Abstract
Charcot-Marie-Tooth disease (CMT) is a hereditary sensory and motor peripheral neuropathy that is one of the most common inherited neurological diseases of humans and may be caused by mutations in a number of different genes. The subtype Charcot-Marie-Tooth disease type 4H (CMT4H) is caused by homozygous mutations in the FGD4 (FYVE, RhoGEF, and PH domain-containing 4) gene. A previous genome-wide association study involving 130,783 dairy cows found 6 novel variants, one of which was a homozygous splice site mutation in the FGD4 gene. Descendants of carriers were genotyped to identify 9 homozygous Holstein Friesian calves that were raised to maturity, of which 5 were euthanized and sampled for histopathology and electron microscopy at 2 and 2.5 years of age. Three control Holstein Friesian animals were raised with the calves and euthanized at the same time points. No macroscopic lesions consistent with CMT4H were seen at necropsy. Microscopically, peripheral nerves were hypercellular due to hyperplasia of S100-positive Schwann cells, and there was onion bulb formation, axonal degeneration with demyelination, and increased thickness of the endoneurium. On electron microscopy, decreased axonal density, onion bulb formations, myelin outfoldings, and increased numbers of mitochondria were present. These changes are consistent with those described in mouse models and humans with CMT4H, making these cattle a potential large animal model for CMT.
Keywords
Charcot-Marie-Tooth disease (CMT) is a hereditary sensory and motor peripheral neuropathy that is one of the most common inherited neurological diseases of humans, with approximately 1 in 2500 people affected around the world. 38 So far, more than 100 different genes have been found to cause different types of CMT, with a variety of different modes of inheritance (dominant, recessive, X-linked). 36
There are 3 main forms of CMT, predominantly demyelinating (eg, CMT1), predominantly myelinated axon loss (eg, CMT2), and intermediate type (eg, CMT dominant intermediate). 38 Recessive forms of CMT are in the CMT4 group, also known as hereditary motor and sensory neuropathy type IV. 38 Demyelinating forms of CMT tend to be due to mutations in Schwann cell genes, while axonal types are due to mutations in neuron and glial-neuron unit genes. 38
Charcot-Marie-Tooth disease type 4H (CMT4H) is caused by homozygous mutations in the FGD4 gene (FYVE, RhoGEF, and PH domain-containing 4) encoding the protein frabin and is considered a demyelinating type of CMT. Mutations in FGD4 are described in several different families.1–4,6,8,9,12,15,17,20,39 Clinically, affected individuals present with delayed age of walking, unsteady gait, absence of deep tendon reflexes, distal muscular atrophy, and mildly reduced sensation to touch.9,15 Some individuals also have scoliosis and foot deformities.1,9,12
In Fgd4-deficient mice, there is dysmyelination in developing peripheral nerves, and later, myelin abnormalities and demyelination. 14 Electron microscopic lesions include myelin outfoldings, redundant myelin loops, myelin degradation, and demyelination. Frabin is an actin filament-binding protein, encoded by Fgd4, which is a component of the Schwann cell cytoskeleton. 24 Frabin regulates Rho GTPase CDC42, which is required for myelin development and maintenance.14,18,24 It is also involved in polyphosphoinositide signaling. 8
A genome-wide association study of 130,783 cattle from the national New Zealand (NZ) dairy herd was designed to test the hypothesis that surrogate phenotypes, such as body weight, could be used to map recessive or deleterious gene variants without any prior disease classification. 31 Using a nonadditive model, 6 novel recessive variants were identified. 31 One of these variants was a homozygous nonsense g.77632752C>T variant in FGD4. Prospective genotyping of calves that were from descendants of known carriers identified 22 homozygous animals with the FGD4 variant, and 9 of these calves, along with control animals, were raised to maturity. Affected cattle had significantly decreased weight gain; 2-year-old animals with the FGD4 variant weighed on average 50 kg less than control animals. 31 In addition, affected animals exhibited stumbling and loss of coordination. The frequency of this cattle FGD4 mutation was found to be remarkably high in the NZ dairy population, where approximately 1 in 590 animals are predicted to be affected, and approximately 1 in 13 purebred Holstein Friesians carry the variant. Detailed descriptions of routine histopathology for CMT are lacking in the literature and electron microscopic descriptions concentrate on myelin changes. Therefore, the aim of this study was to describe the pathology associated with the FGD4 variant in affected cattle using histopathology and electron microscopy.
Materials and Methods
Animals
The details on the prospectively genotyped calves and phenotypic analysis are reported in Reynolds et al’s study. 31 Case (n = 5) and control (n = 3) animals were kept at pasture, grazed, and managed together, on the AgResearch Ruakura research farm in Hamilton, New Zealand. Approval for the study was obtained from the AgResearch Animal Ethics committee, approval number 14508. The study was carried out in accordance with the New Zealand Animal Welfare Act 2018.
