Abstract
Matrix metalloproteinases (MMPs) are a family of enzymes implicated in the degradation and remodeling of extracellular matrix and in vascularization. They are also involved in pathologic processes such as tumor invasion and metastasis in experimental cancer models and in human malignancies. We used gelatin zymography and immunohistochemistry to determine whether MMP-2 and MMP-9 are present in canine tumors and normal tissues and whether MMP production correlates with clinicopathologic parameters of prognostic importance. High levels of pro-MMP-9, pro-MMP-2, and active MMP-2 were detected in most canine tumors. Significantly higher MMP levels were measured in canine tumors than in nontumors, malignancies had higher MMP levels than benign tumors, and sarcomas had higher active MMP-2 than carcinomas. Cartilaginous tumors produced higher MMP levels than did nonsarcomatous malignancies, benign tumors, and normal tissues, and significantly greater MMP-2 than osteosarcomas and fibrosarcomas. Pro-MMP-9 production correlated with the histologic grade of osteosarcomas. The 62-kd form of active MMP-2 was detected only in high-grade, p53-positive, metastatic malignancies. Zymography proved to be a sensitive and quantitative technique for the assessment of MMP presence but has the limitation of requiring fresh tissue; immunohistochemistry is qualitative and comparatively insensitive but could be of value in archival studies. MMP presence was shown in a range of canine tumors, and their link to tumor type and grade was demonstrated for the first time. This study will allow a substantially improved evaluation of veterinary cancer patients and provides baseline information necessary for the design of clinical trials targeting MMPs.
Matrix metalloproteinases (MMPs) are a family of zinc-containing, calcium-dependent, proteolytic enzymes implicated in the degradation and remodeling of extracellular matrix and in vascularization. 34 , 36 Under physiologic conditions, they are expressed by a variety of cell and tissue types, and they are involved in processes such as trophoblast invasion, 14 development, 35 endometrial remodeling, 53 ovulation, 51 angiogenesis, 4 wound healing, 32 and bone resorption. 67 A growing body of evidence indicates that MMPs are also involved in a number of pathologic processes, including tumor invasion and metastasis in experimental cancer models 54 and in human malignancies. 27 , 38
Gelatinases or type-IV collagenases are a group within the MMP family, which includes MMP-2 (gelatinase-A), MMP-9 (gelatinase-B), and membrane-bound metalloproteinase-1 (MT1-MMP). 34 Like all MMPs, MMP-2 and MMP-9 are produced as zymogens and are subsequently activated to become proteolytic enzymes. 60 In terms of their importance in the process of tumor invasion and metastasis, the gelatinases have been shown to be capable of degradation of critical basement membrane and extracellular matrix components including type-IV 44 and type-V 37 nonfibrillar collagens, type-VII collagen, 56 type-X collagen, 71 elastin, 57 and fibronectin. 10
Increased concentrations of MMPs, and gelatinases in particular, are associated with invasion, metastasis, and poor prognosis in numerous human malignancies, 38 including lung, 23 breast, 15 , 66 gastric, 73 colon, 41 and prostatic 59 carcinomas, oral 22 and oesophageal 42 squamous cell carcinomas, neuroblastoma, 1 , 65 ovarian neoplasia, 19 melanoma, 68 and chondrosarcoma. 24 , 52 , 55
Although evidence for the role of MMPs in tumors of domestic animals is sparse, equine MMP-3 and tissue inhibitor of metalloproteinase-1 50 have been cloned and sequenced, and the central role of MMP-2 and MMP-9 activities in the pathophysiology of equine laminitis has been documented. 39 , 40 , 47 A 62-kd canine gelatinase has been isolated and identified. 13 Increased MMP-2 and MMP-9 levels occur in the synovial fluid in canine rheumatoid arthritis 12 and increased MMP-2 levels in canine 11 and equine 8 osteoarthritis, whereas increased pro–MMP-9 levels occur in the myocardium in canine dilated cardiomyopathy. 20 Gelatinase activity was significantly elevated in fluids from eyes of dogs with keratoconjunctivitis. 2 The immunohistochemical distribution of type-IV collagenase in normal, dysplastic, and neoplastic canine mammary gland has been reported 45 and, more recently, MMP-2 and MMP-9 were detected in canine osteosarcoma 28 and cutaneous mast cell tumors 31 at levels that were higher than those found in unaffected stromal tissue.
The aims of this study were to determine whether MMP-2 and MMP-9 are present in canine tumors and normal tissues, whether the production and profile of MMPs correlate with clinicopathologic parameters and p53 status, and whether they are predictive of tumor biologic behavior.
Immunohistochemistry and gelatin zymography were evaluated and compared as methods for accurately demonstrating MMP presence in tumor tissue. Gelatin zymography is an established electrophoresis technique for the detection of gelatinase activity even at low picogram concentrations, 26 , 29 modified to incorporate a gelatin substrate, which enables the observation of gelatinase activity after separation on polyacrylamide gels and which, given the substrate specificities of MMPs, makes this technique virtually exclusive for the detection of gelatinases.
This study provides evidence for the presence of MMP-2 and MMP-9 in a wide range of canine tumors and normal tissues. The involvement of MMPs in canine tumors, to a degree that varies according to the tumor type, and the link of MMP levels to tumor grade are demonstrated for the first time.
Materials and Methods
Tissue samples
A total of 76 fresh canine tissue samples, up to 0.5 cm in diameter, were collected at the time of biopsy, surgery, or during necropsy examination. Tissue samples included tumors, tissues immediately surrounding the tumors, and normal tissues from a range of organs. Where feasible, samples were taken from the marginal and central areas of the tumors.
Samples to be used for zymography were stored rapidly at −70 C until assayed. Part of each tissue sample was fixed in 10% neutral buffered formalin, routinely processed, and embedded in paraffin blocks. Serial sections 5 µm thick were prepared from each tissue block, mounted on silane-coated slides, and used for immunohistochemical staining. Other sections from the same tissue were stained with haematoxylin and eosin (HE) for purposes of diagnosis.
Pathology
Histologic slides were examined by two investigators (P. Loukopoulos, W. F. Robinson). The organs and tissues examined and the diagnoses offered are shown in Table 1. A small number of additional archival cases were used for immunohistochemistry.
Pro-MMP-2, pro-MMP-9, and total active MMP-2 production (in μg/mg) in canine tumors and normal tissues using zymography.
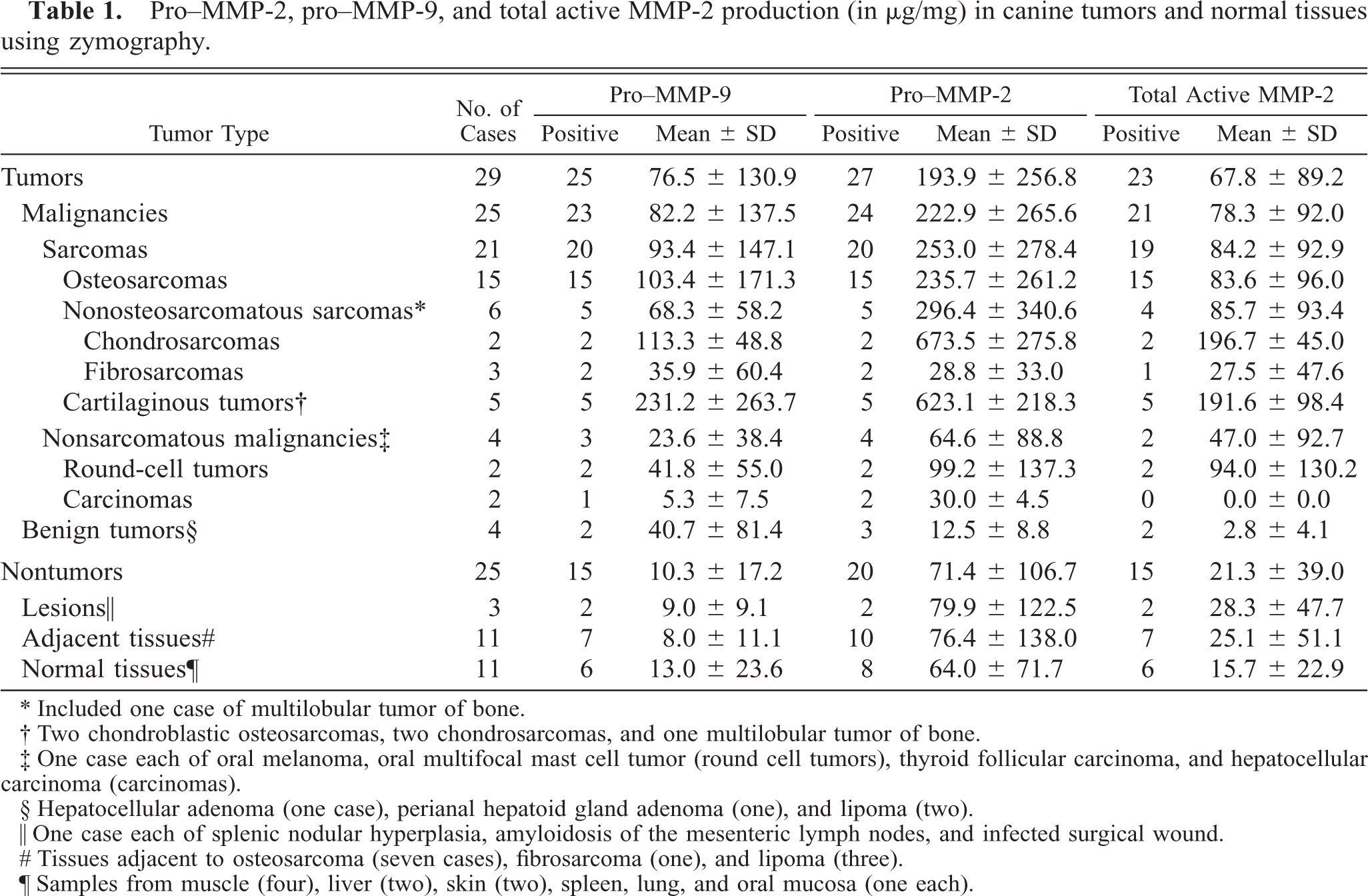


∗ Included one case of multilobular tumor of bone.
† Two chondroblastic osteosarcomas, two chondrosarcomas, and one multilobular tumor of bone.
