Abstract
Gliomatosis cerebri (GC) is a glioma subtype with diffuse neuroparenchymal infiltration without architectural distortion. GC was first used in human neuropathology and remained controversial until its elimination from the diagnostic lexicon in 2016. GC is currently defined as a diffuse growth pattern of glioma rather than a distinct entity. In this article, we characterize 24 cases of canine GC and classify these neoplasms as diffuse gliomas. Selected cases of canine GC were reviewed and immunolabeled for oligodendrocyte lineage transcription factor 2 (Olig2), glial fibrillary acidic protein (GFAP), and 2′,3′-cyclic-nucleotide 3′-phosphodiesterase (CNPase). The mean age of affected dogs was 7 years, and 9 were brachycephalic. Gross lesions (8 cases) consisted mainly of parenchymal swelling. Histologically, of the 24 cases, there was widespread infiltration of neoplastic cells with astrocytic (12 cases), oligodendroglial (8 cases), or mixed morphology (4 cases) in the brain (18 cases), spinal cord (4 cases), or both (2 cases). Secondary structures occurred across different tumor grades and were not restricted to high-grade neoplasms. Astrocytic neoplasms had moderate nuclear immunolabeling for Olig2 and robust cytoplasmic immunolabeling for GFAP. Oligodendroglial neoplasms had robust nuclear immunolabeling for Olig2, moderate or absent cytoplasmic immunolabeling for GFAP, and moderate cytoplasmic immunolabeling for CNPase. Tumors with mixed morphology had robust nuclear immunolabeling for Olig2 and variable cytoplasmic immunolabeling for GFAP and CNPase. Morphologic and immunohistochemical features confirmed a glial histogenesis for all tumors and allowed for their classification as diffuse, low- or high-grade astrocytoma; oligodendroglioma; or undefined glioma. Further research is needed to confirm or refute the hypothesis that canine GC represents an infiltrative growth pattern of canine glioma.
Glioma is the most common primary central nervous system (CNS) neoplasm of humans and one of the most frequently diagnosed primary CNS tumors of dogs and cats. 10,21,29 –31,35,38,39 Gliomatosis cerebri (GC) is a rare glioma subtype in which neoplastic cells diffusely infiltrate the neuroparenchyma without distorting its overall architecture. 28,32 The term GC was first used in human neuropathology in 1938 to describe CNS neoplasms that had widespread infiltration of the telencephalic hemispheres. 25 Over the subsequent decades, the World Health Organization (WHO) Classification of Tumors of the Central Nervous System conventionally defined GC as a diffusely infiltrative neuroepithelial neoplasm with widespread involvement of more than 2 cerebral lobes with or without involvement of deep gray matter structures (such as basal nuclei and thalamus), brainstem, cerebellum, and spinal cord. 28,32,37 Cases with exclusive involvement of deep gray matter structures, brainstem, cerebellum, and/or spinal cord and that had no cerebral lobe involvement were also classified as GC. 19 Based on this previous WHO classification system, most GC cases are classified as a grade 2 or 3 glioma and occur as primary or secondary GC. Primary GC arises de novo and can be further classified as type 1 (diffuse neoplastic infiltration with no distinct tumor mass) or type 2 (diffuse infiltration with an associated tumor mass). Secondary GC is characterized by diffuse spread of neoplastic cells arising from a primary glioma. 28,32 This classification system was based mainly on morphology and lacked more strict criteria for the differentiation between GC and diffusely infiltrating gliomas, resulting in an overlap in diagnosis and little progress in terms of definition, biology, and treatment of this controversial entity. 28
The increased understanding of the molecular basis of human cancer has greatly contributed to the diagnosis, classification, and prognosis of many types of neoplasms, including those of the CNS. 17 The rare frequency, the diagnostic fluidity, and the lack of distinct genetic features unique to human GC still hinder the full understanding of its tumor biology and behavior, but a great proportion of cases have been shown to share key molecular features known to occur in astrocytomas and oligodendrogliomas, shedding light on the glial origin of human GC. 20,24,28 For example, a widespread growth pattern typical of diffuse glioma has been found in isocitrate dehydrogenase 1 (IDH)-mutant astrocytomas, IDH-mutant and 1p/19q co-deleted oligodendrogliomas, and IDH-wildtype glioblastomas. 5,11,18,28 Although the term is still present in the medical literature, such discoveries culminated in the elimination of GC from the 2016 WHO diagnostic lexicon. For these reasons, GC is now defined as a diffuse growth pattern of astrocytoma or oligodendroglioma and no longer as a distinct tumor entity. 18,28
In domestic animals, GC is exceedingly rare and almost exclusively described in dogs, 2,6 –9,13,22,26,27,29,33 with single case reports in 3 cats and a goat. 4,10,31,34 GC affects mainly middle-aged dogs with no sex or breed predisposition. A familial inheritance pattern has been suggested in a group of cases affecting families of bearded collie dogs. 12,33 An astrocytic origin is frequently assumed given the elongate cytoplasm and nuclei of the neoplastic cells. However, this morphology can also mimic a microglial origin, which led to initial descriptions of the condition to be referred to as microgliomatosis. 41 Research on the molecular and genetic basis of glioma in domestic animals is still limited, and thus the diagnosis of these neoplasms relies heavily on histopathology and immunohistochemistry (IHC), mainly oligodendrocyte lineage transcription factor 2 (Olig2) and glial fibrillary acidic protein (GFAP). 2,7 –9,13,22,27,40 Olig2 and GFAP are not specific immunomarkers for oligodendrocytes and astrocytes, respectively, but widespread nuclear Olig2 immunolabeling in a neoplasm with oligodendroglial morphology is considered supportive of a diagnosis of oligodendroglioma. Similarly, strong cytoplasmic immunolabeling for GFAP with variable nuclear immunolabeling for Olig2 is supportive of a diagnosis of astrocytoma in neoplasms with astrocytic morphology. 15 The increasing knowledge of the molecular basis of canine glioma may shed light on distinct growth patterns associated with specific morphological and IHC features of these neoplasms. 1 While a distinct infiltrative pattern of neoplastic glial cells across the neuroparenchyma typically suffices for a diagnosis of GC, the lack of clinical and molecular data precludes a better understanding of the biology and behavior of canine GC and whether it could represent a growth pattern of glioma, as reported in human medicine.
