Abstract
Feline immunodeficiency virus (FIV) infection typically has a prolonged and variable disease course in cats, which can limit its usefulness as a model for human immunodeficiency virus infection. A clade C FIV isolate (FIV-C) has been associated with high viral burdens and rapidly progressive disease in cats. FIVC was transmissible via oral-nasal, vaginal, or rectal mucosal exposure, and infection resulted in one of three disease courses: rapid, conventional/slow, or regressive. The severity of the pathologic changes paralleled the disease course. Thymic depletion was an early lesion and was correlated with detection of FIV RNA in thymocytes by in situ hybridization. The major changes in thymic cell populations were depletion of p55+/S100+ dendritic cells, CD3- cells, CD4+/CD8- cells, and CD4+/CD8+ cells and increases in apoptosis, CD45R+ B cells, and lymphoid follicles. In contrast to thymic depletion, peripheral lymphoid tissues often were hyper-plastic. Mucosally transmitted FIV-C is thymotropic and induces a spectrum of lymphoid lesions and disease mirroring that seen with the human and simian immunodeficiency virus infections.
Keywords
Feline immunodeficiency virus (FIV), a lentivirus identified in 1986, 54 causes a disease syndrome similar to that induced by HIV. The clinical phases of FIV infection parallel those of HIV: 1) an acute phase characterized by a burst of viral replication, flulike illness, lymphadenopathy, and progressive decline in CD4+ T lymphocytes, 1 2 69 2) a prolonged asymptomatic phase featuring viral downregulation and a continued gradual decline in CD4+ cells, and 3) a terminal phase characterized by immunologic decompensation, wasting, hematologic suppression, recrudescence of plasma viremia, and clinical immunodeficiency with opportunistic infection. 55 65 74 75
Development of the FIV model has been limited by its prolonged and variable disease course in infected cats. We identified a virulent clade C FIV isolate, FIV-C-PGammer (FIV-C), which induced high viral RNA titers in plasma and rapid progression to fatal disease. 12–14 Subsequent studies with FIV-C demonstrated that in contrast to intravenous infection studies, mucosal exposure produced a broader range of host–virus relationships. 47 Three provisional categories of FIV-C infection were identified in mucosally infected animals: 1) rapidly progressive/accelerated infection (<5 months) marked by high virus burdens and rapid CD4+ cell depletion, 2) conventional/slow infection (>5 months) featuring slowly progressive CD4+ cell decline, and 3) regressive/transient infection marked by very low virus burdens with no CD4+ cell alterations. Similar disease courses have been reported in HIV-infected humans. 39
The purpose of the present study was to characterize the clinical and pathologic changes associated with the three disease courses induced by mucosal FIV-C infection and to describe the changes in the thymus, the major early target tissue.
Materials and Methods
Animals and sample collection
Thirty-two specific-pathogen-free (SPF) cats from a breeding colony maintained at Colorado State University were inoculated by atraumatic exposure of either oral–nasal, vaginal, or rectal mucosae to FIV-C-PGammer (FIV-C). One-day-old kittens (n = 14; cat Nos. 10–19, 37–40) were inoculated via the oral and nasal mucosae with 200 μl of 200 TCID50/ml (50% tissue culture infective dose; n = 6) cell-free infectious plasma or 300 TCID50/ml (n = 8) cell-free infectious culture. Similarly, 8-week-old cats were inoculated via the oral and nasal mucosae (n = 6; cat Nos. 4–9) or rectum (n = 9; cat Nos. 20–23, 41–45) with 1 ml of 300 TCID50/ml cell-free infectious culture. In addition, 8-week-old female cats (n = 3; cat Nos. 1–3) were inoculated by instillation of 500 μl of 200 TCID50/ml cell-free infectious plasma (n = 1) or 300 TCID50/ml (n = 2) cell-free infectious culture onto the vaginal mucosa. Following administration of the inoculum, each animal was held in a specific position to facilitate exposure of the respective mucosal membrane for a minimum of 20 minutes; cat inoculated oral-nasally were positioned upright and those inoculated rectally/vaginally were positioned with the posterior end elevated. Age- and litter-matched control animals receiving parallel noninfectious inocula consisted of six SPF newborn kittens (cat Nos. 24–29) and seven SPF weanling age cats (Nos. 30–36). The FIV infection status and disease course of these animals has been previously described and is briefly summarized in Table 1. 47
Pathologic changes detected in cats infected mucosally with FIV-C.
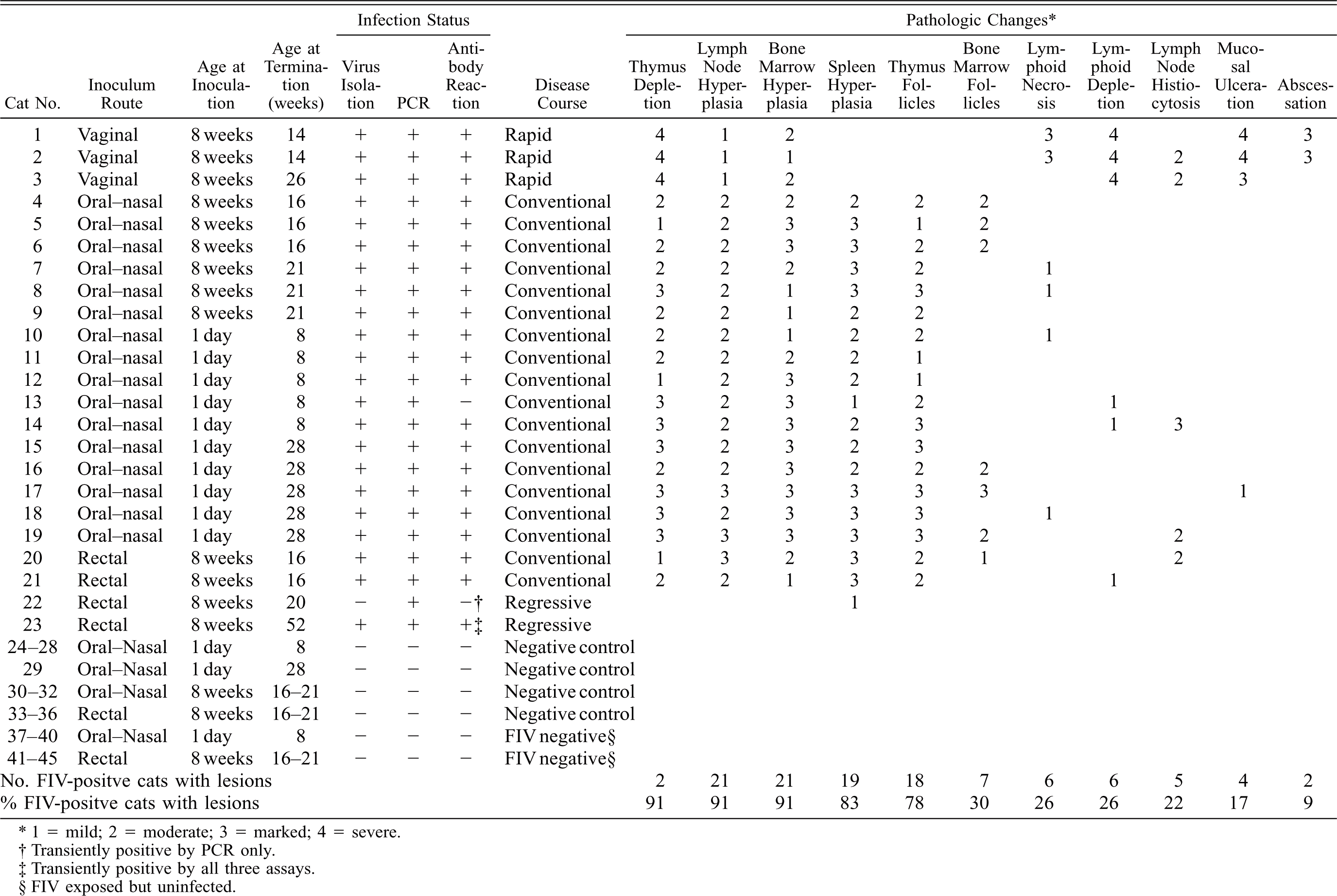
1 = mild; 2 = moderate; 3 = marked; 4 = severe.
Transiently positive by PCR only.
Transiently positive by all three assays.
FIV exposed but uninfected.
At study termination, the animals were euthanatized for necropsy and sample collection. Mucosal, regional, and systemic lymphoid tissues were fixed in 10% buffered formalin for ≤18 hours before histologic processing. Tissues harvested included spleen, bone marrow, thymus, retropharyngeal lymph nodes, submandibular lymph nodes, mesenteric lymph nodes, colic lymph nodes, iliac lymph nodes, tonsils, gastrointestinal tract, rectum, vagina, oral–nasal mucosa, liver, kidneys, lungs, salivary glands, and brain. In addition, body and thymus weights were recorded, and a portion of the thymus was collected for thymocyte isolation.
Virus inocula
The cell-free plasma used as virus inoculum was derived from a pool of cats acutely infected with FIV-C. 14 The cell-free culture used as virus inoculum was obtained by coculture of peripheral blood mononuclear cells (PBMC) from a cat infected with FIV-C with naive PBMC. The plasma and supernatant were frozen in aliquots and infectivity titrated after thawing by virus isolation coculture.
Thymocyte isolation and preparation
Thymocytes were mechanically dispersed by passage through a wire strainer, resuspended in phosphate-buffered saline (PBS), separated by density gradient centrifugation (Histopaque 1077, Sigma, St. Louis, MO), and quantified by trypan blue exclusion. The thymocytes were incubated at a concentration of 5 × 106 cells/ml with antibody combinations (CD4, CD8, and B cell monoclonal antibodies) for 1 hour at 4 C, washed twice with 2% fetal calf serum in PBS, and resuspended in 2% paraformaldehyde in PBS.
Flow cytometry
Four thymocyte populations, CD4+/CD8+, CD4+/CD8−, CD4−/CD8+, and CD45R+ B cells, from 13 mucosally infected cats with conventional FIV disease courses (cat Nos. 4–9, 15–21), seven control cats (Nos. 29–32, 34–36), and one FIV-exposed but uninfected cat (No. 45) were assessed using an EPICS XL-MCL (Coulter Electronics, Hialeah, FL) flow cytometer. Twenty thousand thymocytes were analyzed for each sample by a two-color system using combinations of feline CD4 (clone 7G4.1) and CD8 (clone 31B.5) monoclonal antibodies 51 and an anti-mouse CD45R, B220 (Ly 5), B-cell monoclonal antibody (Cedarlane Laboratories, Hornby, ON, Canada). Thymocyte bitmap gating was based on forward and side light scatter. For each sample, the analysis gates were set to include <2% of thymocytes labeled with isotypic control monoclonal antibodies conjugated to flourescein isothiocyanate or phycoerythrin (Sigma, Cedarlane Laboratories) in the positive analysis regions. Results were expressed as the percentage of cells located in each quadrant.
Lymphocyte subsets were analyzed as previously described. 10 Ten thousand lymphocytes were analyzed using feline CD4 and CD8 monoclonal antibodies. 51
FIV-C RNA in situ hybridization
Plasmids containing the FIV-C gag or env genes were obtained (Dr. J. Mullins, University of Washington, Seattle, WA). 71 The FIV-C genes were isolated via restriction enzyme digestion, 0.7% agarose gel electrophoresis, and Qiaex II gel purification (Qiagen, Chatsworth, CA). The purified, isolated gene fragments (gag = 1,353 bp; envA = 1,255 bp; envB = 776 bp) were subcloned into the pGEM®-7Zf(+) vector (Promega, Madison, WI) containing T7 and SP6 transcription promoter sequences. Sense and antisense riboprobes were transcribed off their respective promoters incorporating digoxigenin-labeled UTP using either AmpliScribe® T7 or SP6 transcription kits (Epicentre Technologies, Madison, WI).
