Abstract
To characterize the immune response associated with Lawsonia intracellularis infection, twenty-eight, 7-week-old pigs were dosed orally with a pure culture of L. intracellularis. Animals were killed 3, 7, 14, 21, 28, 35, and 42 days postinfection. Light microscopic studies were undertaken to immunophenotype the immunologic response using specific antibodies to T-cell subsets (CD3, CD4, and CD8), B cells, major histocompatibility complex class II, cadherin, and macrophages over the course of time. The results indicate that there is a direct association between the presence of L. intracellularis and reduced T-cell and B-cell numbers. For the first time, this provides evidence of the presence of an immunosuppressive mechanism operating in this disease. Furthermore, macrophage marker studies indicated that macrophages may play a more complex and significant role in the disease process than has been previously reported, with activated macrophages accumulating in infected hyperplastic crypts.
Proliferative enteropathy (PE) is an economically important transmissible enteric disease and is a major cause of weight loss and mortality in pigs. The disease has two major clinicopathologic manifestations, porcine intestinal adenomatosis (PIA) and proliferative hemorrhagic enteropathy (PHE), and is associated with infection by the obligate intracellular bacterium Lawsonia intracellularis. 9 , 18
Animals suffering from the primary uncomplicated condition PIA (most often seen in postweaned pigs aged between 6 and 14 weeks) usually exhibit anorexia and wasting. 17 , 19 Macroscopic lesions are restricted to the alimentary tract, and PIA in the classical form is characterized by thickening of the wall of the terminal ileum. In some cases, however, lesions may extend from the mid small intestine into the caecum and colon. These changes may be diffuse, involving the whole mucosa, with discrete nodules or polyps also being present, particularly in the caecum and colon. Marginal cases may be difficult to observe grossly, with confirmation requiring histologic analysis. Microscopically, the normal villus and intestinal crypt structures are replaced by proliferating immature epithelial cells, resulting in the marked thickening that is observed. The villus architecture may be lost, with the glands often becoming elongated and branched. Affected glands are lined with immature epithelial cells, and there is a marked reduction or loss in goblet cells. The epithelial cells remain immature with frequent mitotic figures and a reported lack of associated inflammatory cell infiltration. 3 Immunohistochemistry (IHC) using specific monoclonal antibodies for L. intracellularis demonstrates their presence, free in the apical cytoplasm of the immature crypt epithelium.
In the more acute hemorrhagic form, PHE, the first sign of infection may be the onset of diarrhea with the rapid death of the animal. At postmortem, the mucosa of the terminal ileum is thickened and ridged, with the lumen filled with blood. The epithelium may be covered by a fibrinous membrane under which fresh blood may be trapped. Microscopically, PHE is similar to PIA, with the presence of proliferating immature epithelial cells and L. intracellularis found free in the apical cytoplasm. However, bacteria also are found in macrophages within the lamina propria and submucosa and within epithelial capillaries and lymphatics. 7 An acute inflammatory response is evident in contrast to PIA, and many have credited the differences between the associated low mortality rate of PIA and the more lethal form PHE to this immune response. 7
In the uncomplicated form of PIA, clearance of the bacteria is associated with resolution of the lesion as hyperplasia is reduced. 13 Normal cells regenerate from the base of the crypts and multiply to reform a healthy crypt villus structure. Regeneration also is characterized by degeneration of epithelial cells, apoptosis of macrophages, and reappearance of goblet cells. 13 Information on the inflammatory response in pigs after infection with L. intracellularis is restricted to a handful of publications. 6 , 8 , 12 , 19 There are no recent investigations in this area, and much remains to be ascertained regarding the general immunologic events after infection. To date, there has been no systematic chronological examination in this regard. The available reports suggest that the inflammatory response in PIA is remarkably mild with limited infiltration of CD8+ve and CD25+ve T lymphocytes. 12 A consistent finding in all manifestations of PE is the accumulation of immunoglobulin A (IgA) within enterocytes. 6 , 12 This may be IgA that is directed against the L. intracellularis or may be due to the inability of the immature enterocytes to discharge IgA. 6 Macrophages also are reported to be present in early stages of lesion development; 8 however, their role in controlling infection remains to be defined.
In hemorrhagic lesions, the cellular response is greater but is still considered as moderate with infiltrations of CD8+ve and CD25+ve T lymphocytes, marked cell lysis, and loss of cells into the lumen. 12 Degenerating cells may be responsible for releasing antigens that trigger immediate or delayed hypersensitivity responses similar to those observed in acute bacterial infections and also in type-1 hypersensitivity reactions. Whether this altered immune response is the mechanism instrumental in changes to the vascular supply through increased permeability of blood vessels, and thus restricted blood flow to the mucosa, or whether differences in bacterial strains are responsible is still open to debate.
Here we present the data of detailed characterization of the immune response in pigs infected with L. intracellularis over a time course of 42 days. Specifically, we have investigated T- and B-cell marker expression (CD3, CD4, CD8, and CD79a) and expression of the activated macrophage marker (MAC387), and major histocompatibility complex class II (MHC-II) in association with the characteristic lesions induced in this disease.
Materials and Methods
Animals and infection procedure
Twenty-eight 7-week-old piglets that had been fed on nonmedicated feed containing no growth promoters were obtained from a minimal-disease herd. Before inoculation, fecal specimens from all animals were assessed microbiologically for Brachyspira hyodysenteriae, B. pilosicoli, Yersinia spp. and Salmonella spp. Assessment for L. intracellularis was carried out both by polymerase chain reaction (PCR) of fecal material and by serologic assay on serum extracted from whole blood samples collected before inoculation. 2 All animals were found to be negative (i.e., no organisms cultured, no PCR signal, and no detectable antibody titer) by these techniques for the above pathogens. The animals were randomly selected and penned in seven groups of four each. Animals were inoculated with 5 ml L. intracellularis suspension via orogastric tube, each animal receiving approximately 2 × 108 viable bacteria. Animal weights were recorded on day 0 and again at euthanasia. Four animals were euthanatized at each of the following time points: 3, 7, 14, 21, 28, 35, and 42 days postinfection. A separate group of seven age-matched (7–14 weeks) pigs was used to provide reference data for the immunologic study. Although these reference animals were not euthanatized at time points equivalent to the experimentally infected pigs, they were selected to cover the age range of pigs used in the study.
Preparation of inoculum
Inoculum was prepared as described previously. 5 Briefly, L. intracellularis was cultured in IEC-18 cells for 6 days, then infected monolayers were washed in KCl and lysed in sucrose potassium glutamate (SPG) buffer by scraping and passage through a 19-gauge needle. Suspensions were then centrifuged (100 × g) to remove intact nuclei. An aliquot was removed to estimate bacterial numbers, 5 and the remainder was frozen at −70 C until required for animal challenges. Storage of bacteria at −70 C for over a year results in no significant loss of viability as assessed by LIVE or DEAD stain (Molecular Probes). Bacteria were originally isolated from homogenized intestinal tissue and grown in cell culture in IEC-18 intestinal epithelial cells to a final passage of 11 (isolate LR 189/5/83). Cells were then lysed, and bacterial suspensions were prepared in SPG buffer with 5% fetal calf serum (FCS). 5 To enumerate L. intracellularis, infected cell lysate was serially diluted for infection of IEC-18 cells on 13-mm coverslips (carried out in triplicate). Infectious units ([IU]; equating to viable bacteria in this suspension) were estimated, and detection of L. intracellularis in infected monolayers was carried out as described in Lawson et al. (1993). 5 Purity of bacterial suspensions (including freedom from mycoplasmas and Chlamydia) was confirmed as described previously. 5 Aliquots of suspension containing approximately 2.2 × 108 L. intracellularis infectious units per ml (IU/ml) were stored at −70 C and thawed immediately before inoculation of animals. These were then diluted 1 : 10 to provide 2.2 × 107/ml, and each animal received 10 ml inoculum i.e., approximately 2 × 108 IU/ml.
