Abstract
Nine Gelbvieh calves originating in four herds and clinically presenting with rear limb ataxia/ paresis had histopathologically confirmed peripheral neuropathy and a proliferative glomerulopathy. Degenerative lesions were severe in peripheral nerves, dorsal and ventral spinal nerve roots, and less marked in dorsal fasciculi of the spinal cord. Cell bodies of spinal ganglia were minimally diseased; ventral horn neurons occasionally had central chromatolysis and nuclear displacement. Glomerular lesions ranged from mild mesangial hypercellularity to glomerulosclerosis. Pedigree analysis of affected animals from one herd indicated a strong familial relationship and probable hereditary basis for the syndrome.
Introduction
There are several documented, early onset diseases of cattle characterized by dysfunction of the nervous system. Some are genetically determined, others are nutritionally caused, and yet others may result from infection, toxicosis, or metabolic aberration. Some of the more common genetically determined diseases include progressive degenerative myeloencephalopathy of Brown Swiss, 2,28 spinal muscle atrophy of Braunvieh, Brown Swiss, Holstein-Friesian and Red Danish, 1,10,30 and degenerative axonopathy of Holstein-Friesian. 14 In addition there are progressive spinal myelinopathy, 25 spinal dysmyelination of Brown Swiss × Braunvieh, 12 progressive ataxia of Charolais, 3,6 neuraxial edema of Hereford cattle, 7 a primary demyelination disorder in Limousin, 22 and a peripheral neuropathy in twin Holstein calves. 11 Inherited storage diseases include α and β mannosidoses, galactosidosis, and lipofuscinosis. 4,18,26 Toxins contained in Oxytropis spp. and Astragalus spp. also cause acquired “storage”-type lesions. 16 Gestational and neonatal deficiency of vitamin E, vitamin A, copper, or selenium may result in neurologic disease; 29 citrullinemia and maple syrup urine disease are inherited metabolic disorders whose manifestations include nervous signs. 13,15 Infectious agents capable of causing neuromuscular dysfunction in neonatal cattle include bovine viral diarrhea, bluetongue, Akabane, and Ibaraki viruses. 27
Glomerulonephritis has only occasionally been reported in cattle. One study identified nonclinical glomerulonephritis in 23 of 25 steers; 19 another associated glomerulonephritis in animals persistently infected with the virus of bovine virus diarrhea. 8 Three reports record clinical glomerulonephritis/glomerulopathy in single animals. 9,31,32
We have encountered a syndrome in young Gelbvieh cattle that clinically presents with ataxia and paresis, progresses to recumbency, and histopathologically is characterized by degenerative changes most prominent in peripheral nerves and spinal nerve roots and a glomerulopathy. A similar if not identical syndrome has recently been reported in young Gelbvieh cattle. 21 That syndrome was generally characterized as “a degenerative skeletal muscle disease with vascular, neurologic, and renal lesions and a probable familial distribution….” Myodegeneration was particularly emphasized.
Materials and Methods
The study uses data obtained from nine histopathologically confirmed cases of disease from three sources, A and B from Oklahoma and Source C from California. Five confirmed cases originated from Source A, a herd of 28 breeding-age Gelbvieh cows, one Gelbvieh bull and their immature progeny. Examination of Source A included survey of the premises and examination of live animals during two onsite visits and comprehensive in-hospital examination of two diseased calves. Necropsy of those and two additional animals, and histopathologic examination of abattoir specimens from a fifth animal of Source A were conducted. Source B contributed a single calf from a second Gelbvieh herd that consisted of 30 mature cows and their progeny. This solitary case was available for comprehensive clinical workup and necropsy. Source C included three young Gelbvieh animals that originated in two herds in northern California. Necropsy records and hematoxylin and eosin (HE)–stained slides were provided through the courtesy of the California Veterinary Diagnostic Laboratory, Davis, CA. The aggregate of histologically confirmed cases comprised four males and five females that ranged in age from 5 weeks to 13 months at the time of examination.
During the two visits to Source A the entire herd was observed, and 2 affected and 12 “normal” animals were subjected to limited physical examination. Specimens were collected for routine hematologic, chemical, and urine assay. The premises of Source A were extensively surveyed and samples collected as follows: pond water for total salt, sulfate and nitrate analysis; forage and soil for copper and selenium assay and suspected petroleum-impregnated material for hydrocarbon and organic phosphate assay. Pedigrees were available for four of the five histologically confirmed diseased animals as well as three suspect animals and healthy adult members of the existing herd (suspect calves are those reported by the owner to have been similarly diseased).
The aggregate of complete blood counts (CBC) and serum chemistries on specimens collected during herd visits as well as during hospitalization of animals from Sources A and B was a total of 20 CBCs, 5 serum chemistry panels and 12 urinalyses. Copper and selenium assay of serum, liver, or kidney was obtained on the 3 hospitalized animals, (two from Source A, one from Source B), and 12 normal adults from Source A. (Assays by Oklahoma Animal Disease Diagnostic Laboratory, Stillwater, OK). Serum vitamin E level was available from the calf of Source B. Assay of cerebrospinal fluid (CSF) and electromyograms (EMGs) were obtained from the three hospitalized animals.
Tissues available for histopathologic examination included specimens of major nerve trunks from fore and rear limbs (seven of nine animals), spinal cord (nine of nine animals), kidney (eight of nine animals), skeletal muscle (eight of nine animals), myocardium (seven of nine animals), and numerous other organs and tissues. Specimens were fixed in 10% neutral buffered formalin, embedded in paraffin, sectioned at 4–5μm, and routinely stained with HE. Selected nervous tissues were stained with Luxol fast blue, cresyl echt violet, and by the Sevier-Munger modification of the Bielschowsky method. Sections of kidney were stained with periodic acid-methenamine silver, Masson trichrome, periodic acid-Schiff, and phosphotungstic acid-hematoxylin (PTAH). 5 Glomerular diameters (12–20 glomeruli per animal) of three age-matched control cattle and the nine affected cattle were measured with a calibrated ocular micrometer at 200× magnification. Data are reported as mean glomerular diameter (μm) ± standard error of the means. Means for control and affected cattle were compared using a two-sample t-test. 23
Results
Examination of the premises of Source A revealed a 140-acre pasture that was extremely weedy. Water was available from four ponds. The source of food in the summer was primarily native grass pasture. In winter, prairie hay was provided as a supplement to pasture. Salt and other minerals were available year-round. Approximately 2 acres of the pasture were littered with oilfield equipment that had been present for about 10 years. There was evidence that cattle had been exposed to the content of a large (1,000 ± gal) tank containing petroleum-impregnated solid material.
