Abstract
Equine herpesvirus 9 (EHV-9), a new neurotropic equine herpesvirus, was inoculated intranasally at 107 plaque-forming units in five dogs to assess its pathogenicity. Dogs showed weight loss, pyrexia, anorexia, and neurologic signs on the fourth day. The EHV-9 virus was recovered from the examined brains. Histologically, dogs had a fulminant nonsuppurative encephalitis characterized by severe neuronal degeneration and loss, with intranuclear inclusions, slight glial reactions, perivascular cuffing, and multifocal hemorrhage. The olfactory bulb and the frontal and temporal lobes were predominantly affected. Immunohistochemistry revealed reactivity for EHV-9 antigen in neurons. All dogs had mild bronchopneumonia and various degrees of lymphoid necrosis. These findings indicate that dogs are fully susceptible to EHV-9 and that EHV-9 can cause fulminant encephalitis with high mortality in dogs, as in gazelles and goats.
In 1993, a highly neurotropic herpesvirus named gazelle herpesvirus 1 (GHV-1) was isolated from brains with a fulminant encephalitis in a herd of Thomson's gazelles (Gazella thomsoni) maintained in a zoo. 5 , 11 This new type of virus was closely related immunologically to equine herpesvirus 1 (EHV-1), but its DNA fingerprint differed from those of EHV-1 and other equine herpesviruses. 5 The DNA of the virus was estimated to have 95% homology to EHV-1 and EHV-8 and 60% homology to EHV-4 on the basis of the analysis of the glycoprotein G and the conserved region of glycoprotein B gene sequences. 5 As the newest member of equine herpesviruses, GHV-1 was renamed EHV-9. 5
We previously conducted experimental infections of EHV-9 by the nasal route in rodents and domestic animals to determine the susceptibility and original host. EHV-9 showed strong infectivity in a wide variety of animals, including gazelles, but not in horses. EHV-9 induced fulminant encephalitis in goats 9 and pigs, 7 as in gazelles. 11 On the other hand, horses inoculated with EHV-9 exhibited only moderate encephalitis, which consisted of moderate perivascular cuffing and gliosis; intranuclear inclusion body formation was not observed. 8 Although the natural host of EHV-9 is still uncertain, a member of the Equidae has been suspected as a reservoir. Because the gazelles that died of the disease shared the same field with zebras, the latter were considered to be the most likely reservoir of EHV-9. In carnivorous mammals, including dogs, there has been no information on susceptibility to EHV-9. In this study, we tried to clarify the infectivity of EHV-9 in dogs, a common human companion animal.
Materials and Methods
Six male Beagle dogs, aged 6 months, were obtained from a breeder. Dogs were vaccinated against canine distemper, canine parvovirus, and canine infectious hepatitis. They were handled in accordance with “The Guidelines for Animal Experiment in the Faculty of Agriculture, Gifu University,” and the Japanese Law “The Care and Welfare for Animals.” The experimental protocol was approved by the animal experiment committee in Gifu University. The body weights of the dogs ranged from 9 to 10 kg at the start of the experiment. They were provided with commercial dog chow and tap water twice a day. After a 1-week quarantine, the dogs without observable abnormalities were used for this study. Each dog (Nos. 1–5) was inoculated intranasally with 10 ml of virus solution containing 107 plaque-forming units, using a nebulizer as described previously. 5 The route and dose of virus were chosen according to those used to infect horses 8 and goats. 9 Dog No. 6 was treated with MEM and served as control. The inoculated dogs were kept in isolated rooms. Dogs with severe clinical signs were euthanatized. EHV-9 P19 stock was prepared by propagation in fetal horse kidney cells of virus from the original stock of the fifth passage in Madin-Darby bovine kidney (MDBK) cells. Titration of virus was conducted by plaque formation using MDBK cells and methylcellulose medium for overlay.
