Abstract
In the summer of 2000, proliferative lesions of the skin and oral mucosa were observed in 26 young harbor seals (Phoca vitulina) from a rehabilitation center in Schleswig-Holstein, Germany. Verrucose, roundish nodules, approximately 1–2 cm in diameter, were presented in the oral cavity, especially on the tongue. Some animals developed similarly sized spherical dermal elevations with ulceration on flippers, chest, neck, and perineum. Necropsy of one animal showed multifocal, verrucose nodules in the oral cavity and a mild tonsillitis. Histologically, the nodules were characterized by ballooning degeneration of the outer parts of the spiny layer and stratum granulosum, with large eosinophilic cytoplasmic inclusions and a perivascular to interstitial lymphohistiocytic infiltration accompanied by fibroblastic proliferation and neovascularization. Negative staining of mucosal tissue homogenates demonstrated parapoxvirus-like particles. The presence of parapoxvirus was confirmed by polymerase chain reaction, using primers specific for parapoxvirus of ungulates. By in situ hybridization, using a parapox-specific, digoxigenin-labeled DNA probe, abundant parapoxvirus DNA-positive epithelial cells were detected in the stratum granulosum and the outer parts of the spiny layer. There was no parapoxvirus-positive signal in the adjacent submucosa. Although DNA analysis revealed that the causative agent can clearly be distinct from terrestrial parapoxviruses, lesions resembled parapoxvirus infections in other terrestrial species, and the pattern of virus DNA distribution indicated a direct effect of the virus on keratinocytes. In contrast, changes in the corium may be considered an indirect response mediated by the virus or the immune system.
The family of Poxviridae represents a large group of complex, highly epitheliotropic DNA viruses causing cutaneous and systemic diseases in different animal species and humans. 22 The subfamily Chordopoxviridae consists of several genera with a wide host range of terrestrial and marine wildlife species. 22 Diseases caused by infections with members of the genus parapoxvirus include contagious pustular dermatitis (orf) in sheep and goats and pseudocowpox and bovine papular stomatitis in cattle. 16 Furthermore, parapoxvirus infections have been described in other ungulate species, including reindeer, 3 red deer, 13 , 27 chamois, 16 muskoxen, 18 camels, 16 gazelles, 40 and wild Japanese serows. 32 In nonungulates, disease has been observed in red and gray squirrels, 4 pygmy chimpanzee, 26 and seals. 22 Only orf virus, papular stomatitis virus, pseudocowpoxvirus, parapoxvirus of red deer in New Zealand, and squirrel parapoxvirus are accepted members of the parapoxvirus genus. Tentative species in the genus are auzduk disease virus, chamois contagious ecthyma virus, and sealpox virus. 20 As animal parapoxviruses are zoonotic, dermal lesions also have been reported in man. 16
Transmission can occur via small dermal abrasions through contact with infected animals or contaminated crusts, which can sustain the infectious virus for a long time. 16 The morphology and predilection of parapox lesions vary among the different virus species and range from macules and papules to pustules, with ulcerations of the skin of head, extremities, mammary gland or other areas of sparsely haired skin, and the mucosa of the oral cavity. In contagious pustular dermatitis of sheep, the pustules show exudation and development of marked verrucose crusts. 16 , 22 A papillomatous growth of the parapox lesions has been described in sheep and Finnish reindeer. 3 Rarely, severe disease due to systemic infection has been reported in lambs. 16 In man, the commonly localized lesions are usually restricted to the hands and are characterized by an inflammatory papule, which may progress to a granulomatous lesion before regression. Such lesions may be termed milker's nodules if acquired from cows or orf from sheep or wild artiodactyls. 5 , 22 Histologically, lesions induced by parapoxvirus consist of epithelial proliferation with epidermal cytoplasmic swelling, vacuolation, reticular degeneration, and cytoplasmic inclusion bodies in keratinocytes. In addition, dermal, perivascular, mononuclear infiltrates accompanied by variable numbers of neutrophils are observed. Furthermore, epidermal microabcesses and scale crusts are detected. 15 In pinnipeds, the first identification of a seal poxvirus was reported in a North American California sea lion (Zalophus californianus) in 1969. 36 First, electron microscopic studies of the viral particles revealed that sealpox virus may belong to the paravaccinia subgroup of poxviruses. 39 In the following years, the disease has been reported from several other species, such as South American sea lions (Otaryia bryonia), 38 harbor seals (Phoca vitulina), 37 northern fur seals (Callorhinus ursinus), 10 and gray seals (Halichoerus grypus) 12 from North America. Sealpox infection in European waters was observed in gray seals from the UK 24 , 29 , 30 and the Netherlands. 25 Clinically, the disease is characterized by dermal nodules found on various parts of the skin and oral mucosa. 23 , 29 , 36 In contrast to other pinniped species, different histopathologic manifestations of sealpox virus infection have been reported in northern fur seals from the North American Pribilof Island and South American sea lions. Northern fur seals showed dermal epithelioma-like lesions without connection to the overlying epidermis. 10 In captive South American sea lions, an infiltration of the dermis by a downward proliferative mass of epithelial cells has been reported. 38
Cetaceans are another species of marine mammals susceptible to poxvirus infection. Poxvirus is detected in several captive and free-ranging species of odontocetes and resembles orthopoxvirus ultrastructurally. 2 , 9 , 33 Poxvirus infection in cetaceans usually induces flat or slightly raised or depressed epidermal ring or pinhole lesions. 2 , 9 Advanced lesions, so-called tattoos, are composed of black lines or circles of epidermal hyperpigmentation. In contrast to pox lesions in other animals, there is no nodular proliferation of the infected epidermis. 2
This report describes the clinical and pathologic findings after an outbreak of parapoxvirus infection in captive harbor seal neonates and juveniles from the German North Sea.
Materials and Methods
Seals
In the early summer of 2000, 28 abandoned, young harbor seal pups between 1 and 10 days of age were recovered from the end of May to the end of June at different sites in the Wadden Sea of Schleswig-Holstein, Germany. The pups were transported to a center in Friedrichskoog, Germany, for rehabilitation. Eleven of the animals were males, and 17 were females. The seals were housed in eight outdoor pools with laminated plastic surface and a sand-covered haul-out. Each pool had a separate fresh water supply. Seven smaller basins were used for nursing, and in one larger pool animals were housed before their release back into the wild. The smaller enclosures used for nursing had an average fresh-water capacity of 15 kl, with a daily complete volume exchange, and were cleaned up every day. The basin where the animals were housed before their release back into the wild had a filling capacity of 160 kl and a permanent fresh-water supply and was cleaned every third day. In addition, one exhibition pool, separated from these pools, was occupied by adult seals. The harbor seal pups were divided into small groups of about four animals, according to age and developmental stage, and placed in the rehabilitation pools. Some pups were moved occasionally among the different basins. After the outbreak of parapoxvirus infection, the number of inhabited pools was reduced, and seals were collected in one basin to control the epizootic. The seals were fed with a reconstituted powdered milk ration (Multi melk, PetAg Inc., Hampshire, Illinois, USA) until 10 days of age and later with fresh herring mash until they started to eat whole and live fish. Regular clinical examinations included routine hematology and clinical chemistry. Before identification of the parapoxvirus infection at the facility, a prophylactic application of a paraimmunization stimulant containing inactivated parapox ovis viruses (Baypamun, Bayer AG, Leverkusen, Germany) for prevention of stress-associated infectious diseases was given to several seals after their arrival in the rehabilitation center. To limit disease dissemination, the caretakers wore disposable gloves, and different clothes were used for each enclosure. For prevention of secondary bacterial infection of ulcerated dermal nodules, topical antiseptic biguanide solution (Lavasept, Fresenius AG, Bad Homburg, Germany) was applied regularly.
