Abstract
A 1-year-old intact female German Shepherd Dog was presented to the Veterinary Medical Teaching Hospital of the University of California–Davis for apparent cervical pain of 2 months duration. All vaccinations were current, travel history was restricted to northern California and Idaho, and the animal had been exposed to ticks periodically. No history of trauma was noted. On physical and neurologic examination, the dog was quiet and alert and had pain on movement or palpation of the head and neck. Results from complete blood count (CBC), serum chemistry analysis, urinalysis, thoracic radiographs, abdominal ultrasound examination, joint fluid analysis from several joints, and complete spinal radiographs were within normal limits. Cisternal cerebrospinal fluid (CSF) analysis revealed neutrophilic pleocytosis with increased protein (Table 1). Neutrophils were nondegenerate, and no organisms were seen on direct smears. Aerobic and fungal CSF culture were negative for pathogens. Serologic analyses for common infectious agents such as
A provisional diagnosis of pathogen-free inflammatory CNS disease was made, and symptomatic treatment was initiated with prednisone (1 mg/kg orally twice daily). The animal showed marked clinical improvement over the next 10 days but was presented again after 2 weeks with recurrence of the initial clinical signs. A CBC revealed leukocytosis (32,900 cells/µl, normal = 6,000–17,000 cells/µl) with neutrophilia (23,359 cells/µl, normal = 3,000–11,500 cells/µl) and monocytosis (1,654 cells/µl, normal = 150–1,350 cells/µl). A CSF analysis revealed increases in total nucleated cells and protein (Table 1). Myelography was normal. Aerobic and fungal cultures of the CSF were negative, and anaerobic culture revealed very small numbers of
Cerebrospinal fluid (CSF) analysis of a dog with CNS actinomycosis at presentation (sample 1) and 2 weeks after corticosteroid treatment (sample 2). Total nucleated cell counts and protein levels increased following symptomatic treatment.
On necropsy, the superficial blood vessels of the cerebral cortex were hyperemic, and the cerebellum was mildly coned, protruding slightly through the foramen magnum. There was approximately 1 ml of purulent material in the subdural space, mainly located over the brain stem. The leptomeninges overlying the midbrain, pons, and medulla, and the dura mater lining the corresponding ventral portions of the calvarium were thickened, hyperemic, roughened, and opaque. Sagittal sections of the brain showed symmetrically dilated lateral ventricles, consistent with moderate hydrocephalus. The superficial blood vessels of the spinal cord were diffusely engorged. The head, including oral cavity and teeth, ear canals, tympanic bullae, nasal choanae, and nasal sinuses, was examined, and no evidence of inflammation or trauma was found in any of these locations. The dog also had multiple dark red, 0.5-cm-diameter, consolidated pulmonary nodules located in the periphery of the right and left cranial lung lobes.
