Abstract
Porcine circovirus type 2 (PCV2), a small single-stranded DNA virus, was initially discovered in 1998 and is highly prevalent in the domestic pig population. Disease manifestations associated with PCV2 include postweaning multisystemic wasting disease (PMWS), enteric disease, respiratory disease, porcine dermatitis and nephropathy syndrome (PDNS), and reproductive failure. Although these clinical manifestations involve different organ systems, there is considerable overlap in clinical expression of disease and presence of lesions between pigs and within herds. It is now widely accepted that PCV2 can be further subdivided into different types, of which PCV2a and PCV2b are present worldwide and of greatest importance. This review will focus on PCV2-associated lesions in different organ systems.
Porcine circovirus (PCV) type 2 (PCV2) is a small, non-enveloped virus with a circular-arranged, single-stranded DNA genome 115 that was first described in 1998.1,5 Subsequent retrospective investigations were able to trace PCV2 DNA and antigen back to 1962, 40 but PCV2 has likely been present in the swine population for much longer. Interestingly, recognizable disease outbreaks linked with PCV2 first appeared in the early 1990s and occurred almost simultaneously in France, 59 the USA (Daft B, Nordhausen RW, Latimer KS, et al. Interstitial pneumonia and lymphadenopathy associated with circoviral infection in a six-week-old pig. Proc Am Assoc Vet Lab Diagn 39:32, 1996, Arkansas, USA) and Canada. 30 In subsequent years, PCV2-associated disease became one of the most important and economically significant swine diseases. 75 Development and widespread global usage of PCV2 vaccination since 2006 75 has been remarkably effective in controlling and preventing PCVAD. Although PCV2 vaccination is used in the majority of pigs that enter the market today, PCV2 still remains an important differential diagnosis for various disease manifestations in pigs. As the prevalence of disease directly associated with PCV2 is markedly reduced, it seems even more important to recognize lesions attributable to PCV2 since detection methods to demonstrate PCV2 in lesions such as immunohistochemistry (IHC) either are not used on a routine basis anymore or have been replaced with molecular methods. The overall objective of this review is to summarize the diversity of PCV2-associated lesions in pigs and to compare and contrast lesions and sensitivity and specificity of detection methods (in situ hybridization and immunohistochemistry) in pigs infected with different PCV2 subtypes.
PCV2-Associated Clinical Manifestations
Clinical symptoms attributable to PCV2 have been summarized as PCV disease (PCVD) 103 or PCV-associated disease (PCVAD) 75 depending on geographic origin. For the purpose of this review, PCVAD will be used. The clinical features of PCVAD have recently been described in detail. 102 In brief, the very first recognized clinical PCVAD manifestation was postweaning multisystemic wasting syndrome (PMWS), 32 which resembles, as the name implies, a systemic infection involving several organ systems. Typically, pigs have enlarged lymph nodes and show decreased weight gain or wasting combined with many other nonspecific clinical signs such as dyspnea, diarrhea, pallor, and jaundice. 31 Other clinical signs include coughing, fever, central nervous signs, and sudden death. 31 As this disease manifestation is not only seen in pigs right after weaning but can also affect mature pigs,71,73 systemic PCVAD is perhaps a better term for it.
Respiratory disease is another frequently encountered PCVAD manifestation, often as part of the porcine respiratory disease complex also known as PRDC.33,49 PRDC is a multifactorial disease that typically affects pigs around 12 to 24 weeks of age. Clinically, affected pigs show signs of respiratory disease with fever and varying degrees of sneezing, coughing, nasal discharge, and respiratory distress as well as reduced weight gain (Halbur PG. Porcine respiratory disease. Proc Int Pig Vet Soc Congr 15:1-10, 1998, Birmingham, England).
PCVAD may also manifest as enteric disease that mimics ileitis caused by Lawsonia intracellularis or enteric salmonellosis.41,72 PCV2-associated enteritis is typically observed in grow-finish pigs.
Porcine dermatitis and nephropathy syndrome (PDNS) has been associated with PCV2 94 and is often observed in herds without an active PCV2 vaccination program; however, it has been determined that PDNS can be reproduced with other pathogens independent of the presence of PCV2. 53 PDNS is characterized clinically by acute onset of skin lesions (raised purple skin lesions progressing to multifocal raised red scabs with black centers most prominent on the rear legs), fever, and lethargy, and is commonly fatal.
Finally, reproductive failure is perhaps the least important PCVAD manifestation as this is typically only seen in individual dams 60 and is therefore often not of great economic importance. PCV2-associated reproductive failure was first reported in 1999 in Canada, 121 followed by reports in a number of other countries.55,67 Consistent signs on affected farms include increased numbers of abortions, stillbirths, fetal mummification, and increased pre-weaning mortalities.
PCV2-Associated Lesions in Different Organ Systems
All of the clinical manifestations of PCVAD described in the previous section affect one or more organ systems with substantial overlap. An accurate diagnosis often depends on submission of a complete tissue set. For example, if only lung from a pig with pneumonia and wasting is submitted, the diagnostic evaluation can only determine if PCV2-associated respiratory disease is present in this animal, but not whether the viral disease in this animal is only respiratory or actually systemic (Table 1). In experimentally infected pigs, the peak of viral replication in most tissues, independent of clinical outcome, appears to be around 14 days after exposure, 124 which corresponds also to the peak levels of PCV2 DNA in serum.77,87 The following provides a detailed description of PCV2-associated lesions that have been reported for different organ systems (Fig. 1).