In situ hybridization (ISH) was performed in a manner similar to that previously described. 26 Formalin-fixed, paraffin-embedded tissues were sectioned (4–5 μm) and placed on positively charged glass slides (Fisher Scientific, Pittsburgh, PA). Tissue sections were deparaffinized and rehydrated sequentially with xylene, xylene-ethanol, 100% ethanol, 95% ethanol, and RNase-free water for 5 min each at room temperature. The slides were then incubated with 5 mM levamisole for 20 minutes, washed with standard saline citrate (SSC) buffer (0.15 M NaCl, 0.015 M sodium citrate), incubated in 0.2 N HCl for 20 minutes, and washed again with SSC buffer. The sections were digested with 25 μg/ml proteinase K in buffer containing 10 mM Tris (pH 7.4) and 2 mM CaCl2 for 10 minutes at 37 C. Digestion was stopped with 0.1 M glycine in PBS. Slides were washed with PBS, incubated in 0.1 M triethanolamine–0.25% acetic anhydride solution for 10 minutes, washed in 2× SSC, incubated in 0.1 M Tris (pH 7.4)–0.1 M glycine solution for 15 minutes, and washed in 2× SSC. Prehybridization was done at 50 C for 10 minutes with hybridization solution containing 50% deionized formamide, 1× SSC, 1× Denhardt's solution, 5 mM NaPO4 (pH 6.8), 0.1% sodium dodecyl sulfate, 250 μg/ml salmon sperm DNA, 5% dextran sulfate, and 250 μg/ml tRNA. Riboprobes were added at 1 ng/μl, and the sections coverslipped, heated to 65 C for 5 minutes, chilled for 10 minutes on ice, and hybridized overnight at 55 C. Following hybridization, the coverslips were removed, and the slides washed with 4× SSC–50% formamide for 1 hour at 50 C and then in 2× SSC for 5 minutes. An RNase mixture (1 unit RNase T1, 20 μg/ml RNase A) was added and incubated for 30 minutes at 37 C to digest excess probe, and the wash steps were repeated. Slides were blocked for 1 hour in a buffer containing 2% horse serum, 0.3% Tween 20, 100 mM Tris (pH 7.4), and 150 mM NaCl. Hybridized probe was detected with a sheep antidigoxigenin alkaline phosphatase–conjugated antibody (Boehringer Mannheim, Indianapolis, IN), diluted 1:500 in blocking buffer, for 1 hour at room temperature. After washing, the sections were incubated with 5-bromo-4-chloro-3-indolyl phosphate (BCIP)–nitroblue tetrazolium (NBT) substrate overnight in the dark. Finally, the sections were washed in 10 mM Tris HCl–10 mM ethylenediaminetetraacetic acid (EDTA) solution (pH 8.1), counterstained with nuclear fast red, dehydrated in graded ethanols, cleared in xylene, and mounted with Clear∗Mount® (American Master “Tech” Reagent Co., Lodi, CA). Negative controls included tissue sections from uninfected age- and litter-matched cats and application of sense riboprobes on tissue sections from FIV-infected and uninfected cats.
Terminal deoxynucleotidyl transferase-mediated dUTP nick end labeling
The terminal deoxynucleotidyl transferase (TdT)-mediated dUTP nick end labeling (TUNEL) method was employed to identify individual thymocytes undergoing apoptotic cell death within thymic tissue sections. A protocol based on a previously described method was utilized. 22 Formalin-fixed, paraffin-embedded tissues were sectioned (4–5 μm) and placed on positively charged glass slides (Fisher Scientific). Tissue sections were deparaffinized in xylene and rehydrated in graded ethanols to distilled H2O. The slides were then incubated with 5 mM levamisole for 20 minutes and washed in PBS. The slides were digested with 25 μg/ml proteinase K in buffer containing 10 mM Tris (pH 7.4) and 2 mM CaCl2 for 10 minutes at 37 C. Digestion was stopped with 0.1 M glycine in PBS, and slides were rinsed with PBS. The TUNEL cocktail (20 mM potassium cacadylate, 25 mM Tris HCl, 250 μg/ml bovine serum albumin [BSA], 1 mM CoCl2, 5 μm digoxigenin-dUTP [Boehringer Mannheim], 25 units terminal transferase) was applied using a coverslip, and the slides were incubated at 37 C for 1 hour. Following incubation, the slides were washed several times with PBS and rinsed in 100 mM Tris (pH 7.4)–150 mM NaCl solution. Slides were blocked for 1 hour in a buffer containing 2% horse serum, 0.3% Tween 20, 100 mM Tris (pH 7.4), and 150 mM NaCl. The TUNEL reaction was detected with a sheep antidigoxigenin alkaline phosphatase–conjugated antibody (Boehringer Mannheim), diluted 1:500 in blocking buffer, for 1 hour at room temperature. After washing, the sections were incubated with BCIP/NBT substrate overnight in the dark. Finally, the sections were washed in 10 mM Tris HCl–10 mM EDTA solution (pH 8.1), counterstained with nuclear fast red, dehydrated in graded ethanols, cleared in xylene, and mounted with Clear∗Mount®. Controls included thymic tissue sections from age- and litter-matched uninfected cats and tissue sections processed as described above but omitting the TdT enzyme.
Image analysis
Tissue sections containing ISH-positive cells or TUNEL-positive cells were visualized using an Olympus VANOX-S microscope with a mercury light source. Video images were captured with a low-light cooled charged-coupled device (CCD) camera (Sony Photocamera DKC-5000), the Adobe Photoshop® software program (Adobe Systems, Mountain View, CA), and a Power Macintosh G3® (Apple Computer, Cupertino, CA). The scanned images were imported into the MetaMorph® digital imaging software program (Universal Imaging, Westchester, PA). The positively stained cells were differentiated from unstained cells with the “threshold” and “measure objects” tools of the MetaMorph software. The pixel sizes of stained and unstained cells were determined by “binorizing” the tresholded cells and using the “measure objects” tool to determine the average number of pixels per cell (object). Both the positive threshold (stained cells) and the negative threshold (unstained cells) values were measured for each image and recorded on a Microsoft Excel® spreadsheet (Microsoft Corp., Redmond, WA). From the average pixel size information determined for each cell population, the numbers of positive and negative cells were calculated and compared as a ratio (1 positive cell to number of negative cells). For each tissue section, five different microscopic fields (200× magnification) were evaluated, and the results were averaged.
Immunohistochemistry for cell phenotype
The immunohistochemistry protocol was based on methods previously described. 4 Formalin-fixed, paraffin-embedded tissues were sectioned (4–5 μm) and placed on positively charged glass slides (Fisher Scientific). Tissue sections were deparaffinized in xylene, rehydrated in graded ethanols to distilled H2O, and rinsed in 0.05 M Tris (pH 7.4). The slides were digested with 250 μg/ml proteinase XXIV (Sigma) in 0.50 mM Tris (pH 7.4) for 15 minutes at room temperature. Slides were washed in TBT buffer (50 mM Tris, 150 mM NaCl, 0.5% BSA, 0.3% Tween 20, pH 7.6). Nonspecific proteins were blocked with 5% goat serum, 5% horse serum, 1% BSA, and 1 mg/ml cat IgG (Sigma) in 50 mM Tris HCl–150 mM NaCl (TBS). In addition, endogenous avidin and biotin binding activities were blocked using a commercial kit (Vector Laboratories, Burlingame, CA). The slides were then incubated for 1 hour at room temperature with one of the following primary antibodies. T cells were identified using a polyclonal rabbit anti-human CD3 antibody (Dako Corp., Carpinteria, CA) at 1:100 dilution in TBS. Macrophages were detected with a monoclonal antibody MAC387 (Dako Corp.) at 1:100 dilution in TBS. Dendritic cells were labeled with a rabbit anti-cow S-100 antibody 31 56 68 (Dako Corp.) at 1:250 dilution in TBS and with a mouse anti-p55 actin bundling protein 42 76 (NIH AIDS Research and Reference Reagent Program, Rockville, MD). The primary antibody was then detected using either the mouse IgG (MAC387 and p55) or rabbit IgG (CD3 and S-100) Vectastain® ABC-AP Kits (Vector Laboratories) according to the manufacturer's instructions. In the case of dual immunohistochemistry, p55 was detected using a goat anti-mouse IgG colloidal gold antibody (Pierce, Rockford, IL) at 1:100 dilution in TBS followed by silver enhancement (Boehringer Mannheim). The resulting complexes of immobilized antibody/streptavidin conjugated to alkaline phosphatase were detected with a chromagen, either Vector® Red (Vector Laboratories) or BCIP/NBT. Tissues were counterstained with Vector® Methyl Green (Vector Laboratories), dehydrated in graded ethanols, cleared in xylene, and mounted with Clear∗Mount®. Negative controls included serial sections of tissue to which either irrelevant mouse IgG1 or rabbit IgG had been applied matching the concentration of the primary antibody and processed in parallel with experimental sections.
Statistical analysis
Data were analyzed using the Mann–Whitney test. 59 Significance was defined at P < 0.05.
Results
Histopathologic findings associated with FIV-C disease courses
Following mucosal exposure to FIV-C by oral–nasal, rectal, or vaginal routes, three different disease courses were recognized: 1) rapidly progressive/accelerated infection (<5 months) marked by high virus burdens and rapid CD4+ cell depletion, 2) conventional/slow infection (>5 months) featuring slowly progressive CD4+ cell decline, and 3) regressive/transient infection marked by very low virus burdens with no CD4+ cell alterations. 47
Despite the spectrum of clinical courses observed following mucosal FIV-C infection, the pathologic changes observed in persistently infected animals were similar, varying chiefly in severity. The most commonly occurring lesions (seen in 91% of the mucosally infected cats) were thymic atrophy, generalized lymphoid hyperplasia, and bone marrow hyperplasia (Table 1). The second most common lesions were splenic lymphoid hyperplasia and thymic follicle formation (78–83% of cats). A third cadre of miscellaneous lymphoid and mucosal lesions was confined to ≤30% of cats.
Thymuses from control animals had normal architecture, with clear distinction between cortical and medullary regions and without follicle formation (Fig. 1a). Thymuses from FIV-infected cats with conventional disease course had variable degrees of cortical and medullary dissolution and lymphoid follicle formation in cortical-medullary junction and subcapsular regions (Fig. 2a). Cats with rapidly progressive infection had complete loss of thymus architecture, with minimal thymic tissue discernible amidst connective and adipose tissue (Fig. 3a). In addition, thymuses from all FIV-infected cats had decreased numbers of Hassall's corpuscles as compared with age-matched controls.
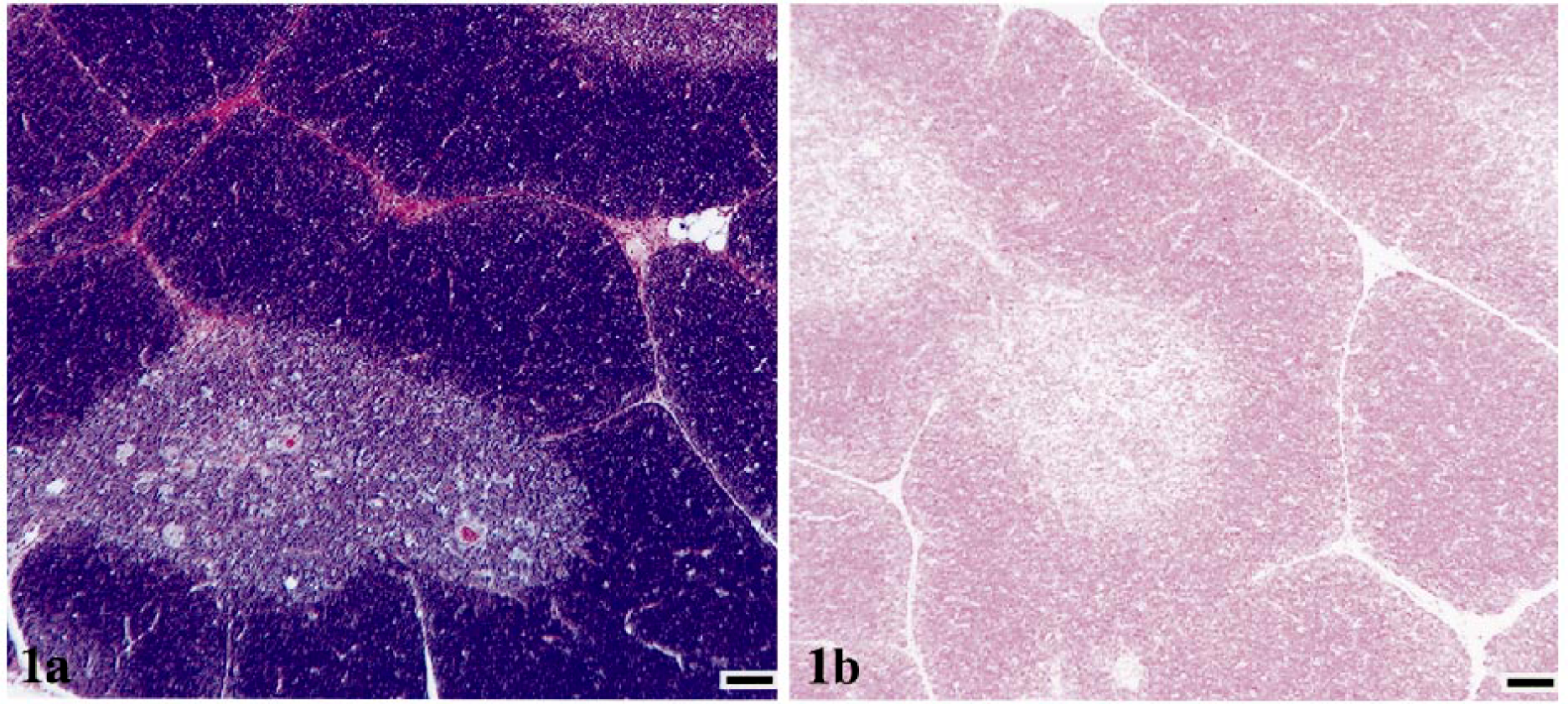
Thymus; cat No. 31, control.