Light and electron microscopy
For histology, tissue samples of skin and mucosa of the oral cavity were collected from three pups (Nos. 1–3) and fixed in 10% nonbuffered formalin. Unfortunately, pup No. 3 died of unknown causes during general anesthesia. Additionally, tissues from various other organs were obtained during necropsy of pup No. 3. Paraffin-embedded sections were processed for light microscopy and stained with hematoxylin and eosin (HE) using standard methods.
Skin samples from seal No. 2 and tissue of the mucosa of the oral cavity from seal No. 3 were prepared for negative staining using 2% (w/v) methylamine tungstate. Small droplets of the nondiluted sample material were placed on pioloform (Plano, Marburg, Germany) and carbon-coated 400 mesh copper-rhodium grids for 5 minutes. After incubation on top of the sample droplets, grids were blotted, and excess liquid was removed by filter paper. Thereafter, the grids were incubated for 60 seconds with 2% (w/v) methylamine tungstate and then air dried. The samples were examined using a Zeiss EM-902 transmission electron microscope at a voltage of 80 kV. Photographs were taken at an instrumental magnification of 31.500×, using a SO-163 electron-image film (Kodak, Rochester, New York, USA).
In addition, formalin-fixed tongue tissue from seal No. 3 was postfixed in 2.5% glutaraldehyde and washed in 0.166 M cacodylate buffer. Postfixation was done in 1% osmium tetroxide (0.166 M cacodylate buffer, pH 7.32) for 24 h at 4 C. After dehydration in acetone, tissues were embedded in Epon 812. For orientation, 0.5- to 1.0-µm-thick samples were sectioned and stained with toluidine blue. Representative sections were selected and cut at 60–90 nm, stained with uranyl acetate and lead citrate, and examined by a Zeiss EM-10 C electron microscope.
In situ hybridization
Tongue sections of seal No. 3 were investigated for parapox-specific DNA using polymerase chain reaction and in situ hybridization (ISH), as described previously. 1 , 7 , 8 , 14 Skin tissue from an unaffected seal, a rabbit with myxomatosis as determined by light microscopy, and a surgical sample from a sheep with infectious pustular dermatitis (orf) served as controls. For exclusion of unspecific signals, tongue sections of pup No. 3 were investigated for morbillivirus-specific RNA, using ISH with a digoxigenin-labeled DNA probe. 7 A universal parapox primer pair, P1 (5′-GTCGTCCACGATGAGCAGCT-3′) and P2 (5′-TACGTGGGAAGCGCCTCGCT-3′; GeneBank accession number U06671), amplifying part of the genomic region encoding the putative virion envelope antigen (B2L gene), which is homologous to the vaccinia virus major envelope antigen p37K, was used to generate a double-strand–specific digoxigenin-labeled DNA probe of about 600 bases. 14 , 31 To improve probe penetration, slides were heated on a hot plate at 96 C for 10 minutes. After proteolytic digestion, postfixation, and prehybridization, hybridization was performed overnight at 52 C in a moist chamber. 7 , 8 The sections were incubated with an antidigoxigenin antibody conjugated with alkaline phosphatase (Boehringer, Mannheim, Germany). For visualization of the color reaction, nitro blue tetrazolium (NBT; Sigma, Buchs, Switzerland) and 5-bromo-4-chloro-3-indolyl phosphate (BCIP) were used.
Bacteriology, immunohistochemistry, and serology
Bacterial culture of various organs from seal No. 3 was performed according to routine standard methods. Immunohistochemistry for detection of morbillivirus antigen was conducted on lung tissue of seal No. 3, using a polyclonal antibody directed against canine distemper virus (CDV) nucleoprotein (rabbit No. 162, kindly provided by Dr. C. Örvell, Sweden) and the avidin–biotin–peroxidase technique. 21 Paired blood samples, taken at the beginning and end of the housing period, were collected from 25 pups, and a virus neutralization assay for detection of canine distemper virus–specific antibodies was performed using the CDV-Onderstepoort strain. Titers were determined by inhibition of virus-induced cytopathogenic effect after 3 days of incubation. Because of cytotoxicity of some sera, titers of 20 or greater were regarded as positive. 6 , 21
Results
Case histories and clinical examination
A young female harbor seal pup (No. 1), approximately 5–6 days of age, was found in mid-June 2000 on the coast of Pellworm/Schleswig-Holstein, Germany, and was taken to the rehabilitation center at Friedrichskoog, Germany. The seal was housed with several other young, healthy pups. One day after the arrival of the seal, the animal developed a discrete red ulcer, 2–3 mm in diameter, on the tip of the tongue. After 6 days, the lesion had increased in size and showed a whitish discoloration, and during the next 3 weeks several similar changes developed in the mouth and on the tongue. Twenty-four days after the arrival of pup No. 1, similar lesions were observed in the corners of the mouth and on the tongue of two other animals. One of these seals was housed with pup No. 1. Thereafter, similar alterations, characterized by ulceration in the oral cavity followed by a verrucose growth, were observed in several other seals (including animal Nos. 2 and 3; Figs. 1, 2). In addition, spheric dermal elevations, approximately 1–2 cm in diameter, with ulceration were found on flippers, chest, neck, and perineum. Seals infected later displayed only dermal lesions and lacked changes on the mucous membranes. By reducing the number of inhabited pools and increasing the stocking density, there was an increase in the incidence of diseased pups. Affected animals displayed various mucous or cutaneous changes (or both), which healed spontaneously in a period of about 3 weeks to 10 months. Most of the animals displayed a regression of dermal lesions between 16 and 25 weeks after infection. Hematology in the initial phases of the infection revealed a moderate lymphocytic leukocytosis and neutropenia in all animals. Fatalities due to parapoxvirus infection were not observed. One female seal pup developed a small, whitish proliferation, 0.5–0.2 cm in diameter, on the underside of the tip of the tongue. Death was attributed to trauma and occurred 7 weeks after the arrival of pup No. 1. One more animal died on the day of arrival, and 1 died 2 days after the arrival of pup No. 1, with clinical signs of catarrhal enteritis of undetermined cause. Both animals lacked lesions characteristic of parapoxvirus infection. Finally, a total of 26 pups were affected by mucous or cutaneous alterations (or both) due to parapoxvirus infection. Though adult seals in the separated exhibition pool had accidental contact with infected seal pups after a short period of time, these animals showed no parapoxvirus-related disease.