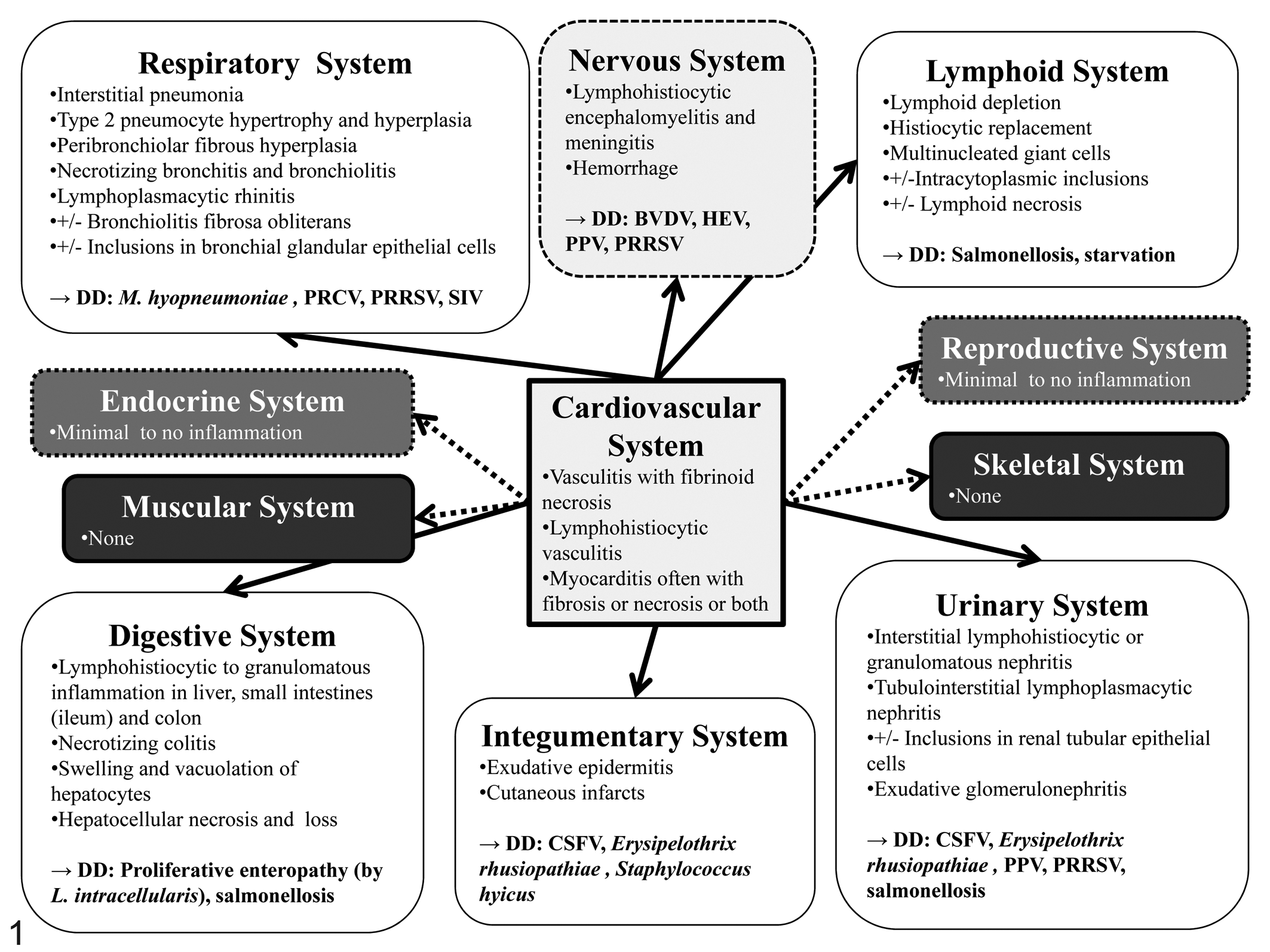
Microscopic lesions commonly encountered with porcine circovirus type 2 infection in different organ systems. Cardiovascular changes can be observed to varying degrees (indicated by regular or dashed arrows) in essentially all other organ systems. DD, differential diagnosis; CSFV, classical swine fever virus; BVDV, bovine viral diarrhea virus; HEV, hemagglutinating encephalomyelitis virus; L. intracellularis, Lawsonia intracellularis; M. hyopneumoniae, Mycoplasma hyopneumoniae; PPV, porcine parvovirus; PRCV, porcine respiratory coronavirus; PRRSV, porcine reproductive and respiratory syndrome virus; SIV, swine influenza virus.
Association of Clinical PCVAD Manifestations and Lesions in Different Organ Systems.
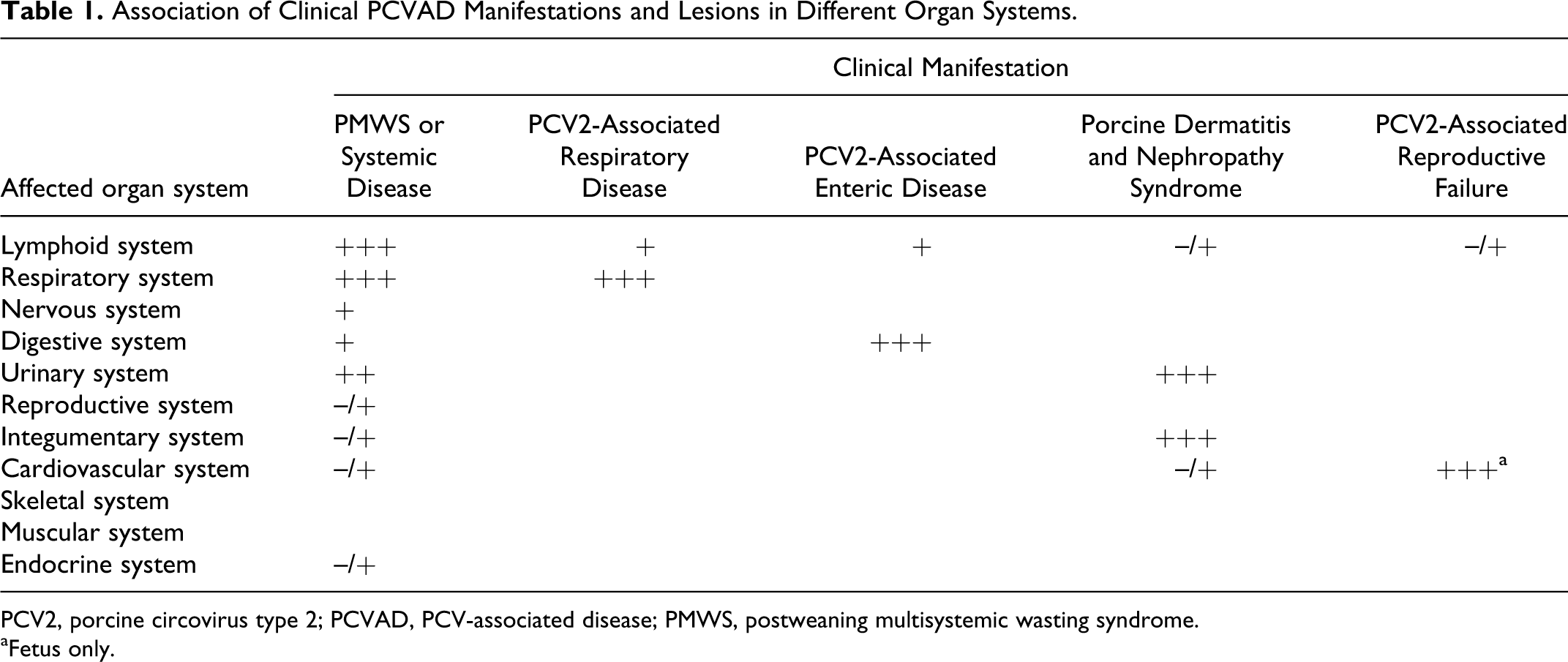
PCV2, porcine circovirus type 2; PCVAD, PCV-associated disease; PMWS, postweaning multisystemic wasting syndrome.
aFetus only.