The herd comprising Source A was constituted in 1984; at the time of study all female adults in the herd except five cows purchased from an outside source were descended from the original purchase group. All offspring of the foundation dams was the get of three sires. The bull in current use had sired three calf crops; all animals from Source A affected with the described syndrome were the offspring of this bull (B) and four cows (Fig. 1). The herd had been regularly vaccinated for blackleg, leptospirosis, and vibriosis; viral respiratory vaccines were used for the first time after the neurologic cases were originally observed. A regular internal and external parasite control program was practiced using ivermectin, albendazole, and diazinon-impregnated ear tags.
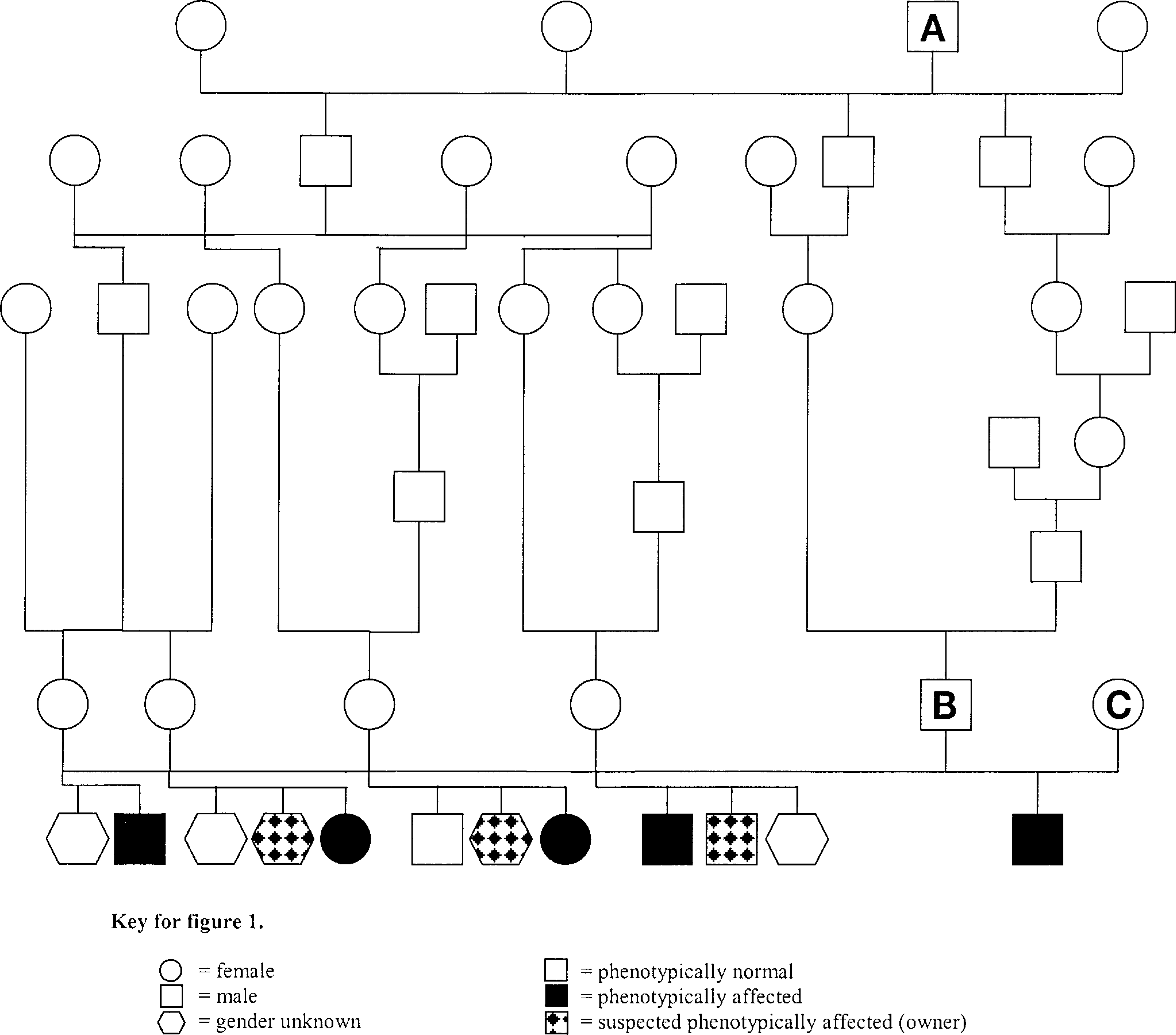
Pedigree of five confirmed affected, three suspect affected, and four phenotypically normal animals which are the progeny of one sire and four or five dams. The dam (C) of one of the five affected calves was not identified. The sire (B) of the affected and suspect calves is a descendant of one sire (A) on both sides of its pedigree. The same sire (A) appears in the pedigree of each of the four dams that produced the seven confirmed or suspect calves.
