Abstract
Response to 3-methylindole (3MI) varies among species. Mice recover from 3MI-induced bronchiolar epithelial injury but sustain persistent olfactory mucosal injury with scarring and epithelial metaplasia. In contrast, 3MI induces obliterative bronchiolitis in horses and ponies, but olfactory mucosal injury has not been reported. To evaluate the effect of 3MI on equine olfactory mucosa, ponies were dosed orally with 100 mg 3MI/kg (n = 9) or corn oil vehicle (n = 6). All ponies treated with 3MI developed obliterative bronchiolitis with mild olfactory injury. By 3 days after 3MI dosing, olfactory epithelium appeared disorganized with decreased and uneven surface height and scalloping of the basement membrane zone. Epithelial cells of Bowman's glands were hypertrophic. Proliferation of olfactory epithelium and Bowman's glands was supported by an increased mitotic index and positive immunohistochemical staining for proliferating cell nuclear antigen as compared with controls. The activity of 11β-hydroxysteroid dehydrogenase, an olfactory mucosal cytosolic enzyme localized to sustentacular and Bowman's glandular epithelial cells, was concurrently decreased. By 9 days postdosing, olfactory mucosal lesions had lessened. Results indicate that 3MI transiently injures equine olfactory mucosa without the extensive necrosis, scarring, or metaplasia seen in murine olfactory mucosa or in equine bronchiolar epithelium.
Keywords
3-Methylindole (skatole, 3MI), a tryptophan metabolite produced in the mammalian digestive tract, 9 , 44 was demonstrated in 1967 to cause acute pulmonary edema and emphysema in cattle that are switched abruptly from sparse to lush pasture. 10 Experimentally, 3MI causes pulmonary edema in goats, 5 , 8 sheep, 4 rats, 22 , 43 and some strains of mice. 11 In these species 3 , 38 , 43 and in horses and ponies 37 3MI selectively targets nonciliated bronchiolar epithelial (Clara) cells, the major pulmonary site of cytochrome P-450. 3MI toxicosis in horses and ponies results in many of the morphologic and pulmonary functional changes noted in equine recurrent airway obstruction (chronic obstructive pulmonary disease, heaves). 6 Cytochrome P-450 isoenzymes of goats, mice, rabbits, and human beings convert 3MI to its reactive intermediate, 3-methyleneindolenine, which damages cells by forming protein adducts, particularly in the lungs. 28 , 31 , 36
In the nasal cavity, 3MI selectively injures olfactory mucosa, sparing respiratory mucosa, at least at lower doses, in mice 39 , 40 and rats. 1 , 25 , 26 In mice, 40 mucosal injury is evident under light microscopy within 6 hours of intraperitoneal injection of 3MI and progresses to diffuse mucosal necrosis by 48 hours. Although mice recover from the bronchiolar epithelial necrosis induced by nonfatal doses, olfactory mucosal injury progresses to epithelial metaplasia, fibrosis, and ossification that persist through 28 days after 3MI injection. 40 Olfactory mucosal damage also persists in rats and is accompanied by a sensory deficit. 26
3MI-induced epithelial necrosis is attributed to a reactive intermediate that binds cell proteins, 28 , 31 , 36 but the pathogenesis of the ensuing nasal fibrosis in rodents is obscure. Aldosterone promotes fibrosis in a variety of tissues 33 , 42 and may function in olfaction by regulating sodium–potassium adenosine triphosphatase 13 but must compete with the more abundant glucocorticoids for the mineralocorticoid receptor (MR). 11β-Hydroxysteroid dehydrogenase type 2 (11βHSD2) inactivates glucocorticoids, thereby facilitating aldosterone binding. 14 Colocalization of 11βHSD2 activity 19 and MR expression 18 is requisite for this aldosterone selectivity and has been demonstrated both in sustentacular cells of olfactory epithelium and in epithelial cells of lamina proprial Bowman's glands. Demonstration of the coexistence of MRs and 11βHSD2 activity in olfactory mucosa could attribute a role for mineralocorticoids in the pathogenesis of 3MI-induced fibrosis.
Although 3MI causes severe olfactory mucosal damage and fibrosis in rodents (in addition to small airways disease), this aspect of 3MI toxicosis has not been evaluated in horses or ponies. Not only would nasal scarring cause decreased performance and exercise intolerance, but any olfactory sensory deficit would also have a major impact on equine behavior. The purpose of this study was to evaluate olfactory mucosal damage in equine 3MI toxicosis by clinical and morphologic evaluation, immunohistochemistry for proliferation markers, and assay of 11βHSD activity.
Materials and Methods
Animals
Fifteen adult grade ponies (aged 2 to approximately 20 years; mean weight, 177.1 kg) were randomly assigned to a treatment (six geldings and three mares) or control group (three geldings and three mares). Initial body weight did not differ significantly between the groups. Nine ponies received a single dose (100 mg/kg body weight) of 3MI in corn oil by nasogastric intubation, a protocol previously shown to induce obliterative bronchiolitis. 37 Six control animals received corn oil only. Heart rate and respiratory rate were monitored daily. Body weight was measured at the beginning of the study and at necropsy. Carotid arterial blood gases were measured at 3 days after dosing for all ponies except one and 6 days after dosing in the ponies still alive at that interval. One pony died 9 days after 3MI dosing. Remaining ponies that received 3MI were euthanatized at 3, 5, and 9 days postdosing; control ponies were euthanatized at 3, 5, and 11 days by intravenous pentobarbital overdose. Complete necropsy examinations were performed on all ponies.
Histology and immunohistochemistry
The left cranial lung lobe was fixed by airway perfusion with phosphate buffered formalin at 30 cm fixative pressure for histopathologic studies. Samples from remaining pulmonary lobes, dorsal and ethmoid turbinates, and other tissues were fixed by immersion in formalin fixative. For each pony, sections of nasal mucosa lined by respiratory epithelium and by olfactory epithelium, brain, heart, lung, liver, kidney, spleen, thyroid gland, adrenal gland, and pancreas were stained with hematoxylin and eosin and examined microscopically. Sections of olfactory mucosa were also immunohistochemically stained for proliferating cell nuclear antigen (PCNA). 27 For this procedure, diaminobenzidine was used as the chromagen; Mayer's hematoxylin was the counterstain. Cell proliferation was quantified by counting the number of mitotic figures and PCNA-immunopositive cells in ten 400× fields of olfactory mucosal epithelium and epithelium of Bowman's glands.
