Abstract
We investigated the embryotoxic and fetotoxic effects of Poincianella pyramidalis (Tul.) L.P. Queiroz (syn. Caesalpinia pyramidalis Tul.) leaves on pregnant rats (Rattus norvegicus). Pregnant rats were divided into 4 groups: G1—fed a ration containing 5% P. pyramidalis leaves beginning on day 1 of pregnancy; G2—fed a ration containing 5% P. pyramidalis leaves commencing on day 6 of pregnancy; G3—fed a ration containing 10% P. pyramidalis leaves starting on day 6 of pregnancy; G4—control group. On day 21 of pregnancy, parameters of reproductive performance were recorded. Fetal growth was measured, and fetuses were subjected to external examination and identification of skeletal anomalies. Rations containing P. pyramidalis resulted in dead or undeveloped fetuses and reduced the number, length, and weight of the fetuses. Rations also increased postimplantation losses and the frequency of skeletal anomalies. Furthermore, P. pyramidalis was also responsible for dose-dependent lesions in the placentas. In conclusion, the ingestion of P. pyramidalis promotes fetal and placental damage.
Introduction
Poincianella pyramidalis (Tul.) L.P. Queiroz (syn. Caesalpinia pyramidalis Tul.) is a tree belonging to the family Fabaceae (Leguminosae), which is endemic to northeastern Brazil, especially in the Caatinga biome. 16 It is popularly known as “catingueira,” “pau-de-rato,” “catinga-de-porco,” or “mussitaiba.” The leaves are used in the preparation of infusions and decoctions by the local population as diuretics and antidyspeptics, as well as for the treatment of fever.1,4 Additionally, the aqueous leaf extract of P. pyramidalis has been used to treat gastrointestinal nematodes in goats, with promising results. 3 Pharmacologic studies have shown that aerial parts of the plant have anti-inflammatory, 18 antifungal, 7 and antimicrobial properties. 19 Its pollen has been proven to be toxic to honeybees (Apis melifera). 15
In northeastern Brazil, the occurrence of malformations, embryonic death, and poor fetal development in ruminants has been associated with the ingestion of the leaves of Senegalia (Mimosa) tenuiflora.8,17 However, in the state of Rio Grande do Norte, also located in the Brazilian semiarid region, some outbreaks of malformations in small ruminants were observed on farms where S. tenuiflora trees were absent (Câmara ACL, Riet-Correa F, unpublished data). On these farms, P. pyramidalis trees are abundant and their leaves are eaten by livestock during drought periods. Therefore, the aim of our study was to determine if the leaves of P. pyramidalis were embryotoxic and fetotoxic when fed to pregnant rats (Rattus norvegicus).
Materials and methods
Mature leaves of P. pyramidalis (Tul.) L.P. Queiroz were collected from a farm where a spontaneous outbreak of malformations in sheep was reported. The farm was located in the municipality of Assu, Rio Grande do Norte, Brazil. The leaves were dried at ambient temperature, protected from sunlight, and ground using a milling machine. The rations for the experimental groups were prepared by weight, with 80% commercial pelleted rat food a ground and mixed with 5–10% ground P. pyramidalis leaves and 10–15% cornstarch in hot water. After mixing the leaves and ration with the starch, pellets were made by hand and dried in the shade. The ration for the control group was prepared in the same way, but with 90% commercial pelleted rat food and 10% cornstarch.
Thirty adult female Wistar rats and 15 adult male Wistar rats were provided by the animal facility of Universidade Federal Rural do Semi-Árido, Mossoró, Rio Grande do Norte, Brazil. The rats were housed in 40 × 50 × 20 cm plastic cages, under a controlled temperature (22–26°C), in a natural cycle of light. All animal procedures were approved by the Bioethics Committee at the Federal University of Campina Grande, Patos, Paraiba (activity CEP 69-2013), and performed under veterinary supervision.
For mating, 1 male and 2 females were housed in a cage for a 12-h period (from 6:00 PM to 6:00 AM). The presence of sperm cells in vaginal washes was regarded as day 1 of pregnancy. After the confirmation of pregnancy, every pregnant female rat was placed alone in a cage until day 21 of pregnancy.