‡ One case each of oral melanoma oral multifocal mast cell tumor (round cell tumors) thyroid follicular carcinoma and hepatocellular carcinoma (carcinomas).
§ Hepatocellular adenoma (one case), perianal hepatoid gland adenoma (one), and lipoma (two).
∥ One case each of splenic nodular hyperplasia, amyloidosis of the mesenteric lymph nodes, and infected surgical wound.
# Tissues adjacent to osteosarcoma (seven cases), fibrosarcoma (one), and lipoma (three).
¶ Samples from muscle (four), liver (two), skin (two), spleen, lung, and oral mucosa (one each).
Osteosarcomas were classified into histologic subtypes, 58 and their mitotic index, degree of nuclear pleomorphism, and tumor necrosis were assessed to classify them into grades, 62 as described previously. The mitotic index (number of mitoses per 10 fields at 400×) was estimated and scored in the following way: 1–10 mitoses = score 1, 11–20 = score 2, 21–30 = score 3, 31 or more = score 4. The degree of tumor necrosis was scored in the following way: 0% = score 0, 1–15% = score 1, 16–50% = score 2, 51–100% = score 3. The degree of nuclear pleomorphism was estimated as mild, moderate, or marked (score 1, 2, or 3). The three scores obtained were added to calculate the “histologic score” (HS) of the tumors (2–10), which was then used to classify the tumors into grades in the following way: HS 1–5 = grade I, HS 6–8 = grade II, and HS 9 or 10 = grade III.
Gelatin zymography
Gelatin zymography was used to detect precursor and active forms of MMP-2 and −9 in fresh, frozen tissues.
Tissues were weighed and homogenized, and MMPs were extracted from the tissues as described previously. 21 , 72 Samples were diluted 1 : 25 in electrophoresis buffer (25 mM Tris–HCl, 200 mM glycine, 0.1% sodium dodecyl sulfate [SDS]), mixed with an equal volume of sample buffer (50 mM Tris–HCl, 2% SDS, 20% glycerol, 0.01% bromophenol blue), and 5 µl aliquots were electrophoresed for 90 minutes with 40 mA current at 10 C on nonreducing 7.5–15% gradient polyacrylamide gels containing 0.1% gelatin as described previously. 48 After 90 minutes, gels were washed in 2.5% Triton X-100 (Sigma-Aldrich Ltd., Sydney, Australia) for 2 × 30 minutes, then incubated for 18 hours at 37 C in incubation buffer: 50 mM Tris–HCl, 5 mM calcium chloride, and 0.02% sodium azide. Gels were then stained for 30 minutes with Coomassie blue G-250, destained with 5% acetic acid and 2% glycerol in water (24 hours), dried using a gel dryer, and scanned using an HP Scanjet 3 C (Hewlett–Packard, Palo Alto, CA) into densitometry analysis software (GelPro; Media Cybernetics, Silver Spring, MD). Standard curves were constructed using recombinant human MMP-2 and MMP-9 standards (Oncogene Research Products, Cambridge, Massachusetts, USA).
Immunohistochemistry
A protocol for immunohistochemistry was developed and optimized for the demonstration of MMP-2, MMP-9, and p53 protein in canine tissues. The same series of tumors was used and a small number of additional archival cases included.
Two human ovarian carcinomas known to be p53 positive from previous studies 17 , 69 were used as positive controls. A canine osteosarcoma shown to produce high levels of both MMP-2 and MMP-9 in zymography was used as positive control for MMPs. For negative controls, the primary antibody was replaced with Tris-buffered saline (TBS), pH 7.6. Nontumorous areas of the positive control slides served as internal negative tissue controls for p53.
Sections were deparaffinized in 100% xylene (3 × 2 minutes) and rehydrated through 100% ethanol (3 × 2 minutes). Sections were then subjected to heat antigen retrieval by microwaving in preheated citrate buffer (pH 6.0) at 95 C for 10 minutes. Sections were allowed to cool for 20 minutes and rinsed with TBS. Sections were treated for 15 minutes with 3% (vol/vol) H2O2 in distilled water to inhibit endogenous peroxidase activity. Nonspecific antibody binding was reduced by incubating the sections with 4% commercial nonfat skim milk powder in TBS for 25 minutes. Sections were then incubated with primary antibody overnight at 4 C. Antibodies used were the polyclonal rabbit anti-human antibody CM-1 (Signet Laboratories, Dedham, Massachussetts, USA), which recognizes both wild and mutant types of p53 protein; the monoclonal mouse antibodies CA-4001, which recognize the 72- and 62-kd forms of MMP-2; A-Gel VC2, which recognizes the 72- and 66-kd forms of MMP-2; VIIC2, which recognizes a 95-kd protein identified as MMP-9 (all Neomarkers, Fremont, California, USA); and the polyclonal mouse anti-human antibodies to MMP-2 (The Binding Site, Birmingham, UK) and MMP-9 (Biogenesis, Poole, UK). The sections were incubated with a sheep anti-rabbit or -mouse biotin–labeled secondary antibody and then a streptavidin–biotin–peroxidase complex (all DAKO, Carpinteria, CA, USA) at room temperature. After washing, 3,3-diaminobenzidine tetrahydrochloride (Sigma) was used as chromogen with H2O2 as substrate. The sections were counterstained lightly with Mayer's haematoxylin (Sigma), dehydrated, cleared in xylene, and mounted on cover slips.
All sections were evaluated independently by two observers (P.L., B.A.M.), without knowledge of the clinical information. Only those tumors containing cells with a distinct nuclear staining for p53 were considered to be positive. The fraction of positively stained nuclei for p53 was scored in the following way: 0% = score 0, 1–10% = score 1, 11–25% = score 2, 26–50% = score 3, 51–100% = score 4. The fraction of positively stained cells for MMPs was noted but not scored. Cell-staining patterns, regional variation, staining specificity, and agreement between multiple tumor sections were also noted.
Statistical analysis
Levels of the different MMPs in their precursor or active forms were related to clinicopathologic parameters and compared between tumor and control groups. To assess the statistical significance of intergroup differences in quantitative data, the Kruskal-Wallis one-way analysis of variance on ranks, the Mann-Whitney rank sum test, or the unpaired t-test were performed.
Additionally, sensitivity and specificity of each MMP were calculated to provide an indicator of the ability of each MMP to discriminate between malignancies and normal tissues. 5 The likelihood ratio for a positive test result (i.e., diagnosis of malignancy) was determined as a fraction of the true positives divided by the fraction of false positives [sensitivity/(1 − specificity)]. 70 Receiver operating characteristic (ROC) curves were then constructed by plotting the probability of true positive (sensitivity) on the y-axis against the probability of false positive (1 − specificity) on the x-axis over the full range of test values. Each point on the curve represented the 2 × 2 table decision matrix at a cut-off point, which corresponds to a particular test value. The optimal cut-off level for each MMP, to discriminate between malignant and normal tissues, was determined on the basis of the ROC curves as the point on the curve closest to the top left-hand corner of the graph, which shows the optimal combination for specificity and sensitivity and where diagnostic accuracy is maximized. This level was used to group the raw MMP values to positive and negative. Data grouped in contingency tables were then analyzed statistically using the Fisher exact test, Student's t-test, or chi-square test for comparison of proportions. Statistical significance was set at 0.05. The data were analyzed using the Jandel Sigmastat® Statistical Software (version 2.0, Jandel Corporation, USA).
Results
Zymography
MMPs were present in almost all tissues examined. The levels were mostly low in normal tissues and much higher in malignancies, particularly sarcomas. Malignancies also produced a wider spectrum of MMPs compared with normal or benign tissues. The gelatinolytic pattern of the samples displayed bands of 92, 82, 72, 66, and 62 kd (Fig. 1). MMP-2 was detected in its precursor (72 kd, pro–MMP-2) and active forms (66 and 62 kd). MMP-9 was detected in its precursor form (92 kd, pro–MMP-9) and its active form (82 kd).
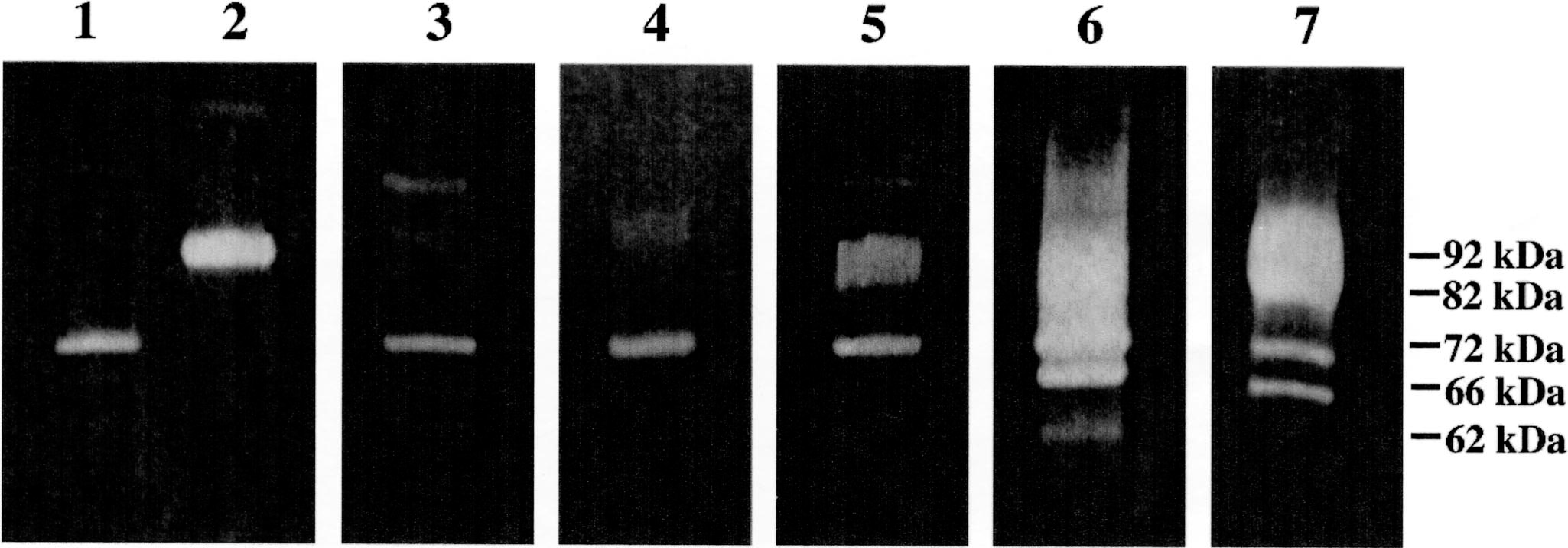
Zymography of canine tumors and normal tissues. Gradient (7–15%) polyacrylamide gel containing 0.1% gelatin. Lane 1 = pure human pro–MMP-2 (72 kd); lane 2 = Pure human pro–MMP-9 (92 kd); lane 3 = normal canine skin; lane 4 = lipoma; lane 5 = skin over osteosarcoma A; lane 6 = osteosarcoma A, p53 positive; lane 7 = osteosarcoma B, p53 negative. The molecular masses of detected MMPs are indicated. The presence of large amounts of MMPs in certain samples resulted in lanes heavily overloaded with MMPs, in which a long smear was apparent in the lanes, such as the ones shown here in lanes 6 and 7, and, to a lesser degree, lane 5. Gelatin zymography was repeated in these samples, after suitable dilution, to allow the production of clear bands and the subsequent densitometric quantification of MMPs
Pro–MMP-2 was detected in 27 of 29 neoplasias (93.1%) as well as in 8 of 11 normal tissue controls (73.2%) (Table 1).