Postmortem sample collection and processing
After euthanasia, postmortem examination of each experimental animal was carried out, and an assessment was made of any gross pathologic change. Intestinal tissue samples (duodenum, upper small intestine, mid small intestine, terminal small intestine [defined for the purposes of this article as 2–3 cm of ileum immediately proximal to the ileocaecocolic junction i.e., terminal ileum], caecum, proximal colon, and distal colon) were removed, and samples from each area were both frozen and fixed in formalin (see below).
Tissue samples were placed in 10% phosphate-buffered formalin for 5 hours. Each tissue sample was further dissected to provide a total of three tissue blocks per sample site before placing them into labeled plastic cassettes and further fixation overnight. Tissue samples were processed for 6 hours using a VIP 2000 vacuum processor (Bayer Diagnostics), dehydrated through graded ethanol, cleared in xylene, and impregnated with Histoplast paraffin wax (Thermo Shandon). Tissue was embedded in fresh Histoplast paraffin wax before cutting.
For generation of frozen sections, fresh tissue samples were frozen at −95 C using a mixture of isopentane (Merck Eurolab) and dry ice. The temperature of the mixture was checked before tissue freezing. Tissue samples were attached to 13-mm-diameter cork disks using OCT compound (Bayer Diagnostics), coated in OCT to prevent desiccation upon storage, then sealed, and stored at −80 C.
Histopathology and immunohistochemistry
Tissue samples were cut at 4 µm and stained with hematoxylin and eosin (HE). Sections were placed onto either standard double-frosted microscope slides for HE staining or onto Biobond-coated microscope slides (British Biocell Ltd.) for IHC studies. Sections were allowed to dry overnight at 37 C before IHC staining.
Fixed and processed tissue sections were dewaxed and rehydrated. Endogenous peroxidase activity was blocked using 1% hydrogen peroxide in methanol for 20 minutes. Nonspecific protein blocking was carried out using a Vectastain Elite Universal ABC kit (Vector Laboratories) as per manufacturer's instructions. The primary antibodies used and the antigen retrieval techniques that were required for formalin-fixed tissues are set out below. Visualization was performed using 3,3′-diaminobenzidine tetra-hydrochloride (DAB) (Vector Laboratories). Negative controls were incorporated for each procedure using monoclonal or polyclonal negative control serum (Dako Ltd.).
For frozen tissue, sections were cut at 6 µm using a 620 M cryostat (Thermo Shandon) and placed onto Biobond-coated slides (British Biocell Ltd). Sections were allowed to air-dry overnight at room temperature. Fixation was carried out using acetone for 10 minutes at room temperature after air-drying for 15 minutes. Air-dried sections were placed into fresh acetone for a further 10 minutes after a final air-drying stage for 15 minutes. Sections were rinsed in 0.1-M phosphate-buffered saline (PBS) (pH 7.4) and placed into Sequenza coverplates (Thermo Shandon) for immunostaining. Nonspecific endogenous staining was blocked using endogenous blocking reagent (Dako Ltd.) for 20 minutes at room temperature. Nonspecific blocking was carried out using a variety of reagents depending on the secondary reagent used (see individual protocols listed below for specific details). Visualization was performed using DAB.
Antibodies
Antibodies specific for L. intracellularis, CD3, CD79, macrophages, MHC-II, and Pan-cadherin were applied to formalin-fixed, paraffin-embedded sections (experimental animals and reference tissue samples). Antibodies for CD8 and CD4 were used on frozen sections (experimental animals only). Details of the protocols used for each antibody are given below. Isotype-specific irrelevant antibodies were used as negative controls in all experiments.
L. intracellularis (VPM53)
Mouse anti–L. intracellularis (Department of Veterinary Pathology, Royal (Dick) School for Veterinary Studies) saturated supernatant diluted 1 : 200 in 0.1 M PBS (pH 7.4) at 37 C for 1 hour after trypsinization of sections using 0.1% trypsin solution (ICN Biochemicals) with 0.1% calcium chloride adjusted to pH 7.8 with 0.1 M NaOH at 37 C for 30 minutes.
CD3
Rabbit anti-CD3 concentrate (Dako Ltd., AO452). Polyclonal antibody diluted 1 : 500 in 0.1 M PBS (pH 7.4) for 1 hour at 37 C after antigen retrieval using 0.01 M citrate buffer (pH 6.0) within a plastic pressure cooker and using a microwave oven as the heat source (800 W) for 4 minutes at full pressure.
CD79
Mouse anti-CD79 (Dako Ltd., M7050). Monoclonal antibodies diluted 1 : 25 in 0.1 M PBS (pH 7.4) for 1 hour at 37 C after antigen retrieval using 0.01 M citrate buffer (pH 6.0) within a plastic pressure cooker and using a microwave oven as the heat source (800 W for 4 minutes at full pressure).
Macrophages
Mouse anti–myloid-histiocyte MAC387 (Dako Ltd., N1570) prediluted 60 minutes at room temperature after trypsinization using 0.1% trypsin solution (ICN Biochemicals) with 0.1% calcium chloride adjusted to pH 7.8 with 0.1 M NaOH at 37 C for 20 minutes.
Major histocompatibility complex class II
Rat anti–MHC-II (Department of Veterinary Pathology, Royal [Dick] School of Veterinary Studies) undiluted saturated supernatant for 1 hour at room temperature. Nonspecific staining was blocked using prediluted goat serum (Vector Laboratories). Secondary labeling was carried out using goat anti-rat IgG horseradish peroxidase (HRP) (Serotec) diluted 1 : 25 in 0.1 M PBS for 1 hour at room temperature.
Cadherin
Mouse anti-pan cadherin (Sigma Chemical Company) saturated supernatant diluted 1 : 200 in 0.1 M PBS pH 7.4 for 1 hour at 37 C after protease digestion of tissue using 0.1% protease type 1 (Sigma Chemical Company) in Tris-buffered saline (pH 7.8) at 37 C for 40 minutes. Given the recognized expression patterns of the cadherin family, this equates to identification of e-cadherin in this anatomical location.
CD8
Mouse anti-pig CD8 (courtesy of Karen Haverson, University of Bristol) undiluted saturated supernatant, for 1 hour at room temperature. Nonspecific staining was blocked using 2.5% normal horse serum (Vector Laboratories) for 20 minutes at room temperature. Secondary labeling using horse anti-mouse IgG conjugated to HRP (Vector Laboratories) was diluted 1 : 200 in 0.1 M PBS for 30 minutes at room temperature.
CD4
Mouse anti-CD4 (courtesy of Karen Haverson, University of Bristol) undiluted saturated supernatant for 1 hour at room temperature. Blocking of nonspecific staining, secondary labeling, and visualization were carried out as per CD8 staining.
Assessment and quantitation of staining
IHC using the mouse monoclonal antibody VPM53 was carried out to locate L. intracellularis (as described previously). After the initial assessment of infection and lesion development, terminal small intestine (TSI) was selected for further detailed immunocytologic analysis because this site provided the most consistent and successful site of bacterial infection. This was as expected and in agreement with the findings of previous studies. 11 , 19 Ten fields from each section were counted at a magnification of 100×; both the number of glands demonstrating the presence of L. intracellularis and the total numbers of glands present were recorded. An average percentage was then calculated based on the total number of glands counted and the number of glands infected using the figures from each set of 10 fields counted. Counting was carried out using a Laborlux microscope (Leitz), JVC TK 1281 Video camera, and Image Pro Plus software.