Two FGD4 variant animals and 2 control animals were euthanized on farm by captive bolt and exsanguination in December 2019, at 2 years of age. A full postmortem examination was performed on the animals immediately after death, and samples (heart, kidney, spleen, thymus, mesenteric and popliteal lymph node, liver, lung, mammary gland, rumen, reticulum, abomasum, omasum, small and large intestine, pancreas, pituitary, brain, and spinal cord; sciatic, femoral, tibial, saphenous, median, and ulnar nerves; and common digital nerves of fore and hind limb, extensor carpi radialis, and gastrocnemius) were collected into 10% neutral buffered formalin.
Four animals (3 FGD4 variants and 1 control) were slaughtered at a local abattoir at 2.5 years of age. The fore and hind limbs and the heart were removed by experienced abattoir staff and delivered immediately to the researchers. Samples of peripheral nerve (sciatic, femoral, tibial, saphenous, median, ulna, and common digital nerves of fore and hind limb), muscle (gastrocnemius), and heart were collected into 10% neutral buffered formalin and 2% glutaraldehyde for histology and electron microscopy, respectively.
Sample Processing
Samples collected into 10% neutral buffered formalin were trimmed, processed, and embedded in paraffin. Sections (3–4 µm) were cut and stained with hematoxylin and eosin (HE). Nerve sections were also stained with Luxol fast blue (LFB). Saphenous nerves were stained with AZAN trichrome (AZT). Slides were examined under a microscope by a single examiner (K.D.), blind to genotype status, and the lesions were described.
Samples of the median nerve and common digital nerve of the forelimb were collected into 2% glutaraldehyde, postfixed in osmium tetroxide, then dehydrated and embedded in resin. Semithin sections (1 µm) were cut and stained with LFB. Thin sections (70-nm thick) were cut and plated on copper grids, followed by impregnation with uranyl acetate and lead citrate. Sections were examined using a FEI Tecnai G2 Biotwin Transmission Electron Microscope by a single examiner at the Manawatu Microscopy and Imaging Centre, Palmerston North, New Zealand.
Immunohistochemistry
Immunohistochemical labeling for S100 was performed on 5-µm sections of paraffin-embedded formalin-fixed saphenous nerve from FGD4 variant and wild-type cows. Antigen retrieval was performed using a citrate buffer (0.01 M, pH 6.0) at 95°C for 30 minutes. The primary antibody was a polyclonal rabbit anti-S100 antibody (Z0311 Dako, Agilent, Santa Clara, CA) and was used at 1:500 dilution. An antirabbit IgG plus horseradish peroxidase conjugated polymer (Biocare Medical, Pacheco, CA) was used for detection of S100 immunostaining and 3,3′ diaminobenzidine chromogen (Betazoid, Biocare Medical, Pacheco, CA) was used to visualize immunoreactivity, with a hematoxylin counterstain. Negative controls included irrelevant antibodies (rabbit IgG) and omission of the primary antibody.
Nerve Measurements
Images of LFB semithin sections were taken from the median nerve and the total number of myelinated fibers was counted using the Adobe Photoshop 2021 Count tool to determine the density of myelinated fibers (/mm2). The area of each nerve fascicle was measured using Fiji Image J 2.1.0/1.53c measure function.
Images of electron microscopy ultrathin sections were taken from the median nerve. Axon diameter, myelinated fiber diameter, axon area, and myelinated fiber area were measured using Fiji Image J 2.1.0/1.53c measure function. G-ratio was calculated as axon diameter divided by myelinated fiber diameter, whereas area ratio was calculated as axon area divided by myelinated fiber area. The G-ratio gives an estimate of the relationship between the myelin thickness and the size of an axon, and the optimal G-ratio is a balance between volume constraints and speed of conduction. 7 Onion bulb formations in peripheral nerves are formed by concentric layers of Schwann cell processes around the central axon. 41 For each axon, the number of Schwann cell lamellae was manually counted, only Schwann cell processes that completely circled an axon were counted as a lamella (1 was the minimum count). 35
Values are reported as mean ± standard error mean (SEM). A linear mixed-effects model, with a random effect to account for repeated measures, was applied to the data using the nlme package 27 (Version 3.1-150) in R-studio (Version 1.3.1093, R Version 4.0.3), 32 to test for significant differences (P < .05) between control and case animals.
Results
Gross Lesions
No differences were noted in the size of the peripheral nerves in control and case animals. Similarly, no differences were noted in organ: liveweight measures (Table 1) or the ratio of left:right ventricular wall thickness. The lack of macroscopic changes in heart weight and ventricular wall thickness suggests the absence of heart disease.
Quantitative data for calves with homozygous splice site mutation in FGD4 compared with controls.