11βHSD assay
For enzyme assays, lung and ethmoid turbinates collected from ponies at 3 and 5 days postdosing were snap-frozen in isopentane chilled with dry ice, and maintained at −80 C until analysis. Frozen ethmoid turbinates and lung were homogenized in Krebs–Ringer phosphate buffer. After determination of protein concentration, 2 aliquots of the homogenized turbinates equivalent to 3 mg total protein were radiometrically assayed for 11βHSD activity, 32 , 34 using corticosterone as substrate and nicotinamide adenine dinucleotide phosphate+ and nicotinamide adenine dinucleotide+ as cofactors for 11βHSD1 and 11βHSD2, respectively. Conversion of substrate corticosterone to its inactive 11-keto product was calculated for each isoenzyme from the initial substrate concentration and the final concentration of radioactive 11-dehydrocorticosterone.
Data analyses
Data sets were compared between treated and control ponies by Student's t-test. The Mann–Whitney rank sum test was used as a nonparametric procedure when observations failed tests for normality or equal variance. Significance level was set at P < 0.05.
Results
Control ponies were clinically normal throughout the duration of the experiment. Ponies dosed with 3MI were lethargic and often recumbent for the first 24 hours. A clinically detectable effect of 3MI on heart rate was not observed. Predosing median respiratory rate was 25/minute in control ponies and 20/minute in test ponies. Twenty-fifth and 75th percentiles were 20 and 30/minute, respectively, in both groups. Subsequent to dosing, median respiratory rate did not exceed 30/minute in control ponies but was 58/minute in 3MI-treated ponies by 1 day postdosing and remained elevated (range 35–60/minute) through the duration of the study. Most 3MI-treated ponies had labored expiration. One pony died 9 days after 3MI; death was attributed to 3MI-induced bronchiolar injury.
Mean Pa
Test ponies lost 8.5 ± 4.8% body weight during the experiment. Control ponies lost 1.7 ± 5.6% body weight.
At necropsy, lungs of ponies treated with 3MI were diffusely overinflated and failed to collapse completely when the thoracic cavity was opened. Treatment-related gross lesions were not observed in remaining visceral organs or tissues.
Treatment-related microscopic lesions were limited to the lung and nasal mucosa. Lung lesions in 3MI-treated ponies were confined to the bronchioles. At 3 days after 3MI dosing, denuded regions within terminal bronchiolar epithelium alternated with regions lined by one to three layers of squamoid and/or low cuboidal epithelial cells. In these regions, approximately one to two mitotic figures per bronchiolar cross section were observed. Neutrophils and macrophages sparsely infiltrated the epithelium and peribronchiolar connective tissue. Some bronchiolar lumina were occluded by fibrinous exudate, hypertrophied and hyperplastic epithelial cells, neutrophils, and mononuclear leukocytes.
At 5 days postdosing with 3MI, bronchiolar mucosa was fully re-epithelialized. Bronchiolar epithelium consisted of one to four layers of cuboidal to polyhedral epithelial cells. Most bronchiolar lumina were devoid of fibrinous exudate, but some lumina were partially occluded by immature granulation tissue with sparse neutrophil infiltration.
Nine days after 3MI dosing, most bronchioles were lined by a single layer of flattened epithelial cells and/or up to five layers of stratified epithelial cells. Some bronchiolar lumina were partially occluded by granulation tissue.
Nasal lesions were confined to areas lined by olfactory mucosa. In control ponies (Fig. 1), olfactory epithelium was arranged in orderly layers of prominent supranuclear (apical) cytoplasm, sustentacular nuclei, neuronal nuclei, and basal cells. Bowman's glands and nerve bundles occupied the bulk of the lamina propria.
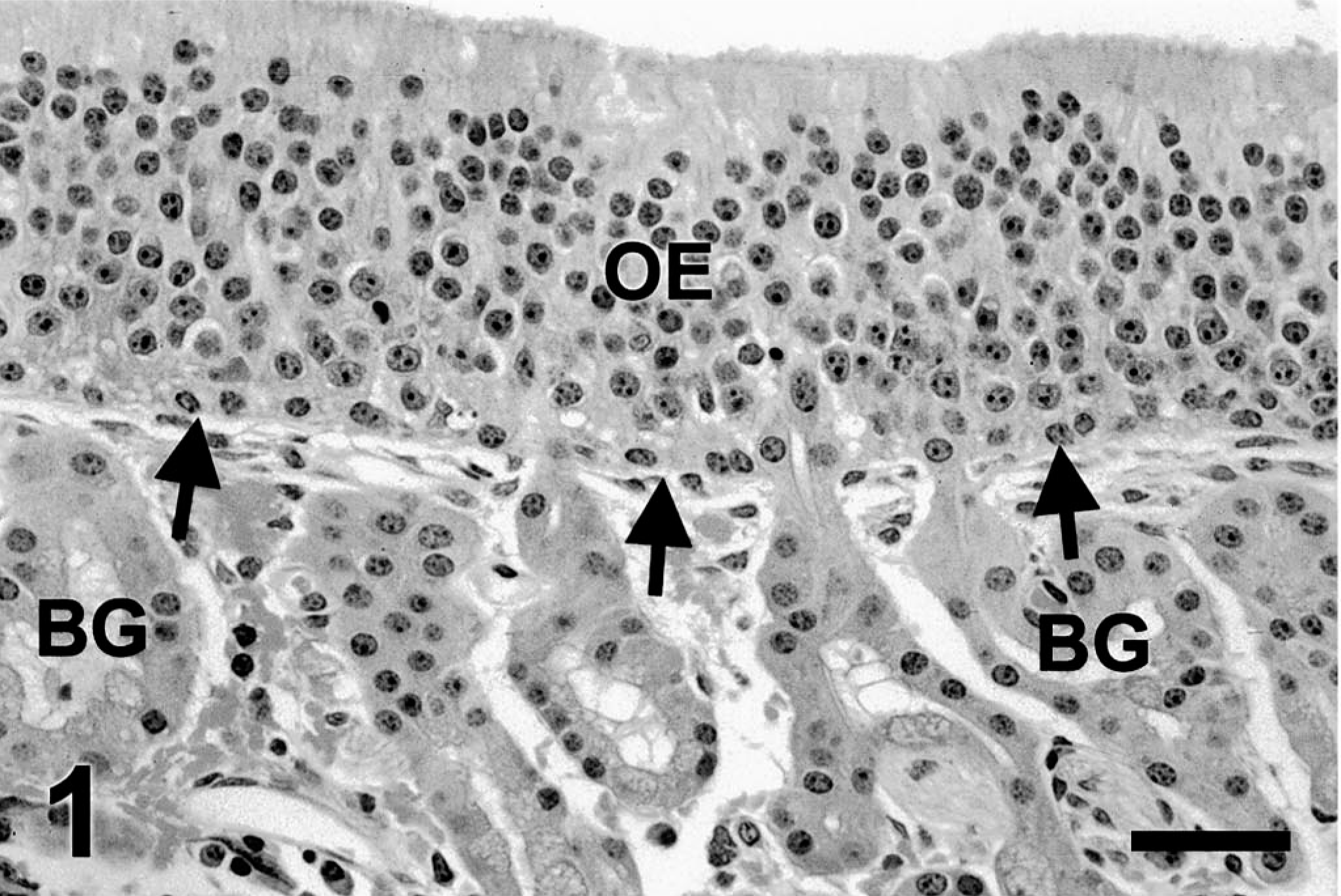
Ethmoid turbinate; control pony. Olfactory mucosa has orderly olfactory epithelium (OE) with straight basement membrane zone (arrows). Bowman's glands (BG) occupy the lamina propria. Mitotic figures are not observed. HE. Bar = 25 µm.