Herein we characterize the morphological and IHC features of 24 cases of canine GC and classify these glial neoplasms according to a recently proposed classification system for canine glioma. 15
Material and Methods
The electronic web-based archives at the Athens Veterinary Diagnostic Laboratory (Athens, GA); Department of Anatomic Pathology, Animal Medical Center (New York, NY); Department of Veterinary Pathobiology, Texas A&M University College of Veterinary Medicine & Biomedical Sciences (College Station, TX); Department of Microbiology, Immunology, and Pathology, Colorado State University College of Veterinary Medicine and Biomedical Sciences (Ft. Collins, CO); and Department of Biomedical Sciences, Section of Anatomic Pathology, Cornell University College of Veterinary Medicine (Ithaca, NY) were searched for cases of GC using the key words “dog,” “gliomatosis cerebri,” and “diffuse glioma.” Neoplasms were included in the study if there was widespread glial cell infiltration of more than 2 cerebral lobes with or without involvement of deep gray matter structures, or if there was exclusive involvement of deep gray matter structures, brainstem, cerebellum, and/or more than one segment of spinal cord. Retrieved cases were reviewed and morphologically evaluated based on a recently proposed classification system for canine glioma. 15 Each neoplasm was classified as low- or high-grade oligodendroglioma or astrocytoma according to its morphologic and IHC features. Tumors with a mixed morphology were classified as low- or high-grade undefined gliomas. Archived paraffin-embedded tissue sections were immunolabeled for Olig2 (rabbit monoclonal, 1:400 dilution at 60 minutes, GeneTex, GTX62440), GFAP (mouse monoclonal, 1:4000 dilution at 60 minutes, Biogen, MU020-UC), and 2′,3′-cyclic-nucleotide 3′-phosphodiesterase (CNPase; mouse monoclonal, 1:2000 dilution at 15 minutes, Abcam, AB6319) for diagnostic confirmation. The IHC for Olig2 and GFAP was performed on an automated stainer (Nemesis 3600, BioCare Medical). Antigen retrieval was performed using Antigen Retrieval Citra Solution 10X (BioGenex) at a dilution of 1:10 for 15 minutes at 110 °C. A biotinylated rabbit (Olig2) and mouse (GFAP) antibody (Vector Laboratories) was used to detect the target, and the immunoreaction was visualized using 3,3-diaminobenzidine (DAB; BioCare Medical) substrate for 12 minutes counterstained with hematoxylin. The IHC for CNPase was also performed on an automated stainer (BOND Max, Leica Biosystems). Antigen retrieval was conducted using bond epitope retrieval solution 1 (Leica Biosystems) for 30 minutes. Target detection was performed using PV-AP-mouse monoclonal antibody (Leica Biosystems) for 10 minutes, and the immunoreaction was visualized using Leica Bond Polymer Refine detection for 10 minutes counterstained with hematoxylin. Positive control tissue consisted of canine brain. The overall immunolabeling of tumor area in each case was scored as 0 (no immunolabeling), 1 (<25% immunolabeling), 2 (25% to 75% immunolabeling), or 3 (>75% immunolabeling). Subsequently, a cumulative and mean value was generated from these IHC scores for each tumor category. 15
Results
Twenty-four cases were included in this study (Table 1). All affected dogs were adults between 2 and 16 years of age (mean age 7.6 years). Nine dogs were brachycephalic. Neurological signs varied according to the localization of the lesions. Dogs were euthanized (21 cases) or died (3 cases) because of the progressive nature of the neurological signs and/or the diagnosis of a CNS tumor. Gross lesions (Table 2) were reported in 8 cases and consisted mainly of subtle neuroparenchymal swelling (6 cases) or cavitation (1 case) within the affected areas (Fig. 1). Secondary changes included cerebellar or subtentorial herniation (2 cases) and hydrocephalus (1 case). A distinct mass was present in the cerebellar white matter in one case. The most common neuroanatomic localization of the neoplasms was brain (18 cases) followed by spinal cord (4 cases) or brain and spinal cord (2 cases).
Clinical and Gross Pathologic Findings in 24 Dogs With Gliomatosis Cerebri.
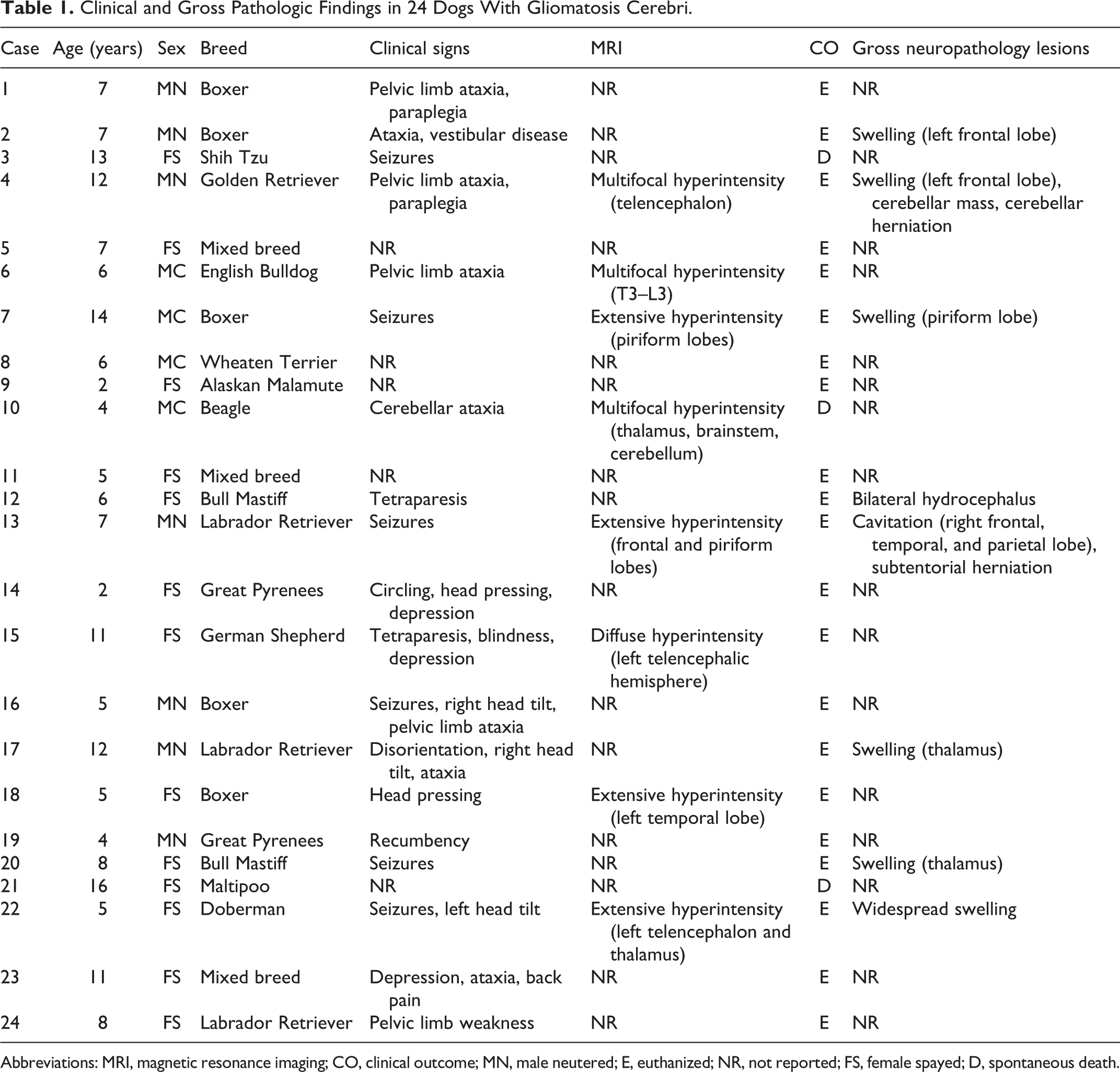
Abbreviations: MRI, magnetic resonance imaging; CO, clinical outcome; MN, male neutered; E, euthanized; NR, not reported; FS, female spayed; D, spontaneous death.
