Abstract
Hemochromatosis was presumptively diagnosed using cytologic examination of liver tissue from an aged male Miniature Schnauzer. The dog was presented after receiving whole blood transfusion every 6–8 weeks for 3 years to treat pure red cell aplasia. The cytologic specimen contained clusters of hepatocytes with abundant intracytoplasmic gold-yellow pigment granules and clumps of extracellular, green-black, globular pigment, both interpreted to be hemosiderin. Histologic sections of liver revealed hepatocellular degeneration with bridging portal fibrosis, lobular atrophy, biliary hyperplasia, and diffuse, severe hemosiderin accumulation. Serum iron and ferritin levels, and dry-weight iron concentrations of liver, heart, and kidneys were markedly increased. Hemosiderin accumulation was confirmed in hepatocytes of cytologic and histologic specimens using Perl's Prussian blue staining. This report is the first description of transfusional hemochromatosis in a dog and is the first to describe its cytologic appearance in a veterinary patient.
Iron overload disorders are classified as hemosiderosis, which is an increased deposition of tissue iron without clinical signs, or hemochromatosis, which manifests clinically as organ dysfunction secondary to iron-induced injury.
2
Hereditary hemochromatosis is a primary disorder of iron metabolism, and in human beings it involves mutations in the
A 13-year-old, intact male Miniature Schnauzer was presented to the Colorado State University Veterinary Teaching Hospital, cachectic and dehydrated. The 10-kg dog had been receiving whole-blood transfusions (200–500 ml) every 6–8 weeks for 3 years as palliative therapy for PRCA. The PRCA, diagnosed 3 years before presentation, was characterized by a moderate to severe nonregenerative anemia accompanied by a marked bone marrow erythroid hypoplasia. A complete blood count revealed a nonregenerative anemia (packed cell volume 13%, reference range 43–58%; reticulocytes 7,840/μl, reference range 0–60,000/μl) and a normal leukocyte count. The serum biochemistry panel demonstrated evidence of hepatocellular injury with increased alanine aminotransferase (361 IU/liter, reference range 10–120 IU/liter) and aspartate aminotransferase (234 IU/liter, reference range 16–50 IU/liter), and cholestasis with increased alkaline phosphatase (214 IU/liter, reference range 18–160 IU/liter) and γ-glutamyl transferase (20 IU/liter, reference range 0–10 IU/liter). Preprandial (61.5 μmol/liter, reference range < 15 μmol/liter) and postprandial (242.1 μmol/liter, reference range < 25 μmol/liter) bile acids were increased, indicating decreased hepatic function. The presence of concurrrent hypocholesterolemia (98 mg/dl, reference range 130–300 mg/dl) and hypoalbuminemia (2.1 gm/dl, reference range 3.0–4.5 gm/dl) suggested that the dog had hepatic failure. Ultrasound of the liver revealed a hyperechoic liver parenchyma, with nodules and irregular borders consistent with chronic fibrosis and cirrhosis.
Liver tissue aspirates were prepared on glass slides and stained with Wright-Giemsa. The samples were moderately cellular, with sheets of hepatocytes that were slightly vacuolated. Abundant intra- and extracellular, fine, gold-yellow pigment granules and large, extracellular, green-black, irregularly shaped globules were interpreted to be hemosiderin (Figs. 1, 2). Several specimens contained sheets of cohesive polygonal cells with a high N:C ratio that were interpreted to be mesothelial cells (oval nuclei with finely stippled chromatin and a pink glycocalyx border) or biliary epithelial cells (darker blue and less abundant cytoplasm) (Fig. 2). Rare aggregates of relatively uniform spindle-shaped cells with ovoid nuclei interpreted to be fibrocytes also were noted (Fig. 3). Perl's Prussian blue staining of liver aspirates revealed a diffuse blue granular pigment within the hepatocyte cytoplasm and scattered in the background, confirming the presence of iron. The large globular pigment stained dark blue to black (Fig. 4). A few mesothelial cells contained small amounts of faint intracytoplasmic blue pigment. A cytologic interpretation of hemosiderin accumulation with possible biliary hyperplasia and fibroplasia, consistent with hemochromatosis, was made.
