Abstract
Nine percent of xenogeneic hybridomas originating from a bovine leukemia virus (BLV)-infected cow secreted monoclonal IgM antibodies with multispecific reactivity. Similar reactivity was evident in some antibodies with an unusually long (>50 amino acids) third complementarity-determining region of the heavy chain. Electron microscopy of hybridomas demonstrated the presence of c-type virus particles consistent with polymerase chain reaction detection of BLV env gene. Some hybridomas contained dilated rough endoplasmic reticulum and cisternae filled with moderately electron-dense granular substance compatible with plasma cells at presecretory stage. The number of chromosomes in xenogeneic hybridomas corresponded to the sum total of mouse and bovine chromosomes. None of the hybridomas showed polyploidy. The immunochemical and genetic analysis of stable bovine immunoglobulin-secreting xenogeneic hybridomas confirms that BLV infection causes polyclonal B cell activation regardless of antigen specificity. Presence of c-type particles in hybridomas suggests that T cell-derived cytokines are not required for sustained BLV expression.
Bovine leukemia virus (BLV), an oncovirus, demonstrates B cell tropism and is often the cause of benign persistent lymphocytosis and lymphosarcoma in adult cattle. 6 The mechanism of BLV-induced leukemogenesis remains obscure. BLV expression in vivo, in both neoplastic and nonneoplastic cells, has been suggested to be blocked at the transcriptional level, 7 , 10 but short term in vitro cell culture permits BLV expression. It should be noted that the BLV enhancer has highest similarity with the murine Cµ immunoglobulin heavy-chain enhancer that functions in a B cell–specific manner. 14 Development of xenogeneic hybridomas provides long-lived monoclonal cell lines that permit cellular, immunologic, and molecular analysis of immune effector cells in species where an appropriate fusion partner is not available. 2 , 4 With the objective of understanding the morphologic, cytogenetic, immunologic, and molecular characteristics of BLV-infected leukemic B cells in cattle, we constructed mouse × cattle xenogeneic hybridomas from peripheral blood lymphocytes (PBL) of a cow afflicted with bovine leukosis. 11 The hybridomas showed morphologic characteristics associated with a secretory lymphocyte that had undergone malignant transformation. Similar to natural antibody secretion in human patients with chronic lymphocytic leukemia (CLL), 3 some of the hybridomas from BLV-infected cattle secreted bovine IgM with polyspecific reactivity to structurally dissimilar antigens such as protein, polysaccharide, nucleic acid, and synthetic haptens. The presence of c-type particles in hybridomas suggests that T cell–derived cytokines are not required for sustained BLV expression.
The PBLs from a 2-year-old Holstein heifer afflicted with persistent lymphocytosis, as a result of natural BLV infection, were stimulated in vitro with mitogen before fusion with X63.Ag8.653 mouse myeloma as described earlier. 11 , 16 The supernatants from cloned hybridomas were tested for bovine IgM secretion in an enzyme-linked immunosorbent assay (ELISA) as described. 16 Positive (bovine serum) and negative (Roswell Park Memorial Institute [RPMI] 1640 medium supplemented with 10% horse serum) controls were included in each test. The hybridomas were frozen in 95% bovine fetal calf serum and 5% dimethylsulfoxide (Sigma Chemical Co., St. Louis, MO), and >90% cell viability was noted on subsequent thawing. The specificity of monoclonal IgM antibodies was determined in an enzyme immunoassay against a panel of antigens, including bovine serum albumin (BSA) fraction V (Boehringer Mannheim GmbH, Ingelheim am Rhein, Germany), bovine thyroglobulin, ubiquitin, trimethyl (p-aminophenyl)-ammonium chloride (TMA) coupled to BSA (substitution of 10 TMA molecules per molecule of BSA), 2,4,6-trinitrobenzenesulfonic acid (TNP) coupled to BSA (substitution of 26 TNP molecules per molecule of BSA), actin (all from Sigma Chemical Co.), mouse IgG1 (Chemicon International Inc., Temecula, CA, USA), and recombinant mycobacterial heat shock proteins hsp65 and hsp70 (courtesy of Dr. J. D. A. van Embden, UNDP/World Bank/WHO program) as described. 15 This panel of antigens includes self- and natural-conserved antigens as well as nonself synthetic haptens that have been used to characterize natural antibody specificities. 3 , 9 The polystyrene plates (Nunc, International, Rochester, NY) were coated at a concentration standardized for each antigen in bicarbonate buffer (pH 9.6) and kept overnight at 4 C. The hybridomas were stained with murine anti-bovine CD5 monoclonal antibody (VMRD Inc., Pullman, WA, USA) and fluorescein isothiocyanate–labeled goat anti-mouse IgG (Southern Biotechnology, Birmingham, AL, USA) for analysis by flow cytometry. Bovine PBLs and murine X63.Ag.653 myeloma were included as positive and negative controls, respectively.