Presenting complaints in calves from each of the three sources consisted of rear limb ataxia progressing to generalized paresis and recumbency. The progression from onset to recumbency was reported to range from 3 days to several weeks. Affected animals were bright and alert and continued to eat and drink normally. Spinal reflexes were consistently hypo- to areflexic. Specific neurologic deficits included loss of conscious proprioception, diminished or absent patellar, triceps, flexor, panniculus and anal sphincter reflexes, and hypotonia. In two cases, analgesia, including loss of deep pain, was noted in all four limbs. The skeletal muscle mass of affected animals ranged from normal to mild or moderate atrophy. EMG evaluation of various muscle groups performed in the three hospitalized cases displayed decreased amplitude and number of motor units and sharp potentials. These findings were interpreted as suggestive of decreased muscle tone or denervation. Total CSF protein was elevated in the single sample collected from one of the three hospitalized animals. Cytologic interpretation of these samples ranged from no cytologic abnormalities to mild pleocytosis with mixed mononuclear cells. Cranial nerve deficit was not observed in any of the hospitalized animals. However, one otherwise healthy adult cow from Source A, the dam of one affected and one suspect calf, did have evidence of neurologic deficit. Her left cheek bulged due to impacted food material. Her tongue was hypotonic and tended to hang out of the mouth when pulled out. Nerve function in other adult animals of the herd appeared essentially normal. Blood counts were in normal ranges except for an inflammatory leukogram in one animal that had a severely ulcerative urinary cystitis. Serum creatine kinase was transiently elevated in three hospitalized, recumbent animals (421, 14,720, 104,000 IU); one was azotemic (blood urea nitrogen = 67 mg/dl, creatinine = 3.6 mg/dl). Three of the four animals from Source A that were subjected to necropsy had proteinuria of 2–4+ (sulfosalicylic acid); histologically, each had advanced glomerulopathy. Levels of liver copper in three animals were 12, 12, and 24 ppm; hepatic selenium levels were 0.238, 0.188, and 0.275 ppm, both copper and selenium values considered marginal to adequate. 24 Serum levels of copper in 12 unaffected herd mates ranged 0.38 to 0.74 ppm. Eight of these specimens were in the deficient and four in the marginal range. 24 Blood selenium in the group of 12 unaffected cattle varied from 0.01 to 0.04 ppm, all deficient values. 24 Serum vitamin E, determined in one animal was 2.97 ppm, an adequate level. 24 Levels of copper and selenium in soil and vegetation were considered normal for Oklahoma (copper 0.94 and 0.76 ppm, respectively; selenium each below detectable limits). Water samples contained total salts, nitrates, and sulfates that were far below hazardous levels. Samples of the suspected petroleum-impregnated material was a fuel oil–type compound and was negative for tri-ortho-cresyl-phosphate (TOCP) and tri-aryl-phosphate–type compounds.
Analysis of the available pedigrees of 11 affected and suspect calves of Source A revealed that each of the calves was the progeny of four dams and a single bull (Fig. 1). Seven of the eleven calves born to these matings during three breeding seasons were either suspects or histopathologically confirmed cases. The pedigree of an additional confirmed case of the disease from Source A was not obtained. Analysis of available pedigrees indicates that the sire of each affected/suspect calf was a descendent of one bull and two cows over three to five generations and that the four dams were descended from the same bull and one cow over three generations (Fig. 1).
Gross lesions were not observed in the central or peripheral nervous systems of any of the nine animals histopathologically confirmed to be diseased. Four animals had diffusely pale renal cortices; three of which had disseminated cortical petechiae. There were focal areas of paleness and fibrosis in epaxial muscles of the cervical and lumbar regions of two animals and generalized paleness of skeletal muscle of two additional animals.
Microscopic lesions occurred consistently in peripheral nerves, the spinal cord, and the kidney; less frequently there were atrophy and focal necrotic lesions in skeletal muscle (Table 1). Degenerative changes without inflammation were present in peripheral nerves, dorsal and ventral spinal nerve roots, the dorsal root entry zone, the dorsal fasciculus, especially the dorsal fasciculus proprius along the course of entering afferent fibers and occasionally, the caudal brain stem. Spinal cord and spinal nerve roots extending from anterior cervical to sacral segments were affected. A single specimen of trigeminal nerve, the sole cranial nerve specimen examined, was severely diseased.
Distribution of histologic lesions and glomerular diameters among nine diseased and three age-matched control calves.∗
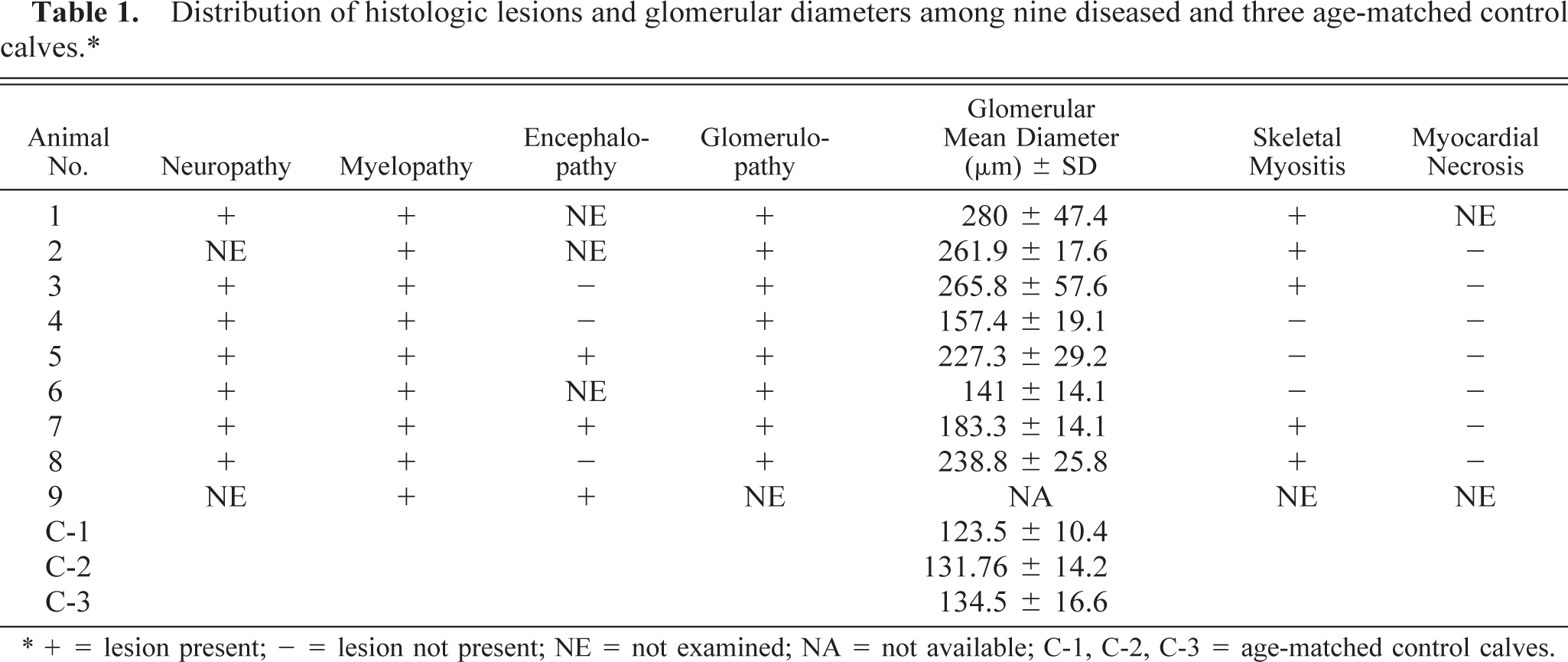
∗ + = lesion present; – = lesion not present; NE = not examined; NA = not available; C-1, C-2, C-3 = age-matched control calves.