Pregnant rats were randomly divided into 4 groups: group 1 (G1) with 7 females fed a ration containing 5% P. pyramidalis leaves starting on day 1 of pregnancy; group 2 (G2) with 8 females fed a ration containing 5% P. pyramidalis leaves starting on day 6 of pregnancy; group 3 (G3) with 7 females fed a ration containing 10% P. pyramidalis leaves starting on day 6 of pregnancy; and the control group (G4) composed of 8 females fed the same ration but without P. pyramidalis leaves. G2 and G3 rats were fed only commercial rat food from day 1 to day 5 of pregnancy to prevent interference with embryo implantation. They were only fed the experimental diets from implantation through major organogenesis (days 6–15) and late pregnancy (days 16–21) periods. 12 Weekly food consumption and water intake were determined on days 7, 14, and 21, and on the same days the rats were weighed.
On day 21 of pregnancy, the rats were anesthetized by intramuscular administration of xylazine (8–15 mg/kg) and ketamine (60–80 mg/kg), and the ovaries and uteri removed after abdominal incision. The incision was enlarged, penetrating the thoracic cavity, and the dams were euthanized by heart perforation. Fragments of lung, liver, spleen, heart, thymus, kidney, uterus, and placentas were collected and fixed in 10% buffered formalin, embedded in paraffin, sectioned at 5 μm, and stained with hematoxylin and eosin for histologic examination. Additionally, fragments of the placentas and uteri were Gram-stained for identification of bacteria.
The whole gravid uterus from each dam was weighed. The number of corpora lutea in each ovary was recorded. The number of implantation sites and resorptions was recorded in both uterine horns, 12 and the fetuses and the placentas were removed. The number and viability of the fetuses were recorded. All fetuses were weighed, cleaned of amniotic fluids, measured, and subjected to external examination. The fetuses were then euthanized with isoflurane and immersed in acetone for 24 h, examined for cleft palate, and eviscerated. For identification of skeletal anomalies, all eviscerated fetuses were submersed in a solution of 0.8% potassium hydroxide with alizarin red S, which was changed daily for 3–4 days, and then cleared and stored in a solution of 40% ethanol, 40% glycerin, and 20% benzyl alcohol. 22
The obtained data were statistically analyzed using R software (version 3.0.3; https://www.r-project.org/). The normality of the data was evaluated using the Shapiro–Wilk test, and the homogeneity of variance was evaluated using the F test. Data on weight, weight gain, food and water intake, and the parameters of reproductive performance were compared using the Kruskal–Wallis test followed by the Student–Newman–Keuls test. The frequencies of malformations were compared using Fisher exact test. P value of <0.05 was considered statistically significant.
Results
The weights of the rats showed a significant difference between the groups only on day 21 of pregnancy, when the G1 and G3 rats weighed less (p < 0.05) and gained less weight (p < 0.05) than the G4 rats (Table 1). The body weights of animals in G3 decreased, and 1 rat died on day 16 of pregnancy as a result of inanition. Feed intake was less (p < 0.05) in G3 rats than in the G2 and G4 groups (Table 1). The intake of experimental ration containing P. pyramidalis leaves was well tolerated by rats in the G1 and G2 groups but poorly tolerated by those in the G3 group. Water intake was less (p < 0.05) in G1 rats than in the G2 and G4 groups. None of the females delivered prematurely during the experiment.