At 3 days postdosing, the olfactory epithelium of 3MI-treated ponies was disorganized and attenuated with patchy necrosis in some areas. Epithelial damage appeared as blebbing of the apical surface of epithelial cells, hyperchromic nuclei, and occasional mitotic figures. Edema expanded the lamina propria. Epithelial cells of Bowman's glands were hypertrophied with hypochromic nuclei, prominent nucleoli, and occasional mitotic figures (Fig. 2).
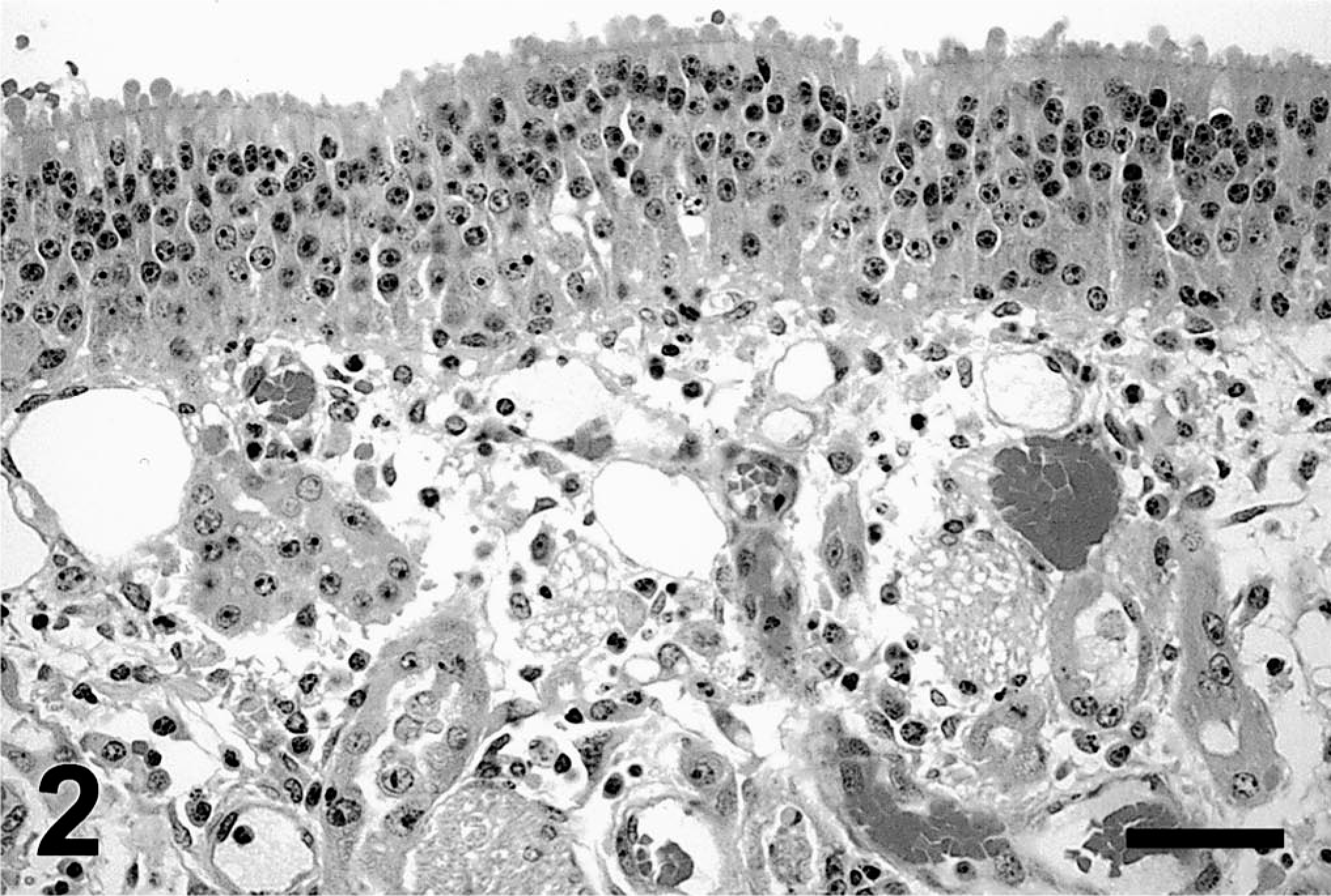
Ethmoid turbinate; pony; 3 days after 3MI dosing. Olfactory mucosal epithelium has apical blebs and hyperchromic nuclei. Lamina propria is expanded by edema. Epithelial cells of Bowman's glands are hypertrophied with hypochromic nuclei and prominent nucleoli. HE. Bar = 25 µm.
Olfactory epithelium, 5 days after 3MI dosing (Fig. 3), was more disorganized than that observed at day 3 postdosing. Increased numbers of hypertrophic cells and mitotic figures were observed in mucosal epithelium and that of Bowman's glands.