The hybridomas were grown in RPMI 1640 medium, 16 and sequential growth characteristics of the 1 × 106 X63.Ag8.653 myeloma and hybridomas were measured at 6-hour intervals by counting viable cells by way of trypan blue dye exclusion.
Total RNA was extracted from hybridomas using Trizol reagent (GIBCO BRL, Gaithersburg, MD, USA), and complementary DNA (cDNA) was synthesized from 7 µg of total RNA using polydT primer (Pharmacia LKB Biotechnology, Uppsala, Sweden). The rearranged bovine VDJ (variable–diversity–joining) heavy-chain immunoglobulin genes were amplified by polymerase chain reaction (PCR) using 5′ (5′-AGCTCGAGATGAACCCACTGTG-3′) and 3′ (5′-AGACTAGTGAGGAGACGGTGACC-3′) primers. 15 , 16 The PCR steps included denaturation at 95 C for 1 minute and annealing and extension at 72 C for a total of 2.5 minutes up to a total of 30 cycles. A 434-bp fragment of the BLV env gene was PCR amplified from genomic DNA and cDNA prepared from hybridomas, using 5′ (5′-GTCTCCCAGATACACCTTGG-3′) and 3′ (5′-GAAGGTTCCCAACATATAGC-3′) primers from the BLV env gene sequence. 13 DNA samples from BLV-infected PBLs and X63.Ag8.653 mouse myeloma cells were included as positive and negative controls, respectively.
Twenty million hybridomas from each cell line were pelleted and fixed with 2.5% glutaraldehyde in 1.0 M phosphate buffer at pH 7.2. Samples were postfixed in 2.0% osmium tetroxide and embedded in epoxy resin. 17 Ultrathin sections (90 nm) were cut with an ultramicrotome and contrasted with uranyl acetate and lead citrate. The sections were mounted on copper grids and examined in a Jeol 100 S transmission microscope at 80 kV. For chromosomal analysis, hybridomas (∼45 passage level) were incubated with 0.1 µg/ml of colcemid for 3 hours in vitro, followed by treatment with hypotonic solution for 10 minutes. Cells were pelleted, fixed in acetic ethanol, and placed on microscopic slides. 18 Chromosomes stained with acetic acid orcein were examined under Photomicroscope I (Carl Zeiss, Göttingen Germany).
Stable monoclonal hybridoma cell lines secreting bovine immunoglobulin during a 5-year period (>50 passages) have been established by xenogeneic somatic cell hybridization of bovine PBLs with murine X63.Ag8.653 myeloma cells. Fusion of 87 × 106 PBLs led to generation of 44 bovine IgM–secreting mouse × bovine hybridomas (Table 1), although at a relatively low frequency (0.5 per 106 lymphocytes) compared with autologous somatic cell fusion. 9 The hybridomas were grown continuously for 3 months between passages. They tested positive for bovine IgM secretion during the 5-year period. None of the 44 hybridomas constructed from BLV-infected cattle tested positive for bovine IgG secretion. The VDJ rearrangements in these cell lines could be readily detected in a PCR assay (Fig. 1). The genetic analysis of the junctions of VDJ rearrangements expressed in the hybridomas indicated their diverse genetic origin. 15 , 16 None of the bovine Ig–secreting hybridomas expressed CD5 glycoprotein on the cell surface. The growth characteristics of xenogeneic hybridomas secreting bovine antibodies are comparable with the murine fusion partner X-63.Ag.653 myeloma (Fig. 2) and consistent with a malignant hybridoma cell. Of 44 IgM-secreting hybridomas from a single cow, four hybridomas (9.1%) showed multireactive binding with structurally dissimilar antigens such as actin, thyroglobulin, ubiquitin, mouse IgG1, rHSP-65, rHSP-70, and BSA and synthetic haptens such as TNP and TMA (Table 1). The pattern of multireactive binding varied among various B cell clones. Two clones, BLV1H12 and BLV5B8, recognized all the antigens in contrast to others that recognized either two (BLV8A5) or five (BLV10H8) test antigens. The majority of the clones, however, did not secrete immunoglobulin that bound to any of the test antigens (Table 1). The commitment of BLV-infected B cells to natural antibody secretion is similar to CD5+ B-cells that expand in CLL of humans. 3 The present study supports earlier observations that BLV infection causes generalized activation of B cells in cattle regardless of antigen specificity. 12 Such a generalized activation is distinct from multispecific binding of normal circulating immunoglobulin to structurally dissimilar antigens observed across species. 1 , 9 Taken together, both genetic and immunochemical evidence suggests a polyclonal origin of leukemic B cells as a result of BLV infection.