The dogs were checked for clinical signs at least three times daily, in the morning, at noon, and in the afternoon. Rectal temperatures were recorded once a day throughout the study. Nasal swabs and peripheral blood samples were taken daily until the completion of the study. White blood cells were counted using an automatic cell counter (Celltack for animal blood, Nippon Koden Kogyo Inc., Tokyo, Japan), and differentials were performed on Giemsa-stained blood smears. Complete necropsies were done immediately after death or euthanasia. The brain, trigeminal ganglia, palatine tonsils, spinal cord, heart, lungs, trachea, nasal mucosa, liver, spleen, lymph nodes, kidneys, urinary bladder, ovaries, uterus, adrenals, thyroid, salivary glands, stomach, small intestine, large intestine, pancreas, skeletal muscle, and skin were collected and fixed in 10% buffered formalin. After fixation, tissue was embedded in paraffin in the usual manner, sectioned (5 μm) and stained with hematoxylin and eosin (HE). The brains were cut coronally into 5-mm-thick slices, and sections were prepared from various levels, including cerebral cortex, hippocampus, caudate nucleus, globus pallidus, thalamus, pons, medulla oblongata, and spinal cord. Brain sections were stained with EHV-9 rabbit antiserum by the avidin–biotin–complex (ABC) immunoperoxidase (IP) method, as described previously, 11 using the ABC kit (Vector Laboratories, Burlingame, CA). The EHV-9 antiserum, made in our laboratory, was used as the primary antibody at dilutions of 1 in 600 and 1 in 1000. Sections were counterstained with hematoxylin. Tissue sections from an EHV-9–infected goat and sera from a nonimmunized rabbit and goat were used as controls.
Virus was isolated from brain (dog Nos. 1 and 3), lungs (dog Nos. 1 and 3), and peripheral mononuclear cells (dog Nos. 1 and 2). Tissues were homogenized and resuspended in MEM. Heparinized peripheral blood was centrifuged at 5000 rpm for 5 minutes to obtain buffy coat samples. Erythrocytes were lysed by treating with distilled water for 1 minute. Leukocytes from the buffy coat and nasal swabs were suspended in MEM, and suspensions were filtered through a membrane with a pore size of 0.45 μm. Isolation was done by inoculation of an MDBK cell monolayer. Inoculated cells were incubated at 37 C for 2–4 days and observed for cytopathic effect. The recovered virus was identified by DNA fingerprinting. 5 The neutralization test was done using the 50% plaque reduction method.
Results
Dog No. 2 was euthanatized on the fourth day to examine initial stages of the disease; it had no apparent signs except for a fever. Dog Nos. 1, 3, 4, and 5 had a slight to moderate decrease in body weight, severe anorexia, and pyrexia of 40 C; they showed neurological signs 4 days after inoculation. The neurological signs consisted of progressive gait and postural abnormalities, hyperreactivity to stimuli, aggressiveness, salivation, tremor, convulsion, and terminal depression and coma. The four dogs with clinical signs were found dead or were euthanatized. In addition, dog No. 5 had a persistently erect penis and visual impairment on the sixth day, and dog No. 3 showed dyspnea on the fourth day and soft feces with bloody mucus on the fifth day.
Blood analysis of dog Nos. 1 and 5 showed a marked increase in the white blood cell count and a slight decrease in lymphocytes on the fourth and fifth days, respectively. A summary of results is shown in Table 1.