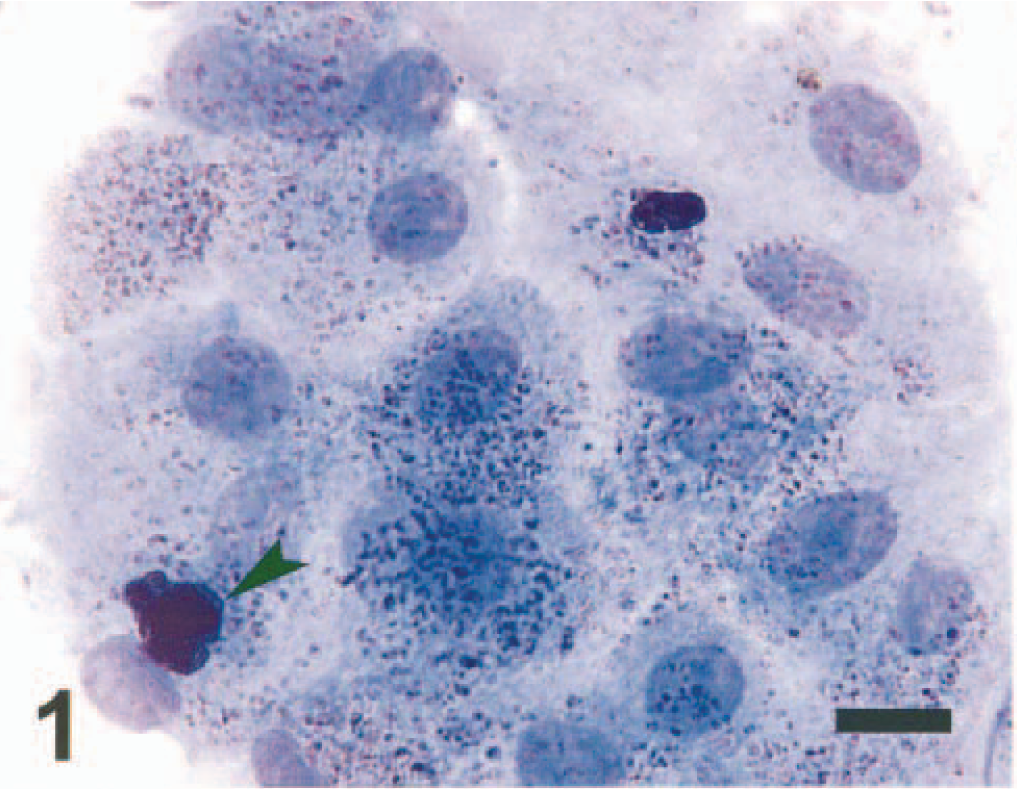
Hepatocytes; dog. A sheet of hepatocytes containing gold-yellow hemosiderin pigment granules and clumps of extracellular, green-black globular hemosiderin pigment (arrowhead). Wright-Giemsa. Bar = 18 μm.
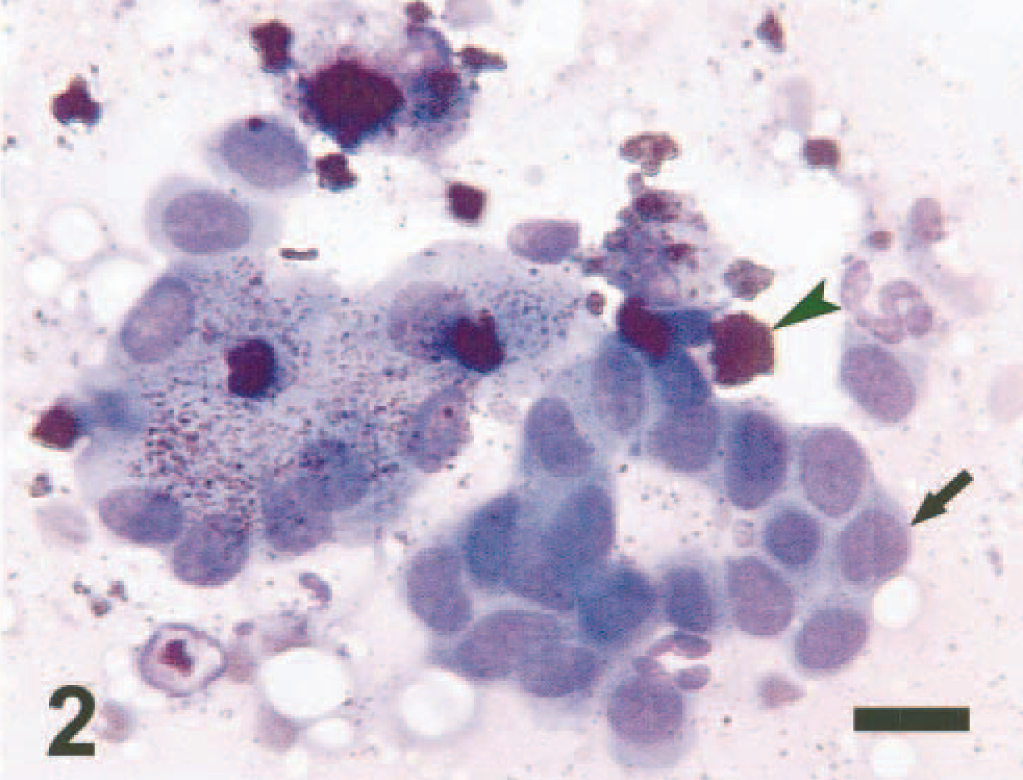
Hepatocytes and biliary epithelial cells; dog. Hepatocytes containing gold-yellow hemosiderin