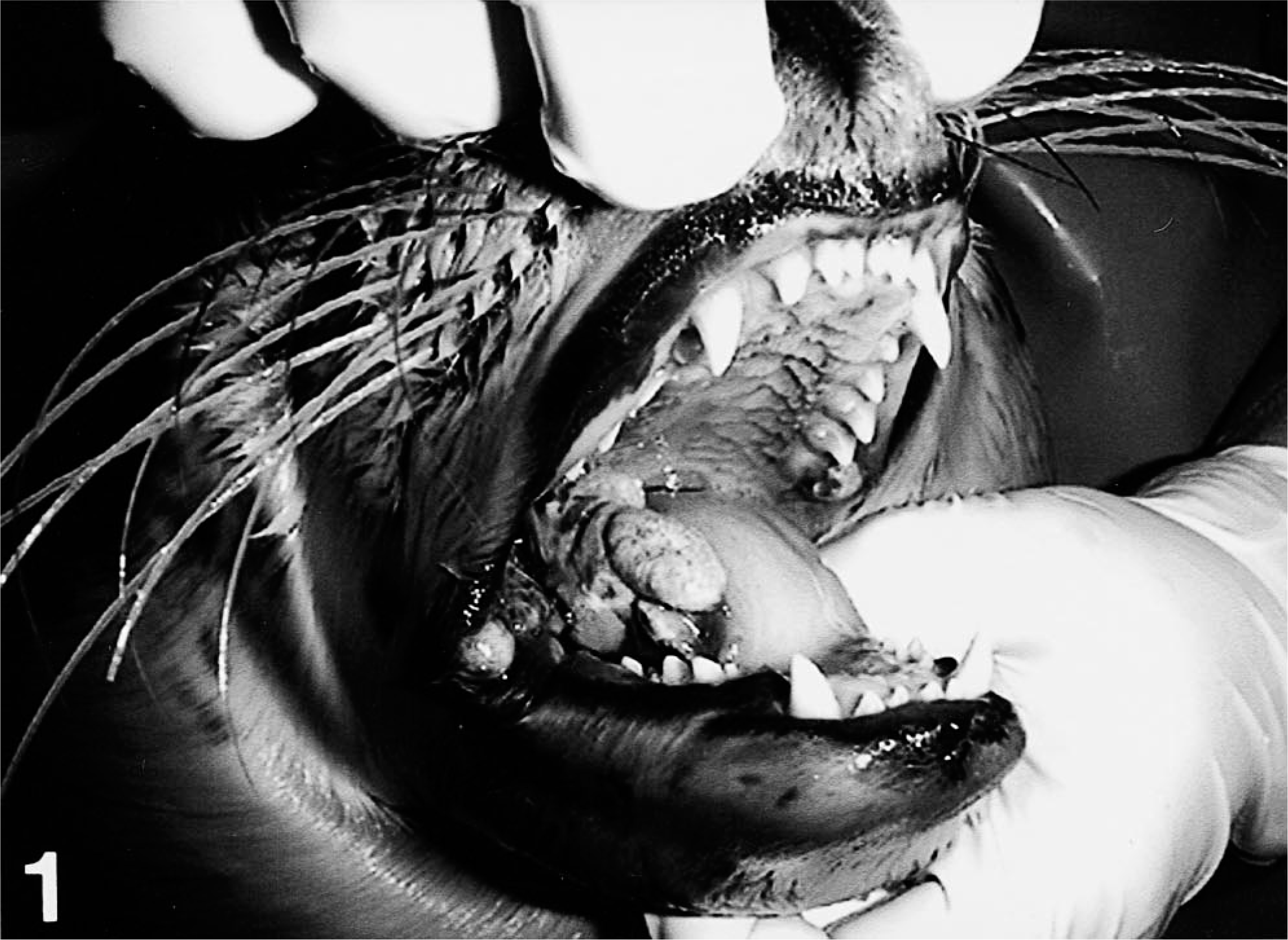
Harbor seal pups showing verrucose, roundish to ovoid nodules in the mouth corner and on the tongue
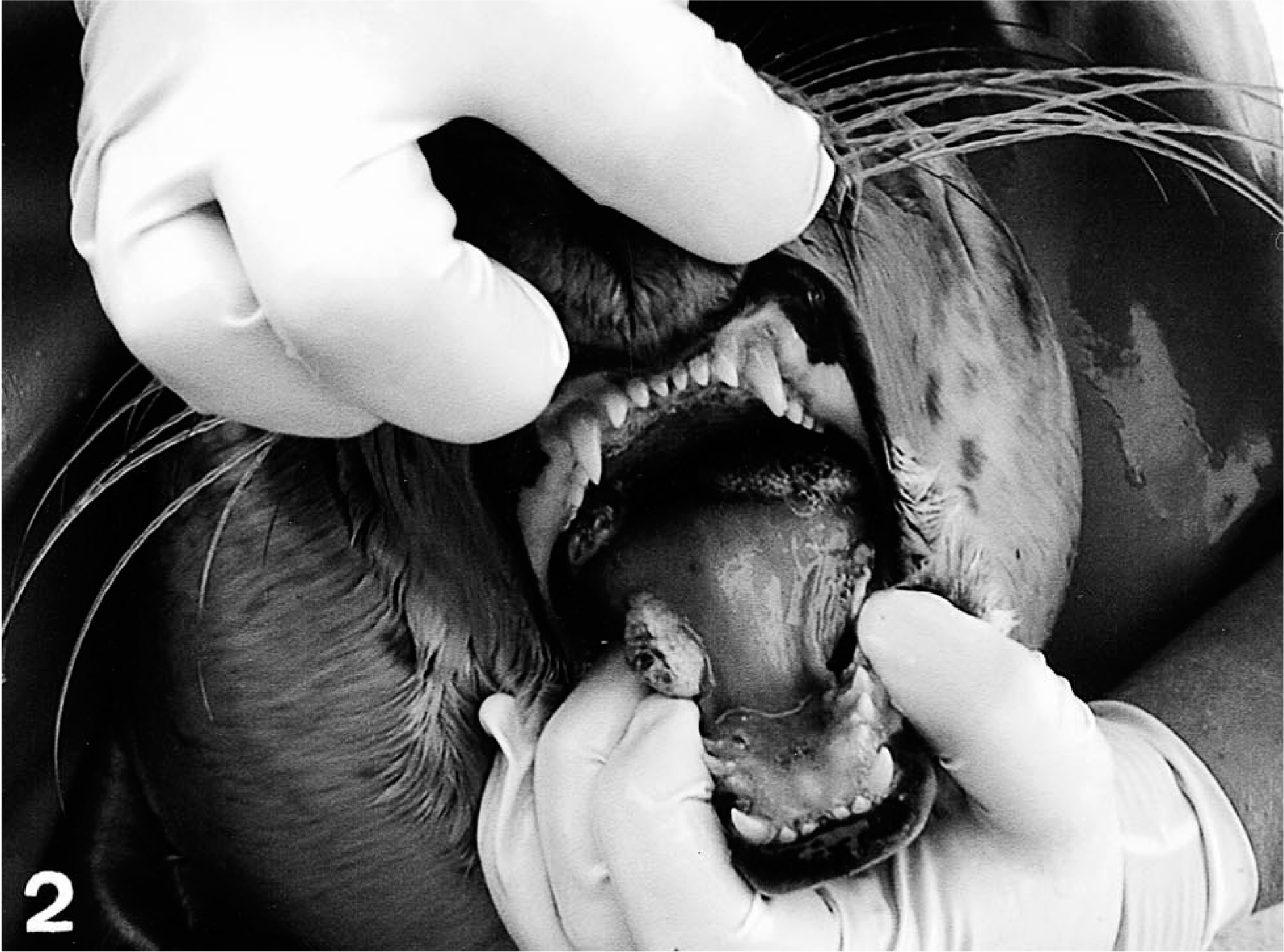
Harbor seal pups showing verrucose, roundish to ovoid nodules in the mouth corner and on the tongue
Necropsy findings
At necropsy, seal No. 3 was 18.5 kg and in good body condition. The thoracic and neck blubber thickness was 21 and 20 mm, respectively. In the oral cavity, multifocal white-gray verrucose nodules, 0.5–1.5 cm in diameter, were observed. In addition, lesions were localized at the base of the tongue, within the oropharynx, and along the soft palate. In the upper jaw, there was one nodule, 0.3–0.3 cm in diameter, at the gum near the right caninus. Additionally, a mild tonsillitis was detected. All other organs were without significant lesions.