Histologic Tumor Neurolocalization of Gliomatosis Cerebri in 24 Dogs.
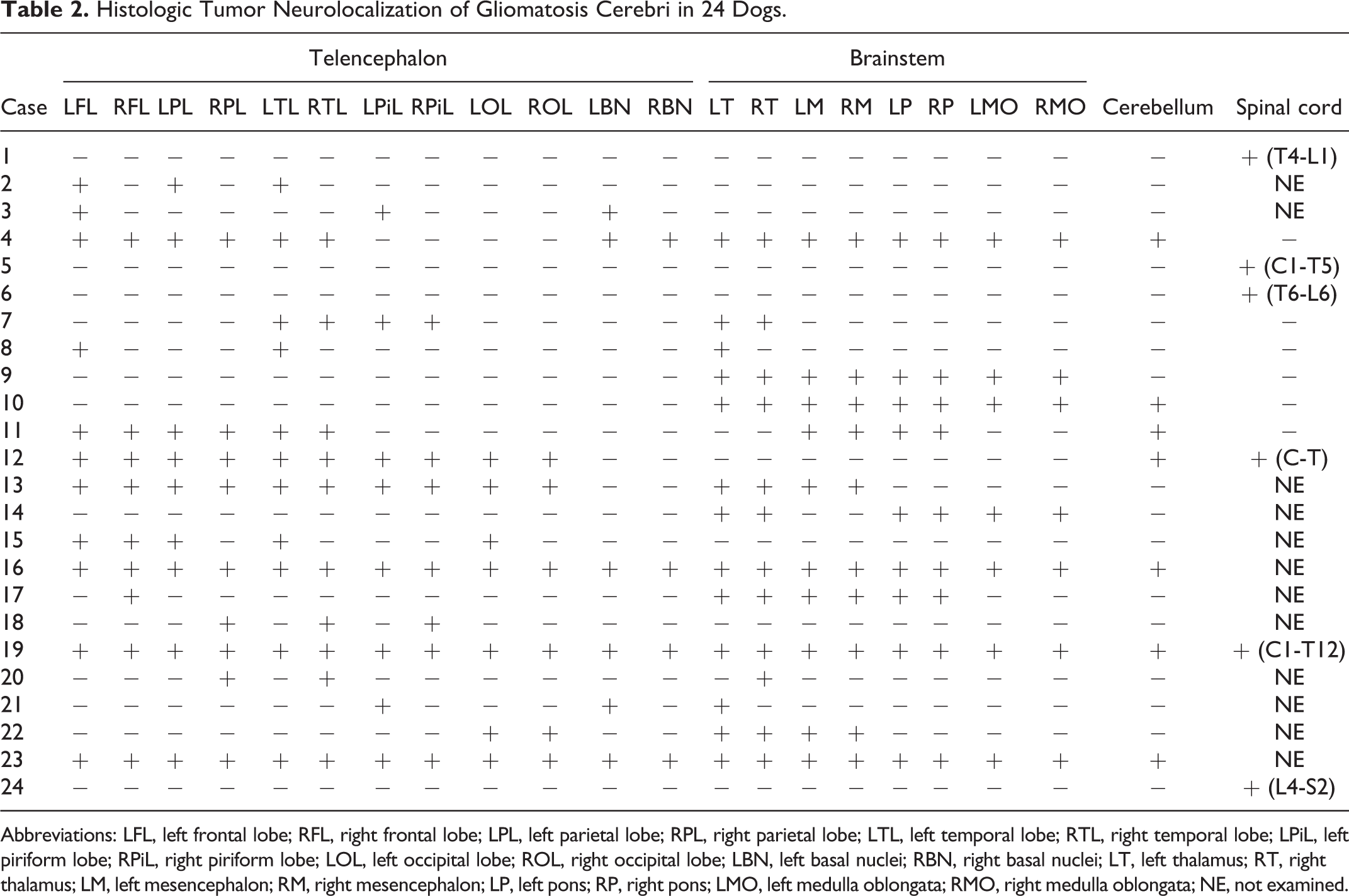
Abbreviations: LFL, left frontal lobe; RFL, right frontal lobe; LPL, left parietal lobe; RPL, right parietal lobe; LTL, left temporal lobe; RTL, right temporal lobe; LPiL, left piriform lobe; RPiL, right piriform lobe; LOL, left occipital lobe; ROL, right occipital lobe; LBN, left basal nuclei; RBN, right basal nuclei; LT, left thalamus; RT, right thalamus; LM, left mesencephalon; RM, right mesencephalon; LP, left pons; RP, right pons; LMO, left medulla oblongata; RMO, right medulla oblongata; NE, not examined.
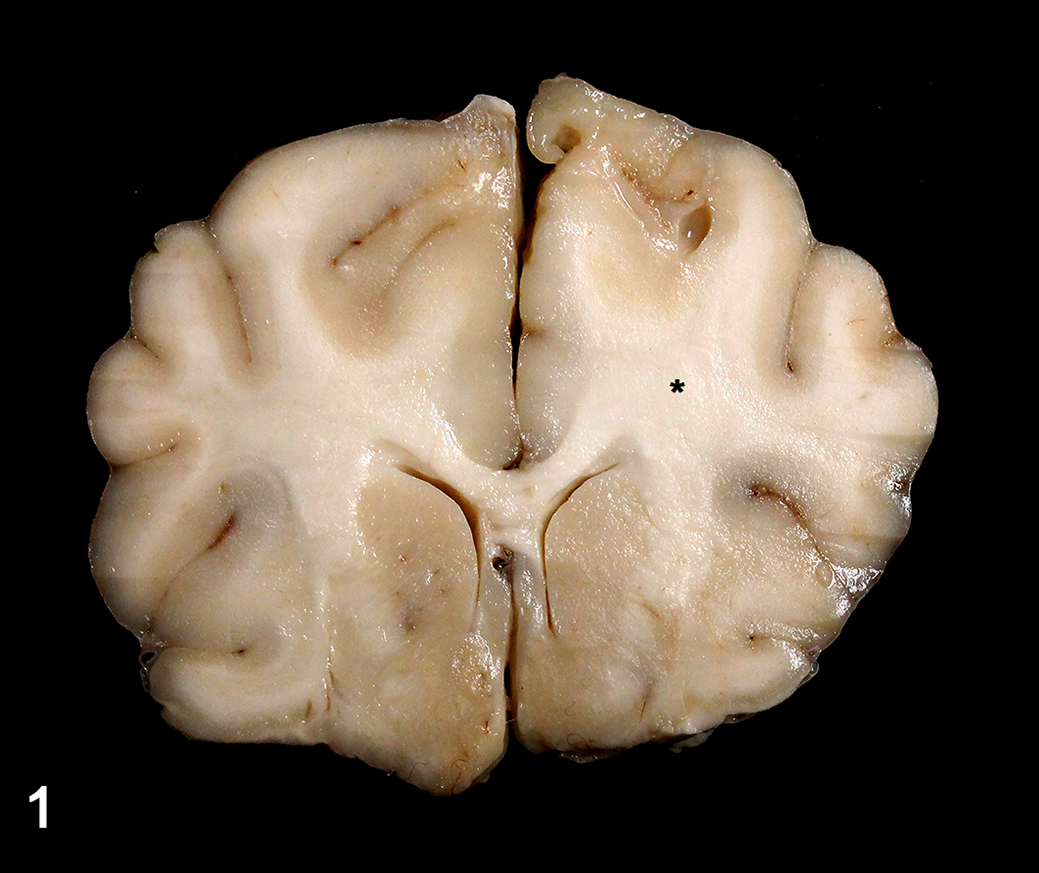
Diffuse glioma, brain, dog, case 4. There is subtle swelling of the left corona radiata and centrum semiovale (asterisk) with no overall disruption of the adjacent neuroparenchyma.