Summary of experimental infection of EHV-9 in dogs.∗
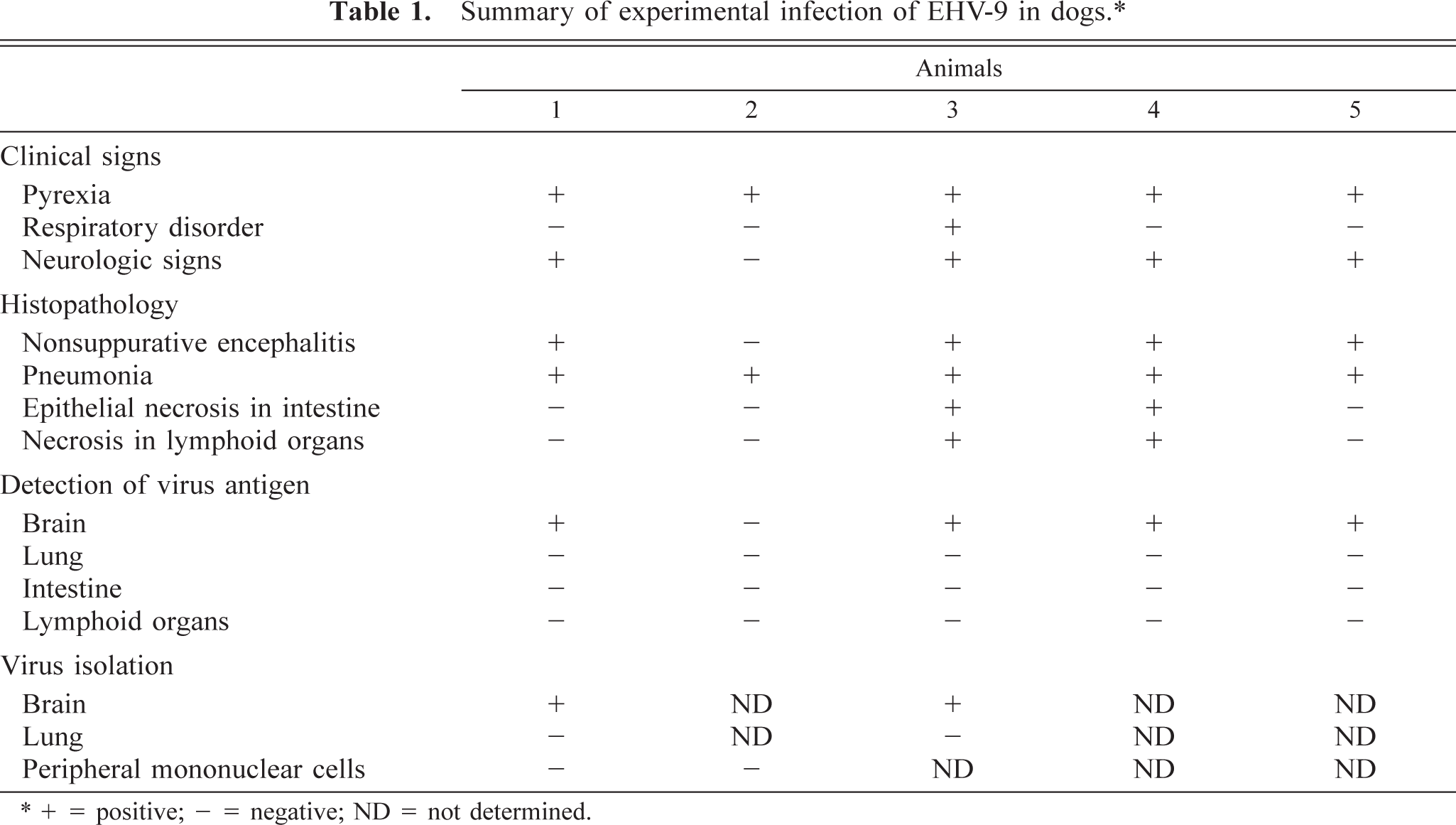
∗+ = positive; − = negative; ND = not determined.
Virus was isolated from the olfactory bulb in dog Nos. 1 and 3. The virus was identified as EHV-9 by DNA fingerprinting. The neutralization test also proved the isolated virus to be EHV-9. No virus was isolated from nasal discharge, white blood cells, or other organs, including lungs.
Grossly, foci of consolidation with congestion were scattered throughout the lungs of all the inoculated dogs. Dog Nos. 1, 3, and 4 had a slight to moderate lymphadenopathy with petechiae on cut surfaces in various lymph nodes. No abnormalities were observed in other organs.