Histology, ultrastructural, and ISH findings
In seal pup No. 3, light microscopy showed a mild to moderate lymphocytic depletion in the spleen. There was a mild follicular hyperplasia of the retropharyngeal lymph nodes with single-cell necrosis. Histologic examination of the nodules from the mucous membranes of the oral cavity of seal Nos. 1 and 3 revealed similar changes. Lesions were characterized by epithelial hyperplasia, acanthosis, ballooning degeneration of the outer parts of the spiny layer and stratum granulosum, and eosinophilic cytoplasmic inclusions most prominent within the stratum granulosum (Figs. 3, 4). Few necrotic and apoptotic cells were detected in the stratum spinosum. A focally extensive, severe, exuberant, perivascular to interstitial infiltration with histiocytes, lymphocytes, and fewer neutrophils accompanied by fibroblastic proliferation and neovascularization was found in the submucosa (Figs. 5, 6). In addition, the dermal nodules of seal No. 2 showed supraepidermal serocellular crusts and bacteria, accompanied by exocytotic degenerated neutrophils, ortho- and para-keratotic hyperkeratosis, and subcorneal microabscessation. Further changes in the epidermis and corium resembled those observed in the epithelium and submucosa of seal pups Nos. 1 and 3.
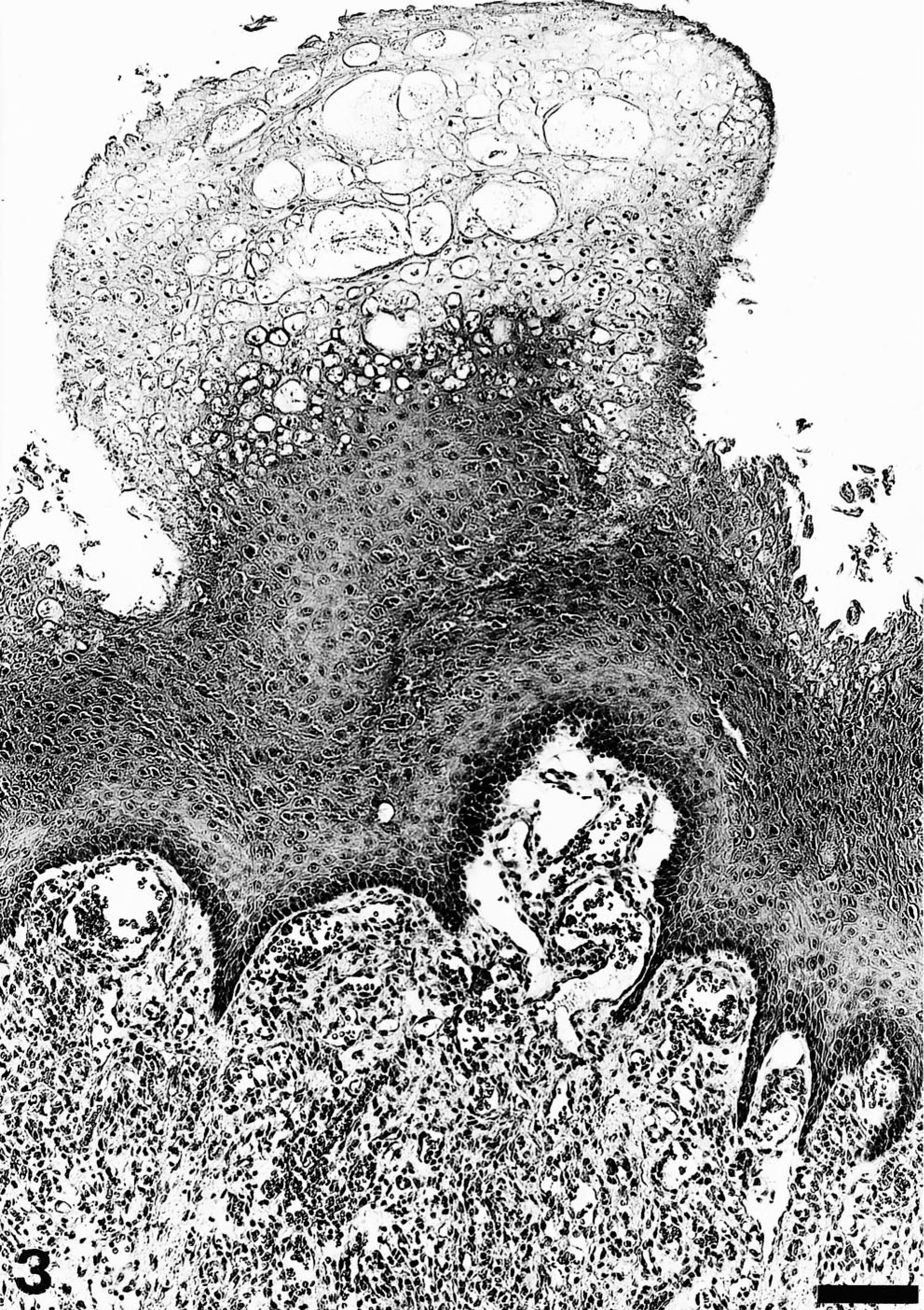
Mucosa of the oral cavity; pup No. 1. Orthokeratotic hyperkeratosis, epithelial hyperplasia, and ballooning degeneration of the stratum granulosum. HE. Bar = 103 µm.
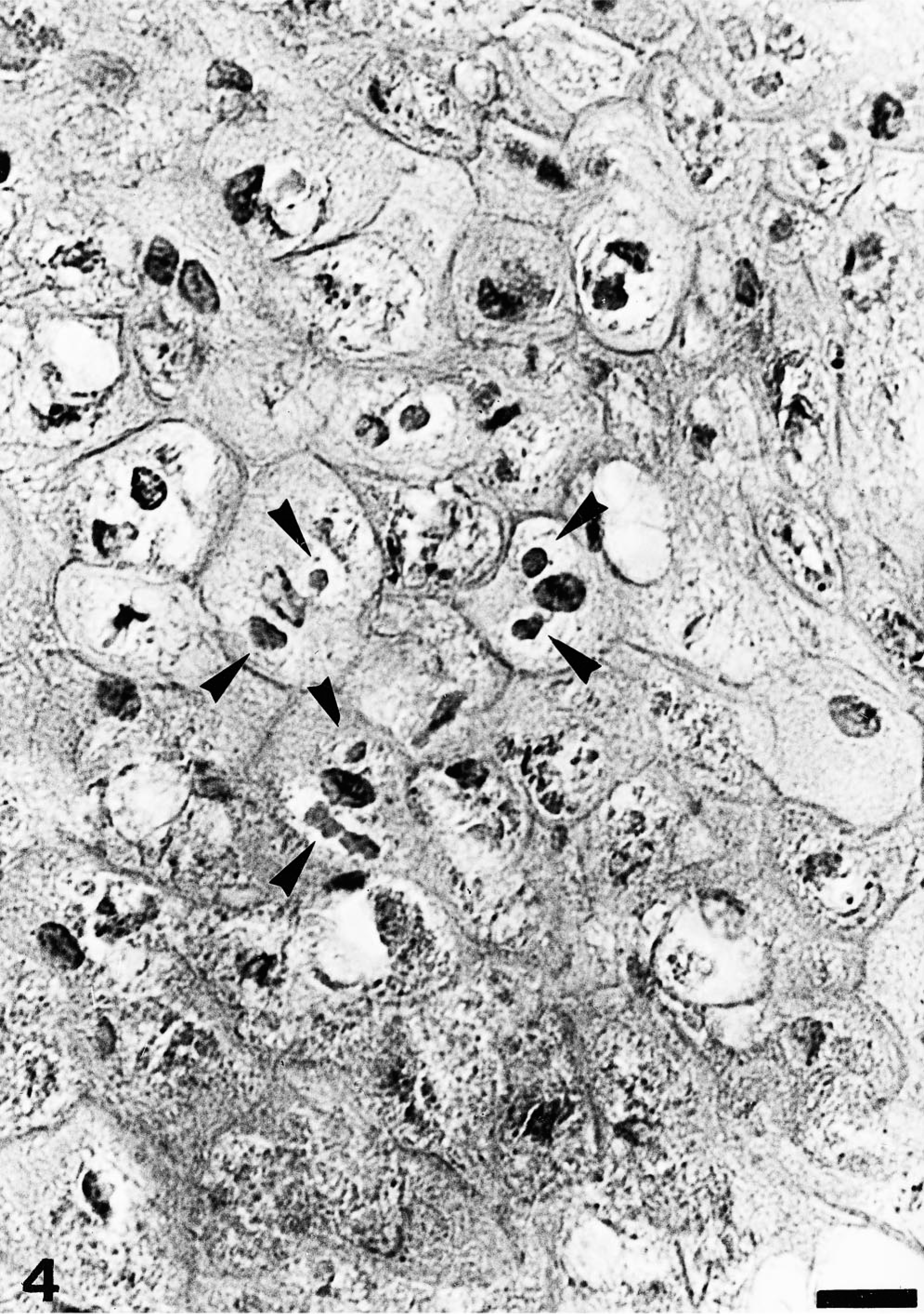
Mucosa of the oral cavity; pup No. 1. Parapoxvirus lesion with degenerating cells of the stratum granulosum and eosinophilic cytoplasmic inclusion bodies (arrowheads). HE. Bar = 16 µm.