Lesions in peripheral nerves occurred in each of the seven animals where specimens were available and were characterized by axonal swelling and loss and the presence of myelin ellipsoids and digestion chambers (Fig. 2). Lesions had a fascicular distribution; many fibers in some fascicles were prominently degenerate whereas few or no lesions might be present in adjacent fascicles. There was marked increase in numbers of Schwann cells associated with fiber degeneration. Similar degenerative changes were present in fibers of dorsal spinal nerve roots (Fig. 3) and the intramedullary segments of axons of dorsal root ganglion cell bodies. Intramedullary lesions were particularly concentrated at the dorsal root entry zone and the dorsal fasciculus proprius and were more sparsely scattered in other areas of the dorsal fasciculus (Fig. 4). Occasional neuron bodies in dorsal root ganglia had chromatolysis, but nuclear displacement was rare. A few neurons had large cytoplasmic vacuoles. Aberrations were not recognized in most neurons of the ganglion nor in the neuron bodies of the dorsal horns of the spinal cord.
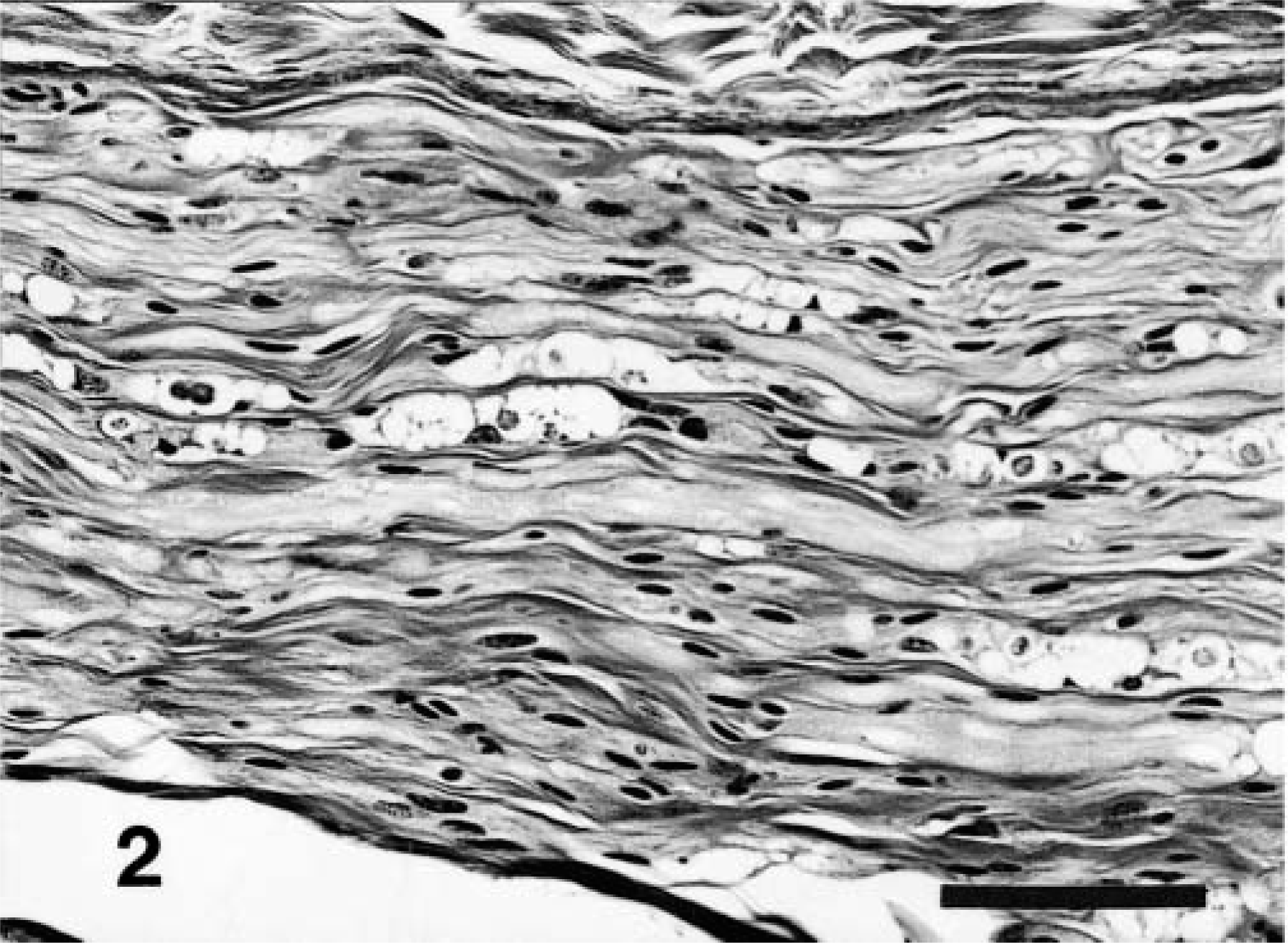
Sciatic nerve; 8-month-old calf, Source C. There are numerous swollen myelin sheaths and myelin ellipsoids often containing macrophages. HE. Bar = 100 μm.
