Abstract
An anaplastic carcinoma was found in one of the two parathyroids of a 2-year-old male Wistar rat, which was sacrificed at the end of a carcinogenicity study. Morphologically, it was characterized by the presence of nodular areas of pleomorphic and dense cells with numerous atypical mitoses and large regions of smaller and dark monomorphic cells devoid of mitoses and forming small cystic spaces. Local invasion of the capsule and pronounced compression of the parenchyma of the thyroid gland were observed. Immunohistochemically, the tumor was markedly positive for the parathyroid hormone and negative for the thyroid transcription factor. The proliferative activity was assessed by immunostaining the endogenous cell proliferation associated-antigen Ki-67, and the proliferating cell nuclear antigen. The diagnosis of carcinoma of the parathyroid was made on the basis of microscopic and immunohistochemical findings.
The function of parathyroid glands is to maintain normal levels of serum calcium through the synthesis and secretion of parathyroid hormone (PTH). Parathyroids of the rat derive from the endoderm of the third pharyngeal pouch, and only one pair of parathyroid glands are formed. 4 In other mammals, including humans, there are two pairs of glands, both deriving from the third and fourth pharyngeal pouches. 2 In rats, the parathyroids are located on the anterior or lateral parts of the thyroid glands, and in females the size of the gland is up to twice that in males, which, depending on the age (in old animals is bigger), measures about 0.9 × 0.6 mm in diameter. The parathyroid glands are separated from the thyroid by a thin capsule of connective tissue. The parenchyma consists of densely packed cords or clusters of polygonal cells adjacent to capillaries and separated by a delicate stroma of reticular and collagen fibers. The principal cell type in the rat is the chief cell, which is uniform with spherical to oval or elongated nuclei, dense chromatin, and scant to moderate eosinophilic cytoplasm. Dark or clear cells that are observed represent differences in the functional state of the chief cells. The mitochondria-rich oxyphilic cell, observed in the human parathyroid, is not present in rat. These cells, which are large with strongly acidophilic cytoplasm, have a poorly developed granular endoplasmic reticulum, Golgi apparatus, and secretory granules, suggesting that oxyphilic cells do not have an active function in the biosynthesis of PTH. Chief cells synthesize and secrete the PTH. This hormone, together with calcitonin and vitamin D, is involved in the regulation of calcium homeostasis and is released in response to low blood Ca++ level.
Proliferative lesions of the parathyroid gland include hyperplasia (focal or diffuse), adenomas, and carcinomas. Neoplasms of parathyroid gland are uncommon in all animal species, including humans. 5 , 8 , 10 , 11 To the best of our knowledge, this is the first time that such investigations have been performed and reported in a parathyroid gland tumor in the rat.
In the present investigation, the affected animal was a 2-year-old male Wistar rat from a long-term study conducted in a contract laboratory organization under barrier maintenance conditions. The animal, from the control group, received orally by gavage the vehicle formulation (sodium carboxymethylcellulose diluted at 1% in purified water) 7 days a week for a period of 104 weeks. Animal care and handling procedures were carried out in complete accordance with the laws on animal protection of France. The rat was sacrificed at the end of the study, and a complete necropsy was performed.