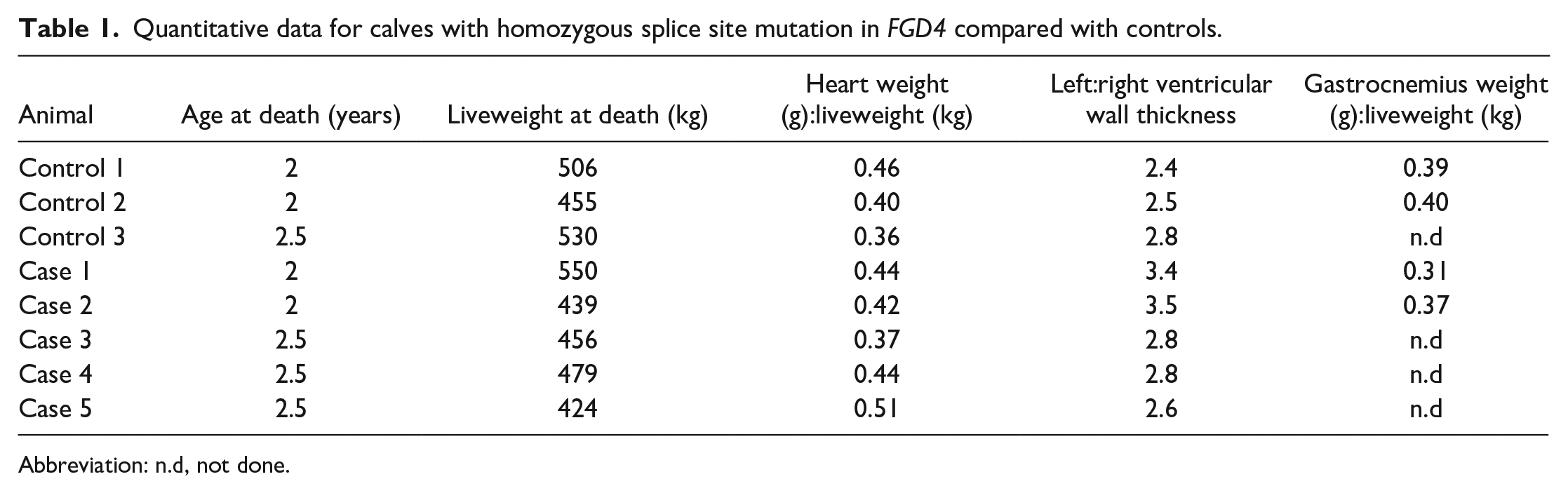
Abbreviation: n.d, not done.
Incidental gross lesions were present in 1 control animal and 1 case animal. In case 2, there was an 11- × 5-cm area of reddened consolidation in the right cranioventral lung lobe, consistent with chronic bronchopneumonia. In addition, multifocally throughout the intestines and mesenteric lymph nodes there were 1- to 2-mm firm cream foci; histologically, these were consistent with parasitic granulomas.
In control 2, a 4- × 3.5- × 4-cm abscess with a 1-cm thick fibrous capsule was present in the right lobe of the liver. Multifocal 1- to 2-mm firm cream foci were also present throughout the intestines which on histology were consistent with parasitic granulomas.
Histopathology and Immunohistochemistry
All peripheral nerves examined from case animals were hypercellular compared with control animals (Figs. 1, 2). Occasionally, Schwann cells were lined up alongside nerve fibers or grouped together in small clusters. Cells that surrounded axons in case animals had positive cytoplasmic and nuclear immunolabeling for S100, confirming their identification as Schwann cells (Figs. 3–5). Other cells with spindle-shaped nuclei in the endoneurial tissue in most cases did not immunolabel for S100 (Figs. 3, 5). In some cases, cytoplasmic processes of Schwann cells swirled around axons (Fig. 6).
Frequently, axons were swollen, and occasional digestion chambers with foamy macrophages (gitter cells) were present (Fig. 7). Axons appeared to be separated by increased amorphous eosinophilic material (Figs. 1, 7). However, this material did not immunolabel for S100 (Fig. 5), and on AZT staining was positive for collagen (Figs. 8, 9). On LFB-stained sections, peripheral nerves showed evidence of demyelination (Figs. 10–12). No microscopic lesions were detected in the spinal cord or brain.