Terminal ileum sample sites were selected and IHC staining for T-cell subsets, B cells, macrophages, and cadherin was carried out as described previously. To allow for differences in the distribution of T-cell populations within ileal villus structures, cells were counted as being either within lamina propria and detailed per square millimeter or associated with epithelial cells and detailed per 100 epithelial cells. Association with epithelial cells was accepted as being either within the epithelial layer or attached to the epithelial basement membrane. Areas of lamina propria were selected to enable measurement of all lamina proprial areas extending from the luminal surface to the muscularis mucosae or Peyers patch excluding islands of glandular tissue. Ten fields were counted per section at a magnification of 100× for areas of lamina propria and 20 fields per section (10 fields of villus surface epithelia and 10 fields of deep crypt gland epithelia) at a magnification of 400× for epithelial-associated cells.
B-cell and macrophage counts were taken per square millimeter ileum based on 10 fields per section at a magnification of 250×.
Statistics
The main aims of the statistical analysis were to investigate the nature and significance of the immune response in animals with classic lesions of PE. For this reason, the major analyses involved comparisons between challenged animals on day 14 (maximum lesions) with selected other time points. Before analysis, whether the parameters were normally distributed was checked. CD3+ve T cells, CD4+ve T cells, CD8+ve T cells, B cells per square millimeter of lamina propria, CD3+ve T cells, and CD8+ve T cells cells per 100 villi and deep crypt cells were normally distributed; however, macrophage numbers required to be log transformed to obtain normally distributed data.
The statistical test used depended on the data being tested. Because different pigs were used on different days, parameter values obtained on one day were independent of each other, and thus, standard analyses could be carried out. To test whether there was any significant difference on day 14 compared with days 3, 7, and 42 for CD3+ve T cells, CD4+ve T cells, CD8+ve T cells, B cells per square millimeter of lamina propria, CD3+ve T cells, and CD8+ve T cells per 100 villi and deep crypts, and log-transformed macrophages, standard two-sample t-tests were used. Two-sample t-tests also were used to test for the differences between CD3 cells per square millimeter of lamina propria, B cells per square millimeter of lamina propria, CD3+ve T cells per 100 villi in uninfected control animals, and those obtained on days 3, 7, and 42.
Additional comparisons were made with counts obtained from the reference material.
Results
Clinical and postmortem results