Dorsal root of spinal nerve; 5-week-old calf, Source B. There are numerous degenerate fibers characterized by swollen myelin sheaths, myelin ellipsoids, macrophages, and numerous Schwann cells. HE. Bar = 200 μm.
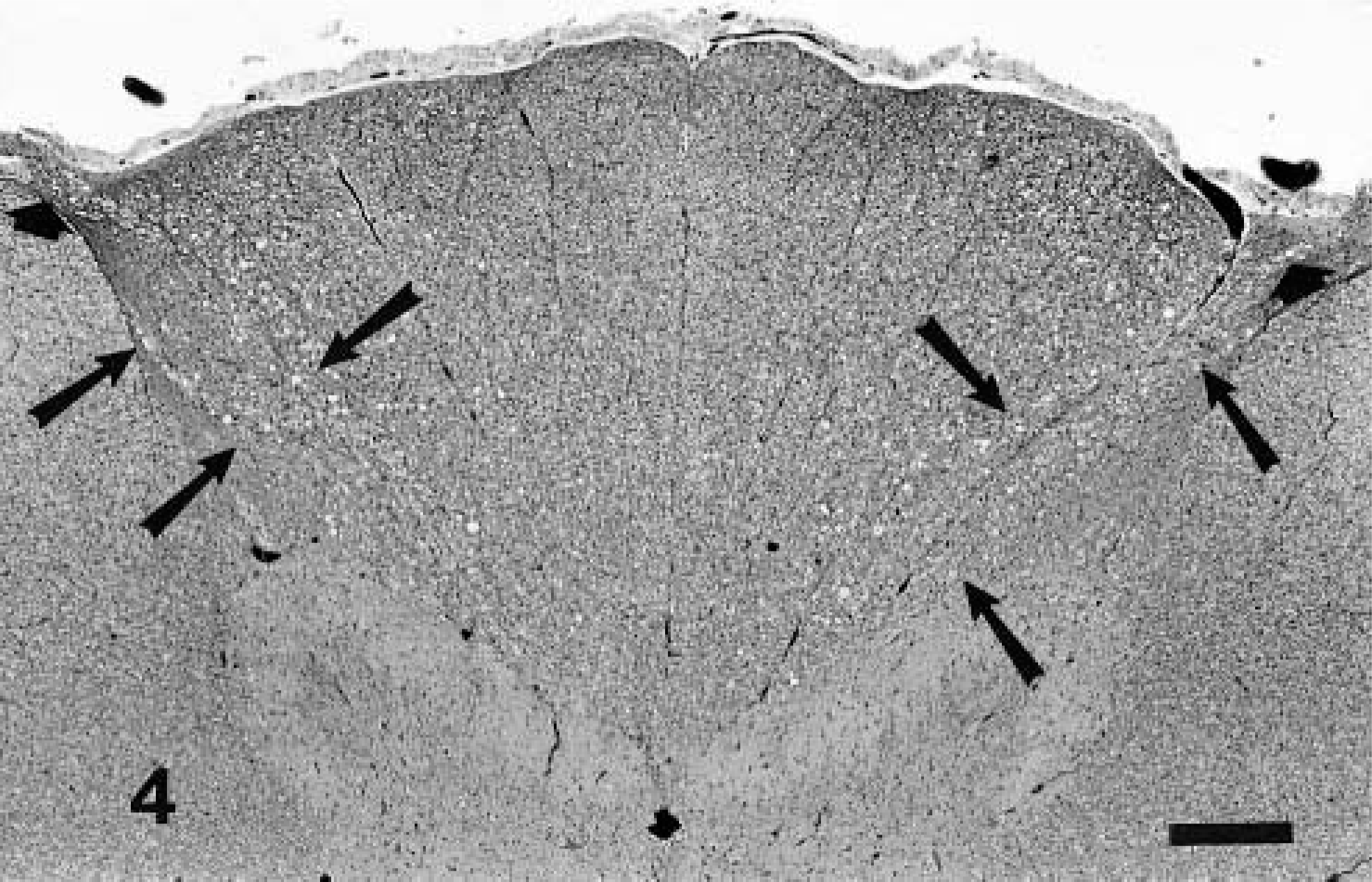
Spinal cord; 10-month-old calf, Source B. Vacuoles (degenerate fibers) in dorsal white matter are concentrated in root entry zone (short arrows) and dorsal fasciculus proprius (arrows). Fewer degenerate fibers are in fasciculus cuneatus and fasciculus gracilis. HE. Bar = 500 μm.
Degenerative changes in ventral spinal roots, although frequently present, seemed generally less intense than those in dorsal roots. Swollen axons in the intramedullary tracts of ventral roots were infrequent as was central chromatolysis of ventral horn cell bodies. Neither necrotic neurons nor glial scars were recognized in ventral horn gray matter. Lesions recognized in the brain included Wallerian-like degeneration of a few scattered fibers in the medulla oblongata, the pons, and subcortical white matter of the cerebellum. Lesions were not recognized in cerebral neurons.
