Abstract
Gliomatosis cerebri is a well-recognized entity in human medicine characterized by unusually widespread infiltration of the neuraxis by neoplastic glial cells with relative preservation of brain architecture. This report describes the pathologic features of the disease in six dogs. The dogs ranged from 3 to 9 years of age (mean 6.1 years) without evidence of breed predilection; five of the six dogs were neutered or intact males. The clinical findings were mixed (including depression, circling, cranial nerve deficits), reflecting the diffuse nature of the disease. Histologically, there was remarkably diffuse infiltration of the white and gray matter of the brain by small numbers of elongated neoplastic cells. Areas of greater cellularity formed grossly visible lesions in four cases. Anisocytosis and pleomorphism were greater in areas of higher cellularity. Other features of tumor growth included subpial accumulation, neuronal satellitosis, perivascular cuffing, and tropism for cranial nerve and brain stem nuclei. Neoplastic cells were negative on immunohistochemical stains for glial fibrillary acid protein (GFAP) and leukocyte markers, reflecting the uncertain histogenesis of these unusual neoplasms.
Gliomatosis cerebri is a rare human disorder characterized by diffuse and widespread infiltration of the central nervous system (CNS) by neoplastic glial cells. 2,9,10 Long-recognized but poorly understood, the World Health Organization Classification of Tumours currently classifies gliomatosis cerebri as a neuroepithelial neoplasm of unknown origin. An astrocytic origin is suspected and, although differentiation from diffuse astrocytomas has been questioned, 4 gliomatosis cerebri is distinguished from astrocytomas by more widespread neuraxial involvement and relative preservation of brain architecture. In reported human cases, the condition is highly variable in regard to age of onset, clinical progression, extent of CNS involvement, and histologic characteristics. 1,3,6 Two general forms are recognized. 9 Type I is the classical form, in which there is diffuse infiltration without the formation of a discrete, grossly visible mass. In type II gliomatosis, the diffuse infiltrate is accompanied by a mass lesion. Reference to gliomatosis cerebri in the veterinary literature are few, 8,14 and to the authors' knowledge, this is the first detailed report of the disease in animals.
The cases in this report were referred to the Cornell University College of Veterinary Medicine between 1992 and 2001 for neuropathologic evaluation. In all cases, the necropsy was performed by the referring veterinarian and the brain was submitted following fixation in 10% neural buffered formalin. Signalment and clinical history information are summarized in Table 1. After gross examination of the brains representative samples extending from the frontal lobes to the medulla were processed routinely, sectioned at 5 μm, stained with hematoxylin and eosin (HE), and examined microscopically. Immunohistochemical stains were performed for glial fibrillary acidic protein (GFAP), CD18, CD3, CD45RA, CD79α, and BLA36 using the streptavidin peroxidase method according to the manufacturer's instructions (Histostain;rM SP Kit, Zymed Laboratories, San Francisco, CA). The primary antibodies were rabbit polyclonal for GFAP (DAKO, Carpinteria, CA) at a dilution of 1:3,000, rabbit polyclonal for CD3 (DAKO) at 1:100 with pepsin pretreatment for 30 minutes at 37°C, mouse monoclonals for CD18 and CD45RA (P. F. Moore, Davis, CA), both at 1:50, and mouse monoclonals for CD79α and BLA36 (DAKO), both at 1:25. For CD18, CD45RA, CD79α, and BLA36, antigen retrieval consisted of two 10-minute microwave treatments in 0.01M citrate buffer at pH = 6. The chromagen was diaminobenzidine and the slides were counterstained with Gill's hematoxylin. Positive controls for the leukocyte markers consisted of canine spleen, lymph node, tonsil, and lung (alveolar macrophages). Reactive microglial cells were recognized with CD18, providing a positive internal control. A positive internal control (glia limitans) was also used with GFAP. For negative controls, the primary antibody was substituted with phosphate-buffered saline, normal rabbit serum, or an isotype-matched irrelevant mouse monoclonal antibody.
Signalment and clinical history for six dogs with gliomatosis cerebri.∗
∗ NM = neutered male; SF = spayed female; CSF = cerebrospinal fluid; CN = cranial nerve; BAER = brain auditory evoked response; CT = computerized tomography.
