Abstract
By using a new member of the neurotropic equine herpesviruses, EHV-9, which induced encephalitis in various species via various routes, an ocular infection model was developed in suckling hamsters. The suckling hamsters were inoculated with EHV-9 via the conjunctival route and were sacrificed after 6, 12, 24, 36, 48, 72, 96, 120, and 144 hours (h) post inoculation (PI). Three horizontal sections of the brains, including the eyes and cranial cavity, were examined histologically to assess the viral kinetics and time-course neuropathological alterations using a panoramic view. At 6 to 24 h PI, there were various degrees of necrosis in the conjunctival epithelial cells, as well as frequent mononuclear cell infiltrations in the lamina propria and the tarsus of the eyelid, and frequent myositis of the eyelid muscles. At 96 h PI, encephalitis was observed in the brainstem at the level of the pons and cerebellum. EHV-9 antigen immunoreactivity was detected in the macrophages circulating in the eyelid and around the fine nerve endings supplying the eyelid, the nerves of the extraocular muscles, and the lacrimal glands from 6 h to 144 h PI. At 96 h PI, the viral antigen immunoreactivity was detected in the brainstem at the level of the pons and cerebellum. These results suggest that EHV-9 invaded the brain via the trigeminal nerve in addition to the abducent, oculomotor, and facial nerves. This conjunctival EHV-9 suckling hamster model may be useful in assessing the neuronal spread of neuropathogenic viruses via the eyes to the brain.
EHV-9, the newest member of the equine herpesvirus family, is a highly neurotropic herpesvirus first isolated in an outbreak of encephalitis in a herd of Thomson’s gazelles (Gazella thomsoni) kept at a zoological garden.13,36 Although the natural host of EHV-9 and the complete host range are still unknown, a member of the equidae is suspected to be a natural host of EHV-9, as domestic horses (Equus caballus) inoculated intranasally with EHV-9 exhibited only mild encephalitis and there were no resulting deaths. 34 In addition, a high sero-prevalence for EHV-9 without any signs was detected among Burchell’s zebras (Equus burchelli) in Serengeti National Park in Tanzania. 2
Recently, it has been reported that the host range of EHV-9 has expanded to include Grevy’s zebras (Equus grevysi) and polar bears (Ursus maritimus),7,31 as well as a giraffe (Giraffa camelopardalis reticulata). 18 EHV-9 is most closely related to the recently re-emergent neurotropic pathogen, EHV-1; however, cleavage by restriction enzymes showed that the DNA fingerprint of EHV-9 is different from that of EHV-1 and other equine herpesviruses. 13
Emerging EHV-9 infections are of special interest because there is a wide range of susceptible hosts, which includes mice and rats, 13 hamsters, 12 goats, 35 pigs, 26 dogs and cats,37,38 and common marmosets. 21 Recently, EHV-9 was used to induce encephalitis in hamsters by different routes of inoculation, including the oral (25%), peritoneal (25%), and ocular (75%) routes; however, the intravenous route failed to induce encephalitis and there were discrepancies in the distribution of EHV-9 antigen immunoreactive cells in the location and severity of the cerebral lesions. This suggests the possibility of transmission of the virus through regional nerves (the non-olfactory route), after initial propagation at the site of viral entry. 9 The exact pathway through which the virus travels from the eye, which proved to have a high incidence of infection (75% of the animals became infected) in comparison to the other routes, to the brain is still unclear.
Thus, the aim of the present study is to elucidate the pathogenesis of EHV-9 ocular-induced encephalitis. Ocular transmission of viruses to the central nervous system (CNS) has been suspected in many viral diseases such as Cercopithecine herpes virus 1 (B-virus) in humans 5 and herpes simplex virus in children with labial vesicles after rubbing of the eye. 14 Fatal infection by B-virus in humans via ocular exposure from biological fluid from macaque monkeys has been reported. 5 A number of viruses have been studied following intraocular inoculation. In general, intraocular inoculation has been found to be effective in inducing CNS infection, with or without inducing specific ocular pathological lesions. Many viruses, including poliomyelitis virus,10,22 rabies virus, 29 vesicular stomatitis virus, 30 fox encephalitis virus, 15 and equine encephalomyelitis, 20 successfully induced brain infection following intraocular inoculation. The ocular route may be important in the transmission of many viruses in the laboratory as well as in the field.