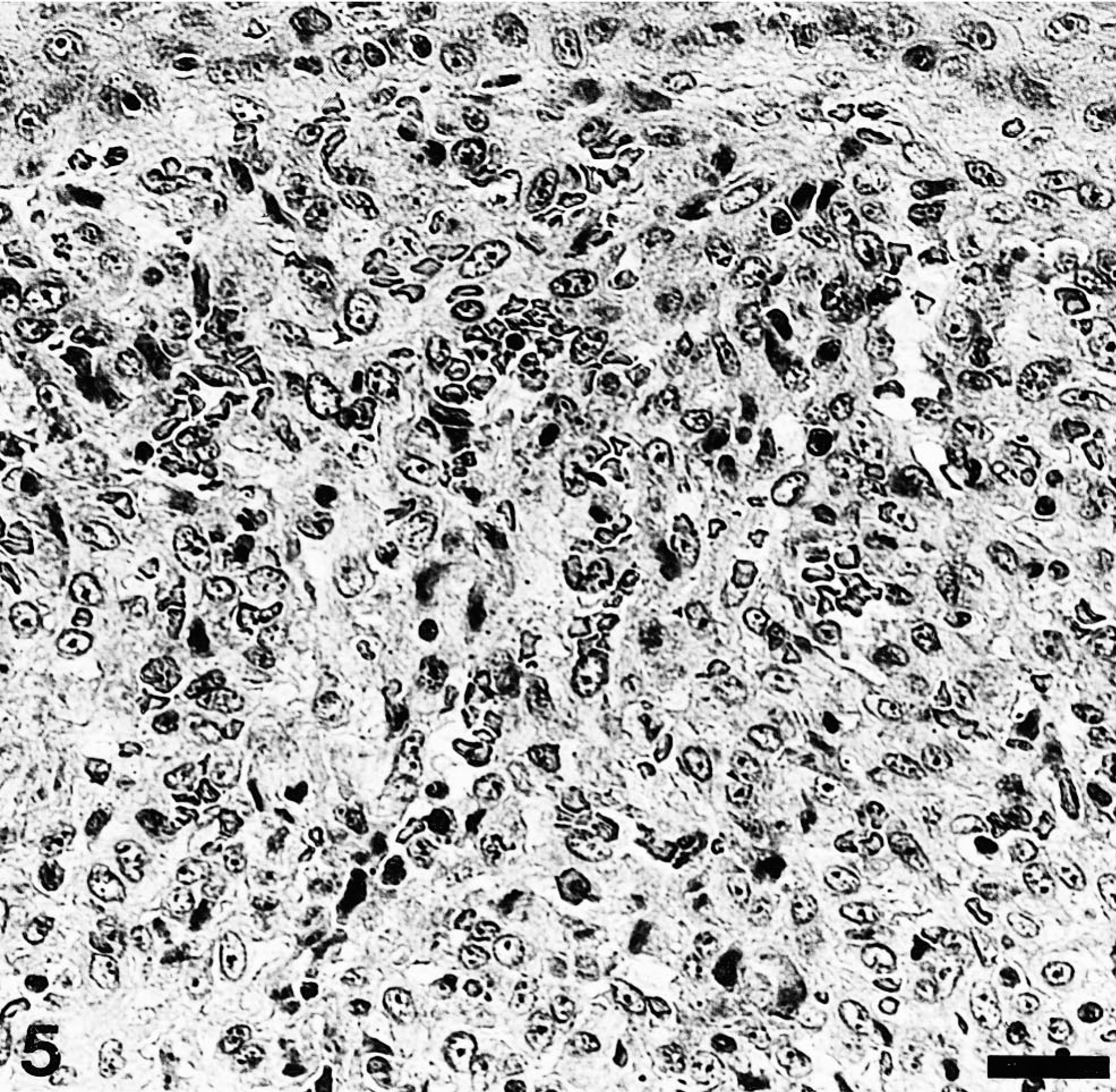
Mucosa of the oral cavity; pup No. 3. Severe lymphohistiocytic infiltration and moderate fibroblastic proliferation of the submucosa. HE. Bar = 26 µm.
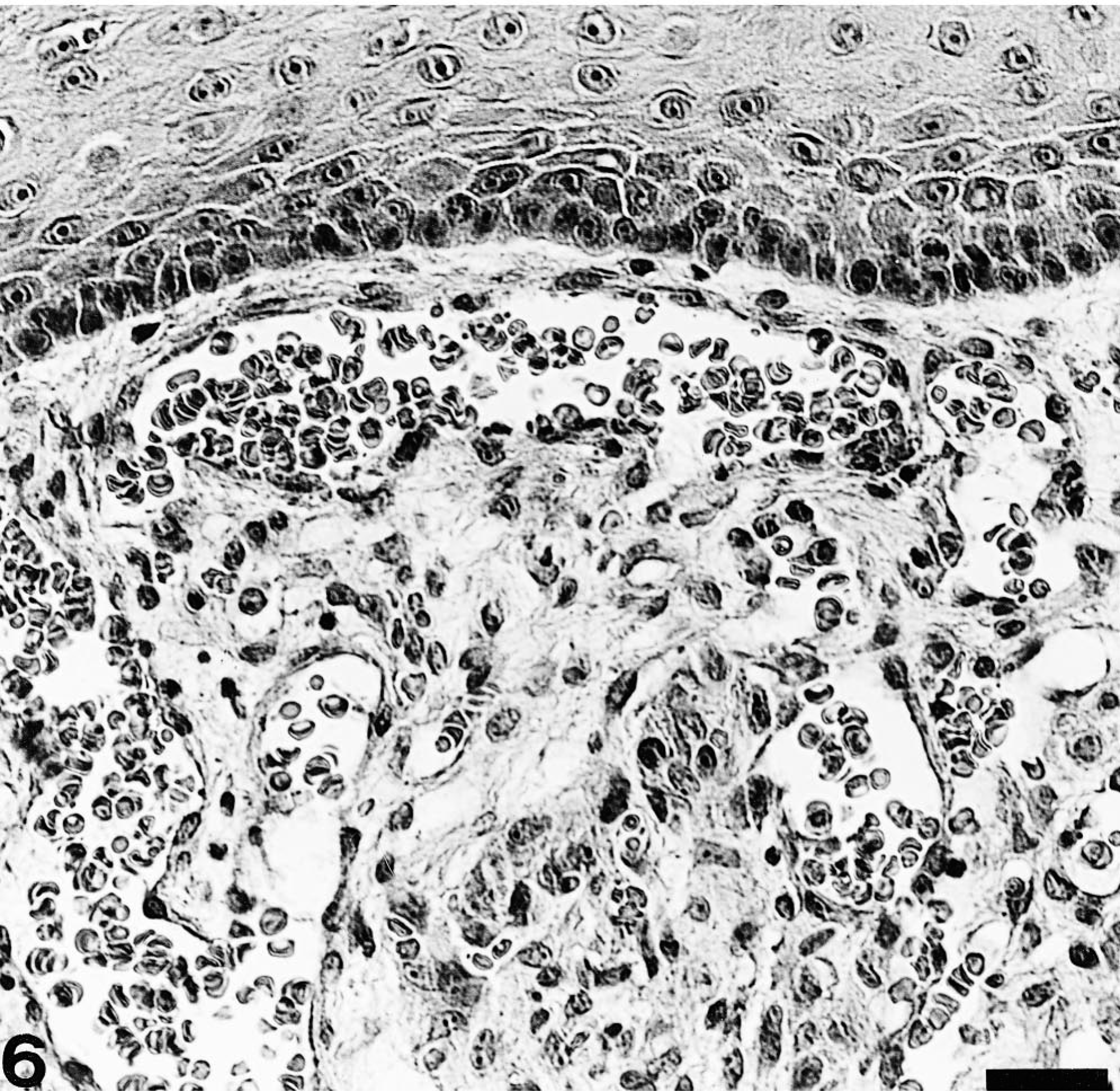
Mucosa of the oral cavity; pup No. 1. Moderate neovascularization of the submucosa. HE. Bar = 26 µm
In formalin-fixed, Epon-embedded samples, keratinocytes displayed cytoplasmic inclusion-like bodies typical of poxvirus (Figs. 7, 8). Occasionally, virions were admixed with electron-dense material. The fine structure of the virions consisted of an oval- to dumbbell-shaped core surrounded by a membrane, lateral bodies, and a surface membrane. Particles had a length of approximately 250 nm and a width of nearly 150 nm. Occasionally, cross sections were detected and measured