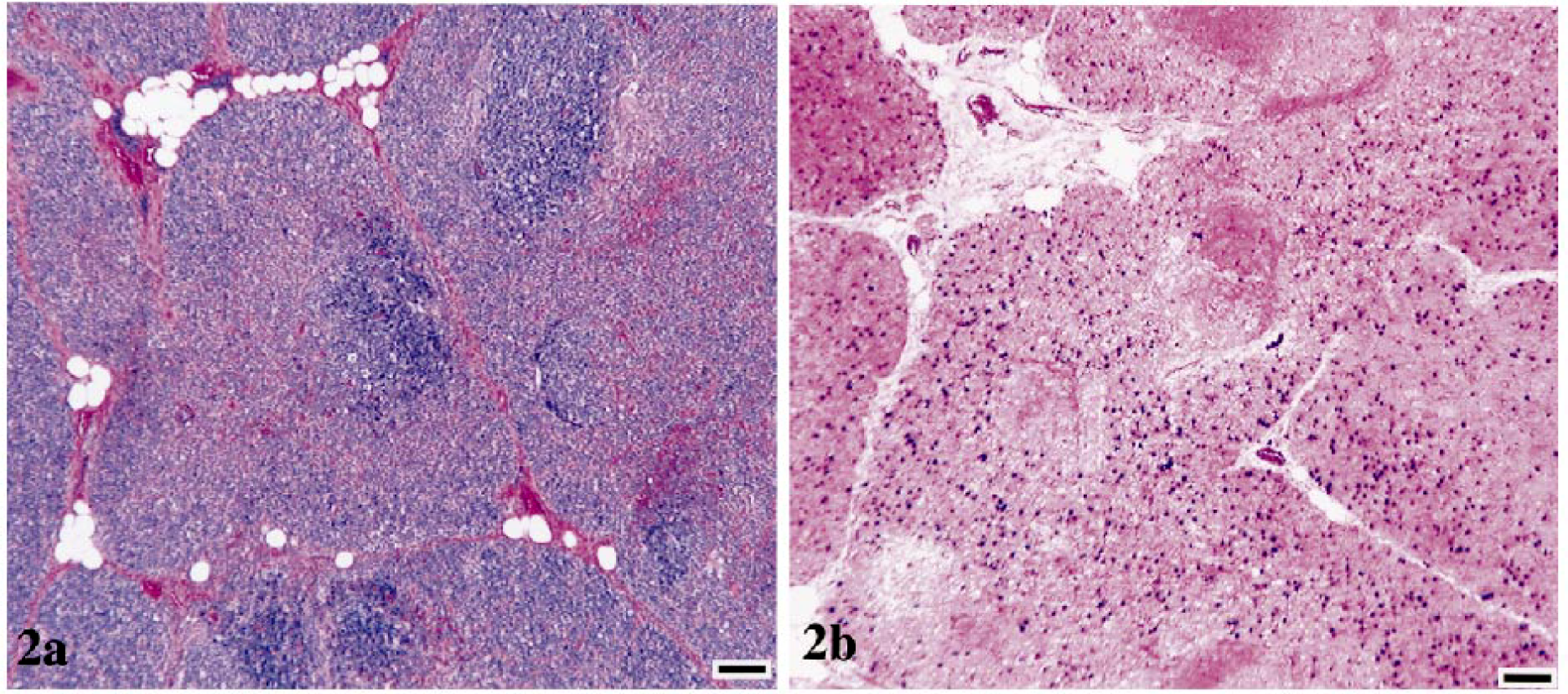
Thymus; cat. No. 15, infected with FIV-C and showing a conventional disease
course.
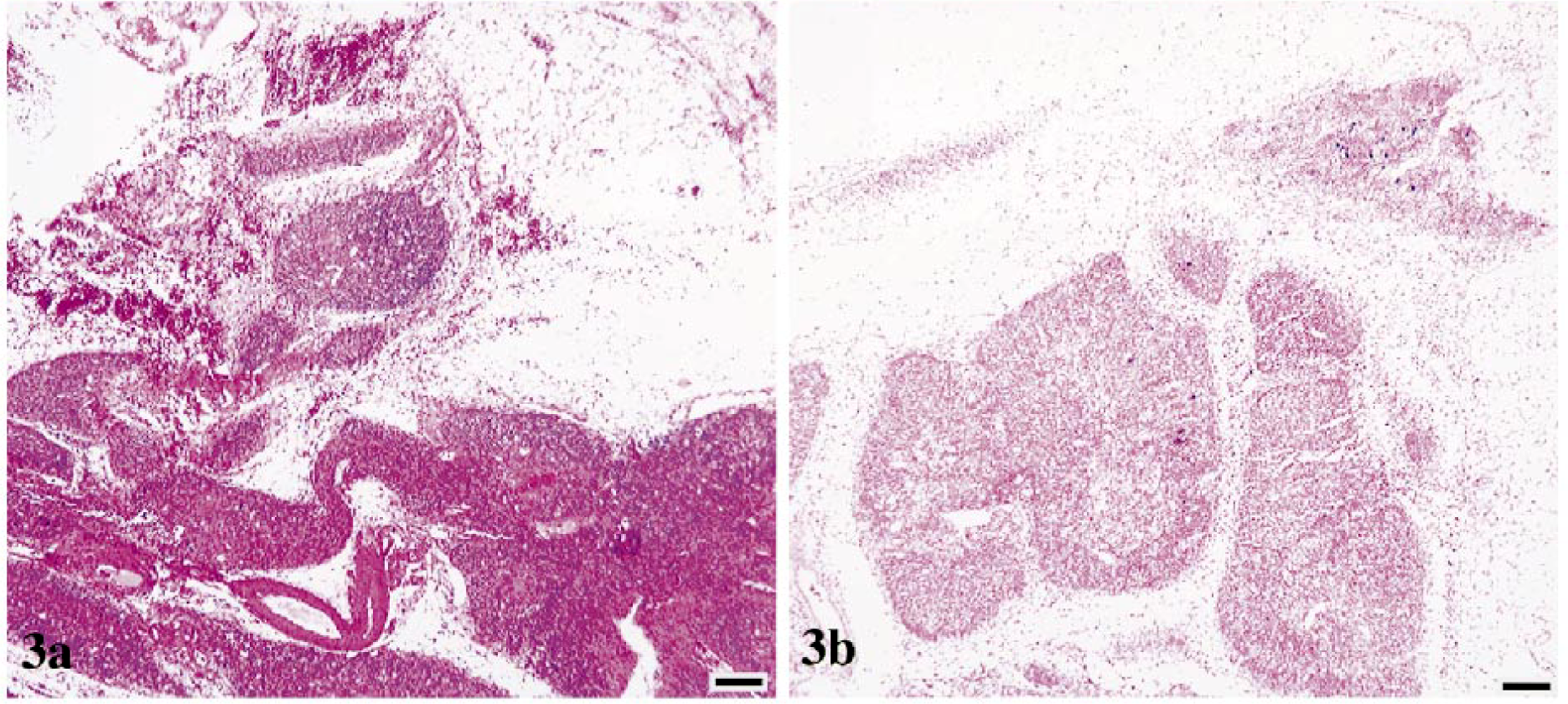
Thymus; cat No. 2, infected with FIV-C and showing a rapid disease course.
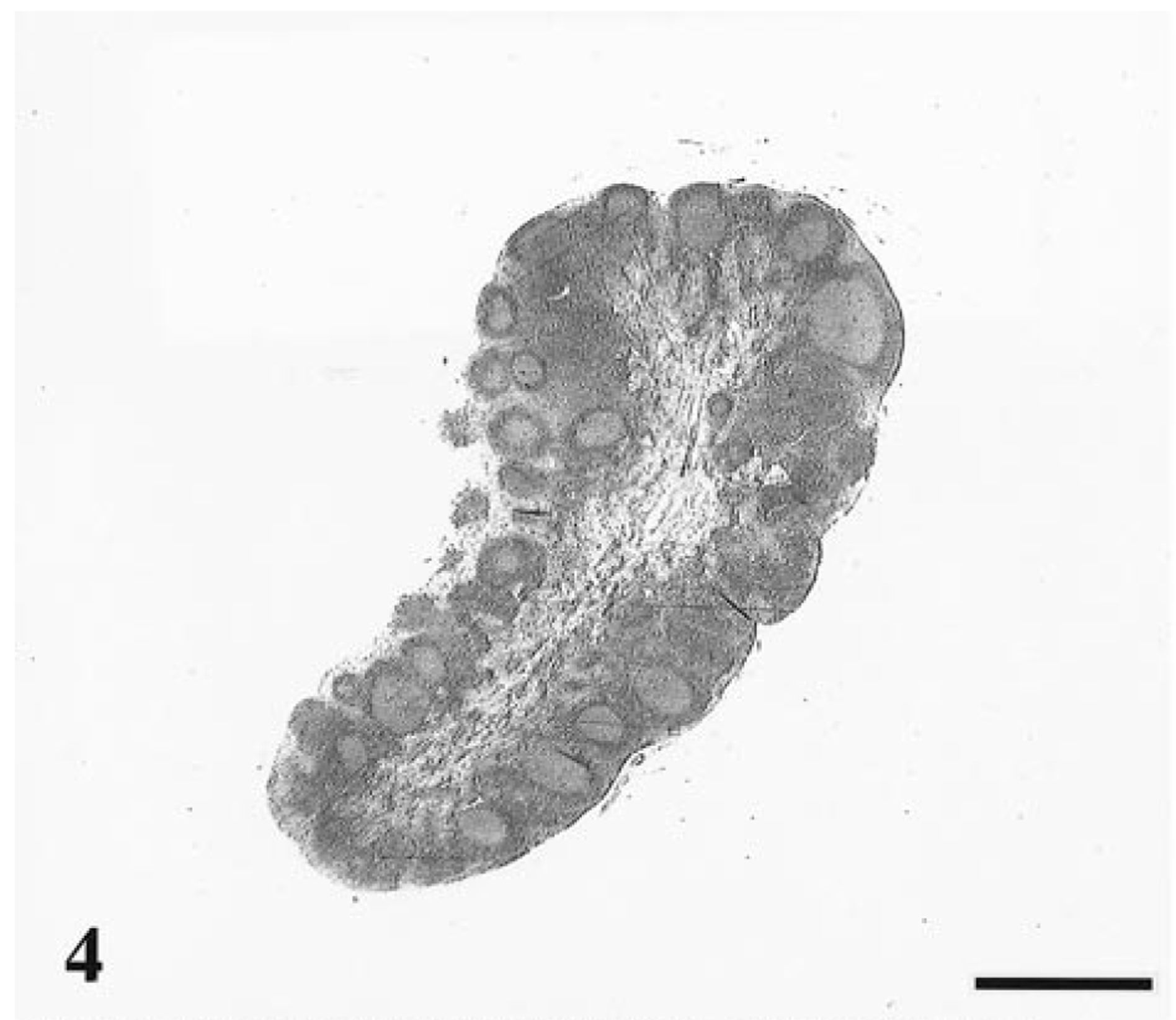
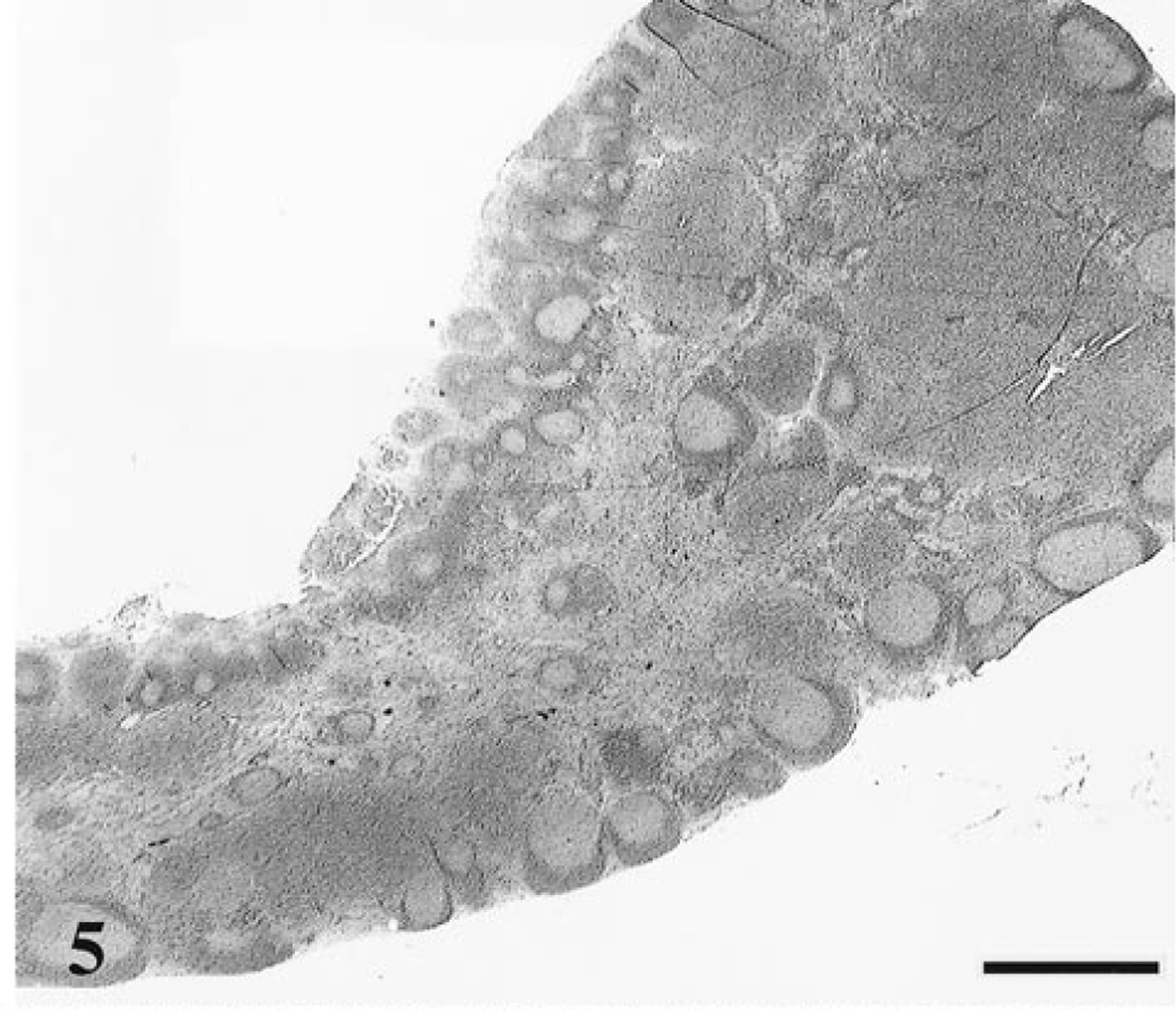
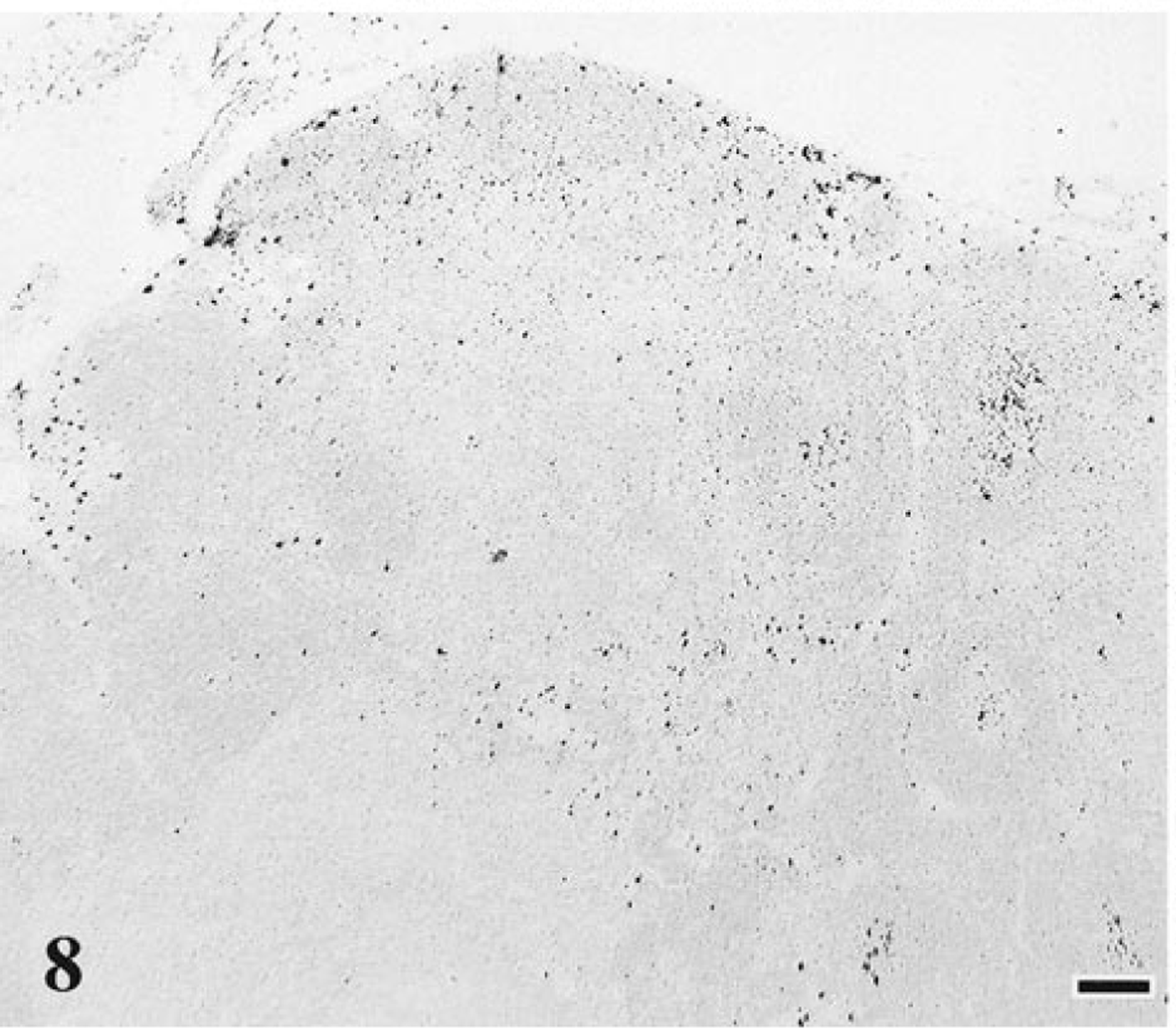
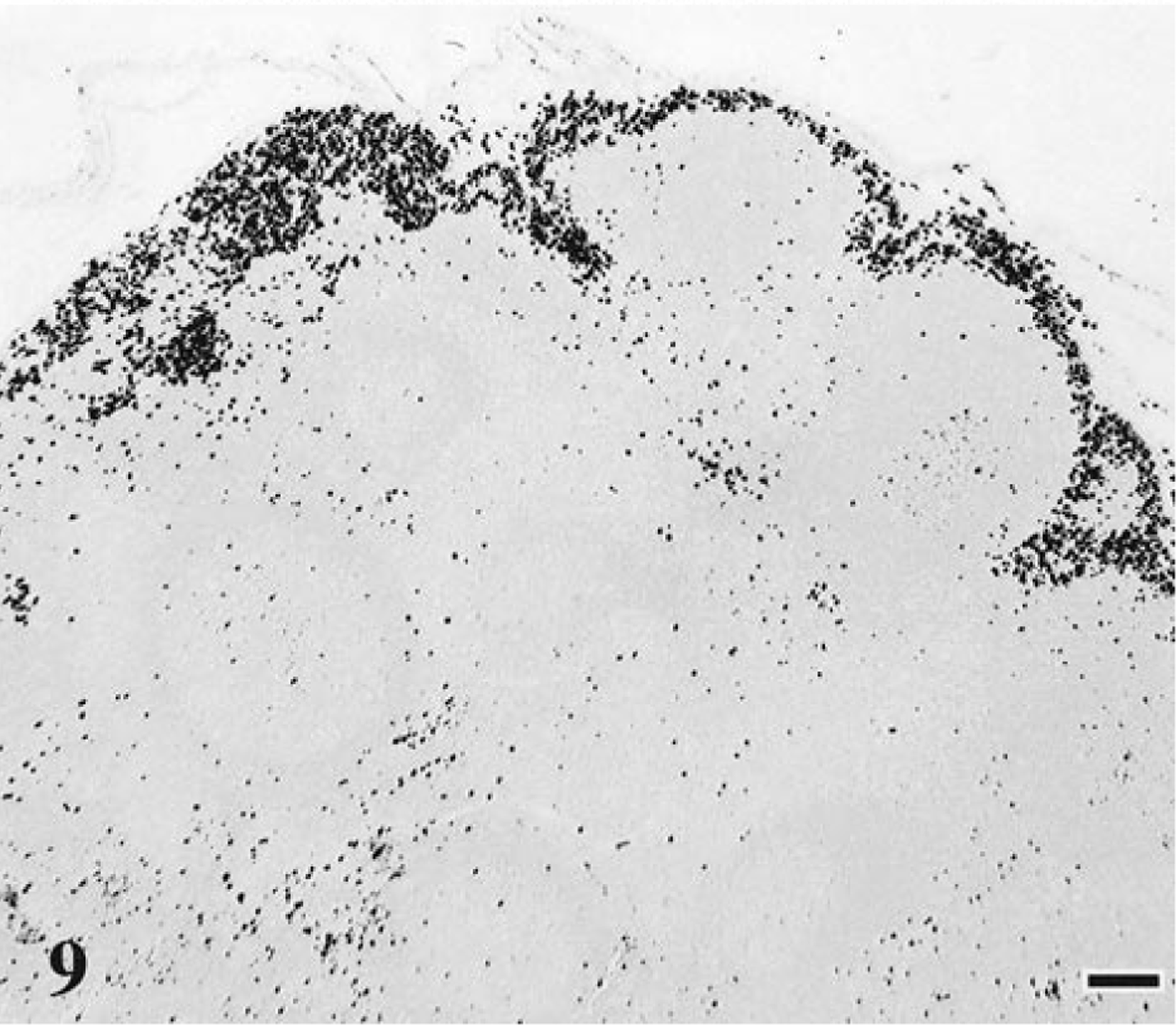
All of the 18 cats with the conventional FIV disease course exhibited thymic atrophy and thymic follicle formation and varying degrees of lymphoid hyperplasia in lymph nodes, bone marrow, and spleen (Table 1, Figs. 4, 5). In lymph nodes, three of these 18 cats (17%) had sinus histiocytosis (Figs. 8, 9), three (17%) had mild lymphoid depletion, and four (22%) had multifocal areas of necrosis (mesenteric nodes only). Lymphoid follicle formation in bone marrow was detected in seven of the 18 cats (39%). One cat had focal ulcerative dermatitis.
Internal iliac lymph node; cat No. 25, control. Normal lymph node size and architecture. HE. Bar = 1,000 μm.
Internal iliac lymph node; cat No. 15, infected with FIV-C and showing a conventional disease course. Enlarged, hyperplastic lymph node containing numerous secondary follicles, parafollicular lymphoid expansion, and distended hypercellular medullary cords. HE. Bar = 1,000 μm.
Mesenteric lymph node; cat No. 24, control. Normal distribution and number of dark-staining macrophages. Monoclonal antibody MAC387, ABC–alkaline phosphatase detection method, Vector® Red chromagen, methyl green counterstain. Bar = 100 μm.
Mesenteric lymph node; cat No. 14, infected with FIV-C and showing a conventional disease course. Increased number of dark-staining macrophages in the subcapsular sinuses. Monoclonal antibody MAC387, ABC–alkaline phosphatase detection method, Vector® Red chromagen, methyl green counterstain. Bar = 100 μm.