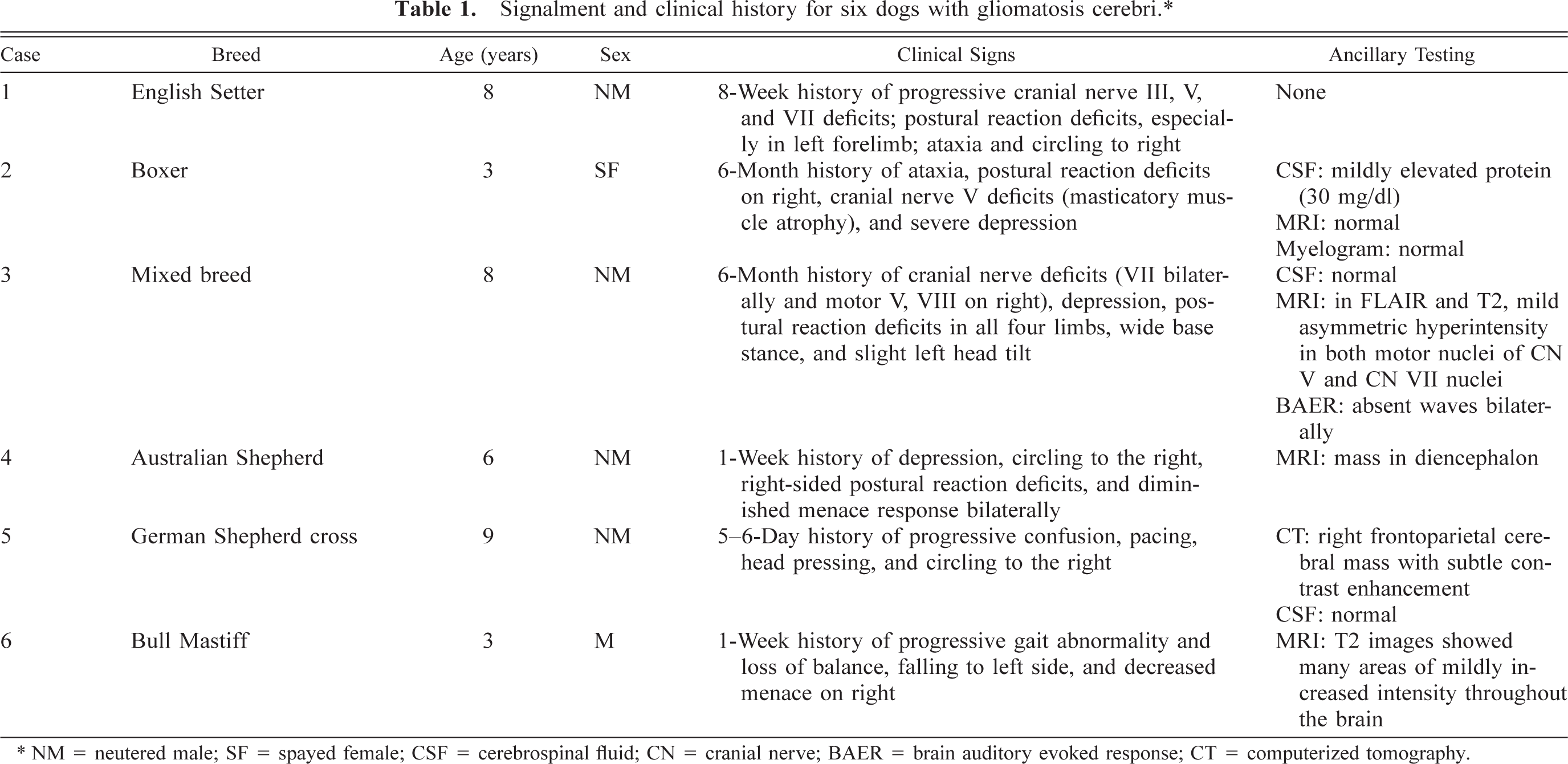
Gross abnormalities of the brain were detected in four dogs. In dog No. 1, there was a slightly soft, gray, irregular area in the left brain stem that extended from the caudal mesencephalon to the caudal medulla. Dog No. 2 had mild diffuse enlargement of the medulla with loss of structural detail and mottled discoloration of the mesencephalon. Dog No. 5 had a gray swollen area in the frontoparietal region of the right cerebrum, centered in the internal capsule and extending into the centrum semiovale and corona radiata (Fig. 1). A discrete mass was evident in dog No. 4. The mass was 15 mm × 12 mm, pale yellow to light gray, centered on the body of the fornix, and bulging into the lateral ventricle on the right side. Hydrocephalus was noted in two dogs (Nos. 2 and 4). Two brains (Nos. 3 and 6) were grossly normal.
Cerebrum; dog No. 5. Neoplastic infiltration has expanded the right hemisphere, producing a mild midline shift. Tumor effaces the centrum semiovale and infiltrates the corpus callosum, caudate nucleus, and internal capsule.
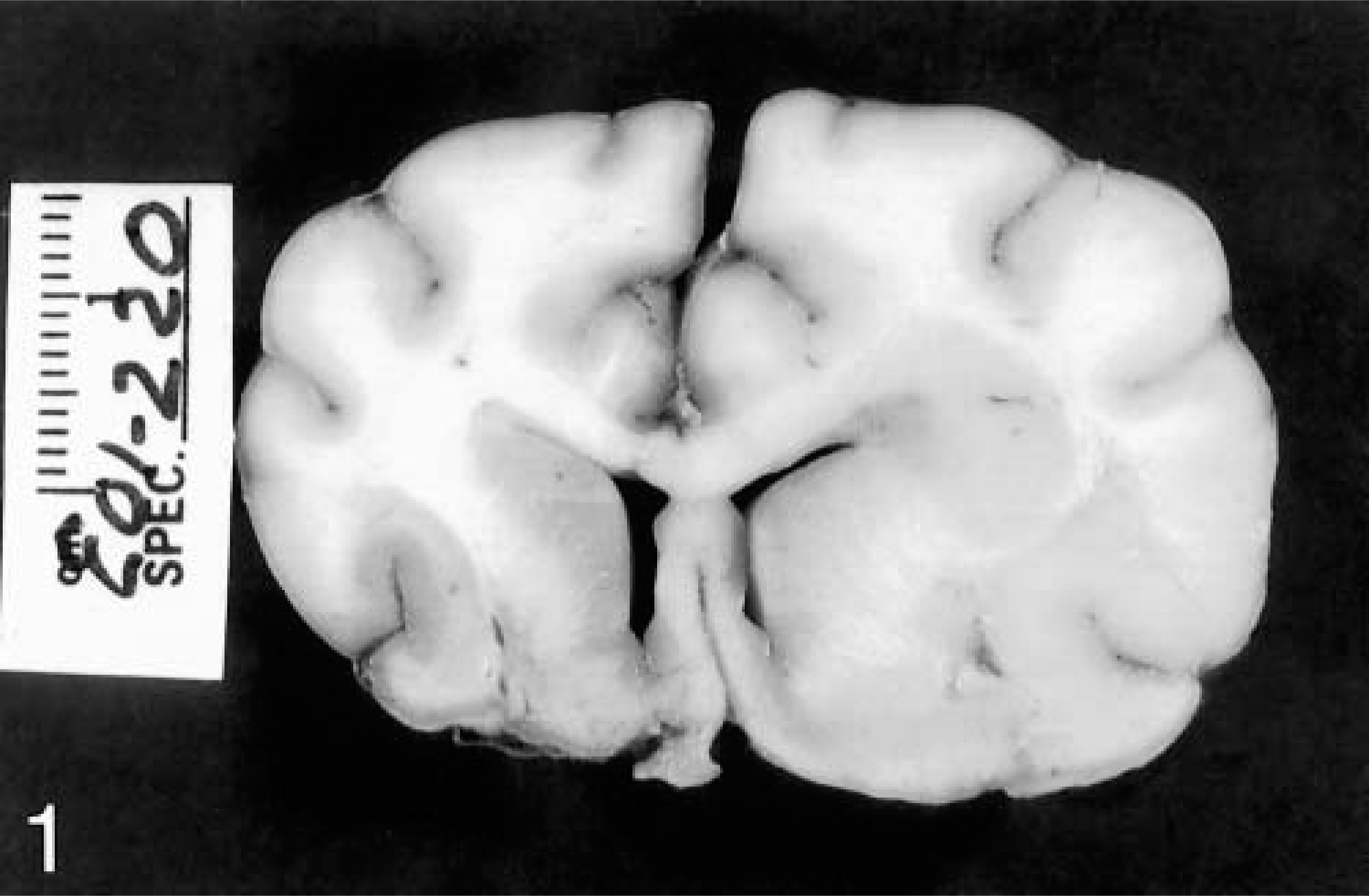
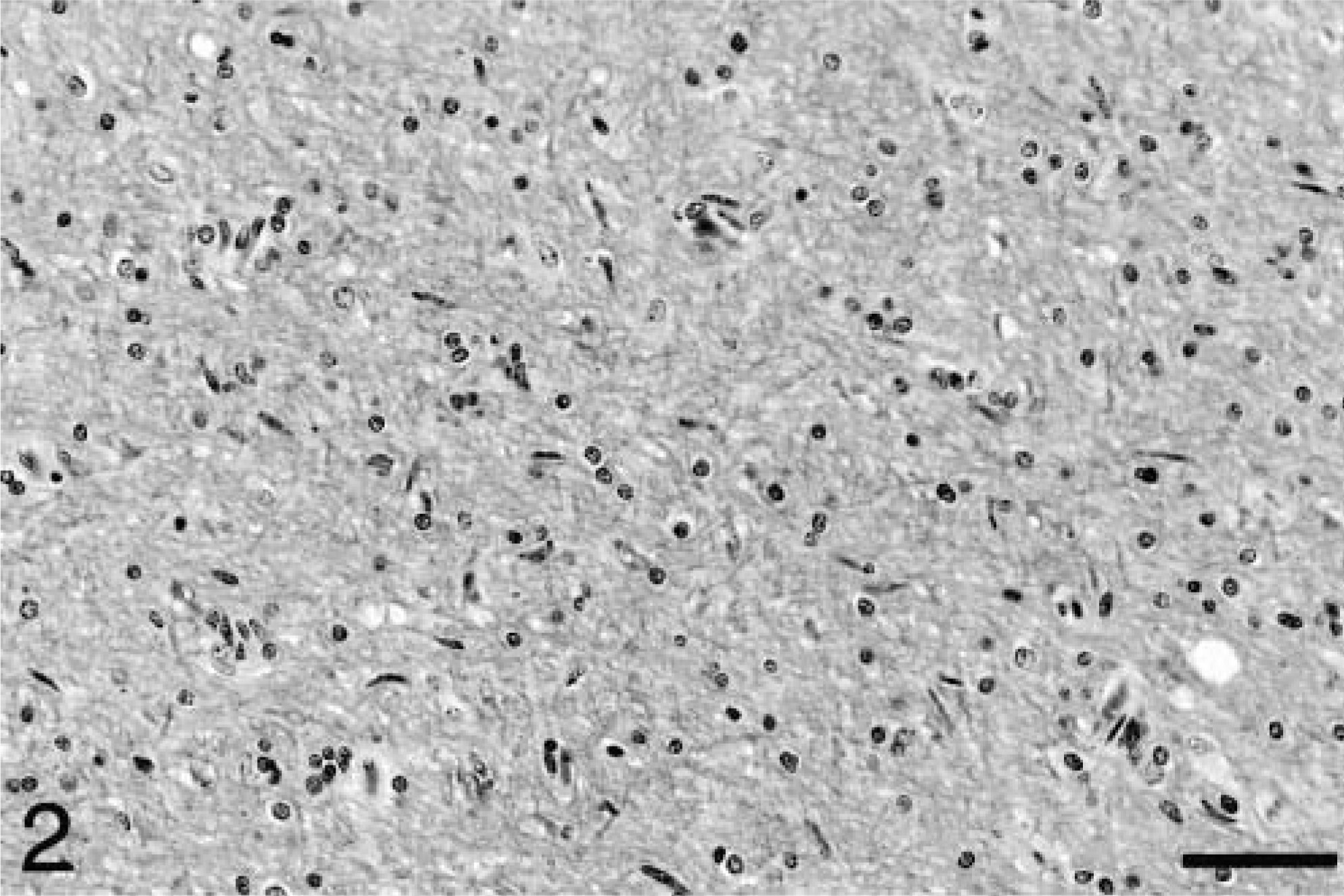
Histologic examination revealed remarkably diffuse infiltration of both white and gray matter by elongated neoplastic glial cells. In all cases, the degree of brain involvement seen histologically was much more widespread than anticipated during gross examination. In five of the six cases, neoplastic cells were present in every section of brain examined, ranging from barely discernible (Fig. 2) to occasionally dense (Fig. 3). In most brain sections, neoplastic cells were scattered in small numbers diffusely, with no evidence of necrosis or disruption of normal