Parathyroid glands were fixed together with the thyroid gland by immersion in 10% buffered formalin, dehydraded in a graded ethanol series, and embedded in paraffin wax. Serial sections were cut at 4- to 5-μm thickness and were either stained with hematoxylin and eosin (HE) or used for immunohistochemistry. Immunohistochemical reactions were performed using the streptavidin-peroxidase (StreptABC/HRP) method with the following mouse monoclonal (mAb) antibodies: 1) anti-PTH mAb (Novocastra Laboratories Ltd, Newcastle upon Tyne, UK, clone OP4, lot 213104, diluted 1:200); 2) anti–thyroid transcription factor (TTF-1) mAb (Dako Diagnostic AG, Zug, Switzerland, clone 8G7G/1, lot 128, diluted 1:50); 3) antiproliferating cells Ki-67 mAb (MIB-5, Immunotech, Milan Analytica AG, La Roche, Switzerland, lot 1297, diluted 1:1,200) and 4) anti–proliferating cell nuclear antigen (PCNA) mAb (PC10, Dako, lot 043, diluted 1:10,000). All sections were deparaffinized in xylene, rehydrated through graded ethanol, and endogenous peroxidase activity was blocked with hydrogen peroxide (0.5% in methanol for 30 minutes). Sections prepared for Ki-67 and PCNA antibody immunoreactions were placed in a Coplin jar containing an antigen retrieval solution (Biogenex, San Ramon, CA, USA) and heated eight times (5 minutes each) in a microwave processor at 95 C. Primary antibodies were applied for 18–22 hours at 4 C. For the StreptABC/HRP method, biotinylated goat anti-mouse/rabbit immunoglobulins (Dako, lot 068, diluted 1:100) and StreptABComplex/HRP (Dako, lot 068, diluted 1:100) were successively applied at room temperature (RT) for 25 minutes each. The immunohistochemical reactions were developed in freshly prepared 3,3′-diaminobenzidine tetrahydrochloride (Amersham Laboratories, Buckinghamshire, UK), 10 mg in 13 ml of 0.05 M Tris buffer containing 0.015% of hydrogen peroxide, 1–10 minutes at RT. Negative controls were obtained by omitting the primary antibodies.
Cell proliferation was assessed using Ki-67 and PCNA. The proliferating index (PI) was defined as the percentage of labeled cells divided by the total number of cells. The total number of cells (labeled and unlabeled cells together) counted for each immunoreaction ranged between 3,000 and 3,800. Tumor size (mm) was determined by measurement of the two greatest tumor dimensions in HE sections using a Zeiss microbar (1 mm) under the light microscope.
Postmortem examination disclosed an enlargement of spleen and a firm grayish/whitish mass in the forestomach beneath the mucosa, whereas no macroscopic alterations were noted in the neck and thyroid region. Microscopically, the mass in the forestomach was a fibrosarcoma. The few other microscopic findings reported corresponded to the background changes occurring in ageing rats of this strain.
The tumor observed in the parathyroid had an oval shape and measured 2 × 1.5 mm in diameter. Under light microscopy, two different patterns of cellular morphology were present: the predominant area of the tumor (accounting for about 60% of the total; Fig. 1) was composed of rather monomorphic and dark chief cells with round or oval small nuclei with dispersed chromatin, clearly evident nucleoli, and scant amounts of cytoplasm (Fig. 2). In this area, mitoses were not detectable. The neoplastic cells were arranged in a trabecular pattern (Fig. 1), palisading around the capillaries, or forming acinar structures within a bed of fine fibrovascular tissue. Small and focal hemorrhages and scanty hemosiderin pigment were also present. The second cellular pattern consisted of nodules of pleomorphic neoplastic cells with numerous atypical mitoses (Fig. 2), up to 10 per 40× high-power field. Cells were larger with more abundant lightly eosinophilic cytoplasm, pleomorphic and vesiculated nuclei of irregular size, and one to three prominent nucleoli. A few apoptotic cells were observed in these areas. The nodules compressed the other predominant area of the neoplasm, in which they were embedded, and the tumor compressed the surrounding thyroid gland, the remnants of the normal parathyroid and its capsule (Figs. 1, 2). Focal infiltration of the capsule was also present.