Antibody specificity of bovine IgM secreted by mouse × cattle xenogeneic hybridomas.

∗ Solid-phase ELISA based on optical density, (OD, 405 nm); OD >2.0 (++++), 1.0–2.0 (+++),0.5–1.0 (++),0.1–0.5 (+).
† Thirty-nine other IgM-secreting clones from the same animal had unknown binding specificity.
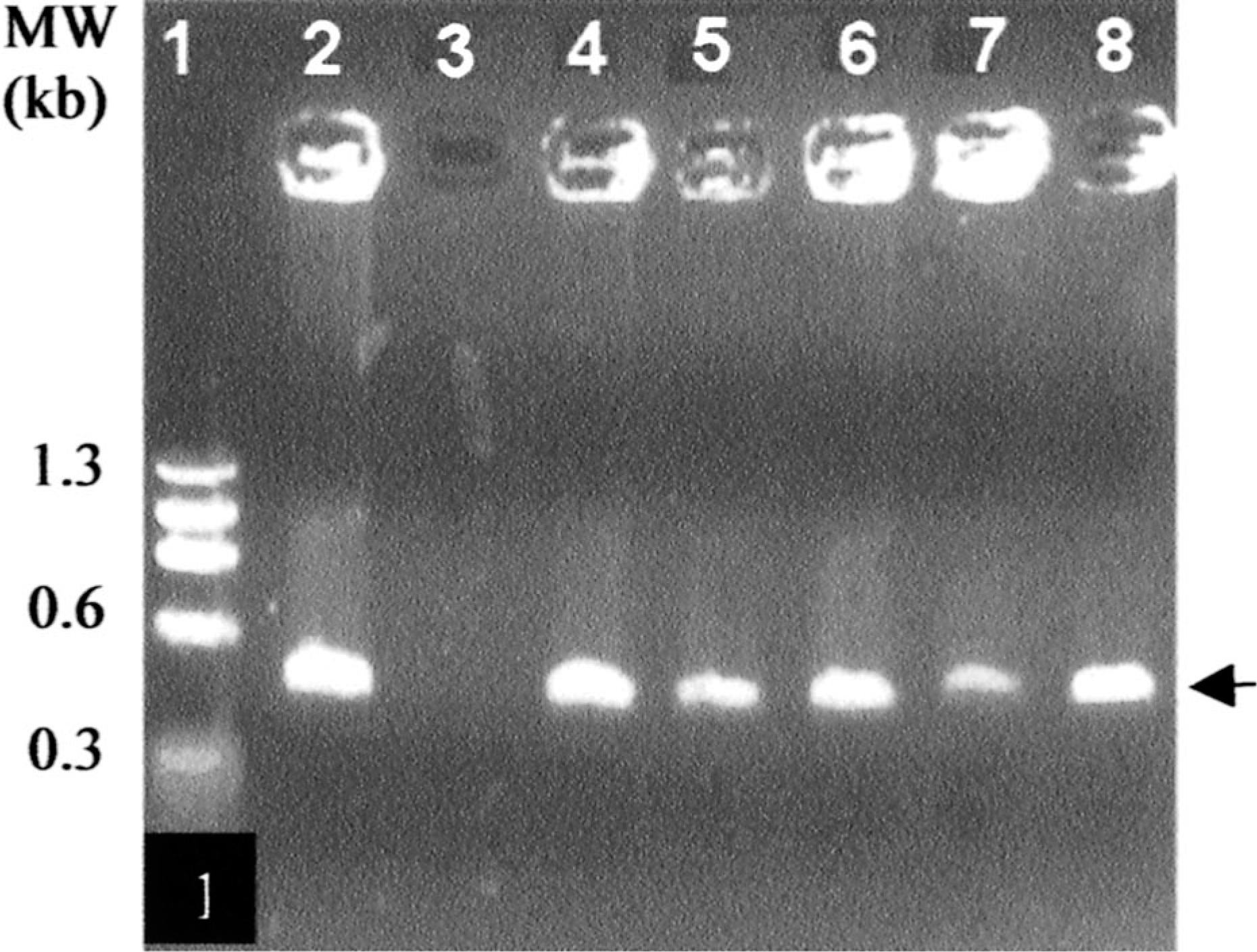
PCR amplification of VDJ rearrangements (arrow ∼450 bp) in hybridomas originating from PBL of BLV-infected cow. Lane 1, molecular weight. Lane 2, positive control, 9907 hybridoma. Lane 3, negative control, mouse myeloma. Lane 4, BLV9B8. Lane 5, BLV9C12. Lane 6, BLV9H3. Lane 7, BLV 10H2. Lane 8, BLV11F3
