Abstract
An outbreak of classical herpetic infection causing vesicoulcerative stomatitis in a family group (eight animals) of Callithrix jacchus is described. In all eight infected animals, human herpesvirus 1 (HHV-1) was identified as the causative agent. This was confirmed by histologic, immunohistologic, and molecular biologic investigations, as well as by virus isolation. The clinical picture, the macroscopic appearance, and the histologic results indicated a herpes infection as the cause of mortality. Alterations of the oral mucous membranes were erosive to ulcerative with typical intranuclear inclusions. Immunohistologic and molecular biologic techniques clearly identified the HHV-1 virus and excluded other possible primate herpesviruses such as B-virus, SA8, HVP-2, and Herpes tamarinus. The significance of this herpesvirus infection for colony management is discussed.
Keywords
Nonhuman primates are primary hosts to a number of herpesviruses. 3 These viruses generally cause mild or inapparent infections in their natural host, but some are associated with severe disease when transmitted to other species. The well-known spontaneous interspecies transmissibility of herpesviruses is responsible for a high zoonotic risk and may result in fatal diseases of either humans or animals. Human herpesvirus 1 (HHV-1) is one of the best-characterized viruses transmissible from human to nonhuman primates, but spontaneous infections in monkeys appear to be rare. Humans are the original host and the reservoir; they exhibit typical clinical symptoms such as acute gingivostomatitis during the primary infection. In Old World primates, natural HHV-1 infection has been described in gorillas (Gorilla gorilla), 7 chimpanzees (Pan troglotydes), bonobos (Pan paniscus), 16 and white-handed gibbons (Hylobates lar). 6 , 21 , 22 In these species, the disease remains localized to mucocutaneous tissues. It seems to be comparable with the human disease and usually does not become a systemic infection. New World monkeys and prosimians are more susceptible to infection and disease. The disease course is severe, leading to death in most cases. In spontaneously infected owl monkeys (Aotus trivirgatus), the generalized febrile disease is characterized by erosions and ulcers of oral mucous membranes and mucocutaneous junctions of the lips, accompanied by focal necrosis and hemorrhages in all organs, including the cerebral cortex. Death occurs within 16 days after the onset of symptoms. 14 , 17 Similar gross lesions are observed in naturally infected tree shrews (Tupaia glis), 15 lemurs, 12 and most experimentally infected marmoset and tamarin species (Callithrix sp., Saguinus sp.). 10 Spontaneous HHV-1 infection was described in a fatal case in black tufted-ear marmosets (Callithrix (C.) penicillata) in a conservation unit of the State park of Serra da Tiririca, Niteroi, in Brazil, as well as in four young pet marmosets (C. jacchus) belonging to private owners in Spain and Brazil. 5 , 11 , 18
Our report describes a HHV-1 outbreak in a group of C. jacchus housed as a family group after contact with a person who had recrudescent HHV-1 disease. We describe the clinical appearance and pathomorphologic alterations in the disease and virus detection using immunohistologic and molecular techniques as well as virus isolation.
The infected animals belonged to a colony consisting of up to 45 animals kept in a greenhouse or in huts with outdoor facilities. All animals were housed in family groups (breeding couple with up to four generations of offspring). The eight members of the infected family were housed in mesh wire cages in the greenhouse, with both natural daylight and artificial light. The animals were fed a pap in the morning, vegetables, fruits, and animal protein source in the evening, and water ad libitum. In this greenhouse, there were up to 10 different families living without direct contact. Visual contact between family groups was prevented by long curtains, but indirect contact with bodily fluids or other material through caretakers, scientists, and exchanged food or water bowls was possible.
Clinical Findings
On the first day of the outbreak, the α-male (marmoset No. 1) showed apathy, anorexia, weakness, and marked salivation in the morning and died the same afternoon. At this time, no other animals in the group showed clinical signs. The following morning one young adult (marmoset No. 2) and the α-female (marmoset No. 3) developed excessive salivation and apathy (Table 1). Closer examination showed ulceration of the lips (Fig. 1) and the oral cavity. All seven animals were treated with Baypamun® (inactivated Parapoxvirus ovis D1701, Bayer, Leverkusen, Germany) 0.5 ml/animal s.c. and 5 mg/kg marbofloxacin Marboxyl® (Vetoquinol, Oberursel, Germany). The next day, the young adult and the female were found dead. All other animals developed the same symptoms and were euthanatized. In summary, all eight animals had a 1–2 day history of vesicular to ulcerative gingivitis and stomatitis accompanied by severe salivation, serous nasal discharges, anorexia, and depression. Three animals developed lesions covered with fibronecrotic exudate at the mucocutaneous junctions (marmoset Nos. 3, 4, and 7).
Animals and clinical signs of infection.