Lymphoid System
Lesions in the lymphoid system are commonly observed in the tonsil, spleen, Peyer’s patches, and lymph nodes. In some cases, pigs with PCVAD may have all lymphoid tissues affected while in other cases only individual lymph nodes may be affected, so it is important that a full set of lymphoid tissues is examined. In lymphoid tissues, PCV2 induces very characteristic microscopic lesions that are essentially pathognomonic and include lymphoid depletion, histiocytic replacement of follicles 108 (Fig. 2), and infiltration of parafollicular areas by histiocytes; however, PCV2-associated lymphoid depletion is not necessarily correlated with expression of clinical disease and can also be observed in subclinically infected pigs. 91 It has been determined that the lymphocellular depletion affects both lymphoid follicles and parafollicular zones. 94 Mild to severe granulomatous inflammation in lymphoid tissues is also commonly observed (Fig. 2).2,4,108 Multinucleated giant cells can be seen frequently in all lymphoid tissues, including Peyer’s patches in the ileum. 94 Macrophages in affected lymphoid tissues may contain sharply demarcated, spherical, basophilic cytoplasmic inclusion bodies (Clark EG. Post-weaning multisystemic wasting syndrome. Proc Am Assoc Swine Pract 28:499-501, 1997, Quebec, Canada). 94 The inclusions are either large and single or smaller and multiple with groups of up to 12 inclusions. 94 Interestingly, these inclusion bodies are much less commonly seen today than when the disease first emerged (P. G. Halbur, personal communication). The alterations in the lymphoid tissues can be of varying severity depending on the state of infection. Different lymphoid lesion grading systems have been described (Fig. 2).69,98 In pigs experimentally infected with PCV2, lymphoid depletion typically can be seen between 14 and 21 days after infection and starts to resolve at 35 days post infection.37,68,79
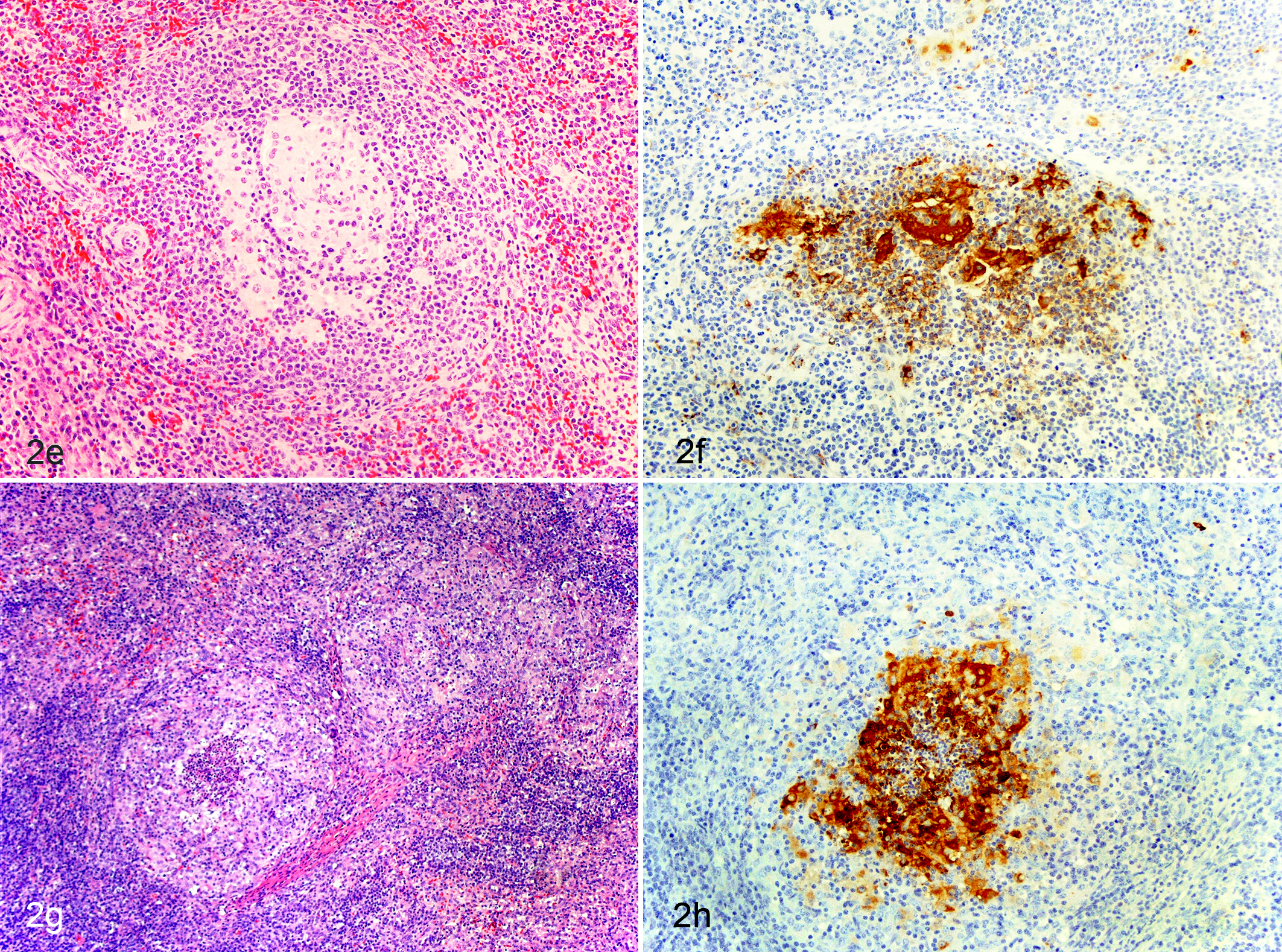
Pig experimentally inoculated with porcine circovirus type 2 (PCV2), lymph node. Different stages of lymphoid depletion and histiocytic replacement associated with varying amounts of PCV2 antigen.
Studies on pigs with naturally acquired disease revealed that absence of follicles and depletion of lymphocytes were associated with a reduction in the number of interfollicular dendritic cells and interdigitating cells, as well as a reduction or absence of B cells and CD4+ T lymphocytes. 98 Through immunohistochemical assays, increased numbers of macrophages and partial loss and redistribution of antigen-presenting cells were identified in the lymphoid tissues of the affected pigs when compared to healthy control pigs. 15 In particular, reduction or loss of CD79α and CD45RA-positive cells and diminution in T-cell areas of CD3-positive lymphocytes was evident. Additionally, there was increase of subcapsular and peritrabecular lysozyme-positive macrophages and partial loss and redistribution of SLA-DQ-positive antigen-presenting cells throughout lymphoid tissues. 15
Occasionally, necrotizing lymphadenitis has also been associated with PCV2. 48 The main lesion in this case is characterized by necrosis in the center of prominent lymphoid follicles (Fig. 3) and PCV2 can be detected within the necrotic foci. 48 Though initially suspected, 48 apoptosis or programmed cell death was later found not to be involved in lymphocyte depletion in cases with PCV2-associated necrotizing lymphadenitis. 28
Microscopic lesions in lymphoid tissues of fetuses, if present, are very similar to those observed in growing pigs: Lymph nodal and splenic lymphocyte depletion with occasional multinucleated giant cells11,61,65 or lymph node follicular hyperplasia 10 have been described.