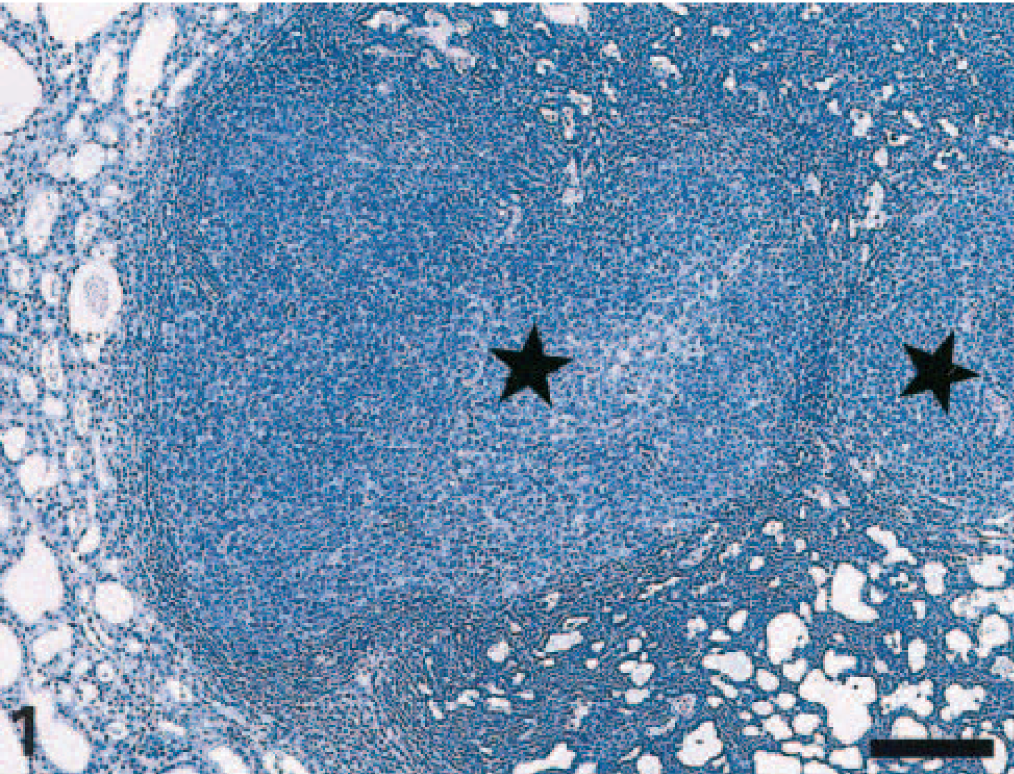
Parathyroid gland carcinoma; Wistar rat. Note the presence of several neoplastic nodules (stars) exhibiting a solid cellular pattern surrounded by large areas of monomorphic tumor cells displaying a trabecular pattern with acinar formations. The adjacent thyroid parenchyma (left) is compressed by the neoplasm. HE. Bar = 160 μm.
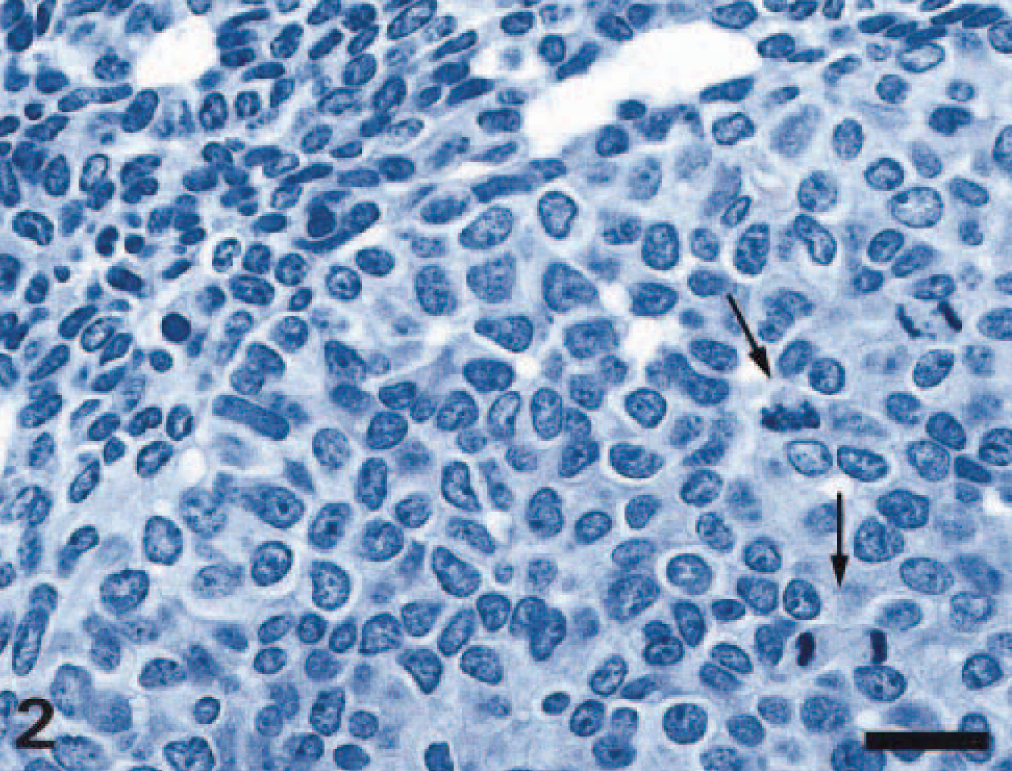
Parathyroid gland carcinoma; Wistar rat. Higher magnification of Fig. 1. Nodular neoplastic formation containing pleomorphic cells with large irregular nuclei, with prominent nucleoli, and abundant cytoplasm. Numerous mitoses are present (arrows). Area of smaller, dark, monomorphic tumor cells (upper left) forming acini appear to be compressed by the large nodule. HE. Bar = 20 μm.