All experimentally infected pigs appeared clinically normal with the exception of two pigs, which contracted the hemorrhagic form of the disease and were euthanatized on days 13 and 14.
The weight of the animals was recorded on day 0 immediately before inoculation and again at necropsy. Weight loss in animals challenged with L. intracellularis was not a notable feature in this experiment. The pigs having detectable bacterial infections gained weight by 660 grams/day on average compared with an average of 760 grams/day gained in animals with no detectable bacteria. These results were not statistically significant (data not shown).
At postmortem, lesions were not obvious grossly in the majority of pigs, although evidence of intralumenal hemorrhage was apparent in two pigs on 14 days postinfection with mesenteric lymphadenopathy in one pig at this time point. Thickening of the terminal ileum was apparent in one pig on day-42 postinfection (Table 1.
Summary of major gross and histopathologic features for each experimental animal (pigs 1–28 [P1–P28]). DPI = days postinfection; MLN = mesenteric lymph node; NAD = no abnormalities detected; TSI = terminal small intestine. Unless otherwise stated, inflammation was mixed—macrophages, lymphocytes, neutrophils.
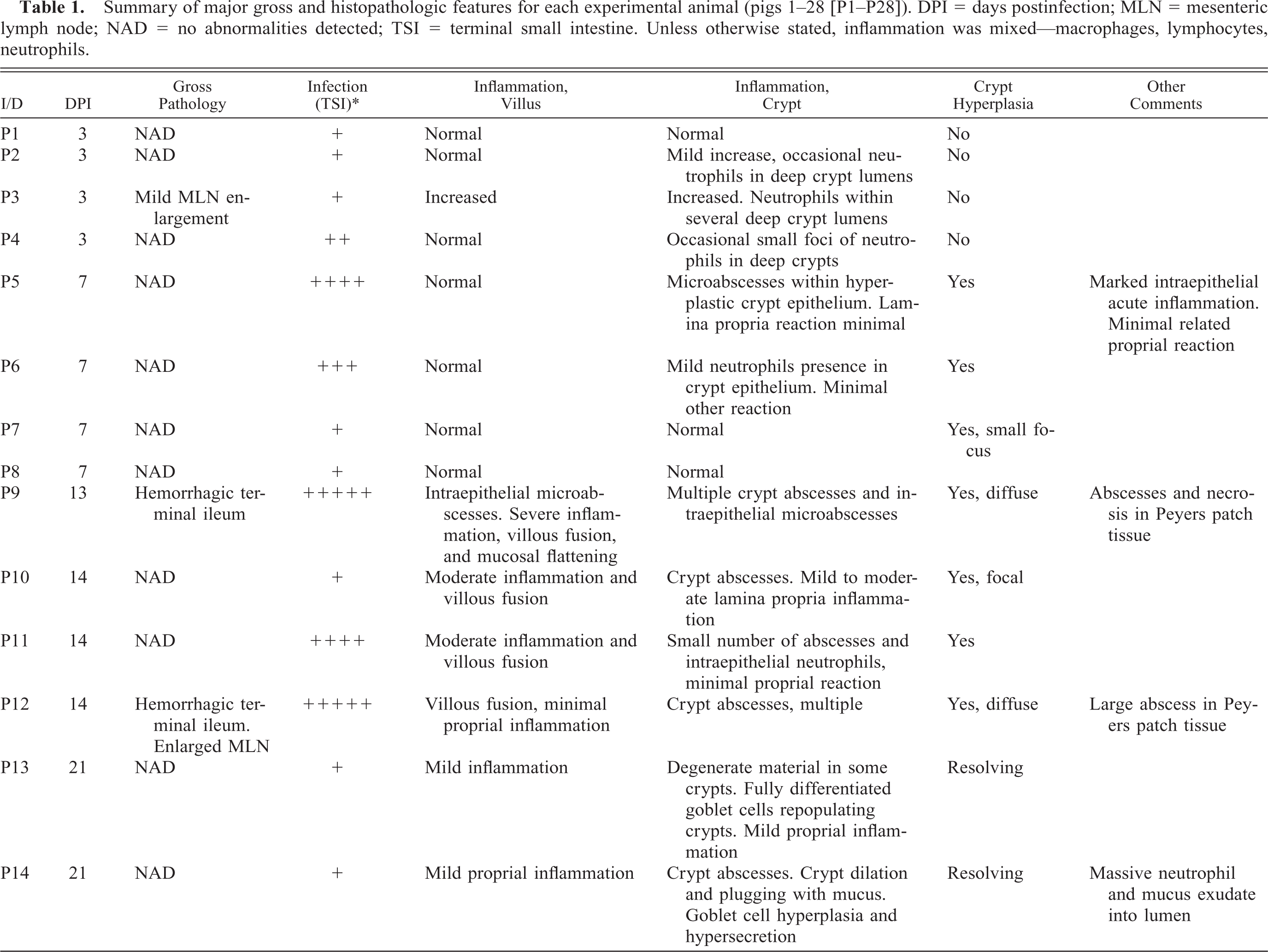
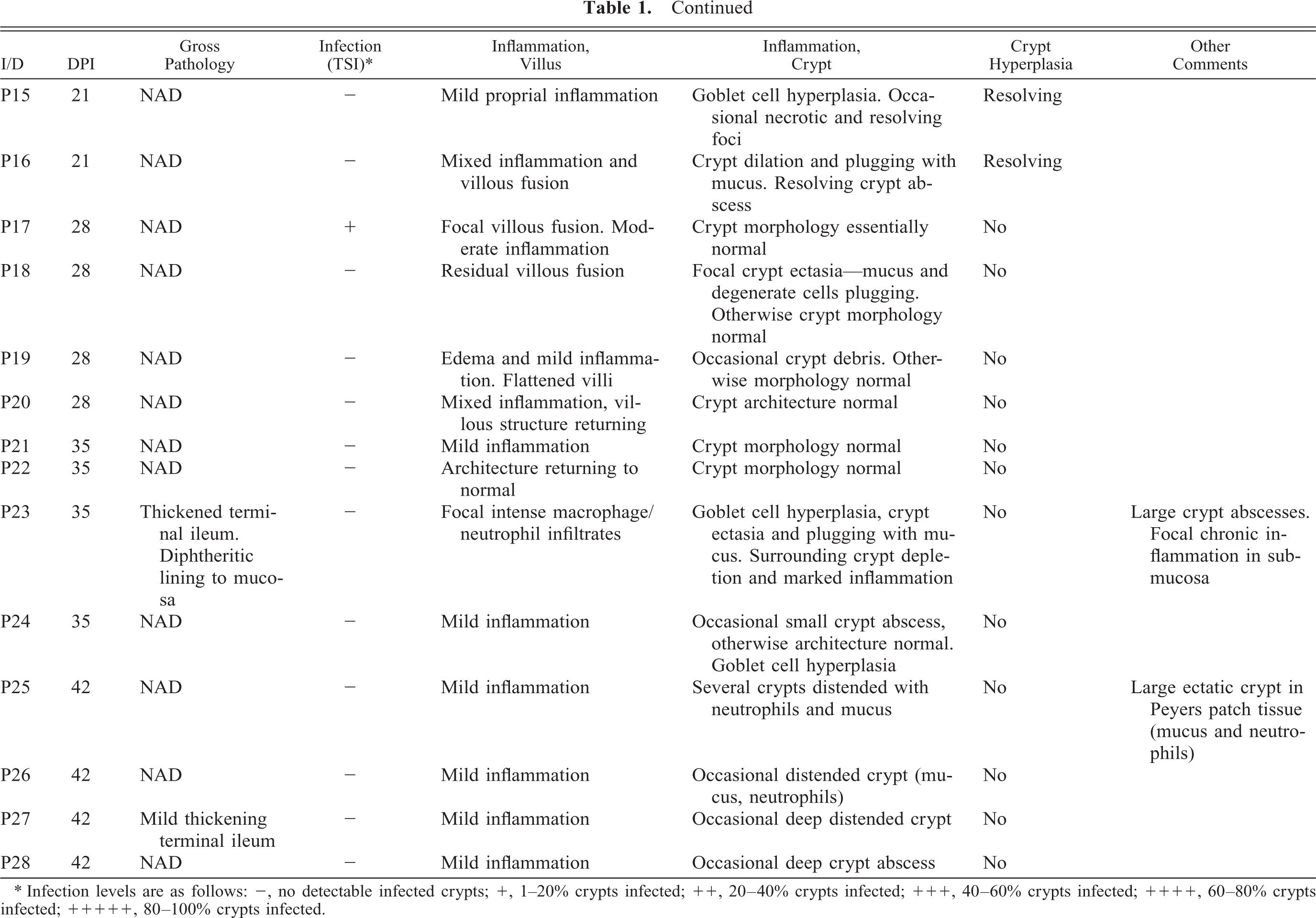
∗ Infection levels are as follows: -, no detectable infected crypts; +, 1–20% crypts infected; ++, 20–40% crypts infected; +++, 40–60% crypts infected; ++++, 60–80% crypts infected; +++++, 80–100% crypts infected.
Histopathology
Results of histopathology also are summarized in Table 1; pertinent findings are described in more detail below.
On 7 days postinfection in two out of four pigs, crypt epithelial hyperplasia was evident with associated neutrophil infiltrate between crypt epithelial cells. The inflammatory reaction was targeted to the intraepithelial compartment with minimal change in the lamina propria.
On day 14, gross changes were evident in two of the pigs associated with the development of extensive crypt hyperplasia, and crypt and intraepithelial abscess formation (Fig. 1). On day 21, the hyperplastic response was resolving with a return of goblet cells to deeper crypts. The trend through the remaining time points was of a return to normal crypt morphology passing through a phase of goblet cell hyperplasia and hypersecretion (Fig. 2).
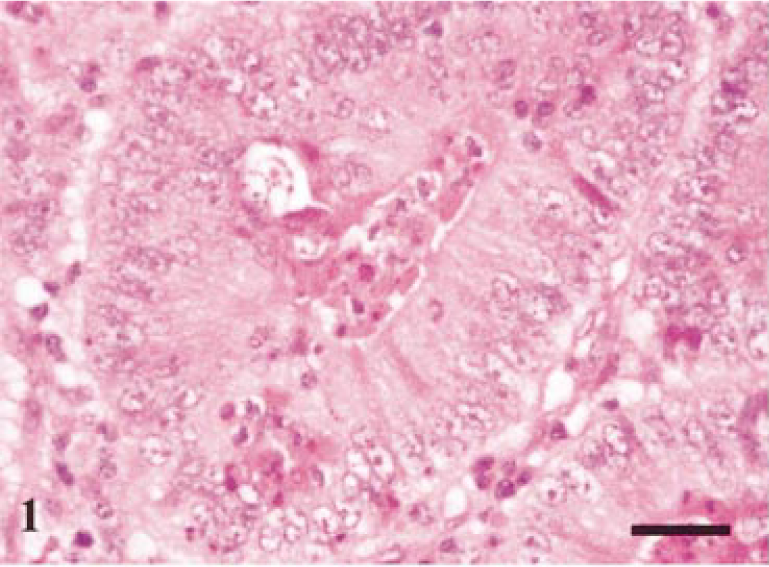
Terminal ileum. Typical histologic lesion seen on day-7 postinfection. Mild crypt hyperplasia and neutrophil infiltrate into crypt with minimal reaction in surrounding lamina propria. HE. Bar = 40 µm.
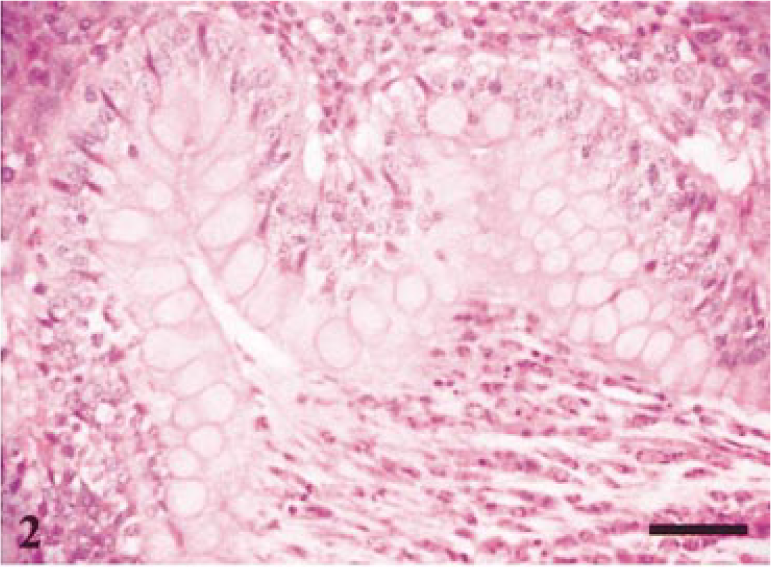
Terminal ileum. Typical histologic lesion seen on day-21 postinfection. Crypt lumen distended with mucus and debris. Hyperplasia of goblet cells. HE. Bar = 60 µm.