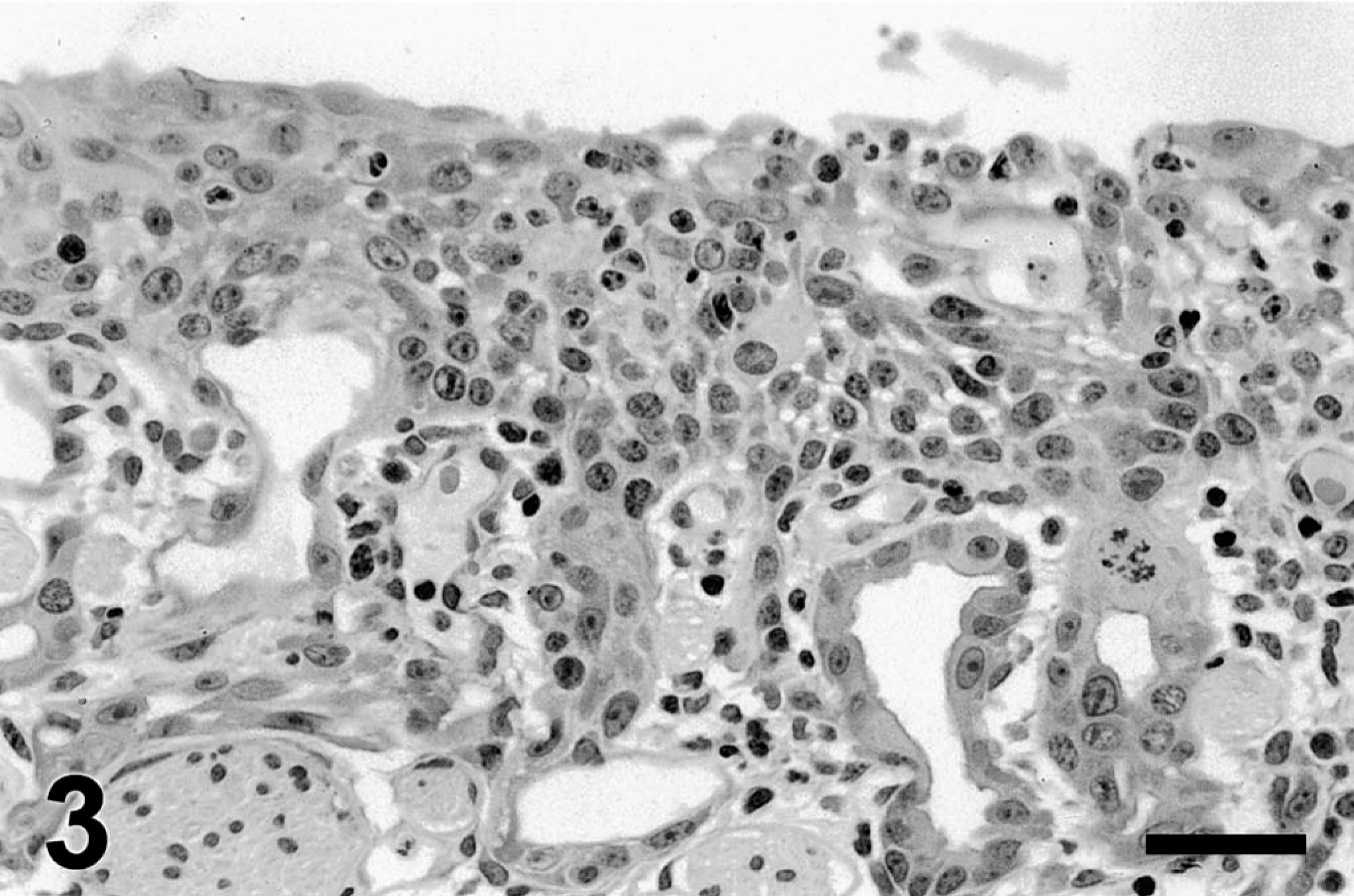
Ethmoid turbinate; pony; 5 days after 3MI dosing. Olfactory epithelium and Bowman's gland are hyperplastic and poorly organized with mitotic figures. HE. Bar = 25 µm.
Nine days after 3MI dosing, minimal change was apparent in the olfactory mucosal epithelium and Bowman's glands in two of three ponies. However, areas with epithelial disorganization, cellular hypertrophy, and/or mitotic figures (Fig. 4) were observed in the olfactory mucosa of all three ponies.
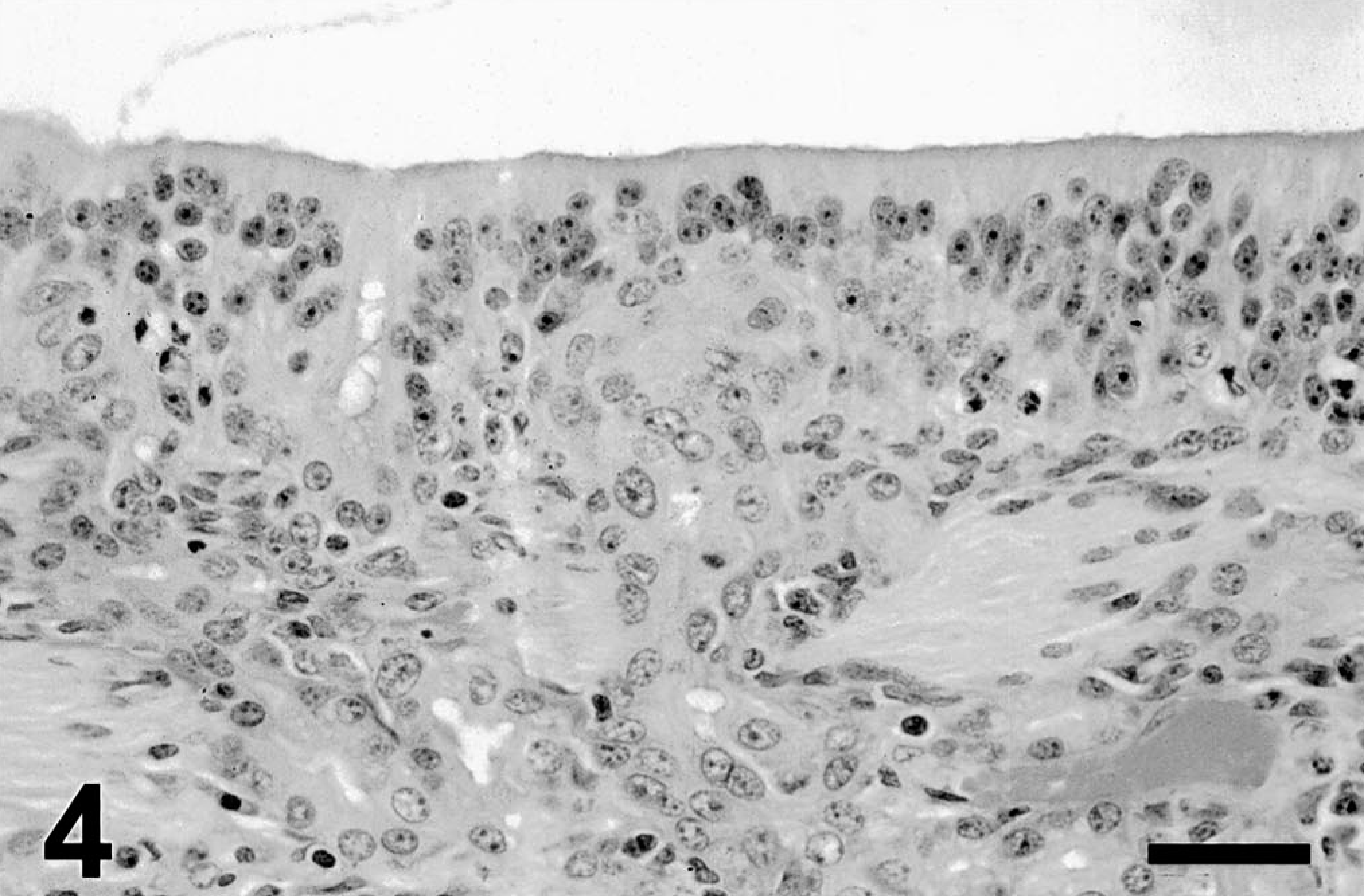
Ethmoid turbinate; pony; 9 days after 3MI dosing. Hyperplasia is milder than that at earlier intervals. Olfactory epithelium and Bowman's glands appear disorganized. HE. Bar = 25 µm
The mitotic indices in olfactory epithelium and Bowman's glands did not have normal distribution, so data are reported as median values with 25th and 75th percentile points (Fig. 5). Mitotic figures were not observed in olfactory epithelium or Bowman's glands of control ponies at any dosing interval. At 3 days after 3MI dosing, a median value of three mitotic figures per 10 high-power (400×) fields was determined in olfactory epithelium but was not significantly different from the mitotic index in control ponies and was diminished at subsequent sampling intervals. The mitotic indices in epithelium of Bowman's glands were significantly higher than in control ponies at 3 and 5 days after 3MI dosing.