Active MMP-2 was detected in 23 of 29 neoplasias (79.3%) (Table 1). The 66-kd form of active MMP-2 was detected in 21 of 29 neoplasias (72.4%), including 18 of 21 sarcomas, a mast cell tumor, and two adenomas. Normal tissue samples from the skin and muscle contained this MMP, whereas carcinomas and samples from the oral mucosa, spleen, liver, and lung did not.
Zymographic detection of the smaller (62 kd) form of active MMP-2 was linked with the malignant phenotype. Only high-grade (II or III), p53 positive, metastatic malignancies produced this MMP form, including a case each of soft tissue fibrosarcoma and oral multifocal mast cell tumor, an osteosarcoma lung metastasis, and two osteosarcoma bone metastases.
Pro–MMP-9 was detected in 6 of 11 (54.5%) normal tissue controls (liver, oral mucosa, skin, and muscle) and 25 of 29 neoplasias (86.2%), including 20 of 21 sarcomas (95.2%), 3 of 4 nonsarcomatous malignancies (75%), and 2 of 4 benign tumors (50%) (Table 1).
The active form of MMP-9 was detected in three cases, one nonmetastatic fibrosarcoma, one hepatocellular adenoma, and a normal lung sample.
Tumors had significantly higher total MMP (P < 0.01), pro–MMP-9, total MMP-2 (P < 0.05), and total active MMP-2, but not pro–MMP-2, levels than did nontumors (Table 2 and Figs. 2–4). Malignancies and sarcomas produced significantly higher total MMP, total pro-MMP, and individual MMP levels than nontumors and benign tumors (Table 2). Although MMP levels were not statistically different when sarcomas were compared with nonsarcomatous malignancies (Table 2) (P = 0.09 for total MMP), sarcomas produced significantly greater active MMP-2 (P < 0.05) when compared with carcinomas alone. Similar statistical correlations, as with sarcomas overall, were observed when osteosarcomas alone were compared with the above tumor or tissue groups (Table 2 and Figs. 2–4). MMP-2 production was higher in chondrosarcomas compared with fibrosarcomas (P < 0.05).
P values for pro-MMP-9, pro-MMP-2, and active MMP-2 resulting from the comparison of various tissue types. Characters in bold indicate statistical significance. Tissue groups in the parameter 1 column exhibit higher MMP values. Active MMP-2 values include both forms of active MMP-2.
∗ Excluding two chondroblastic osteosarcomas.
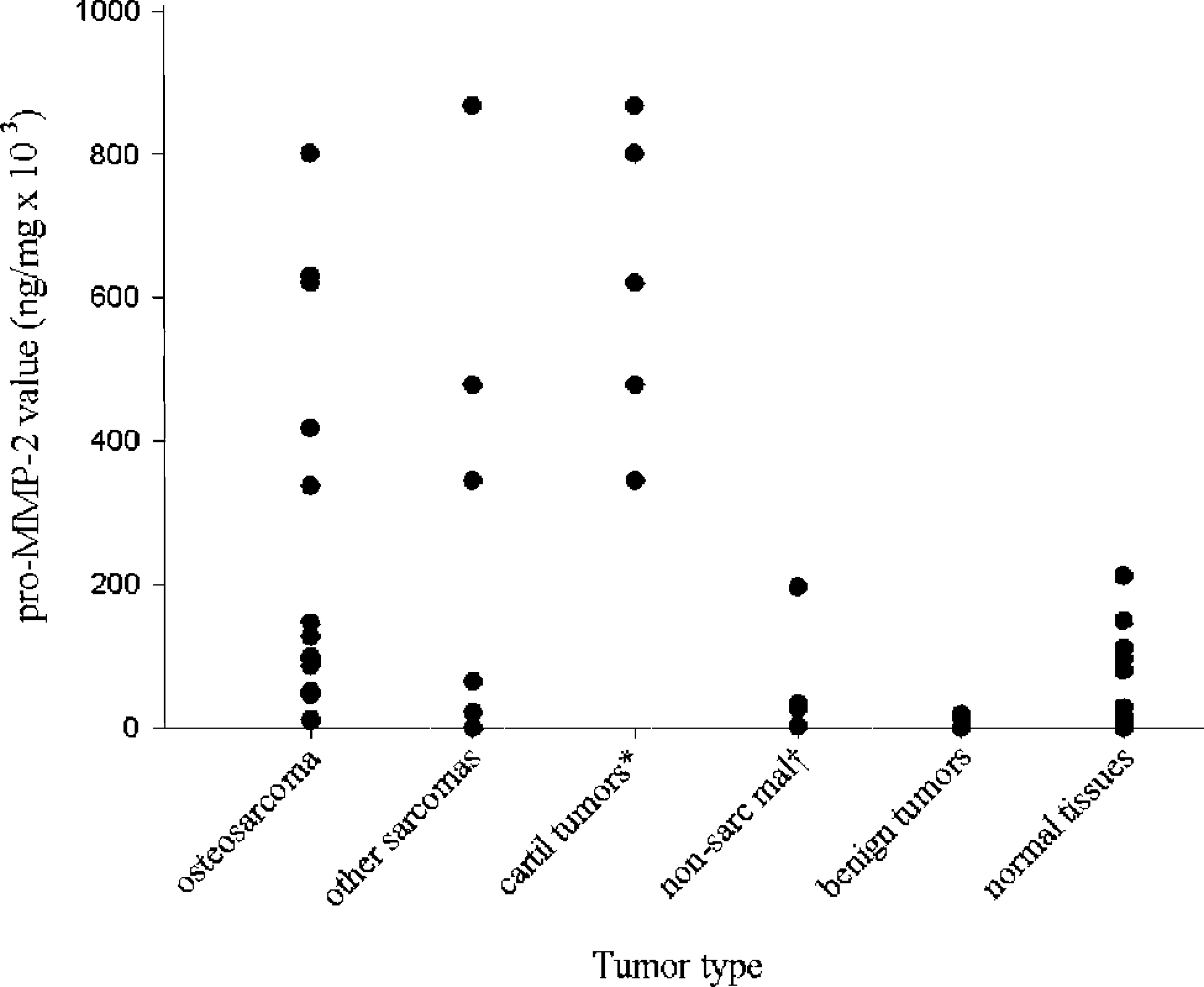
Distribution of pro–MMP-2 values in 29 canine tumors and normal tissues from 11 dogs. Pro–MMP-2 values were significantly greater for osteosarcomas compared with benign tumors and normal tissues; they were also significantly greater for cartilaginous tumors compared with osteosarcomas, nonsarcomatous malignancies, benign tumors, and normal tissues. ∗ cartilaginous tumors; † nonsarcomatous malignancies
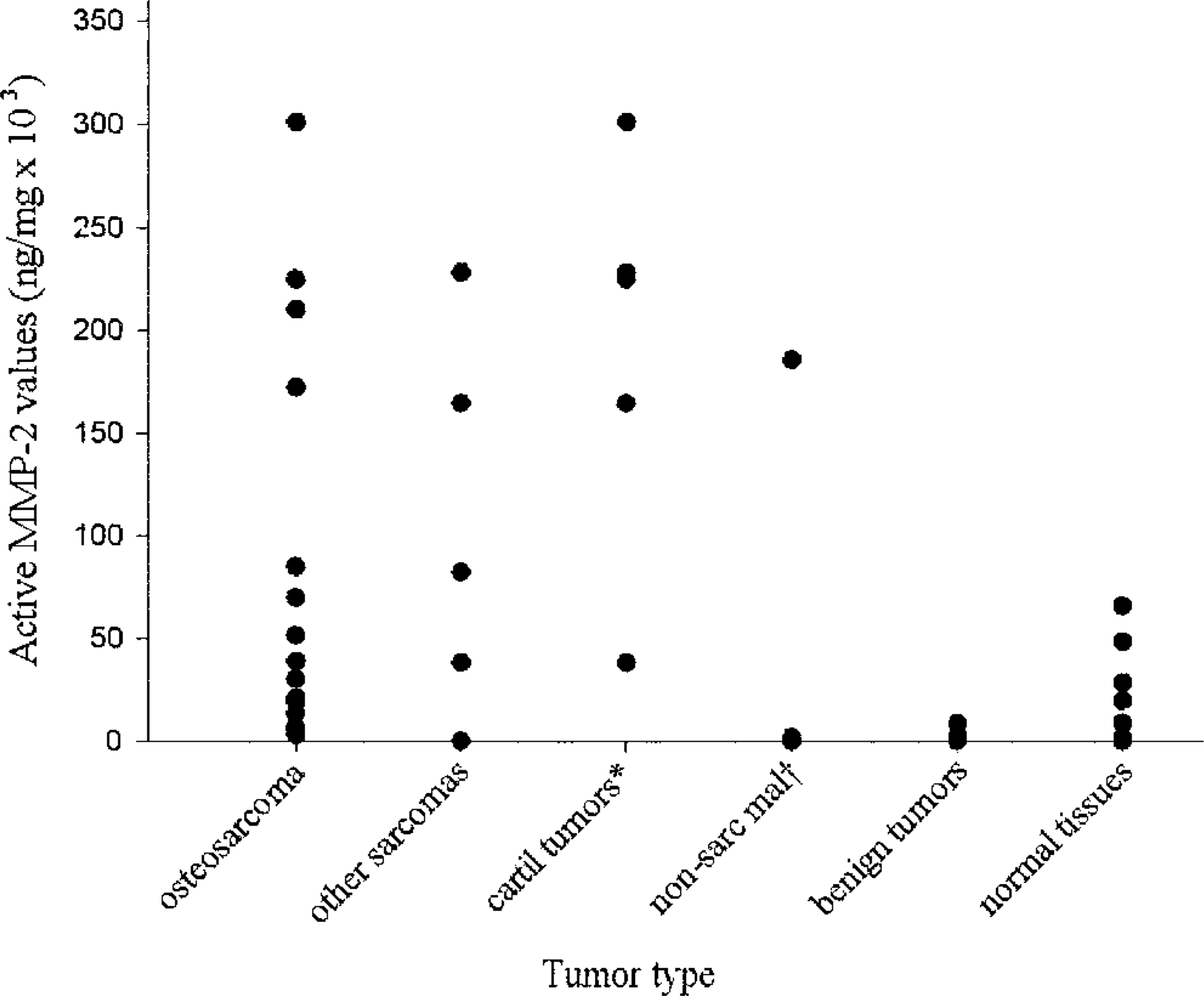
Distribution of active MMP-2 values in 29 canine tumors and normal tissues from 11 dogs. Active MMP-2 values were significantly greater for osteosarcomas compared with benign tumors and normal tissues; they were also significantly greater for cartilaginous tumors compared with osteosarcomas, benign tumors, and normal tissues. ∗ cartilaginous tumors; † nonsarcomatous malignancies