Weights, weight gains, and food and water intakes in rats fed rations containing Poincianella pyramidalis leaves (G1: 5% from day 1 of pregnancy; G2: 5% from day 6 of pregnancy; G3: 10% from day 6 of pregnancy; and G4: control group).*
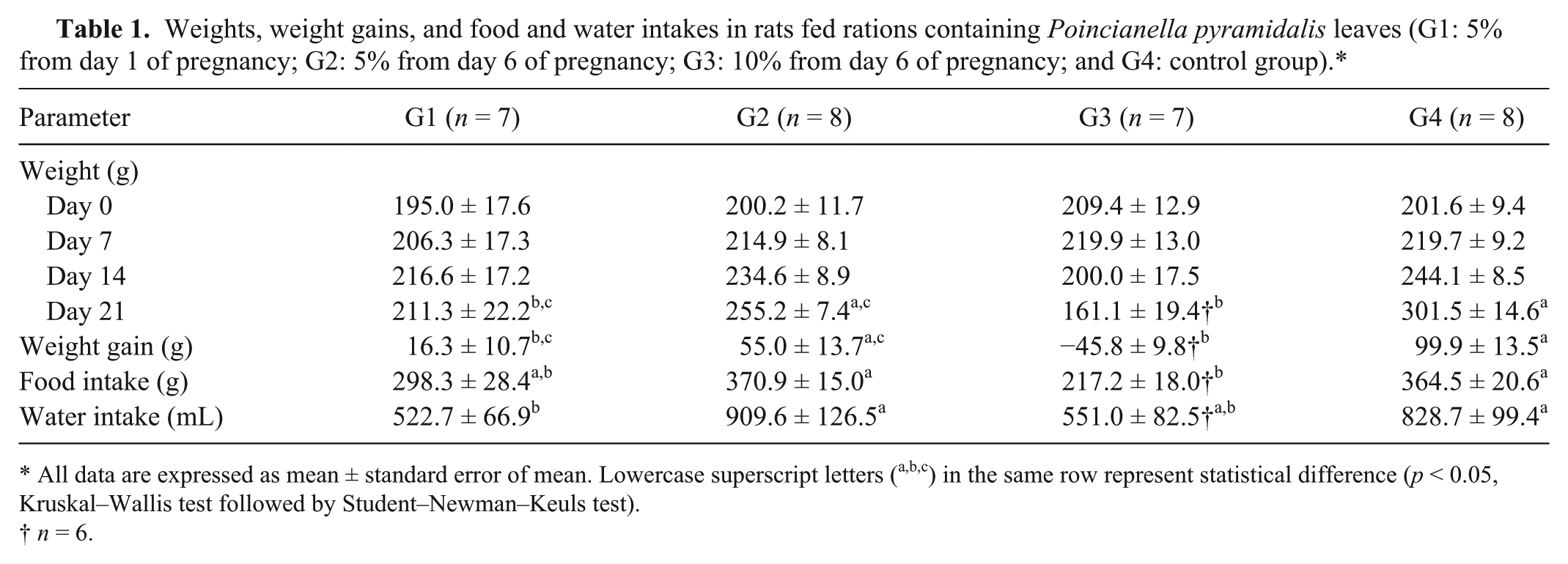
All data are expressed as mean ± standard error of mean. Lowercase superscript letters (a,b,c) in the same row represent statistical difference (p < 0.05, Kruskal–Wallis test followed by Student–Newman–Keuls test).
n = 6.
Uterine weights were less (p < 0.05) in G1 and G3 rats than in G4 rats, and the weights of rats without the uterus were less (p < 0.05) in G3 rats than in G2 and G4 rats (Table 2). There were also no significant differences in the numbers of corpora lutea and implantations. There were fewer (p < 0.05) live fetuses in the G1 group than in the G4 group, and the G3 group had no live fetuses. Dead fetuses were observed in all P. pyramidalis experimental groups, but no dead fetuses were observed in the G4 group. The weights and lengths of the live fetuses and the weights of the placentas were less (p < 0.05) in the G1 group than in the G2 and G4 groups. There were no significant differences in preimplantation losses between the different groups, but postimplantation losses in the 3 treated groups (G1, G2, and G3) were greater (p < 0.05) than in the G4 group.