In the present study, we describe the use of a suckling hamster model to assess primary infection in the eye, followed by the spread of the virus through neuronal routes to the brain. The suckling hamsters were useful in assessing viral kinetics and pathogenesis because panoramic horizontal views of the head, including the eye, orbital cavity, nasal cavity, cranial nerves, and brain, are very similar to MRI (magnetic resonance imaging) in human radiography. In addition, the eyes were still closed at this age in the suckling hamsters, which enabled the precise delivery of the exact inocula to an enclosed site in the conjunctival sac between the eye and eyelid, which is not available in adult animals with open eyes. Suckling hamsters have been used in the past to elucidate the pathogenesis of various viruses, including the mumps virus, via intracerebral inoculation,23,33 and recently, the suckling hamster model was used to study the spread of EHV-9 from the nasal cavity to the brain by nasal inoculation. 8
Materials and Methods
Animals
Nine pregnant Syrian hamsters (Mesocricetus auratus) dams at gestational day 10 were purchased from a commercial breeder (SLC Inc., Hamamatsu, Japan). The animals were housed in an isolated biohazard cabinet and were fed a basal pellet diet (Oriental MF, Oriental Yeast Co., Tokyo, Japan) and bottled water ad libitum until they gave birth, after which they were left to rear the pups until the end of the experiment. This experiment was conducted in accordance with the pertinent laws and regulations on the treatment and use of laboratory animals. The experiment protocol was approved by the Animal Experiment Committee for Animal Welfare in the Faculty of Applied Biological Science at Gifu University.
Inoculation of virus
Twenty-nine 6-day-old suckling hamsters were bilaterally inoculated via the conjunctival route with 10 μL (106 PFU) of EHV-9 virus solution. The eyelid was grasped, and virus solution was injected into the conjunctival sac between the eyelid and eye (the eyes are still closed in animals at this age). Three each of the inoculated animals were sacrificed at 6, 12, 24, 36, 48, 72, 96, 120, and 144 h postinoculation (PI), respectively, or when they were moribund (one animal after 96 hr and another after 120 hr PI). Five uninfected animals were kept as a control group and were inoculated in the conjunctival sac with a sham inoculum that consisted of sterile cell culture medium (MEM). The animals were checked for clinical signs at least twice daily, in the morning and afternoon. Two inoculated animals died after 6 h PI and their data were excluded from the experiment as we believed that the animals died as a result of the inoculation procedure.
Necropsy, histopathology, and immunohistochemistry
After necropsy, the heads were separated from the bodies and were fixed in 10% neutralized buffered formalin. The fixed heads were sectioned on the horizontal plane at two levels (the first section extending cranially from the supraorbital area passing through the cerebral cortex, the second section extending cranially through the eye and brain; from the second section, ventral [Fig. 1a] and dorsal [Fig. 1b] sections were obtained) to obtain the connection between the eye and brain at the same section and were then dehydrated and embedded in paraffin wax by routine methods. They were then sectioned at 3 μm, stained with hematoxylin and eosin (HE), and examined by light microscopy. The bones, including the skulls, of the suckling hamsters were soft enough to prepare histological slides with good quality without decalcification.