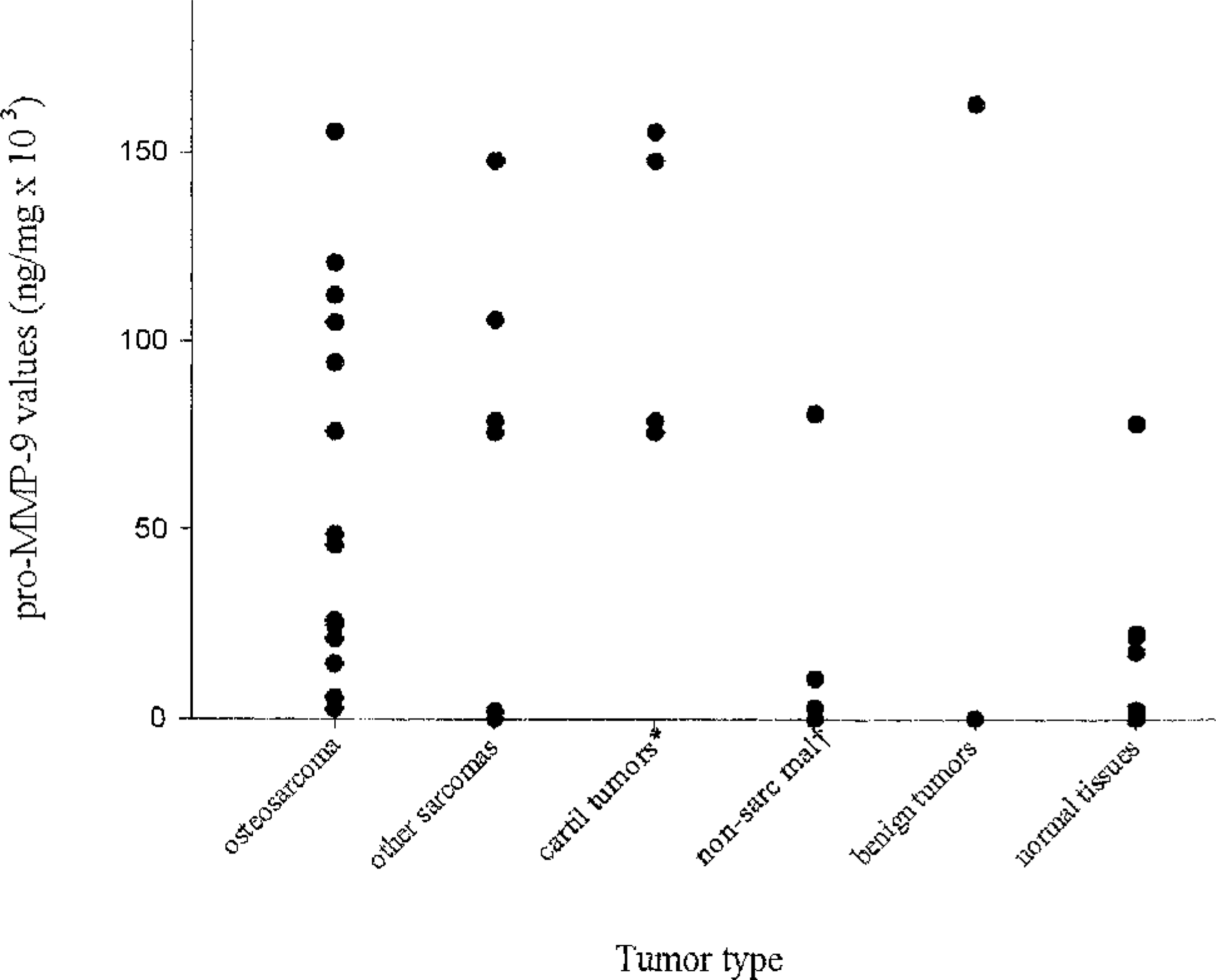
Distribution of pro–MMP-9 values in 29 canine tumors and normal tissues from 11 dogs. Pro–MMP-9 values were significantly greater for osteosarcomas compared with normal tissues; they were also significantly greater for cartilaginous tumors compared with nonsarcomatous malignancies, benign tumors, and normal tissues. ∗ cartilaginous tumors; † nonsarcomatous malignancies
Bone tumors containing abundant cartilage (two chondroblastic osteosarcomas, two chondrosarcomas, and one multilobular tumor of bone) produced significantly higher MMP levels than benign tumors and nontumors, pro–MMP-2 levels than nonsarcomatous malignancies (Table 2), and higher pro- and active MMP-2 and total MMP levels when compared with fibrosarcomas and osteosarcomas (with or without the two chondroblastic ones).
There were no significant differences for any individual MMPs or MMP groups when nonsarcomatous malignancies, round cell tumors, carcinomas, benign tumors, normal tissues, lesions, tissues adjacent to tumors, or nontumors overall were compared pairwise (Figs. 2–4).
MMPs were detected in a range of normal tissues including the liver, lung, muscle, lymph node, skin, bone marrow, and oral mucosa; among these, the lung was the most productive tissue. No MMPs were detected from the splenic samples. The MMP levels in the normal tissues were generally much lower than the levels produced by the tumors.
The active MMP-2/pro–MMP-2 ratio value was linked to the tumor type; it was, in descending order, higher in osteosarcomas (0.41), malignancies (0.40), neoplasias (malignant or benign) (0.36), tissues adjacent to tumors (0.31), normal tissues (0.26), lesions (0.15), and benign tumors (0.14).
Pro–MMP-9 production correlated with the histologic grade of osteosarcomas; total and pro–MMP-9 values were significantly greater for grade III compared with grade I (P < 0.05), grade II compared with grade I (P < 0.01), and high grade (II or III) compared with low grade (grade I) (P < 0.05) osteosarcomas. Pro–MMP-2 and total MMP-2 values were also markedly, but not significantly, higher for grade III compared with grade I or II osteosarcomas (P = 0.07 and P = 0.08, respectively) (Fig. 5). Only high-grade osteosarcomas produced the 62-kd MMP.
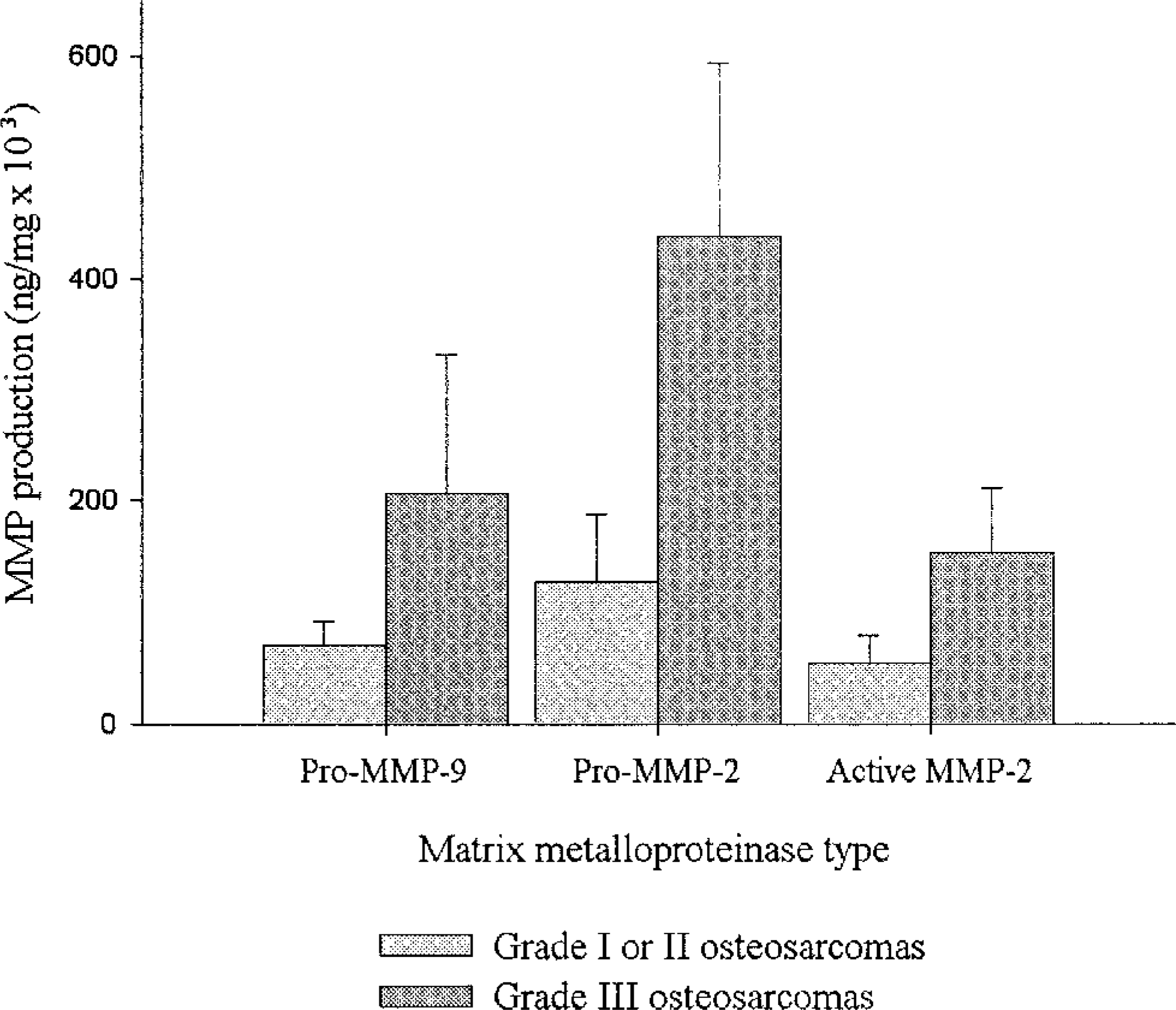
MMP production in relation to tumor grade in osteosarcomas. Grade III osteosarcomas produced very high levels of MMPs, as assessed by zymography
Only metastatic malignancies (3 of 7) produced the 62-kd form of active MMP-2. All metastatic osteosarcomas produced active MMP-2 (66 or 62 kd). Metastatic malignancies produced, on average, approximately five times higher levels of pro–MMP-9 and 1.6 times higher levels of pro–MMP-2 compared with nonmetastatic malignancies; these differences were not, however, statistically significant (Fig. 6).