Measurements associated with reproduction in female rats fed rations containing Poincianella pyramidalis leaves (G1: 5% from day 1 of pregnancy; G2: 5% from day 6 of pregnancy; G3: 10% from day 6 of pregnancy; and G4: control group).*
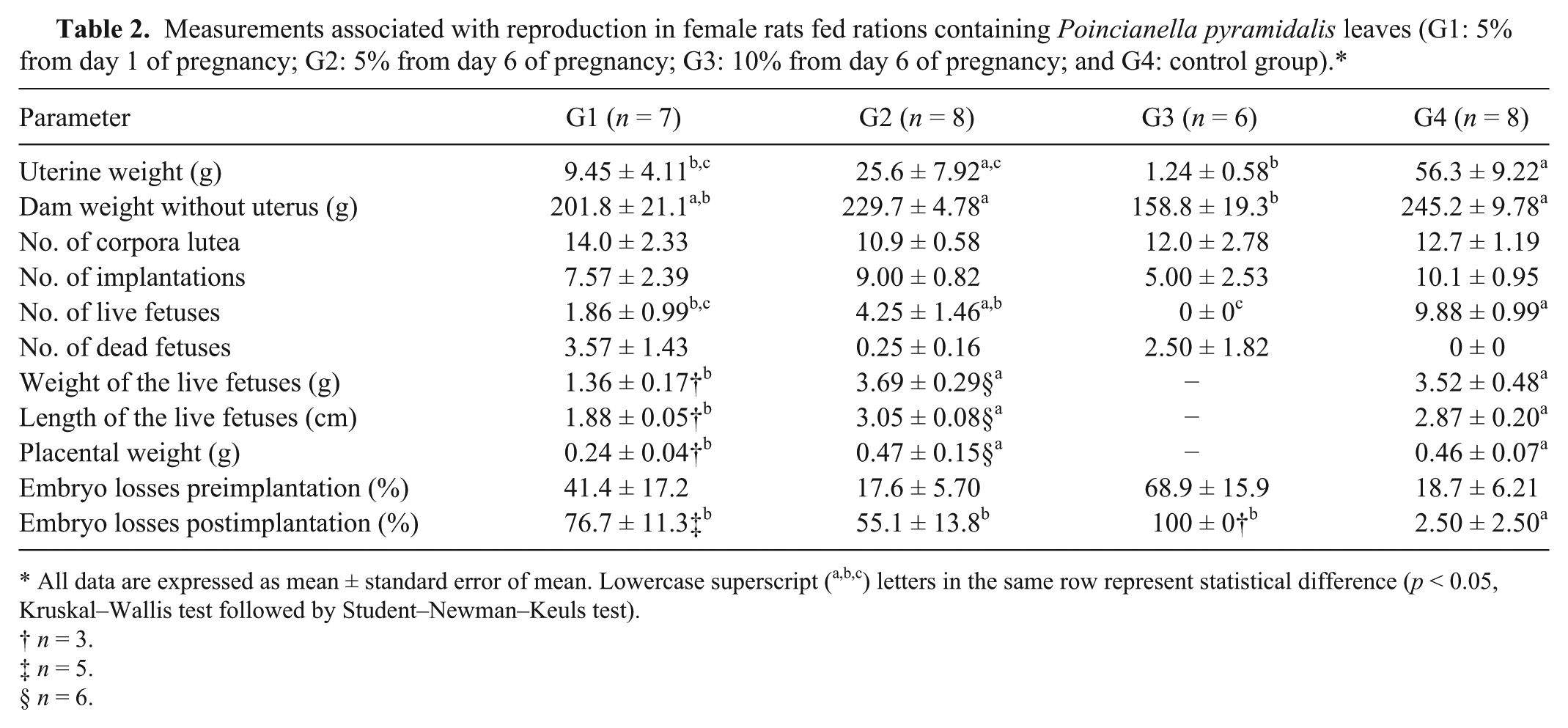
All data are expressed as mean ± standard error of mean. Lowercase superscript (a,b,c) letters in the same row represent statistical difference (p < 0.05, Kruskal–Wallis test followed by Student–Newman–Keuls test).
n = 3.
n = 5.
n = 6.
Forty-one bone malformations were observed in 31 of 57 fetuses in the experimental groups, and 7 malformations were observed in 7 of 79 fetuses in the control group (Table 3). Only 1 fetus had a cleft palate and this was in the G2 group. The G3 group was not included in the evaluation of skeletal anomalies because the females had no fetuses of adequate size for analysis. The frequencies of aplasia of sternebrae, arthrogryposis, wavy ribs, and fusion of cervical vertebrae showed significant differences (p < 0.05).