Respiratory System
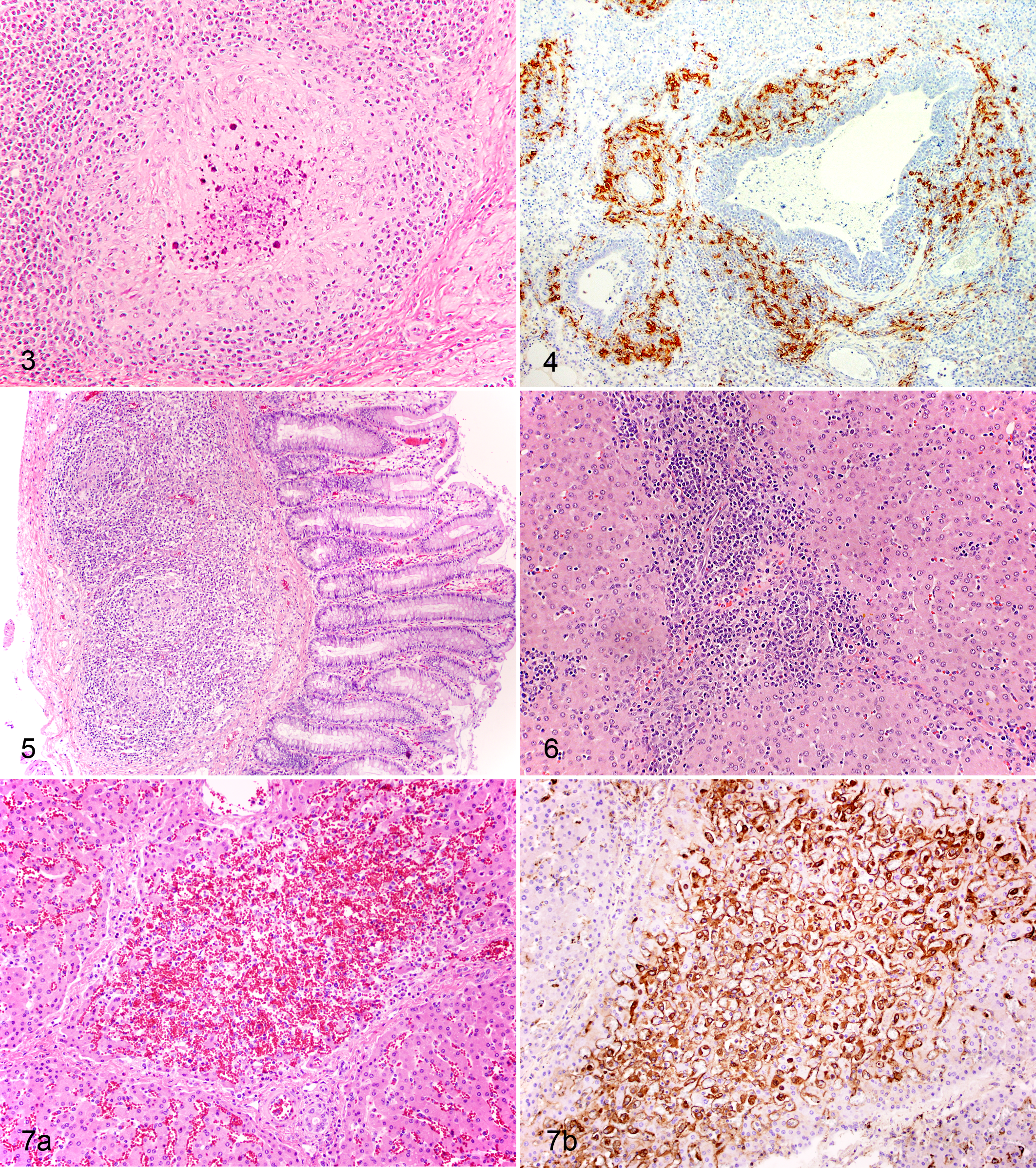
A characteristic microscopic lesion of PCV2 in the respiratory system is peribronchiolar fibrous hyperplasia, often associated with abundant amounts of PCV2 antigen (Fig. 4). Interstitial pneumonia with bronchiolitis was initially reported in association with PCV2 in field cases. 22 There can be partial to complete airway epithelial sloughing and mucosal or submucosal replacement by fibroplasia and diffuse lymphohistiocytic infiltration (Clark EG. Post-weaning multisystemic wasting syndrome. Proc Am Assoc Swine Pract 28:499-501, 1997, Quebec, Canada). Bronchointerstitial pneumonia was reproduced in conventional pigs inoculated with PCV2, 63 and mild multifocal interstitial pneumonia was seen in cesarean-derived colostrum-deprived (CDCD) pigs inoculated with PCV2. 9 Furthermore, mild interstitial pneumonia and lymphoplasmacytic rhinitis were induced in conventional pigs experimentally inoculated with PCV2. 96 Mild granulomatous bronchointerstitial pneumonia was reproduced in conventional pigs using an infectious DNA clone of PCV2. 23 Recently, it was reported that intracytoplasmic botryoid inclusion bodies that displayed PCV2-specific staining were observed in bronchial and bronchial glandular epithelial cells. 38 Mild nonsuppurative interstitial pneumonia51,82,88 or broncho-pneumonia,10,123 perhaps due to concurrent bacterial infection, have been reported in fetuses and newborn piglets infected with PCV2 in utero. PCV2 antigen was also associated with intraluminal polyps in bronchiolitis obliterans organizing pneumonia. 13 In addition, proliferation of type II pneumocytes and myofibroblasts, extracellular collagen matrix deposition, and fragmentation of elastic fibers were identified. 13 Proliferative and necrotizing pneumonia was diagnosed and associated with PCV2 antigen in Hungarian pigs. 113 Severe acute interstitial pneumonia characterized by the presence of abundant hyaline membrane in the alveoli and fibrin in the bronchioles was also described. 113
PCV2 has been associated with a variety of lesions in respiratory tissues, many of which can also be seen with other respiratory pathogens such as porcine reproductive and respiratory syndrome virus (PRRSV), swine influenza virus (SIV), porcine respiratory coronavirus (PRCV), and Mycoplasma hyopneumoniae or any combination thereof. In experimental infected pigs, microscopic lung lesions are rare with singular PCV2 infection.37,68,76 Under influence of concurrent M. hyopneumoniae infection, recognizable peribronchiolar hyperplasia is present between 21 and 35 days post infection. 79
Nervous System
PCV2 was initially associated with congenital tremors and PCV2 antigen was demonstrated in neurons or macrophages in newborn pigs in the Midwestern United States. 110 A follow-up study found no evidence of PCV2 infection in 40 pigs with congenital tremor from Spain, the United Kingdom, Ireland, and Sweden. 45 PCV2 antigen has been demonstrated in neurons, cerebral perivascular mononuclear cell infiltrates, fibroblast-like cells, and endothelial cells in the meninges, choroid plexus, cerebrum, and cerebellum from infected pigs.3,23,44,110 Meningoencephalitis and encephalitis have been linked to PCV2 infection too.23,94,111 In pigs experimentally infected with PCV2 and bovine viral diarrhea virus type 1 (BVDV1), mild to moderate lymphohistiocytic inflammation and hemorrhage were observed in the brain stem and cerebellum. 58 PCV2 antigen was identified by IHC stains in cerebellar lymphohistiocytic vasculitis combined with hemorrhage in 11.0% (50/456) or with lymphohistiocytic meningitis in 5.0% (23/456) of 3- to 5-month-old Brazilian pigs. 17 A portion of these pigs (15.1%; 69/456) had multiple hemorrhages in the cerebellar gray matter associated with lymphohistiocytic vasculitis and fibrinoid degeneration in vessels of the cerebellum and periventricular areas. The presence of PCV2 antigen was confirmed in the cytoplasm and nuclei of intralesional perivascular macrophages and endothelial-like cells. 17 Similar lesions (acute hemorrhages and edema of cerebellar meninges and parenchyma due to necrotizing vasculitis) were also identified in 16 nonrelated pigs that were part of a different study. 101 PCV2 DNA and antigen were demonstrated in endothelial cells in affected areas, and numerous apoptotic endothelial cells were also demonstrated in these lesions. 101 Recently, multifocal lymphohistiocytic to granulomatous meningoencephalomyelitis with giant cell formation was associated with PCV2 in slaughter-age pigs in Canada. 20
Neural lesions associated with PCV2 are seldom described and considered to be of minor importance. This may be due to true low incidence, lack of thorough microscopic examination, lack of using PCV2 detection methods on neural tissues, or a combination of any of these.