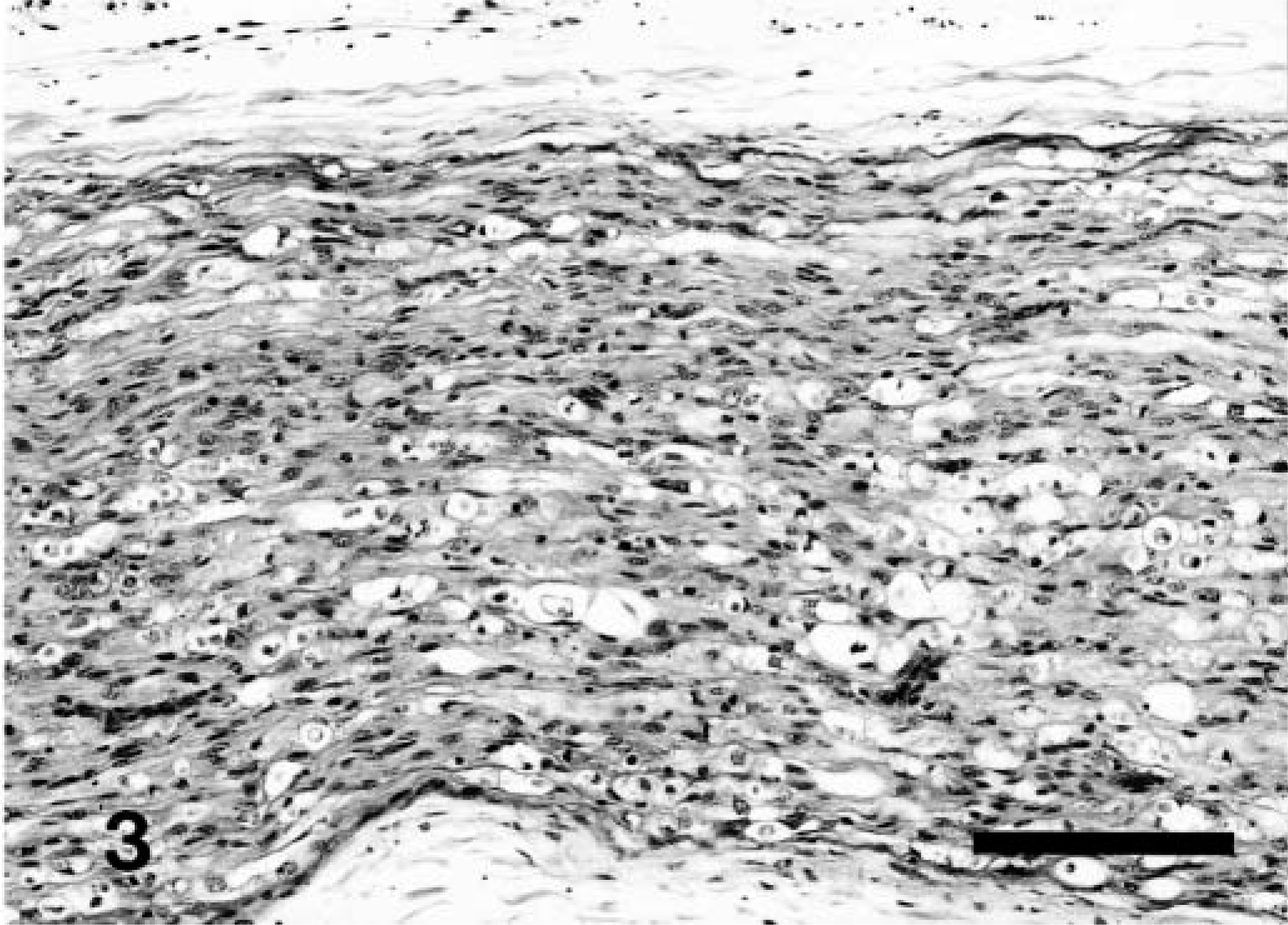
Histologic examination of the kidney revealed a glomerulopathy of variable intensity in each animal. The least severe lesion was characterized by mild glomerular hypercellularity associated with hypertrophy and hyperplasia of mesangial cells and minimal hypertrophy of visceral epithelial cells (Fig. 5). More advanced lesions included greater increase in size and number of mesangial cells, minimal to mild hypertrophy of visceral epithelium, a few scattered neutrophils in and excessive lobulation of tufts. In further advanced cases there were often thickening and fibrosis of Bowman's capsule. Epithelial cells lining Bowman's capsule were usually normal but had plump nuclei when periglomerular fibrosis was severe (Fig. 6). Mean glomerular diameter was significantly smaller (P = 0.01) for control cattle (129.9 ± 3.3 μm) than for affected cattle (208.8 ± 15.6 μm). In six of the nine affected calves, diameters were increased 47–126%.
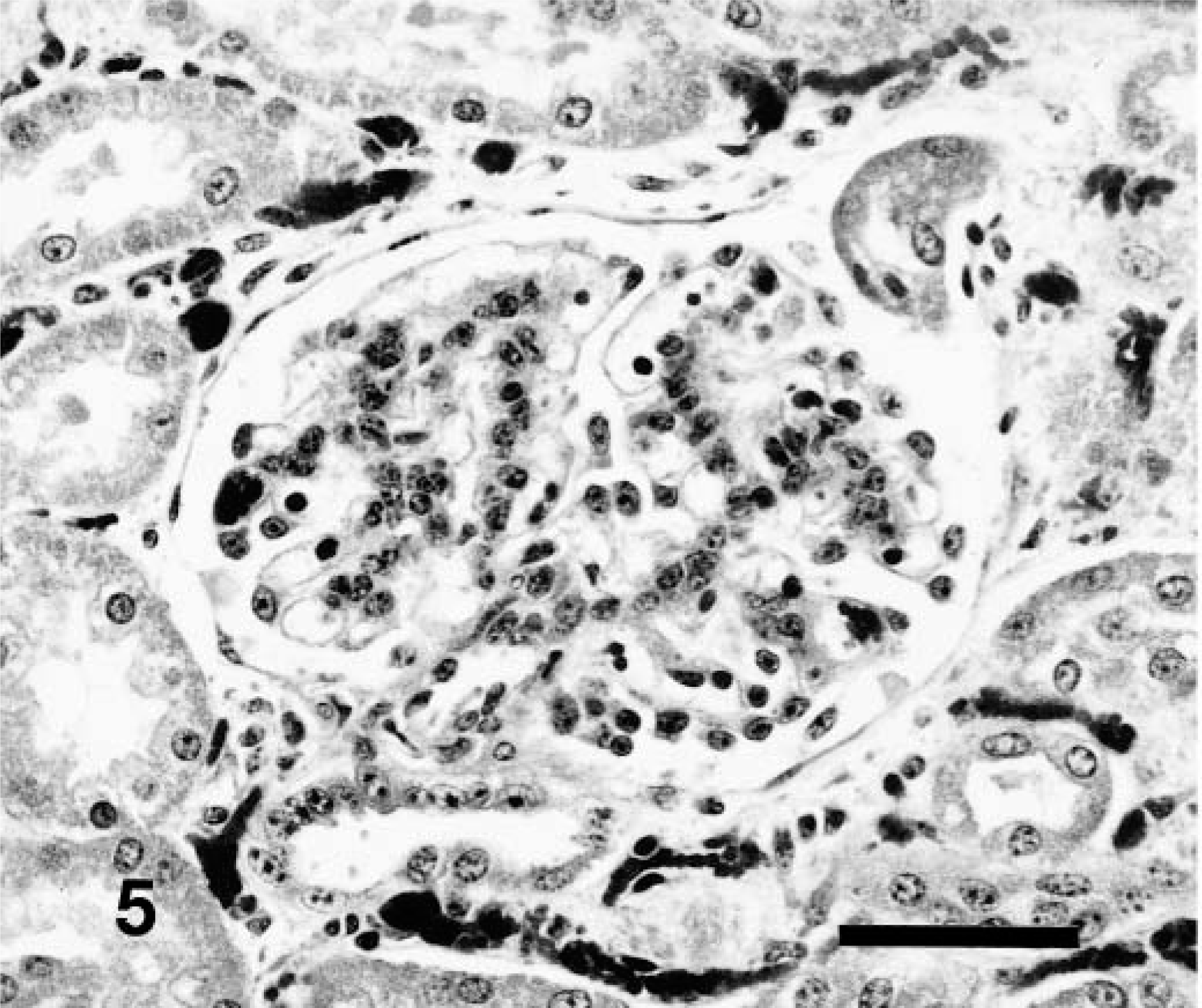
Kidney; 6-week-old calf, Source B. Early stage glomerulopathy characterized by hypertrophy and hyperplasia of mesangial cells and possibly swelling of visceral epithelial cells. HE. Bar = 50 μm.
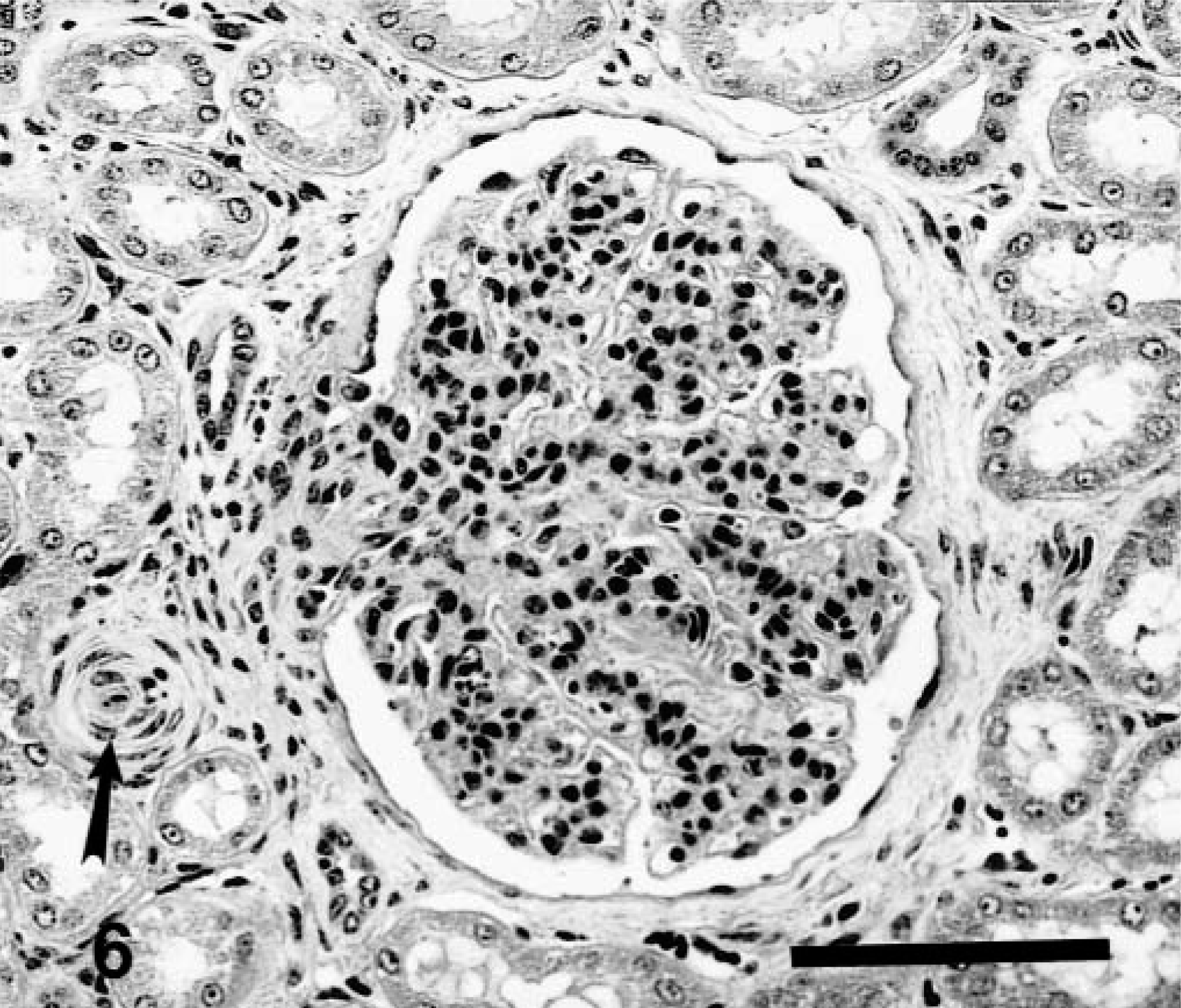
Kidney; 6-month-old calf, Source C. Relatively advanced stage glomerulopathy. Mesangial regions are hypercellular with mesangial cells and scattered granulocytes. There is minimal or lack of involvement of visceral and parietal epithelium. Bowman's capsule and vascular pole are thickened and fibrotic. Note arteriolar muscular hypertrophy near vascular pole (arrow). HE. Bar = 100 μm.