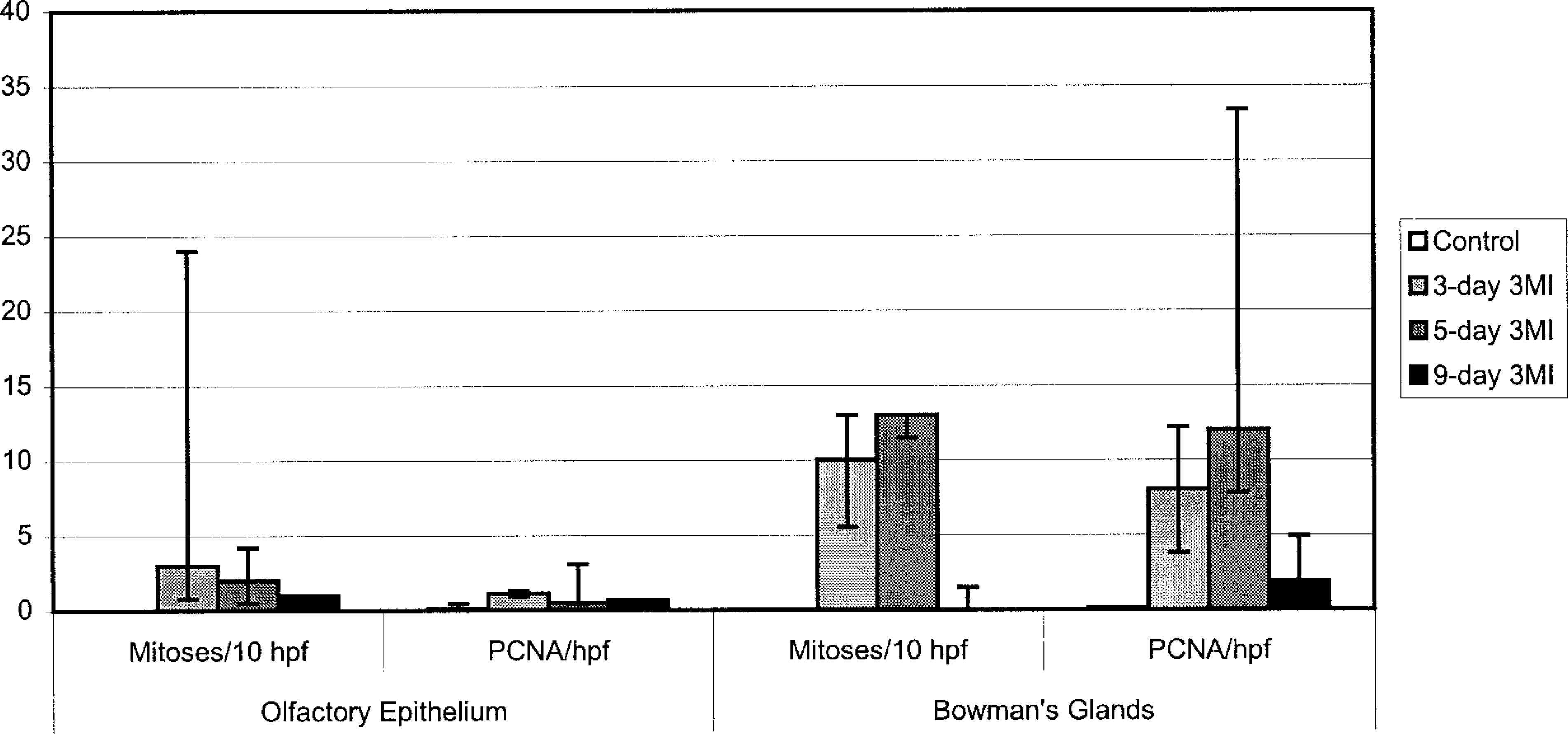
Median (minus 25th plus 75th percentiles) mitotic index and PCNA labeling in olfactory mucosa of ponies after 3MI dosing
Values for PCNA immunohistochemistry (Fig. 5) are expressed as for mitotic indices. In control ponies, PCNA-immunopositive cells were seldom observed in olfactory epithelium or Bowman's glands (Fig. 6). PCNA labeling did not vary with time after dosing in control ponies. PCNA labeling in olfactory epithelium was maximal at 3 days after 3MI dosing (Fig. 7) and significantly higher than that in control ponies. PCNA labeling was always more frequent in epithelial cells of Bowman's glands than in the mucosal epithelium and was elevated by 3 days (Fig. 7), maximal at 5 days (Fig. 8), and still elevated 9 days after 3MI dosing (Fig. 9). Values were significantly greater than those in control ponies at 5 and 9 days after 3MI dosing.