Digestive System
The most frequent PCV2-associated lesion in the digestive system is granulomatous enteritis, characterized by increased numbers of macrophages and scattered multinucleated giant cells expanding the mucosa and submucosa of the ileum (Fig. 5) and occasionally the colon and cecum. 50 These lesions have also been reproduced under experimental conditions. 72 Interestingly, in a field study involving 63 pigs carried out in Brazil, necrotizing colitis was the main lesion observed in 95.2% (60/63) of the pigs. 126 PCV2 antigen was identified in crypt epithelial lesions in these animals, accompanied by depletion of goblet cells. Concurrent infection with Salmonella sp. was identified in 38.1% (24/63) of the cases. In another study conducted in Spain, PCV2-associated colitis was seen in 3% (13/396) of the investigated cases. 105 Under experimental conditions, PCV2-associated granulomatous enteritis, although recognizable already by 14 days post infection, is more severe at 28 days post infection. 72
Gastric ulceration is commonly observed in PCVAD cases, 30 and PCV2 can often be demonstrated in the affected gastric mucosa. However, it is often unclear if PCV2 is initiating this lesion or is invading a developing lesion as an opportunist. Experimentally, gastric ulceration was observed in specific pathogen-free pigs infected with PCV2. 54
PCV2-associated hepatic lesions have been described as lymphohistiocytic infiltration in portal zones (Fig. 6) and single cell necrosis, cytoplasmic swelling and vacuolation, and karyomegaly of hepatocytes (Clark EG. Post-weaning multisystemic wasting syndrome. Proc Am Assoc Swine Pract 28:499-501, 1997, Quebec, Canada). In a field study investigating 100 livers from pigs with clinical PCVAD, PCV2 nucleic acid were detected in 70% of the samples by in situ hybridization (ISH) and associated with hepatocytes, Kupffer cells, and inflammatory infiltrates indicating hepatocytes as a target cell for PCV2 infection and replication. 93 PCV2-associated hepatitis has been reported in experimentally infected colostrum-deprived (CD) pigs co-infected with PCV2 and porcine parvovirus (PPV),3,44 and necrotizing hepatic lesions were reproduced in CDCD pigs inoculated singularly with PCV2 or dually with PCV2 and PRRSV. 34 Similarly, moderate to severe necrotizing and granulomatous hepatitis was observed in PCV2 and PCV2/PPV co-infected gnotobiotic piglets. 52 Acute hepatitis with centrilobular necrosis of hepatocytes (Fig. 7). was observed in 24-day-old CD pigs experimentally infected with a tissue homogenate from a clinically affected pig from the field. 36 Moderate to severe hepatic atrophy associated with nonsuppurative cholangiohepatitis was observed in experimentally PCV2-infected pigs. 54 In fetuses, hepatic lesions were described as congestion with hepatocellular loss 121 and nonsuppurative hepatitis with periacinar necrosis. 10
Lesions in the digestive system associated with PCV2 are important to recognize. While these lesions are often seen in connection with systemic infection, it is also possible that enteric lesions represent the only manifestation of PCV2 infection. Hepatic lesions have been observed mainly in gnotobiotic pigs and often contribute to death in this type of pig. Interestingly, lesions of similar severity are rarely seen in conventional pigs.
Urinary System
In an investigation involving 24 pigs naturally infected with PCV2, interstitial nephritis lesions were further characterized and PCV2 infection was associated with tubulointerstitial, lymphoplasmacytic nephritis (38.5%) (Fig. 8), interstitial granulomatous nephritis (45.8%) (Fig. 9), or both (20.8%). 99 Mild pyelitis and interstitial nephritis with focal lymphohistiocytic infiltrates can be seen in the deep medulla and pelvis of pigs experimentally infected with PCV2. 44 With concurrent PCV2 and PPV infection, tubular epithelial necrosis and regeneration and peritubular fibroplasia may be observed. 44 Intracytoplasmic botryoid inclusion bodies can rarely be observed in the renal tubular epithelium in pigs with systemic PCVAD. 38 Renal tubular epithelial necrosis with extensive interstitial edema and hemorrhage; lymphoplasmacytic, eosinophilic, and histiocytic tubulointerstitial nephritis; and numerous botryoid intracytoplasmic inclusions within the renal tubular epithelium and interstitial macrophages were identified in a pig with acute renal failure. These lesions were associated with PCV2 antigen by IHC and PCV2 virus particles by electron microscopy indicating a causative relationship. 39
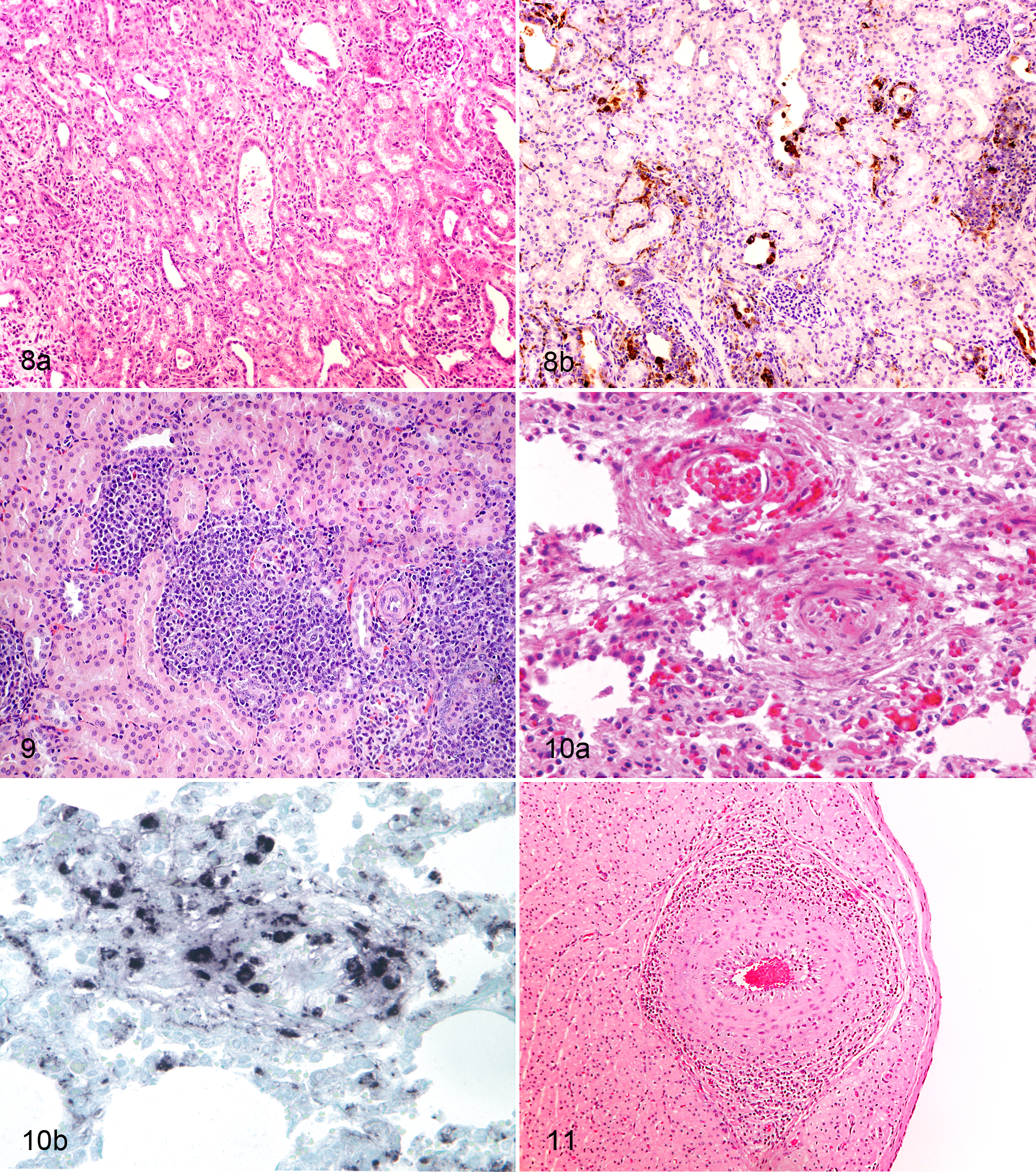
Renal lesions, if identified, often indicate chronicity of PCV2 infection and sometimes can be the only clue for a contribution of PCV2 to the disease in an affected pig. In experimentally infected pigs, renal lesions usually are absent by 21 days post infection but can be seen with increasing prevalence by 35 days post PCV2 infection. Interestingly, in studies terminated by 69 87 or 140 77 days post infection, renal lesions were not seen or had a low prevalence, indicating that resolution of this specific lesion eventually occurs.
