Abstract
Vasculitis is a hallmark lesion of the severe form of systemic porcine circovirus–associated disease (PCVAD). In 2 experimental studies with porcine circovirus type 2 serogroup b (PCV2b), 2 pigs developed fatal PCVAD with acute vasculitis, and 5 related pigs developed chronic lymphohistiocytic and plasmacytic peri- and endarteritis. Five of these pigs (1 with acute vasculitis and 4 with chronic vasculitis) had also been inoculated with bovine viral diarrhea virus type 1 (BVDV1) or BVDV1-like virus. Vascular lesions were similar, independent of whether pigs had been inoculated singly with PCV2b or dually with PCV2b and BVDV1 or BVDV1-like virus. The acute vasculitis was accompanied by marked pulmonary and mesenteric edema and pleural effusion. In situ hybridization demonstrated abundant intracytoplasmic porcine circovirus type 2 (PCV2) nucleic acid in endothelial, smooth muscle–like, and inflammatory cells within and around affected arteries. The pigs with lymphohistiocytic and plasmacytic vasculitis had lesions of systemic PCVAD, including multisystemic lymphoplasmacytic and histiocytic or granulomatous inflammation. PCV2 nucleic acid was detected in renal tubule epithelial cells, mononuclear inflammatory cells, and rare endothelial cells in noninflamed vessels in multiple tissues of these animals. The 2 pigs with acute vasculitis had no PCV2-specific antibodies (or a low titer of), whereas the pigs with lymphohistiocytic and plasmacytic vasculitis developed high antibody titers against this virus. These observations suggest that (1) acute vasculitis observed in the current studies is directly caused by PCV2b, (2) chronic vasculitis may in part be mediated by the subsequent immune response, and (3) host factors and viral strain may both contribute to vasculitis in animals infected with PCV2b.
Keywords
Vasculitis is an important component of a variety of infectious diseases in animals, but its pathogenesis is often unknown. 21 Two commonly proposed mechanisms are direct endothelial injury by the pathogen and damage caused by the immune reaction. Porcine circovirus–associated disease (PCVAD) is a group of disease manifestations caused by or associated with infection by porcine circovirus type 2 (PCV2). Systemic vasculitis is a hallmark lesion of one of the forms of PCVAD, porcine dermatitis and nephropathy syndrome (PDNS). 8,26,33 Currently, it is hypothesized that high PCV2 antibody titers may trigger the development of this vasculitis through the deposition of immune complexes, possibly in combination with a cell-mediated immune response. 37
Excluding PDNS cases, vasculitis was a rarely reported finding in association with PCV2 infection in the past; 2,25 however, it has been identified as a significant component of the severe form of PCVAD in North American outbreaks since the fall of 2004. 7,11,16 Severe vascular lesions have also recently been reported from Europe and South America in pigs naturally infected with PCV2. 6,9,32,35 Here we describe similar acute and chronic vascular lesions in pigs experimentally infected with PCV2 serogroup b (PCV2b), and we identify potential underlying pathogenic mechanisms.
Materials and Methods
Seven pigs from 2 experimental studies with PCV2b and ruminant pestivirus (I. Langohr, G. Stevenson, E. Nelson, S. Lenz, H. Wei, R. Pogranichniy, unpublished data, 2008) were selected for this post hoc study. Selection criteria were the presence of vascular lesions and gross and histologic lesions characteristic of systemic PCVAD. In the first study, 2 partially characterized virus preparations were inoculated into 24-day-old germ-free pigs either alone or with PCV2 serogroup a. These inocula were filtered tissue homogenates from diseased pigs known to contain either (1) PCV2b and a bovine viral diarrhea virus (BVDV) type 1–like virus or (2) the same pestivirus but not PCV2. A vaccination protocol against BVDV, which included a cytopathic strain of BVDV type 1 (cpBVDV-NADL) as a booster, was tested as a possible measure to prevent development of clinical disease. Animals were sacrificed at 10 and 21 or 22 days postinoculation (PI). In the second study, 27-day-old cesarean-derived/colostrum-deprived (CD/CD) conventionally reared pigs from 4 litters were inoculated with PCV2b and cpBVDV-NADL, either alone or in combination. Inoculation of cpBVDV-NADL preceded that of PCV2b by 4 days. A similar vaccination protocol was used. The study was terminated 5 weeks after inoculation of PCV2b. Both experimental protocols were approved by the Institutional Animal Care and Use Committee.
