Abstract
Fifty-one meningiomas obtained from 28 dogs and 23 cats were selected for this study to investigate the immunohistochemical expression of matrix metalloproteinase (MMP)-2 and MMP-9 and to compare it to the reverse transcriptase subunit of human-telomerase, progesterone receptor expression, and the proliferative index of the tumors, expressed by Ki67 and proliferating cellular nuclear antigen. Paraffin-embedded tumor tissue was obtained from biopsy samples (28 cases) and at necropsy (23 cases). The most common histotype was malignant in dogs (12/28) and transitional in cats (12/23). Slides immunolabelled for MMPs showed a diffuse cytoplasmic pattern. Twenty-one cases (19 dogs and 2 cats) did not express MMP-2, while only 2 cases were completely negative for MMP-9. The highest values of MMP-2 and MMP-9 were observed in a psammomatous and meningothelial tumor, respectively. On statistical analysis, MMP-2 expression did not show a significant correlation with MMP-9. Moreover, both MMP expressions failed to show significant variance among histologic patterns of the tumor and correlation with the proliferative index. MMP immunolabeling showed an inconstant correlation with progesterone receptor expression. No significant correlation was found between MMP and reverse transcriptase subunit of human-telomerase expression. In feline meningiomas, the MMP-2 value was significantly higher than in canine tumors and the MMP-9 value tended to be low for meningiomas with a follow-up duration from the 23rd month to the 44th month. In cats, the longer the time from surgery, the lower the proliferative index seemed to be. In dogs, we failed to find a correlation between MMP expression and the follow-up duration.
Introduction
Meningioma is a common neoplasm of the central nervous system in both human beings and domestic animals and the most common brain tumor in dogs and cats. 44,47 In spite of the highly variable morphologic and immunophenotypical patterns 5,29 due to the mesenchymal and epithelial origin of meninges, the biologic behavior of meningiomas is generally considered benign in human beings and in dogs and cats, except for the anaplastic histotype, and is associated with secondary effects in the adjacent nervous tissue. Consequently, surgery is the treatment of choice for canine and feline meningiomas, although this is usually reserved for accessible tumors, and postexcision prognosis is generally quite satisfactory. 3,4,8,10 However, in a number of cases (18–21%), there is local recurrence, despite apparently complete resection and benign histology. 8,10 Because of its high incidence and the large number of morpho- and immunophenotypes, there has been great interest in studying meningioma and in establishing an improved classification and grading in relation to its biologic behavior so that both prognosis and advanced therapeutic applications could draw advantages in veterinary oncology. Assessment of anaplasia for meningiomas is still based on morphologic criteria of malignancy, first including high mitotic rate and cellularity, with uninterrupted patternless growth, extensive necrosis, nervous tissue invasion, and metastasis. 18 Nevertheless, today in assessing the malignant potential of meningioma, the difference between cytologic and biologic malignancy should be stressed. To date, several studies have significantly correlated the biologic behavior of meningioma with the proliferative activity of meningeal neoplastic cells in man 1,31 and domestic carnivores, 25,38 but the relationship between the proliferative activity and the recurrence of the tumor seems to be rather inconstant. Therefore, other biologic marker factors, such as peritumoral edema, vascular endothelial growth factor, and vascular permeability factor, have been investigated as predictor markers in human 9,15,37,40 and canine meningioma 7,39 but have not yielded conclusive results. It has been reported that a high proliferative index in the tumor is associated with low progesterone receptor (PR) concentration, suggesting that PR expression in canine and feline meningiomas is a reliable prognostic factor in the evaluation of this tumor, as well as in humans. 2,12,25,46 More recently, canine and feline meningiomas have been tested for telomerase immunoreactivity 22,26 and have produced contradictory results as to the correlation with the proliferative index.
In recent years, in both human and veterinary oncology, 19,20,35 there has been considerable interest in studying metalloproteinase activity as a possible independent prognostic marker and target for therapeutic options. Matrix metalloproteinases (MMPs) are a family of zinc-containing proteolytic enzymes implicated in the degradation and remodeling of extracellular matrix and in vascularization. 27,28 Growing evidence indicates that MMPs are also involved in a number of pathologic processes, including tumor invasion and metastases, in experimental cancer models 43 and in human malignancies. 17,32
Normal arachnoid cells have been shown to synthesize several of the extracellular matrix glycoproteins, including fibronectin, laminin, collagen type IV, and procollagen type III 42 , and it has been seen they are able to produce matrix metalloproteinases (MMP-9) also in bacterial meningitis and encephalomyelitis. 21 MMP-2 and MMP-9, known as the type-IV collagenases, 27–41 have been investigated with immunohistochemistry in both benign and malignant human meningiomas to evaluate the recurrence and invasion potential of this tumor. 6,33,48 Both collagenases have been proved to be directly related to the histologic malignancy and to the tumor recurrence or regrowth. 6,33 To date, MMPs have been investigated in a wide range of canine tumors 19,20,35 but have not been reported in meningioma.
The aim of this study was to investigate MMP-2 and MMP-9 expression in canine and feline meningiomas. We also tried to correlate metalloproteinase expression with other biologic markers of meningioma, such as the proliferative index, the progesterone receptor and telomerase expression, and time from surgery.
