 The morbilliviruses isolated from these Serengeti carnivores were comparable to CDV, and the authors concluded that the dominant CDV strain has frequently crossed host species among the four Serengeti carnivores tested. 15
The lion morbillivirus should be classified as a biotype of CDV. These morbilliviruses should be identified as CDV followed by the species from which the virus was isolated, country of origin, year, and strain designation, e.g., CDV/Lion/Africa/1994 (strain A94-11/13).
Footnotes
Acknowledgements
We thank the faculty and staff of the Washington Animal Disease Diagnostic Laboratory, the USDA Research Unit, and the Virology Laboratory at the James A. Baker Institute at Cornell University for valuable technical assistance and support for this investigative study. Special thanks are extended to the Biomedical Communications Unit for assistance with graphics and photography and to Amy Wood for assistance with the manuscript.