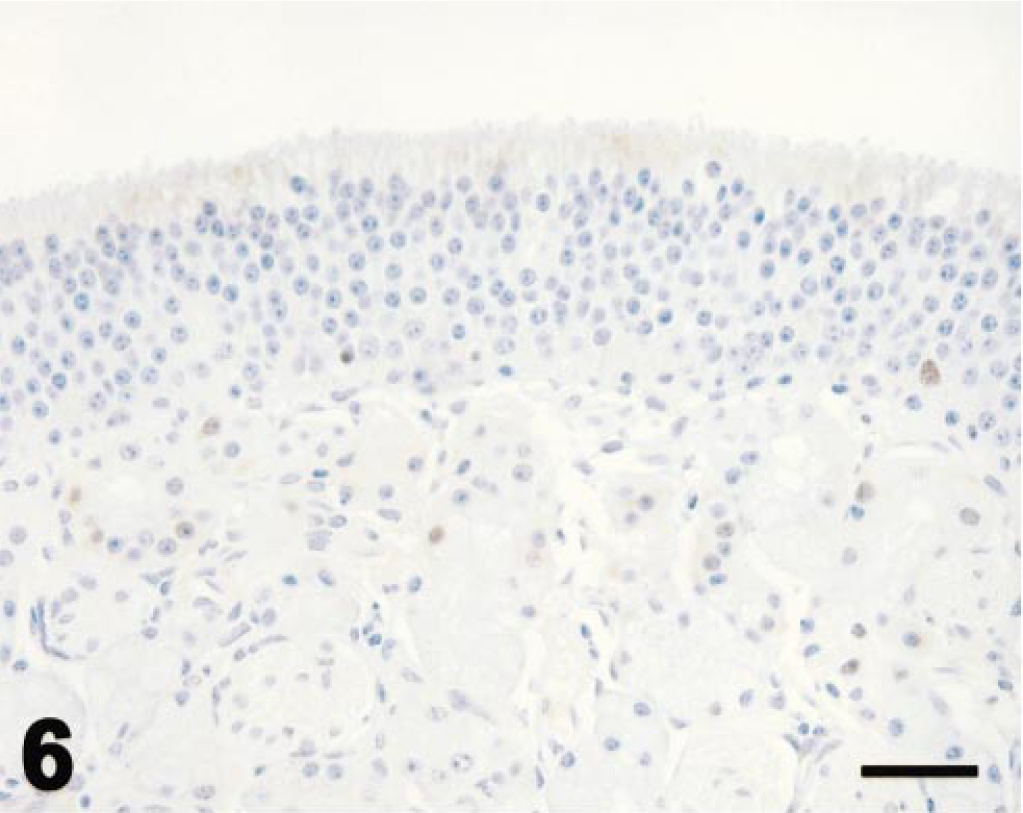
Ethmoid turbinate; control pony; immunohistochemistry for PCNA. Olfactory mucosa has orderly olfactory epithelium and prominent Bowman's glands. Epithelial cells are seldom labeled for PCNA. Avidin–biotin immunoperoxidase with hematoxylin counterstain. Bar = 25 µm.
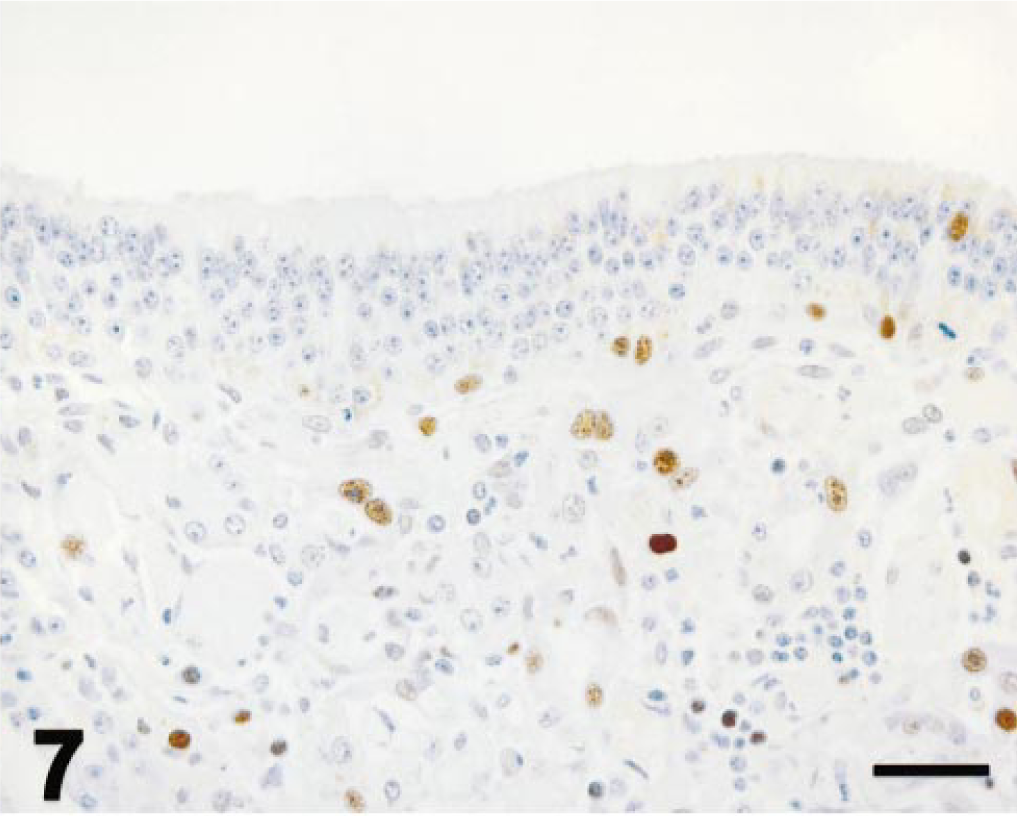
Ethmoid turbinate; pony; 3 days after 3MI dosing; immunohistochemistry for PCNA. Epithelial height is decreased. Scattered cells in olfactory epithelium and Bowman's glands are labeled by antibody to PCNA. Mayer's hematoxylin counterstain. Bar = 25 µm.
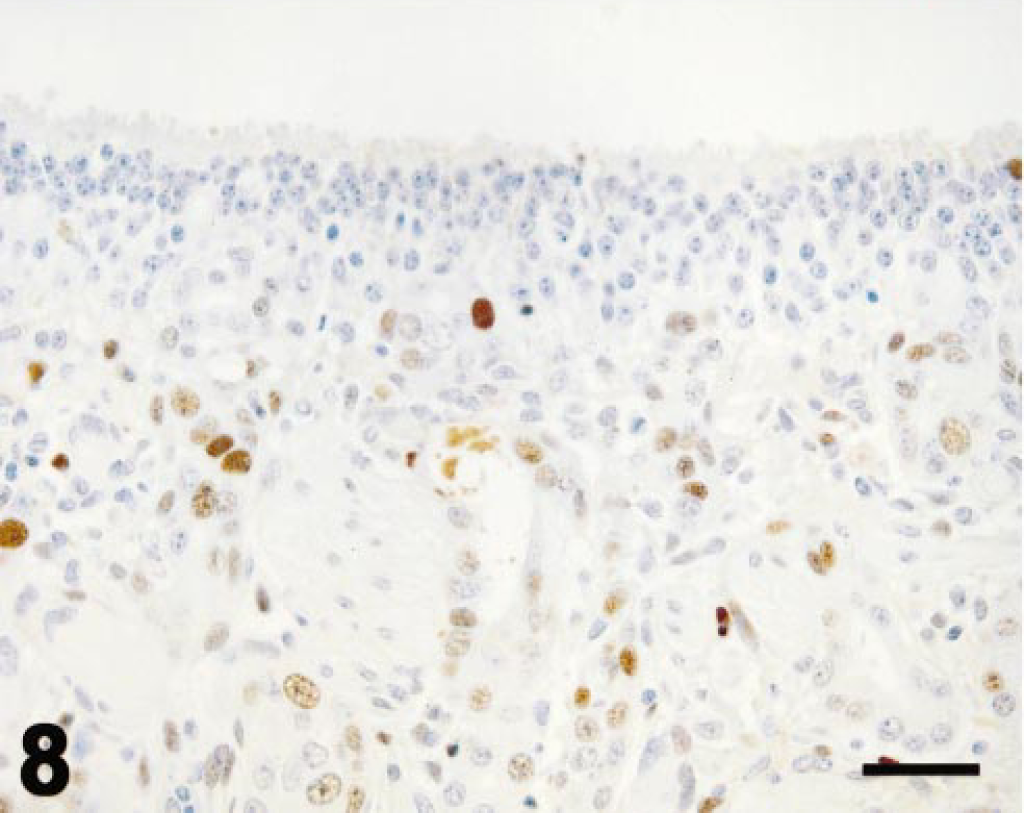
Ethmoid turbinate; pony; 5 days after 3MI dosing; immunohistochemistry for PCNA. Scattered epithelial basal cells and more numerous Bowman's gland epithelial cells are labeled by immunohistochemistry for PCNA. Mayer's hematoxylin counterstain. Bar = 25 µm.