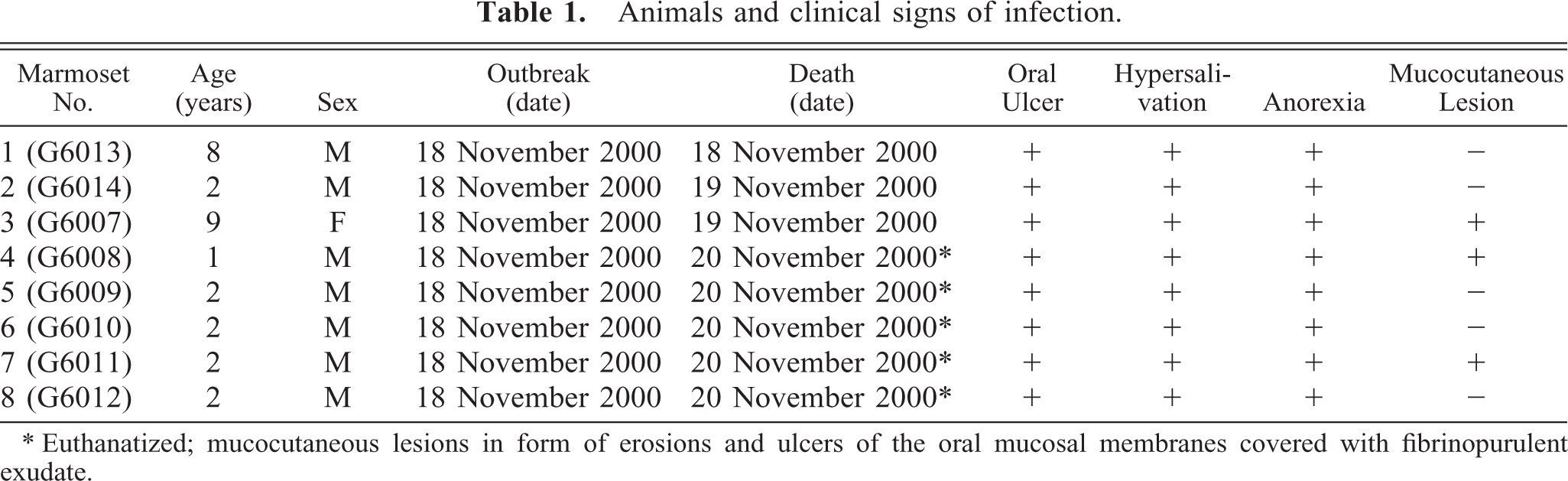
∗ Euthanatized; mucocutaneous lesions in form of erosions and ulcers of the oral mucosal membranes covered with fibrinopurulent exudate.
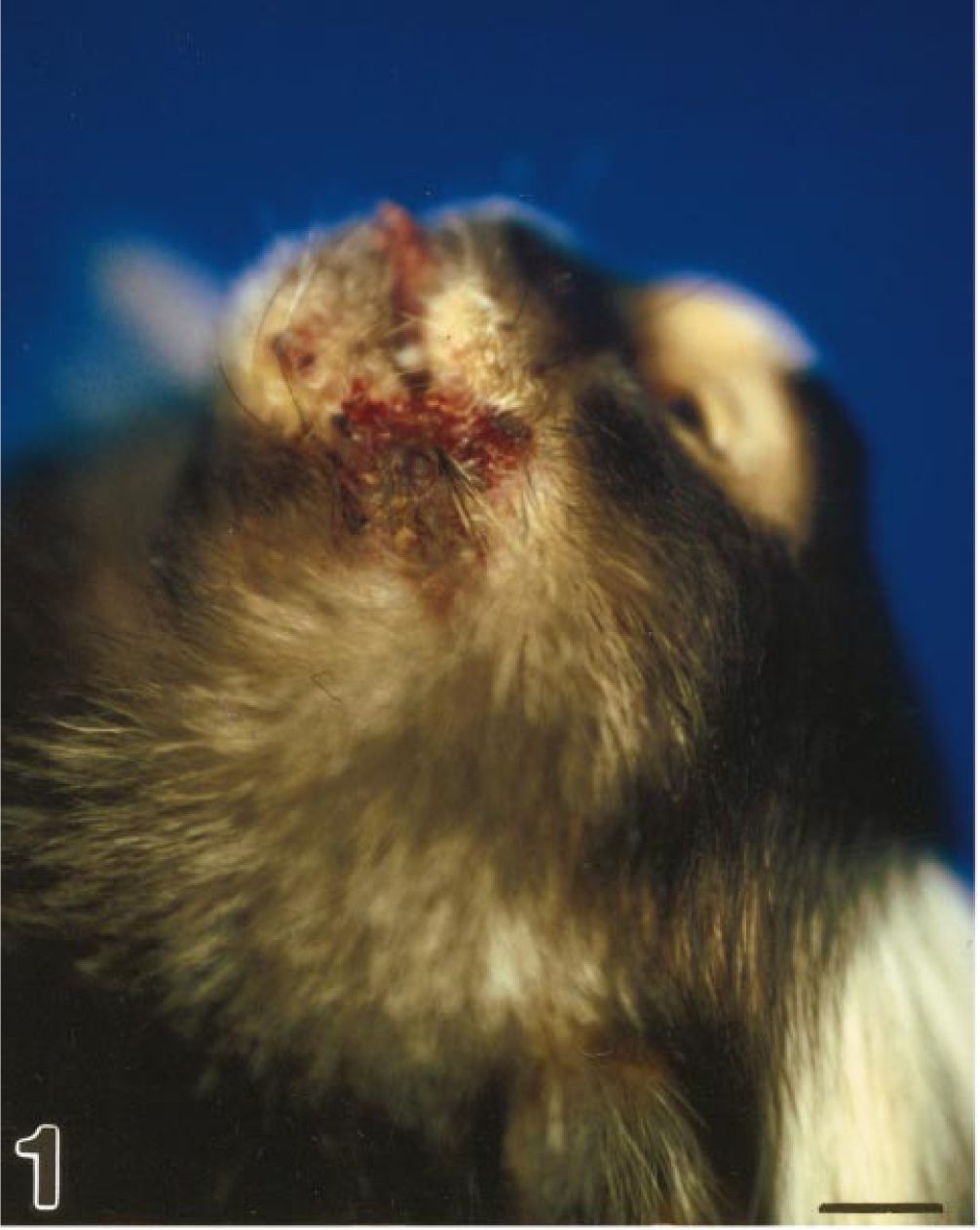
Head; marmoset No. 3. Severe cutaneous lesions covered with fibronecrotic exudate at the mucocutaneous junctions.