Histopathologically, severe nonsuppurative encephalitis was observed in the central nervous system (CNS) in four of the five dogs (Figs. 1–4). The CNS lesion was characterized by prominent diffuse neuronal degeneration and loss, with frequent intranuclear inclusion bodies. There were varying degrees of diffuse glial reaction, perivascular cuffing, hyperemia, and multifocal perivascular hemorrhage. Affected neurons were characterized by pyknosis with shrinkage, diffuse to central chromatolysis, and karyorrhexis. Degenerating neuronal cells often had Cowdry type A intranuclear inclusion bodies compatible with those in other herpesviral infections. Inflammatory changes were observed mainly in the olfactory bulb, the frontal, occipital, temporal, and parietal lobes, the basal ganglia, and the hippocampus (Table 2). No abnormalities were observed in the cerebellum, spinal cord, or trigeminal nerve. Lesions were usually limited to the gray matter, and white matter was relatively unaffected. Among the affected cerebral areas, the rhinencephalon and the olfactory bulb had the most severe lesions. In the olfactory bulb, all layers showed marked, diffuse neuronal loss with numerous intranuclear inclusion bodies, in addition to an intense inflammatory reaction. No CNS lesions were detected in dog No. 2. EHV-9 immunohistochemistry showed reactivity in the cytoplasm and intranuclear inclusions in degenerating neurons and occasionally in cells that appeared normal in the brains. Positive cells were seen in and around glial nodules in the olfactory bulb and the cerebrum, mainly in the frontal and temporal lobes. Neurons containing EHV-9 antigen were frequently found in the olfactory bulbs. No reactivity was detected in the cerebellum, spinal cord, spleen, lymph nodes, thymus, tonsil, or intestine.
Distribution and severity of histopathologic lesions in the central nervous systems of affected dogs.

∗− = negative; + = slight; ++ = moderate; +++ = severe.
†− = absent; + = present.
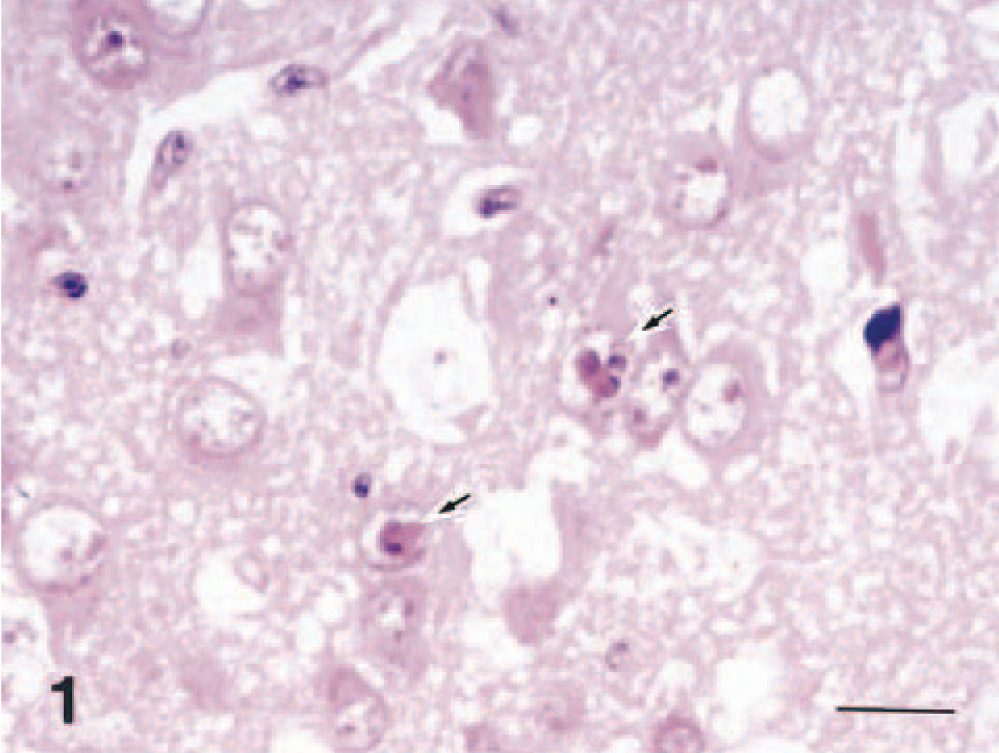
Brain; dog No. 1. Degenerating neuronal cells in the olfactory bulb. Frequent intranuclear inclusion bodies are seen (arrows). HE. Bar = 12 μm.