Immunohistochemistry (L. intracellularis)
The presence of L. intracellularis was confirmed by IHC. In addition to the ‘classic’ intraepithelial location of organisms, bacteria also could be observed free within macrophages and the lamina propria in ileum and proximal colon (Fig. 3). Discrete concentrations of L. intracellularis also were observed within aggregates at the basal area of the enterocyte appearing to protrude out into the lamina propria (Fig. 4). Infection was confirmed in 15 of the 28 inoculated pigs with the peak of infection occurring 14 days postinoculation (Table 1. A notable feature was that on 14 days postinfection there was evidence of extensive infection as demonstrated by both the number of infected crypts present and the number of sites infected. An illustration of the infection rate at each time point is incorporated in Fig. 5.
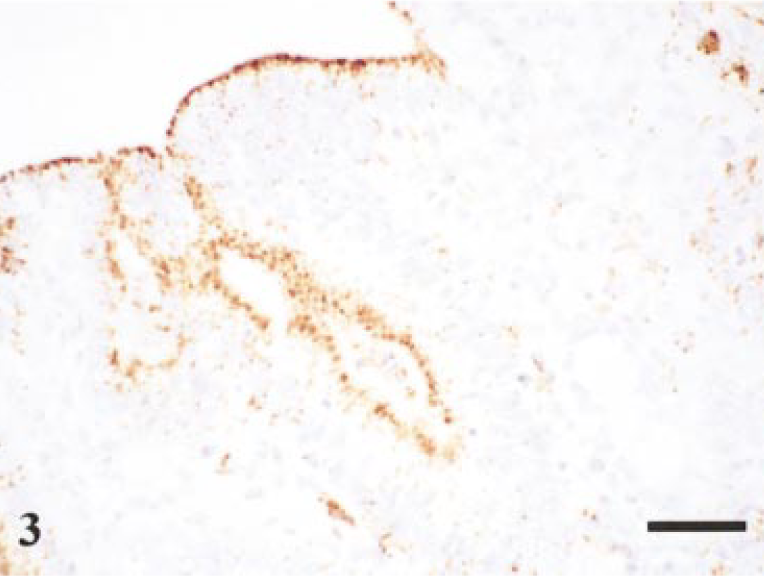
Terminal ileum. Staining of L. intracellularis organisms (mAb VPM53) within epithelial cells and within the lamina propria. Day-14 postinfection. Avidin–biotin–peroxidase complex method. Mayer's hematoxylin counterstain. Bar = 50 µm.
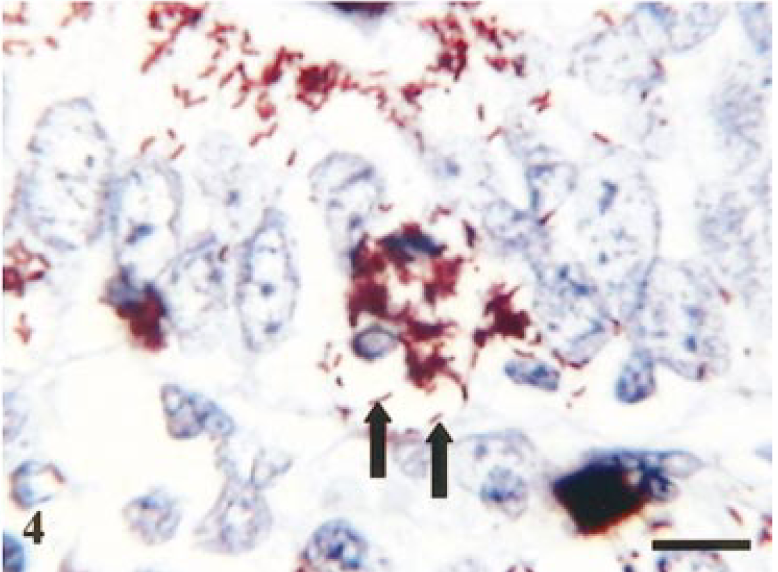
Terminal ileum. Staining of L. intracellularis organisms (mAb VPM53) within apical cytoplasm of epithelial cells and congregating at the basal cytoplasm (arrows). Day-14 postinfection. Avidin–biotin–peroxidase complex method. Mayer's hematoxylin counterstain. Bar = 15 µm
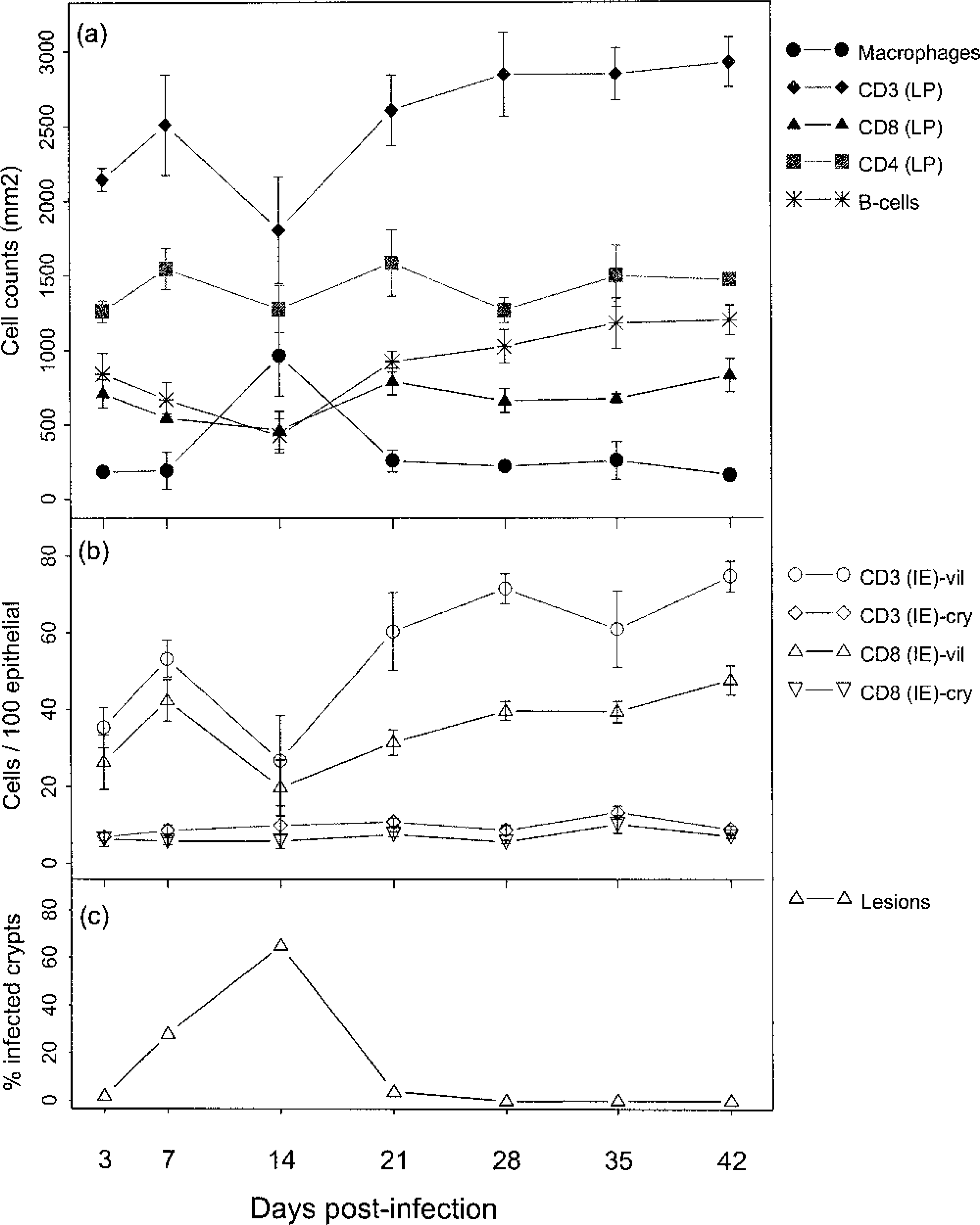
Summary of alterations in immune parameters over time in association with levels of infection in terminal small intestine. Fig. 5a. Cell counts are number of cells per 1,000 mm 2 lamina propria and represent the mean data for four pigs at each time point (the data from the pig dying on day 13 is included in the day-14 group) with standard error bars. LP = lamina propria. Fig. 5b. Cell counts are per 100 epithelial cells and represent the mean data for four pigs at each time point (the data from the pig dying on day 13 is included in the day-14 group) with standard error bars. IE = intraepithelial; vil = villous; cry = crypt. Fig. 5c. Infection rates are percentages of crypts infected (terminal ileum)
Immunohistochemistry (phenotyping of infiltrate)
Figure 5 summarizes the major alterations in immune populations over time, with corresponding levels of infection at each time point. The most obvious trends occurred on day 14 at which point lesions (Table 1) and percentage of infected crypts (Fig. 5c) also were maximal. There was a significant reduction in lamina propria CD3-positive cells on day 14, relative to resolved lesions on day 42 (P = 0.029, t = 2.85; Fig 5a), with mean levels of 1,802 and 2,924 respectively. In addition, there was a significant reduction in villus intraepithelial CD3-positive cells (P = 0.008, t = 3.89; Fig 5b), relative to resolved lesions on day 42. The reduction in CD3 cells also is illustrated histologically by comparing Fig. 6 (reference) and Fig. 7 (day-14 postinfection). Further analysis of this reduction in T-cell numbers using CD4 and CD8 markers (Fig 5a) revealed no significant difference in CD4 numbers (P = 0.269, t = 1.22). Similarly, no significant difference was found in CD8 cells in the lamina propria (P = 0.074, t = 2.164). However, analysis of the CD8 villus intraepithelial population revealed a significant difference when compared with resolved lesions on day 42 (P = 0.014, t = 3.42; Fig 5b). This change also is illustrated histologically in Fig. 8 (reference) and Fig. 9 (day-14 postinfection). B-cell numbers also were significantly reduced on day 14 when compared with resolved lesions on day 42 (P = 0.002, t = 5.119).