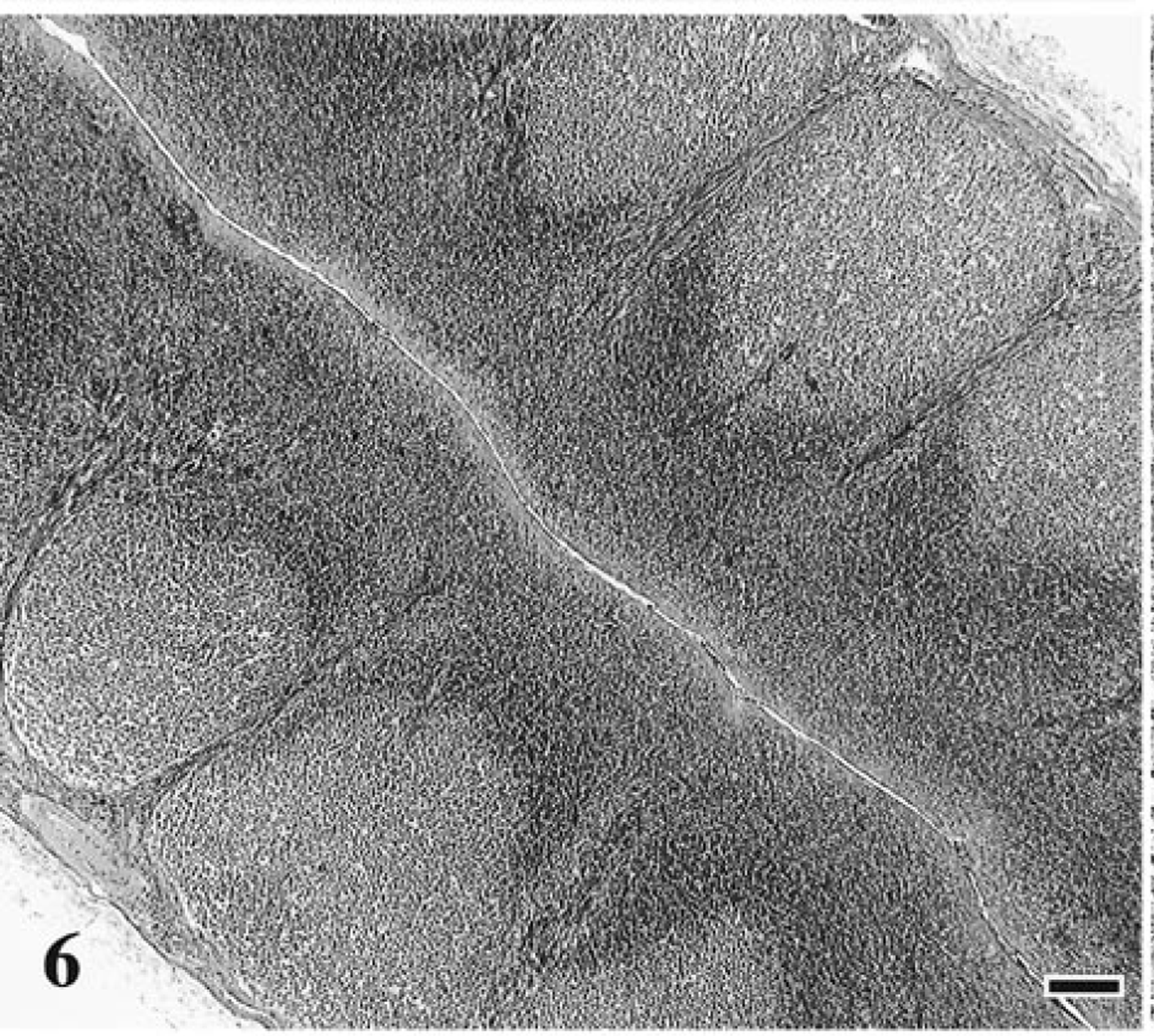
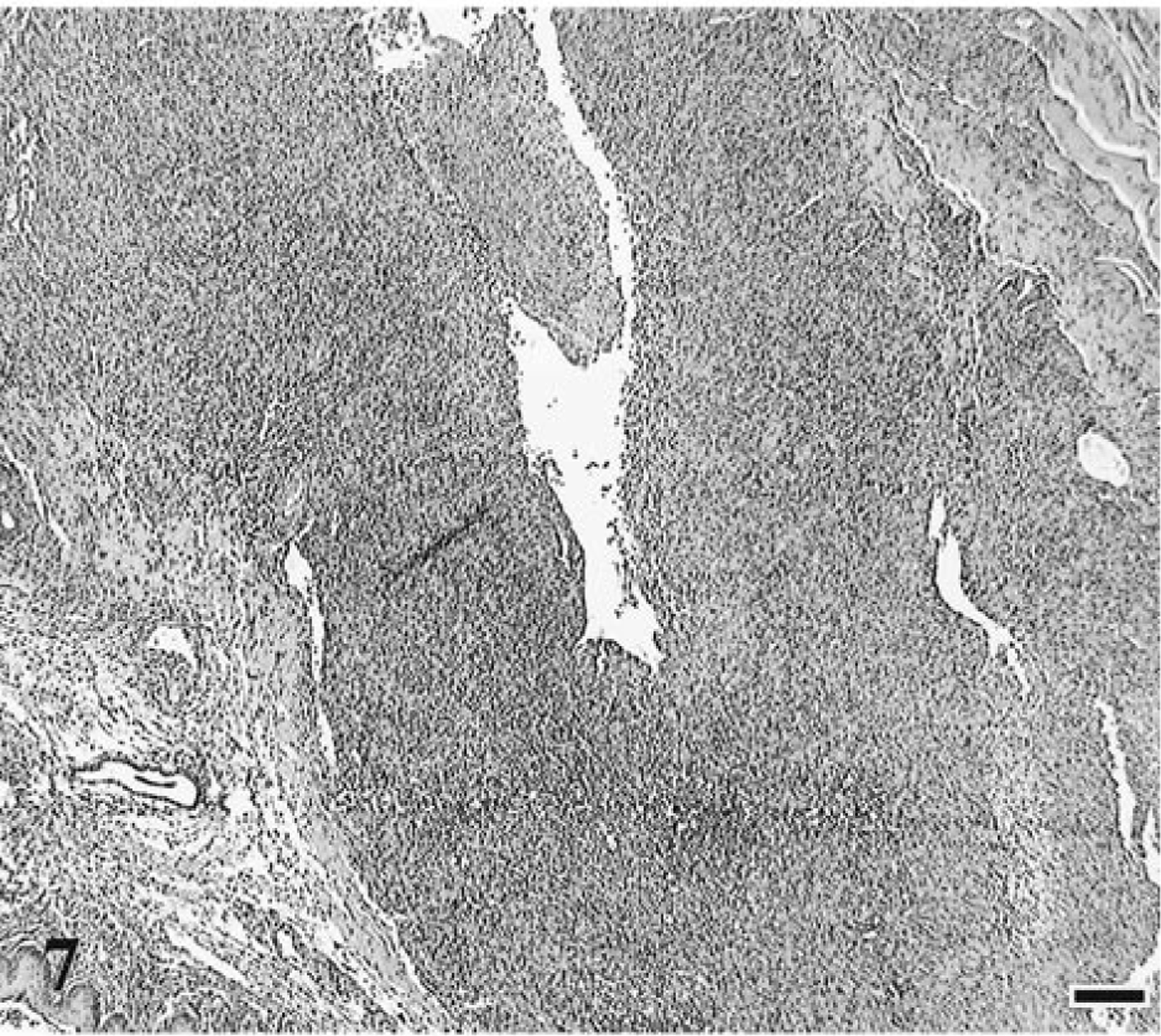
The rapidly progressive FIV disease course was characterized by a mixture of lymphoid hyperplasia and depletion in multiple lymphoid tissues (spleen, lymph node, tonsil, and mucosal-associated lymphoid tissue) in all three infected cats (Table 1, Figs. 6, 7). By contrast, lymphoid hyperplasia unaccompanied by depletion was seen only in the lymph nodes from cats with conventional/slow FIV disease progression (15 of 18 cats). In all instances, rapid progression was correlated with severe thymic atrophy, bone marrow hyperplasia, and lymphoid necrosis in tonsils, cecum, or jejunum. In addition, suppurative necrotizing lesions were detected in several organs, including oral mucosae, jejunum, cecum, submandibular lymph node, uterus, and lung. Sinus histiocytosis was detected in lymph nodes from two of three cats (67%) with rapidly progressing disease.
Tonsil; cat No. 31, control. Normal tonsilar architecture. HE. Bar = 100 μm.
Tonsil; cat No. 1, infected with FIV-C and showing a rapid disease course. Severe tonsilar depletion with complete loss of normal architecture. HE. Bar = 100 μm.
Similar to the control animals, the two cats with regressive FIV infection did not exhibit any significant gross or histologic lesions. However, one of these cats (No. 22) had mild splenic lymphoid hyperplasia.
ISH
Figures 1b, 2b, and 3b demonstrate frequency of virus-infected thymocytes by RNA ISH. Thymic tissue from cats with conventional FIV infection had numerous FIV gag/env RNA-bearing cells throughout the atrophic parenchyma (Fig. 2b). Image analysis was used to determine the frequency of FIV-bearing thymocytes (Table 2). In particular, thymic tissue from a cat with a conventional FIV disease course (cat No. 17, Fig. 2b) contained 21 unstained cells for every FIV-positive (FIV+) thymocyte. By contrast, thymic tissue from a cat with rapidly progressive FIV disease course (cat No. 2, Fig. 3b) contained approximately one FIV+ thymocyte for every 218 unstained cells. Overall, the severely atrophic thymic tissue from cats with rapidly progressive disease had fewer virus-bearing cells (average, 1:276) compared with thymuses from cats with conventional FIV disease course (average, 1:115), probably reflecting the terminal stage of disease at tissue collection. A significant difference was confirmed (P < 0.05, Mann–Whitney test) for thymic virus burden in FIV-infected cats with a conventional disease course versus cats with rapidly progressive disease. Thus, viral replication was prominent in thymic tissue of FIV-infected cats that was not already severely depleted of thymocytes.
Image analysis results of FIV-positive (FIV+) cells in thymuses of FIV-infected cats as identified by in situ hybridization.
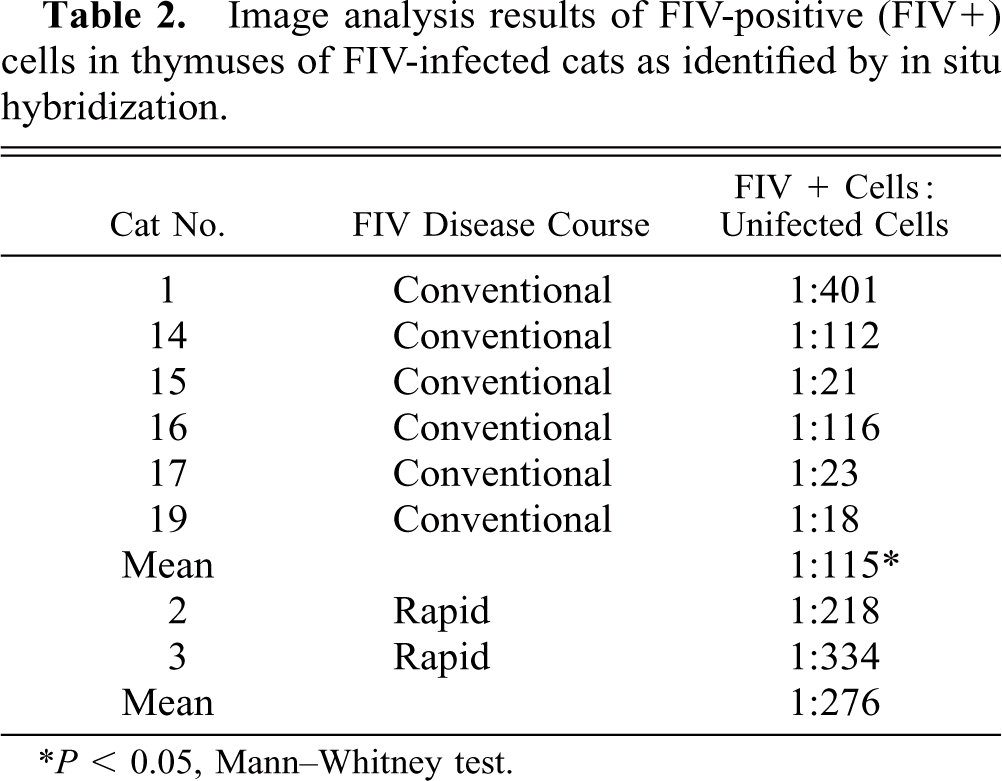
P < 0.05, Mann–Whitney test.
Immunohistochemistry
Immunohistochemical staining for CD3+ thymocytes demonstrated loss of cortical-medullary architecture and highlighted the presence of unstained B-cell follicles. In control thymuses, immature (CD3−) thymocytes were primarily in the cortices of lobules and mature (CD3+) thymocytes were in medullary regions (Fig. 10). However, in cats with both conventional and rapid disease courses, the number of unstained CD3− thymocytes was decreased, leaving primarily CD3+ thymocytes framing B-cell follicles (Fig. 11).
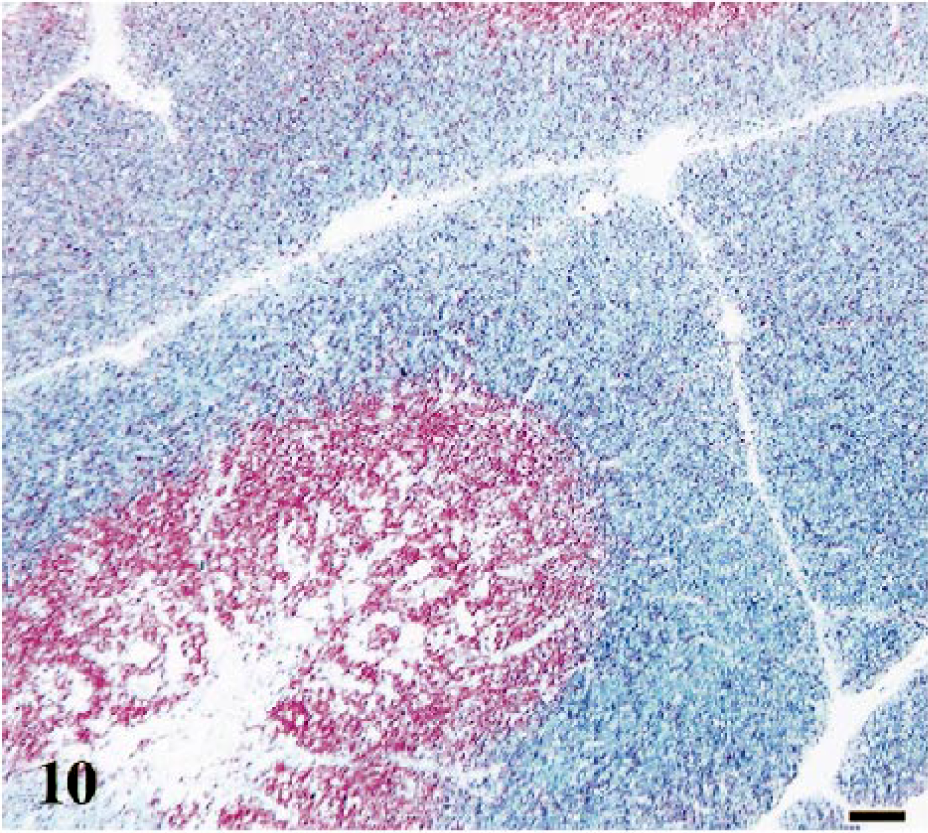
Thymus; cat No. 24, control. Normal distribution and number of red-staining CD3-positive and nonstaining CD3-negative thymocytes. Polyclonal anti-CD3 antibody, ABC–alkaline phosphatase detection method, Vector® Red chromagen, methyl green counterstain. Bar = 100 μm.