At necropsy, selected tissues were collected, fixed in 10% neutral buffered formalin for 36 to 48 hours, embedded in paraffin, sectioned at 4 µm, and stained with hematoxylin and eosin, Verhoeff–van Gieson for elastin fibers, phosphotungstic acid–hematoxylin for fibrin, and periodic acid–Schiff for fibrinoid deposits.
Two digoxigenin-labeled oligonucleotide probes (DIG-5′-AAGTTCAGCCAGCCCGCGG-3′ and DIG-5′-CATTCCAACGGGGTCTGATTGCTGG-3′) were designed for in situ hybridization based on highly conserved regions of the genome of PCV2 (nucleotide position Nos. 491-509 and 811-835, respectively). The technique was carried out using a manual capillary-action work station (MicroProbe, Fisher Scientific, Pittsburgh, PA) as previously described 12 but with modifications. Sections of lymph node and kidney of a pig naturally infected with PCV2 were used as positive control.
Immunohistochemical testing for one of the fragments of the cleaved third complement component (C3c) and for porcine immunoglobulins M and G (IgM and IgG) was performed on formalin-fixed, paraffin-embedded sections of tissues with vascular changes. The primary antibody used for C3c was a rabbit polyclonal antibody (ab15980, Abcam Inc., Cambridge, MA), diluted 1:200 and with an incubation at room temperature for 60 minutes. Antigen retrieval was performed by pretreatment with 0.05% proteinase K (Invitrogen, Carlsbad, CA) for 5 minutes. A labeled streptavidin-biotin detection system was used, and diaminobenzidine was used as chromagen. Immunohistochemistry for IgG and IgM was performed by Jan Shivers at the Minnesota Veterinary Diagnostic Laboratory, St. Paul, MN; sections of kidney from a pig with porcine dermatitis and nephropathy syndrome and sections of porcine tonsil, respectively, were used as positive controls.
Serum samples from sows collected before cesarean section and from germ-free pigs and conventionally reared CD/CD pigs at different time points during the studies were tested by a PCV2 enzyme-linked immunosorbent assay (ELISA) based on the recombinant open reading frame 2–capsid protein of PCV2. 22 Positive results were confirmed by indirect fluorescent antibody (IFA) test. 29 A virus neutralization assay for BVDV was performed in 96-well microtitration plates as previously described 23 but with some modification, using Madin-Darby bovine kidney (MDBK) cells as the indicator and a 1:4 starting dilution. Serum of sows and pigs was also tested by ELISA for the presence of specific antibodies to porcine parvovirus (PPV; PPV-Ab Svanovir, Svanova, Uppsala, Sweden), porcine reproductive and respiratory syndrome virus (HerdChek PRRS, IDEXX Laboratories Inc., Westbrook, MA), and Mycoplasma hyopneumoniae (HerdChek Mhyo, IDEXX Laboratories Inc., Westbrook, MA). Positive results for PPV by ELISA were confirmed by IFA.
Quantitative real-time polymerase chain reaction (PCR) for PCV2 and pestiviruses was performed on serum and multiple tissues using sequence-specific oligonucleotide primers as previously described. 14,27 Serum samples collected from the pigs were tested by quantitative real-time reverse transcriptase PCR (RT-PCR) for porcine reproductive and respiratory syndrome virus (PRRSV) using a commercially available one-step multiplex RT-PCR test (Tetracore Inc., Rockville, MD) that can detect both North American and European strains of the virus. 36 In addition, the serum was tested for PPV by gel-based PCR as previously described. 4 Each reaction included negative and positive controls with, respectively, no added template nucleic acid and one or more samples known to contain the virus that was to be detected. In the real-time assays, progressive dilutions of a known copy number of viral genomic nucleic acid served to generate a standard curve. A real-time PCR was considered positive if the cycle threshold level was obtained at ≤ 40 cycles.
Pooled samples of lymph nodes and tonsil from each pig included in this study were processed for virus isolation. Porcine kidney–15 cells that were free of porcine circovirus and to which D-glucosamine was later added were used for isolation of PCV2. Virus growth was confirmed by immunofluorescence microscopy as previously described. 29 For isolation of BVDV, the tissue homogenate from pigs inoculated with cpBVDV-NADL (pig Nos. 4, 5, 6, 7) was inoculated into a suspension of MDBK cells. Cells were evaluated for cytopathic effect as an indication of virus growth, which was confirmed by IFA using anti-BVDV polyclonal antiserum conjugated with fluorescein-isothiocyanate (VMRD Inc., Pullman, WA).