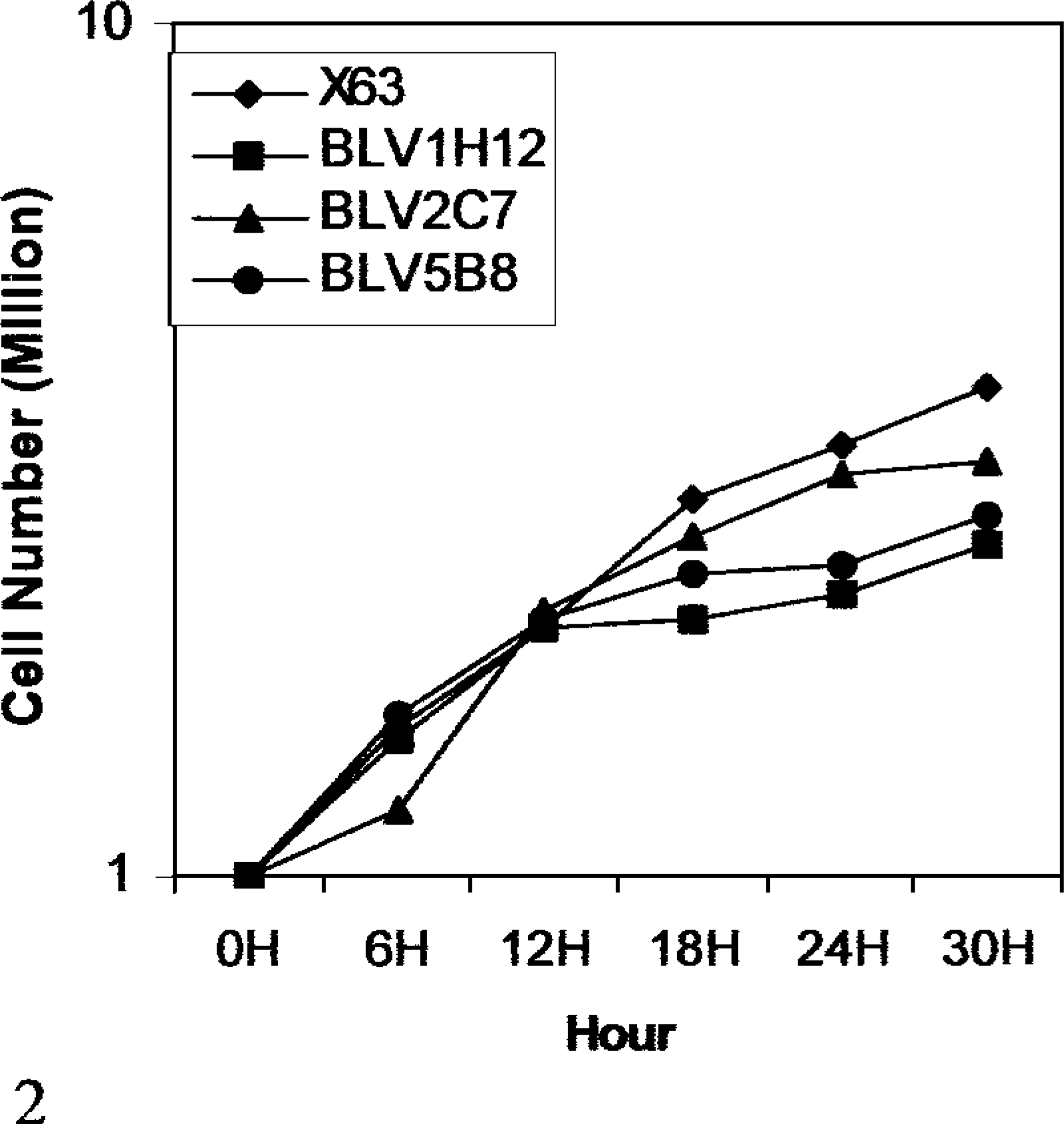
Growth characteristics of mouse × bovine xenogeneic hybridomas
As expected, the three hybridoma cell lines examined contained approximately 90 chromosomes, consistent with the total number of chromosomes in mouse and bovine cells. The marker metacentric chromosome of the mouse myeloma cells was found in all the hybrid cells (Fig. 3). The chromosome number of mouse myeloma cells ranged from 38 to 42, including the metacentric marker chromosome. The ability to retain approximately 90 chromosomes and secrete bovine immunoglobulin during the 5-year period reflects stable establishment of xenogeneic hybridomas.