Immunohistochemically, the neoplastic cells displayed strong positivity for PTH and remained negative for the TTF-1 (Figs. 3, 4), whereas the surrounding thyroid gland was positive for TTF-1 and negative for PTH (Figs. 3, 4). Immunostaining either with Ki-67 or with PCNA varied between the two different areas of the tumor (Figs. 5, 6): the pleomorphic nodules showed a strong positivity for both markers with a proliferative index of 8.5% for Ki-67 and of 44.9% for PCNA, whereas only few scattered positive Ki-67 cells were detected in the nonpleomorphic area (PI 1%). In the latter, some more positive cells were seen with the PCNA immunoreaction (PI 2.4%). Based on the morphological and immunohistochemical findings, the diagnosis of parathyroid gland carcinoma was made.
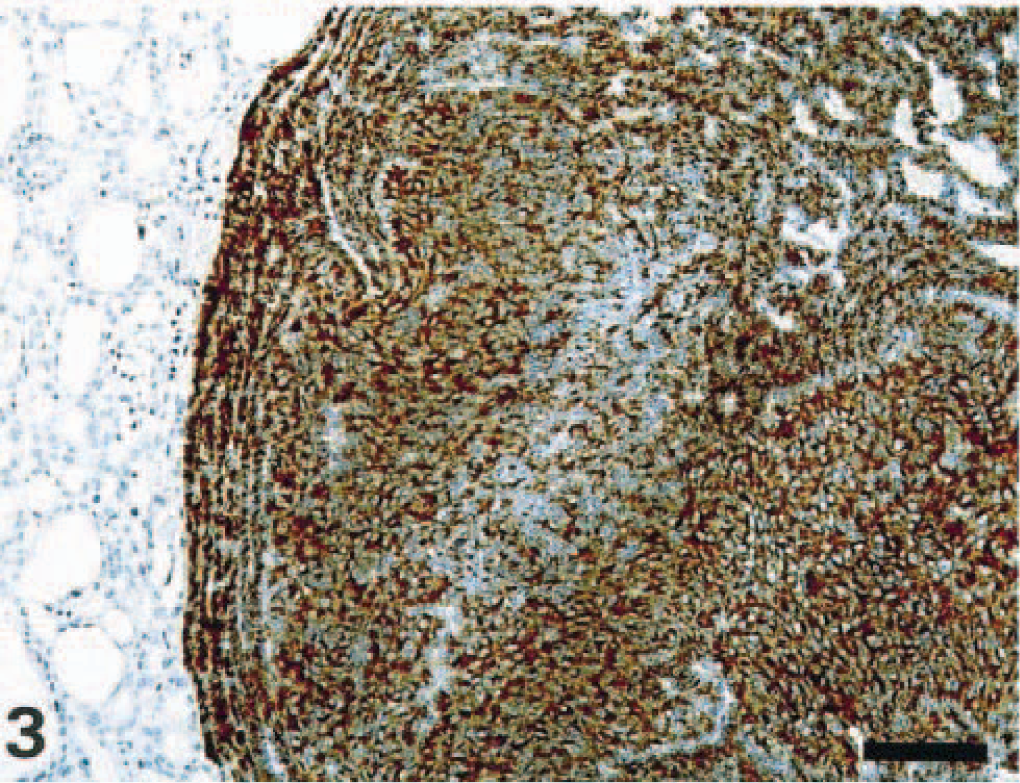
Parathyroid gland carcinoma; Wistar rat. Anti-PTH immunostaining. All neoplastic cells express intense PTH immunoreactivity, whereas the thyroid gland remains PTH negative (left). StreptABC/HRP method for PTH, hematoxylin counterstain. Bar = 100 μm.