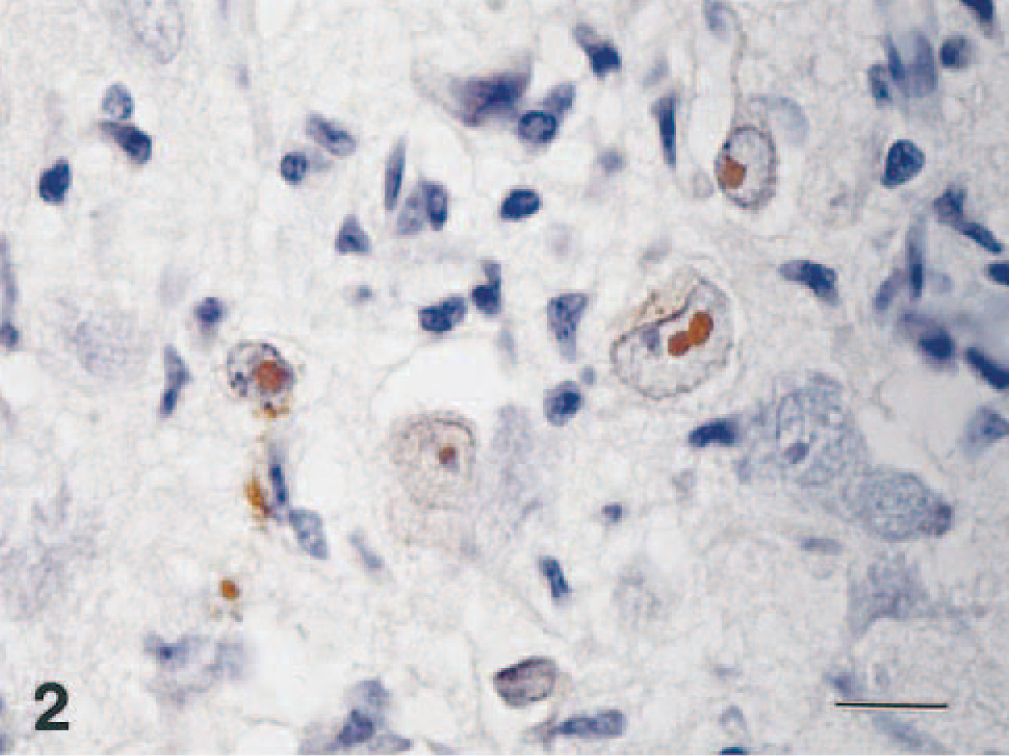
Brain; dog No. 6. A positive reaction for EHV-9 is seen in neuronal cells in the temporal lobe. ABC method. Bar = 12 μm.
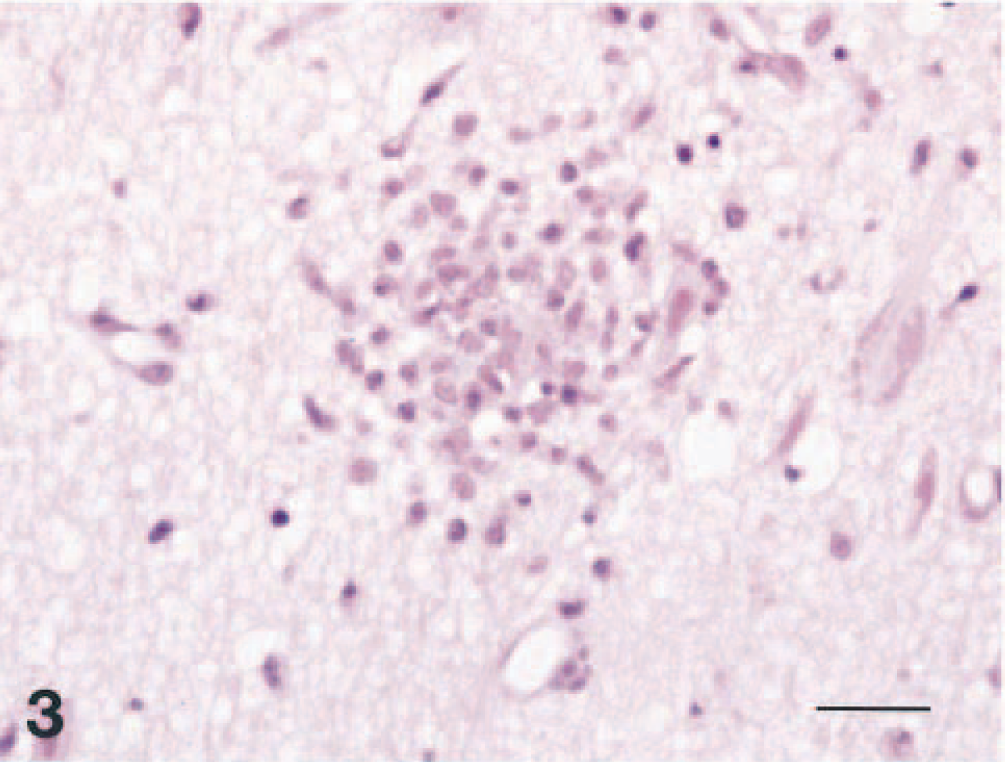
Brain; dog No. 1. Focal proliferation of glial cells is seen in the temporal lobe. HE. Bar = 24 μm.