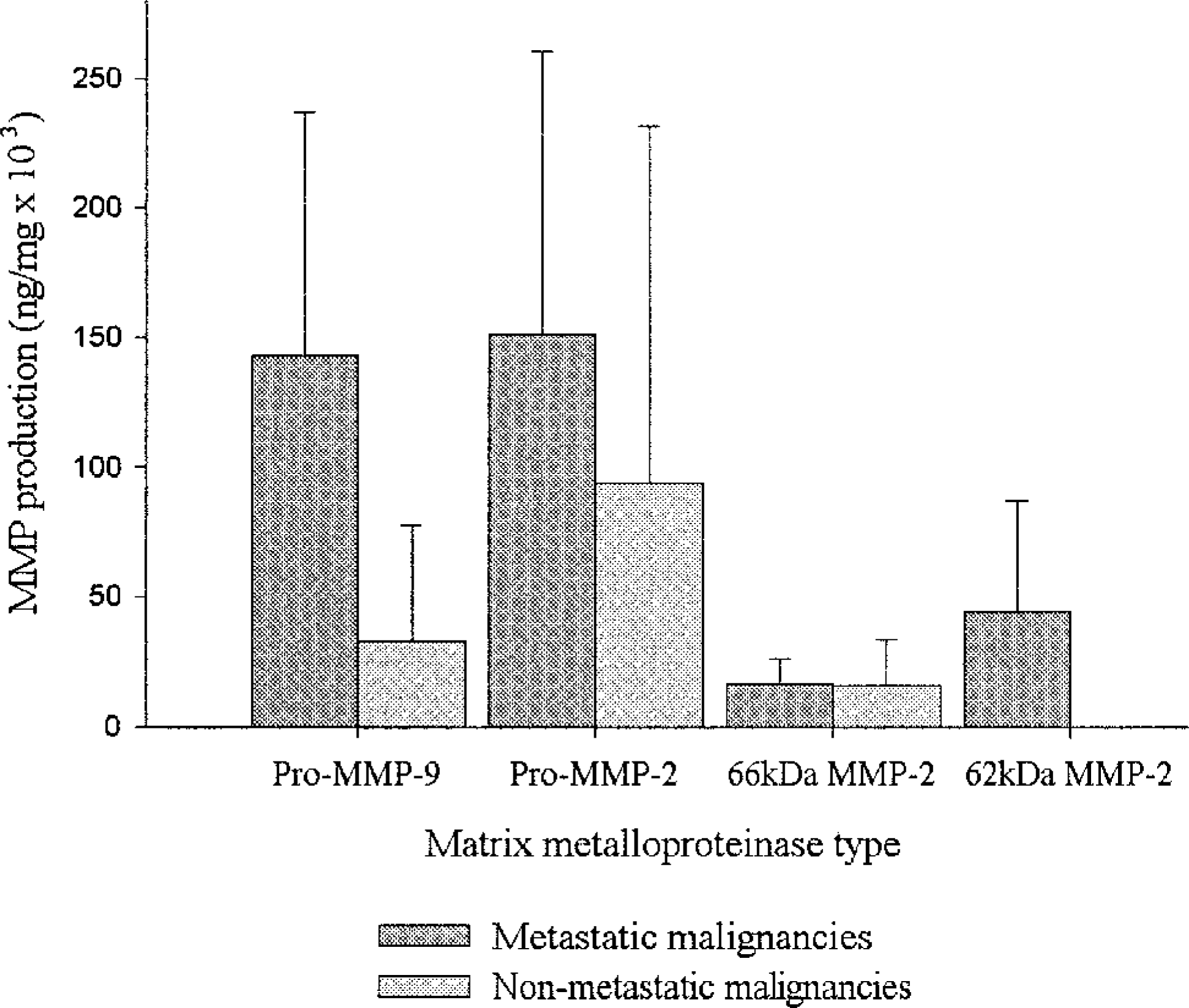
MMP production in relation to metastatic status. Metastatic malignancies produced very high levels of pro–MMP-9 and pro–MMP-2, as assessed by zymography. Only metastatic malignancies produced the 62-kd form of active MMP-2
Sarcomas that showed p53 overexpression (n = 8) produced markedly higher levels of pro–MMP-9 (three-fold) and pro–MMP-2 than did p53 negative sarcomas (n = 6) (Figs. 1 and 7). However, these differences were not statistically different (P = 0.08). p53-positive cases with a fraction of positive nuclei (FPN) of 3 or 4 produced, on average, five-fold higher pro–MMP-9 levels than p53-negative cases and two-fold higher total pro-MMP levels than cases with an FPN = 2 (data not shown). Only p53-positive sarcomas produced the 62-kd form of active MMP-2.
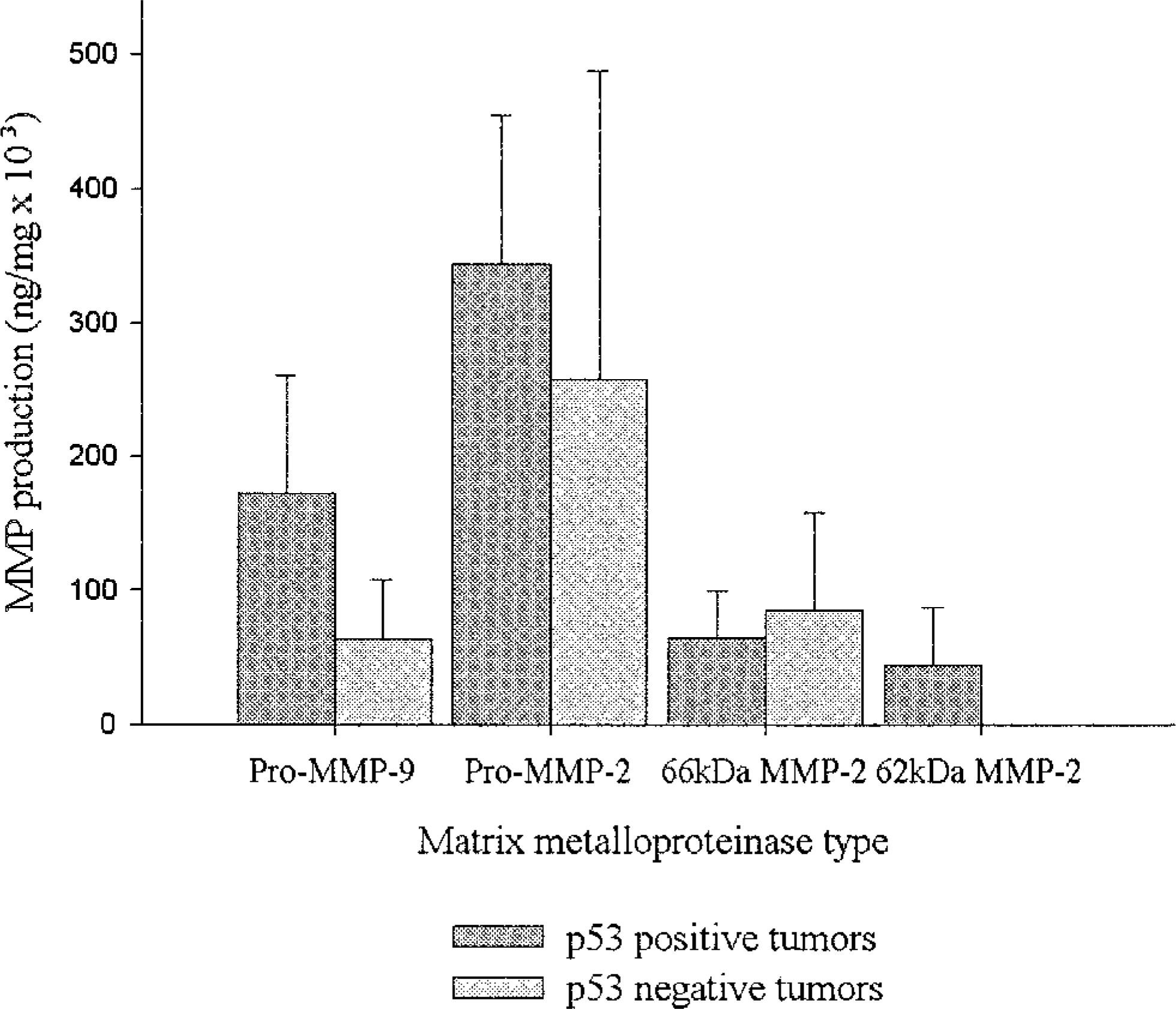
MMP production in relation to p53 status in sarcomas, as assessed by zymography. p53-positive cases produced higher levels of pro–MMP-9 and pro–MMP-2 than p53-negative cases; however, these differences were not statistically different (rank sum test, P = 0.08). Only p53-positive sarcomas produced the 62-kd form of active MMP-2
Samples were analyzed from both the advancing edge and the center of seven malignancies. MMP production was higher, to a variable degree, in the tumor margin compared with the central part of the tumors in five of seven cases. Similarly, MMP production was higher in six of seven osteosarcomas compared with samples taken from tissues immediately surrounding the respective tumors, whereas they were lower in one fibrosarcoma and two lipomas that were also examined.