Materials and Methods
Fifty-one selected meningiomas obtained from 28 dogs (3–17 years old; mean age = 9 years) and 23 cats (5–16 years old; mean age = 10.6 years) were used for this study. Dog breeds included mixed breeds (n = 12), German Shepherd Dogs (n = 8), Boxers (n = 2), a Pastore Maremmano (n = 1), an Epagneul Breton (n = 1), a Schnauzer (n = 1), a Rottweiler (n = 1), a West Highland White Terrier (n = 1), and a Lhasa Apso (n = 1). Fifteen dogs were female and 13 were male. Cat breeds included domestic short hairs (n = 20), Persians (n = 2), and British Shorthairs (n = 1). Sixteen cats were male and 7 were female. In 28 cases (17 dogs and 11 cats), the tumors were collected as surgery biopsy samples, and in the remaining cases (11 dogs and 12 cats), they were collected during necropsy. In the latter cases, the animals had not been previously submitted to surgery. Animals submitted to surgery did not receive further radiation treatment. Meningiomas affected the brain and spinal cord in 42 and 7 cases, respectively. In the remaining 2 cases, they developed in the retrobulbar region. Tumor tissue was fixed for 5 days in 10% neutral buffered formalin for biopsy samples and up to 3 weeks for necropsy samples. Tumor tissue was then embedded in paraffin wax, sectioned at 5 µm, and stained with hematoxylin and eosin. Histomorphologically, the tumors were classified as transitional (15 cases), meningothelial (11 cases), fibroblastic (4 cases), psammomatous (4 cases), and malignant (17 cases) based on the WHO Domestic Animal Classification of Meningioma (Table 1). 18
Summary of cases with image and statistical analysis values.∗
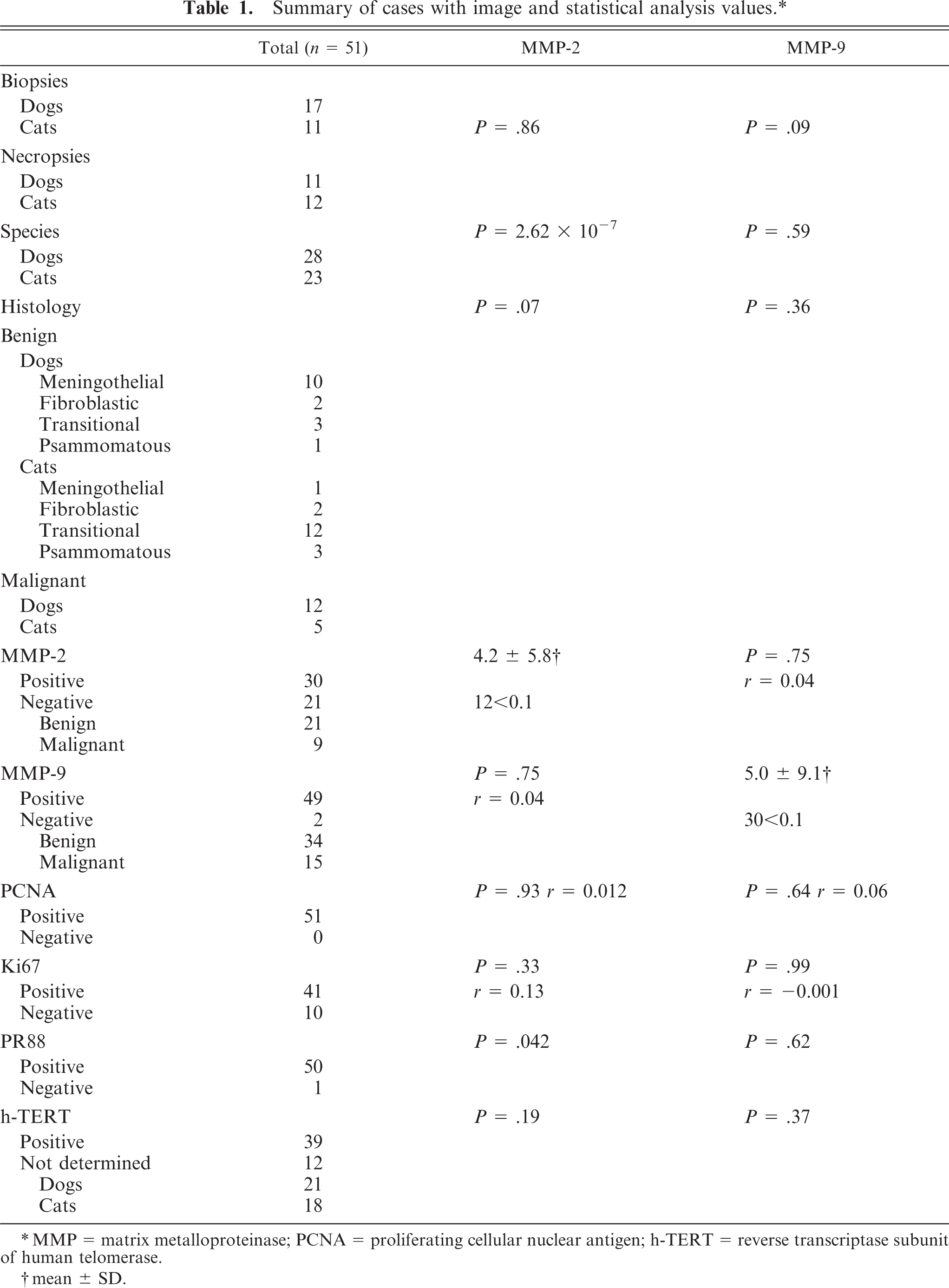
∗ MMP = matrix metalloproteinase; PCNA = proliferating cellular nuclear antigen; h-TERT = reverse transcriptase subunit of human telomerase.
†mean ± SD.
Five-micrometer serial sections were submitted to immunolabeling performed with streptavidin-biotin peroxidase complex method (ABC, Dako, Milan, Italy) for MMP-2 and MMP-9. All cases were also tested for PR. In 39 cases, including 18 cats and 21 dogs, 20 biopsies and 19 necropsy samples, reverse transcriptase subunit of human-telomerase (h-TERT) was also investigated. To estimate the proliferative index of the tumors, immunohistochemistry was also performed in all cases for Ki67 protein and proliferating cellular nuclear antigen (PCNA), as summarized in Table 2. Endogenous peroxidase was neutralized by peroxidase block (3% H2O2 for 5 minutes); slides were dipped in citrate buffer solution (pH 6.0) and Tris-EDTA (pH 9.0) for Ki67, PCNA, PR, h-TERT, MMP-9, and MMP-2. Antigen unmasking was obtained in a microwave oven at low power cycles, except for MMP-2, in which case proteinase K was used as antigen retrieval. The reaction products were visualized with aminoetylcarbazole. Carazzi's hematoxylin was used as a counterstain.
Primary antibodies and antigen retrieval methods used in immunohistochemical evaluation of canine and feline meningiomas.