Pathology
At necropsy, all animals showed an acute gingivitis and stomatitis. The tongues had multifocal single to coalescing lingual erosions with rough borders up to 0.5–1.0 cm in diameter. Some lesions appeared to be lingual or gingival ulcers (marmoset Nos. 3, 4, and 7) covered by a fibrinopurulent exudate (Fig. 2). No other gross lesions were seen except a mild splenomegaly and severe lymphadenopathy of the regional lymph nodes. Tissue specimens were fixed in 4% neutral buffered formalin or frozen in liquid nitrogen. Light microscopy was performed on formalin-fixed, paraffin-embedded specimens. Histopathologic changes in the mucosal lesions were similar in all animals. There was severe vesiculation and ulceration of squamous epithelia of the tongue with acantholysis, parakeratosis, coagulation necrosis, and polykaryocytosis. Necrotic plaques extended from the submucosa to the surface. Epithelial cells located at the ulcer margins showed varying degrees of degeneration and necrosis. Intranuclear inclusion bodies were seen, particularly at the borders of vesicles and ulcers (Fig. 3). These inclusions filled the nucleus or were surrounded by a clear halo. They appeared red-blue in Feulgen-stained sections. A marked neutrophilic and lymphohistiocytic infiltration was evident in the underlying submucosa. Furthermore, all animals had a mild mixed cellular infiltration of the adrenal cortex. Three animals (marmoset Nos. 1–3) exhibited a mild interstitial hepatitis characterized by multifocal lymphohistiocytic inflammation. Inclusion bodies were not observed in these lesions. All other organs investigated were unexceptional. On the basis of the clinical signs and gross and microscopic findings, infection with a herpes virus was suspected. This was confirmed by the methods discussed below.
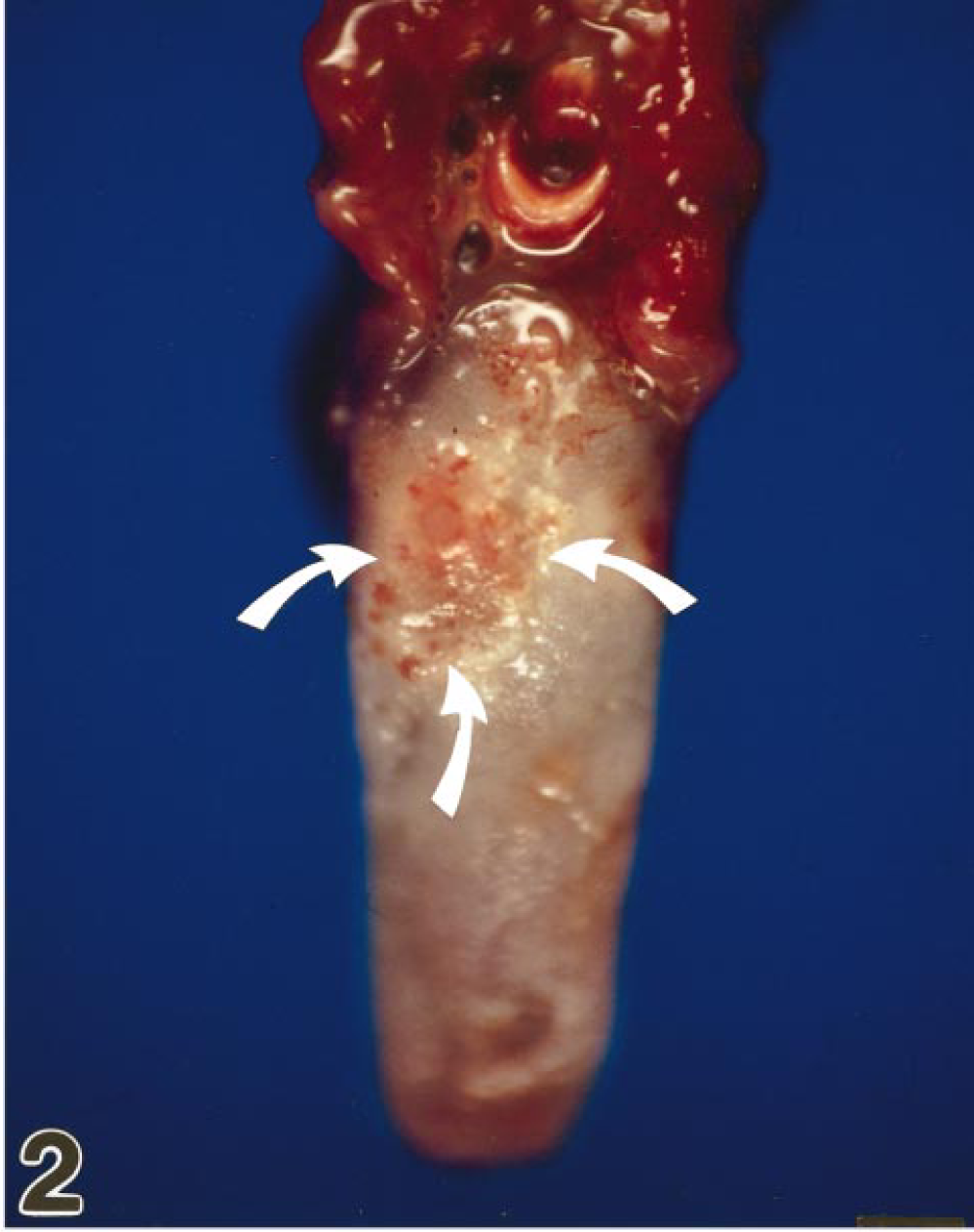
Tongue; marmoset No. 3. Acute severe glossitis with multifocal discrete to coalescing lingual erosions and ulcers with rough borders (arrows).