ROC curves were constructed and the optimal cut-off level was determined for each MMP (Fig. 8). The values of 1,000, 20,000, and 1,000 ng/mg tissue were chosen as the optimal cut-off levels for pro–MMP-9, pro–MMP-2, and active MMP-2, respectively. Using these values, the ability to discriminate between malignant and normal tissues was found to be very good. The specificity and sensitivity of this test were determined as 82.1% and 92% for pro–MMP-9, 77.7% and 84% for pro–MMP-2, and 77.7% and 84% for active MMP-2. Specificity of 100% was guaranteed when the 80,000, 220,000, and 70,000 ng/mg were chosen as the cut-off levels for pro–MMP-9, pro–MMP-2, and active MMP-2, respectively, i.e., all cases that produced an MMP value above these levels were malignant; sensitivity, however, decreased proportionately.
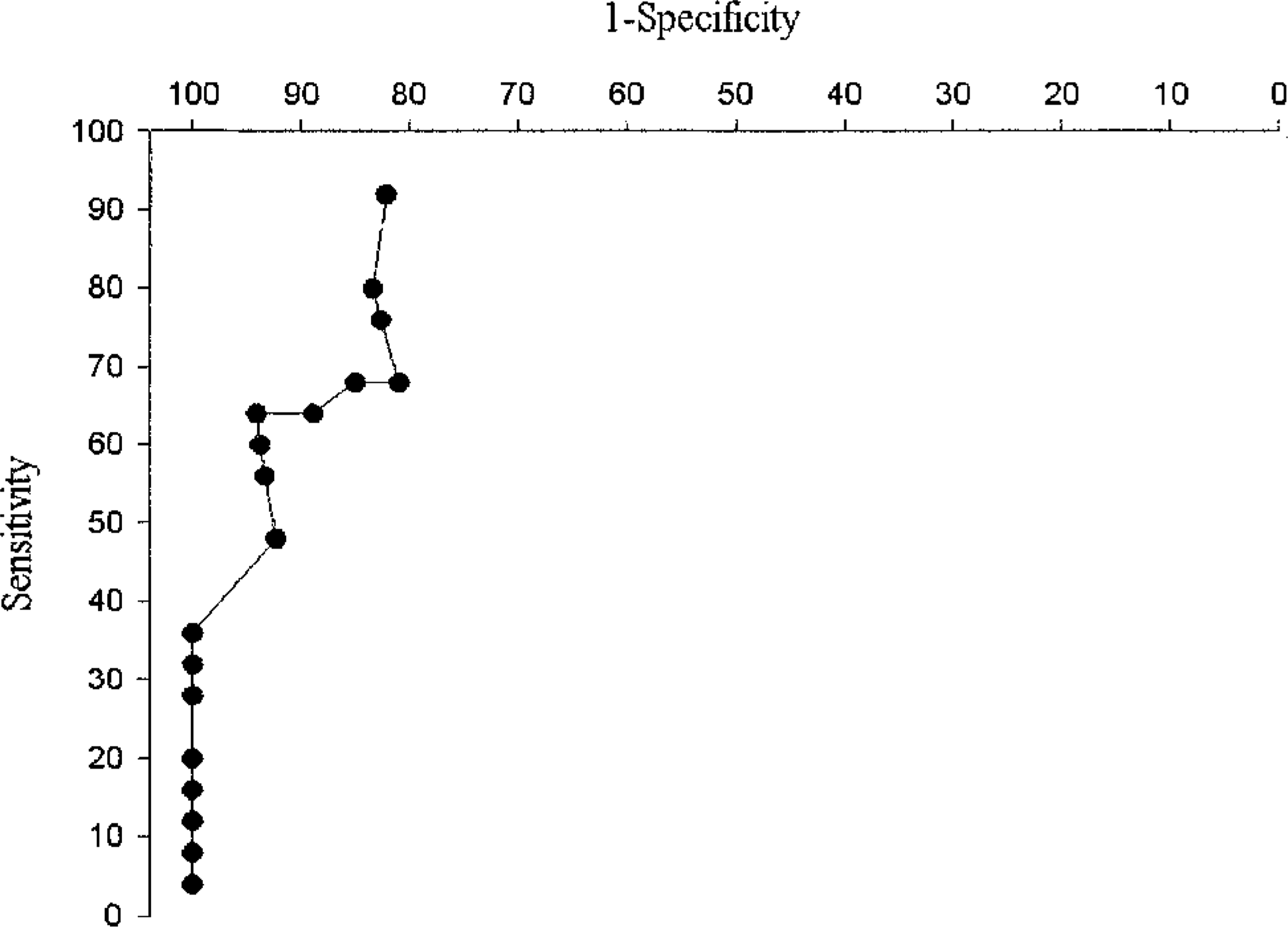
ROC curve for pro–MMP-9. Similar ROC curves were constructed for each MMP after zymographic analysis of MMP presence in canine tissues. Sensitivity and specificity of each MMP were calculated to determine the ability of each MMP to discriminate between malignancies and normal tissues. Each point on the curve corresponds to a particular test value. The cut-off level for a positive test result (i.e., diagnosis of malignancy) was determined on the basis of the optimal combination of specificity and sensitivity, which, in the case of pro–MMP-9, was the value of 23,000 ng/mg
Immunohistochemistry
The immunohistochemical expression of MMP-2 and MMP-9 was evaluated in the same series of tumors (Figs. 9–11). Tumor cells were found to be more immunoreactive to MMPs compared with normal cell populations of surrounding tissues or normal tissues and appeared to be the main source of MMP-2 and MMP-9 in neoplasias. The contribution of stromal cells to the production of MMPs, in general, was minimal or absent. The degree of immunoreactivity showed no correlation with tumor type or grade.
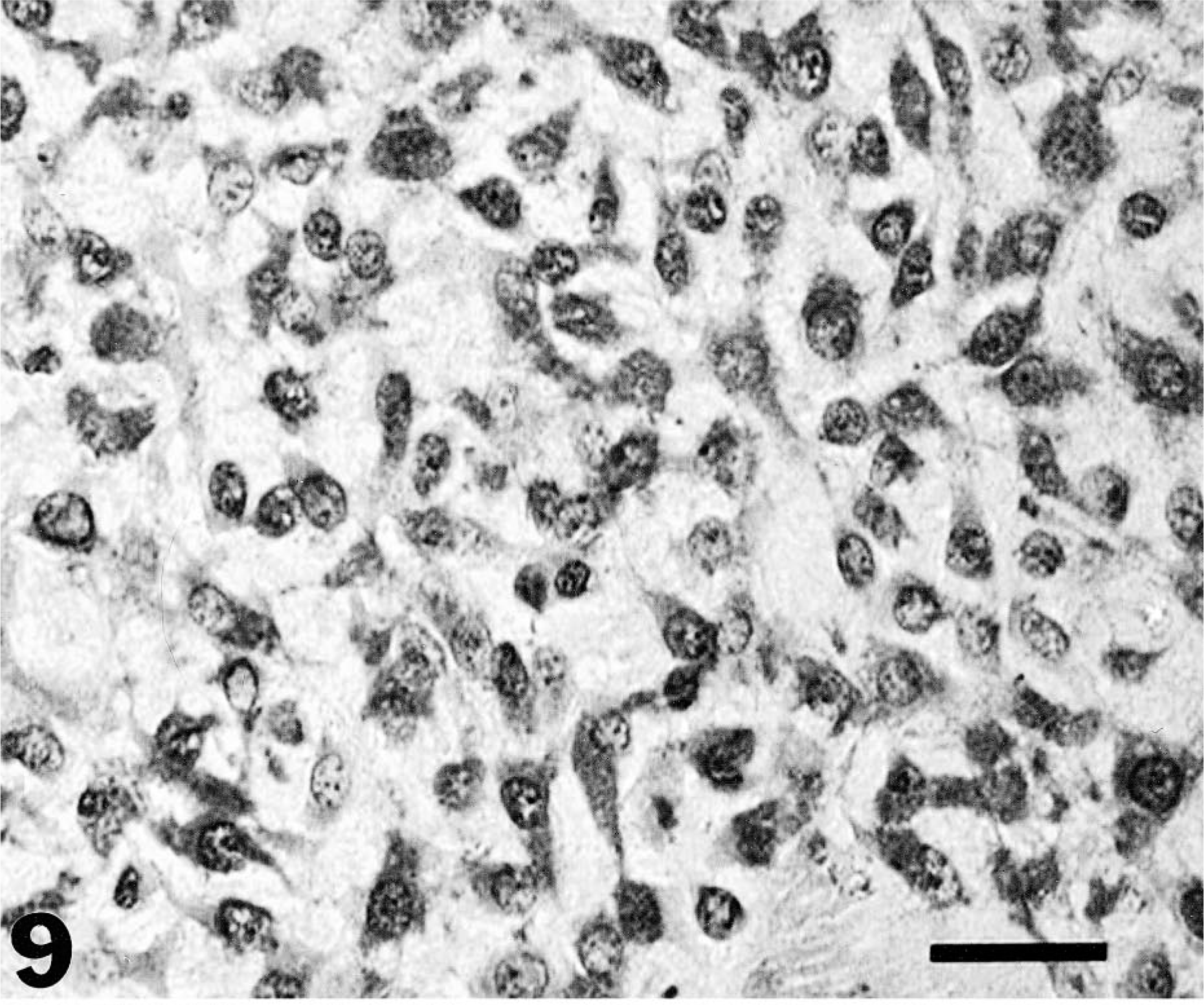
Canine myxoid chondrosarcoma of the mandible. Immunohistochemical localization of MMP-2 protein: note the intense and specific cytoplasmic staining of most tumor cells and the absence of nuclear staining. Streptavidin–biotin–peroxidase, Mayer's hematoxylin counterstain. Bar = 30 µm.