Frequencies of skeletal anomalies in live and dead fetuses from rats fed rations containing Poincianella pyramidalis leaves (G1: 5% from day 1 of pregnancy; G2: 5% from day 6 of pregnancy; and G4: control group).*
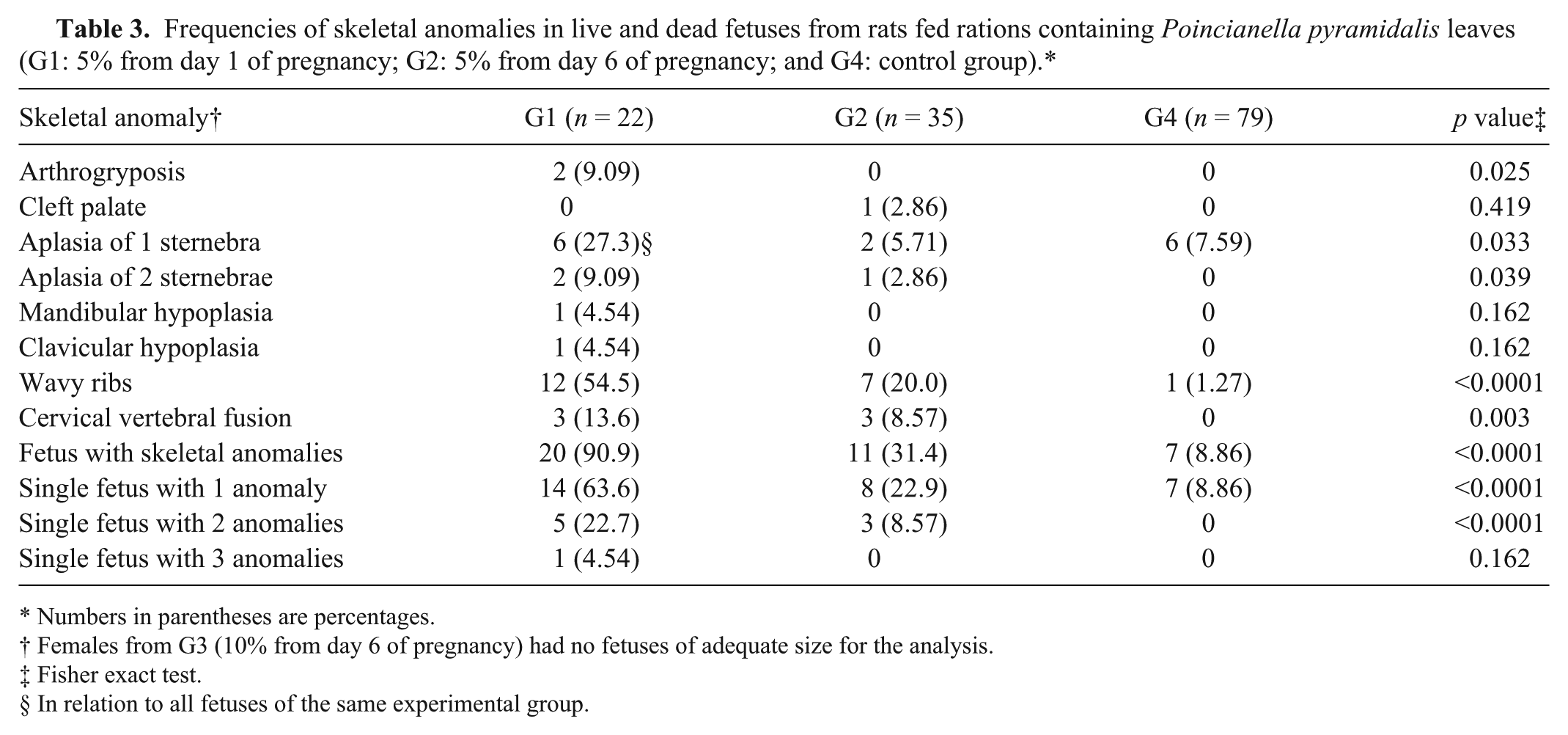
Numbers in parentheses are percentages.
Females from G3 (10% from day 6 of pregnancy) had no fetuses of adequate size for the analysis.
Fisher exact test.
In relation to all fetuses of the same experimental group.