architecture. Heavier concentrations were most often present in the brain stem and were often widely separated. In dog No. 1, a densely infiltrated area extended from the left mesencephalon to medulla. Intense multifocal infiltrates were present in the cerebrum, midbrain, pons, and medulla in dog No. 2 and in the midbrain and pons in dog No. 6. In addition to the mass lesion in the fornix, dog No. 4 had intense infiltration of the hippocampus. In dog No. 5, the neoplastic infiltrate was concentrated in the right frontal lobe and extended caudally to the thalamus. In all cases, intense areas of cellularity merged gradually into the surrounding parenchyma, with indistinct margins between normal and abnormal tissue. In all but dog No. 5, the cerebellar white matter and cortex were diffusely infiltrated. White matter was generally more severely affected than gray matter, but nuclear tropism was evident in four cases. Dog No. 3 exhibited marked nuclear tropism, with dense neoplastic infiltrates in the cerebellar nuclei, medial geniculate nucleus, and nuclei of cranial nerves V, VIII, X, and XII (Fig. 4) Cranial nerve nuclear involvement in this case corresponded to the deficits observed clinically. The cerebellar nuclei were also infiltrated in dogs No. 1 and No. 2, while the motor nucleus of cranial nerve V and medial geniculate nucleus were infiltrated in dogs No. 2 and No. 6, respectively. Spinal cord involvement, including both white and gray matter, was evident in the cervical spinal cord of dog No. 3, the only case in which spinal cord was examined.
Cerebrocortical white matter; dog No. 5. The white matter is diffusely infiltrated by a small number of elongated neoplastic glial cells. HE. Bar = 57 μm.