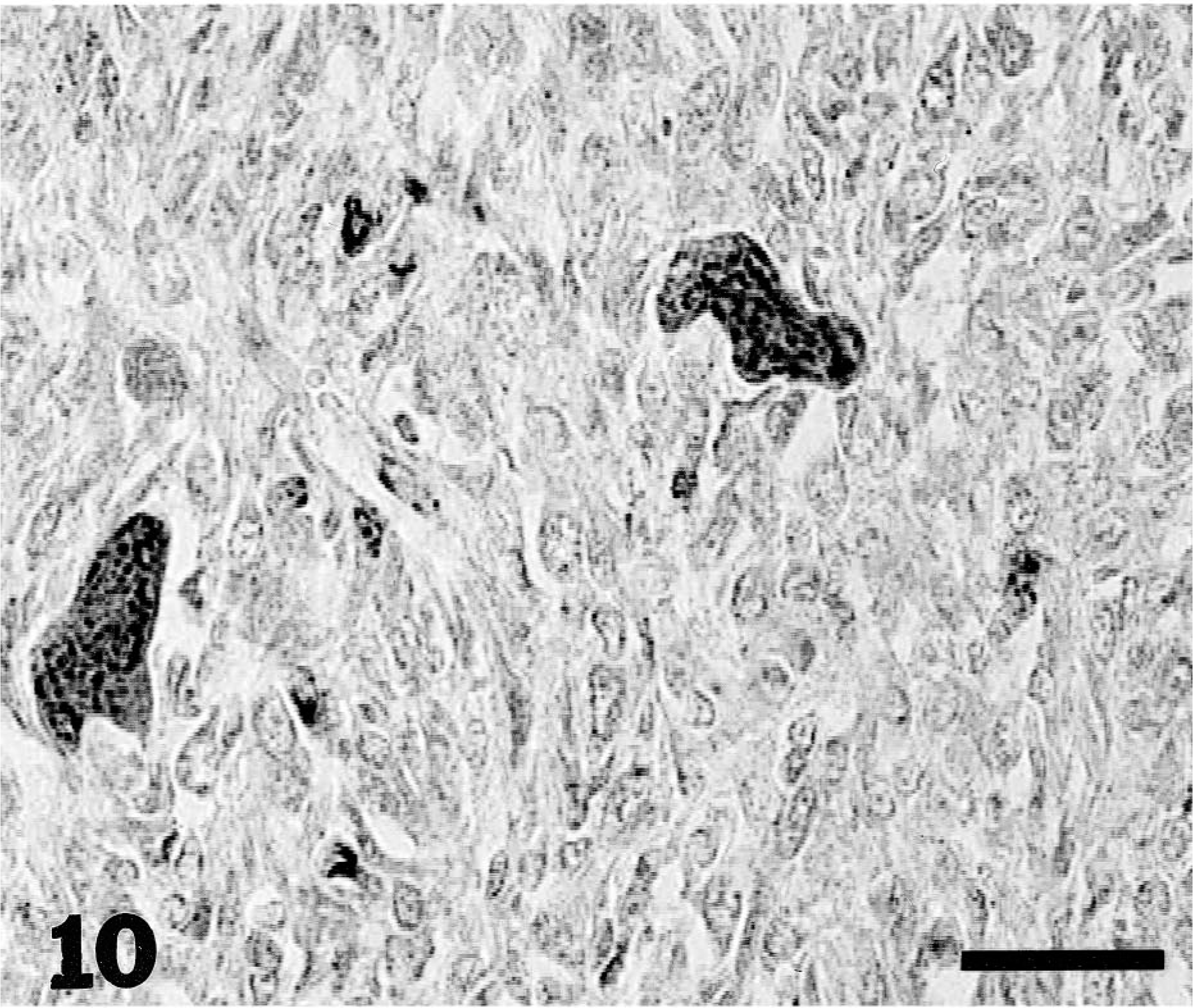
Canine osteosarcoma of the proximal humerus. Immunohistochemical localization of MMP-9 protein: note intense cytoplasmic staining of osteoclasts and a small number of tumor cells. Streptavidin–biotin–peroxidase, Mayer's hematoxylin counterstain. Bar = 30 µm.
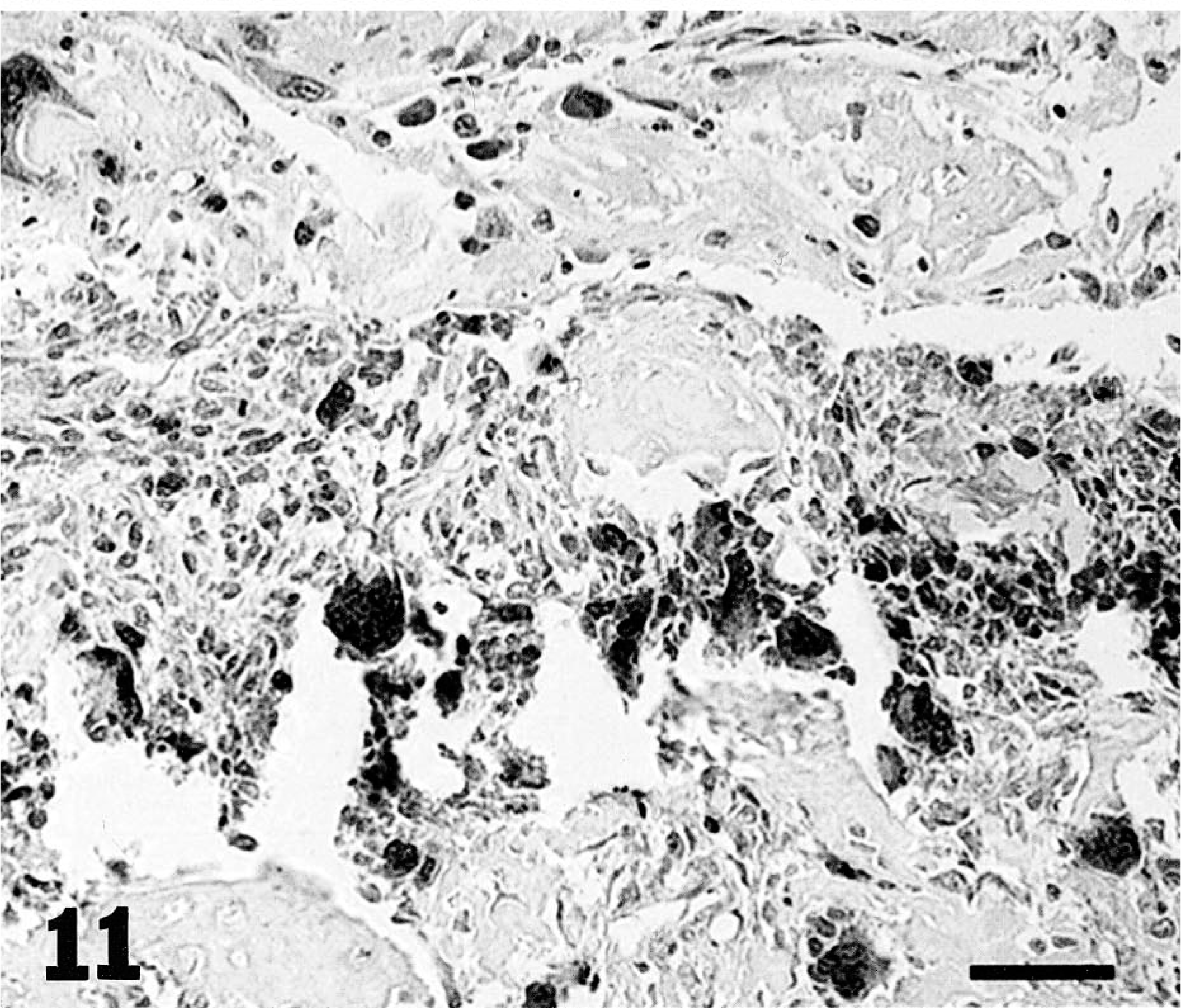
Canine osteosarcoma. Immunohistochemical localization of MMP-9 protein: note intense cytoplasmic staining of most osteoclasts and of a considerable number of tumor cells. Streptavidin–biotin–peroxidase, Mayer's hematoxylin counterstain. Bar = 60 µm
Among the tumors examined (Table 3), 9 of 27 (33.3%) were positive for MMP-2 and 9 of 28 (32.1%) were positive for MMP-9 with the use of the polyclonal antibodies. None were positive with the use of the monoclonal A-Gel VC2 (MMP-2) and VIIC2 (MMP-9) antibodies, whereas only one chondrosarcoma was positive for MMP-2 with the use of the CA4001 antibody.
Immunohistochemistry results for MMP-2 and MMP-9 in canine tumors and normal tissues.
∗ One case each of fibrosarcoma, multilobular tumor of bone, hepatocellular carcinoma, melanoma, mast cell tumor, lipoma, amyloidosis, and samples from infected surgical wound, spleen, kidney (one each), and muscle (two) were negative for both MMP-2 and MMP-9 by immunohistochemistry.
In all positive cases, for both MMPs, immunoreactivity was diffusely located in the cytoplasm or, less frequently, the extracellular matrix. The cells that showed immunoreactivity were mainly malignant osteoblast-like cells and neoplastic chondroblasts within the cartilaginous lobules. Lymphocytes, alveolar macrophages, bronchial lining cells probably representing intraepithelial lymphocytes, granulocytic cells in the bone marrow, keratinocytes, and osteoclasts also stained moderately to intensely. In a small number of cases, osteoid and chondroid produced by malignant cells showed weak to moderately intense staining for both MMP-2 and MMP-9.
Discussion
In the present study, the presence of MMPs is demonstrated in a number of canine tumors and normal tissues. Evidence is provided for the interrelation of MMP levels and profile with the tumor type and clinicopathologic parameters such as the tumor grade.