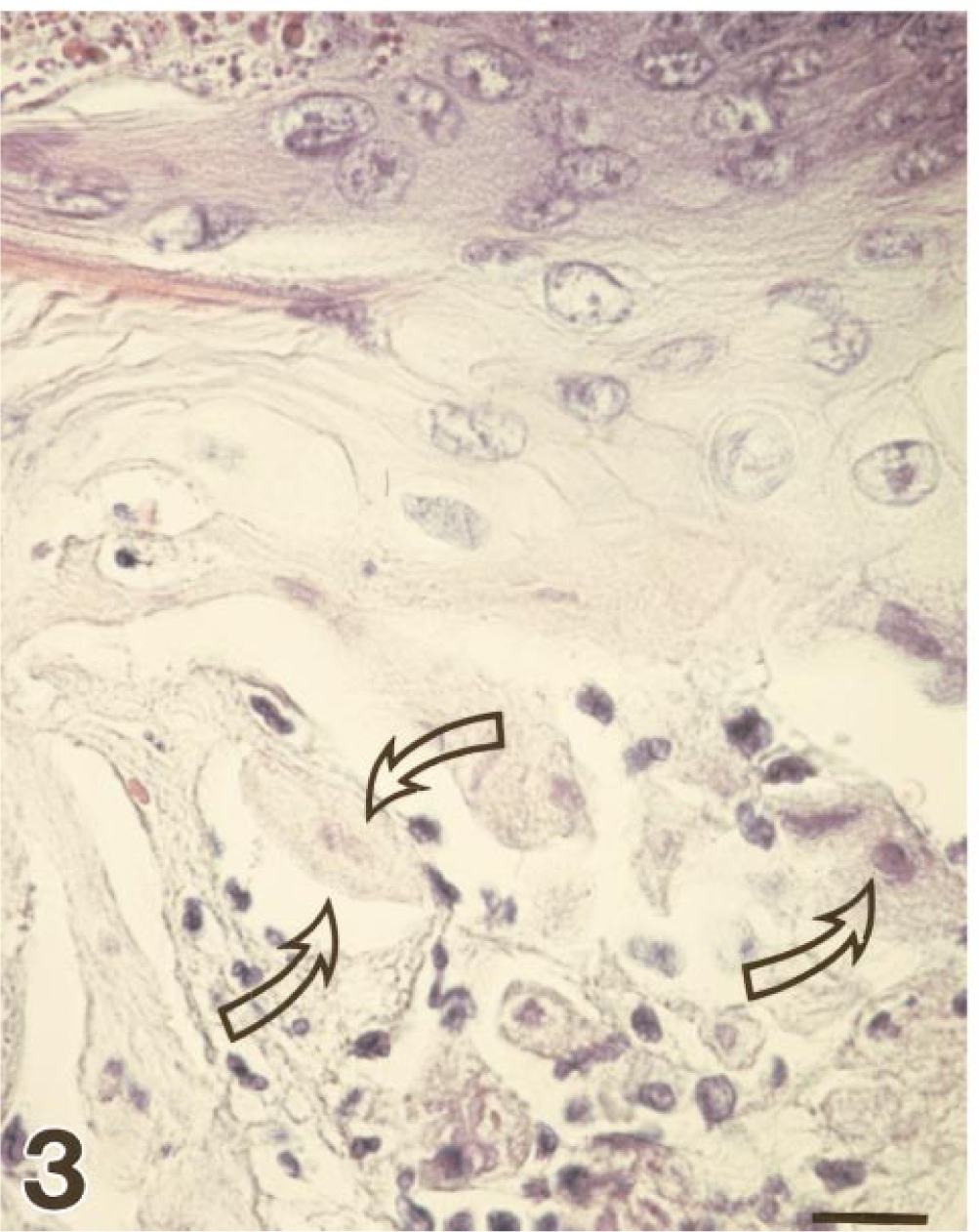
Tongue; marmoset No. 3. Histologic demonstration of tongue lesions with acantholysis, parakeratosis, coagulation necrosis, and intranuclear inclusions in the border area of the vesicle region (arrows). HE. Bar = 17 µm.
Immunohistochemistry for HHV-1 virus was performed on formalin-fixed, paraffin-embedded sections using a monoclonal antibody directed against HHV-1 and −2 antigen (Biogenex, cat No., MU086-UC) at a dilution of 1 : 200, after protease XXIV (Ventana) pretreatment. Immunohistochemistry revealed a specific antigen-antibody reaction in the lingual lesions of all animals (Fig. 4). The whole ulcer region, especially the degenerate epithelial cells, was strongly positive. Furthermore, virus antigen could be detected in the liver and brain tissue of some animals (marmoset Nos. 4–8). In the liver, virus-positive cells were found within inflammatory cell infiltrates. The central nervous system contained single neurons positive for herpesvirus antigen. The adrenal glands were negative for herpesvirus antigen.