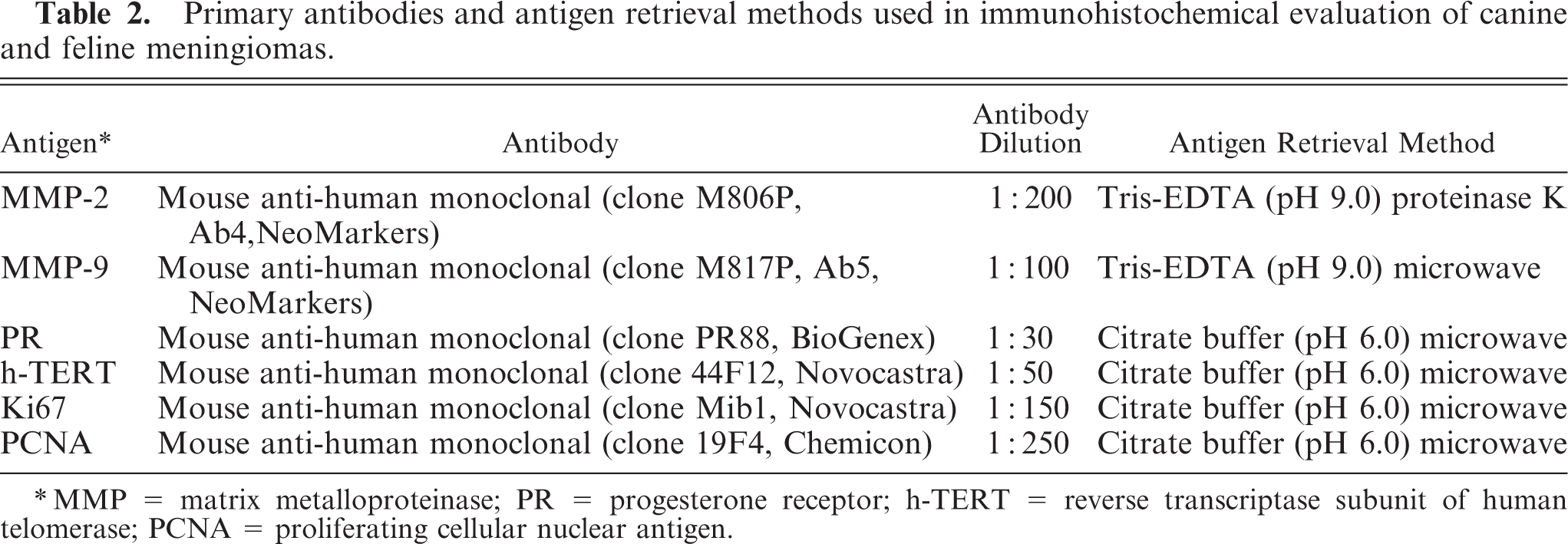
∗MMP = matrix metalloproteinase; PR = progesterone receptor; h-TERT = reverse transcriptase subunit of human telomerase; PCNA = proliferating cellular nuclear antigen.
Positive control samples for MMP-2, MMP-9, Ki67, and PCNA consisted of paraffin wax–embedded feline and canine osteosarcoma, while canine solid mammary carcinoma was used as a positive control for PR. 25 Canine normal testis was used as a positive control for h-TERT. 26 An isotype-matched IgG negative control was run on each section.
Image analysis quantification of immunoreactive cells was performed with an image analyzer NIS-Elements Br-2 (Nikon, Japan) for MMP-2, MMP-9, PR88, Ki67, and PCNA on 10 selected fields (40×). Fields free from aspecific staining showing areas of high and low immunohistochemical expression compared with negative control were chosen. For each tumor it was expressed as the mean value of the percentage of positive area for MMP-2 and MMP-9 and of positive cells for PR88, Ki67, and PCNA. The h-TERT index was assessed by an image analyzer (Lucia 32G/Mutech, Nickon, Japan) on 10 fields (20×) and expressed as the percentage of positive cells. For MMP-expression, 0 was considered negative, while <1.0 was considered low-intensity expression.
Subsequently, statistical analysis was carried out using 1-way analysis of variance, with log transformation for data not normally distributed, to compare MMP expression to species (dog/cat), meningioma types (benign/malignant), and sample type of tumor (biopsy/necropsy). Five percent was the assumed significance level. Then, linear regression analysis was used (R2.7 version software) to evaluate 1) the correlation between the expression of MMP-2 and MMP-9, 2) the correlation between the metalloproteinase expression and neoplasm proliferative index, and 3) the correlation between MMPs and telomerase and PR expression. The assumed significance level for fitting model regression was P ≤ .05 and for correlation R = ±0.7.
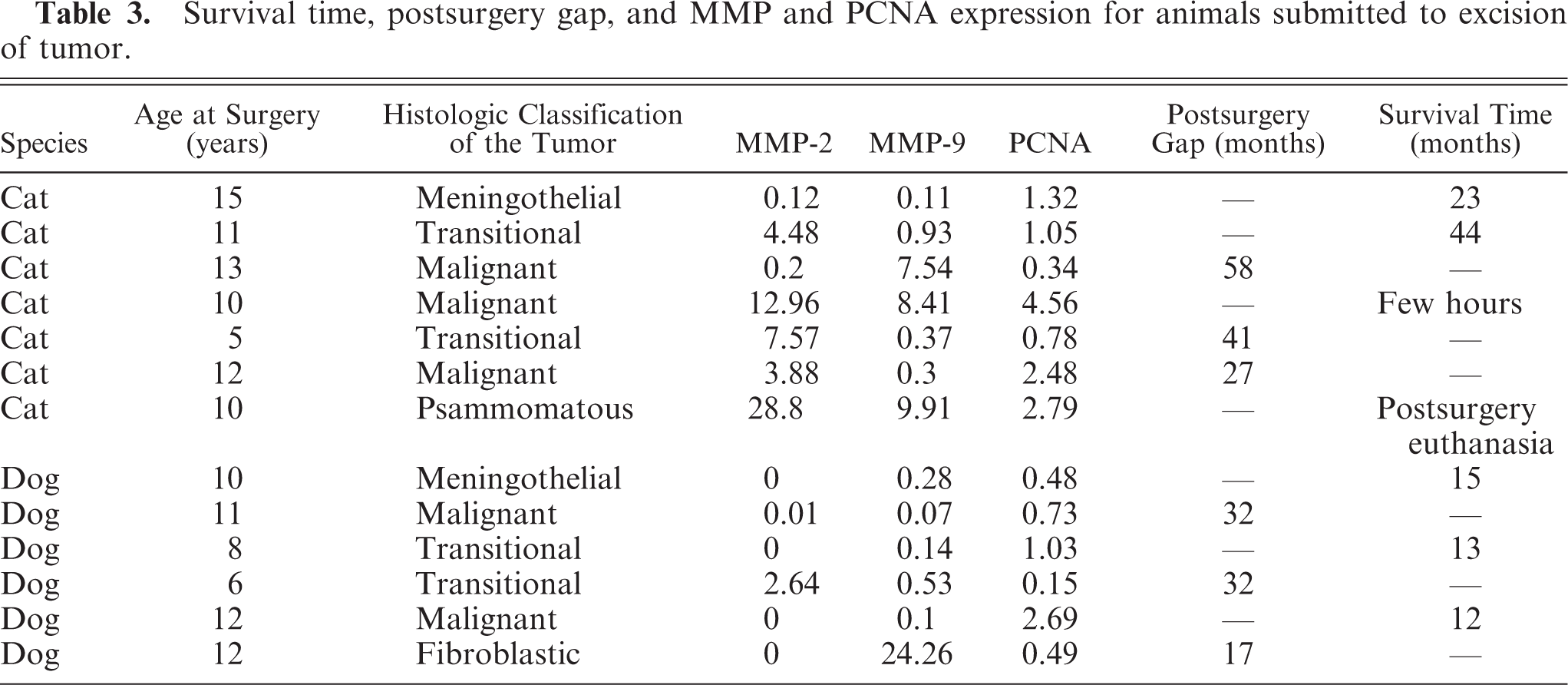
Finally, in 13 cases (6 dogs and 7 cats), the findings collected were correlated with the duration of follow up, defined as the time from the day of surgery expressed in months. It included survival time for dead animals and postsurgery gap for those still alive at the time of investigation (Table 3).