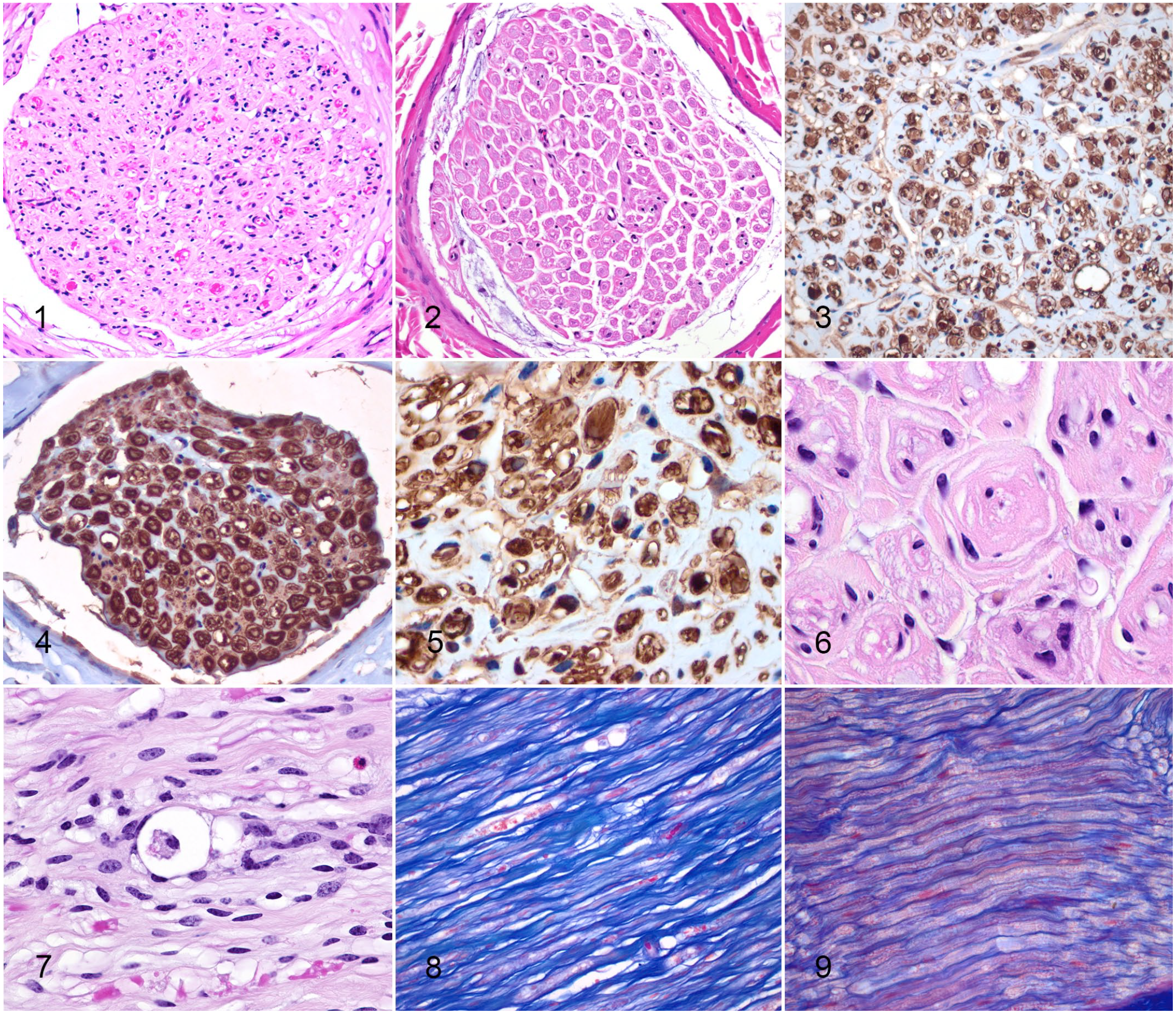
Charcot-Marie-Tooth disease type 4H, nerve, cow.
On LFB semithin sections from case animals, myelinated nerve fibers were markedly reduced in number and widely separated compared with control animals (Figs. 13, 14). There was marked variation in myelinated fiber size due to the presence of axons encompassed by excessive focally folded myelin sheaths (Figs. 15, 16). Other axons had no myelin sheath or an extremely thin layer of myelin (Fig. 16). The nerve fascicles of case animals were hypercellular compared with control animals, and this was due to Schwann cell hyperplasia (Figs. 16–18). Onion bulb formations were also present (Fig. 16). Increased, slightly fibrillar material was present surrounding axons and Schwann cells (Figs. 16, 18), which when combined with the positive staining for collagen on AZT was consistent with increased thickness of the endoneurium.