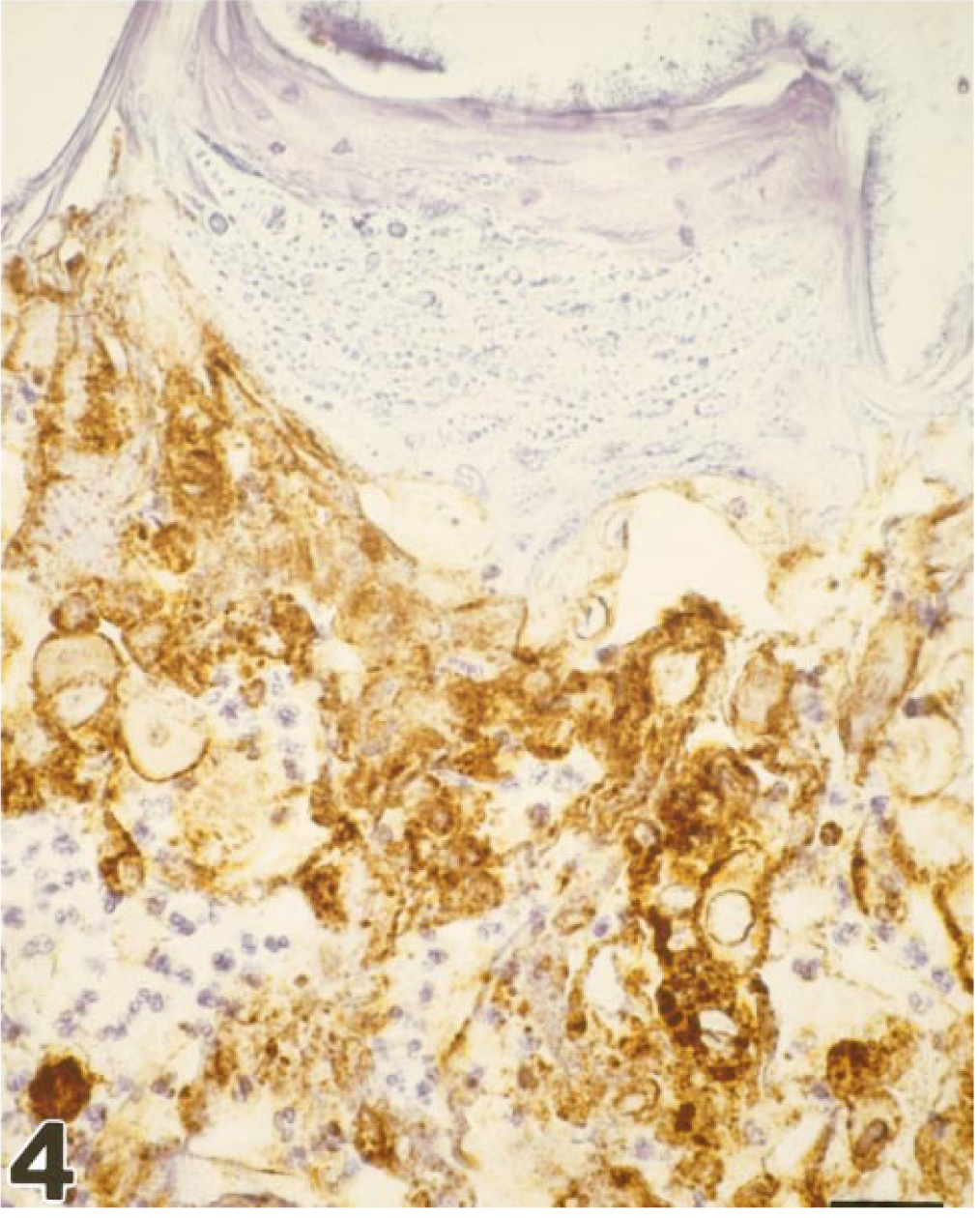
Tongue; marmoset No. 3. Immunohistochemical demonstration of Herpes simplex antigen within the tongue lesion, membranous staining pattern. Avidin–biotin–peroxidase complex method, Mayer's hematoxylin counterstain. Bar = 26 µm
Intranuclear viral nucleocapsids, either empty or filled with electron dense material, were observed by electron microscopy within epithelial cells. Large numbers of enveloped virions in the cytoplasm and intercellular spaces could be demonstrated (Fig. 5). Intranuclear particles measured 80–100 nm; the enveloped particles had an outside diameter of 150 mm or more. Many of the virus particles had the hexagonal configuration characteristic of members of the herpesvirus group. Size, location, and morphologic features were consistent with the identification of the virus as a member of the herpesvirus family.
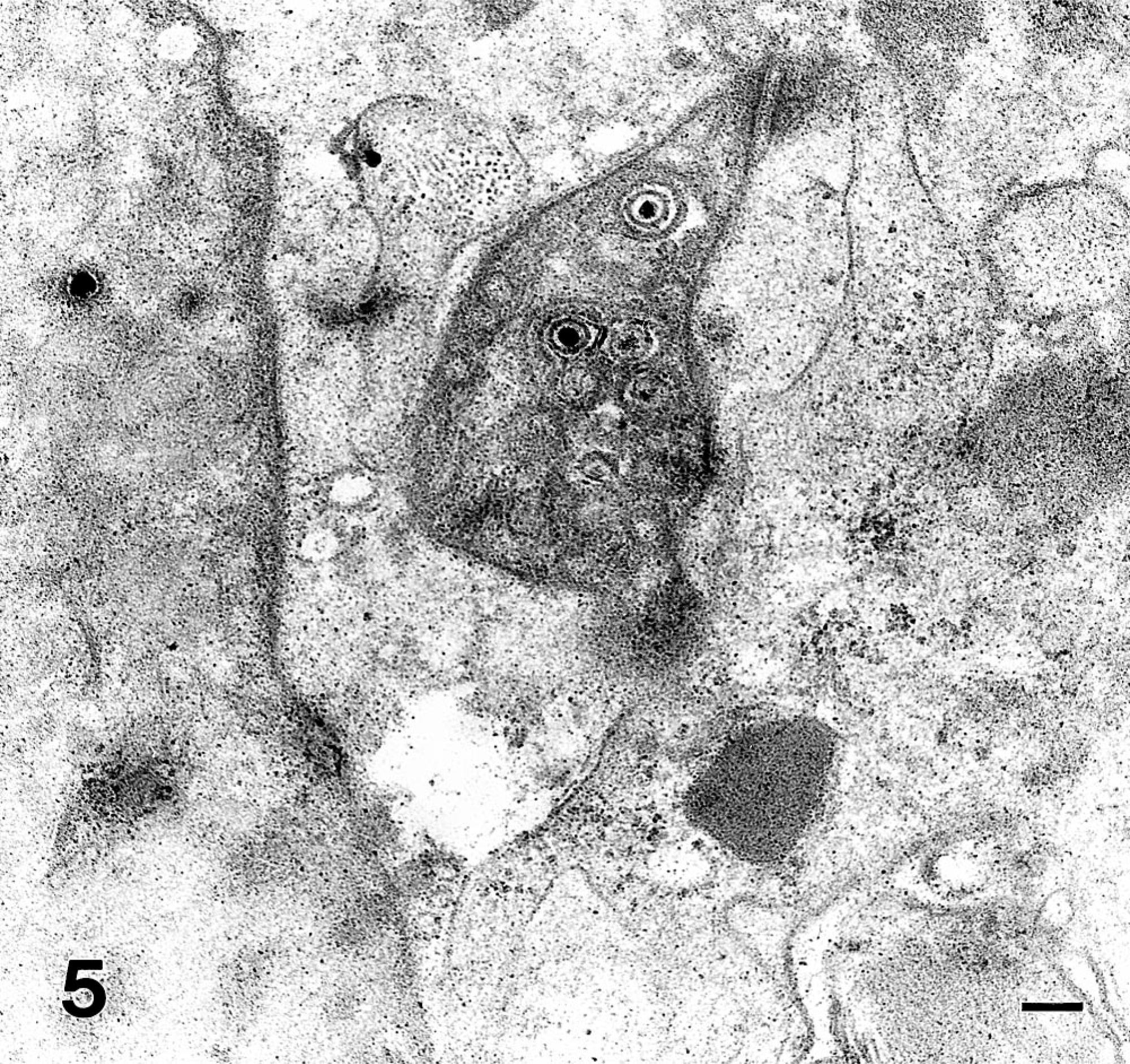
Tongue; marmoset No. 3. Electron microscopic demonstration of herpesvirus particles showing numbers of enveloped virions 140 nm in diameter in the cytoplasm and intercellular spaces. Transmission electron microscopy. Bar = 160 nm
Virus culture from swabs of the oral cavity (marmoset Nos. 3–8) was performed on BHK-21 (a gift from Dr. D. Daniels, New England Regional Primate Research Centre, Southborrough, NE, USA), Vero (Biowhittaker/Boehringer Mannheim, Germany), and chimpanzee primary fibroblast cell lines. Cells were cultivated at 37 C in Dulbecco modified Eagle medium (Life Technologies, Scotland) supplemented with 1× glutamine (GIBCO-BRL, Life Technologies, Scotland), 1× penicillin/streptomycin (GIBCO-BRL, Life Technologies, Scotland), and 10% fetal calf serum (GIBCO-BRL, Life Technologies, Scotland). Daily screening of the cultures revealed small plaques in the monolayer of Vero, BKH, and chimpanzee primary fibroblast cell lines after 1 day of culture. After 3–4 days, the monolayer of the cells had completely disappeared; all cells had detached from the bottom of the flask and were floating in the culture medium. Growth characteristics of the isolated virus were consistent with those of α-herpesviruses. DNA was isolated from infected BHK, Vero, and chimpanzee primary fibroblast cell lines. Viral sequences were amplified by nested polymerase chain reaction (PCR), which is essential for the amplification of primate α-herpesviruses, as described by Black and Eberle. 