about 150 nm. In addition, nuclear swelling and chromatolysis of keratinocytes were observed. Because of poor preservation of formalin-fixed samples, further investigation of the ultrastructural characteristics of the affected mucosa was not possible. Transmission microscopy of negative-stained suspensions prepared from mucosal lesions of seal No. 3 led to the detection of a small number of cocoon-shaped viral particles with convex ends consistent with parapoxvirus morphology. The particles had a length of approximately 240 nm and a width of nearly 140 nm, with regularly arranged surface tubules.
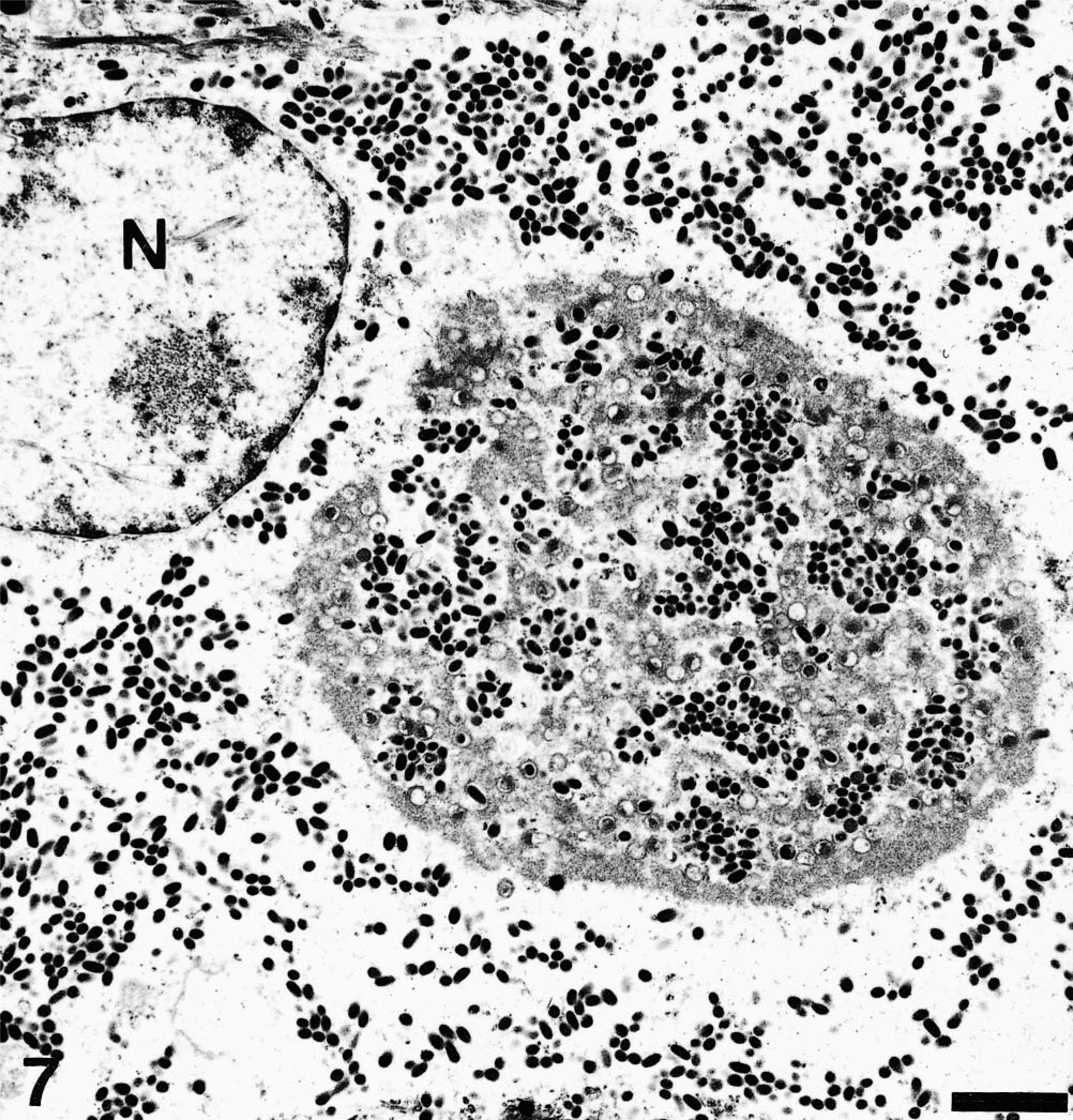
Electron micrograph. Tongue; pup No. 3. Keratinocyte with cytoplasmic parapoxvirus particles. N = nucleus. Uranyl acetate and lead citrate counterstain. Bar = 1.13 µm.