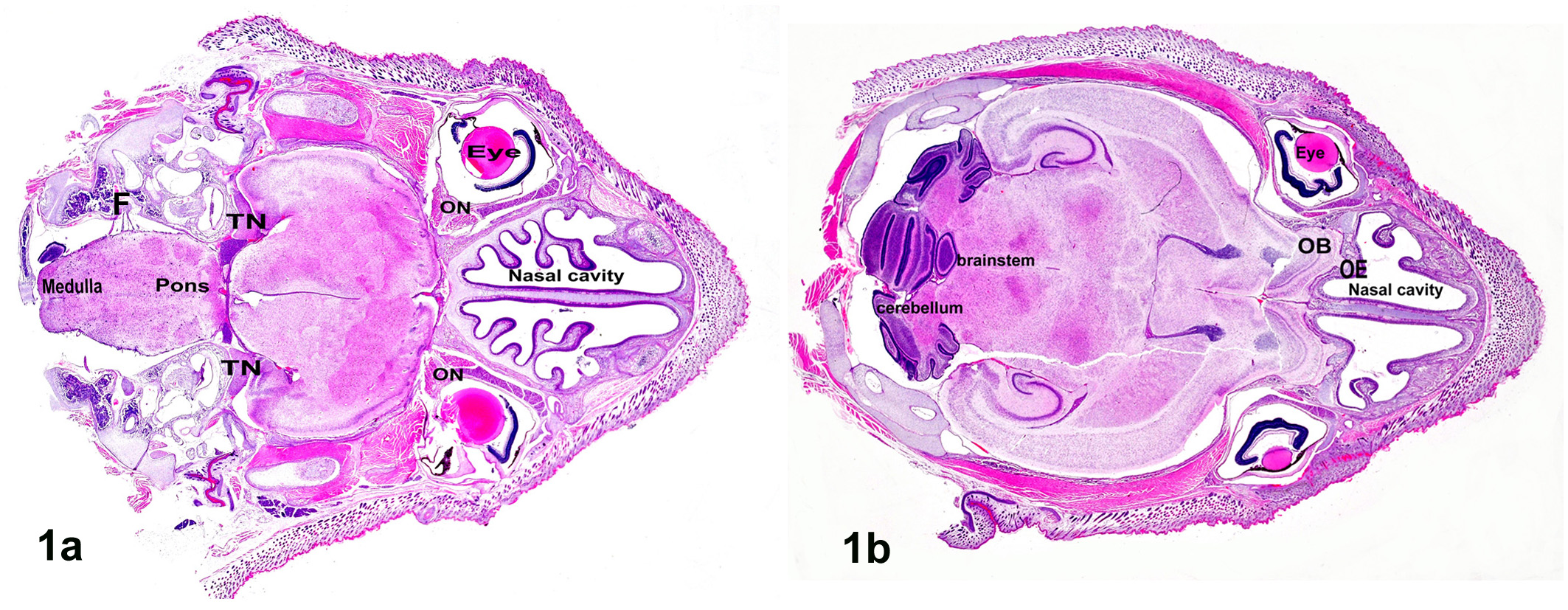
a. Hamster, brain, horizontal section showing the head from the eye to the whole brain from the ventral view; TN, trigeminal nerve; ON, optic nerve; F, facial nerve. Hematoxylin and eosin (HE). b. Hamster, brain, horizontal section showing the head from the eye to the whole brain from the dorsal view at the level of the forebrain, brainstem, and nasal cavity; OE, olfactory epithelium; OB, olfactory bulb. HE.
Paraffin-embedded sections of the head, including the brain and eyes, were immunolabeled with EHV-9 rabbit antiserum by the avidin-biotin-complex (ABC) immunoperoxidase method, as described previously, 38 with ABC kits (Vector Laboratories, Burlingame, CA, USA). The primary antibody was EHV-9 antiserum (1:800, Veterinary Microbiology Laboratory at Gifu University) followed by application of a secondary antibody (biotinylated anti-rabbit IgG, DAKO Cytomation, USA) with Liquid DAB Substrate Chromogen System (DAKO Cytomation, USA) used as the chromogen and hematoxylin counterstain. Tissue sections from confirmed EHV-9–infected hamsters were used as positive control samples, and sera from a nonimmunized rabbit and goat were used as a negative control treatment on positive control specimens.
Results
Clinical Findings
The inoculated hamsters showed various degrees of clinical signs, including depression and uncoordinated movements, starting at 96 h PI. By 120 h PI, all of the animals exhibited severely uncoordinated movement, as well as paralysis, sometimes unilaterally. The animals were unable to crawl normally, were dysphagic, became depressed, and eventually died from the neurologic disorder.