Autopsies of G1 rats revealed brown vaginal discharge (n = 3), dark red foci in uterine implantation sites (n = 2), and a uterus with hemorrhagic content (n = 1). G2 rats revealed white discoloration on the superficial portion of fetal placentas (n = 3), placental discs with hemorrhagic black surface (n = 2), and dark red foci in uterine implantation sites (n = 2). Females from the G3 group showed similar but more severe placental and uterine changes than the G1 group, and also edematous and friable uteri (n = 3); 1 dam had intestinal tympany. Histologically (Table 4), rats from the G1 and G2 groups had primary lesions restricted to the placenta, characterized by moderate-to-severe trophoblast giant cell necrosis (Fig. 1A) and mild-to-severe suppurative inflammation of adjacent decidua (Fig. 1B); in some animals, reduction of glycogen cells in the spongiotrophoblast was also observed (Fig. 2). Females from the G3 group had more severe placental changes than G1 and G2 rats. Furthermore, in G3 rats, mild-to-severe neutrophilic inflammation and edema in uterus mucosa were observed (Fig. 1B). Bacteria were not present in histologic sections from the placentas and uteri stained with Gram stain. Significant microscopic changes were not observed in the liver, spleen, lungs, heart, and kidneys in any group. No gross or histologic lesions were observed in females in the G4 group.