Survival time, postsurgery gap, and MMP and PCNA expression for animals submitted to excision of tumor.
Results
The most common histotype observed in canine meningiomas was malignant (12/28), followed by meningothelial (10/28), transitional (3/28), fibroblastic (2/28), and psammomatous (1/28). Among feline meningiomas, transitional histotype was the most frequently observed (12/23), followed by malignant (5/23), psammomatous (3/23), fibroblastic (2/23), and meningothelial (1/23).
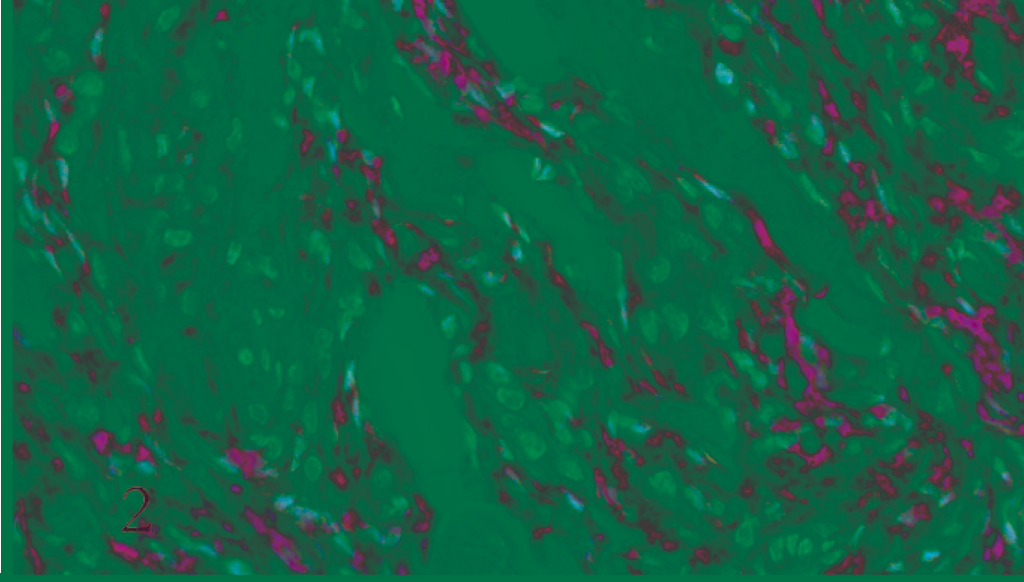
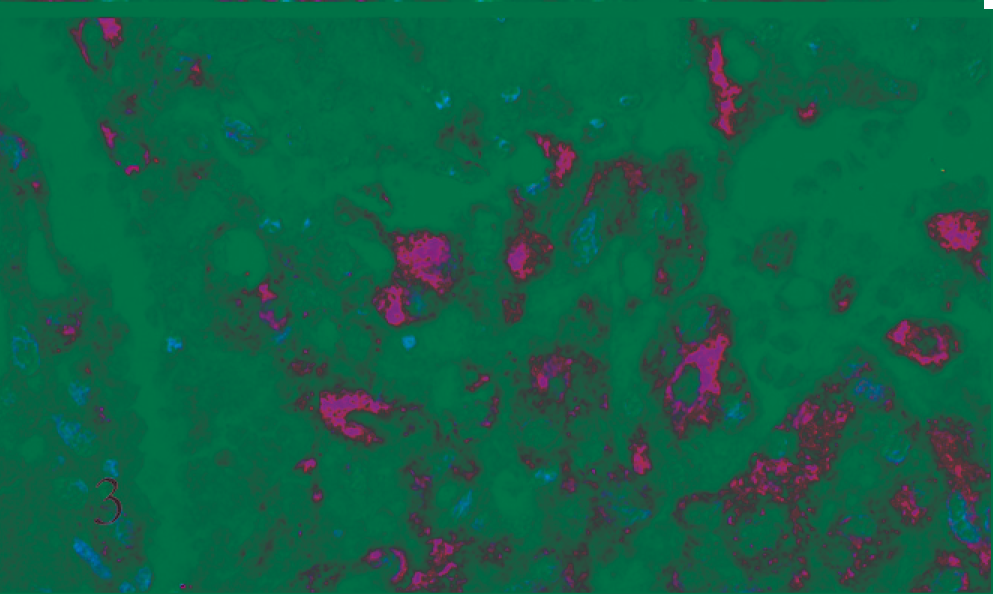
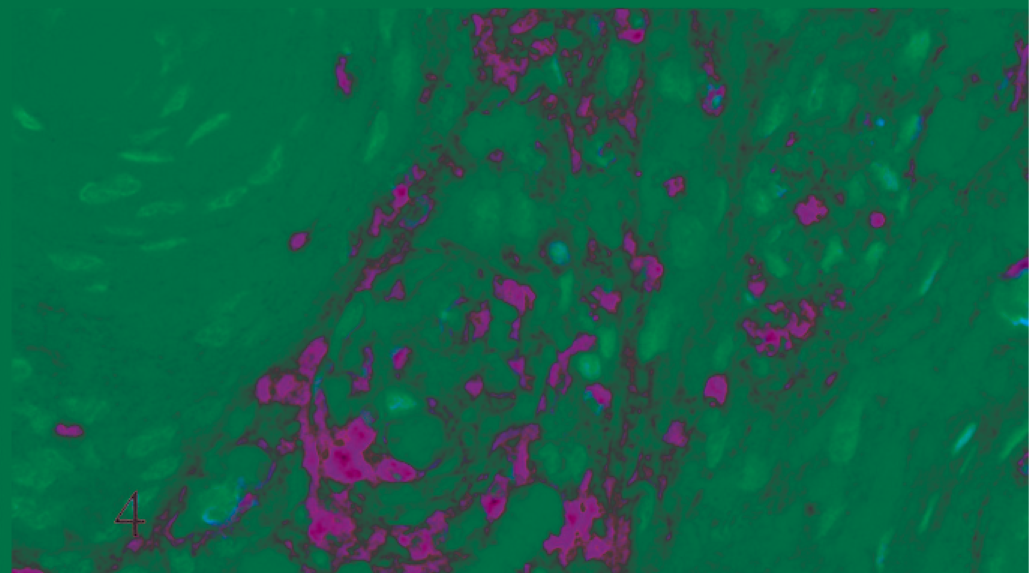
A preliminary histologic examination of the MMP-2 and MMP-9 immunolabelled slides showed a diffuse cytoplasmic immunoreaction pattern. MMP-2 and MMP-9 were expressed by both whorl and fibroblastic patterns in benign meningiomas (Figs. 1, 2). In the malignant type they were expressed in a number of neoplastic cell clusters (Figs. 3, 4) mainly localized in the outer part of the tumor.