Gross observation and histopathology
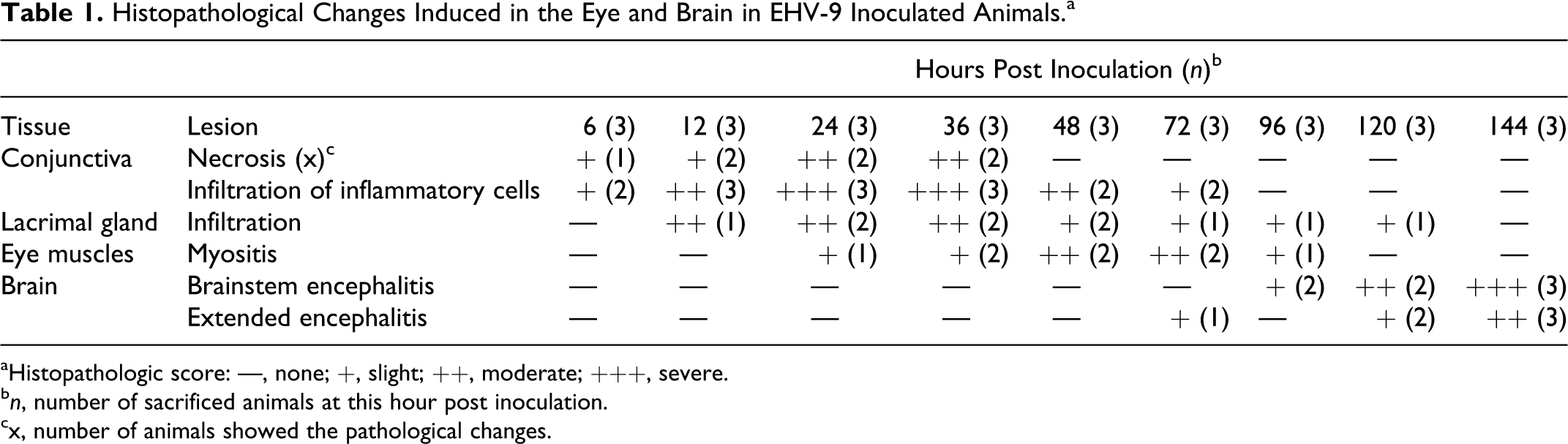
Grossly, there were no apparent abnormalities in the organs or tissues of the inoculated animals. Horizontal sectioning of the head in the suckling hamsters made it easy to view the orbital cavity, including the eye with all of its compartments, and most of the cranial nerves connections to the brain and brain itself were visible in a histological section (Figs. 1a, 1b). No microscopic abnormalities were noted in the five uninoculated control mice at any interval of the experiment. Histopathological alterations in the eye and brain in animals inoculated with EHV-9 via the conjunctival route are summarized in Table 1 and as follows:
Histopathological Changes Induced in the Eye and Brain in EHV-9 Inoculated Animals.a
aHistopathologic score: —, none; +, slight; ++, moderate; +++, severe.
bn, number of sacrificed animals at this hour post inoculation.
cx, number of animals showed the pathological changes.

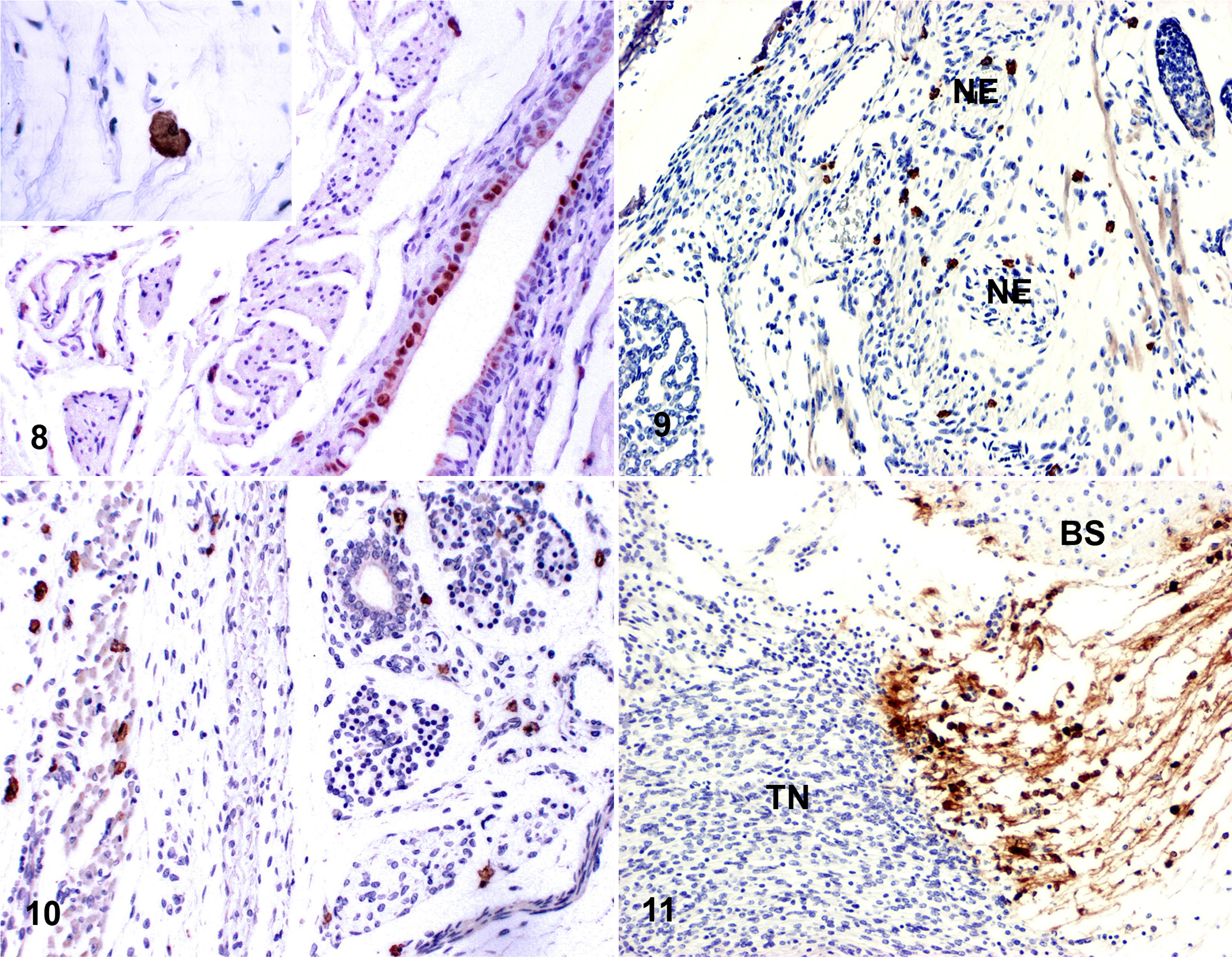
Immunohistochemistry
Immunohistochemical staining with EHV-9 antibody was used to determine the viral kinetics of transmission from the eye to the brain. The immunohistochemical detection of EHV-9 in the eye and the brain is summarized as follows:
The distributions of the viral antigen immunoreactivity in the horizontal sections of the brain after 96 h and 144 h PI are depicted in Figures 12a and 12b, respectively. At 96 h, EHV-9 antigen immunoreactivity was detected only in the brainstem at the level of the pons close to the entrance of the sensory branch of the trigeminal nerve; however, at 144 h PI EHV-9 antigen immunoreactivity was detected in the medulla oblongata, close to the entrance of the abducent nerve, as well as the pons and extended to frontal and temporal lobes of the cerebral cortex.
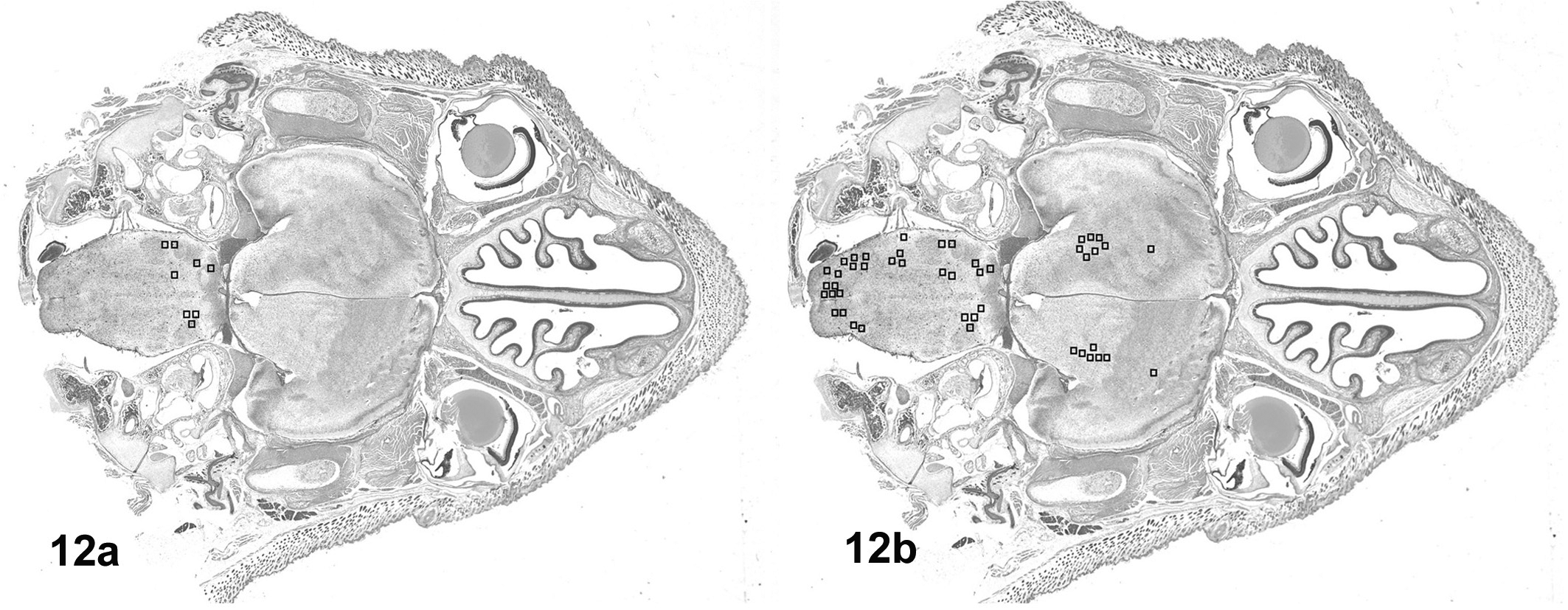
a. Suckling hamster, brain, distribution of EHV-9 antigen immunoractivity. At 96 h post inoculation (PI), viral antigen was detected in the pons. b. Suckling hamster, brain, distribution of EHV-9 antigen immunoractivity. At 144 h PI, viral antigen was found in the pons, cerebellum, medulla oblongata, and the frontal and temporal lobes of the cerebral cortex.
Discussion