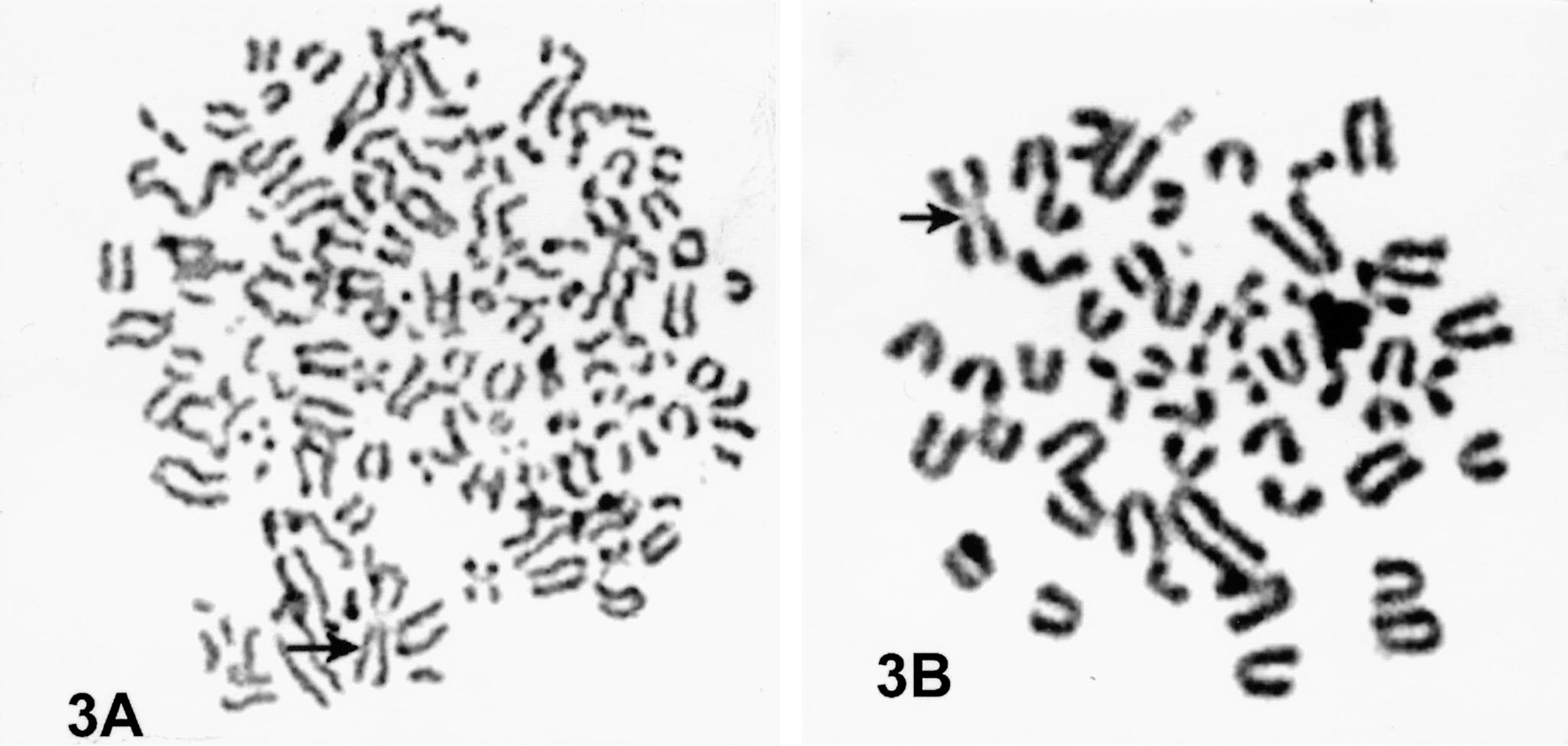
Fig. 3A. Chromosomal spread of mouse × bovine xenogeneic BLV1H12 hybridoma showing the number and type of chromosomes indicative of murine and bovine cells. The metacentric marker chromosome (arrow) confirms the murine complement. Fig. 3B. Chromosomal preparation of myeloma cell exhibiting metacentric chromosome (arrow).