2 A BLAST search in the GenBank, EMBL, DDJB, and PDB sequence databases showed a 99% sequence identity of the PCR fragment with HSV-1 sequences.
Furthermore, PCR analysis was performed on several specimens obtained at necropsy and frozen at −80 C. A primer set designated B9/B10 was used to identify the herpesvirus. B9/B10 are located within the highly conserved C1 and C2 regions of the glycoprotein gB encoded by HHV-1, HHV-2, B-virus, SA8, and HVP2 and amplify the highly divergent D2 region of gB (360 bp for HHV-1 [KOS], KO1760, PubMed, genebank). Restriction digestion of the PCR product with HaeIII results in a virus-specific restriction pattern. 2
DNA from liver, heart, lung, tongue, lymph node, kidney, spleen, and blood (marmoset No. 2), liver, lymph node, tongue, kidney, spleen, heart, and blood (marmoset No. 5), and liver, lung, spleen, tongue, heart, kidney, and blood (marmoset No. 6) was isolated by using the QIAamp Mini Kit (Quiagen, Hilden, Germany). PCR with DNA from the tongue as template resulted in an amplification product of 360 bp, with that obtained with the HHV-1–positive control (Fig. 6). PCR of all other DNA samples did not result in any amplification product. Addition of dimethyl sulfoxide (5%), betaine (1 M), or both did not improve these results. In a gradient PCR with different annealing temperatures ranging from 48 to 60 C, only DNA from the tongue of three animals was positive. Higher annealing temperatures lead to nonspecific bands in all samples. To identify the origin of the PCR amplification products, the purified PCR products were digested with the restriction enzyme HaeIII. As shown in Fig. 7, the restriction fragments had the molecular sizes expected for HHV-1, about 175 bp, 126 bp, and minor bands in the range of 40 and 20 bp (computer predicted). 2 They comigrated with the restriction fragments of the HHV-1 control DNA, indicating that the animals were infected with HHV-1.