EHV-9, the newly emerging neurotropic herpesvirus, has been studied with respect to pathogenesis and route of brain infection including the nasal, ocular, peritoneal, oral, and intravenous routes. The nasal and ocular routes show higher incidences of infection (100% and 75% of animals became infected, respectively) in comparison to the other routes. 9 The ocular route for the transmission of neuropathogenic viruses is gaining in importance in the laboratory as well as in the field. Fatal infections with Cercopithecine herpes virus 1 (B-virus) via ocular splashes with biological fluid from macaque monkeys without percutaneous injury have been reported in humans,4,5 and in this case, conjunctivitis developed 10 days later after exposure, and the victim then died from B-virus–induced encephalomyelitis 6 weeks later. Possible infection in the CNS following ocular inoculation was reported with respect to many viruses, such as EHV-9 in hamsters, 9 poliomyelitis virus,10,22 rabies virus, 29 vesicular stomatitis virus, 30 fox encephalitis virus, 15 and equine encephalomyelitis. 20 However, the detailed kinetics and pathogenesis in terms of access to the CNS are still unclear because of difficulties in tracing the entrance of the cranial nerves to the brain through the cranial bone. The suckling hamster model following conjunctival inoculation with EHV-9 was developed to assess the kinetics and pathogenesis of EHV-9 using a panoramic view that includes the eye, olfactory epithelium, olfactory nerve, and optic nerve as well as other cranial nerves and the brain by using horizontal sections. Horizontal sections at two levels were prepared in the present study to assure easy tracking from the viral inoculation site of the eye to the main affected site of the brain. Based on histology and immunohistochemistry, conjunctival inoculation with EHV-9 first results in conjunctivitis characterized by conjunctival epithelial cell necrosis, mononuclear cell infiltrations in the lamina propria of the eyelid, as well as myositis of the eyelid muscles and infiltration of lacrimal glands, which may be an essential stage for propagation of the virus in the entry site for further travel to the CNS. At the early stage of infection (6 hr PI), the conjunctival epithelial cell as well as macrophages exhibited immunoreactivity to EHV-9 antigen, especially around the fine nerve endings, and this extended later to the eyelid muscles, lacrimal and extraocular muscles, as well as the nerves supplying those muscles. At the initial stage of infection, the macrophages migrating from surrounding tissue may play a very important role in the propagation and transmission of EHV-9 to the nerves of the eyes. Previously, the importance of macrophages in the pathogenesis of virus infections was reviewed by Mires. 