MMPs were detected in all but a few of the tissues examined. MMP-2 (gelatinase A) was detected in its precursor (72 kd) and active forms (66 and 62 kd), and MMP-9 (gelatinase B) was detected in its precursor form (92 kd) and its active form (82 kd). Coexpression of the MMPs was noted, suggesting their coregulation. However, all three cases that expressed the active form of MMP-9 did not produce any active forms of MMP-2.
Most tumors produced very high levels of pro–MMP-9, pro–MMP-2, and one or both forms of active MMP-2. The fact that the active form of MMP-9 (82 kd) was only present in three cases indicates that it does not play a major role within the primary tumor or established metastasis environment. Although the possibility of it having a role in other stages of the metastatic cascade cannot be excluded, active MMP-9 appears to be of little prognostic or pathogenetic value. In contrast, zymographic detection of the smaller (62 kd) form of active MMP-2 was invariably linked with malignancy. Only high-grade, p53 positive, metastatic malignancies produced this form of active MMP-2.
MMP production levels and profile correlated with the tumor type and biologic behavior. Tumors produced significantly higher levels of MMP-2 and MMP-9 than nontumors, malignancies significantly higher total MMP levels than benign tumors, and sarcomas significantly higher active MMP-2 than carcinomas.
Cartilaginous tumors of bone (including chondroblastic osteosarcomas, chondrosarcomas, and multilobular tumors of bone) produced significantly higher MMP-2 and MMP-9 levels than nonsarcomatous malignancies, benign tumors, and nontumors, including normal tissues, lesions, and tissues adjacent to tumors. MMPs may have a role but were clearly not significant in the small number of nonsarcomatous malignancies, benign tumors, and lesions that we examined.
One of the most interesting findings of the study was that the production of each MMP, with the exception of pro–MMP-9, was significantly greater in cartilaginous tumors even when they were compared with osteosarcomas and fibrosarcomas. Previous studies have reported that the expression of MMP-2 is enhanced to a greater degree in the cartilage of osteoarthritis than in normal cartilage 33 and that increased immunohistochemical expression of MMP-1 and MMP-9 and decreased expression of MMP-3 are associated with the malignant phenotype of the human cartilaginous tumors, 24 and the expression of MMP-2 was also found to play a crucial role in invasion in the high-grade components of human dedifferentiated chondrosarcoma. 52 Nowadays, there is also a tendency for high-grade human chondrosarcoma to be considered the most malignant bone tumor because of its resistance to chemotherapy and consistently poor prognosis compared with osteosarcoma and Ewing's sarcoma, whose prognoses have been considerably improved recently. 24 Canine osteosarcomas, however, are considered to be more aggressive than chondrosarcomas, and chondroblastic osteosarcomas are generally not seen as a distinct, more malignant clinicopathologic entity. Our findings may suggest that high metalloproteinase activity within chondrocytes, malignant chondroblasts, and chondroid-producing areas is not exclusively associated with malignancy but may in part be attributed to the increased rate of matrix turnover, tissue remodeling and ossification within cartilaginous tumors, and the apparently central role MMPs play in these processes.
We detected MMPs in a range of normal canine tissues, although their levels were generally much lower and the spectrum more limited than the ones produced by the tumors. The lung was the most productive tissue among them, the alveolar macrophages being the presumed primary source of MMPs. Absence of MMPs from the splenic and some liver samples may be explained by the destruction of MMPs by other peptidases related to digestion or possibly suggests that the rate of synthesis is very low in these tissues. Lymphocytes, alveolar macrophages, and keratinocytes showed increased immunoreactivity in normal tissues, whereas osteoclasts, malignant osteoblast-like cells, and neoplastic chondroblasts were the most immunoreactive cells in the tumor sections examined. Although MMP expression by osteoclasts has been well documented in previous studies, 43 , 49 our immunohistochemical study demonstrates their involvement in MMP production in canine osteosarcoma and chondrosarcoma, which is in accord with their role in bone resorption 9 and matrix turnover.
Both in zymography and immunohistochemistry, we found that MMP production was higher in the advancing edge of canine malignancies compared with the tumor center, a fact that is in agreement with the suggested role of MMPs in human cancer invasion. MMP production was also higher in osteosarcomas compared with tissues immediately surrounding them; however, in many cases, the host tissue immediately surrounding the tumor did produce MMPs at varying levels, supporting the notion that tumor cells may facilitate invasion by inducing MMP production or activation in host tissues.
MMP-9 production correlated with the histologic grade of osteosarcomas. Histologic grading of osteosarcomas is of known prognostic significance 25 and is routinely used to make decisions on the clinical management of the tumors, 62 particularly with regard to chemotherapy protocols. 3 , 63 , 64 MMP-9 production therefore may be useful as an additional indicator to those traditionally used in the prognostic evaluation of osteosarcoma cases.
Despite the fact that all metastatic osteosarcomas produced active MMP-2 and that only metastatic malignancies produced the 62-kd form of active MMP-2, the presence of metastases did not generally appear to influence MMP production, presumably because only a small number of metastatic neoplasias were examined. Future studies may prove the contrary, particularly with regard to active MMP-2.
The tumor suppressor gene p53 is a negative regulator of tumor angiogenesis, 7 a process paramount for malignant growth and metastasis and in which MMPs play an integral part. 34 , 36 In our study, a small number of cases was examined for p53 expression using immunohistochemistry, which only allows the detection of abnormal p53 product. Again, although there tended to be a clear link between MMP production and p53 overexpression, future analysis of a sufficient number of cases will allow firm conclusions to be drawn.
The use of ROC curves after zymographic analysis of MMP presence in canine tissues provides an accurate and adequately specific and sensitive test with very good discriminatory power between malignant and normal tissues. The cut-off level for every MMP can be determined based on the optimal or desired combination of specificity and sensitivity. The development of a clinical test based on the above, in conjunction with other established or proposed prognostic and diagnostic indicators, may potentially be of great importance in practice; it may also be used, for example, to determine or confirm whether a bone biopsy was successful in obtaining tumor material or, if guided, whether the biopsied lesion is tumorous or inflammatory.
It has become increasingly apparent that MMPs are integral to the behavior of many tumor types in humans. Previous studies on human tissue 16 have revealed an association between gelatinase expression and malignant behavior, but in many cases, it is an increase in the level of active MMP-2 that correlates with the invasive phenotype to a greater degree than total MMP-2 expression. For that reason, we examined the active MMP-2/total MMP-2 ratio value in our study and found it to be linked to the tumor type and indicative of malignant behavior because with more malignant behavior of the tumor type, the ratio was reliably higher. However, pro–MMP-2 and pro–MMP-9 levels as well as total gelatinase levels were equally indicative of malignant behavior.
In keeping with the findings of our study, Lana et al. 28 also found that MMP-2 and MMP-9 were produced in canine osteosarcoma in levels higher than in unaffected stromal tissue. However, no other tumor types or normal tissues were examined in that study, the specific MMP profile of osteosarcomas was not analyzed, MMP levels were expressed in arbitrary units, and the results were not examined in relation to clinicopathologic parameters or compared with immunohistochemical results.
Given the potential importance of MMPs in pathogenesis and prognosis, MMP involvement has been otherwise reported in a strikingly limited number of canine conditions, including rheumatoid arthritis, 12 osteoarthritis, 11 dilated cardiomyopathy, 20 keratoconjunctivitis, 2 and cutaneous mast cell tumors. 31
We evaluated and compared immunohistochemistry and gelatin zymography as methods for accurately demonstrating MMP presence in tumor tissue. Immunohistochemistry provides one method for the assessment of the presence of MMPs; however, it is a relatively insensitive and qualitative method. On the contrary, the method of gelatin zymography proved to be both sensitive and quantitative but has the limitation of requiring fresh tissue. We would recommend zymography as the technique of choice, however, immunohistochemistry could be of some value in archival studies.
The use of immunohistochemistry allowed us to demonstrate the patterns of tissue distribution and cellular localization of MMPs in different canine tumors for the first time. Because MMPs are secreted in vivo and are not stored intracellularly in most cell types, it is usually difficult to demonstrate MMPs by immunohistochemical methods. Cancer cells appeared to be the main source of MMP-2 and MMP-9 in canine neoplasias.
Each of the antibodies used to detect MMPs recognizes the active or the precursor forms of the enzymes, although they have not been tested in canine tissues previously. MMP presence could not be demonstrated immunohistochemically in a large number of tumors that were shown to produce MMPs when zymography was used. This may be attributed to the possibly prolonged decalcification time of the predominantly osseous tumors that were examined, the usually prolonged fixation time, the use of 10% formalin for tissue fixation rather than a weaker formalin solution that may have altered the conformation of MMPs, or the relative lack of species cross-reactivity of the antibodies used. Furthermore, MMP immunoreactivity did not discriminate between the various forms of precursor or active MMPs.
On the basis of our study, it is clear that MMP-2 and MMP-9 are present in a wide range of canine tumors and normal tissues, and it appears that MMPs are particularly involved in one or more stages of the pathogenesis or clinical advancement of osteosarcoma. Their significance in tumor advancement and their link to tumor type and grade are demonstrated for the first time. The study provides baseline information for a number of commonly encountered clinicopathologic entities in dogs and links the biologic behavior of the tumors with the presence or absence of MMP expression. It, therefore, will allow a substantially improved prognostic and diagnostic evaluation of veterinary cancer patients, particularly if used in conjunction with other established or potentially useful indicators, such as the tumor grade, p53 expression, or proliferating cell nuclear antigen expression.
An unquestioned need has arisen to examine the effects of MMP inhibitors on canine tumor development. MMP inhibitors have been tried in experimental models of human malignancies 6 , 30 and equine laminitis, 48 whereas some inhibitors have entered human clinical trials. 46 , 61 The use of MMP inhibitors as a new approach to the treatment of canine tumors shows enormous potential for clinical application. 6 , 18 The demonstration of MMP involvement in canine neoplasias was a necessary first step that may consequently lead to the design of clinical trials on canine cancer patients, with MMPs being the therapeutic target.
Footnotes
Acknowledgements
This study was supported by grants from the Australian Companion Animal Health Foundation and the Perrett Bequest Fund. We thank Dr. S. J. More for statistical advice, Dr. C. C. Pollitt and Dr. K. Verkest for constructive comments on the manuscript, and Dr. M. Lau and Ms. C. Wilkinson for supplying tumor samples. Preliminary results from this study were presented at the WSAVA/FECAVA World Congress 2000.