Four animals had scattered glomeruli that were markedly enlarged, with capillaries that were greatly engorged. Occasionally one or more lobules in thus-affected glomeruli were necrotic. The mesangium of scattered glomeruli contained variably sized eosinophilic protein droplets. Arteriolar walls at the glomerular hilus and elsewhere in the cortical interstitium appeared thickened. Glomerular hyalinization was present in two of nine animals, and sclerotic glomeruli were scattered in two. Eosinophilic hyaline casts were present in medullary tubules of eight of nine animals; hyaline droplets were prominent in three of nine animals. In those cases with more severe glomerular lesions, radial streaks of tubular atrophy and mild lymphoplasmacytic infiltrates and fibrosis existed in the cortical interstitium.
Specimens of skeletal muscle for histologic examination, generally from two but as many as eight individual muscles, were available from eight of the nine animals. Individual hypereosinophilic fibers, sometimes swollen but retaining striation, were scattered in sections from most animals. Such changes are considered to be fixation artifacts. Myodegeneration/necrosis was recognized in five of the eight animals. Muscle lesions in one animal were represented by a single fiber that had segmental necrosis and local infiltration of inflammatory cells. Two animals had a single, well-defined focus of fibrosis surrounding scattered clusters of severely atrophic fibers. Hemosiderophages and a minimal inflammatory cell infiltrate were scattered within areas of fibrosis. Two additional animals had discrete areas of coagulative myonecrosis; in these, not only was the entire muscle component necrotic, but stromal elements including fibroblasts and vasculature also were necrotic. In each of the five animals, lesions were single (monofocal), and they appeared to have been the result of a single incident of injury (monophasic). Mineralization of degenerate myofibers was not observed.
Muscle atrophy was difficult to discern histologically. Occasional fascicles contained fibers of angular configuration and considerably reduced cross-sectional dimension but these were not numerous. Nerve fascicles within apparently normal skeletal muscles occasionally contained degenerate fibers. Many areas of skeletal muscle grossly suspected to be abnormal were not histologically confirmed to have lesions. Neither degenerative change nor necrosis was recognized in the myocardium of any of the nine animals; sarcocysts were present in three of eight. Numerous other tissues from eight of nine animals were examined histologically; significant lesions were not recognized.
Discussion
Major and distinctive lesions in animals of this study existed in peripheral nerves, the extension of afferent fibers into the spinal cord, and in renal glomeruli. Our opinion that the primary locus of neural lesion is peripheral nerve representing a peripheral neuropathy is based on 1) the severe degeneration of both afferent and efferent peripheral fibers accompanied by quite limited lesion in the spinal cord and 2) by degeneration of both arms of bipolar axons, whereas dorsal root ganglion neurons are generally without lesions. 17,29 Further, we believe the neuropathy is the primary neurologic lesion. Though we have not observed signs of cranial nerve dysfunction conclusively attributable to this disease, we did recognize lesions in trigeminal fibers of the only animal from which a cranial nerve was examined, and we noted dysfunction of the tongue in a live, mature cow, the dam of two affected calves.
The renal lesion is clearly centered on glomeruli. Glomerular lesions appear to be progressive, often leading to glomerulosclerosis, tubular atrophy, and interstitial fibrosis. Though both neurologic and renal lesions are consistent manifestations of the syndrome, we are unaware of a disease mechanism that might account for pathologic changes in both. Whatever the mechanism(s) responsible for the glomerulopathy observed in this group of animals it/they occur uncommonly in cattle because glomerular disease in cattle is unusual.
Skeletal muscle contained scattered but limited areas of fiber atrophy compatible with neurologic damage. Myodegeneration and myonecrosis were recognized in five of eight animals. Lesions observed in our study were discretely monofocal, appeared to be uniformly monophasic, and in some instances included intralesional necrosis of arterial and other tissue components. Despite indications of marginal to deficient levels of tissue and blood selenium, the observed lesions are much more likely to be the result of trauma, injection, ischemia, or profound local muscular fatigue rather than related to systemic metabolic or toxic causes. 20
The syndrome in young Gelbvieh cattle recently reported by Moisan et al. 21 appears to be essentially identical to the one reported here. Both have familial if not well-defined hereditary occurrence, and both have similar lesions in a similar organ distribution. However differences, real or interpretive, exist in the magnitude and distribution of skeletal muscle lesions and the primary locus and cause of neurologic and renal lesions. Although Moisan et al. 21 emphasize the myelopathy, our observations lead us to conclude that the primary neurologic lesion is a peripheral neuropathy with secondary lesions in the spinal cord. Further, in contrast to Moisan et al., 21 we recognized vasculitis and vascular necrosis exclusively in foci of myonecrosis rather than as a disseminated lesion.
Clinical and pathologic features of most other congenital and early onset neurologic syndromes of cattle differ from the presently described syndrome in breed occurrence, clinical signs, course to death, and primary or specific locus of the nervous system affected. The occurrence of disease exclusively and repeatedly in offspring of a single bull and only 4 of 28 breeding females in successive pregnancies implies genetic factors as causal or strongly contributory. We doubt that environmental factors (toxic, nutritional, infectious) contributed to the syndrome we report. We are unable to speculate constructively on a molecular basis for the lesions that characterize the disease.
Footnotes
Acknowledgements
Cases from Source C were provided through the courtesy of Dr. B. C. Barr, California Veterinary Diagnostic Laboratory, Davis, CA. The assistance of Ms. Diana Moffeit with word processing and Ms. Jeanenne Duffy with histotechnologic expertise is gratefully acknowledged.