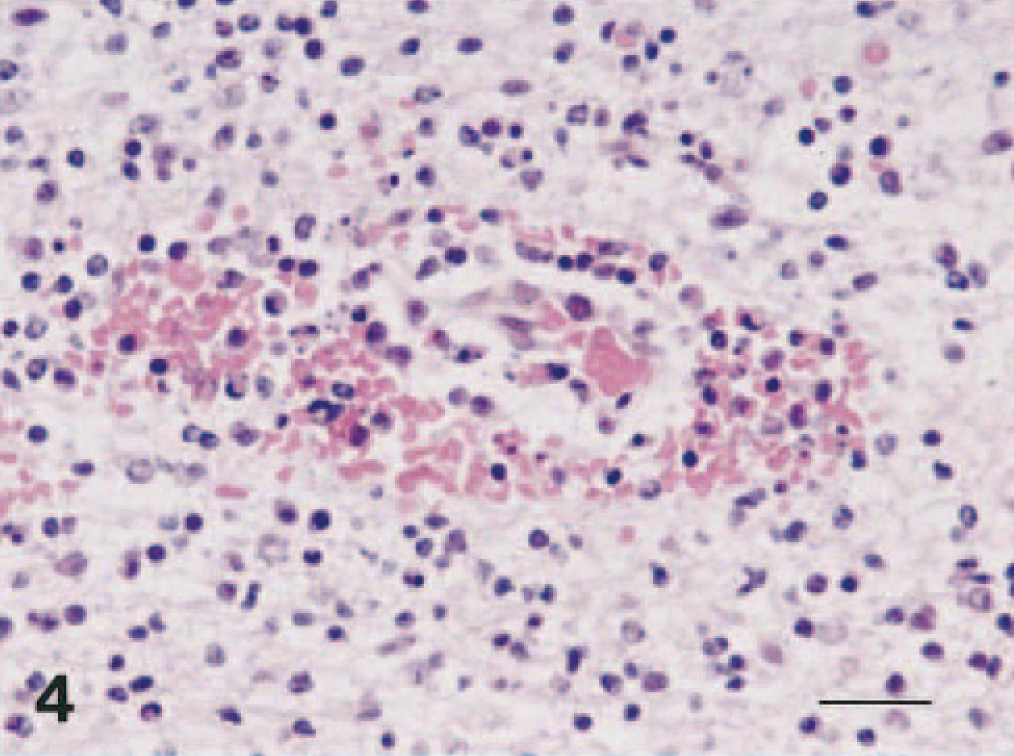
Brain; dog No. 4. Multifocal hemorrhages around small vessel in the olfactory bulb. HE. Bar = 24 μm.
Dog Nos. 2, 3, 4, and 5 had focal to diffuse interstitial pneumonia characterized by moderately thickened alveolar septa with variable numbers of macrophages and neutrophils (Fig. 5). Dog No. 1 had slight bronchopneumonia. No lesions were observed in the nasal mucosa. In all lymphoid organs, there was follicular necrosis and atrophy (Fig. 6). Dog No. 4 had marked necrosis and desquamation of epithelial cells in the duodenal mucosa and necrosis of lymphocytes in the lamina propria. No abnormalities were found in other organs examined.