Corona radiata; dog No. 4. In this area of greater cellularity, neoplastic cells are arranged parallel to the direction of the fibers of white matter tracts. HE. Bar = 57 μm.

Medulla oblongata; dog No. 3. Neoplastic cells are concentrated within the motor nucleus of cranial nerve V (arrowheads). Vacuolation of neurons in this nucleus in the dog is an incidental finding. HE. Bar = 286 μm.
Occasional neuronal satellitosis by neoplastic cells was present in all areas of the brain and was most evident in areas of more intense infiltration (Fig. 5) Neuronal degeneration was uncommon. Evidence of axonal degeneration was lacking along the corticospinal tract, providing indirect evidence of neuronal preservation. Mild to moderate edema, evident as separation of the neuropil, was often present in intensely infiltrated areas and in two dogs (Nos. 1 and 2), there was mild cavitation with the accumulation of small numbers of gitter cells. In affected white matter, elongated neoplastic cells were often arranged parallel to the fiber tracts (Fig. 3). In the cerebrum, subpial infiltration was noted, being mild in dog No. 6 and marked in dog No. 5. In the cerebellar cortex, minimal to small numbers of neoplastic cells were present beneath the pia mater in five of the six dogs (Fig. 6), a feature also characteristic of primitive neuroectodermal tumors. Perivascular cuffing by neoplastic cells was often present in areas of dense infiltration. Dogs No. 3 and 5 had small subependymal accumulations of neoplastic cells. Small lymphocytic or lymphoplasmacytic perivascular cuffs were noted in three dogs. Other unique features of dog No. 5 included rare glomeruloid vessels and multifocal extension of neoplastic cells into the leptomeninges (Fig. 7).