Results
Table 1 lists the clinical and principal gross and histologic lesions in the animals selected for this study. A single pig of the germ-free study, from the group vaccinated against BVDV and inoculated with PCV2b and BVDV type 1–like virus (pig No. 1), had acute respiratory distress followed by death at 21 days PI. In the conventional study, clinical signs of PCVAD were observed in a single animal, inoculated only with PCV2b (pig No. 2). This pig had growth retardation, rough hair coat, generalized pallor, and mild lethargy over 12 days, followed by acute respiratory distress and death 30 days PI. Lesions in both animals resembled the severe form of systemic PCVAD recently observed in the field, characterized by pulmonary interstitial edema histologically associated with multisystemic acute arteritis. Pig No. 1 also had marked mesocolonic edema and mild chyloabdomen associated with prominent mesenteric lymphatics with a beaded appearance, whereas pig No. 2 had additionally marked hydrothorax. Five other PCV2b-inoculated CD/CD pigs (Nos. 3 to 7) did not have clinical signs related to the inoculated viruses. Lesions in these 5 animals were restricted to multisystemic lymphoplasmacytic and histiocytic or granulomatous inflammation typical of the classic form of systemic PCVAD accompanied by chronic vascular inflammatory lesions. All pigs that developed vasculitis in the second study were the offspring of 2 Landrace sows with the same dam.