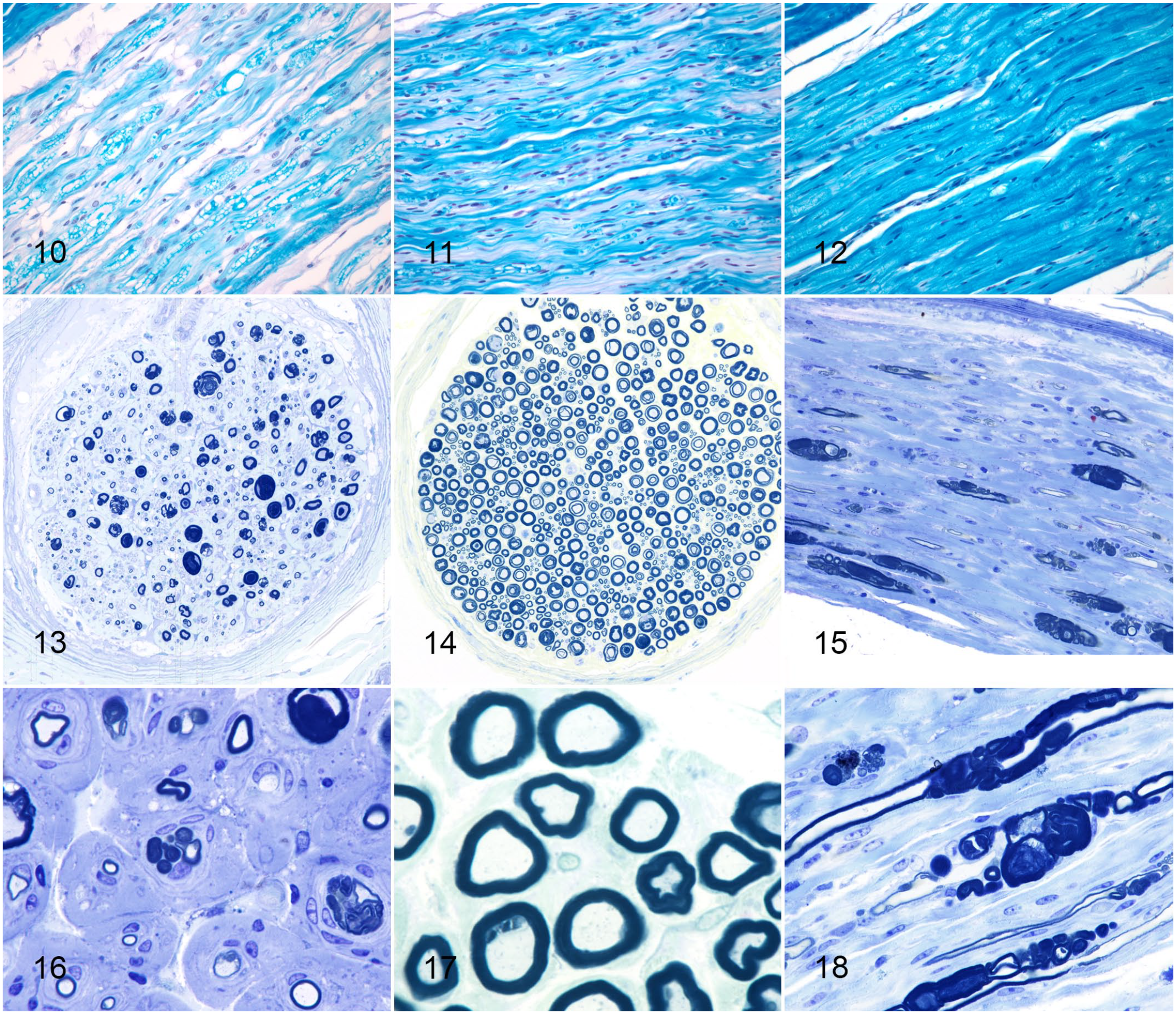
Charcot-Marie-Tooth disease type 4H, nerve, cow.
Electron Microscopy
A variety of different myelination patterns were present on electron microscopy. In some cases, axons lacked a myelin layer (Fig. 19) or the myelin layer was extremely thin, consistent with demyelination. Other axons had myelin outfoldings ranging from simple to highly complex (Fig. 20), some protruding into the axon, in comparison with normal myelinated axons (Fig. 21). On longitudinal sections, abnormal myelin outfoldings appeared to be associated with nodes of Ranvier (Fig. 22).
Schwann cells were markedly hypertrophied and hyperplastic with multiple Schwann cells around a single axon (Figs. 23, 24) and concentric layers of Schwann cell processes (onion bulb formations) surrounding myelin sheaths (Fig. 25); the maximum number of lamellae present around an axon was 6. Within the cytoplasm of Schwann cells, there were increased numbers of mitochondria, some abnormal with loss of cristae, and endoplasmic reticulum (Figs. 26, 27); this was particularly marked in some Schwann cells. Similarly, increased numbers of mitochondria were also present within axons. In addition, within the cytoplasm of some Schwann cells there were accumulations of electron dense material consistent with degenerate myelin (Fig. 27).