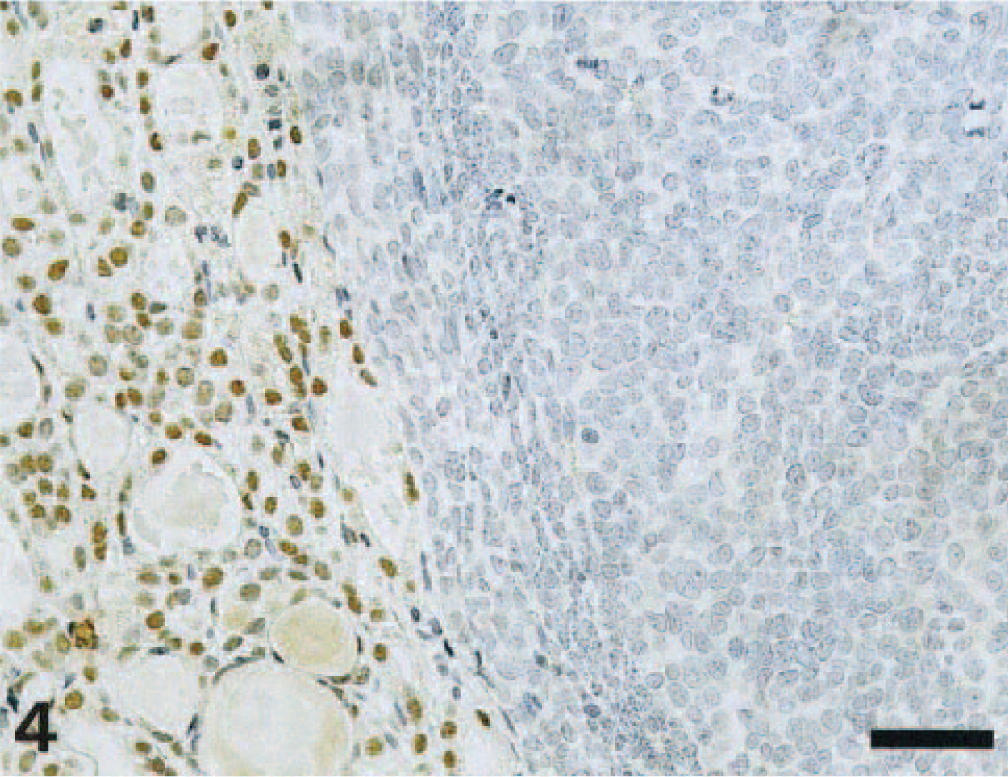
Parathyroid gland carcinoma; Wistar rat. Anti–TTF-1 immunostaining. Nuclei of the thyroid follicular cells are positive (left), whereas the parathyroid neoplastic cells are negative. StreptABC/HRP method for TTF-1, hematoxylin counterstain. Bar = 50 μm.