Electron microscopy of mouse myeloma cells demonstrated pleomorphism and high mitotic activity, commonly observed in the tumor cells during in vitro culture. The cytoplasm had a few vesicles containing fragments of membranous substance consistent with swollen mitochondria with disrupted cristae. Free ribosomes were abundant throughout the cytoplasm. Although the cells contained multilobed nuclei, none were multinucleated. The tumor cells appeared to have stabilized during in vitro culture because they remained uninucleated. Interestingly, 30–50% of mouse × cattle hybridoma cells were large with multiple nuclei. The multinucleated giant cells also contained a large number of ribosomes in the cytoplasm, similar to those observed in the mouse myeloma cells. Mitochondria were slender and found in relatively small numbers. There were a few vesicles containing finely particulate material. Hybridoma cells contained dilated rough endoplasmic reticulum (RER), and cisternae were filled with a moderately electron-dense substance consistent with the appearance of plasma cells in presecretory stage (Fig. 4). 5 Such characteristics, however, varied among individual cell lines. The granular appearance of the substance in the cisternae may be related to the molecular size of the bovine IgM antibody because some hybridomas secreted IgM with an exceptionally long third complementarity-determining region of the heavy chain (CDR3H), e.g., BLV1H12 (61 amino acids) and BLV5B8 (56 amino acids) as compared with others such as BLV2C7 (19 amino acids). 15 The contents of the cisternae in different hybridoma cells did not show obvious differences in coarseness. Higher-resolution and immunoelectron microscopy may yield additional information regarding the morphologic differences between hybridomas secreting antibodies with exceptionally long or short CDR3H regions. The fact that the hybrid cells remained stable through cell divisions without becoming polyploid indicates that they are permanently established cell lines. Moderate numbers of particles, with a dark peripheral ring and clear center, were observed extracellularly as well as in the peripheral cytoplasm of hybridoma cells (Fig. 5). These particles are consistent with the electron microscopic appearance of c-type virus particles. 8 Indeed, the presence of c-type viral particles was confirmed by PCR amplification of a 434-bp segment of BLV env gene from these hybridomas (Fig. 6). The presence of c-type viral particles in the hybridomas, in the absence of either T cell–derived cytokines or phorbol ester mitogen, suggests their ability to replicate during in vitro culture. Nevertheless, it remains unclear how viral replication is initiated. The contribution of phorbol ester to initiation of viral replication cannot be excluded because BLV-infected B cells were stimulated with the mitogen before cell fusion.
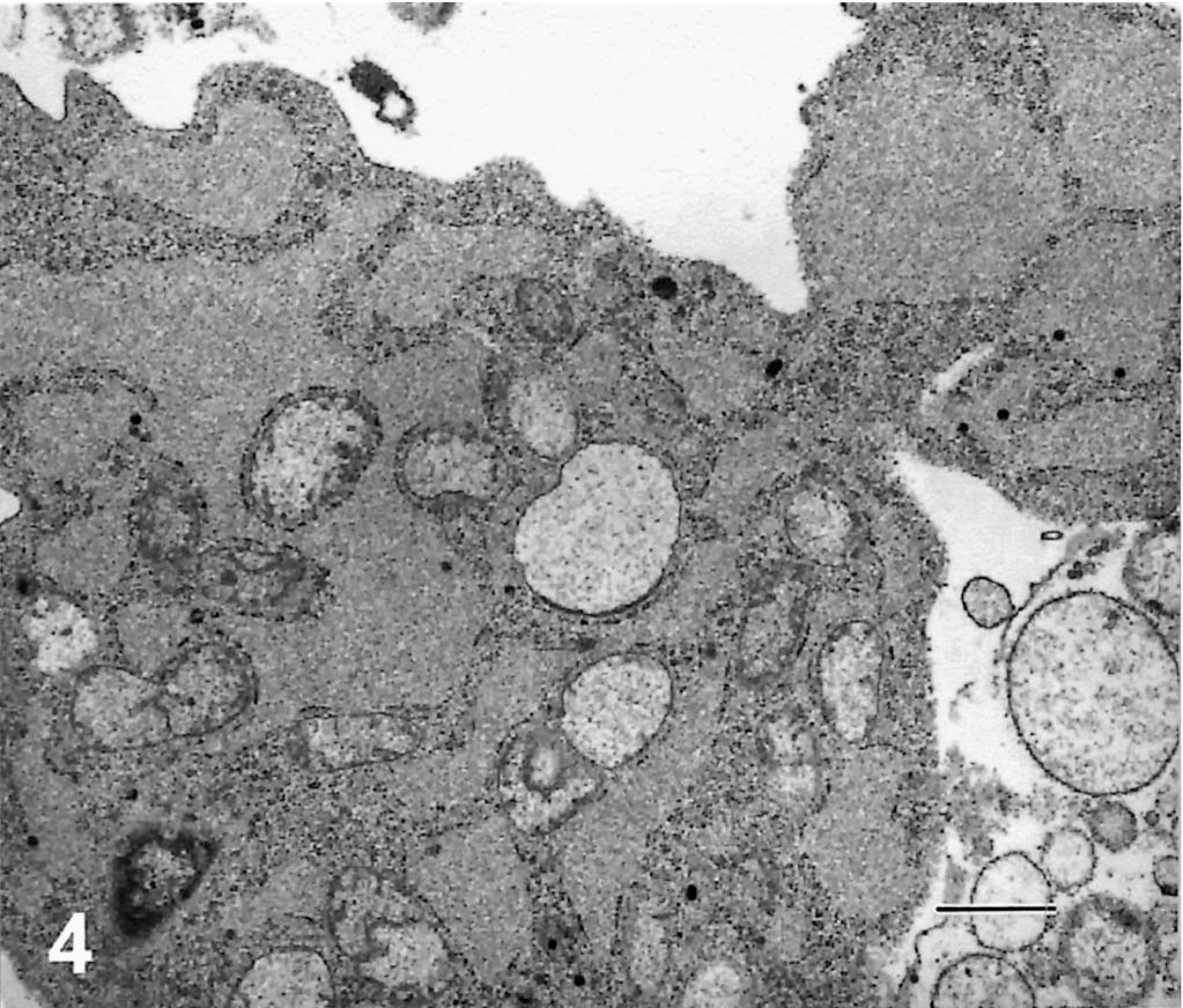
Cytoplasm of BLV5B8 hybridoma cells is mostly occupied by RER. The dilated cisternae of the RER are filled with a moderately granular substance. Electron-opaque small particles are scattered in the cytoplasm. Bar = 1 µm.