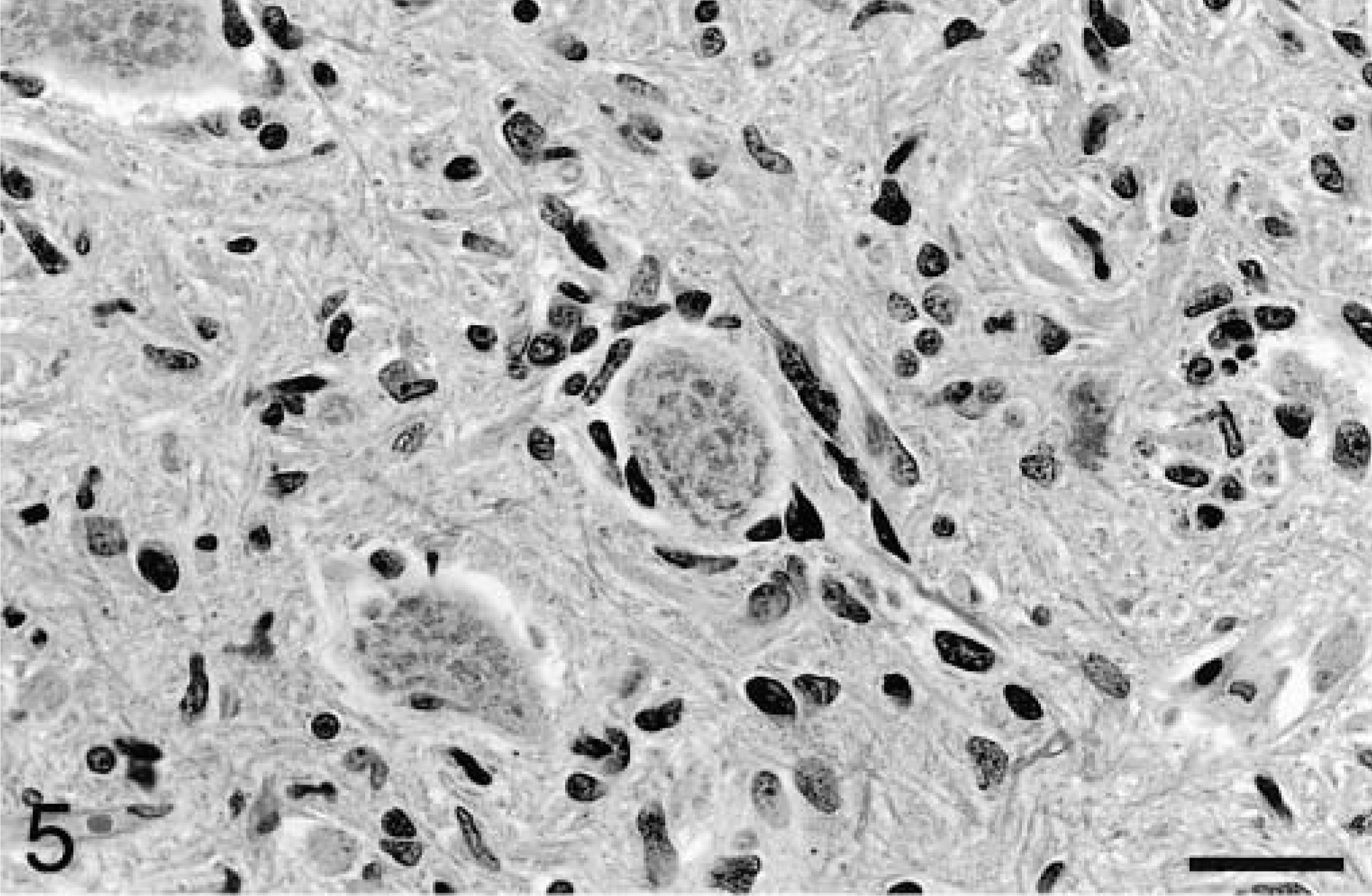
Medulla oblongata; dog No. 3. Neurons are occasionally surrounded by neoplastic cells. HE. Bar = 29 μm.
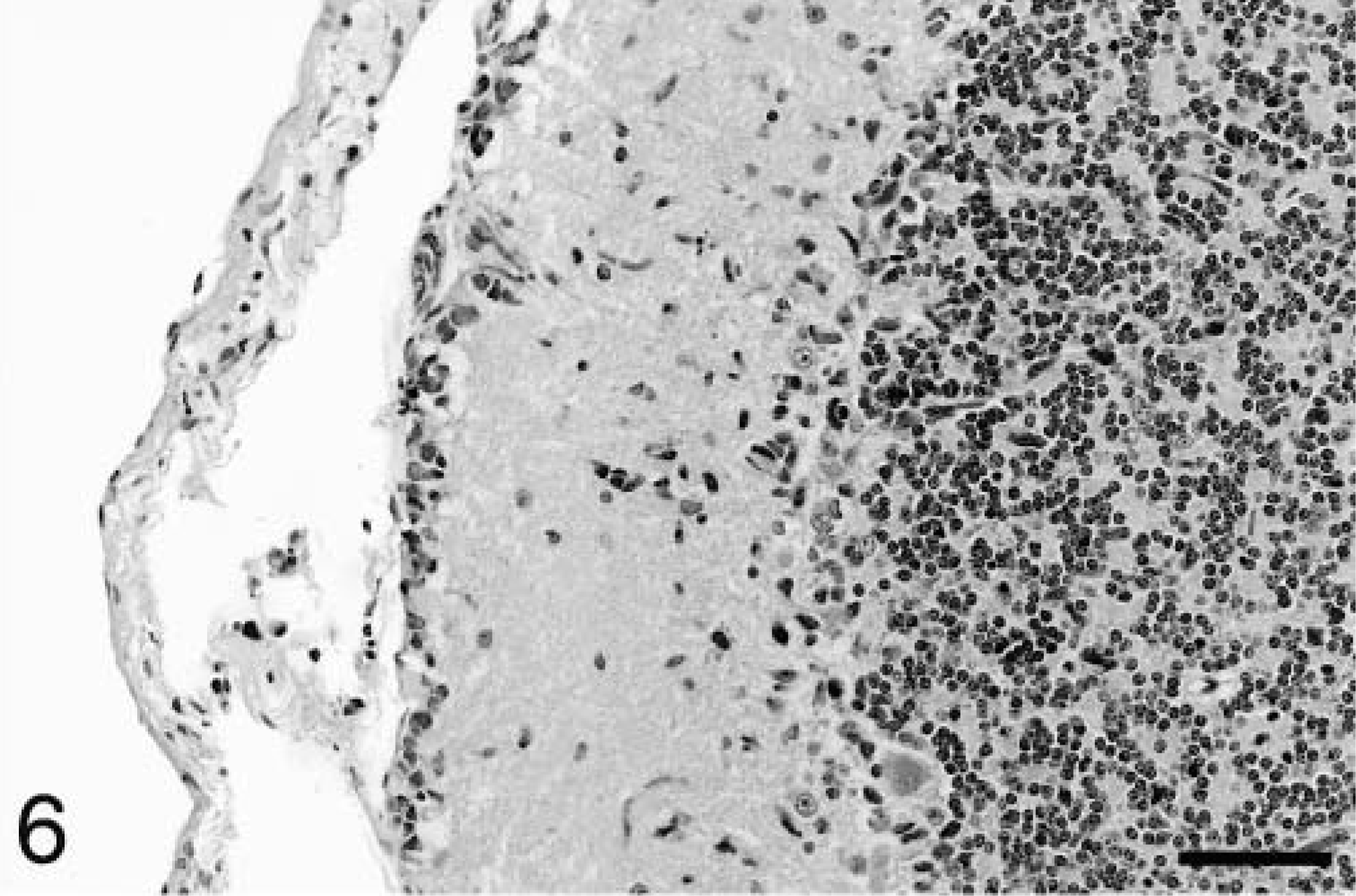
Cerebellum; dog No. 2. A narrow band