Reproductive System
PCV2-associated microscopic lesions have not been reported in dams with PCV2-associated reproductive failure, and reproductive organs and placental sections are usually normal.61,92 When ovaries and uteri of 83 culled sows were investigated for PCV2 DNA and presence of endometritis, no association of PCV2 with endometritis was detected. 92 In a naturally infected boar, abundant PCV2 antigen and PCV2 nucleic acid were present in the parenchyma of the seminal vesicles; however, no remarkable inflammation was observed in the reproductive tract. 71 PCV2 DNA was detected in semen (47%; 8/17) and testicles (35.3%; 6/17) of healthy boars from six boar studs. 92 Nonetheless, PCV2-associated lesions in the male reproductive system can be considered as incidental and rare.
Integumentary System
PCV2 is not a primary cause of skin lesions but has been associated with exudative epidermitis and concurrent presence of Staphylococcus hyicus in a high health Swedish pig herd. 118 Severe, diffuse exudative epidermitis was seen in 42% (8/19) of CDCD pigs experimentally infected with PCV2, and the simultaneous presence of Staphylococcus hyicus was confirmed by bacterial culture. 34 The epidermitis lesions appeared 3 to 7 days post experimental PCV2 infection and progressed to a severe and diffuse form between 10 to 21 days. 34 In a different study, PCV2 was found in 8.5% (12/142) of exudative epidermitis cases, PPV was found in 11.3% (16/142), and both PCV2 and PPV were found in 42.3% (60/142) cases. 47 Abundant PCV2 antigen has been identified in sections of ears associated with ear tip necrosis obtained from clinically affected pigs in France (G. M. Allan, Am Assoc Swine Vet server email communication, October 21, 2011). Typically, however, PCV2 is not associated with inflammatory lesions of the skin. Most cutaneous lesions associated with PCV2 are due to necrotizing vasculitis and are described in the cardiovascular system section.
Cardiovascular System
Severe interlobular pulmonary edema associated with arteritis (Fig. 10) and focal alveolar hemorrhage have been described in a pig experimentally infected with PCV2 and PPV. 44 Specifically, there was necrosis and proliferation of endothelial cells and mononuclear cell infiltration of the media and intima. 44 Severe diffuse segmental to circumferential lymphohistiocytic and plasmacytic periarteritis and endarteritis (Fig. 11) in several organs was also observed in a pig naturally infected with PCV2. 70 PCV2 antigen was demonstrated in endothelial cells and inflammatory cells in the arterial walls. 64 In three pigs experimentally infected with PCV2, viral antigen was associated with obliterated blood vessels in areas of granulomatous and necrotizing lymphadenitis. 70 A study further attempting to characterize vascular lesions found that lymphoid necrosis in clinically affected pigs suffering from systemic PCVAD may be related to hypertrophy and hyperplasia of high endothelial venules, and necrotizing lesions may develop in affected lymph nodes following vascular damage with thrombosis and subsequent follicular necrosis. 28 Recently, acute necrotizing and chronic lymphohistiocytic and plasmacytic vascular lesions were also observed in pigs experimentally infected with PCV2, several of which had also been experimentally co-infected with BVDV1.57,58 It was determined that PCV2-infected endothelial cells displayed an activated phenotype, characterized by an increase in cell surface procoagulant activity. 64 Moreover, the PCV2-infected endothelial cells pretreated with exogenous thrombin displayed an increased viral load. 64
Degeneration of endothelial cells, perivascular and intramural edema, fibrinoid necrosis, vasculitis, perivasculitis, and vascular thrombi associated with PCV2 antigen were also observed in lung tissues in 140 Hungarian pigs. 112 The viral antigen was present in intravascular mononuclear cells, endothelial cells, myocytes, and infiltrating inflammatory cells in 13% to 100% of the pulmonary lymph and blood vessels in 78% of the respiratory cases in these pigs. Detection of PCV2 antigen was positively correlated with the presence of the vascular lesions, and the probability of detecting vascular lesions in PCV2-infected lungs was higher than in lungs infected with porcine reproductive respiratory syndrome virus, Pasteurella multocida, or Streptococcus spp. 112 The increasing observation of field PCVAD cases in North America with diffuse distention of the pulmonary interlobular septa by edema and fibrinoid necrosis of the blood vessel wall12,16 has led to the proposal of a new peracute PCVAD syndrome: acute pulmonary edema (APE).
In PDNS, the characteristic cutaneous and renal result from necrotizing and neutrophilic vasculitis of dermal and hypodermal capillaries and arterioles and of glomerulonephritis, respectively, accompanied by thrombosis and infarction of the circumjacent tissues. 42 In addition to necrotizing vasculitis in the skin and kidney, vascular lesions in PDNS cases can also be seen in other tissues, most prominently in the mesentery and spleen.6,42,89,95,103,114
In the heart, abundant intracytoplasmic PCV2 antigen was associated with myocardiocyte swelling or necrosis, or myocardial fibrosis (or both) in three naturally infected pigs aged 4 to 7 weeks from three different farms. 70 Cardiomegaly, especially on the right side of the heart and with pericardial effusion, was reported in clinically affected Spanish pigs, and PCV2-associated myocarditis was found to be the primary cause of congestive heart failure in these cases (Marco E. PMWS control – European style. Proc Swine Disease Conf Swine Pract 10:83-90, 2002, Ames, Iowa).
In fetuses, microscopic lesions associated with PCV2 are most prominent in the cardiovascular system, especially in the heart, making this the organ of choice for diagnostic work-up of PCV2-induced abortion. Often, myocardiocytes are degenerate, necrotic, or lost and replaced by fibrous connective tissue and mineralization.61,67,88 In addition, there is myocarditis associated with infiltration by lymphocytes, plasma cells, and macrophages of varying degrees; occasionally multinucleated giant cells are also present. Botyroid intranuclear inclusion bodies can be observed in mycocardiocytes. Often, abundant PCV2 antigen can be demonstrated in these lesions.60,65,67,88 The exact timing of myocardial lesions in fetuses is difficult to determine as time of exposure of the fetus depends on dam viremia, spread of PCV2 to the uterus, and transmission between fetuses. When fetuses were directly infected with PCV2 and harvested 21 days later, PCV2 was present in cardiomyocytes at higher levels in fetuses infected at 57 and 75 days of gestation compared to fetuses infected at 92 days of gestation or cesarean-derived piglets infected one day after birth. 97
Based on the body of evidence present in the literature, including the ability of PCV2 to replicate in endothelial cells, vascular changes appear to be very important in the pathogenesis of PCVAD. Thus, future research should perhaps focus on this organ system.