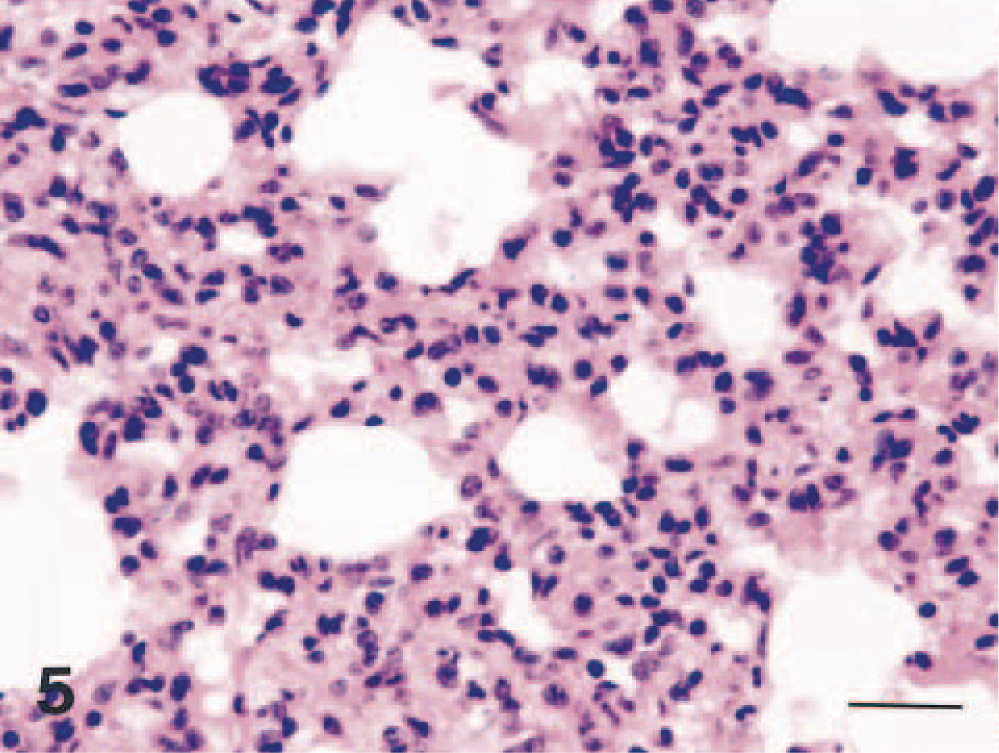
Lung; dog No. 4. Interstitial pneumonia in the lung. Note thickening of alveolar septa. HE. Bar = 24 μm.
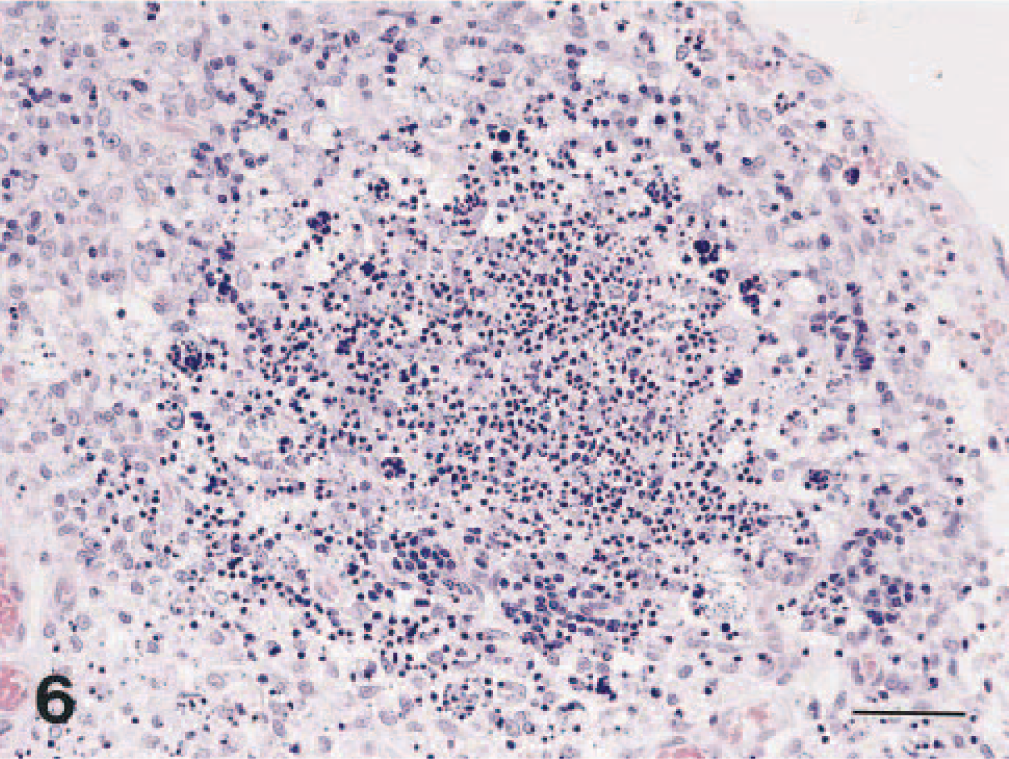
Mesenteric lymph node; dog No. 4. Marked lymphoid follicular necrosis. HE. Bar = 48 μm.