of neoplastic cells is present beneath the pia mater. HE. Bar = 57 μm.
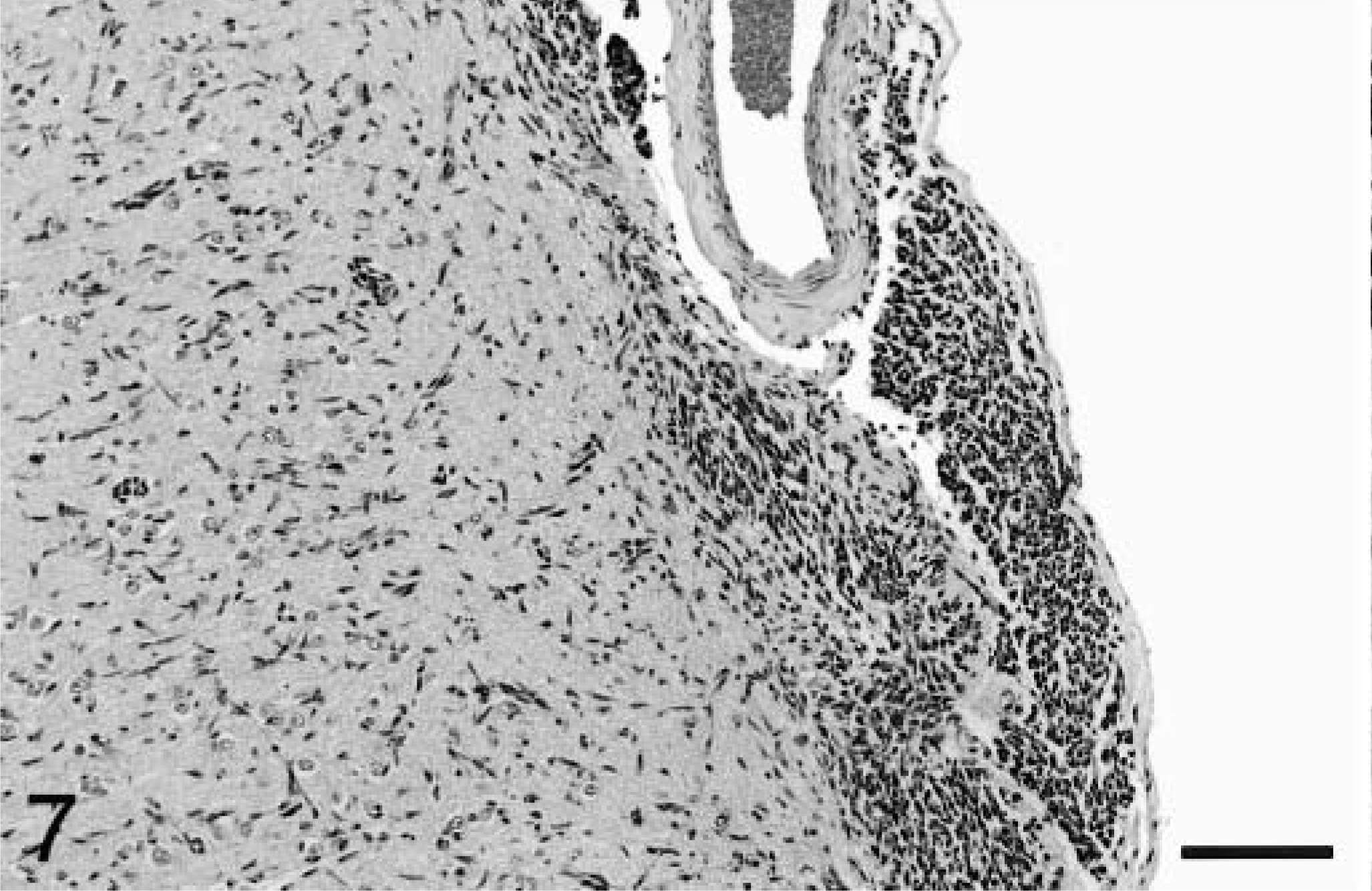
Cerbral cortex; dog No. 5. Neoplastic cells extend into the leptomeninges from the neocortex. HE. Bar = 114 μm.