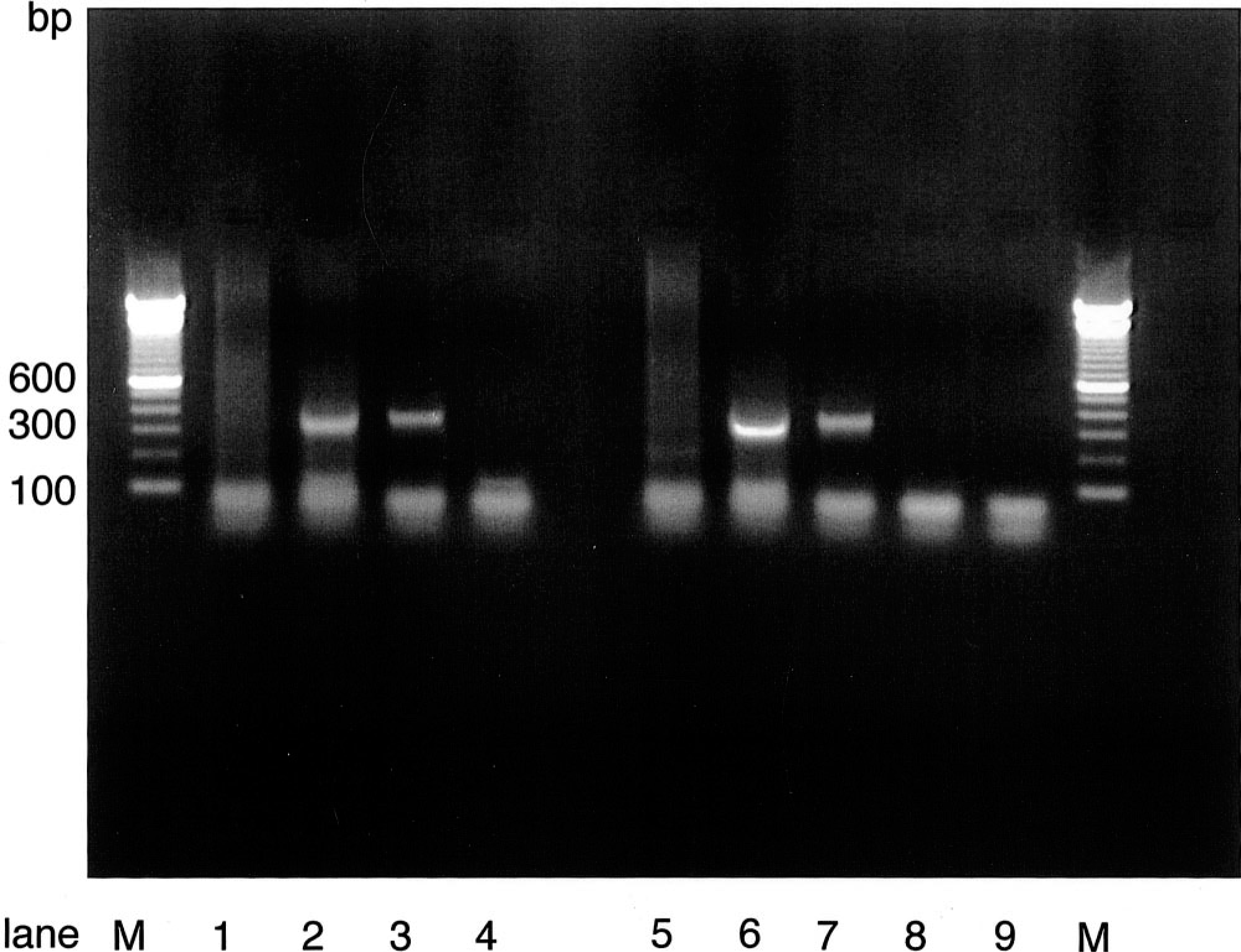
PCR amplification products. Tongue; marmoset No. 6. Lanes 1 and 5, spleen; lanes 2 and 6, tongue; lanes 3 and 7, HHV-1 positive control; lanes 4 and 8, HHV-1 negative control; lane 9, PCR negative control with water instead of DNA. To the samples in lanes 5–8 betaine was added to a final concentration of 1 M. M, marker DNA—100-bp ladder. The expected amplification product of about 360 bp was observed only using the DNA's of the tongue (lanes 2 and 6) and the HHV-1 positive control (lanes 3 and 7). The template DNA from other specimens also was negative after addition of betaine (lanes 5 and 8)
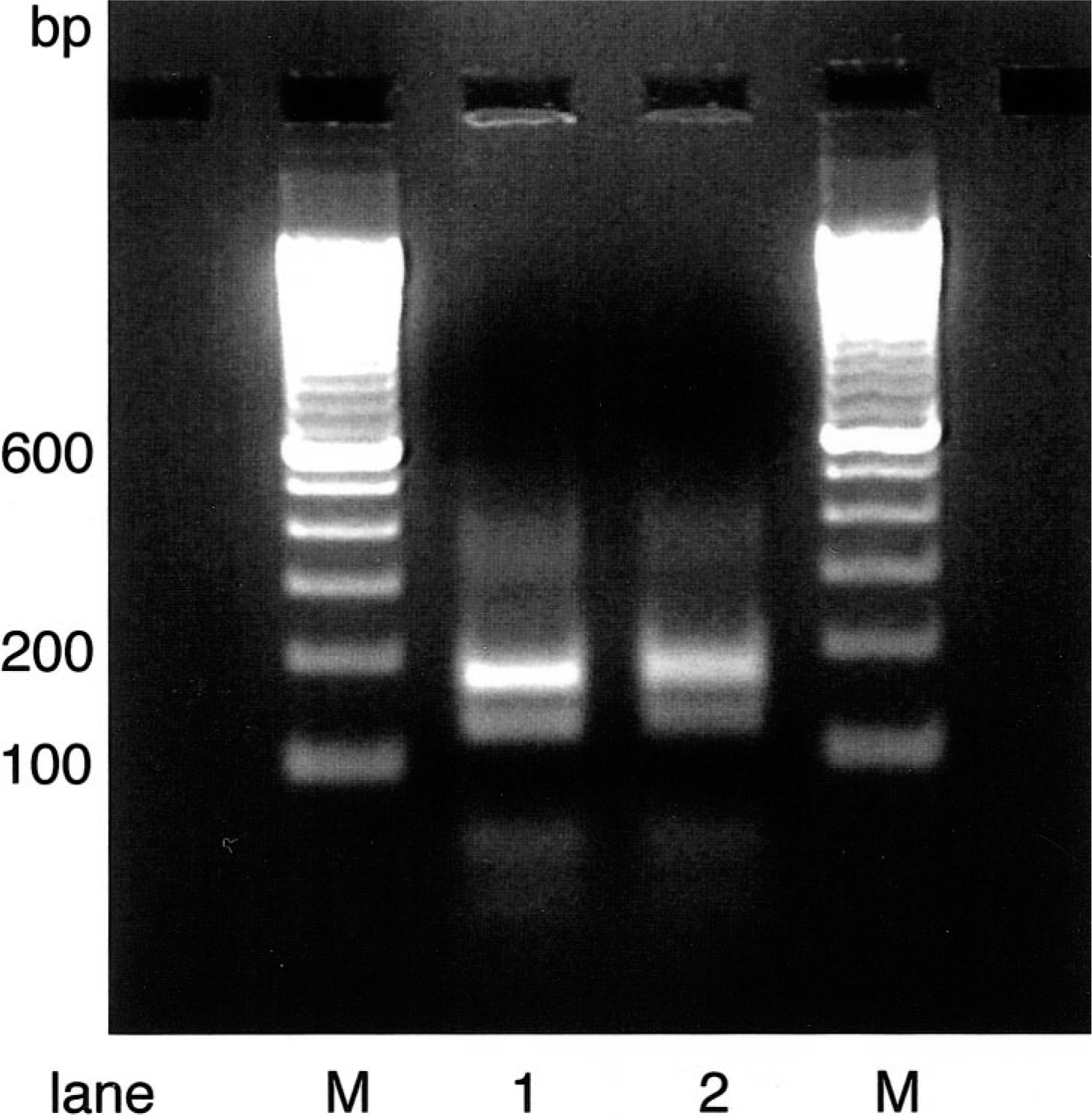
Tongue; marmoset No. 6. HaeIII digestion pattern of the PCR amplification products from the tongue (lane 1) and the HHV-1 positive control (lane 2). M, marker DNA—100-bp ladder. The samples were electrophoresed on 2% agarose gels. The predicted computer analysis bands of 175, 126, 43, and 20 bp for HHV-1 are detectable. They are specific for a distinct region of the glycoprotein gB of α-herpesviruses
New World monkey species are regarded as highly susceptible to HHV infections compared with Old World monkeys such as macaque species and great apes. Natural infections of callithrichids with HHV-1 seem to be rare and most available data are based on experimental infections. 20 This may be misinterpreted as a low infection risk. The HHV-1 outbreak in callithrichids described in this case report clearly demonstrates the fulminant progression of herpesvirus infection to disease in marmosets and shows the high susceptibility of this species to HHVs. The animals developed the classical clinical signs of herpesvirus infection. The clinical course of this outbreak was dramatic, leading to the sudden death of three animals within 3 days after the onset of the first symptoms. Alterations in the oral mucous membranes were erosive to ulcerative. Histologically, massive epithelial damage was noted. Typical intranuclear inclusions were found. Results of all investigations were indicative of HHV-1 infection. According to the results of immunohistochemistry and PCR, HHV-1 DNA sequences and antigens were present in oral tissues of all infected marmosets. The most severe alterations were found on the mucocutaneous regions of the mouth and especially on the tongue of each animal. Interestingly, none of these animals developed a severe necrotizing hepatitis or a multifocal nonsuppurative meningoencephalitis, alterations that are commonly found in herpesvirus infections of nonhuman primates. 4 , 20 In the cases described by Juan Salle, the animals developed lesions in the central nervous system. 11 A possible explanation for this observation is the course of disease in the described outbreak. Three of the eight animals rapidly died in the acute phase of infection, the remaining five animals were euthanatized for humane reasons. The liver lesions found in three of the animals might be interpreted as an early stage of a systemic infection; the other animals died at the stage of a severe local mucosal infection. The three animals that died first were regarded as the animals primarily exposed to the infectious agent.