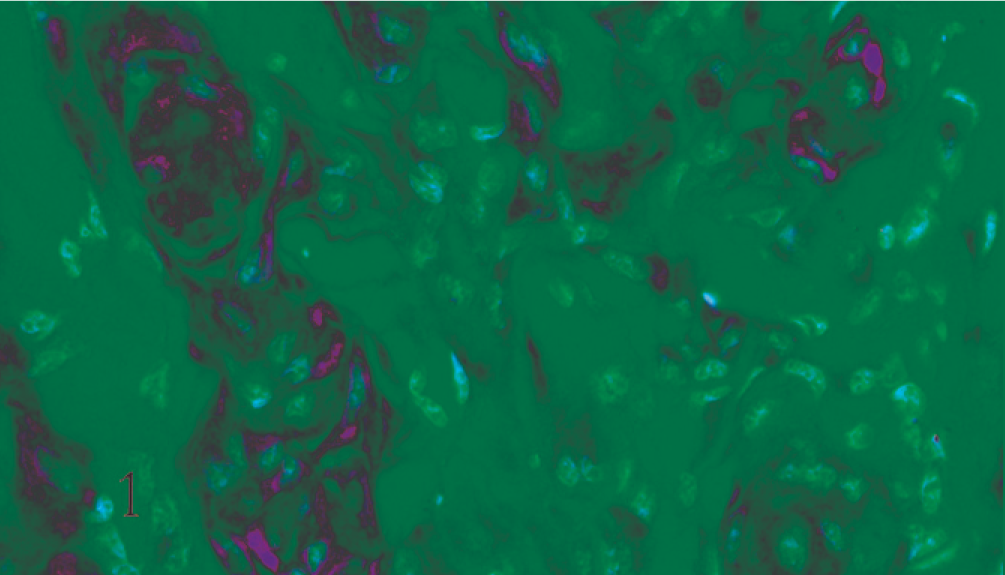
Dog. Psammomatous meningioma. Neoplastic cells arranged in whorls exhibit a marked positive cytoplasmic immunohistochemical staining for MMP-2 (ABC, Carazzi's hematoxylin counterstain).
Cat. Fibroblastic meningioma. Marked cytoplasmic immunoreaction to MMP-9 of neoplastic spindle cells (ABC, Carazzi's hematoxylin counterstain).
Dog. Malignant meningioma. Scattered clusters of neoplastic cells show granular cytoplasmic immunoreactivity to MMP-2 (ABC, Carazzi's hematoxylin counterstain).
Cat. Malignant meningioma. Neoplastic cells arranged close to a vascular element show MMP-9–positive cytoplasm (ABC, Carazzi's hematoxylin counterstain).
MMP-2 expression was observed in 30 of 51 cases (58.8%), of which 21 cases were in the benign group (70%) and 9 in the malignant group (30%). Meningiomas expressing MMP-2 showed a percentage of positive area ranging from 0.01 to 28.8 (mean value = 4.2; SD = 5.8), the higher value corresponding to a psammomatous tumor. Twelve cases were considered of low-intensity expression (< 1.0). MMP-9 expression was observed in 49 of 51 cases (98%), of which 34 cases belonged to the benign group (69.3%) and 15 to the malignant group (30.7%). Cases expressing MMP-9 showed a percentage of positive area ranging from 0.02 to 43.92 (mean value = 5.0; SD = 9.1), the higher value corresponding to a meningothelial tumor. Thirty of these showed a low-intensity expression (< 1.0) (Table 1). In 20 cases, MMP-2-positivity was higher than MMP-9, while in 29 cases MMP-9-positivity exceeded MMP-2. Metalloproteinase expression appeared to be higher in feline meningiomas than in those deriving from dogs.