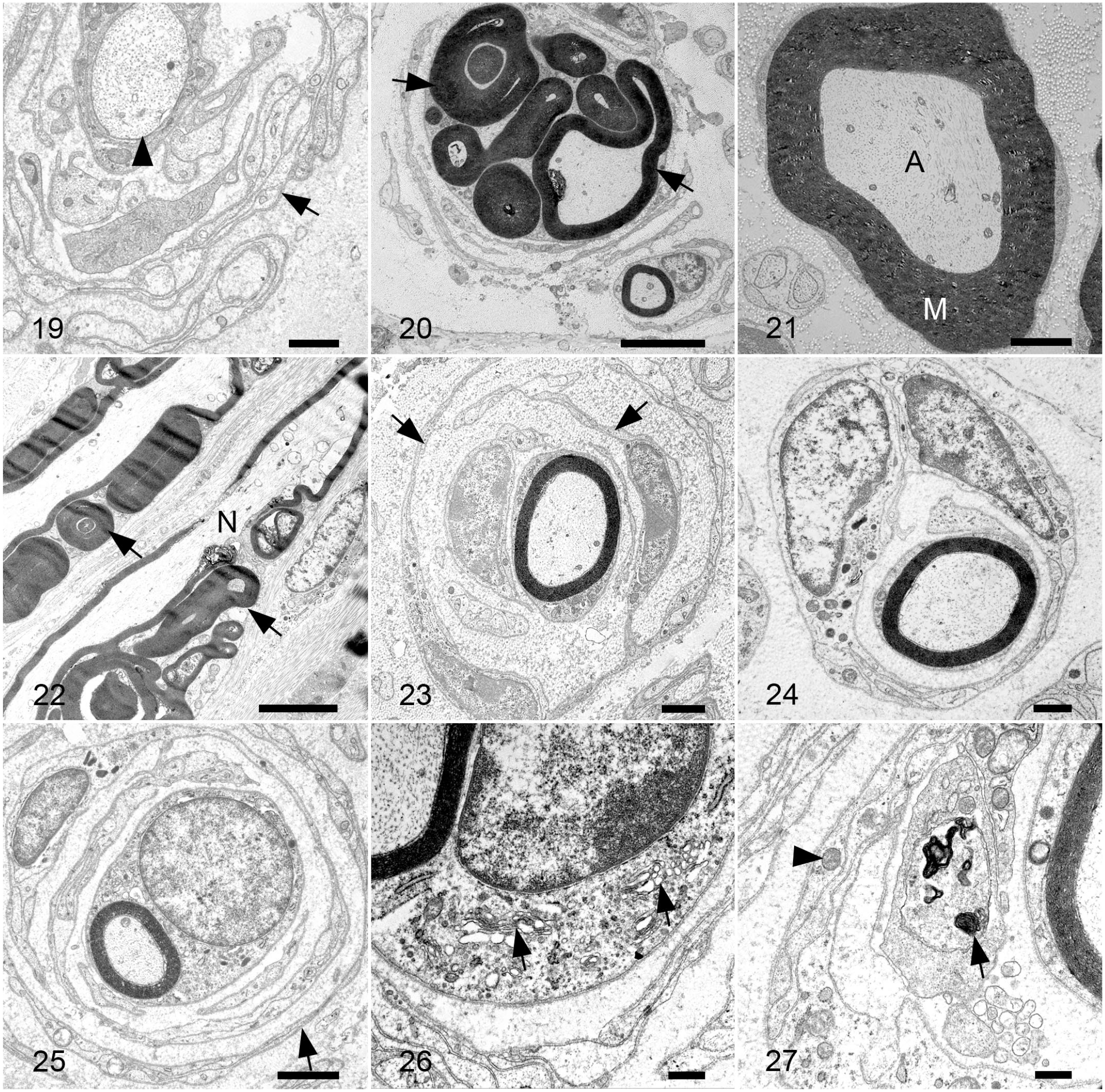
Charcot-Marie-Tooth disease type 4H, median nerve, cow. Transmission electron microscopy.
Nerve Measures
Case animals had a significantly greater G-ratio (mean ± SEM, 0.76 ± 0.01, n = 5) compared with control animals (mean 0.60 ± 0.01, n = 3, P = .0048), due to decreased myelin thickness in case animals. Similarly, case animals had a significantly greater area ratio (mean 0.53 ± 0.02, n = 5) compared with control animals (0.27 ± 0.01, n = 3, P = .0052), due to decreased myelin area in case animals. Case animals had significantly decreased myelinated fiber density (10.73 ± 2.58 myelinated fibers/mm2, n = 5) compared with control animals (37.67 ± 0.37 myelinated fibers/mm2, n = 3, P = .0002). All case animals had onion bulb formations, while no onion bulb formations were seen in control animals. The mean number of Schwann cell lamellae per onion bulb in case animals was 2.6 ± 0.1 lamellae, while all control animals had just 1 Schwann cell lamella surrounding each axon.
When the age of affected animals was examined, younger animals (2 years of age) had greater fiber density (12.40 ± 3.92 myelinated fibers/mm2, n = 5) compared with older animals (2.5 years of age, 7.39 ± 0.97 myelinated fibers/mm2, n = 3); however, this difference was not significant (P = .1541).