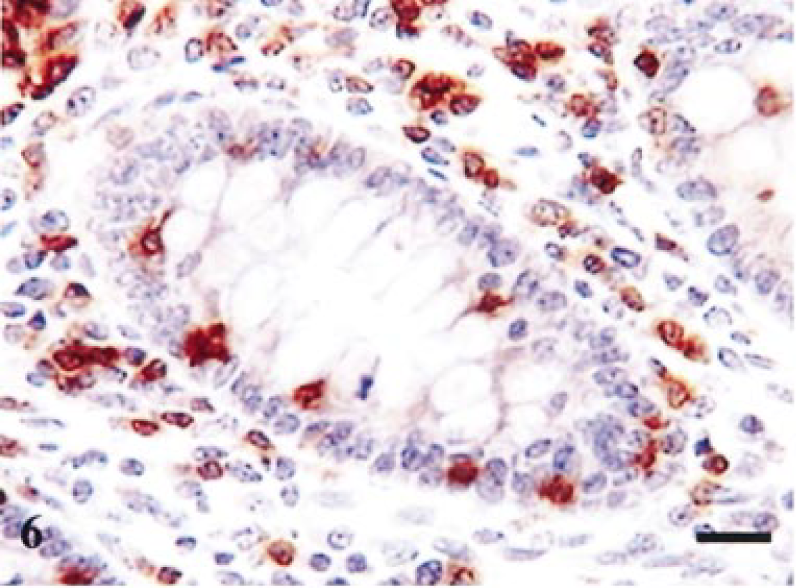
Terminal ileum, reference tissue. Staining for CD3 in crypt region. Positive cells in lamina propria and intraepithelial compartment. Avidin–biotin–peroxidase complex method. Mayer's hematoxylin counterstain. Bar = 40 µm.
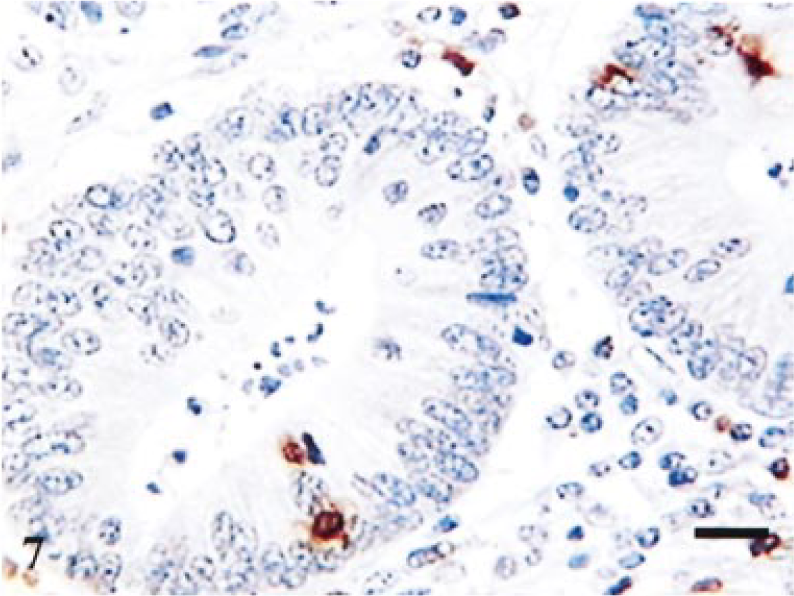
Terminal ileum; infected pig. Day-14 postinfection. Staining for CD3 in crypt region. Few positive cells in lamina propria and intraepithelial compartment. Avidin–biotin–peroxidase complex method. Mayer's hematoxylin counterstain. Bar = 40 µm.
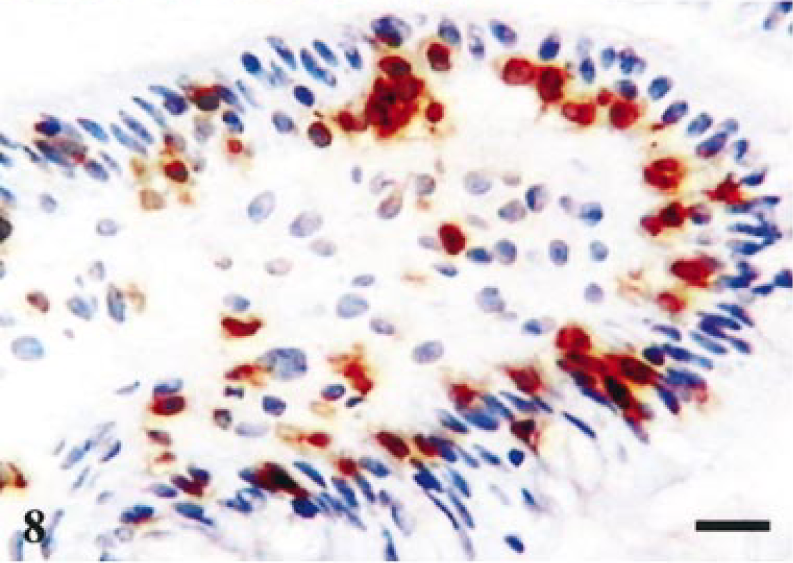
Terminal ileum, reference tissue. Staining for CD8 in villi. Positive cells in lamina propria and intraepithelial compartment. Avidin–biotin–peroxidase complex method. Mayer's hematoxylin counterstain. Bar = 40 µm.