24 Blood clearance studies suggested that most viruses are taken up by macrophages in the same way as other colloidal materials.3,24 Since macrophages move freely through tissues, it has also been suggested that they play a primary role in the dissemination of viruses. 11 Therefore, the susceptibility or resistance to viral diseases could depend in large part on the macrophage-virus interaction. In an in vitro study on macrophages in suckling mice, herpesvirus was shown to have spread to adjacent cells of various types by macrophages. 17 The migrating macrophages may pick up the EHV-9 and transport it to the nerve endings supplying the eyelid and eye, including the ophthalmic branch of the trigeminal nerve and the abducent, oculomotor, and facial nerves. EHV-9 may then travel by way of these nerves to the brainstem causing encephalitis at the level of the pons and cerebellum. CNS lesions in these animals were restricted to the pons, medulla oblongata, and cerebellum, suggesting viral transport through the trigeminal, abducent, and facial nerves. A similar transneural passage has been described in porcine Pseudorabies virus (PRV) infection where virus gained access to the brain via the trigeminal and oculomotor nerves. 32 In cases of herpes simplex virus infection in rabbits, the inoculated virus was passed serially onto the scarified cornea, producing fatal encephalitis, where the virus did not enter the eye after inoculation onto the cornea, but passed to the conjunctiva and thereafter to the brain via the sensory branches of the trigeminal nerve. 14 Keeble et al. described lesions of natural B-virus infection in monkeys where CNS lesions were minimal and consisted of localized involvement of the pons and medulla oblongata. 19 In addition there were glial and lymphocytic infiltrations around the nerve roots of the trigeminal and facial nerves, in the nucleus and tract of the descending branch of the trigeminal nerve and in the solitary tract. 19 Encephalitis restricted to the brainstem was reported previously in cases of human herpes simplex virus 1 infection16,25 and herpes simplex virus 2 infection.6,28
At the final stage EHV-9 virus expanded rostrally to the cerebrum as well as caudally to the spinal cord. There were some discrepancies in severity and distribution of the lesions. In some cases, the encephalitis was restricted to one-half of the brain, or there was bilateral involvement. The ocular EHV-9 suckling hamster model may be useful in pathogenesis studies of neurotropic virus such as B-Virus, especially those concerned with how virus gains access to the brain.