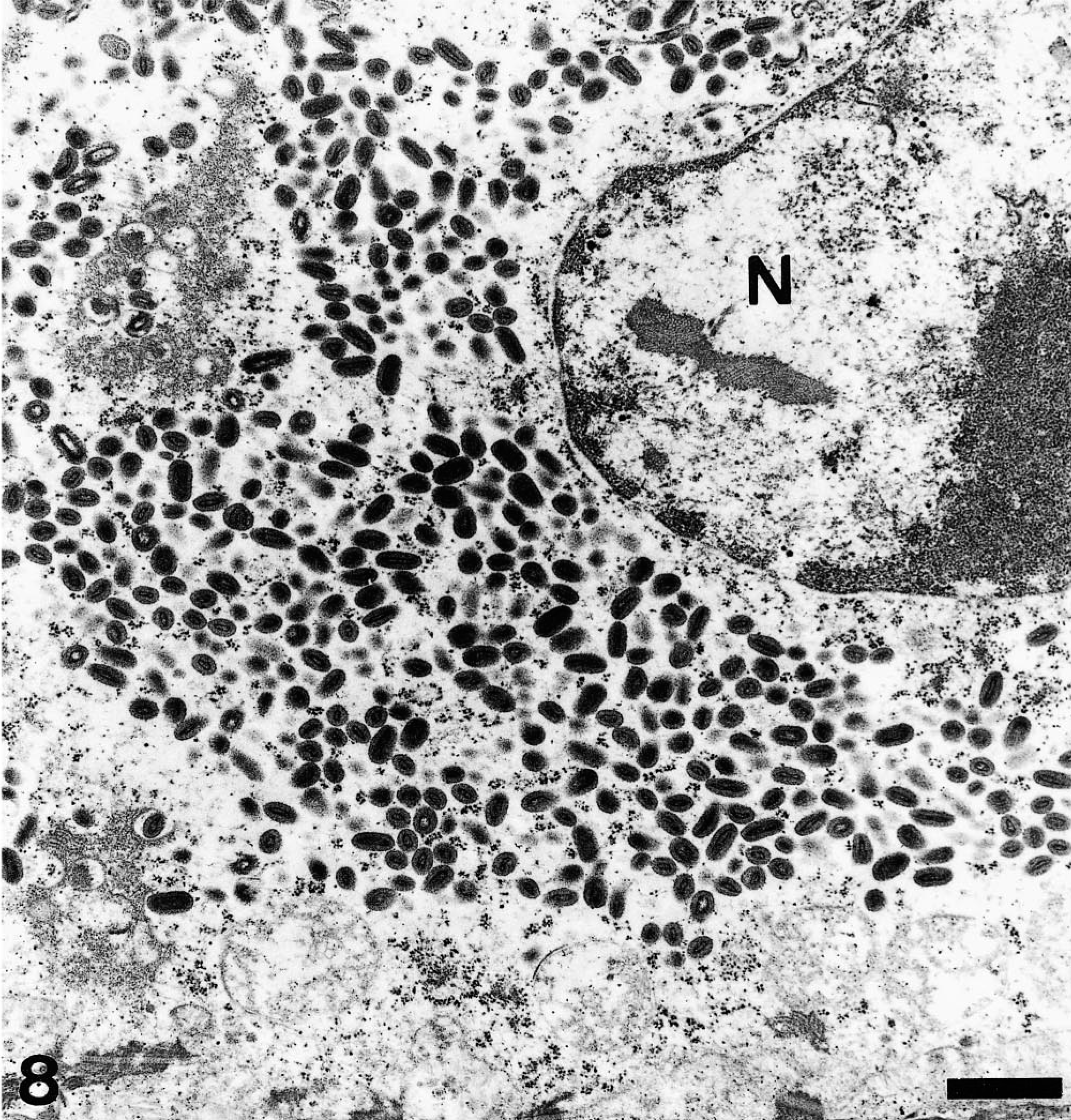
Electron micrograph. Tongue; pup No. 3. Keratinocyte with cytoplasmic parapoxvirus particles. N = nucleus. Uranyl acetate and lead citrate counterstain. Bar = 0.55 µm
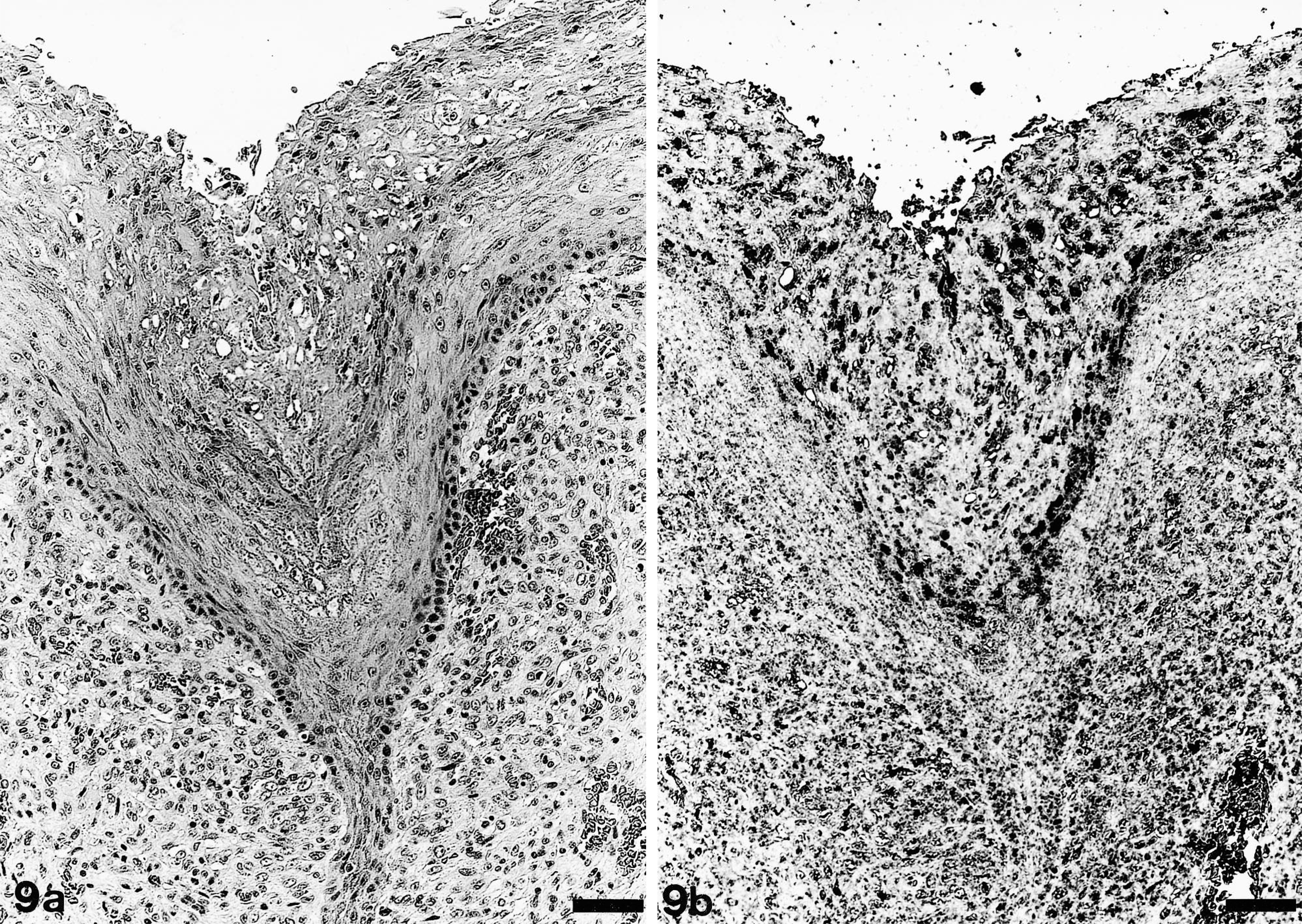
In situ hybridization demonstrated abundant parapoxvirus DNA–positive epithelial cells, especially in the stratum granulosum and the outer parts of the spiny layer corresponding to the cells with ballooning degeneration (Fig. 9a, b). In contrast, basal cells and deeper layers of the stratum spinosum lacked a positive signal. No parapoxvirus DNA–positive cells were detected in the mucosa adjacent to the proliferative epithelial lesion. Furthermore, there was no positive signal for morbillivirus RNA in the mucous mem-branes of the tongue from pup No. 3. In the skin from an unaffected seal and a rabbit with myxomatosis, no parapoxvirus DNA–positive signal was detected. In the skin from a sheep with ovine contagious pustular dermatitis, parapoxvirus DNA–positive signals were observed in keratinocytes.