Discussion
EHV-9 causes nonsuppurative encephalitis in a wide variety of animals, including rodents and domestic ruminants. A fulminant encephalitis with marked neuronal signs was found in mice, 5 hamsters, 4 and goats 9 after nasal infections of EHV-9, as in the Thomson's gazelles. 11 However, racing horses inoculated with EHV-9 intranasally had only moderate encephalitis and survived, 8 which suggests that horses might be a natural host for EHV-9. Similar to pseudorabies virus infection in dogs 6 or B virus infection in humans, 1 EHV-9 might be more virulent in unusual hosts. In the present study, dogs infected intranasally with EHV-9 showed rapid progression of disease with a high mortality rate and prominent neurological signs. Clinically, infected dogs had a 4-day latent period before the onset of clinical signs. Signs observed were similar to those observed in hamsters 4 and goats, 9 which demonstrated a high rate of mortality and rapid disease progression. Dogs infected with EHV-9 had prominent neurological signs, including gait abnormalities and aggressiveness, tremor, convulsion, and depression.
Histologically, dogs had severe meningoencephalitis. CNS lesions induced by EHV-9 were similar to those induced in hamsters 4 and goats, 9 although multifocal hemorrhage predominated in dogs. These perivascular hemorrhages were found around small vessels and capillaries and were accompanied by no apparent destruction of the blood vessel walls. EHV-9 is closely related genetically to EHV-1, which has strong vascular tropism instead of neural tropism. 2 , 3 , 10 Although EHV-9 has strong neural tropism in rodents, 4 , 5 goats, 9 pigs, 7 and gazelles, 11 there is apparently some vascular tropism in dogs.
In dogs infected by the nasal route, the CNS lesions occurred mainly in the olfactory bulb and forebrain, expanding to the basal ganglia, thalamus, and hippocampus, as in gazelles, 11 goats, 9 and rodents. 4 , 5 Virus was detected in the olfactory bulb and cerebrum in dogs and hamsters by immunohistochemistry. The virus may travel along the olfactory pathway and spread regionally.
Interstitial or bronchopneumonia was observed in all dogs, as in gazelles. In addition, two of five dogs had follicular necrosis in lymphoid organs, but EHV-9 was not recovered from the lung and lymph nodes.
There are two possible routes by which EHV-9 virus may reach the CNS in dogs infected intranasally. First, extension from the nasal mucosa to the olfactory bulb must be considered as a possible route because the olfactory bulb had the most prominent inflammation. The respiratory route is another possibility, given that EHV-9 was isolated from lungs with pneumonia in two cases.
The present study showed that EHV-9 presents a considerable hazard to carnivorous animals as well as to wild and domestic ruminants and rodents. Because EHV-9 induced fatal fulminant encephalitis in dogs, a popular companion animal, the question of infectivity to humans is a significant concern. Further study is needed to clarify the pathogenesis of infection and the infectivity to other animals and humans.
Footnotes
Acknowledgements
We are particularly grateful to Ms. Cate Swift for conscientious proofreading of this manuscript. This study was supported in part by Grants-in-Aid for Basic Scientific Research from the Ministry of Science, Sports, and Culture of Japan (COE).