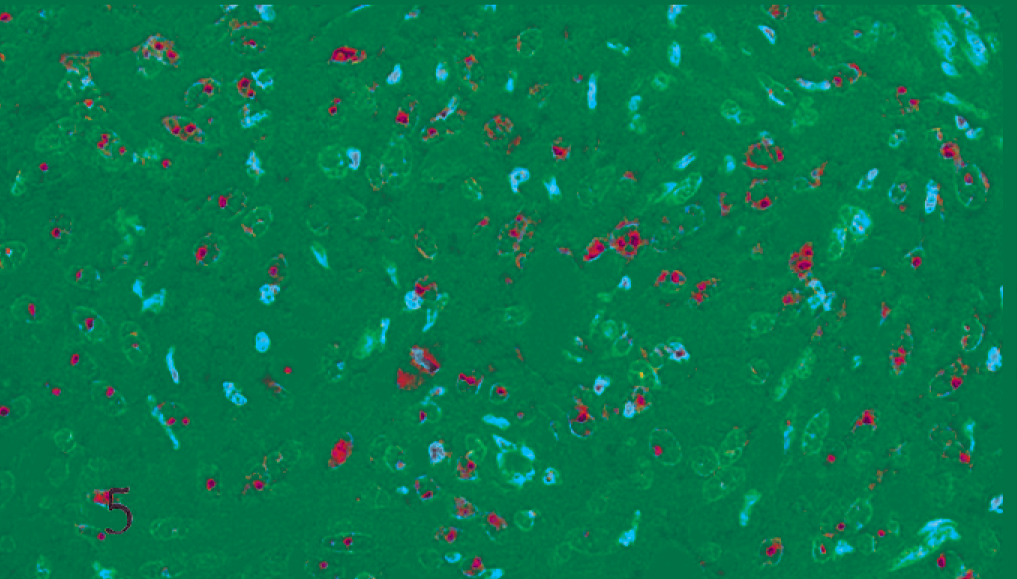
h-TERT protein expression was localized in the nucleus, notably in the nucleolus (Fig. 5). Occasionally it was observed also in the cytoplasm of neoplastic meningothelial cells. In 3 cases, no expression was detected; in the remaining samples, staining intensity ranged from mild to strong, being highly variable from case to case. Progesterone receptors were expressed as a nuclear reaction in all but one case (Fig. 6). PCNA was detected in all the tumors (Fig. 7), while Ki67 was not expressed in 10 cases (4 of them were malignant meningiomas) (Fig. 8). Compared with the related PCNA-positive cases, they were suspected to be false negative and excluded from further evaluations (Table 1).
Cat. Malignant meningioma. Diffuse nuclear and nucleolar immunoreactivity for transcriptase reverse human telomerase (ABC, Carazzi's hematoxylin counterstain).
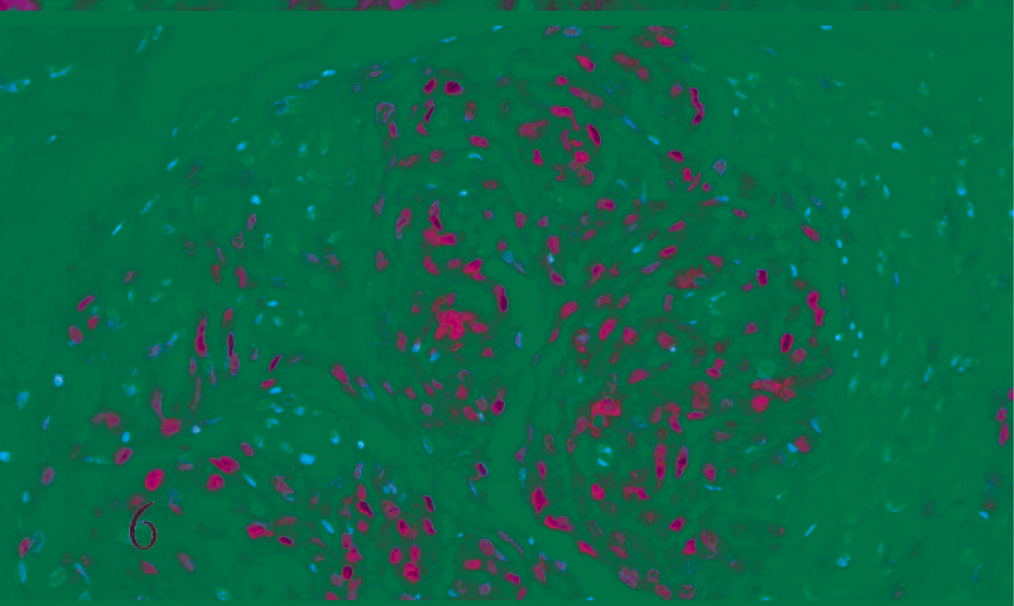
Cat. Transitional meningioma. Diffuse nuclear positive expression for progesterone receptors in ovoid to spindle neoplastic cells (ABC, Carazzi's hematoxylin counterstain).
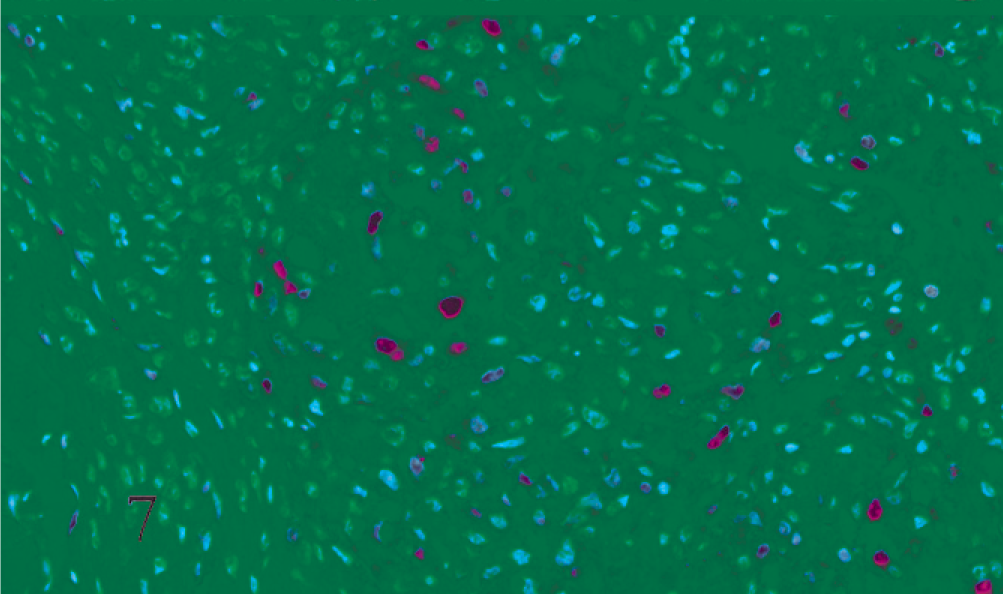
Dog. Malignant meningioma. Positive nuclear immunoreaction to proliferating cell nuclear antigen in scattered neoplastic cells of a solid area (ABC, Carazzi's hematoxylin counterstain).