Discussion
The key features of the pathology of CMT4H in Holstein Friesian dairy cattle were demyelination and remyelination, as shown by decreased myelinated fiber density, Schwann cell hyperplasia and hypertrophy, and onion bulb formations. These findings are consistent with those described in humans with CMT4H and a FGD4−/− mouse model.8,14
Microscopic lesions in HE-stained histologic sections were suggestive of a peripheral neuropathy, with hypercellular nerves that immunohistochemistry for S100 confirmed was due to Schwann cell hyperplasia. Furthermore, LFB staining on routine sections showed decreased myelination of the nerves. However, full characterization of the myelin changes required semithin LFB-stained sections, where the excessively folded myelin sheaths, onion bulb formations, and decreased myelin fiber density were obvious, and case and control animals were easily distinguished.
The different types of CMT in humans may present with different types of nerve lesions. 37 There are 3 forms of CMT: predominantly demyelinating, predominantly loss of myelinated axons, and mixed axonal and demyelinating (dominant intermediate). In demyelinating forms of CMT such as CMT1a, there is decreased myelinated fiber density, both decreased and increased myelination, and a high percentage of onion bulb formations. 37 Whereas, in CMTX1, a mixed axonal and demyelinating form, there is normal density of myelinated fibers, but myelin is thin, small fibers predominate, and there are few onion bulb formations; instead, cluster formations are present. 37 CMT4H is considered a demyelinating form of CMT, and this was consistent with the microscopic findings, with hypo- and hyper-myelination and a large number of onion bulb formations, similar to the findings of CMT1a (the prototypical demyelinating CMT).
CMT4H in humans has an early onset (often before 3 years of age) but progresses slowly. 3 Affected individuals often develop scoliosis, foot deformities, and atrophy of distal muscles. 12 About a third of the CMT4H disease-causing variants described in humans are splice site mutations,2,8,12,17,39 like that described in affected cattle in this study. 31 However, there appears to be some variability in clinical presentation, depending on the location of the FGD4 mutation. 3 The cattle c.1671 +1G>A mutation leads premature truncation of the protein (p.L626X) and the loss of 3 conserved domains, including 3 FYVE, RhoGEF, and PH domains, one with a putative zinc-binding site and another with a putative phosphatidylinositol 3-phosphate-binding site, suggesting severe consequences for protein function. However, clinical signs in affected animals were mild, as stumbling and loss of coordination were not noted until close to 2 years of age (early adulthood). In addition to stumbling, affected animals regularly collapsed to kneeling when in a crush. 31 Anecdotally, in herds with affected animals, they were more likely to be culled for decreased weight gain and milk production rather than clinical signs of neurological disease. Humans with a milder phenotype of CMT4H have been described, whereby clinical signs did not occur until late adolescence or adulthood. In one of those affected individuals, although a truncated protein was likely to be produced (c.2211dupA), this protein was likely to contain all the functional domains, perhaps explaining the milder phenotype. This, however, does not appear to be the case with the affected cattle.
In humans, a common feature of CMT4H is delayed walking but this is not a feature of the cattle disease. Cattle as a precocial species differ developmentally at birth compared with an altricial species such as humans. The spinal cord, nerves, and limb muscles develop relatively early during development in both precocial and altricial species. 22 However, rats (an altricial species) have fewer motor neuron dendritic bundles, neuromuscular connections, muscle maturation, and afferent feedback from the periphery at birth compared with chickens (a precocial species). 22 Likewise, while the progression/order of myelin formation is similar in precocial and altricial rodent species, the altricial species have an earlier onset of myelination, meaning myelin is more mature at a given postconception age. 40 This difference in myelin development at birth may perhaps explain differences in developmental delays in humans and cattle with CMT4H.
Affected cattle of 2 different ages were examined in this study (2 and 2.5 years of age). Older cattle appeared to have decreased myelin fiber density compared with younger cattle, although this difference was not significant likely due to the small sample size. The decrease in myelin fiber density in only 6 months suggests that perhaps once the cattle develop clinical signs, the lesions may progress more quickly than in humans. Examination of greater numbers of animals and more age ranges would be required to confirm this. In addition, serial biopsies in humans are not routinely performed and so progression of histological lesions is not described. In a Fgd4−/− mouse model, myelin outfoldings increased in number and complexity with age. Similarly, the G-ratio also increased with age, indicating thinner myelin. 14 Interestingly, in the Fgd4−/− mice, while progression of demyelination/remyelination was steady up to 60 days of age, between 60 and 80 days there was a rapid increase in features of demyelination/remyelination. 14 The authors suggested that demyelination may proceed quickly if a certain threshold is reached. In contrast to the affected cattle, Fgd4−/− knockout mice did not have any primary or secondary axonal loss.