Morphology of the neoplastic cells was similar in all six dogs, with most cells having ovoid to slender nuclei, with coarsely stippled chromatin and indistinct cytoplasm. In general, cells within more intensely infiltrated areas were larger and more pleomorphic than those found elsewhere. These cells were elongated, often with curved, angular, and bizarrely shaped nuclei (Fig. 8). The mitotic rate was 2–4 mitoses per 40× field in highly cellular areas. The mass lesion centered on the fornix in dog No. 4 was composed of short parallel bundles of elongated neoplastic cells. In all cases, neoplastic cells were negative for GFAP, CD18, CD3, CD45RA, CD79α, and BLA36. Small numbers of scattered GFAP-positive cells with prominent branching processes were interpreted as reactive astrocytes. Scattered CD18 and BLA36 positive cells were occasionally present and interpreted as reactive microglial cells. Widespread cellular and white matter staining with CD79α was interpreted as nonspecific and spurious. During case selection, one putative case of gliomatosis cerebri was excluded due to positive staining with CD45RA and BLA36 and was reclassified as a B cell lymphoma.
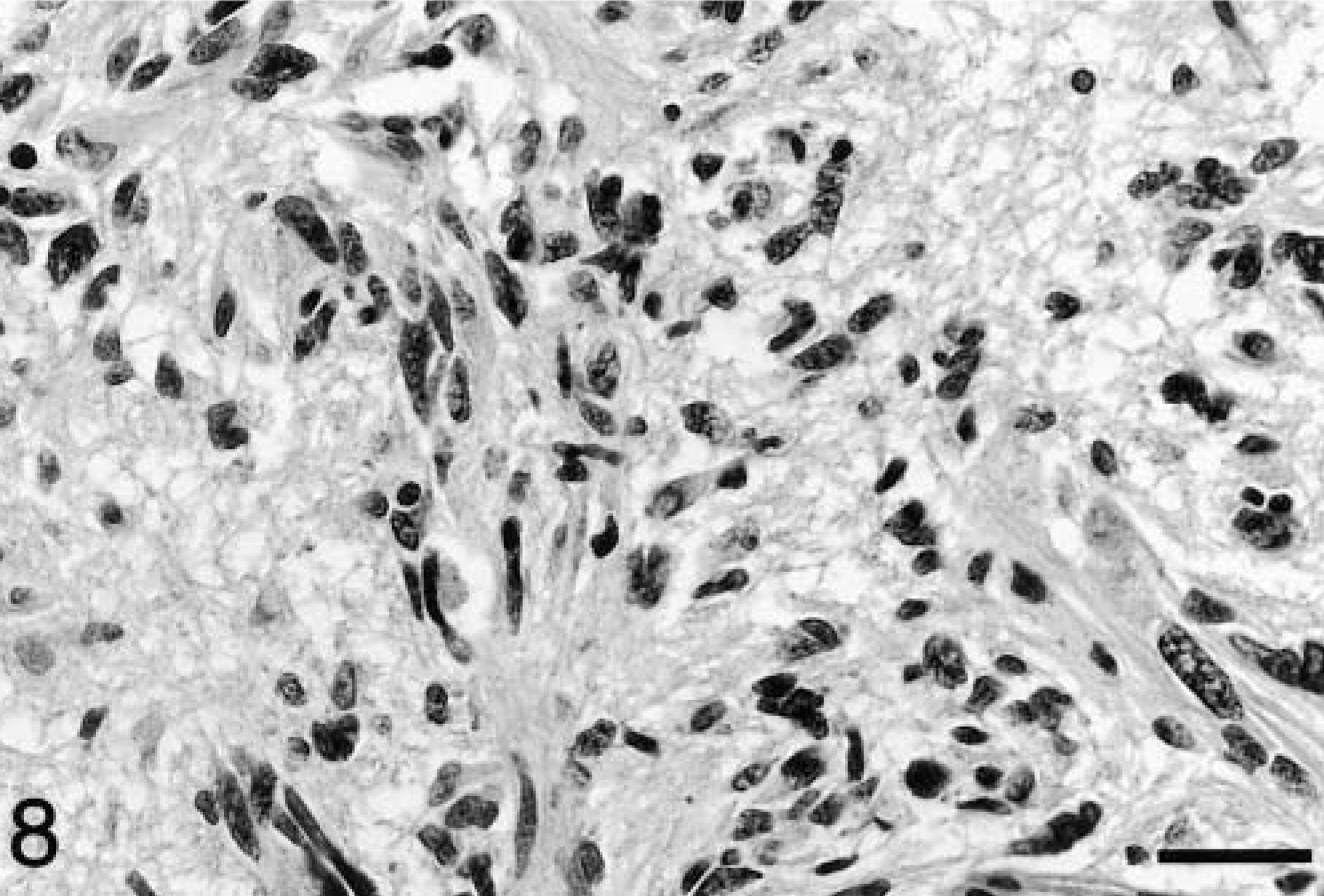
Body of fornix; dog No. 4. The presence of larger, pleomorphic neoplastic cells is a feature in areas of greater cellularity. HE. Bar = 29 μm.