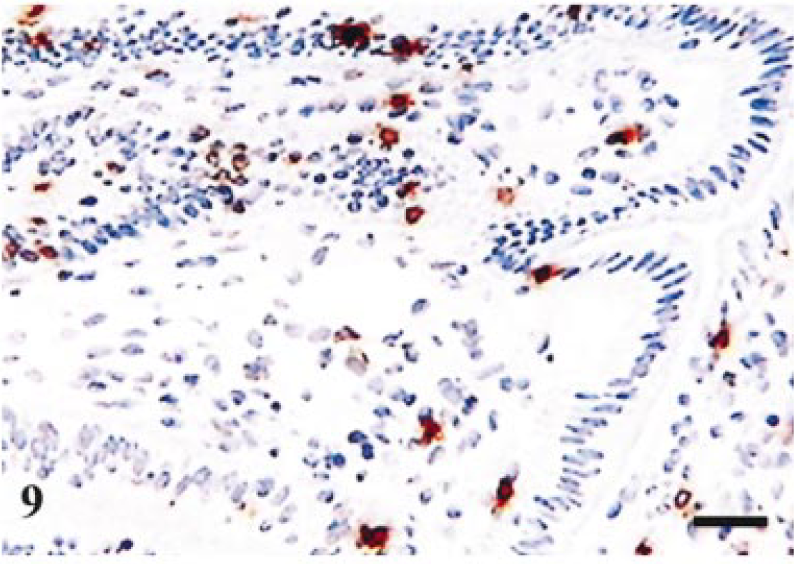
Terminal ileum; infected pig. Day-14 postinfection. Staining for CD8 in villi. Few positive cells in lamina propria and intraepithelial compartment. Avidin–biotin–peroxidase complex method. Mayer's hematoxylin counterstain. Bar = 80 µm.
Analysis of macrophage numbers revealed a significant difference between days 14 and 42 (P = 0.005, t = −4.318; Fig. 5a). Staining for activated macrophages in reference tissue revealed negligible staining. The typical changes seen are illustrated in Figs. 10, 11, where the macrophages can be observed in an intraepithelial location.
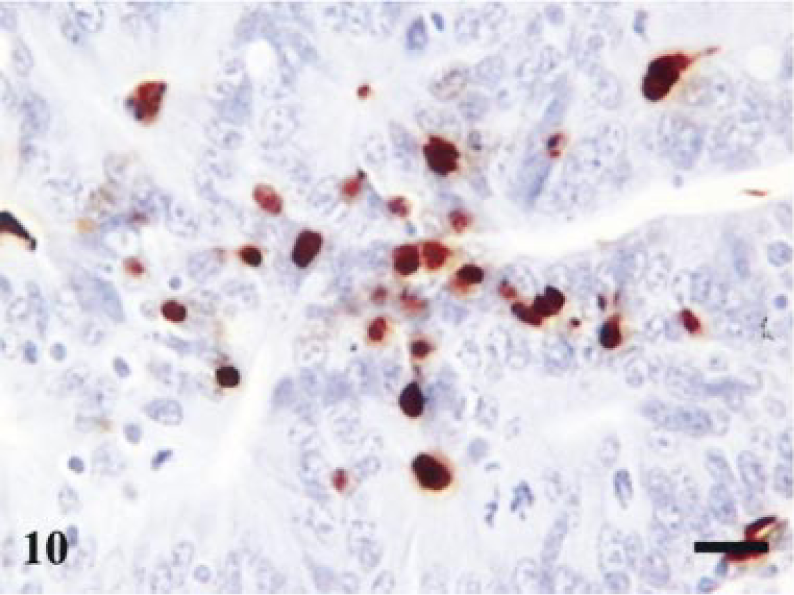
Terminal ileum; infected pig. Day-7 postinfection. Staining for macrophages. Macrophages trafficking into the epithelial compartment. Avidin–biotin–peroxidase complex method. Mayer's hematoxylin counterstain. Bar = 30 µm.
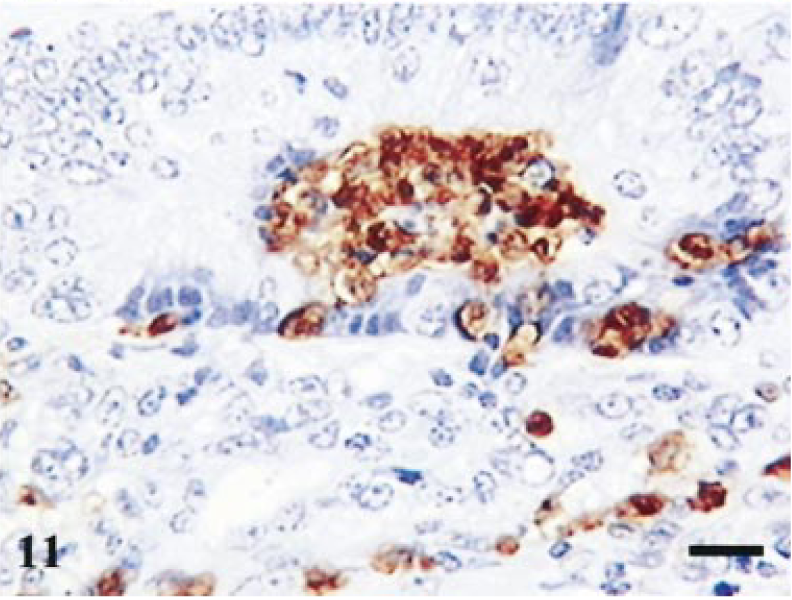
Terminal ileum; infected pig. Day-14 postinfection. Staining for macrophages. Intraepithelial macropahges forming large aggregates in crypt lumen. Avidin–biotin–peroxidase complex method. Mayer's hematoxylin counterstain. Bar = 40 µm
In addition to these comparisons between maximal and resolved lesions, comparisons also were made with counts on the reference tissue (for CD3 and CD79 only). This revealed significant differences between the day-14 time point and reference material for intraepithelial CD3 cells (P = 0.004, t = −3.905), with mean levels of 26.68 and 62.52, respectively (positive cells per 100 epithelial cells). B-cell numbers also were significantly reduced on day 14 in comparison with reference material (P = 0.001, t = −4.918), with mean levels of 424.3 and 1,006.5, respectively (counts per 1,000 m3 lamina propria).
Staining for MHC-II showed a concentration in the core of the villi and in the lamina propria around the deep crypts. In heavily infected pigs on day 14, there was an apparent reduction in the overall amount of MHC-II expression relative to the reference tissue; however, this was not quantified.
Staining for e-cadherin was carried out on samples of terminal ileum at time points 3, 7, 14, 21, and 28. On day-3 postinfection, all pigs exhibited strong surface staining for e-cadherin. On day 7, two pigs were negative and two positive. On day 14, all pigs were negative except one showing mild and variable staining. On days 21 and 28, one pig was negative in both groups. Age-matched reference showed positive staining in five of the seven pigs.
Discussion
In this article, we present the first prospective study to follow the histopathologic and immunologic changes occurring after experimental infection of pigs with L. intracellularis. Animals were obtained from a minimal disease herd with no history of L. intracellularis infection. As an obligately intracellular bacterium, it is not practical to cultivate L. intracellularis from fecal samples; indeed, it is difficult to do so from tissue. However, before inoculation, fecal specimens from all animals were assessed microbiologically for B. hyodysenteriae, B. pilosicoli, Yersinia spp., and Salmonella spp. Assessment for L. intracellularis was carried out both by PCR of fecal material and serologic assay on serum extracted from whole blood samples collected before inoculation. 2 All animals were negative by these techniques for the above pathogens. The light microscopic studies undertaken over the 42-day time course of the infection have provided a comprehensive and detailed account of the cellular events occurring after inoculation with L. intracellularis. These results underline the difficulty of detecting infection grossly at postmortem and highlight the usefulness of histopathology and IHC for definitive organism identification.