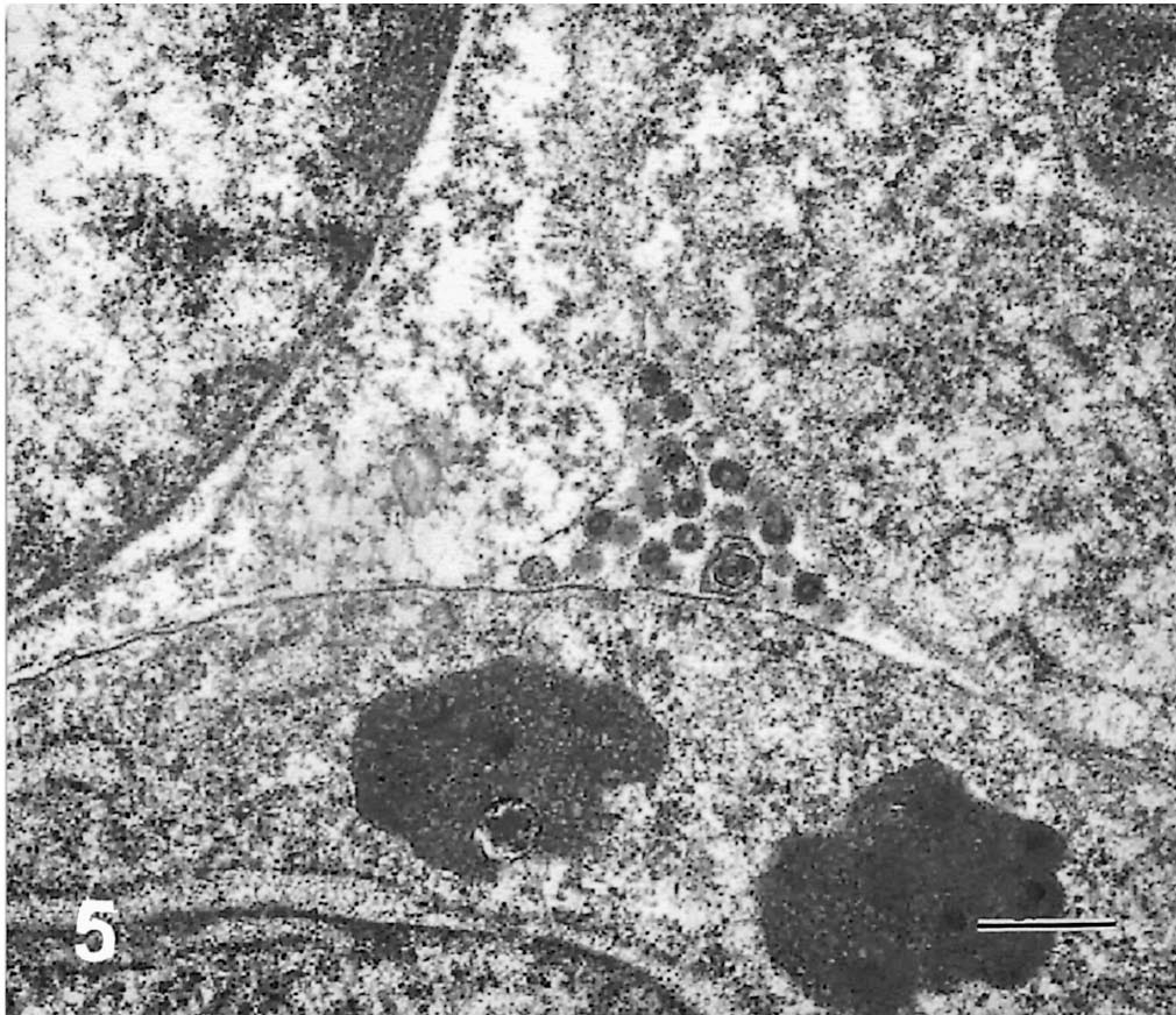
BLV5B8 hybridoma cells contained c-type virus particles in the cytoplasm. Electron-opaque ring with lucent center is a characteristic appearance of the retrovirus. Bar = 0.5 µm
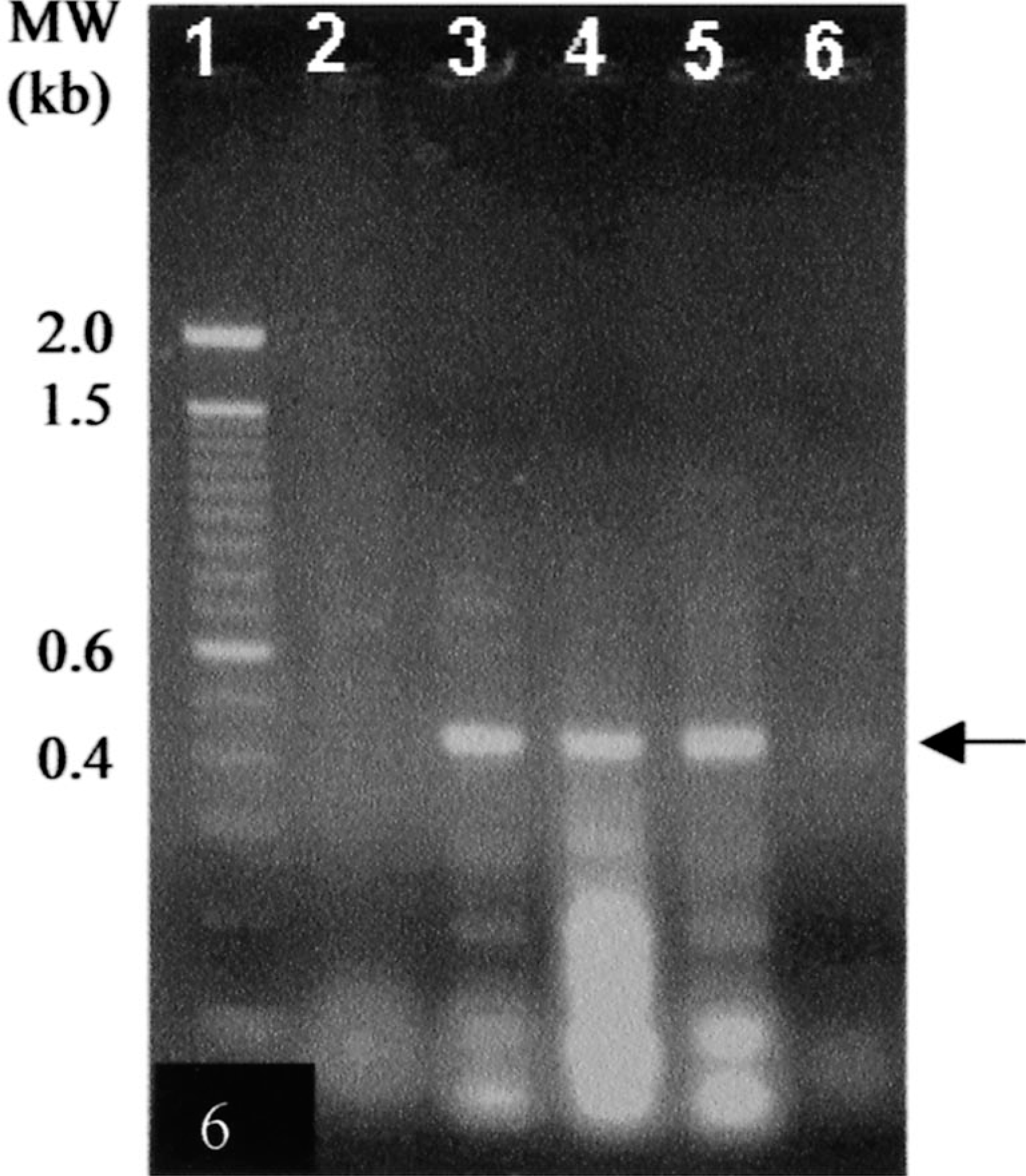
PCR amplification of env gene of BLV (arrow ∼434bp) in IgM-secreting xenogeneic hybridomas. Lane 1, molecular weight. Lane 2, myeloma cells. Lane 3, PBLs from BLV-infected cattle. Lane 4, BLV1H12 hybridoma. Lane 5, BLV5B8 hybridoma. Lane 6, BLV2C7 hybridoma
Overall, these experiments demonstrate establishment of long-term mouse × cattle hybridomas from BLV-infected B cells that secrete monoclonal antibody of various specificities. The BLV-infected leukemic B cells in circulation are of polyclonal origin and capable of secreting natural antibodies. Further, presence of c-type particles in the hybridomas suggests that T cell–derived cytokines are not required for sustained BLV expression.