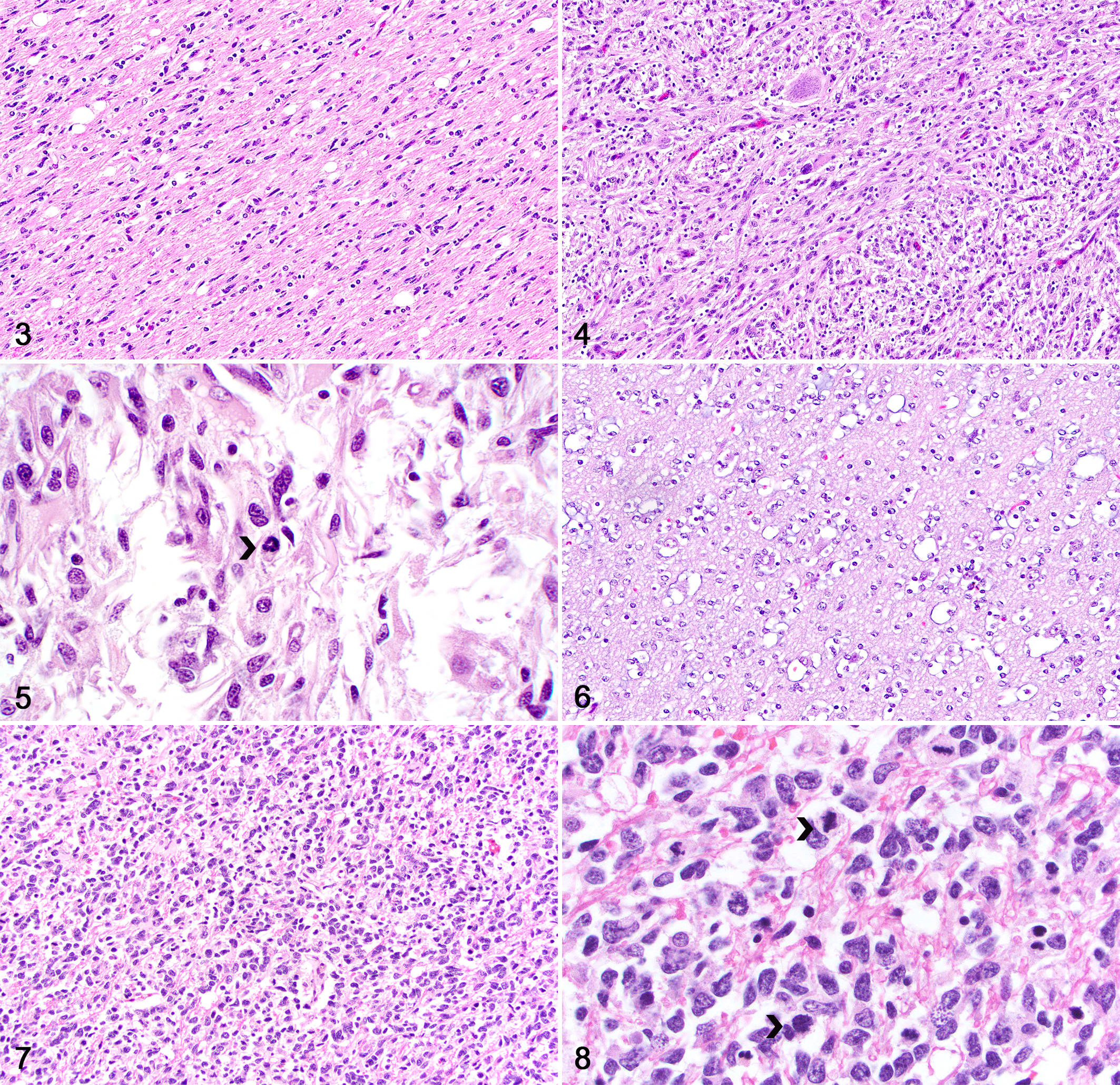
Histologically, all cases had neoplastic glial cell infiltration expanding the white and gray matter and causing no disruption of the adjacent neuroparenchyma (Fig. 2). Neoplasms with astrocytic morphology (12 cases) were characterized by neoplastic glial cells arranged in poorly defined streams and bundles supported by an eosinophilic, fibrillar, often neuropil-like stroma. Low-grade astrocytic tumors (6 cases) were sparsely cellular, with low cellular and nuclear pleomorphism (Fig. 3). Neoplastic cells were elongate and had scant, eosinophilic, fibrillar cytoplasm. Nuclei were oval to elongate and had dense to finely stippled chromatin with 1 to 3 nucleoli. Mitoses were absent. High-grade astrocytic tumors (6 cases) were densely cellular with increased cellular and nuclear pleomorphism (Fig. 4). Neoplastic cells were elongate and had a small to moderate amount of eosinophilic cytoplasm. Nuclei were large and irregular, with coarse chromatin and 1 to 3 nucleoli. The mitotic count ranged from 4 to 9 per 2.37 mm2 (equivalent to 10 FN22/400X fields; Fig. 5).