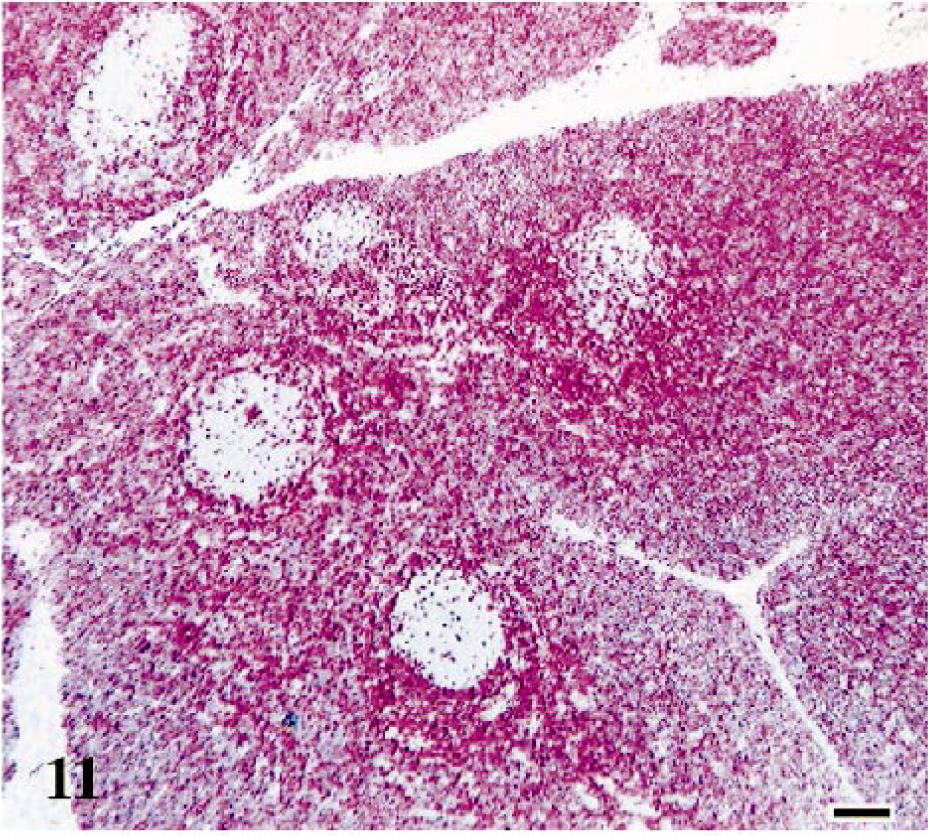
Thymus; cat No. 14, infected with FIV-C and showing a conventional disease course. Atrophic thymus contains predominately red-staining CD3-positive thymocytes, suggesting a loss of nonstaining CD3-negative cortical thymocytes. Nonstaining lymphoid follicles are highlighted. Polyclonal anti-CD3 antibody, ABC–alkaline phosphatase method, Vector® Red chromagen, methyl green counterstain. Bar = 100 μm.
No evidence of B-cell follicles was seen in thymic tissue from controls (Fig. 12). However, the follicles in FIV-infected cats stained for the B-cell marker CD45R, B220 (Ly5) (Fig. 13). These results corroborate the increased number of B cells seen with flow cytometry in FIV-infected cats versus controls.
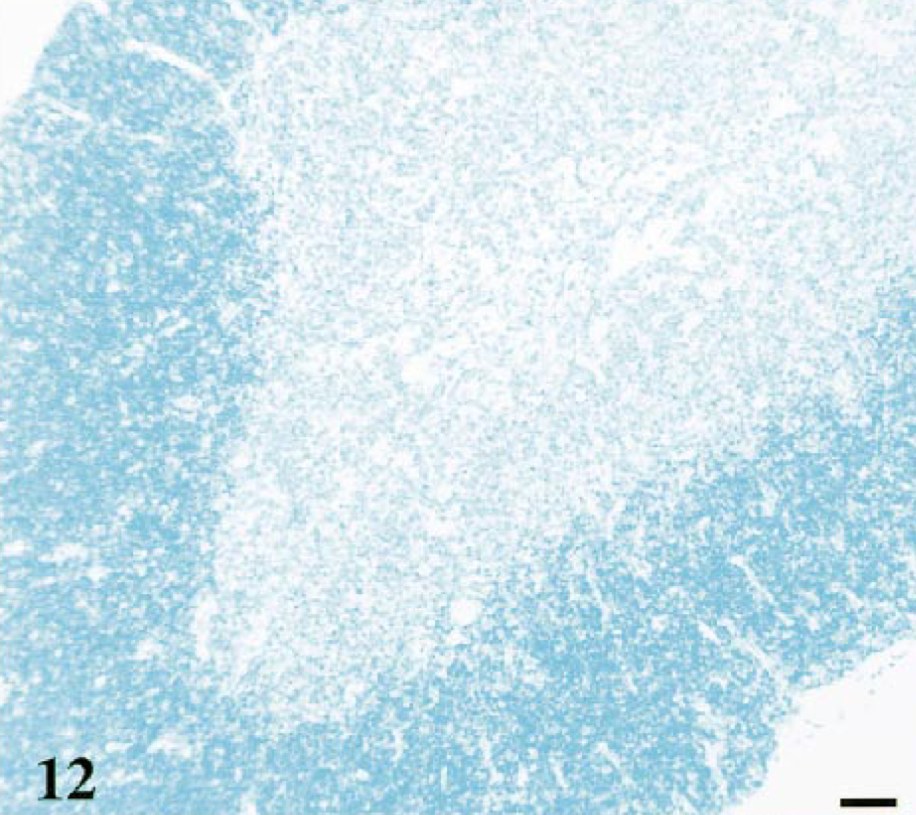
Thymus; cat No. 24, control. Normal thymic architecture without dark blue-staining B-cell follicles. Monoclonal anti-CD45R, B220 (Ly5) antibody, ABC–alkaline phosphatase detection method, BCIP/NBT chromagen, methyl green counterstain. Bar = 40 μm.
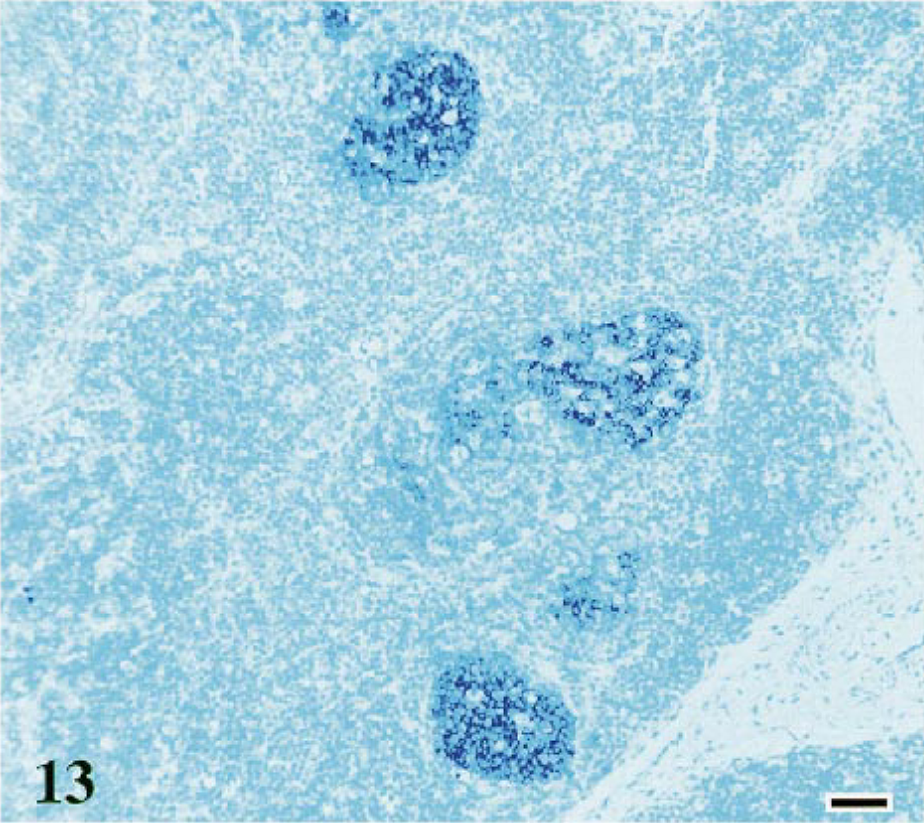
Thymus; cat No. 14, infected with FIV-C and showing a conventional disease course. Atrophic thymus contains multiple prominent dark blue-staining B-cell follicles. Monoclonal anti-CD45R, B220 (Ly5) antibody, ABC–alkaline phosphatase method, BCIP/NBT chromagen, methyl green counterstain. Bar = 40 μm.
Dual labeling with p55 and S100 antibodies demonstrate decreased numbers of dendritic cells in thymic tissue from cats with conventional (Fig. 15) and rapid FIV disease courses compared with controls (Fig. 14). Thymuses from control cats contained numerous dendritic cells in the medullary regions with lesser numbers in the lobule cortices. In both FIV-infected and control tissues, the p55 antibody stained a larger population of dendritic cells than did the S100 antibody.
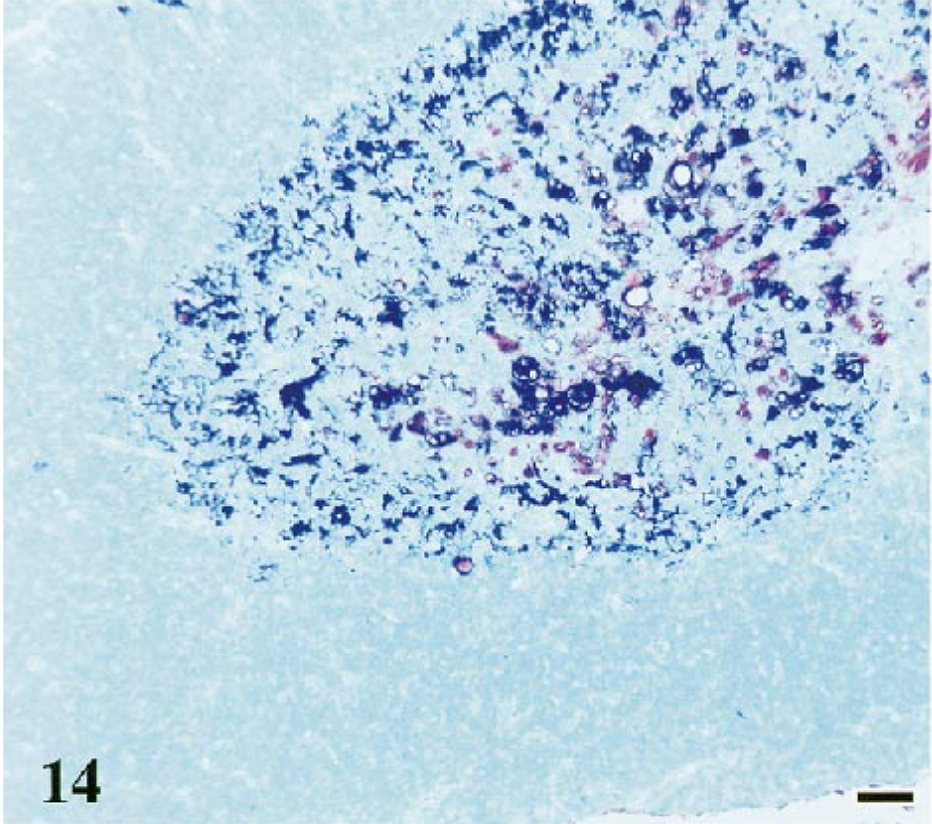
Thymus; cat No. 24, control. Normal distribution and number of black-staining p55-positive and red-staining S100-positive dendritic cells. Polyclonal antibody to S100, ABC–alkaline phosphatase detection method, Vector® Red chromagen. Monoclonal antibody to p55 followed by anti-mouse IgG–colloidal gold with silver enhancement. Methyl green counterstain. Bar = 40 μm.