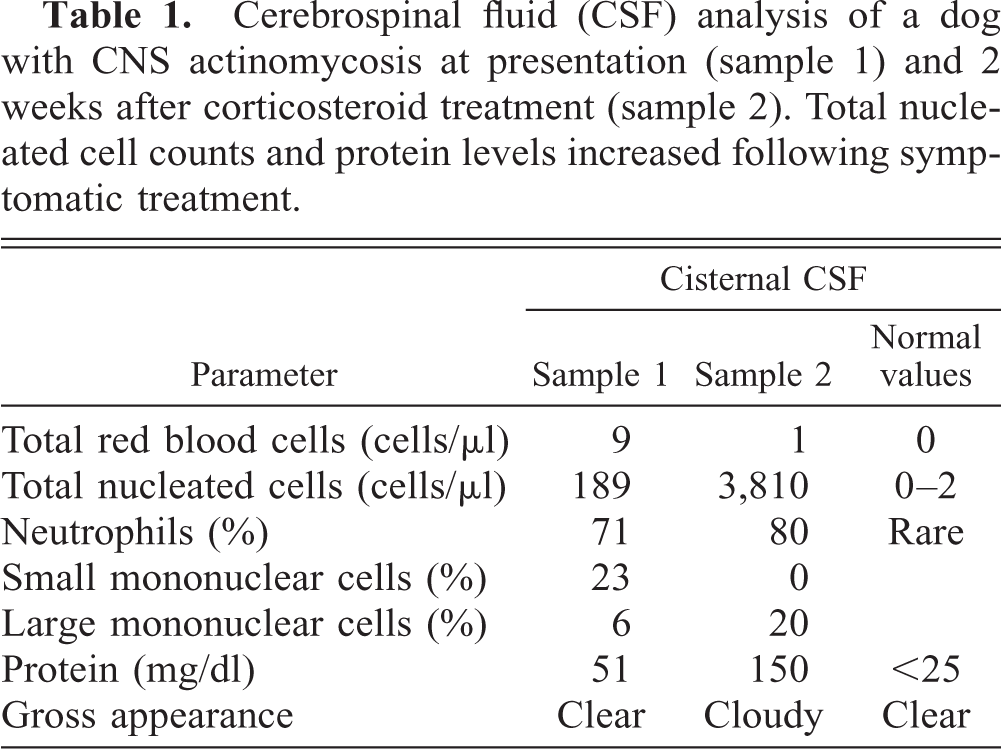
Cytology smears of the CSF obtained from the subdural space and stained with Diff Quik (Baxter Scientific Products, Mc Gaw, IL) showed abundant neutrophils and a few aggregates of degenerated neutrophils (Fig. 1). These aggregates were characterized by radiating gram-positive, acid-fast–negative filamentous organisms surrounded by a corona of elongated club-shaped neutrophils. Culture of the CSF was performed in 5% sheep blood agar with 5% carbon dioxide at 37C, and a pure culture of
Cerebral spinal fluid: dog. Central radiating aggregate of
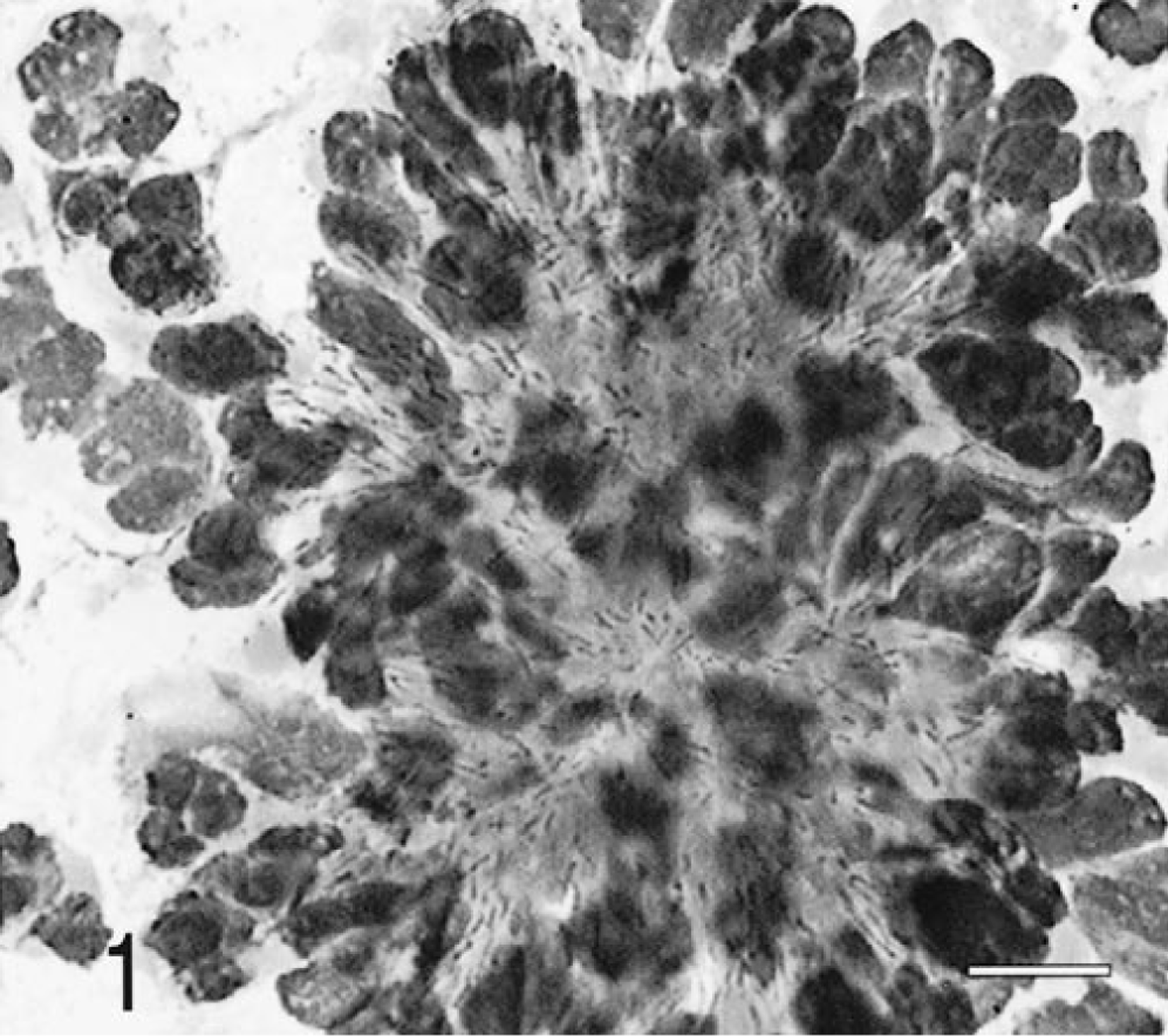
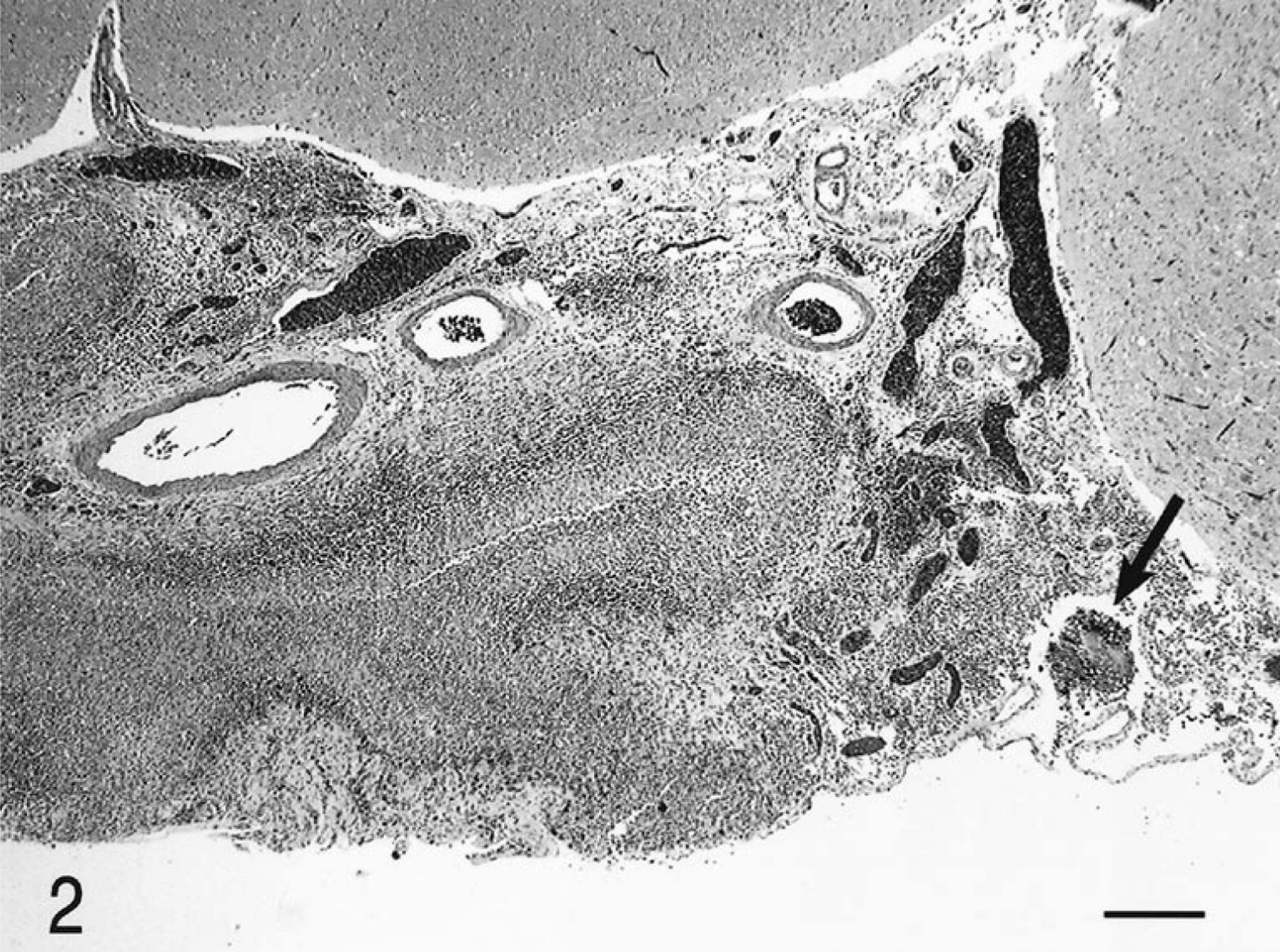
Histologically, there was severe suppurative and granulomatous meningoencephalitis and ventriculitis that involved the