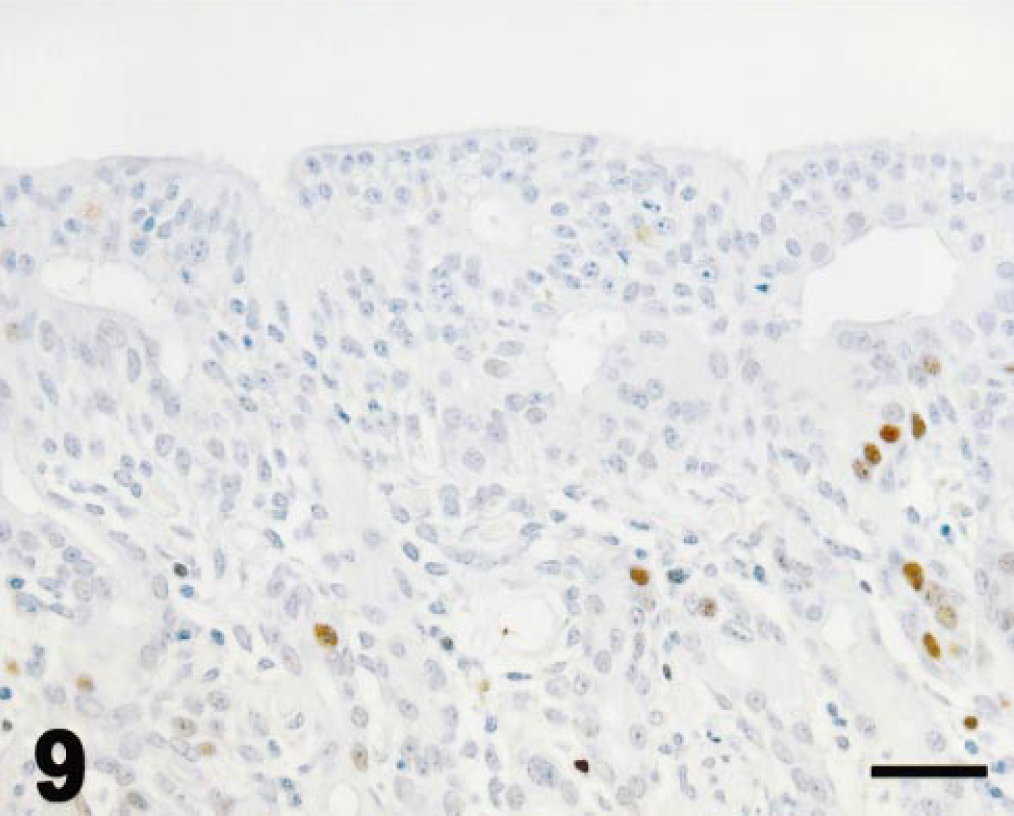
Ethmoid turbinate; pony; 9 days after 3MI dosing; immunohistochemistry for PCNA. Mucosal epithelium remains disorganized; scattered epithelial cells mainly in Bowman's glands are labeled by immunohistochemistry for PCNA. Mayer's hematoxylin counterstain. Bar = 25 µm
Changes in 11βHSD activity in ethmoid turbinates are depicted in Fig. 10. 11βHSD1 activity was significantly lower 5 days after 3MI dosing than the value in control ponies. 11βHSD2 activity was significantly lower at 3 and 5 days after 3MI dosing than in the control ponies. Activity of pulmonary 11βHSD1 or 11βHSD2 in 3MI-treated ponies did not differ significantly from that in control ponies (data not shown).
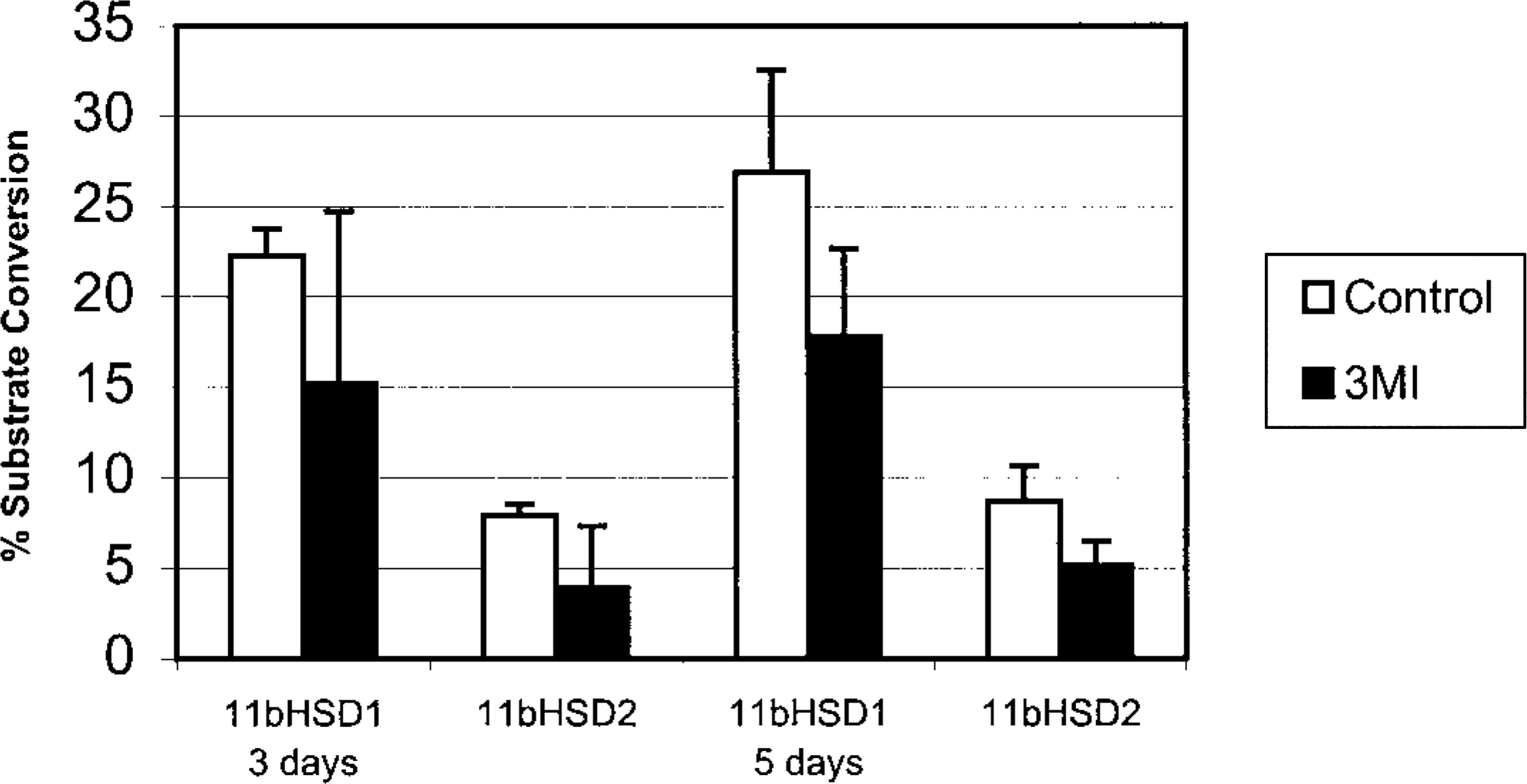
11βHSD activity in ethmoid turbinates of ponies after treatment with 3MI. Values are mean (SD) percent conversion of corticosterone to inactive 11-keto product
Discussion
3MI-induced bronchiolar lesions resembled those reported in ponies and horses 37 and were more severe than those reported in mice. 38 , 39 We cannot completely eliminate the possibility that arterial samples were contaminated with venous blood; however, decreased arterial oxygen tension in 3MI-treated ponies correlated with increased respiratory rate and expiratory dyspnea typical of heaves or equine recurrent airway obstruction. The clinical changes in respiration and decreased arterial oxygen tension in 3MI-treated ponies were attributed to bronchiolar rather than olfactory mucosal injury.
Nasal mucosal lesions have not been previously reported in horses or ponies. In this study, nasal mucosal damage was confined to areas lined by olfactory epithelium and was most severe 3 and 5 days after dosing with 3MI. Cell proliferation was evident by an increased mitotic index and positive immunohistochemistry test results for PCNA as early as 3 days after 3MI dosing, which diminished by 9 days postdosing. Olfactory mucosal injury had subsided by 9 days after 3MI dosing; however, scattered areas of epithelial disorganization with cellular hypertrophy, mitotic figures, and PCNA-labeling persisted. Immunohistochemistry was more sensitive than mitotic indices in detecting a proliferative response to cell injury, particularly in the epithelium of Bowman's glands.
In contrast to mice, in which 3MI induces obliterative nasal fibrosis but only temporary bronchiolar damage, ponies developed obliterative bronchiolitis but sustained only transient olfactory mucosal injury without scarring and epithelial metaplasia. Olfactory injury in ponies resulted in epithelial attenuation followed by mild hyperplasia, and disorganization with decreased severity of lesions by 9 days after 3MI dosing.