Thymus; cat No. 14, infected with FIV-C and showing a conventional disease course. Loss of black-staining p55-positive and red-staining S100-positive dendritic cells in atrophic thymic tissue. One follicle (F) has black-staining p55-positive follicular and perifollicular dendritic cells. Polyclonal antibody to S100, ABC–alkaline phosphatase method, Vector® Red chromagen. Monoclonal antibody to p55 followed by anti-mouse IgG–colloidal gold with silver enhancement. Methyl green counterstain. Bar = 40 μm.
TUNEL
Numerous apoptotic cells in atrophic thymic tissue of FIV-infected cats were identified with the in situ TUNEL assay (Fig. 17). Thymic tissue from age- and litter-matched control cats displayed the expected pattern (cortex and cortical-medullary junction staining 66 ) and the degree of apoptosis expected in weanling animals (Fig. 16). Image analysis (utilizing MetaMorph® software) revealed that for each TUNEL-positive (TUNEL+) apoptotic thymocyte in control cats there were an average of 877 nonapoptotic thymocytes (Table 3). By contrast, thymuses of FIV-infected cats contained an average of one TUNEL+ apoptotic thymocyte for every 97 nonapoptotic thymocytes. The apoptotic thymocytes were primarily located in the cortex and follicular germinal centers. A significant difference was confirmed (P < 0.05, Mann–Whitney test) between the degree of apoptosis occurring in thymuses of FIV-infected cats and that in controls.
Image analysis results of TUNEL–positive (TUNEL+) apoptotic thymocytes in FIV–infected cats.
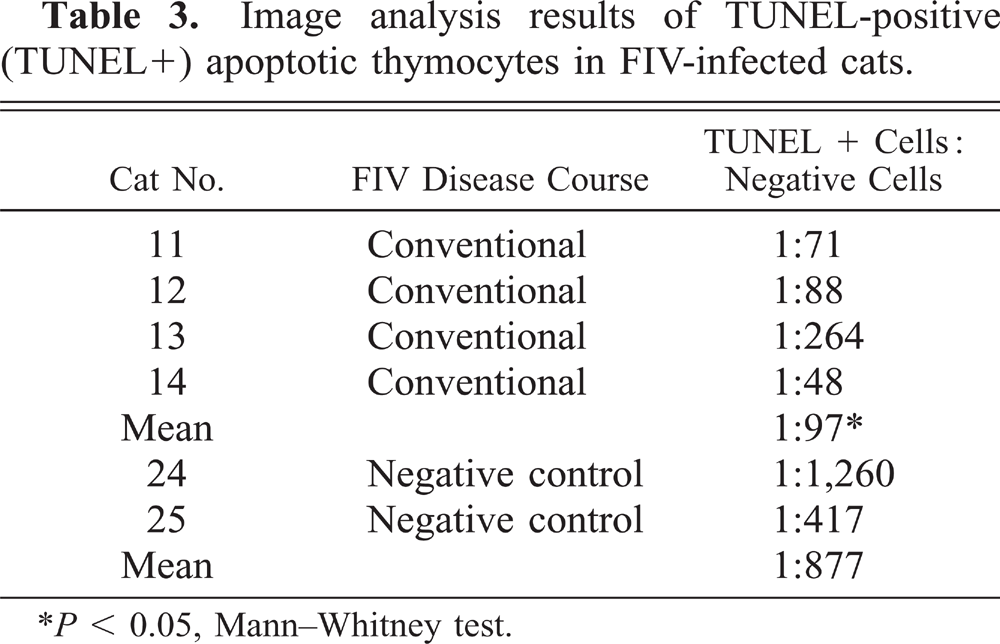
P < 0.05, Mann–Whitney test.
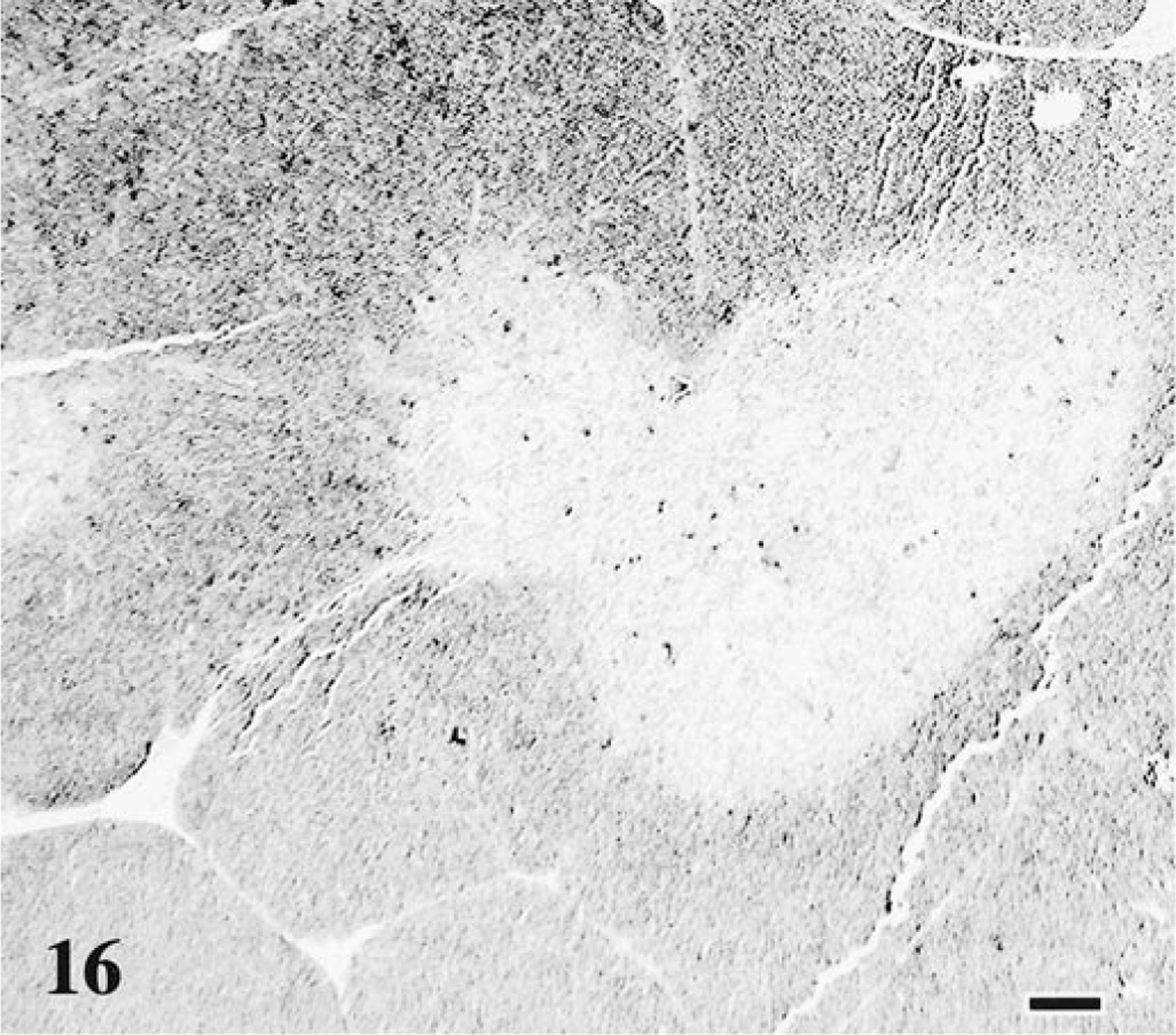
Thymus; cat No. 24, control. Normal distribution and number of dark-staining apoptotic thymocytes. TUNEL method. Digoxigenin-labeled nucleotides detected with an anti-digoxigenin–alkaline phosphatase antibody, BCIP/NBT chromagen, nuclear fast red counterstain. Bar = 100 μm.

Thymus; cat No. 14, infected with FIV-C and showing a conventional disease course. Increased number of dark-staining apoptotic thymocytes in lymphoid follicles and remnant cortical tissue. TUNEL method. Digoxigenin-labeled nucleotides detected with an anti-digoxigenin–alkaline phosphatase antibody, BCIP/NBT chromagen, nuclear fast red counterstain. Bar = 100 μm.
Thymus weight/body weight comparisons
Thymic depletion was a prominent feature of FIV-C infection. The average thymus weight:body weight ratios and terminal CD4+ cell counts of cats having either conventional or rapidly progressive FIV infections were significantly different than those of controls (P < 0.05, Mann–Whitney test), whereas ratios of FIV-exposed but uninfected cats and cats with regressive infections did not differ significantly from ratios of controls. By comparing thymus weight:body weight ratios and terminal CD4+ cell counts, cats with accelerated or conventional FIV infections were significantly different from both age-matched controls and FIV-exposed but uninfected cats (P < 0.05, Mann–Whitney test). However, cats with regressive FIV infections did not differ significantly from age-matched controls or FIV-exposed but uninfected cats (Fig. 18). Additionally, cats with rapidly progressive FIV infections were significantly different (P < 0.05, Mann–Whitney test) from cats with conventional FIV infections, as indicated by the more severe thymic atrophy and lower CD4+ cell counts seen in the rapid progressors.
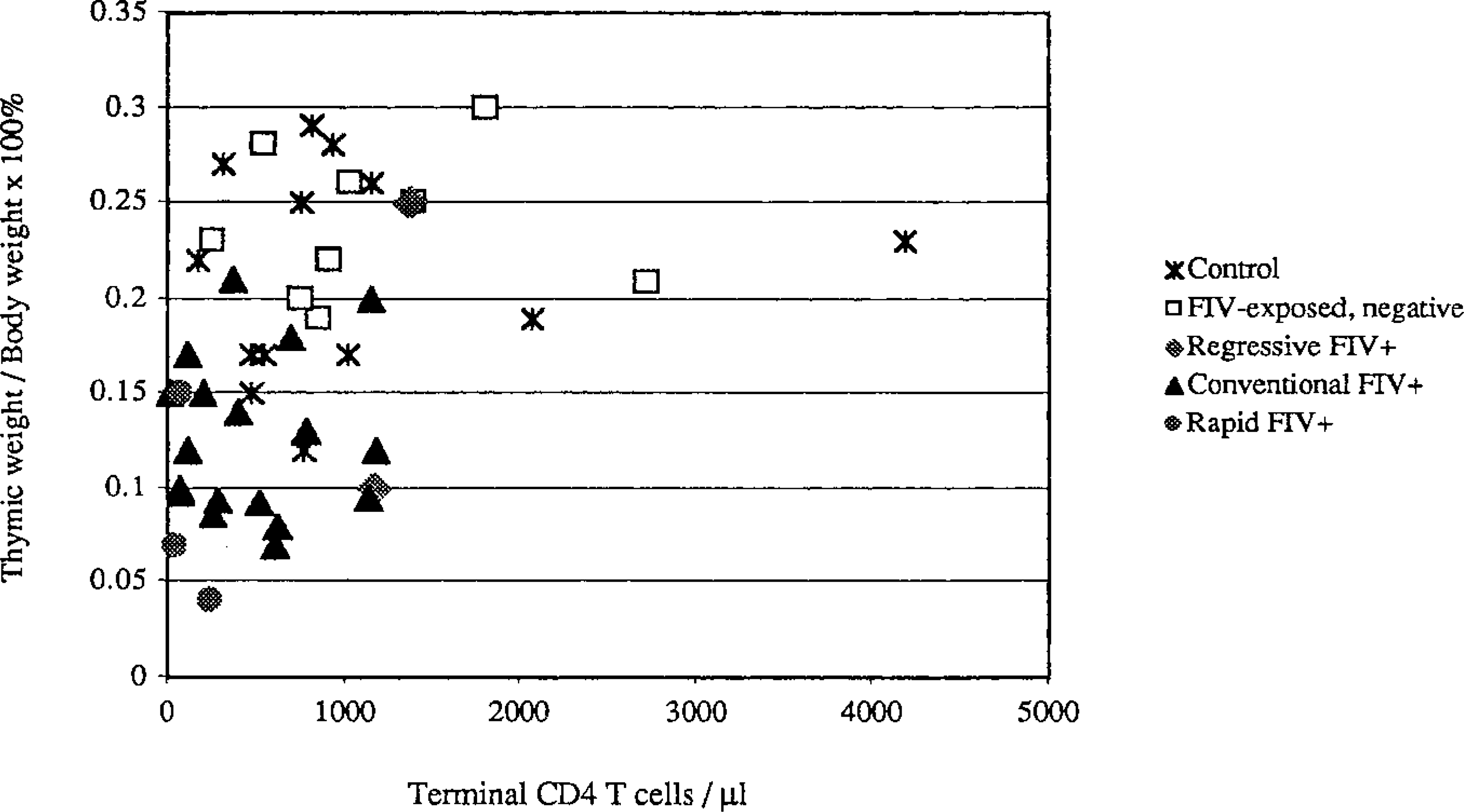
Thymus weight:body weight ratio plotted against terminal CD4 T-cell count for each study animal: conventional FIV+ (▴), rapid FIV+ (○), regressive FIV+ (⋄), FIV exposed but uninfected (□), and control (★). Cats persistently infected with FIV (▴, ○) and uninfected cats (★, □) cluster in different areas of the scatter plot. The FIV-infected cats lie within the lower left quadrant, reflecting lower terminal CD4 T-cell counts and lower ratios because of smaller thymuses.