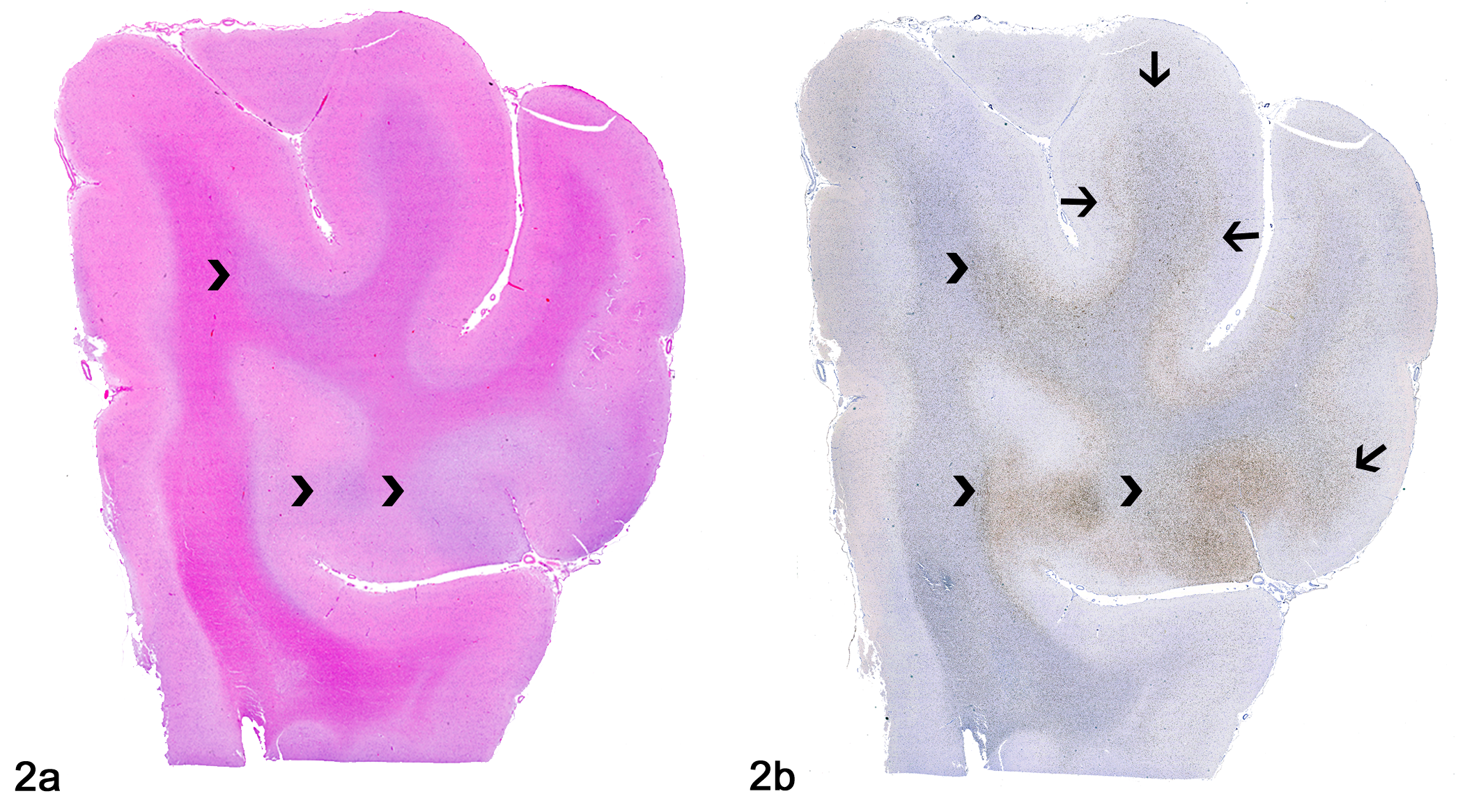
Diffuse glioma, brain, dog, case 2. (A) Neoplastic glial cell infiltration consistent with a diffuse glioma expanding the gray and white matter (arrowheads). Hematoxylin and eosin. (B) Olig2 immunolabeling highlights the neoplastic glial cells shown in A (arrowheads) and reveals additional widespread infiltration dorsally (arrows).
Diffuse glioma, brain, dog.
Neoplasms with oligodendroglial morphology (8 cases) consisted of neoplastic cells arranged in sheets supported by preexisting neuroparenchyma. Low-grade tumors (6 cases) were sparsely cellular with low cellular and nuclear pleomorphism (Fig. 6). The tumor stroma had a rich mucinous background and a network of branching small capillaries (chicken-wire pattern). Neoplastic cells were round and had scant, usually retracted cytoplasm (fried-egg or honeycomb appearance). Nuclei were round and had dense to finely stippled chromatin with no evident nucleoli or finely stippled chromatin and 1 to 2 nucleoli. Nuclear rowing was frequent. Mitoses were absent. Two high-grade oligodendroglial tumors (Fig. 7) were densely cellular and consisted of neoplastic cells with moderate cellular and nuclear pleomorphism. The cytoplasm was scant and retracted and nuclei were large and irregular, with coarse chromatin and 1 to 3 nucleoli. The mitotic count was 10 per 2.37 mm2 (Fig. 8) in one case; mitoses were not seen in the second case but areas of microvascular proliferation were present.
Four cases consisted of a mixed population of neoplastic cells with astrocytic and oligodendroglial morphology (30% to 40% each) and were classified as low-grade (Fig. 9) or high-grade (Fig. 10) undefined gliomas (2 low-grade and 2 high-grade tumors) based on degree of cellularity, cellular and nuclear pleomorphism, and mitotic count.
Diffuse glioma, brain, dog.
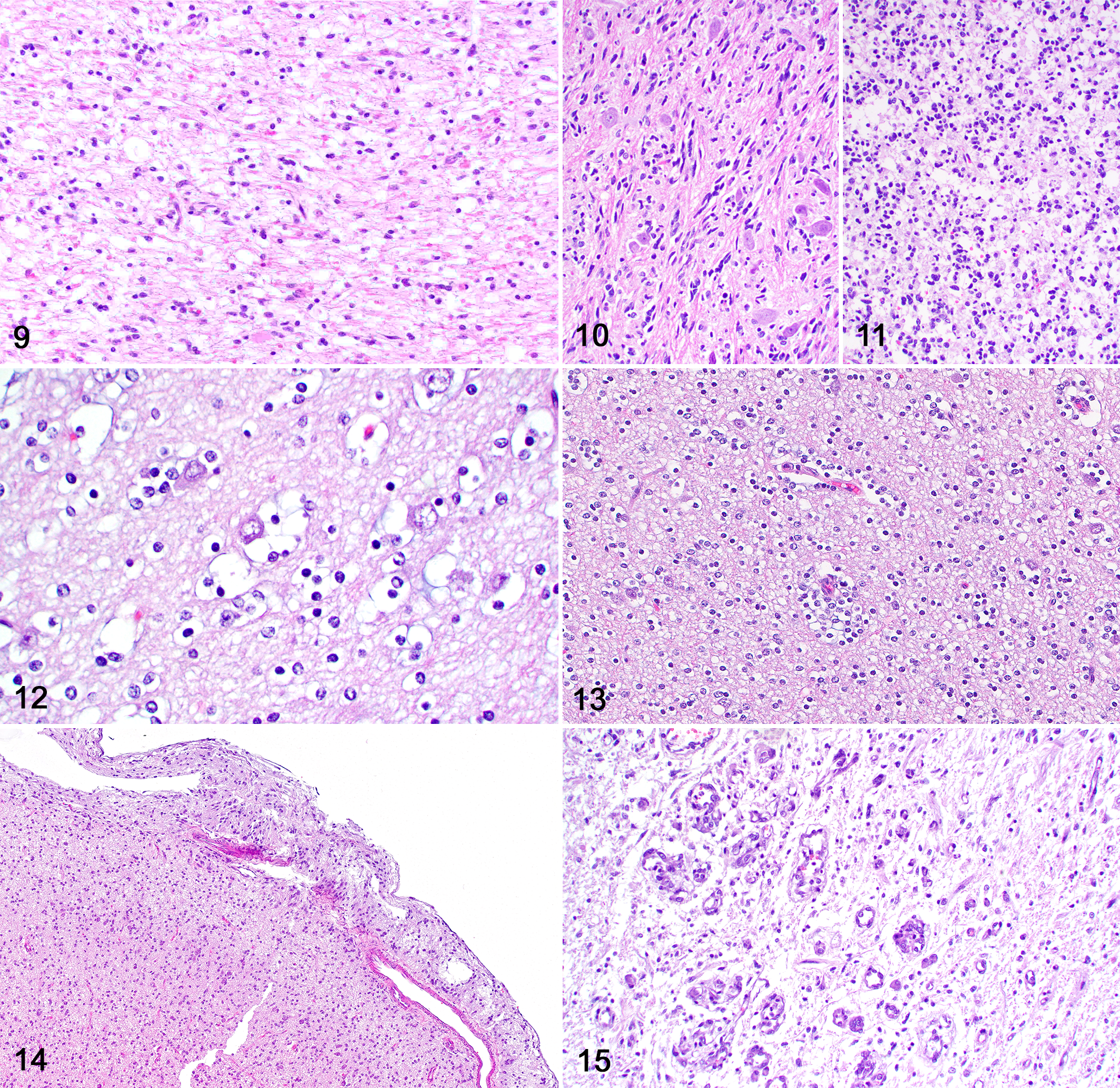
Regardless of the overall cell morphology and tumor grade, secondary structures were present in 16 neoplasms, including perineuronal (Fig. 12) or perivascular (Fig. 13) clusters of neoplastic cells and leptomeningeal infiltration by neoplastic cells (Fig. 14). One high-grade oligodendroglial neoplasm (case 20) had areas of microvascular proliferation (Fig. 15) and one high-grade undefined glioma (case 16) had areas of necrosis. A main tumor mass consistent with a high-grade astrocytoma with neuronal differentiation was present in the cerebellar white matter in one dog (case 4).