The course of infection was broadly in line with predictions from previous experimental infections in which lesions were evident most commonly in the terminal ileum and the upper colon between 7 and 21 days postinfection. 1 Immunocytologic detection of L. intracellularis proved highly sensitive with levels of infection as low as one crypt in 500 of those clearly detectable, thus permitting the progress of the infection to be studied from the earliest infection stages with less than 1% of crypts infected (as assessed by IHC) through the peak of infection with more than 90% of crypts infected to the recovery phases and the return toward normal cell populations.
These results support the previous findings that the crypts in the terminal ileum, caecum, and upper colon are the preferred sites of infection. 17 The reasons for preference of L. intracellularis for the crypt cells rather than the mature epithelium remain speculative. Brush-border development may play a role in the affinity of L. intracellularis as the immature enterocytes at the base of crypts have a poorly developed brush border compared with the villus epithelium; similarly, epithelial cells in the upper colon have poorly developed brush borders. In this study, it is interesting to note that on day 14 bacteria could be observed in association with the lumenal surface epithelium in the ileum and colon in addition to the deep crypts. Bacterial association and possible entry via the colonic surface epithelium is an event that was not reported previously. In addition to the possible role of the immature brush border in this phenomenon, these cells also have a higher turn over rate. Results from cell culture experiments suggest that bacterial replication is entirely dependant upon proliferating cells. 4 , 9 Further studies into the permissiveness of colonic and ileal epithelium are required to provide definitive answers for the tropism of L. intracellularis.
Histologic examination showed that infection, lesions, and changes in immune populations peaked on day 14 postinoculation. The earliest lesions identified were on 3 days postinfection and were characterized by neutrophil infiltrates in the absence of detectable hyperplasia.
The two pigs exhibiting the hemorrhagic form of the disease demonstrate clear differences when compared with the uncomplicated form of disease: notably, L. intracellularis was observed in large numbers, free in the lamina propria. Speculation has existed as to the mechanism by which the bacteria are released from the enterocytes and whether this results from cell rupture or as a consequence of active escape by the bacteria from the enterocytes. This study has provided evidence that the latter may be the likely mechanism with the observation of aggregates of bacteria at the basal end of the enterocytes in what appears to be membrane-bound vesicles (Fig 4); this theory would require further ultrastructural examinations for confirmation. Previous studies have reported the presence of bacteria-filled cell protrusions in cell culture experiments. 5 , 10 An additional report documented the presence of bacteria-filled balloonlike protrusions and bacteria-filled epithelial cell protrusions in ileum of experimentally infected pigs. 13
This study has provided the first detailed analysis of the components of the immune response associated with L. intracellularis infection. Although clearly not optimal, the features of the immune population quantified in the age-matched reference pigs together with data from our own and other laboratories 15 established a reference population with which to compare T-cell and B-cell numbers in the challenged animals.
The reduction in villus epithelial–associated CD3+ve T-cell numbers was unexpected with the results from a previous study, indicating a slight increase in T-cell numbers. 12 However, this study is the first comprehensive systematic analysis to be undertaken and provides evidence of a significant reduction in CD3+ve T-cell numbers occurring on 14 days postinoculation. The reduction in CD3+ve T cell in villus epithelium was a reflection of the loss of the CD8+ve T-cell villus epithelial population with CD8+ve T cells accounting for approximately 84% of T cells associated with villus epithelium. 20 Although cytolytic CD8+ve T cells respond to antigens presented in association with MHC class I molecules, any reduction in their numbers would be expected to affect the host's ability to mount a cellular response to intracellular pathogens. In contrast to these significant alterations in the CD8 population, the CD4 population remains relatively unchanged.
Furthermore, the reduction in B-cell numbers was an unexpected result because accumulations of IgA within enterocytes and adjacent to intracellular bacteria have been observed previously in this disease. 6
As a result of the observed loss of T cells from the intraepithelial compartment, IHC for pan-cadherin (to detect cell surface expression of e-cadherin) also was carried out to test the hypothesis that there may be down regulation of the ligand for intraepithelial lymphocytes associated with the disease (data not shown). Modulation of e-cadherin expression is a recognized feature in many enteric diseases 16 , 21, and we sought to investigate whether e-cadherin modulation was involved in the reduced CD8-positive T-cell numbers in the epithelium in PE. Although some heavily infected pigs showed apparent down regulation of e-cadherin, variable expression of this marker was present in control animals examined making full interpretation of these results unreliable. Nevertheless, investigation of the underlying mechanisms for the dramatic loss of cells from the intraepithelial compartment in association with this disease is clearly an area worthy of future study.
The results in this study of MHC-II expression were in agreement with the majority of workers in that pig intestinal epithelial cells do not express MHC-II molecules. 14 MHC-II expression was concentrated in the villous core resembling the known normal T-cell distribution in the pig gut, with MHC-II–+ve CD4 T-cells concentrated in the villus core and MHC-II–−ve CD8 T-cell populations immediately below the basement membrane of epithelial cells.
Macrophages were present in larger numbers in crypts showing hyperplastic lesions rather than those infected but not hyperplastic. This massive infiltration of activated macrophages, peaking on day 14, during this study has not been reported previously. The presence of large numbers of macrophages and their observed preference for hyperplastic crypts may reflect the release of macrophage chemo-attractants by hyperplastic crypts; however, this area requires further investigation.
High numbers of bacteria can be observed within the lumenal macrophages and are identifiable as L. intracellularis by IHC. In the hemorrhagic form of the disease, large numbers of bacteria can be observed within the lamina propria and in macrophages. This may suggest that either the macrophages are able to engulf large numbers of bacteria very quickly or that bacteria are able to multiply within the macrophages. In vitro experiments involving L. intracellularis growth in macrophages appear to support this view with evidence of an increase in the numbers of bacteria in some macrophage cell lines (S. C Mitchell, D. G. E. Smith, unpublished).
The presence of the high levels of macrophages may be a key factor in the development of the hemorrhagic form of the disease. Although the increase in macrophage numbers may be important in restitution, the potential release of excessive cytokines may lead to the endothelial damage and changes in vascular permeability observed in PHE. Alternatively, the hyperplastic cells themselves may release cytokines that lead to massive attraction of macrophages. Severe disease (PHE) is characterized by large numbers of bacteria that escape into the lamina propria where they are engulfed by macrophages. The presence of L. intracellularis in macrophages may lead to the release of cytokines such as tumor necrosis factor α causing increased vascular permeability and hemorrhage. This remains speculative at present, and further examination of the cytokine responses during L. intracellularis infection is required to provide insight into immune mechanisms involved in precipitating PHE.
In conclusion, this study has provided further information on the pathogenesis and associated immunologic alterations associated with L. intracellularis infection. More specifically, we have provided important data that suggest for the first time that infection corresponds to induction of an apparently immunosuppressive phenotype with down regulation of the adaptive immune response through a reduction in CD8+ve T cells and B cells. Results obtained during these experiments also have provided details of a significant macrophage involvement not previously reported, which may provide some of the answers to the enigma of the hemorrhagic form of the disease.
The consequences of L. intracellularis infection are undoubtedly complex, and further work will be required to elucidate mechanisms underlying the observed immunologic and cellular changes.
Footnotes
Acknowledgements
We are grateful to Joanne Hannigan, Lorraine Henderson, and Gordon Glass for their technical assistance. We gratefully acknowledge the generous gifts of monoclonal antibodies from Dr. Karen Haverson, University of Bristol. This work was supported by funding obtained from the BBSRC (Ref.15/A08055) and Institute of Medical Laboratory Sciences. D. J. Shaw is funded by the Wellcome Trust as part of their International Partnership Research Awards in Veterinary Epidemiology. Scottish Agricultural College receives financial support from the Scottish Executive Environment Department.