It seems likely that close contact with persons suffering from an oral herpesvirus infection was the source of virulent HHV-1. In the case reported here, the animals had contact with a caretaker suffering from a herpes labialis–like illness a few days before the outbreak occurred. In most of the reported infections in various nonhuman primates species, it was indicated that contact with humans was the source of infection. 6 , 12 , 15 , 17 , 22 Visitors, students, or caretakers might be able to infect the animals, for example, by passing partly eaten food into the cage. Most reported cases of herpesvirus infection in callithrichids occurred in animals kept by private persons, often in close association with the family. Close contact seems to be necessary for transmission of the infection. The route of transmission in spontaneous infections in nonhuman primates is unknown, but it seems likely that direct contact or aerosol was involved. Once brought into a colony, the disease spread rapidly and had high morbidity and mortality. 9 Because humans are the natural or reservoir host for the virus, contact with symptomatically and subclinically HHV-infected humans should always be avoided. Infected persons can excrete the virus even in the absence of visible lesions. The use of appropriate protective clothing and face masks for humans handling nonhuman primates should greatly reduce the risk of infection.
The most characteristic gross findings in the reported cases of HHV-1 infection were discrete vesicles, necrotic plaques, and erosions or ulcers of the oral mucous membranes and mucocutaneous junctions. These lesions cannot be differentiated from herpesvirus T infection, which leads to a systemic infection with identical gross and microscopic lesions. 1 , 9 Herpes tamarinus (HVT) was originally isolated from moribund marmosets (tamarins) and owl monkeys. Squirrel monkeys are the natural host for this virus, 13 which is distantly related to the other primate herpesviruses (HHV-1, HHV-2, SA8, and B virus). 8 , 19 A definitive diagnosis can only be made by virus isolation and identification or by immunostaining techniques and molecular investigations as described in this study. Recently, the sequence of the glycoprotein B of squirrel monkey herpesvirus 1 was made public (accession number AYO95366, Breshears et al. 2002). Computer analysis of the primer pair B9/B10 demonstrated the distant relationship of Herpes T to HHV-1. No PCR product could be expected when the animals were infected with Herpes T. Alignments of the primer pair B9/B10 with different computer programs showed no significant homology to the published sequences of HVT. Therefore, the digested PCR products, which were detected in the tongue of three animals, were specific for HHV-1.
In conclusion, callithrichids may represent a highly susceptible host for human-borne herpesvirus infections. The contact with symptomatically and subclinically HHV-infected humans should always be avoided to reduce the risk of infection.
Footnotes
Acknowledgements
We are grateful to W. Henkel, K. Kaiser-Jarry, N. Knöchelmann, H. Gilhaus, and H. Zuri for their excellent technical assistance and to W. Bodemer for his generous advice and support.