Immunohistochemically (Table 3), neoplasms with astrocytic morphology exhibited moderate nuclear immunolabeling for Olig2 (Figs. 16, 17) and robust cytoplasmic immunolabeling for GFAP (Figs. 18–21). The mean IHC values for Olig2 and GFAP in astrocytic tumors were 2.1 and 2.9, respectively. CNPase immunolabeling was negative in all tumors with astrocytic morphology. Neoplasms with oligodendroglial morphology had robust nuclear immunolabeling for Olig2 (Figs. 22, 23) and patchy or absent cytoplasmic immunolabeling for GFAP. The mean IHC values for Olig2 and GFAP in oligodendroglial tumors were 2.8 and 0.5, respectively. Cytoplasmic CNPase immunolabeling was moderate in neoplasms with oligodendroglial morphology (Figs. 24, 25), with a mean IHC value of 1.1. Undefined gliomas (Figs. 26, 27) exhibited robust nuclear immunolabeling for Olig2 (mean IHC value of 2) and variable cytoplasmic immunolabeling for GFAP (mean IHC value of 1) and CNPase (mean IHC value of 0.5).
Morphological and Immunohistochemistry Features of Gliomatosis Cerebri in 24 Dogs.
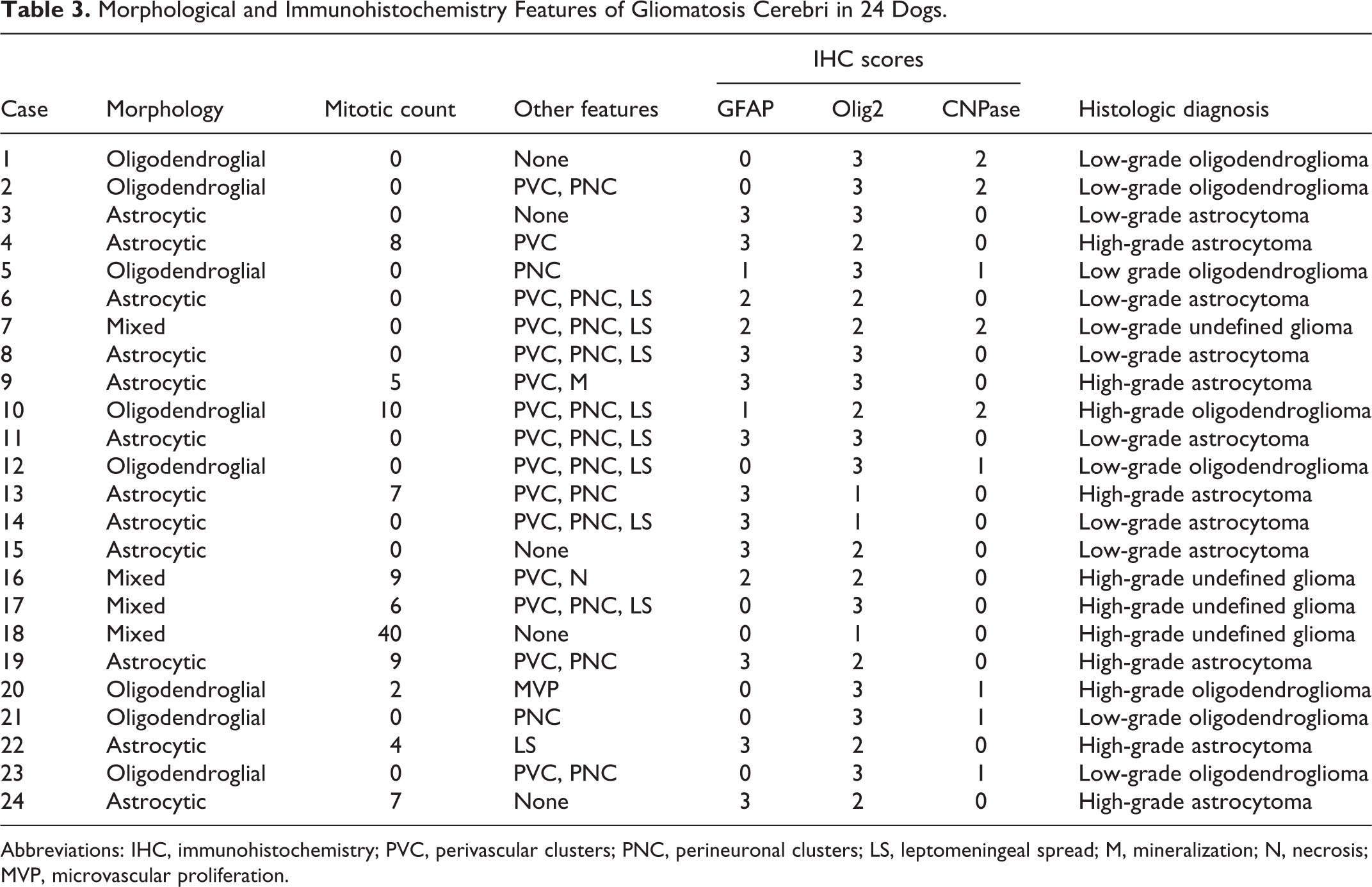
Abbreviations: IHC, immunohistochemistry; PVC, perivascular clusters; PNC, perineuronal clusters; LS, leptomeningeal spread; M, mineralization; N, necrosis; MVP, microvascular proliferation.