The reports of gliomatosis cerebri in humans have many similarities to the cases presented here. 1,2,6,7,10,13 As in the dog, reports of human cases indicate remarkable variation in the area of brain involvement, but extensive tumor burden is the hallmark of the disorder. The cerebrum is most consistently affected, but the brain stem is often involved concurrently. There is relatively less involvement of the cerebellum in man. As in the dog, white matter is typically more severely affected than gray matter, and diffusely infiltrated areas are interspersed with denser focal accumulations. Neuronal satellitosis, subpial and subependymal accumulations, perivascular cuffing, and parallel arrangement of neoplastic cells within white matter tracts are other features common to both human and canine cases. Nuclear tropism, as seen in several of the canine cases, has not been noted in human cases. Cytologically, human cases are pleomorphic, with cells ranging from well-differentiated protoplasmic and fibrillary astrocytes to elongated, more poorly differentiated glial cells. As with canine cases, pleomorphism and anisocytosis are more marked in densely infiltrated areas of the brain. Immunohistochemical staining for GFAP in human cases has been highly variable. Well-differentiated neoplastic cells have often stained positively, but less differentiated, elongated forms have typically been negative.
The clinical signs exhibited by these dogs were extremely variable, reflecting the diffuse nature of the disease. Likewise, the human disease is characterized by a wide variety of clinical presentations. Common symptoms in human cases include corticospinal tract deficits, dementia, headaches, mental and behavioral changes, and seizures. 1,3,9 Signs and symptoms may begin abruptly or progress slowly for weeks or months. In affected dogs, the duration of illness ranged from 1 week to 6 months. In human cases, there is no evidence of sex predisposition and all ages are affected, with the peak incidence between 40 and 50 years of age. 9 Similarly, four of the six dogs in this study were 6–9 years of age. No breed predisposition was evident, but five of the six cases were in males. In man, magnetic resonance imaging (MRI) is the diagnostic modality of choice, revealing hyperintensity of T2-weighted images. 12 Clinical signs included cranial nerve deficits in three of these dogs, and in dog No. 3, lesions in cranial nerve nuclei were evident by MRI. While the diagnosis can be made with the use of MRI and brain biopsy, gliomatosis cerebri remains a difficult antemortem diagnosis. 9,12
With the unusually widespread infiltration of the brain by this neoplasm, there is a limited differential diagnosis. CNS lymphoma is a viable differential, and we eliminated one case that had been diagnosed as gliomatosis cerebri following positive immunohistochemical staining for B cell markers. In retrospect, morphologic differences included more rounded nuclei and more widespread perivascular and leptomeningeal involvement in CNS lymphoma. Neoplastic cells in primitive neuroectodermal tumors may be fusiform and show subpial infiltration but are commonly limited to the cerebellum, with a predilection for young animals. 8
The cell of origin of gliomatosis cerebri is a subject of debate. Efforts to determine the histogenesis have been hindered by the limitations of current immunohistochemical techniques. The majority of human cases are thought to be of astrocytic origin, based on morphology and, in some cases, positive immunohistochemical staining with GFAP. In many human cases, the neoplastic cells exhibit a range of differentiation. Oligodendroglial forms of gliomatosis cerebri have been reported, 9 and rare cases exhibit both astrocytic and oligodendroglial differentiation. 1 The canine cases reported here exhibit less morphologic evidence of an astrocytic origin than reported human cases. Also, the lack of GFAP staining in canine cases fails to support an astrocytic origin. Similarly, negative GFAP staining was seen in six of seven cases of gliomatosis cerebri in a previous immunohistochemical study of canine neuroectodermal tumors. 14
The term microgliomatosis has long been used to describe diffuse infiltrative tumors composed of neoplastic cells of putative microglial origin and so may also contribute to gliomatosis cerebri. Since the advent of immunohistochemisty, it has been recognized that most previously reported cases of human microgliomatosis were actually primary central nervous system lymphomas. 11 Tumors of microglial cells appear to be extremely rare, with the diagnosis depending on the demonstration of macrophage-specific markers such as Ham-56 or CD68. 5 In the few reported cases of microgliomatosis in dogs, the diagnosis was based strictly on cellular morphology. 15,16 Morphology alone, however, appears to be unreliable, as both microglial cells and astroglial cells are capable of assuming elongated forms. Tumor cells in the canine cases reported here were negative for CD18, which is expressed by cells of monocyte/macrophage lineage (P. F. Moore, personal communication). This finding does not support a microglial origin.
Neoplasms grouped under the term gliomatosis cerebri may actually represent tumors of more than one cell lineage or perhaps an origin from uncommitted progenitor cells. Until more specific techniques become available, the histogenesis will remain speculative. Despite the controversy surrounding its classification and cellular origin, gliomatosis cerebri is a distinct entity and should by included in the differential diagnosis of widely disseminated canine intracranial neoplasms.
Note added in proof: In a recent study of a human gliomatosis cerebri case, similar genetic aberrations were identified in tumor cells at multiple random locations throughout the brain, supporting the concept of monoclonal tumor proliferation. (Kross JM, Zheng P, Dinjens WNM, Alers JC: Genetic aberrations in gliomatosis cerebri support monoclonal tumorigenesis. J Neuropathol Exp Neurol
Footnotes
Acknowledgements
The authors thank Pat Fisher and Joy Cramer for help with immunohistochemistry, Dr. Sean McDonough for help with interpretation of immunohistochemical stains, and Alexis Wenski-Roberts for assistance with photographs.