Clinical Signs and Postmortem Lesions of 7 Pigs Experimentally Inoculated With Porcine Circovirus Type 2 Serogroup B a
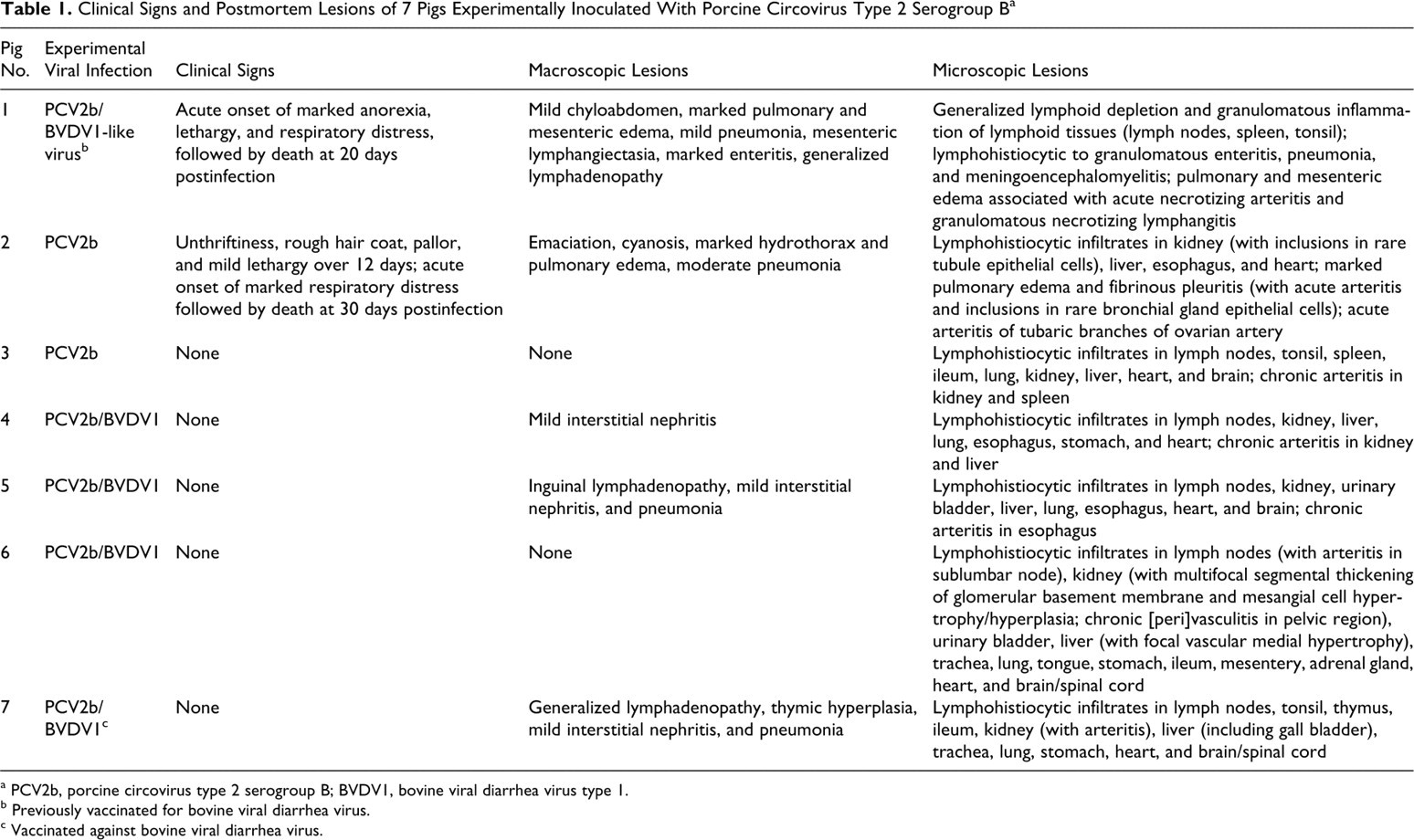
a PCV2b, porcine circovirus type 2 serogroup B; BVDV1, bovine viral diarrhea virus type 1.
b Previously vaccinated for bovine viral diarrhea virus.
c Vaccinated against bovine viral diarrhea virus.
In pigs Nos. 1 and 2, many pulmonary arterioles and small- and medium-sized arteries had degeneration of endothelial cells, intramural and perivascular edema and hemorrhage, fibrinoid necrosis of the vascular wall, and thrombi (Fig. 1). In tubaric branches of the ovarian arteries of pig No. 2, the tunica intima and, to a lesser extent, the tunica media were expanded by acute edema and hemorrhage, superimposed on intimal spindle cell proliferation (Fig. 2). In some cross sections of this artery, the proliferated spindle cells delimited multiple small vascular channels. Inflammatory cell infiltrates within and around affected vessels were minimal. Similar lesions were observed in few small arteries in the mesentery, small intestine, and brain of pig No. 1.
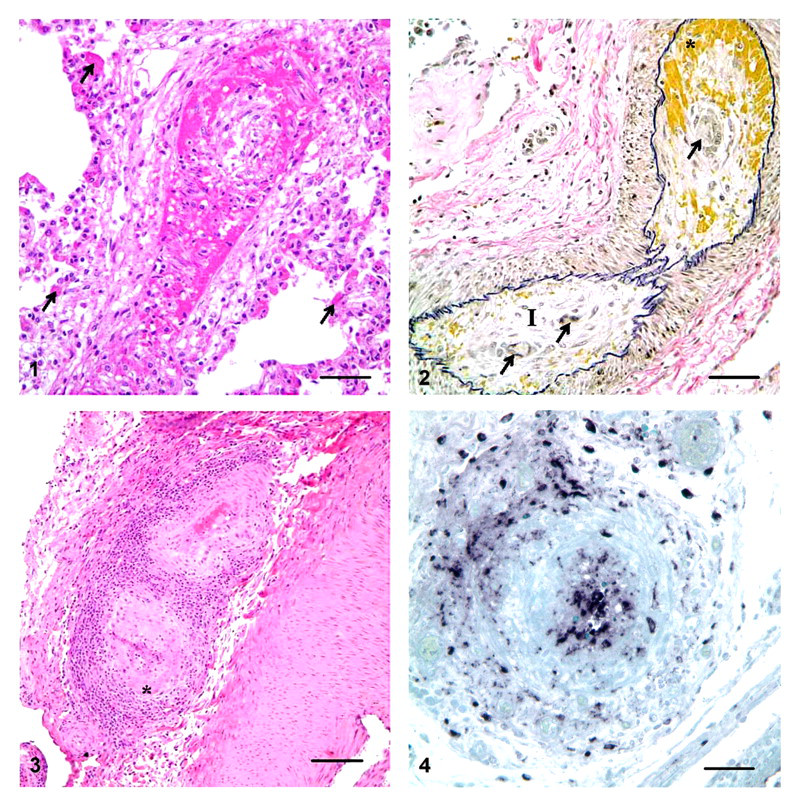
Granulomatous and necrotizing lymphangitis was a prominent additional finding in pig No. 1. Histologically, macrophages admixed with karyorrhectic debris effaced the wall of mesenteric and pulmonary lymphatics. The mesenteric lymphatics were markedly distended by fluid between the valves, which accounted for their beaded appearance observed grossly.