Skeletal System
Infectious PCV2 has been demonstrated in bone marrow; 76 however, in experimentally infected pigs, both myeloid (M) and erythroid (E) cell series were present and followed a normal maturation sequence. 80 M:E ratios in these animals were within reference intervals for the species, and megakaryocytes were observed in all specimens indicating no PCV2 attributable changes in the bone marrow. 80 To the authors’ knowledge, PCV2 has not been demonstrated or associated with any direct changes in bone or cartilage. In 2007, a syndrome affecting neonate calves characterized by high case fatality rates and later linked to vaccine-induced alloreactive antibodies 7 was described in Germany, followed by reports in Belgium, England, France, Germany, Italy, the Netherlands, Scotland, and Wales. The syndrome termed bovine neonatal pancytopenia is characterized by marked pancytopenia, panmyelophthisis, widespread hemorrhage, and absence of megakaryocytes. 7 Interestingly, although involvement of infectious factors or toxins has been ruled out, PCV2 was initially associated with this syndrome, 43 but this was not confirmed by others. 122
Muscular System
Although PCV2 DNA can be demonstrated in skeletal muscle by polymerase chain reaction (PCR) and is infectious in a pig bioassay, 76 muscle-specific lesions have not been reported to the authors’ knowledge. Likely, detection of PCV2 in this system is due to presence of PCV2 in the vascular system rather than in the actual myofibers, which is further supported by the lack of detectable PCV2 antigen by IHC stains. 76
Endocrine System
Granulomatous lesions associated with intralesional PCV2 antigen have been reported in pancreas, thyroid, and adrenal glands of pigs experimentally infected with either PCV2 alone or a combination of PCV2 and PPV. 44
PCV2-Associated Lesions: Effect of PCV2 Subtype
Taxonomy of PCV2
Based on a consistent difference in 6 amino acids spanning sequence in the capsid better known as the signature motif, PCV2 isolates can be further subdivided into PCV2a and PCV2b. 14 Another difference between these 2 main PCV2 subtypes is the length of the PCV2 genome, which is 1767 nucleotides for PCV2a and 1766 nucleotides for PCV2b. 14 PCV2c has been identified in archived tissues in Denmark. 21 Although PCV2d and PCV2e 117 have been described to be present in China, there is ongoing discussion if these represent true PCV2 subtypes due to the usage of a different classification system. 19
Comparison of Lesions Induced by PCV2a or PCV2b in Field Cases
There is no convincing evidence to support the differentiation of PCV2a- and PCV2b-induced lesions. To complicate this, a high rate of concurrent infection with PCV2a and PCV2b in the same pig is suspected. 46 Six PCV2 strains (open reading frame 2; ORF2) from cases of respiratory disease in Hungarian pigs were sequenced, 1 of 6 was consistent with PCV2a and 5 of 6 with PCV2b. 112 Similar results were also obtained in Serbia 100 and Portugal. 35 In China, more than one PCV2 subtype was identified in 32.2% (38/118) pigs with PCVAD, but only one PCV2 strain was detected in any of 41 clinically healthy pigs, implying that coexistence of several PCV2 strains may contribute to severity of disease. 125
Comparison of Lesions Induced by PCV2a or PCV2b in Experimentally Infected Pigs
With the emergence of PCV2b in recent years 12,14,27 and global shift from PCV2a toward PCV2b, 86 there have been concerns that PCV2b is more virulent compared to PCV2a. However, experimental studies using single PCV2 infection8,24,29,56,78 or PCV2 and PRRSV co-infection 107 were not able to support this hypothesis.
Typically, in conventional pigs experimentally infected with PCV2a or PCV2b alone, clinical disease is not observed, and although some pigs developed severe lesions especially in lymphoid tissues, there are no significant differences among groups. Similar findings were also noted in conventional pigs co-infected with PCV2a-PRRSV or PCV2b-PRRSV. 107 In CDCD pigs infected with PCV2a or PCV2b, microscopic lesions were similar. 8 In gnotobiotic pigs, no differences in lesions in pigs infected with PCV2a or PCV2b were found. These pigs had severe vacuolar degeneration and necrosis of hepatocytes with multifocal areas of hemorrhage and marked interlobular edema.29,56 Apoptotic rates in hepatic tissues were comparable in pigs infected with PCV2a or PCV2b. 106
When tissues from pigs experimentally infected with either PCV2a (n = 5) or PCV2b (n = 5) were examined by veterinary pathologists blinded to the treatment status for evidence of vasculitis lesions as indicator for PCV2b infection, 2/5 and 1/5 PCV2b pigs were correctly identified, respectively; however, there was no recognizable difference between the tissues obtained from the other PCV2a- and PCV2b-infected pigs, indicating that microscopic evaluation of tissues is not sufficient to differentiate between PCV2a and PCV2b.
Detection of PCV2
It has been recommended that diagnosis of systemic PCVAD requires the presence of microscopic lesions and PCV-antigen or nucleic acids associated with the microscopic lesions. 108 Such an association can be done by using immunohistochemical assays, in which specific antigens in tissues are stained with antibodies labeled with certain detectors, or ISH, in which DNA or RNA specific probes are used to detect a complementary often very short nucleic acid sequence in the tissue. Both methods have advantages and disadvantages such as differences in sensitivity, specificity, processing times, and cost. In recent years, with technical advances of molecular assays such as automation, increased speed, and lower cost, there has been a trend toward testing tissues for PCV2 with PCR rather than conventional IHC or ISH methods.
Effect of Detection Method
There is some concern that not all tissue-based detection assays are equal, especially in detecting different PCV2 subtypes. Previously, comparison of ISH and IHC did not indicate differences between these methods. 15 In order to re-evaluate the previous findings on tissues from pigs experimentally infected with PCV2a or PCV2b, a small study was conducted by the authors using two IHC assays that differed in the antigen used for immunization of the rabbits during the production of the polyclonal antiserum and ISH done as described 94 with the following probe sequence: DIG-5′-AGT TCA GCC AGC CCG CGG AAA-3′. The production of the polyclonal antiserum in rabbits was essentially as previously described. 109 The antigen used was either a whole-virus form of PCV2a (strain 40895) or the capsid protein expressed by the ORF2 of the same viral strain. Lymphoid tissues from gnotobiotic and conventional pigs inoculated with PCV2a or PCV2b that were part of previously published experiments56,78 were used in this comparative study. The slides were evaluated for degree of staining as described, 79 and the results are summarized in Table 2. Nine of 10 pigs were correctly identified as positive by one of the three methods while all three assays were negative on one PCV2b-infected pig. The overall results indicated that the ISH stain was easy to read and resulted in equal or higher numbers of stained cells compared to the two IHC stains. The two IHC stains appeared very similar on the tissues examined. There were no differences in detection rates of pigs infected with PCV2a or PCV2b.
Prevalence Expressed as Number of Positive/Total Number Tested (Mean Group Score ± SE) of Correctly Identified Porcine Circovirus Type 2 (PCV2) Positive Lymphoid Tissues (Pool of Tonsil, Spleen, Lymph Nodes) by Using 1 of 3 PCV2 Detection Methods, Including Immunohistochemistry (IHC) 109 and In Situ Hybridization (ISH) 94 on Tissues Obtained From Pigs Experimentally Infected With PCV2a or PCV2b.