Flow cytometry
In FIV-infected cats, CD4+/CD8+ and CD4+/CD8− thymocytes were significantly decreased and CD45R+ B cells were significantly increased (P < 0.05, Mann–Whitney test) (Fig. 19). The CD4−/CD8+ populations in FIV-infected cats were slightly increased compared with controls, but this difference was not significant. Thymocytes from one FIV-exposed but uninfected cat (No. 45) were also examined, and the numbers mirrored control values.
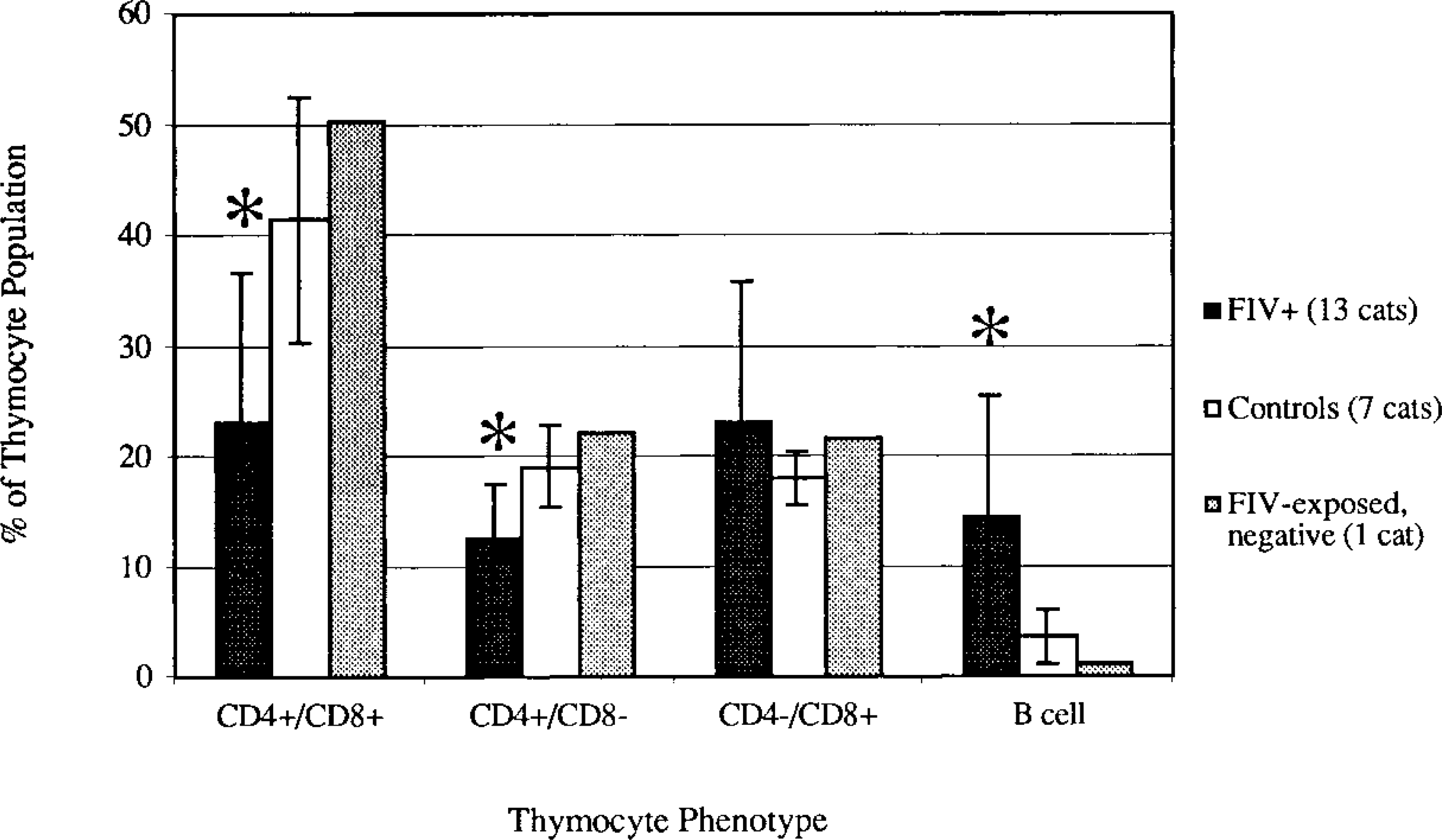
The CD4+/CD8+, CD4+/CD8−, CD4−/CD8+, and CD45R+ (B cell) thymocyte populations of 13 FIV-C–infected cats having conventional disease course (▪), seven control cats (▪), and one FIV-exposed but uninfected cat (□) were assessed by flow cytometry. CD4+/CD8+ and CD4+/CD8− thymocyte populations were significantly decreased, and B cells were significantly increased in FIV-C–infected cats compared with age- and litter-matched control animals. ∗ P < 0.05 (Mann-Whitney test).
Discussion
Previous work in our laboratory demonstrated that rapid intravenous passage of a clade C FIV isolate (FIV-C PGammer) resulted in rapidly progressive, fatal disease in >50% of infected cats. 12–14 In the present study, mucosal inoculation with FIV-C also resulted in rapidly progressive disease but in just 8.3% of infected animals. Instead, a wider spectrum of disease courses was observed, spanning from rapid to regressive. The three FIV-C disease courses and the lymphoid lesions detected were directly related.
The thymus was a major target tissue in FIV-C infection. In particular, the CD4+/CD8− and CD4+/CD8+ thymocyte populations were selectively decreased. These findings are consistent with those of other studies in the FIV and simian immunodeficiency virus (SIV) systems. 33 34 50 72 In addition, selective depletion of CD4-bearing thymocytes has been observed in HIV-infected adults and in children. 6 8 58 67 As with FIV infection, the severely depleted thymic tissue from children dying of AIDS contained minimal evidence of virus replication. 30 63 It appears that loss of CD4+ cells spared only the least susceptible thymocyte populations (CD4−/CD8+ and CD3+) and infiltrating CD4−/CD8+/CD3+/CD1− inflammatory cells. 25 50 58 72 73 In both conventional and rapid FIV infections, the thymic cortex was comprised of primarily CD3+ thymocytes (single positive mature thymocytes/lymphocytes 8 ), whereas in controls a large population of unstained cortical cells (CD3− immature thymocytes 8 ) was present. Immature human CD4+/CD8+ thymocytes express the highest levels of both CD4 and CXCR4 and are preferentially infected and depleted by syncytium-inducing (SI) HIV-1 strains in vitro. 32 SI isolates appear to replicate faster, produce higher titers, and are more cytopathic in immature and mature CD4+ thymocytes than are non-SI isolates. 70 In that FIV-C-PGammer is also T-cell cytopathic in vitro (unpublished data), this isolate may be somewhat analogous to T-cell tropic HIV SI strains. The immunohistochemistry, flow cytometry, and ISH findings suggest that FIV-C has preferential tropism for immature thymocytes, which would contribute to elimination of peripheral CD4+ cells and subsequent immunodeficiency.
Decreased p55+/S100+ medullary dendritic cells and decreased Hassall's corpuscles were observed in FIV-C–infected cats as compared with controls. Although no references utilizing the S100 or p55 antibodies for the identification of feline dendritic cells are available, other researchers have utilized these antibodies to identify dendritic cells in human tonsil, 16 adenoid, 17 and thymic 21 56 tissues. Vacuolization, shrinkage, and cytolysis of thymic cortical epithelial and interdigitating dendritic cells have been reported early in the course of SIV infection. 44 FIV-C–induced damage to thymic dendritic and epithelial cells could also contribute to thymic depletion and peripheral T-cell loss.
The number of cells containing FIV-C mRNA as revealed by ISH did not correlate with the degree of thymic atrophy. Viral cytopathicity is one mechanism by which these findings can be explained. The thymic target cells could have been eliminated early in the course of virus infection, leaving only the least susceptible thymocytes, as previously discussed. In contrast, loss of CD4+/CD8+ thymocytes independent of cell infection has been reported in HIV disease, and multiple HIV studies have demonstrated an association between apoptosis and cell loss. 23 24 40 77 Thymic tissue from FIV-C–infected cats had significantly higher levels of apoptosis when compared with age- and litter-matched control cats. Apoptotic cells were scattered throughout the atrophic thymic tissue but were also prominent in the follicular germinal centers, as described for the lymph nodes of HIV-infected individuals. 45 Similarly, a higher apoptotic index was found in the thymic cortex (especially in the follicle germinal centers) of FIV-infected cats versus controls. 60 In SIV-infected macaques, increased apoptosis was seen in the thymic cortex by 7–14 days postinfection and was followed by depletion of thymocyte progenitors (CD34+ and CD4+/CD8+) by day 21. 73 Thymic dendritic cells release, upon infection with primary HIV isolates and laboratory strains, soluble factors that induce the killing of primary CD4+ and CD8+ thymocytes and activated but not resting PBMC. This cytotoxic factor–mediated cell death involves the direct/indirect contribution of FasL and tumor necrosis factor α (TNF-α). 3 In addition, disease progression in FIV-infected cats is associated with production of TNF-α, which has been shown to induce apoptosis in a cell line chronically infected with FIV. 36 37 48 Thus, apoptosis is likely to be another mechanism contributing to thymic CD4+ depletion caused by FIV-C and other lentiviruses.
A spectrum of peripheral lymphoid cell alterations ranging from hyperplasia to depletion was observed in FIV-C–infected cats. At one extreme were cats with regressive FIV-C infection, in which little or no alterations were detected. At the other extreme were cats with rapidly progressive disease, in which lymphoid hyperplasia was variably mixed with lymphoid depletion and necrosis in the tonsils and mucosa-associated lymphoid tissue. Follicular hyperplasia followed by involution and depletion has been described in HIV, SIV, and FIV infections. 1 5 7 9 27 29 53 57 62 75 Thus, as with the primate lentiviruses, lymph node histology reflects clinical stage of disease in FIV-C infections.
Many of the FIV-C–infected cats with conventional disease courses had bone marrow lymphoid follicle formation and lymph node sinus histiocytosis. Similarly, expansion of the paracortical macrophage population and B-cell hyperplasia accompanied by sinus histiocytosis have been described in lymph nodes of HIV-infected patients. 11 28 41 43 Thus, the lymph node reactive changes observed in FIV-C–infected cats parallel those reported in HIV-infected people.
In addition to profound lymphoid depletion, cats with rapidly progressive FIV-C infections had intestinal and mucosal ulcerations with necrosis and disseminated bacterial infections. 14 Rapidly fatal infection characterized by severe intestinal mucosal damage has been reported in macaques infected with SIVsmmPBj14. 18–20 In contrast to the cats with accelerated FIV-C disease, SIVsmmPBj14-infected macaques die within 21 days with a systemic shocklike syndrome and severe diarrhea. 14 18–20 Rapid disease progression with lymphoid depletion more analogous to FIV-C disease has been described in SIV-infected macaques, which succumb to AIDS after a few months (versus weeks) of infection. 15 38 52 Although there have been occasional reports of individual cats developing rapid disease following FIV infection, 9 49 54 prior to FIV-C transmission studies this response was rare. As with SIVsmmPBj14 infection, 64 rapid FIV-C disease occurred less frequently after mucosal than after intravenous infection. In those animals affected, however, the resulting disease was equivalent to that associated with intravenous passage, indicating that the virulent genotype can cross the mucosal barrier. The rapid progression in SIVmac239 and SIVsmmPBj14 infections involves amino acid substitutions in the nef gene, leading to enhanced infection of resting T cells. 35 46 61 Better understanding of the viral genetic basis of the rapid FIV-C disease awaits generation of an infectious molecular clone.
In conclusion, although the pathogenic FIV subtype C isolate PGammer maintained its thymic tropism and pathogenicity following mucosal transmission, three different infection courses were observed. The severity of the disease course produced by mucosal FIV-C infection was correlated with the severity of the thymic lesions.
Footnotes
Acknowledgements
We thank Dr. James Mullins for the partial molecular clones of FIV-C PGammer, Dr. Richard Reyes for his assistance with riboprobe generation, and Dr. Kevin Keane for assistance with image analysis. This research was funded by grants K08-A1-01403 and R01-A1-33773 from the Division of AIDS, NIAID, NIH.