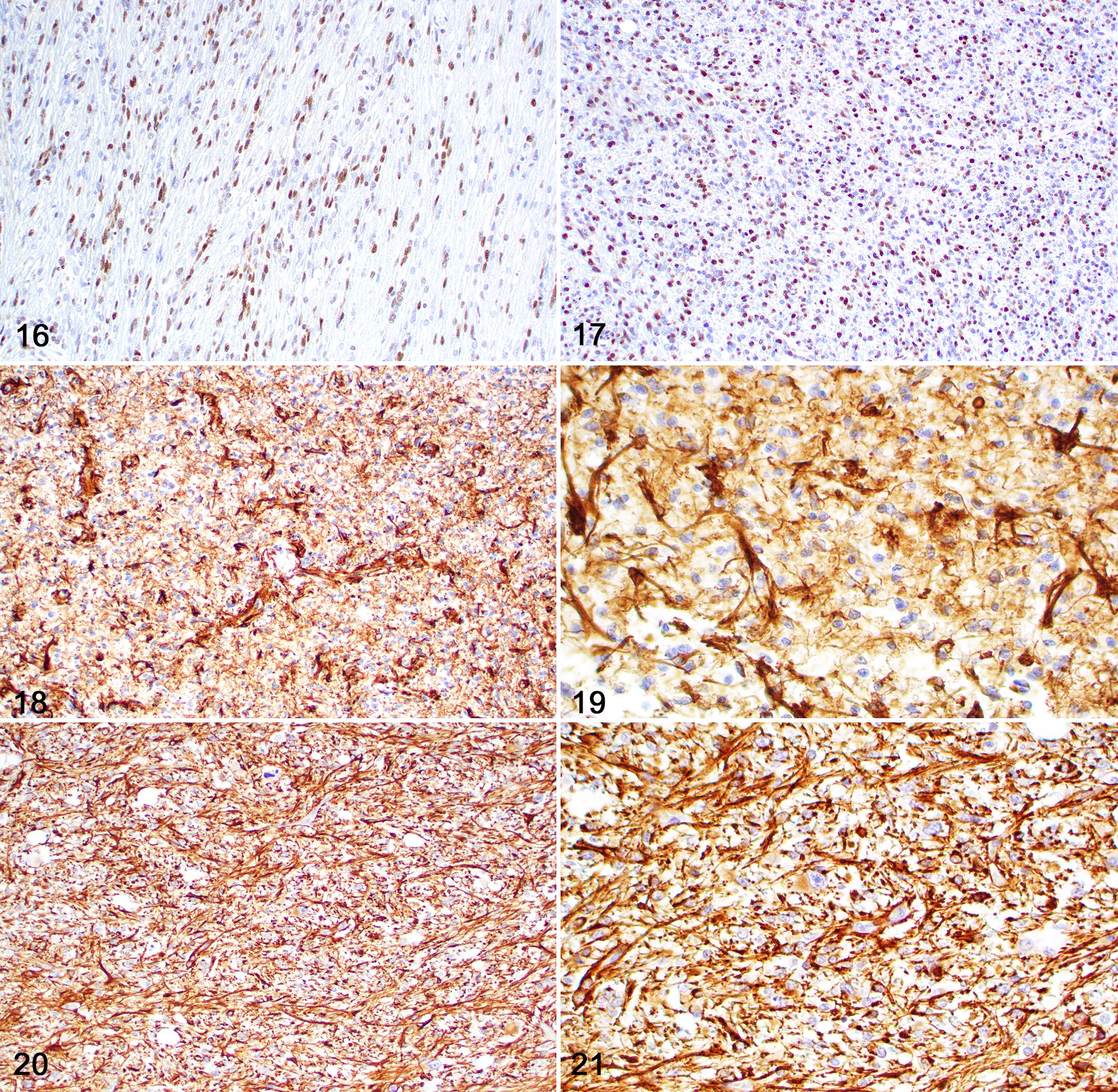
Diffuse glioma, brain, dog. Immunohistochemistry.
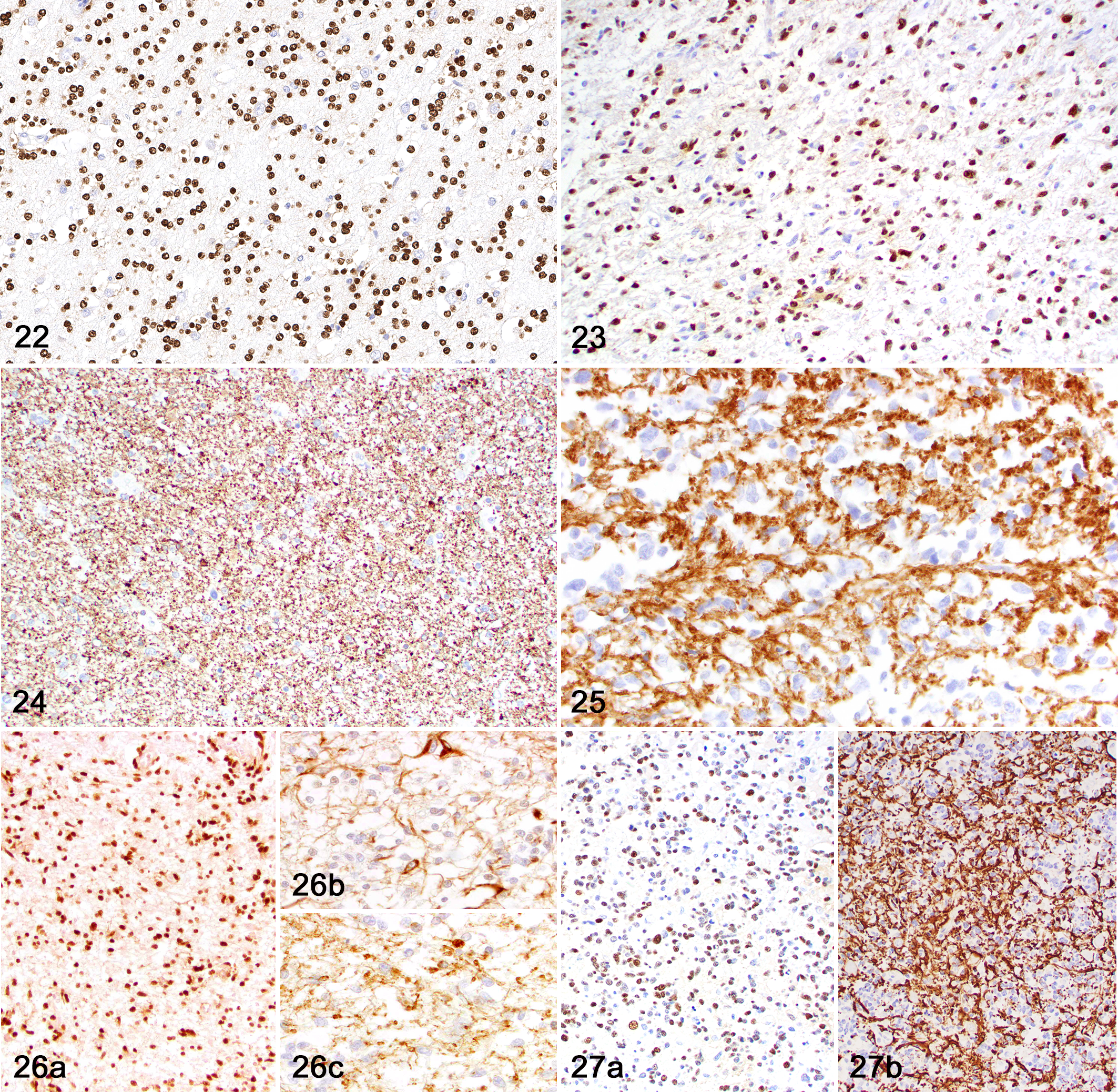
Diffuse glioma, brain, dog.