On HE-stained histologic sections, increased eosinophilic amorphous material wrapped around axons. The material did not immunolabel for S100, suggesting it was not Schwann cell cytoplasmic processes. However, it did stain positively for collagen with AZT suggesting the endoneurium was thickened. Increased peri- and endoneurial type I and III collagen has been demonstrated in biopsies from CMT1 patients, thought to be associated with axon loss and the duration of disease. 26 Increased extracellular matrix molecules (such as fibronectin and laminin) and collagens such as type I, III, IV, and V show increased deposition in CMT1, and encircle myelinated fibers. 26
Cardiac disease, including cardiac arrhythmias, cardiomyopathy, ischemic heart disease, and mitral valve prolapse, has been described in humans with CMT.11,19 However, the direct association of cardiac disease with the peripheral neuropathy is controversial. Morphometric heart measurements and microscopic examination of the heart failed to detect any evidence of heart disease in cattle with CMT4H.
Secondary axonal degeneration can be a feature of demyelinating neuropathies, 21 and axonal degeneration with the presence of gitter cells was visible on HE-stained sections of nerves from affected cattle. Schwann cells produce myelin, but also have other important roles such as protecting axons from toxic insults, organizing axon cytoskeleton and microtubule transport, and providing trophic (neurotrophin-3, insulin-like growth factor 1, ciliary neurotrophic factor, and erythropoietin) and metabolic support. 21 In humans with CMT1A (mutations in peripheral myelin protein 22, PMP22), secondary axon degeneration is thought to contribute to clinical disability. 21 Possible suggested mechanisms for secondary axon degeneration in CMT1A include altered microtubule cargo transport and cytoskeleton organization, and reduced ciliary neurotrophic factor and neurotrophin-3 expression.21,23,33,34
The pathogenesis of the myelin lesions, particularly the myelin outfoldings, in CMT4H is not clear. Some evidence suggests the disease may be a combination of dysmyelination in early development of the peripheral nervous system and then demyelination later in life. 14 FGD4 encodes the frabin protein which is a guanine nucleotide exchange factor specific for CDC42 Rho GTPase 8 and is expressed in early postnatal development of the peripheral nervous system, with expression persisting in adult Schwann cells. 39 FGD4 may be involved in a crucial step of peripheral nerve system development known as radial sorting of axons, a process whereby Schwann cells instigate events that ultimately result in 1 Schwann cell ensheathing 1 axon segment. 29 Defects in axon sorting lead to dysmyelinating peripheral neuropathies. Laminin-211 (also known as merosin) is considered a key controller of axonal sorting, and both CDC42 and frabin are in a pathway downstream from laminin-211. 28 Mutations in the LAMA2 gene that encodes laminin-211 cause a dysmyelinating neuropathy (along with muscular dystrophy) with both focally thickened myelin and thinner myelin, although onion bulbs are not reported. 30 Frabin cross-links actin, which interacts with laminin-211 via periaxin and dystroglycan, thus mutations in FGD4 disturb the cytoskeleton, as evidenced by decreased formation of filopodia-like microspikes with an abnormal curled shape. 8
Disrupting CDC42 in Schwann cells produces a similar myelin phenotype to that of Schwann cells with mutant FGD4. 14 CDC42 is involved in several processes in Schwann cells, including endocytosis for regulation of protein and lipids, vesicular transport, proper matching of axon-Schwann cell units, and rearrangement of the cytoskeleton in radial sorting.5,10,13,25,30 FGD4 is also required for efficient endocytosis. 14 In addition, frabin has FYVE and PH domains that suggest it could interact with myotubularin-related proteins (MTMR2) which are required for membrane trafficking. 39 Indeed, humans with MTMR2 mutations and CMT4B1 also have myelin outfoldings.16,42 Therefore, the myelin outfoldings are suggested to result from dysregulation of the cytoskeleton and membrane transport processes, such as endocytosis, due to impaired Rho GTPase signaling.38,39
In summary, cattle homozygous for the FGD4 mutation, despite not having severe clinical signs, had lesions on semithin sections and electron microscopy consistent with CMT4H in humans and mouse models. Given the high frequency of the FGD4 mutation in the NZ dairy population, and the similarity of the microscopic lesions among animals and humans homozygous for the FGD4 mutation, the cattle may be a useful large animal model for CMT4H and provide abundant material for future research.
Footnotes
Acknowledgements
The authors would like to thank Bryce Ilton for technical assistance, Petru Daniels and Evelyn Lupton for preparing histology slides, Raoul Solomon and Yanyu He at the Manawatu Microscopy and Imaging Centre for preparing EM sections, Jonathan Marshall for statistical advice, and R.D. Jolly for reading the article.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: C. Neeley and M. Littlejohn are paid employees of Livestock Improvement Corporation, a breeding company and patent applicant for testing applications of the FGD4 mutation. The remaining author(s) declare no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Funding from an MBIE Endeavour grant titled Next Generation Genomic Selection (LICX1802).