In pig Nos. 3 to 7, affected blood vessels had marked endothelial cell swelling, with multifocal disruption of the intima and expansion of the subendothelial layer by edema. Lymphocytes, macrophages, and plasma cells surrounded and variably infiltrated the vascular wall. Some of these vessels had segmental areas of medial necrosis and replacement by fibrinoid deposits (Fig. 3). Others had hydropic degeneration of the smooth muscle cells. The internal elastic membrane was intact. In general, the vascular lesions affected small- and medium-sized muscular arteries predominantly in the kidney (at the corticomedullary junction and the medullary crest) and, less commonly, in the spleen, liver, esophagus, and lymph nodes.
Large amounts of intralesional PCV2 nucleic acid were detected in both the nucleus and the cytoplasm of endothelial cells, smooth muscle–like cells, and inflammatory cells within and around acutely inflamed blood and lymph vessels of the 2 pigs that exhibited clinical disease (Fig. 4). No viral nucleic acid was detected within the chronically inflamed vessels of the other 5 animals; however, renal tubule epithelial cells, mononuclear inflammatory cells elsewhere, and rare endothelial cells of noninflamed vessels in multiple tissues from these animals had positive labeling for PCV2.
Deposition of the cleaved C3c and the IgM and IgG was not detected within the wall of the inflamed vessels in any of the animals, whereas the positive control tissue had appropriate staining.
Pig No. 1 (acute vasculitis) had a low antibody titer (1:80) to PCV2, which was first detected at 21 days PI. Pig No. 2 (acute vasculitis) was seronegative for PCV2 at 4 and 14 days PI. No serum sample was available from this animal thereafter. Samples of thoracic and lung fluid, collected at 30 days PI, were tested instead, but antibodies to PCV2 were not detected. In pigs with chronic vascular lesions (pig Nos. 3 to 7), antibodies to PCV2 were first detected at 35 days PI. The antibody titer varied between 1:320 and 1:1280. Of the pigs inoculated with BVDV type 1–like virus (pig No. 1) and the cytopathic strain of BVDV type 1 (cpBVDV-NADL) (pig Nos. 4 to 7), only pig No. 1, which had been vaccinated against this virus, had antibodies to BVDV at the day of inoculation (day 0). In pigs No. 5 and Nos. 4 and 6, neutralizing antibodies to cpBVDV-NADL were first detected at 18 and 39 days PI, respectively. Pig No. 7 did not develop any neutralizing antibody to this virus. Pig No. 6 seroconverted against PPV at day 18 PI. At the termination of the study, the other 2 animals of the same group (pig Nos. 4 and 5) had also seroconverted to this virus. All pigs tested negative for PRRSV and M. hyopneumoniae–specific antibodies.
PCV2 DNA was detected in serum and tissues of all pigs. Viremia for PCV2 peaked at 14 days PI in pig Nos. 4, 5, and 6 but was still increasing at the termination of the study in pig Nos. 1, 2, 3, and 7. Serum and tissue homogenates were positive by quantitative real-time PCR for BVDV in pig Nos. 1, 4, 5, and 6. Viremia for this virus was transient in all pigs: It peaked at 8 days PI (pig Nos. 4–6) or 10 days PI (pig No. 1) and disappeared by the end of the studies. Pigs Nos. 2, 3, and 7 were negative for BVDV by PCR on serum and tissues. PCR for PPV was positive on serum of pig No. 4 at 35 days PI. All pigs tested negative for PRRSV nucleic acid.
PCV2 was isolated from tissue homogenate prepared from pooled samples of lymph nodes and tonsil of all pigs included in this study. The isolation test for BVDV was negative.
Discussion
Acute necrotizing arteritis accompanied by marked pulmonary and mesenteric edema and hydrothorax was seen at 21 and 30 days PI (PCV2b) in 2 pigs included in this study (1 of each experiment). Five other related animals infected with the same viral strain and necropsied 35 days PI developed classic microscopic lesions of PCVAD accompanied by lymphohistiocytic and plasmacytic periarteritis and endarteritis. The former animals presented clinical signs of PCVAD, whereas the latter remained healthy until the termination of the study.