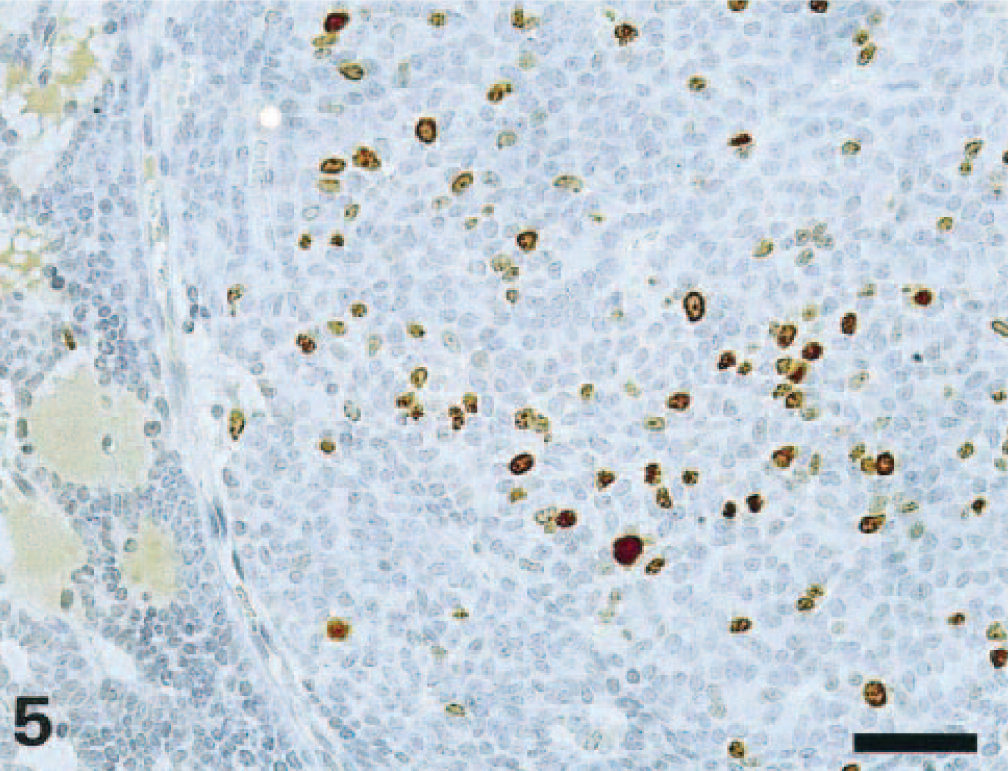
Parathyroid gland carcinoma; Wistar rat. Numerous neoplastic cells, mainly in the solid area, express Ki-67 immunoreactivity. StreptABC/HRP method for Ki-67, hematoxylin counterstain. Bar = 50 μm.
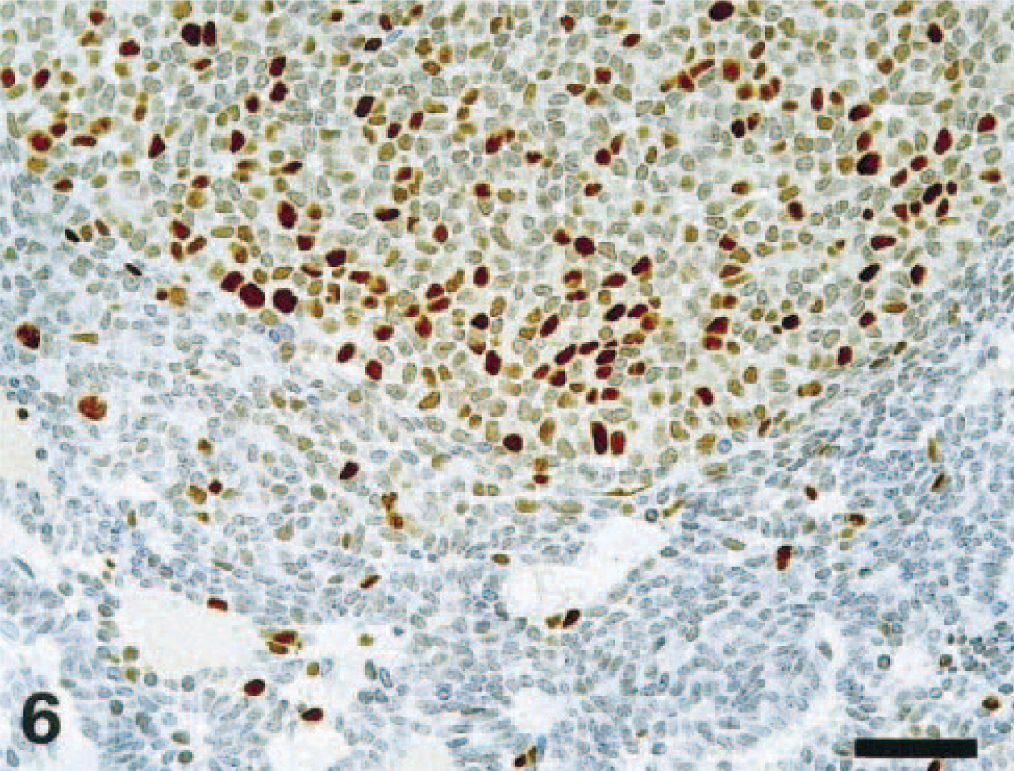
Parathyroid gland carcinoma; Wistar rat. A large number of neoplastic cells express PCNA immunoreactivity. The number of PCNA-positive cells is much higher in the solid neoplastic area. StreptABC/HRP method for PCNA, hematoxylin counterstain. Bar = 50 μm.