pigment granules are intermixed with biliary epithelial cells (arrow). Clumps of extracellular, green-black hemosiderin pigment granules can be seen in the background (arrowhead). Wright-Giemsa. Bar = 12 μm.
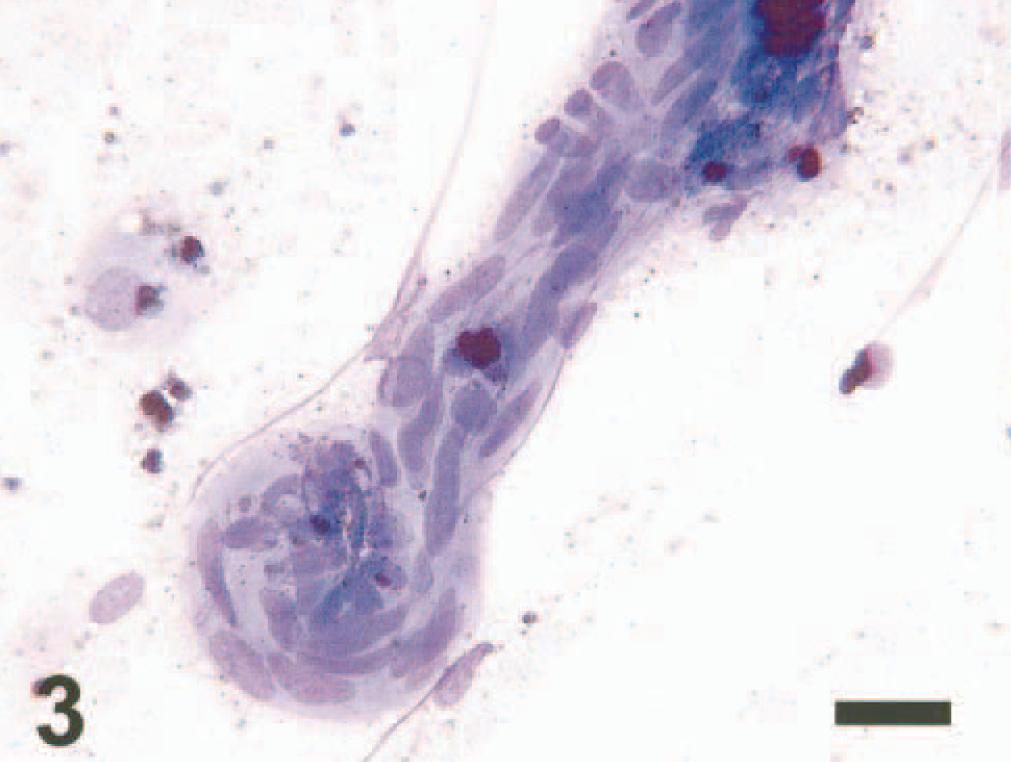
Fibrocytes; dog. Aggregate of fibrocytes and one macrophage containing clumped hemosiderin pigment. Wright-Giemsa. Bar = 15 μm.