The optic nerve is considered to be part of the central nervous system, which is completely encased by three layers of meninges and is protected from contact with macrophages carrying the virus. With some viruses, such as the lymphocytic choriomeningitis virus (LCM), multiplication of the virus in the eyes does not induce ocular pathological changes following intraocular inoculation except mild transitory hyperemia; 27 however, in the present study marked conjunctivitis was noted. In our previous work with EHV-9 after nasal inoculation, we found that the virus traveled through the olfactory nerve after initial propagation in the olfactory receptor neurons, and that area of the brain that was affected earliest and to the greatest degree was the olfactory bulb; 8 however, in the present study, the pons and cerebellum were affected earliest.
There is still a possibility of other routes of transmission of EHV-9 after conjunctival inoculation, via the oral or nasal routes, because the eyes and the oral and nasal cavities are connected by means of the lacrimal (tear) duct. After conjunctival inoculation, (1) that virus may secondarily infect the olfactory nerve via the infection of the lacrimal gland, and (2) virus may also spread to the nasopharynx with the tear film via the nasolacrimal duct, providing an alternate method of infecting the olfactory nerve and accounting for the rhinitis noted in one animal with possibly dominant and earliest lesions in the olfactory bulb.
The suckling hamster model, using horizontal sections of the head following conjunctival inoculation, is very useful in elucidating the mechanism of EHV-9 transmission from the eye to the brain. Following conjunctival inoculation, EHV-9 appears to result in local conjunctivitis and infection of local macrophages, which transport the virus to local fine nerve endings; because of this, EHV-9 is able to invade the brain via the trigeminal, abducent, oculomotor, and facial nerves, inducing meningitis, encephalitis, and myelitis.
Footnotes
Acknowledgements
We thank Miss Cate Swift for her conscientious proofreading of grammatical aspects.
Declaration of Conflicting Interests
None of the authors of this paper has a financial or personal relationship with other people or organizations that could inappropriately influence or bias the contents of the paper.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported in part by Grant-in-Aid for Scientific Research from the Ministry of Health, Labor and Welfare of Japan, Hokkaido University, Research Center for zoonosis control, and a grant from Ono Pharmaceutical Co, Ltd.