Tongue; pup No. 1. Affected part of the mucous membrane processed with HE (
Bacteriology, immunohistochemistry, and serology
Bacteriology of seal No. 3 revealed no growth of specific pathogenic bacteria in liver, spleen, kidney, lung, esophagus, stomach, and intestine. Lung tissue specimens of seal No. 3 were negative for morbillivirus antigen by immunohistology. All 25 seals were seronegative for canine distemper virus–specific titers (<10) at the beginning and end of the rehabilitation period.
Discussion
Ulcerative to proliferative, nodular, cutaneous, and mucosal lesions caused by an endemic outbreak of parapoxvirus infection in a rehabilitation center were diagnosed in 26 captive seal pups from the German North Sea. Histologically, the lesions were characterized by epidermal and mucosal ballooning degeneration, with eosinophilic, cytoplasmic inclusion bodies and a dense dermal and submucosal lymphohistiocytic perivascular to interstitial infiltrate, fibroblast proliferation, and neovascularization. The morbidity in seal pups was 100%, and disease-related mortality was 0%. The clinical signs and the exophytic growth of the dermal changes resembled those observed in common seals from North America and other pinniped species. 12 , 29 , 36–39 However, the long clinical course differed from the duration of the disease reported in harbor seals from North America. Regression of nodules was reported to start after the fourth week of infection in harbor seals between 4 and 15 months of age. Most nodules were resolved within 15 weeks, and a few persisted for as long as 18 weeks. 37 The prolonged clinical course in the harbor seals investigated, 16–25 weeks and up to 10 months in select cases, might be due to the age or immune status of the seal pups at the time of infection. Presumably, the immune system is less competent than in older animals. First clinical signs in young seals from the rehabilitation center were observed 24 days after contact with an infected seal pup in captivity. Based on the sequence of events, an incubation period of 3 weeks can be assumed. Similarly, an incubation period of 3–5 weeks has been reported in captive harbor seals from North America. 37 Pup No. 1 expressed oral lesions first, indicating that this animal was likely infected in the wild or during suckling or nursing. The outbreak of the disease may be due to immunocompromising circumstances in combination with an insufficient protection by maternal antibodies against infection with parapoxvirus, as has been reported in lambs. 17 The frequent clinical presentation of the first pox lesions in the oral cavity of the initial seal pups of the rehabilitation center may be due to gavage with possibly contaminated flexible tubes. Later, when seals started to eat by themselves, contact with contaminated surfaces of the pools may have facilitated cutaneous viral transmission between animals, leading to the development of dermal nodules at different sites. Additionally, in California sea lions, social behavior such as head and neck rubbing has been discussed as a possible cause of transmission. 2
Histopathology of the lesions in affected pups was characterized by marked orthokeratotic hyperkeratosis, acanthosis, epidermal and mucosal hyperplasia, and degenerated ballooning cells of the stratum granulosum with cytoplasmic eosinophilic inclusion bodies. In addition, exuberant histiocytic dermal and submucosal infiltration, fibroblastic proliferation, and neovascularization were observed. These findings paralleled those of pox lesions reported in harbor seals from North America and other pinniped species. 12 , 29 , 36 , 37 A different histologic appearance has been reported from South American sea lions 38 and northern fur seals. 10 In these species, downward growth of epithelial cells and dermal epithelioma-like lesions have been described. Whether the alterations in these pinnipeds are due to an infection with a different species of poxvirus remains to be determined. In the epithelium, poxvirus replication is considered to induce degenerative and proliferative changes. 15 Hyperplastic changes resembling a tumor-like growth have been reported for different poxviruses, including fowlpox virus, shope fibroma virus, molluscum contagiosum virus, and Yaba virus. 16 , 19 In the latter, the lesions consisted of a dense histiocytic infiltrate resembling cutaneous histiocytomas. 16 Similarly, in the present study, a focally extensive histiocytic dermal and submucosal infiltration was observed. Early poxvirus genes seem to encode epidermal growth factor–like products or counteract host defences and may play an important role in the pathogenesis of viral lesions. 22 Different viral-defense molecules were detected in ortho-, lepori-, and molluscipoxviruses and consisted of virokines, viroceptors, and intracellular proteins. The viral proteins comprise inhibitors of type I or II interferons and interferon transduction pathways, and complement regulatory proteins, interleukin (IL)-18–binding proteins, tumor necrosis factor and IL-1 receptor homologues, serine protease inhibitors and inhibitors of cytokine processing, and chemokine and apoptose inhibitors. 19 Ovine parapoxviruses contain a vascular endothelial growth factor homologue, which dysregulates the cell cycle by inducing mitogenesis and upregulation of the cellular metabolism for virus replication. 28 Additionally, the secretion of several immune-modulating proteins related to ovine IL-10 and inhibitors of granulocyte-macrophage colony-stimulating factor, IL-2, and interferon-induced kinase have been reported in orf virus infection. 5 Moreover, proliferative nodule formation in sheep is assumed to be the consequence of the massive accumulation of major histocompatibility complex Class II–positive dendritic cells. 11 In the seals of this study, the dermal and submucosal proliferation seemed to be a consequence of histocytic infiltration and fibroblastic proliferation. Neovascularization may be due to a virus-derived growth factor homologous to orf virus infection.
The ISH findings correspond to experimental studies of ovine parapoxvirus infection, which show that in abraded sheep skin the newly proliferating keratinocytes in the outer stratum spinosum are the target-cell population for parapoxvirus infection. 17 Parapoxvirus is known as the causative agent for contagious pustular dermatitis in sheep and occurs in bovine and wildlife animals. 22 Although transmission of seal parapoxvirus to humans may occur, 12 there was no evidence of a high zoonotic potential for man in the present study, and none of the caretakers developed parapox-like lesions.
By electron microscopy, lesions seemed to be due to a solitary parapoxvirus infection. There was no evidence of other viruses like orthopox- and caliciviruses, as has been reported in gray seals. 25 , 30 These findings support previous studies that demonstrated that seal poxvirus belongs to the genera parapoxvirus. 39 Sealpox virus is considered a tentative member of the parapoxvirus genus. However, first comparative sequence analysis between the different established parapoxvirus species and the parapoxvirus isolated from the animals described in this study revealed that sealpox virus differed significantly from orf virus, bovine papular stomatitis virus, pseudocowpoxvirus, and parapoxvirus of red deer in New Zealand. 1 Accordingly, it can be concluded that the seal pups of this study were not infected with a known ovine parapoxvirus. Parapoxvirus infection in seals seemed to be due to an infection through a solitary pathogenic agent likely of marine origin. Subclinically infected seals probably served as a viral reservoir. Infections with poxviruses also have been described in different captive and free-ranging species of odontocetes 2 , 9 , 33, but the unclassified viruses seem to belong to the genus orthopoxvirus. 33 However, the relationship between sealpox and poxvirus infection in cetaceans requires further investigation.
In the past, several surveys demonstrated a widespread occurrence of nodular lesions due to poxvirus infection in captive and wild pinnipeds from North and South America and in seals in the UK. 30 , 34 , 35 Using serologic methods, parapoxvirus-specific antibodies were detected in sera of harbor and gray seals from the UK and the Netherlands. 24 Gray seals from the coast of Cornwall 29 and northwest Europe 25 that were infected with parapoxvirus may act as carriers of the infection from these areas to the northern part of the German Wadden Sea. The disease outbreak seemed to be associated with immunocompromising circumstances including capture and lack of protective maternal antibodies. In the present cases, there was no evidence of immunosuppressive conditions such as morbillivirus infection. More importantly, the lack of affected adult wild animals in the German North Sea suggested that this population is protected against parapoxvirus infection possibly because of a previous subclinical infection or a more intact, well-developed immune system.