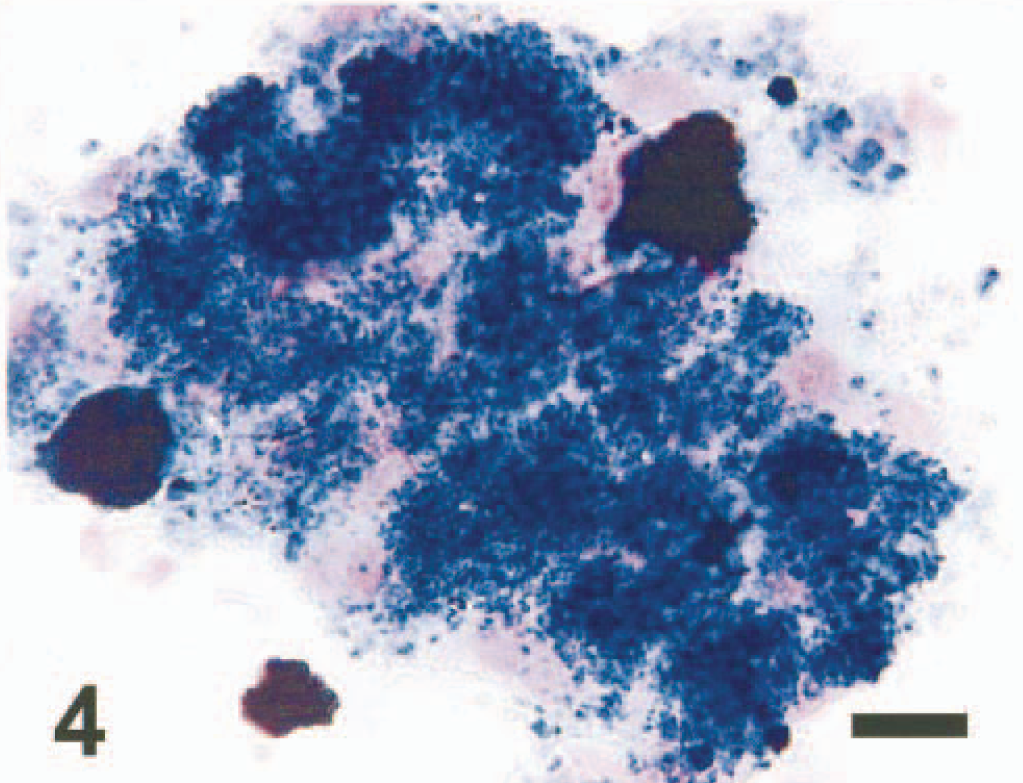
Hepatocytes; dog. Hepatocytes containing hemosiderin pigment granules and clumps of extracellular, globular hemosiderin pigment. Perl's Prussian blue. Bar = 18 μm.
Further support for iron overload with hemosiderin accumulation was based on an increased serum iron of 583 μg/dl (reference range 77–253 μg/dl), with a total iron-binding capacity of 588 μg/dl (reference range 275–443 μg/dl), an unbound iron-binding capacity of 5 μg/dl (reference range 75–313 μg/dl), and an iron saturation of 99% (reference range 20.5–72.5%). A diagnosis of liver failure due to iron toxicity was made, and the animal was started on iron chelation therapy with deferoxamine mesylate (Novartis Pharmaceuticals Co., East Hanover, NJ) infusions; however, the animal's condition continued to deteriorate, and it was euthanatized 6 weeks later.
At necropsy mild subcutaneous icterus was found. The left medial liver lobe was irregularly rounded. The mitral and tricuspid valves had moderate to severe endocardiosis with thickening of valve leaflets. Cross sections of ribs and long bones showed decreased marrow with replacement by adipose tissue. All other organ systems examined were free of gross lesions. Tissue samples were fixed in 10% neutral buffered formalin, routinely processed, and embedded in paraffin. Tissues from liver, heart, kidney, brain, and lung were sectioned to 5 μm and stained with hematoxylin and eosin (HE). Samples of liver, heart, and kidney were analyzed for dry-weight concentrations using the flame atomic absorption method (CSU Diagnostic Laboratory, Fort Collins, CO), and the results are listed in Table 1.