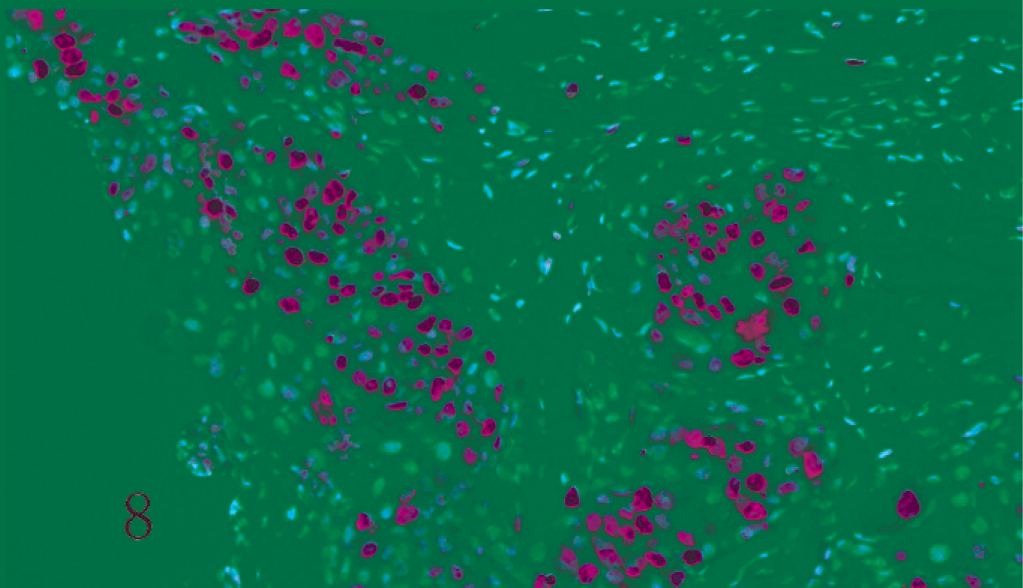
Dog. Meningothelial meningioma. Solid sheets of neoplastic cells reveal a high mitotic activity expressed by Mib-1 nuclear immunoreactivity (ABC, Carazzi's hematoxylin counterstain).
On statistical analysis, MMP-2 expression did not show a significant correlation with MMP-9 expression (R = 0.044, P = .75). Moreover, neither of the metalloproteinases showed a significant variance in the different histologic patterns of the tumor (benign and malignant; P = .07 for MMP-2 and P = .36 for MMP-9) and no correlation with the proliferative index expressed by PCNA (R = 0.012 and P = .93 for MMP-2, R = 0.06 and P = .64 for MMP-9) or by Ki67, for cases expressing it (R = 0.13 and P = .33 for MMP-2, R = −0.001 and P = .99 for MMP-9). In addition, MMP-2 and MMP-9 did not show a significant variance when associated with biopsy or necropsy samples (P = .86 for MMP-2, P = .09 for MMP-9) (Table 2).
MMP-2 immunolabeling showed a significant correlation with PR expression (P = .042), whereas no significant correlation was found between MMP-9 and PR-expression (P = .62). Again, both metalloproteinase expressions failed to show a significant correlation with h-TERT immunoreactivity (P = .19 for MMP-2 and P = .37 for MMP-9) (Table 2).
MMP-2 expression, but not MMP-9 expression (P = .59), showed a significant deviation only when associated to species. Indeed, in the feline meningiomas the MMP-2 value was significantly higher than in the canine tumors (P = 2.62 × 10−7) (Table 2).
In the feline group, animals were submitted to surgery from 5 to 15 years of age (mean = 10.8 years). Three cats were still alive, with a postsurgery survival time ranging from 27 to 58 months. Two cats died immediately after surgery, and the remaining 2 cats died 23 and 44 months after surgery (mean survival time = 33.5 months). The first died at 17 years of age without any further neurologic signs, whereas the second showed balance loss and severe motor deficits 3 months before death. In the canine group, animals were submitted to surgery from 6 to 12 years of age (mean = 9.8 years). Three dogs were still alive at the time of investigation, with postsurgery survival time ranging from 17 to 32 months. The remaining three dogs died 12 to 15 months after surgery (mean survival time = 13.3 months) (Table 3). One died of a neoplasia developing out of the central nervous system, and the remaining 2 dogs presented status epilepticus prior to death. In all cases with presumed recurrence of meningioma, the owners did not allow further resonance magnetic imaging investigations.
In the canine cases, meningiomas with a follow-up duration of 12 to 17 months did not express MMP-2 at all, and the remaining cases, showing the longest postsurgery gap (32 months), corresponded to an expression of MMP-2 below the mean value. MMP-9 expression was of low intensity in all cases except for one, with a 17-month follow up that expressed MMP-9 above the mean value. In cats the meningiomas with the shortest (23 months) and the longest (58 months) observation time showed a MMP-2 expression of low intensity, whereas MMP-9 expression proved to be constantly low from the 23rd to the 44th month of survival time. The case with the highest postsurgery gap (58 months) showed the highest MMP-9 value (7.54). Moreover, in feline meningiomas, but not in canine tumors, the proliferative index expressed by PCNA tended to decrease as follow-up duration increased.
Discussion
This study was performed on a selected group of meningiomas including a proportionate number of canine and feline tumors. Biopsy and necropsy samples were collected to investigate the weight different clinical approaches may have in the evolution of the tumor's biologic behavior. In our series, canine meningiomas were more frequently found to be malignant (42.8%), although the meningothelial and transitional types are reported as the most commonly diagnosed. 5,29,36,45 In a recent study based on WHO classification of human nervous system tumors, canine atypical meningioma was identified in about 43% of cases. 45 Their morphologic features provide sufficient criteria to classify these tumors as malignant based on WHO Domestic Animal Classification of Meningioma. On the other hand, the most frequent feline meningiomas we found were of the transitional type, confirming recently published data. 26,39
In this study we observed MMP-2 and MMP-9 expression in canine and feline meningiomas, showing an immunohistochemical reaction of the cytoplasm of tumor cells, as previously reported. 6,33 Nuclear staining was completely absent. Twenty-one cases did not express MMP-2 (41.2%), and 2 cases did not express MMP-9 (2%). Although some meningiomas failed to express metalloproteinase-2 in the 60% of human cases, 30 we could not completely exclude considering our negative cases as possible false negatives, especially the malignant types. 34 MMP-2 and MMP-9 were expressed with a value higher than 1 (low intensity) in 35.5% and 37.3% of meningiomas, respectively. In human beings, 23.2% and 39.3% of meningiomas are reported to show a high expression of MMP-2 and MMP-9, respectively. 33
Whether the sample was taken as a biopsy or at necropsy does not seem to play a role in MMP-2 and MMP-9 expression either, suggesting that sampling time (in life or postmortem) is not a sufficient criterion to consider the biologic stage of the tumor. In fact, in our experience necropsy material does not always indicate the tumor with the most severe biologic behavior, because owners may request euthanasia because of a dramatic clinical progression or as soon as a cancer is diagnosed, thereby refusing support therapy or feasible surgery.