Histologic changes in the placenta and uterus from female rats fed rations containing Poincianella pyramidalis leaves (G1: 5% from day 1 of pregnancy; G2: 5% from day 6 of pregnancy; G3: 10% from day 6 of pregnancy; and G4: control group).*

+ = presence of pathologic changes; – = absence of pathologic changes.
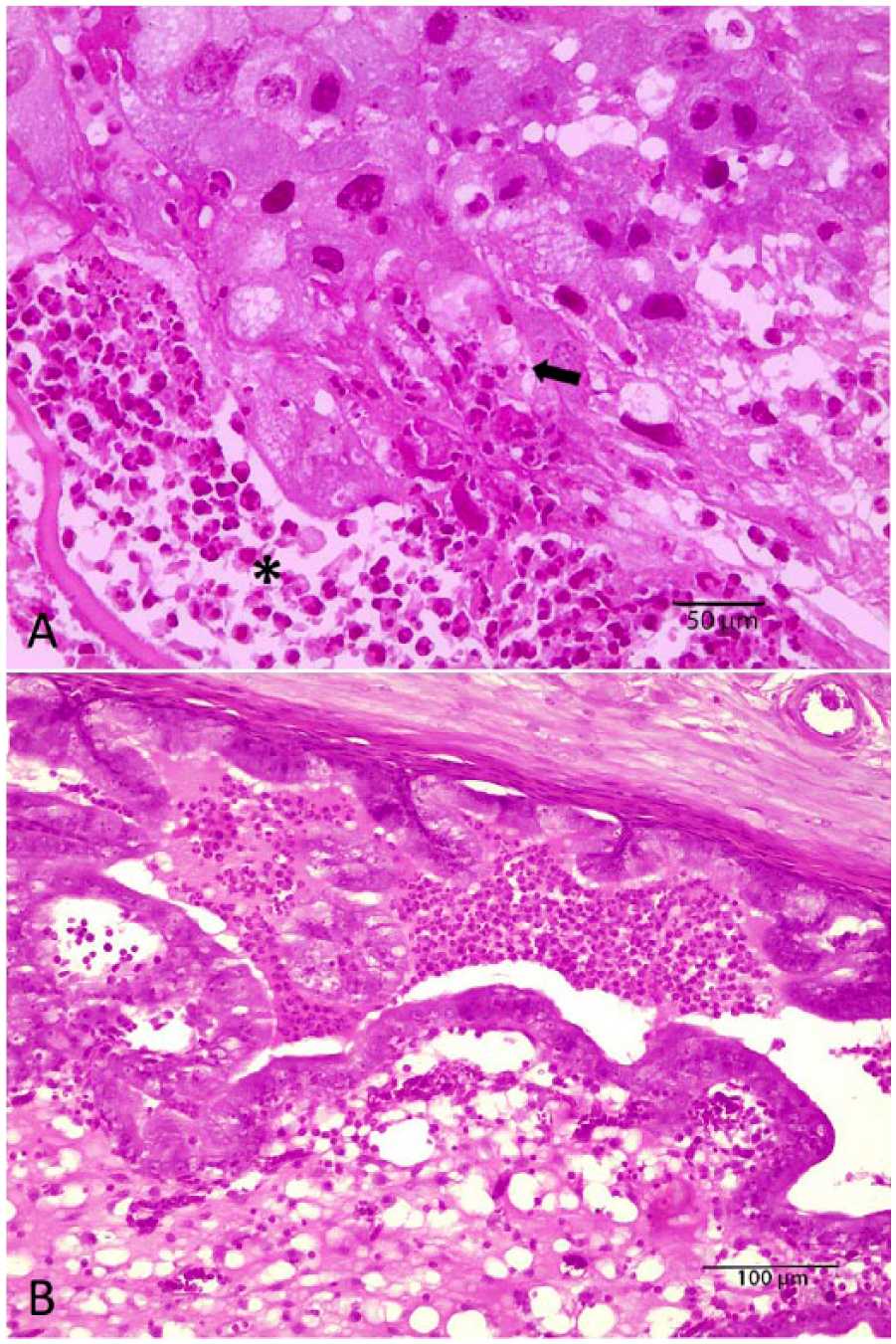
Placenta showing (
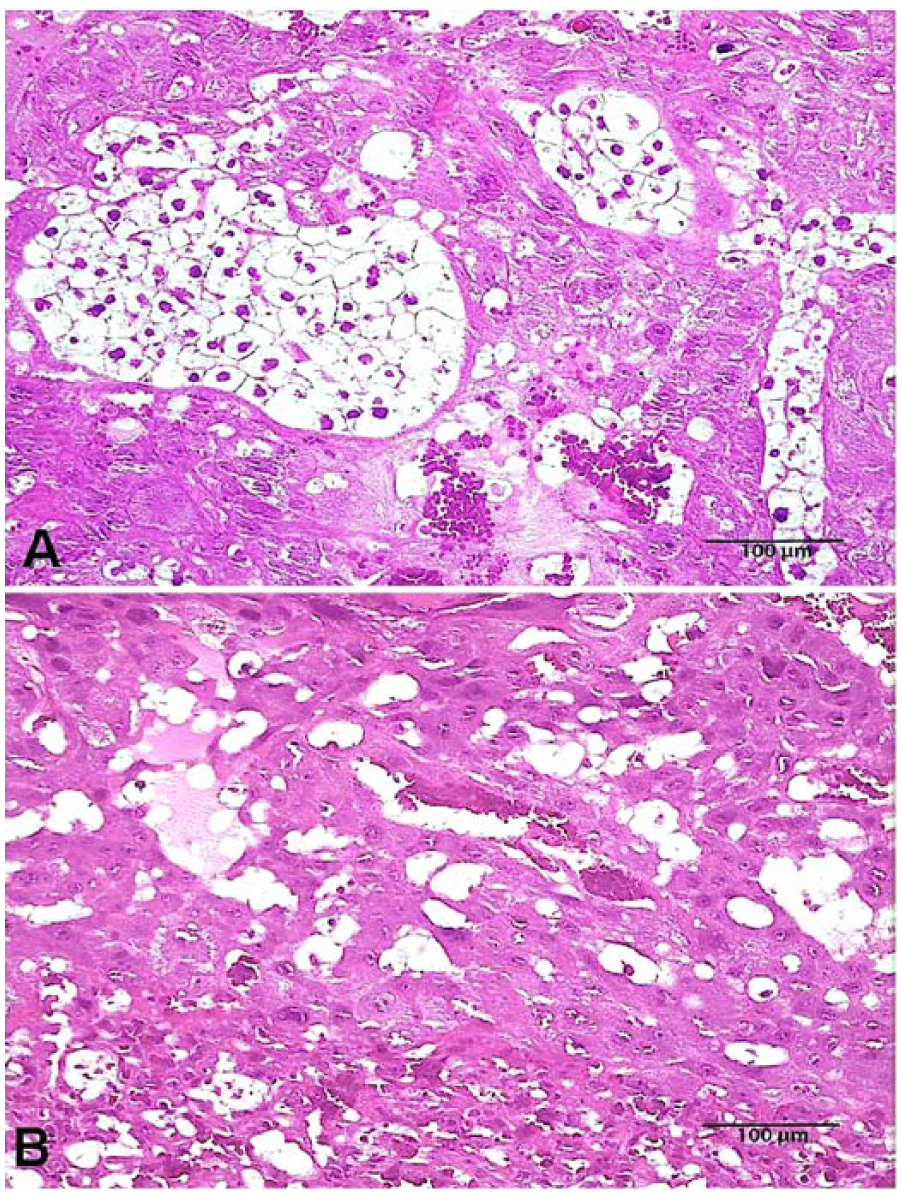
Normal (
Discussion
It is well known that maternal toxicity can interfere with maternal–fetal interactions, negatively affecting fetal development.2,5,13 In our study, the consumption of ration containing 10% P. pyramidalis leaves for 15 days caused severe maternal toxicity, whereas an inclusion rate of 5% P. pyramidalis leaves for 15 and 20 days caused only slight maternal toxicity. However, the overall results show 10% of leaves for 15 days, 5% of leaves for 20 days, and 5% of leaves for 15 days caused very severe, severe, and moderate disruption of fetal development, respectively, showing an apparently dose and time interaction of toxic effects in fetuses.