Carcinomas of the parathyroid glands are very rare tumors in all animal species, including humans. 5 , 8 , 10 , 11 One carcinoma was reported in the dog 8 associated with hypercalcemia and local invasion. To our knowledge, there are only two reports on parathyroid carcinoma in rats. 10 , 14 In our case, although there were no distant metastases, all other criteria for malignancy used in humans were met: high number of mitoses with atypia, high Ki-67 and PCNA proliferative index (8.5% and 44.9% respectively), cellular and nuclear pleomorphism, and invasion of the capsule. 13 According to some authors, 3 , 13 high number of mitoses and Ki-67 antigen expression in more than 6% of the neoplastic cells are the most valuable criteria for distinguishing malignant from benign parathyroid tumors. According to these criteria, in our case, a diagnosis of parathyroid carcinoma, therefore, is appropriate. As a differential diagnosis, a tumor arising from the thyroid parenchyma had to be excluded. Immunostaining for the PTH and TTF-1 antigens revealed that the neoplasm was positive for PTH and negative for TTF-1, whereas thyroid gland was positive for TTF-1 and negative for PTH.
Regarding the assessment of the proliferative activity of endocrine tumors, previous studies have shown that Ki-67 as marker for cell proliferation is superior to PCNA. 1 , 7 , 9 , 12 So, it appears that high Ki-67 expression can be highly indicative of malignancy. The difference in expression of Ki-67 and PCNA in the present case was unexpected because both antigens are expressed during the same phases of the cell cycle. However, a review of the literature, in addition to our personal experience with other proliferative lesions, has indicated that PCNA expression in tumors is variable. 1 , 7 , 9 , 12 The main reason seems to be that PCNA can persist in the cells even after the mitotic phase throughout the resting phase (G0) and, therefore, may overestimate proliferative activity.
Parathyroid tumors can be either functional or nonfunctional depending on the ability of chief cells to secrete PTH. Excessive secretion of this hormone leads to hyperparathyroidism. In rats, diffusely hyperplastic parathyroid glands are observed in animals, principally in males, with progressive nephropathy that leads to renal loss of calcium and phosphorus, which in turn stimulate the continuous overproduction of PTH, resulting in diffuse hyperplasia of the gland. 6 A secondary mineralization due to overstimulation of parathyroid may be seen in several tissues, including kidneys, lung, gastrointestinal tract, and media of large arteries. Fibrous osteodystrophy may also occur in the most severe cases of progressive nephropathy.
The present case was probably a nonhyperfunctional tumor because no signs of hyperparathyroidism were observed. Blood biochemistry values were not investigated. In rats and mice, considering the rare occurrence of neoplasms of the parathyroid, biochemistry data regarding their functionality (PTH secretion) do not exist. In humans, it is reported that most of the parathyroid neoplasms are functional and associated with hyperparathyroidism.
Footnotes
Acknowledgements
We thank Mrs. Elisabeth Perentes and Christine Bresser for the immunohistochemical work and Mr. Philippe Scheubel for the photographic support.