Toxicological analysis of liver, heart, and kidney.
∗ Tissue from one normal adult dog.
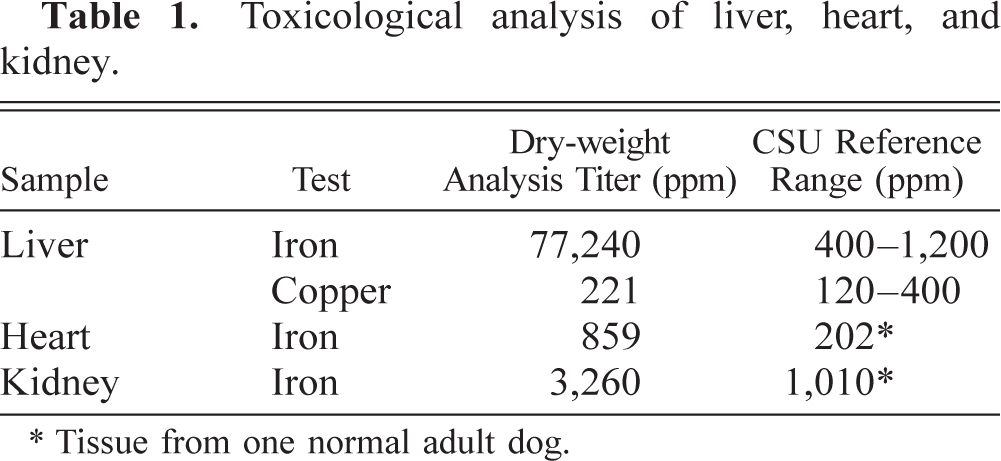
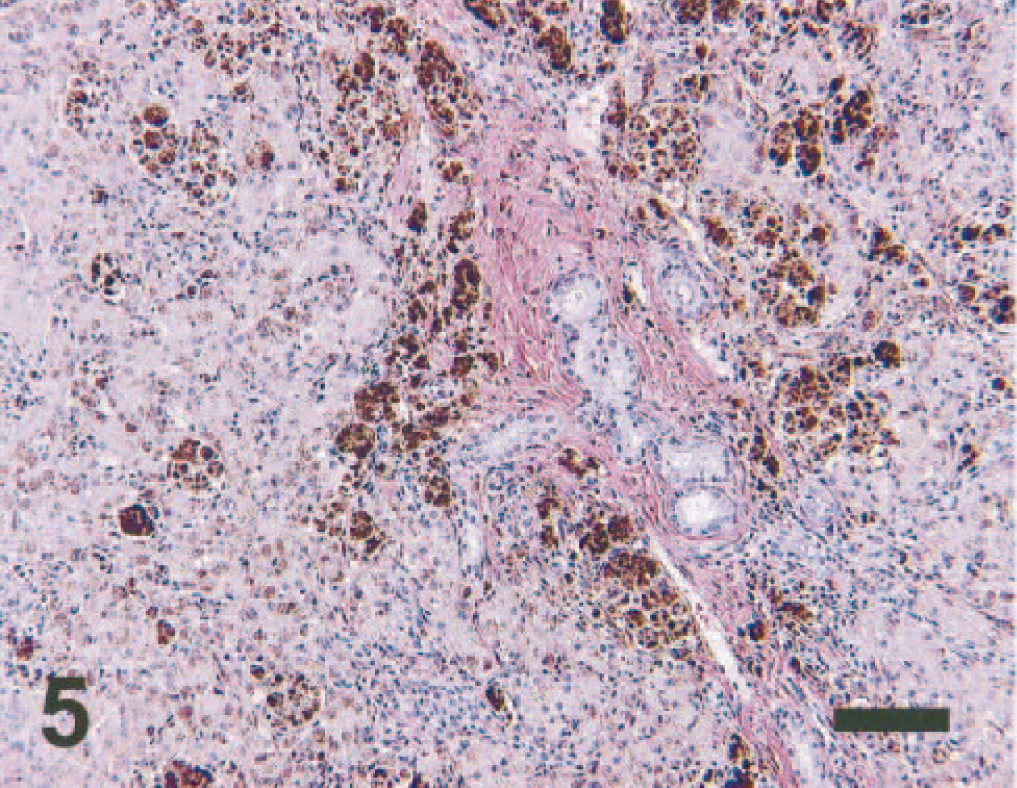
Histologic examination of liver revealed hepatocellular degeneration with bridging portal fibrosis, lobular atrophy, biliary hyperplasia, and diffuse, moderate to severe hemosiderin accumulation (Fig. 5). Lobular architecture was disrupted by fibrosis, atrophy, and clusters of macrophages and Kupffer cells that contained abundant dense dark brown to black globular pigment. Hepatocytes had intracytoplasmic, finely granular, dark brown to black pigment associated with the portal regions and extending into the surrounding lobules. Kidneys had subacute, mild to moderate tubular degeneration and necrosis with minimal lymphocytic, plasmacytic interstitial nephritis and scattered brown globular pigment. The medullary cords and subcapsular sinuses of peripheral lymph nodes were expanded by macrophages with dense dark brown to black globular pigment. The spleen had follicular hyperplasia with increased plasma cells and numerous pigment-laden macrophages in the white and red pulp. A section of pancreas contained disorganized pancreatic lobules with accumulations of dark brown to black pigment granules in macrophages and pancreatic acinar cells. There was marked atrophy of the thyroid gland, with many follicular epithelial cells containing brown, finely granular pigment. Positive staining for iron using Perl's Prussian blue staining method correlated with granular and globular pigment identified on HE-stained sections (Fig. 6). In support of the toxicology results, liver biopsy samples were negative for staining to detect copper using rubeanic acid stain.
Liver; dog. Fibrosis, lobular atrophy, biliary hyperplasia, and clusters of Kupffer cells and macrophages with expanded cytoplasmic borders and dense dark brown to black globular hemosiderin pigment. Hepatocytes contain intracytoplasmic, finely granular hemosiderin pigment. HE. Bar = 100 μm.
Liver; dog. Diffuse hemosiderin pigment in hepatocytes and globular hemosiderin pigment in macrophages and Kupffer cells. No hemosiderin is detected in biliary cells. Perl's Prussian blue. Bar = 100 μm.