The two most common mechanisms responsible for the development of vasculitis are direct injury by infectious agents and immune-mediated inflammation. 19 In pigs, vasculitis can be caused by PRRSV, African swine fever virus, classical swine fever virus, BVDV, 17 ovine herpesvirus 2 (sheep-associated form of malignant catarrhal fever), 1,18,34 pseudorabies virus, 21 and bacteria capable of causing multisystemic infections, such as Salmonella choleraesuis, Streptococcus suis, and Erysipelothrix rhusiopathiae. 19 Additional differential diagnoses for necrotizing vasculopathy in pigs include edema disease and cerebrospinal angiopathy, caused by shiga-like toxins produced by Escherichia coli, and vitamin E and selenium deficiency (mulberry heart disease), for which the vascular alterations can be a useful indicator. 19
Neither African nor classical swine fever occur in the USA. Pseudorabies was declared eradicated from commercial swine herds in the USA in 2004. 28 PCR and serologic testing for PRRSV were negative in all animals of the current studies. Tissue homogenates tested positive for BVDV by PCR in most animals experimentally inoculated with this virus; however, vascular lesions were also seen in animals that lacked infection with this virus. In addition, arteritis was morphologically similar irrespective of whether pigs were infected singly with PCV2b or dually with PCV2b and BVDV type 1 or BVDV type 1–like virus. Multisystemic lesions in the animals of this study did not indicate infection by any of the above-listed bacteria. In edema disease, epidemiologic aspects (disease most commonly seen 5 to 14 days postweaning) and preferential distribution of vascular lesions (gastric and colonic submucosa and cerebellar folia) 19 are different from the lesions observed in these studies. Liver selenium and vitamin E concentrations were not tested in these cases; however, in cases of mulberry heart disease, vascular lesions occur preferentially in the heart. 19 In the current studies, cardiac vasculature was not affected by the systemic vasculitis.
PCV2b infection was the only common feature among all the affected animals in our studies. In the pigs with acute arteritis, abundant PCV2 nucleic acid was detected within the lesions. Similar lesions and PCV2 nucleic acid or antigen distribution were recently reported in the brain and lung of pigs naturally infected by PCV2, often in the absence of any other major porcine pathogens. 9,32,35 The reported vascular lesions were associated with viral antigen in endothelial cells, smooth muscle cells, and infiltrating inflammatory cells in lymph and blood vessels. This suggests that the acute vasculitis in PCV2-infected animals is directly caused by the virus.
In the pigs with chronic arteritis, no viral nucleic acid was observed in lesioned vessels; however, PCV2 was detected within scattered endothelial cells in nonaffected vessels and within mononuclear cells that composed the inflammatory infiltrate in multiple organs of these animals. Multisystemic lymphohistiocytic and plasmacytic periarteritis and endarteritis in these pigs resembled those previously reported in some pigs naturally infected with PCV2. 2,25 In some of the reported cases, PCV2 antigen was demonstrated by immunohistochemistry in endothelial cells, smooth muscle–like cells, and inflammatory cells located mainly in the intima and media of affected arteries. These findings suggest that the chronic vasculitis may, at least in part, be mediated by the host immune response that follows the viral infection.