Equine olfactory mucosal injury was accompanied by decreased 11βHSD activity at 3 and 5 days after 3MI dosing. 11βHSD type II (11βHSD2) is known to inactivate glucocorticoids (cortisol or corticosterone), thereby preventing their inappropriate saturation of MRs and facilitating aldosterone binding. 15 Glucocorticoids have an antimitogenic effect on fibroblasts 12 and counteract the fibrogenic activity of aldosterone. Expression of MR and 11βHSD2 in the same cell is requisite for aldosterone selectivity and has been demonstrated in epithelial cells of olfactory mucosa 18 , 19 and intrapulmonary airways. 7 , 16 , 35 The presence of MR and the “gatekeeper” 11βHSD2 in olfactory and bronchiolar mucosa suggests a role for corticosteroids in modulation of the granulation tissue formation and fibrosis that can follow injury in these tissues. The decreased activity of 11βHSD2 in olfactory mucosa is not surprising because 3MI toxicosis targets the cellular sources of this enzyme. This decrease in 11βHSD2 activity might permit glucocorticoids to block fibroplasia, but fibroplasia would not have been expected, given the mildness of 3MI-induced olfactory mucosal injury in ponies.
Synthetic glucocorticoids are frequently administered by inhalation to treat nasal and pulmonary disease and have been shown to downregulate cytokine expression in fibroblasts, 23 to prevent nasal polyp formation and growth, and to diminish olfactory deficits. 26 Nevertheless, dexamethasone reportedly enhanced 3MI-induced olfactory receptor cell injury in rats, 20 which was attributed to cytochrome P450 induction. In murine olfactory mucosa damaged by 3MI, 11βHSD activity peaked during maximal granulation tissue formation, which was preceded by destruction of the epithelial sites of 11βHSD2, suggesting an alternative source of enzyme activity. 24
In horses, glucocorticoids are administered by injection or inhalation 17 , 21 , 29 , 30 to treat recurrent airway obstruction. Granulation tissue formation is a major component of the lesion of 3MI-induced bronchiolitis in horses, which contributes to small airway obstruction 37 , 41 and might be expected to respond to glucocorticoid therapy. 11βHSD2 has been identified and cloned from equine lung (Ganjam VK, Zhou MY, Johnson PJ, Chen YJ, Gomez-Sanchez CE: 11 Beta-hydroxysteroid dehydrogenase in the horse. Ann Meeting Endocrinol Soc, 1999). In our ponies, 3MI treatment did not alter pulmonary 11βHSD activity. Thus, we found no evidence of a role for glucocorticoids or mineralocorticoids in the pathogenesis of 3MI-induced bronchiolitis.
Results of this study indicate that bronchiolar epithelium is the major target of 3MI toxicosis in ponies. Olfactory mucosal lesions are deemed mild and presumably transient. Although selectivity for olfactory epithelium and Bowman's glands with sparing of nasal respiratory epithelium mirrors those lesions found in murine 3MI toxicosis, the mild degree of olfactory injury in the pony is in striking contrast to that in the mouse and suggests interspecies variation in binding and/or metabolism of this toxin.
Footnotes
Acknowledgements
This work was supported in part by the USDA Animal Health Formula Fund. We thank D. Dubé and D. Hatfield for technical support and H. Wilson and D. Connor for assistance in photographic preparation.