Discussion
All cases in this study were originally diagnosed as GC based on the widespread neoplastic glial cell infiltration within the gray and white matter of multiple lobes of the brain, brainstem, cerebellum, and/or spinal cord without disruption of the adjacent neuroparenchymal architecture. 2,27 Affected dogs were all adults and 9 were brachycephalic, similar to what has been widely reported for canine glioma. 3,15,23,36 Oligodendrogliomas are more frequent in brachycephalic breeds, especially Boston terriers, Bulldogs, and Boxers. 15 In the current study, 50% of dogs with oligodendroglial tumors were brachycephalic, corroborating these findings. Gross anatomic lesions in the CNS consisted mainly of local or widespread neuroparenchymal swelling. The absence of an associated tumor mass was consistent with type 1 GC in 23 cases, whereas a distinct cerebellar astrocytoma resulted in the diagnosis of type 2 GC in one case. 28,32 These findings reflect previous reports of GC in dogs, which are characterized mainly by CNS swelling, 2,8,9,22,27,33,40 with rare cases displaying a discrete tumor mass. 2,27,33
The lack of consensus in terms of definition, morphology, molecular profile, and treatment of human GC has culminated in its elimination from the 2016 WHO CNS tumor classification system. Currently, GC is no longer considered a distinct tumor entity in humans, but rather a type of growth pattern that is rarely found in gliomas. 5,11,18,28 Although a similar lack of agreement in terms of definition and morphology also exists in veterinary medicine, GC still stands as a rare tumor entity of undetermined or conflicting histogenesis. 2,7 –9,22,26,27,33
All neoplasms in the current study and many of those reported in the veterinary medical literature displayed the characteristic widespread invasion pattern and key morphological features described in human gliomas formerly classified as GC. 7 –9,18,22,26,27,29 A diagnosis of diffuse glioma in humans is routinely achieved by magnetic resonance imaging (MRI) and further confirmed by tumor morphology and molecular analysis. 20,28,32 The lack of MRI data in the majority of the cases in our study hindered a detailed assessment of this diagnostic tool in our case series. The main factors limiting our understanding of canine GC and other types of glioma are the lack of information on clinical outcome and the insufficient data regarding the molecular basis of these tumors. 1,16 The vast majority of dogs are euthanized soon after a diagnosis is made, which precludes a detailed assessment of the progression of clinical signs and the efficacy of specific therapeutic protocols in each tumor group. 2,7 –9,15,22,26,27 In humans, diffuse gliomas with GC growth pattern affect mainly adults and cause insidious clinical signs that are highly dependent on the location of the lesion. 20 Despite aggressive treatment, including surgery, radiation therapy, and chemotherapy, these neoplasms have a poor prognosis because of their clinical, morphological, and molecular heterogeneity, which hinders attempts to assess the efficacy of specific therapies. 28 In contrast, tumor diagnosis in canine patients relies heavily on morphology and IHC, and the possible clinical differences, tumor behavior patterns, and genetic alterations associated with each tumor type or grade remain widely unknown. 2,13 –15,27,29
Based on a simplified classification system for canine glioma, all cases in our study were tentatively classified as gliomas with astrocytic (12 cases) or oligodendroglial (8 cases) cell morphology. 15 Furthermore, the mixed population of neoplastic cells in 4 cases was consistent with a diagnosis of undefined glioma. 15 These findings are similar to those of human diffuse gliomas with GC growth pattern, which exhibit mainly astrocytic and less often oligodendroglial or mixed cell differentiation. 20 Neoplastic cell infiltration in our cases occurred in multiple areas of the telencephalon, brainstem, and cerebellum, or across multiple segments of the spinal cord. Neoplastic cells infiltrated the gray and/or the white matter, as reported elsewhere. 2,8,9,22,33,37,40 Other findings common to canine glioma (including GC), such as microvascular proliferation and necrosis, 27 leptomeningeal spread, 2,7,27 and perineuronal, perivascular, or periventricular clustering of neoplastic cells (secondary structures) 2,8,27 were found in the current cases. Of these features, only microvascular proliferation and necrosis have been associated with high-grade gliomas in a revised diagnostic classification scheme of canine glioma. 15 Interestingly, microvascular proliferation and necrosis were absent in most of the diffuse gliomas in this study and the majority of high-grade neoplasms were diagnosed based on increased cellular and nuclear pleomorphism and mitotic count. These findings, associated with the diffuse infiltration across the neuroparenchyma, could indicate that these tumors may have intrinsic molecular features that are distinct from those in other glioma subtypes. 1 Contrary to what has been described for canine gliomas under the previous WHO grading scheme, 41 secondary structures occurred across different tumor grades in the current study and were not restricted to higher grade diffuse gliomas. 15
The lack of a systematic IHC assessment of GC in the veterinary medical literature has likely contributed to inconsistencies in the reported immunolabeling patterns in these neoplasms. 2,13 –15,27,29 However, the widespread availability of specific antibodies has allowed for further characterization and diagnostic guidance. 14 Although subsets of GC have been thought to be of astrocytic origin based on morphology and immunopositivity for GFAP, 7,13 other cases have had an oligodendroglial profile or inconsistent morphological and IHC features. 2,8,9,13,22,26,27
Olig2 and GFAP are not specific immunomarkers for oligodendrocytes and astrocytes, respectively. 14 However, a robust and widespread nuclear immunolabeling with Olig2 is highly consistent with a glial histogenesis, as observed in all cases in our study. 15 Furthermore, as previously demonstrated in canine and feline glioma, neoplasms with oligodendroglial morphology in the current study had stronger and widespread nuclear Olig2 immunolabeling associated with a less consistent or absent immunolabeling with GFAP. 14,15,29,31 In contrast, robust cytoplasmic GFAP immunolabeling was evident and more consistent in neoplasms with astrocytic morphology or in those classified as undefined glioma, a feature that is also shared with other canine gliomas. 14,15 CNPase can be used to confirm oligodendroglial differentiation in cases in which morphology associated with Olig2 and GFAP immunostaining is inconclusive. 15 However, as seen in our case series and other studies, CNPase immunostaining tends to be inconsistent and less reliable in autopsy cases because of its increased susceptibility to a delay in tissue fixation. 14,15 These results indicate that CNPase IHC could be a more reliable diagnostic tool for biopsy cases. 14,15 Our findings indicate that the diagnosis of canine GC can be achieved based on a systematic morphological evaluation of the neoplastic cells in combination with an IHC panel consisting of Olig2, GFAP, and CNPase, similar to what has been reported for other canine gliomas. 15 After morphological and IHC assessment of the current cases, all neoplasms had features that were consistent with a low- or high-grade astrocytoma, oligodendroglioma, or undefined glioma. 11,15,18
Our results confirm that canine GC shares key morphological and IHC features with other canine gliomas and can be subjected to the same classification system applied for canine glioma. Although these findings could suggest that GC represents a diffuse growth pattern found in subsets of canine glioma rather than a separate entity, as reported in human medicine, this hypothesis should be further confirmed or refuted by future research on the molecular aspects of canine glioma. 16
Footnotes
Acknowledgements
The authors thank Dr Mark Troxel of Massachusetts Veterinary Referral Hospital for submission of case 22.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