brain stem and the subependymal white and gray matter along the lateral third and fourth ventricles (Fig. 2). Rare radiating bacterial colonies surrounded by neutrophils and a Splendore-Hoeppli reaction were found within the meninges of the caudal brain stem (Fig. 3). The pulmonary nodules were areas of bronchopneumonia, characterized by hemorrhage, and neutrophilic infiltrates surrounding small airways. There also was a single small granuloma within the pulmonary parenchyma, characterized by an accumulation of epithelioid macrophages surrounded by a rim of lymphocytes. No organisms were seen within this granuloma with Giemsa, Brown and Brenn, or acid-fast (Fites Faraco) stains.
Brain and meninges; dog. Severe pyogranulomatous meningitis with a bacterial colony (arrow). HE. Bar = 700 µm.
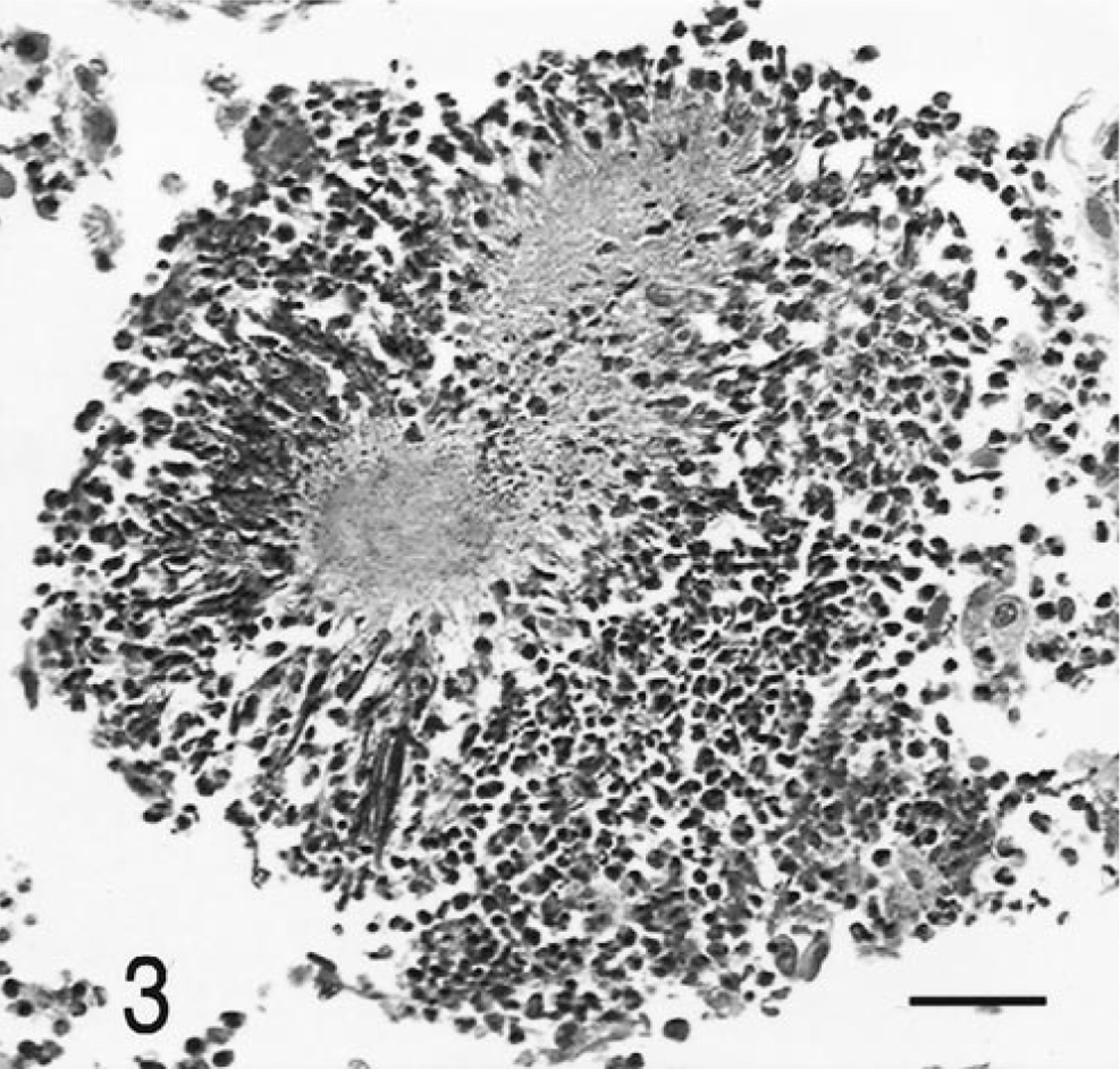
Meninges; dog. Higher magnification of Fig. 2, with radiating aggregate of
Actinomycosis of the CNS in animals has not been previously confirmed by culture of the organism. In the previously reported cases of meningitis in a fox and a dog, the diagnosis was assumed based on the histologic appearance of the filamentous organisms. Furthermore, both animals had facial or head bite wounds that probably were the source of infection. The present case is unusual in the absence of previous history of trauma or other sites of infection. The multifocal bronchopneumonia affected mainly cranioventral portions of the lungs and was interpreted to be due to aspiration, secondary to seizures. These lesions were acute and not considered to be associated with the actinomycosis. The single small pulmonary granuloma was a resolving lesion that was considered unrelated because of its chronicity and the absence of microorganisms.
This case is also unusual because of the disparity of bacterial culture results ante- and postmortem.
Because there was no history of trauma, a migrating foxtail (seed of
Hydrocephalus was common to both this case and the previously reported canine case and is likely secondary to blockage of the mesencephalic aqueduct by the florid pyogranulomatous ventriculitis. Therefore, some of the chronic, progressive, poorly localized neurologic signs may be caused by secondary hydrocephalus in dogs with CNS actinomycosis.
Footnotes
Acknowledgements
We thank Dr. R. J. Higgins for valuable assistance with photomicroscopy.