Administration of P. pyramidalis to pregnant rats was responsible for impaired fetal development, skeletal anomalies in the fetuses, and prenatal mortality. Reduced fetal weight and viability are some of the most important parameters measured in studies of developmental toxicity. 6
Skeletal anomalies promoted by P. pyramidalis in our study included sternebral aplasia, cervical vertebral fusion, wavy ribs, and arthrogryposis. Another plant that promotes malformations similar to these is S. tenuiflora. In rats, the reported malformations include sternebral malformations, cleft palate, scoliosis, and cranial anomalies (concavity of parietal, interparietal, and supraoccipital bones; hypoplasia of nasal bone; and microphtalmia). 14 The malformations promoted by S. tenuiflora are attributed to the tryptamine alkaloids N-methyltryptamine and N,N-dimethyltryptamine. 10 It is not known whether the leaves of P. pyramidalis contain these compounds.
Fetuses with skeletal contracture and cleft palate may be a result of chemicals that reduce fetal movement. This effect might be produced by the activation and prolonged desensitization of fetal muscle–type nicotinic acetylcholine receptors. 11 N,N,N-trimethyltryptamine and 5-hydroxy-N,N,N-trimethyltryptamine, which are produced after N-methylation of N-methyltryptamine and N,N-dimethyltryptamine, are considered agonists of nicotinic acetylcholine receptors. 20 Thus, it is feasible that N-methyltryptamine and N,N-dimethyltryptamine or their products of transformation could reduce fetal movement.
Several compounds are known to promote placental damage, which may interfere with embryonic and fetal development.9,21 In the present study, the damage to placentas promoted by P. pyramidalis was evidenced macroscopically by the reduced placental weight, the presence of white discoloration on the superficial portion of the fetal placenta, and placental discs with hemorrhagic black surfaces. Furthermore, microscopic lesions in the placentas of treated rats supported this observation, showing moderate-to-severe trophoblast giant cell necrosis and reduction of glycogen cells in the spongiotrophoblast. Mild-to-severe suppurative inflammation of adjacent decidua could be attributed to the response to necrosis of tissues because no bacteria were observed within the lesions. Histopathologic changes observed, especially the trophoblast giant cell layer necrosis, were possibly associated with impaired fetal development and prenatal mortality. In addition, the unknown toxic substance contained in the leaves of the plant could also cause embryotoxic changes promoting malformation and contributing to prenatal mortality. Therefore, the toxic effects of P. pyramidalis leaves on the fetuses could be partially attributed to damage induced in the placenta affecting the transport of nutrients, oxygen, and waste products between mother and fetus.
Footnotes
Authors’ contributions
ACL Câmara, ICN Gadelha, MB Castro, RMT Medeiros, F Riet-Correa, and B Soto-Blanco contributed to conception and design of the study, and contributed to acquisition, analysis, and interpretation of data. ACL Câmara drafted the manuscript. MB Castro, F Riet-Correa, and B Soto-Blanco critically revised the manuscript. ACL Câmara, MB Castro, RMT Medeiros, F Riet-Correa, and B Soto-Blanco gave final approval. ACL Câmara agreed to be accountable for all aspects of the work ensuring that questions relating to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
a.
Labina, Purina, São Lourenço da Mata, Pernambuco, Brazil.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was funded by National Council of Scientific and Technological Development (CNPq), National Institute of Science and Technology (INCT) for the study of the control of poisoning by plants.