Although MMP-2 expression seemed to be generally higher than MMP-9 expression, no significant relationship was found between them, probably due to an independent activation of these molecules.
The most relevant result in our study was that neither metalloproteinase expression was correlated to malignant morphologic patterns, contrary to human oncology, in which the high expression of MMP-2 and MMP-9 is significant in nonbenign meningiomas 33 Based on growing evidence indicating that MMPs are involved in tumor progression and malignancy, our findings would appear to be difficult to interpret.
In human meningiomas controversy exists in the literature regarding the level of MMP expression in meningiomas of differing grades reported by WHO classification. 23 In some studies, MMP-2 expression increases progressively from WHO grade I to II and III, while MMP-9 expression shows an increase in the number of positive tumor cells in grades I and II but decreases significantly when progressing from WHO grade II to III. 48 Conversely, other studies reported that only MMP-9 shows a real tendency to increase with increasing tumor grade. 34 Providing criteria for grading malignancy potential could help in understanding tumor progression and biologic changes also for domestic animal meningiomas. 7,38,45
A statistically significant value found in metalloproteinase expression was related to species. In fact, conversely to our expectations, MMP-2 appeared to be significantly more expressed in feline meningiomas, which are normally known to be less aggressive than canine meningiomas and thus more responsive to surgery. 8,10
In our study we found that metalloproteinase expression is also independent of proliferative potential, as commonly observed in human meningiomas. 33 Only recently, a positive correlation was found between MMP-9 expression and proliferative index. 34 The proliferative index failed to be assessed by Ki67 expression in a number of cases, most likely due to a prolonged storage in formalin or in paraffin blocks responsible for subcellular modifications. 16,31 On the other hand, PCNA immunostaining was more prominent and constant than Ki67 in all tumors, even though it is a less specific marker of proliferation. 14
We also confirmed that canine and feline meningiomas express h-TERT, as previously reported. 22,26 Nevertheless, no significant correlation was found between telomerase and metalloproteinase immunoexpression, probably because they follow different and independent activation pathways.
The correlation between MMP and PR expression seemed quite contradictory. In fact, statistical analysis showed a significant correlation between PR and MMP-2 immunolabeling, but not for MMP-9 expression. This finding seems to support the fact that metalloproteinase activity is not that strictly related to the proliferative index of neoplastic meningial cells, as conversely demonstrated for PR88. 25,46
The follow-up data showed a survival time of up to 58 months for feline meningiomas submitted to surgery and for canine meningiomas of up to 32 months, even though they were malignant. These findings are more encouraging than those reported in the literature. 3,8,10,19 Patients still alive or with a long survival time were indifferently both young and old, with a mean age of about 10 years. This finding indicates that the survival time was not apparently influenced by the age of the animals.
Indeed, in feline more than in canine meningiomas, survival time was well correlated to the proliferative index. In fact, a lower proliferative index seemed to correspond to a longer survival time. On the contrary, the relationship between the metalloproteinase expression and follow-up data proved to be contradictory. Although the limited data in the present study must be interpreted with caution, in the feline meningiomas MMP-9 expression seemed to be quite constant and of low intensity (< 1) for tumors with 2 to 4 years of survival. The higher value of MMP-9 in the cat with a postsurgery gap of over 4 years might simply represent a singular case of no sound prognostic judgment. In the canine cases, we failed to find a correlation between MMP-2 and MMP-9 expression and follow-up duration.
To date, the role played by MMP-2 and MMP-9 expression in canine and feline meningiomas does not appear to be a consistent prognostic factor, because MMP expression was not related to the proliferative potential of meningiomas, and neither were MMP-2 and MMP-9 expressions related to histologic malignancy, making the morphologic evaluation of these tumors' biologic behavior quite arbitrary. In human beings, although various histopathologic features have been associated with more aggressive behavior and recurrence of meningiomas, there is little agreement as to which features are the most important to be considered. They include loss of architecture, hypercellularity, nuclear pleomorphism, prominent nucleoli, high nuclear/cytoplasmic ratios, increased mitotic activity, hypervascularity, necrosis, and invasion of the brain. 13,23 The lack of consensus regarding the characteristics associated with aggressive cancer behavior and recurrence of meningiomas makes it difficult for histopathologists and neurosurgeons to predict recurrence. The fact that Ki67 expression corresponds to WHO classification and tends to be correlated with the biologic behavior of these different groups (i.e., benign, atypical, and malignant) does not indicate whether the tumor has a tendency to recur. 24
In canine and feline meningioma, the finding of MMP expression calls for further investigations into the role played by MMPs, perhaps in relation to a revised morphologic malignancy grading and to the recurrence of intracranial or spinal meningioma, whether benign or malignant, along with other factors influencing tumor invasion into the brain or spinal cord and its recurrence, such as location, extent of surgery, intraoperative findings, radiographic aspects, age of the patient, edema, cytokine production, and possibly the chromosomal alteration.
Finally, we believe MMP-2 and MMP-9 expression should be further investigated as an additional prognostic factor in canine and feline meningiomas, apparently independent of proliferative potential, h-TERT, and PR expression but more likely dependent on factors we still need to define. At the moment, the high variability between morphologic and biologic malignancy we observed remains to be explained. At the same time we believe more exhaustive and careful follow-up data should be collected for patients with meningiomas showing an increased expression of MMPs, to reduce at least the difference between cytologic and biologic malignancy.
Footnotes
Acknowledgements
The authors thank Dr. Giovanni Ricci and Dr. Silvia Capuccini for their technical assistance in histochemistry and Dr. Andrea Onofri in statistical analysis.