Hemochromatosis has been reported in a variety of mammalian and avian species. Hepatic cirrhosis resembling hemochromatosis has been induced in dogs given chronic, large doses of parenteral iron sorbital or iron dextran for several years. 6 Iron overload leading to liver failure also has been described in Basenjis, secondary to pyruvate kinase deficiency. 3 Transfusional hemochromatosis has been described in people with refractory anemias associated with several disorders, including pure red cell aplasia, aplastic anemia, and leukemias. 5 , 8 This report is the first description of transfusional hemochromatosis in a dog.
The pathogenesis of iron overload in this case likely resulted from infusion of red blood cells, with absence of blood loss and subsequent accumulation of stored iron (hemosiderin). 1 Parenchymal cells become targets of iron deposition after transferrin has become saturated, which, in humans, can occur with only 10–15 units of transfused blood. 7 The mechanism of iron toxicity in the liver is not completely understood; however, it has been shown that iron, which can exist in both oxidized and reduced forms, can act as a potent catalyst in the production of hydroxyl radicals, resulting in lipid peroxidation of organelle membranes. 4 Fibrosis and collagen deposition is understood less well, and a causal relationship between iron deposition and fibrosis has not been firmly established. The diagnosis of hemochromatosis in this case, rather than hemosiderosis, was made because there was liver damage and decreased function secondary to iron deposition.
Cytologic impressions of hemochromatosis in liver have not been reported previously. In this case, hemochromatosis was presumptively diagnosed based on Wright-Giemsa–stained cytologic preparations of liver tissue aspirates, and there was a good correlation between cytologic and histologic interpretations. Cytologic preparations stained with Perl's stain can confirm the presence of intrahepatocyte hemosiderin and direct further diagnostic tests to verify the diagnosis of hemochromatosis.
Footnotes
Acknowledgements
We thank Dr. Martin David for critical review of the manuscript.