The pigs that died naturally had evidence of impaired immune response as indicated by marked lymphoid depletion and absent or low antibody titers to PCV2. Large amounts of PCV2 nucleic acid in multiple tissues, including in foci of arteritis, suggest that unchecked replication of the virus at a high level was the likely determinant of the progression from subclinical PCV2 infection to fatal disease in these animals. In the other animals, the presence of a high antibody titer to PCV2 suggests that these animals were able to mount an immune response that cleared infection of the virus in many tissues (including the vascular wall) and inhibited manifestation of clinical disease. Reduced amount of PCV2 nucleic acid within inflammatory lesions typical of those described for PCVAD is consistent with resolving lesions. 26
The exact pathogenesis of the vascular lesions in PCV2-infected pigs remains to be established. The mechanism by which viruses trigger vasculitis can be divided into two basic categories: (1) direct damage by invasion of endothelial cells and (2) indirect damage through immune mechanisms in genetically susceptible individuals with some level of immune dysregulation. 10 Many viruses are endotheliotropic, including those that cause classical swine fever, bovine viral diarrhea, and infectious canine hepatitis. 19 There is strong evidence that PCV2 is similarly capable of infecting endothelial cells and directly inducing vascular damage. Vasculitis may also result from local immune reactions. Virus-infected endothelial cells can express major histocompatibility complex class II molecules and secrete cytokines that recruit T cells and macrophages, leading to vasculitis with lymphocytic predominance such as that seen in malignant catarrhal fever and border disease. 19
In addition to direct invasion and damage by the virus or subsequent cell-mediated injury to the vessel wall (type IV hypersensitivity reaction), another mechanism for viral infection–associated vasculitis is humoral-mediated injury. Type III hypersensitivity reaction caused by the deposition of immune complexes in vessel walls, possibly in association with PCV2 infection, has been discussed as the underlying mechanism of PDNS. 8,26,33 The presence of immunoglobulins or complement in and around affected vessels is often used as an argument for an underlying immune complex pathogenesis, although these immune reactants may be nonspecifically entrapped in already compromised vessels. 19 In the pigs of this study, no immunoglobulins (IgM and IgG) or complement (C3c) was found within affected vessels. This, however, does not rule out immune complex vasculitis, because immune reactants can reportedly disappear rapidly. 19
Three of the pigs with chronic vasculitis in this study had seroconverted to PPV, a known cofactor of PCVAD. 3 Hydrothorax, pulmonary interstitial edema, and mesocolonic edema were previously observed in pigs experimentally infected with both PCV2 and PPV. 3,15 In only one of these studies was segmental vasculitis reported and this in tissues of the dual-inoculated pigs, where numerous cytoplasmic inclusions and positive labeling for PCV2 were observed in smooth muscle and endothelial cells of affected vessels. 15 These authors discussed the possibility of E. coli infection as the underlying cause for the edema of the mesocolon and other tissues, but they did not rule out the possibility that the lesions resulted from direct PCV2-induced vascular damage. Lesions induced by PPV alone have been reported only in pregnant sows and their fetuses. There is no evidence that PPV causes lesions in growing pigs—particularly, vasculitis. 20
In most of the reported cases with vasculitis in naturally and experimentally PCV2-infected pigs, vascular lesions predominated in the kidney (lobar, interlobar, and arcuate arteries), 2,25,32 mesocolon (mesenteric vessels and lymph node), 13,15,26,32 lung, 25,35 heart, 25 lymphoid tissues, 15,25 brain, 6,9,25,32 and intestinal submucosa. 15 This tissue distribution correlates with that of inflammatory lesions seen in systemic PCVAD. 26,31 Thus, it is possible that multiplication of the virus in endothelial cells of blood and lymph vessels provides a mechanism for viral dissemination. It may also account for the prolonged viremia observed in some animals infected with PCV2. 5,30
Interestingly, all pigs with vasculitis in the second study were the offspring of 2 related Landrace sows, suggesting a genetic predisposition to the development of the vascular lesions. Genetic variations influencing the immune response might determine the intensity of the systemic inflammatory response to the viral infection and, consequently, the development and severity of disease. This is consistent with previously reported evidence that host genetics influence the susceptibility of animals to PCV2-associated lesions. 24 It is possible that many animals experience transient vascular damage early in the infection with PCV2 but recover after mounting an immune response against the virus. Presence of adequate immunity against PCV2 possibly leads to clearance of the virus from the infected vascular wall, followed by uneventful recovery of any viral-induced damage. Some pigs, owing to as-yet-unknown factors, develop chronic inflammation with long-lasting vascular injury, as seen in pig Nos. 3 to 7 in this study. These vascular lesions may represent a persistent cell-mediated immune reaction following viral infection of vascular endothelium and smooth muscle cells. PCV2 may be cleared, or the quantity may be below detection limits in such lesions. Finally, those animals with inadequate immune response against the virus, such as pig Nos. 1 and 2 described herein, likely allow unchecked viral replication leading to extensive tissue damage, clinical disease, and death. The occurrence of vasculitis in pigs experimentally infected with PCV2b further indicates the importance of the vascular system in the pathogenesis of PCV2-associated diseases in swine.
Footnotes
Acknowledgement
Special thanks go to Jan Shivers (Minnesota Veterinary Diagnostic Laboratory, St. Paul, MN) for performing the immunohistochemistry assays for porcine immunoglobulins G and M.
The authors declared that they had no conflicts of interests with respect to their authorship or the publication of this article.
The animal experiments from which the pigs of this study were selected were funded by the National Pork Board.