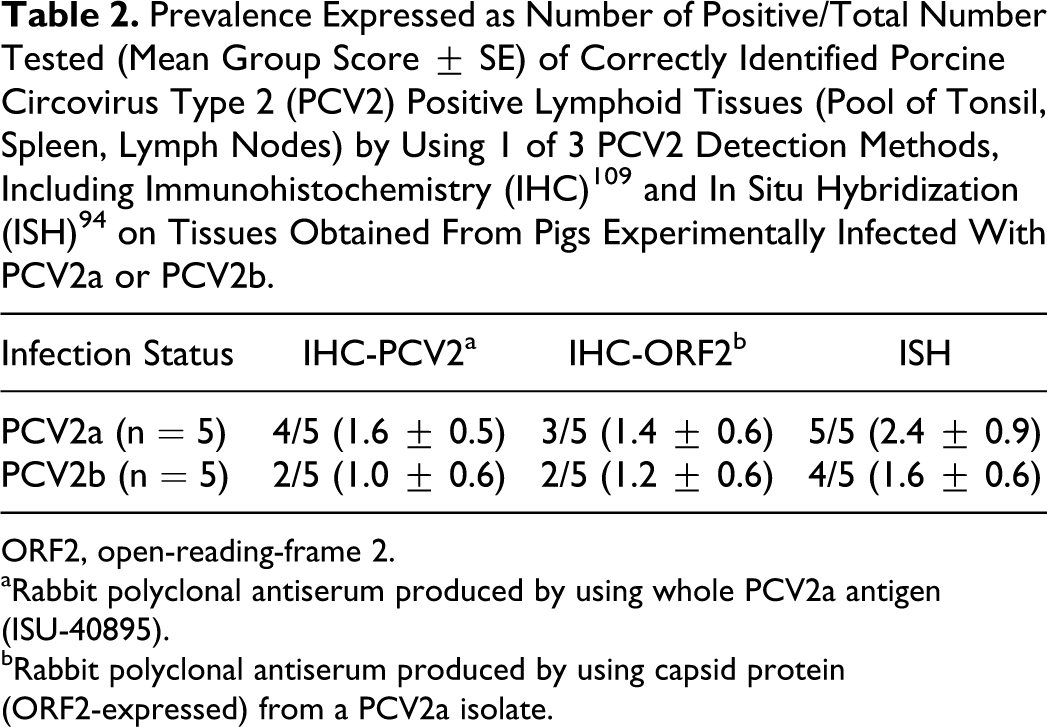
ORF2, open-reading-frame 2.
aRabbit polyclonal antiserum produced by using whole PCV2a antigen (ISU-40895).
bRabbit polyclonal antiserum produced by using capsid protein (ORF2-expressed) from a PCV2a isolate.
Effect of Storage Time of Formalin-Fixed Tissues
Most laboratories use neutral-buffered 10% formalin for tissue fixation, which introduces cross-linking of immune reactive sites in the tissues. Furthermore, formaldehyde overfixation can result in a reversible or nonreversible loss of immunoreactivity by masking or damaging antibody binding sites. 120 Previously, it has been determined that a delay in processing formalin-fixed tissues results in reduction or even prevention of detection of PRRSV viral antigen by IHC. 116 PRRSV antigen in lungs of experimentally infected pigs was no longer detectable 6 days after fixation. 116 Although RNA viruses such as PRRSV are more prone to be damaged by formaldehyde compared to DNA viruses such as PCV2, there is concern that formalin fixation could interfere with detection of PCV2 antigen or nucleic acids. In order to further evaluate this, the authors examined 4 representative lymph nodes from a pig experimentally infected with PCV2a (PCV2 isolate 40895) and necropsied 21 days post experimental inoculation. The pig was part of an experiment that was described previously 74 and had moderate to severe lymphoid lesions associated with moderate amount of PCV2 antigen (scores 2-3; score 0 = not detectable, score 3 = large amount of PCV2 antigen present). The selected lymph nodes were stored in 10% neutral-buffered formalin in a lightproof container at ambient room temperature. Periodically, sections of the lymph nodes were cut into blocks, routinely processed, and paraffin embedded. This was done at the following time intervals after initial necropsy and fixation: 7 days, 14 days, 1 month, 2 months, 4 months, 9 months, 16 months, and 5 years. All blocks were further processed at the same time by cutting 3 5-μm-thick sections of each block and subjecting the slides to 1 of 4 detection methods: IHC-PCV2 using a polyclonal rabbit hyperimmune serum based on whole PCV2a virus; IHC-ORF2 using a polyclonal rabbit hyperimmune serum based on the capsid protein, expressed by ORF2 of PCV2a; and ISH using a single-stranded, 21-base, oligonucleotide DNA probe end-labeled with digoxigenin targeting the ORF2 gene. The slides were evaluated for degree of staining as described, 79 and the results are presented in Table 3. The results indicate that all assays were successful in detecting PCV2 antigen in tissues stored in formalin for 5 years. While both the PCV2-IHC stain and ISH had similar results, the overall staining intensity was reduced with the ORF2-IHC stain. There was a weak but noticeable decrease in the signals over time. One possible explanation for this is difference in amounts of the target in the various sections cut from the block, which could explain the disparity seen with the ORF2-IHC stain of the tissues fixed in formalin for 16 months and 5 years. Alternatively, the accessibility of the target may have been decreased through continued formaldehyde group binding to the tissue. 25 Regardless of the perceived preservation of PCV2 in this small investigation, it is important that tissues should not remain in formalin for extended periods of time as also determined for other antigens and nucleic acids.66,119
Amount (–, None; +, Low; ++, Moderate; +++ Abundant) of Porcine Circovirus Type 2 (PCV2) Antigen or Nucleic Acids Detected in Lymphoid Tissues From a Pig Experimentally Infected With PCV2 Stored in 10% Neutral-Buffered Formalin for Different Intervals of Time Using 1 of 3 PCV2 Detection Methods, Including Immunohistochemistry (IHC) 109 and In Situ Hybridization (ISH). 94

ORF2, open-reading-frame 2.
aRabbit polyclonal antiserum produced by using whole PCV2a antigen.
bRabbit polyclonal antiserum produced by using capsid protein (ORF2-expressed) from a PCV2a isolate.
Other Detection Methods to Determine PCV2 Status of a Herd or Pig
Nowadays, PCR detection methods are widely used to determine virus ecology within herds and to determine pathogen freedom of pigs or porcine products such as semen that are transferred between farms. PCV2 as a ubiquitous virus can be easily detected in serum, 104 tissues, 11 oral fluids, 90 and environmental samples.85,86 In addition, PCV2 was found to be shed in semen from experimentally infected boars for at least 90 days. 62 Moreover, multiplex assays capable of differentiating between PCV2 subtypes have been described 26 and are available in most veterinary diagnostic laboratories. Attempts to correlate the amount of PCV2 in different sample types with clinical disease stage of a pig 11 have shown that there are many variables that need to be considered when interpreting results, such as extraction method, PCR assay used including target areas or primers and probe, standards used for quantification, sample type, and disease stage of the pig. Furthermore, pooling of samples, a common practice done to reduce overall test cost, can interfere with interpretation. 18
Other detection methods that have been used to determine the PCV2 status of a herd include serological assays. However, the value of this assay type for determining disease status is questionable as most herds are subclinically affected with PCV2 81 and most pigs will be seropositive. Recent comparisons of in-house and commercially available anti-PCV2 antibody detection methods found that the included assays were not able to differentiate between antibodies against different PCV2 subtypes but that they correctly identified pigs infected with PCV2 versus pigs infected with PCV1 or noninfected negative controls.83,84
Summary and Conclusions
PCV2-associated lesions in pigs can involve many organ systems and be of varying severity. Differentiation of PCV2 subtypes by the type of microscopic lesions induced is difficult if not impossible. Current detection methods on tissue samples appear to be sufficient to detect PCV2 of different subtypes; however, differences in amount of staining exist, and care must be taken by comparing results obtained with different methods.
Footnotes
Acknowledgement
We thank Dr Patrick Halbur for critical review of this manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
