Abstract
Riboflavin (vitamin B2) deficiency in young chickens produces a demyelinating peripheral neuropathy. In this study, day-old broiler meat chickens were fed a riboflavin-deficient diet (1.8 mg/kg) and killed on posthatch days 6, 11, 16, 21, and 31, while control chickens were given a conventional diet containing 5.0 mg/kg riboflavin. Pathologic changes were found in sciatic, cervical, and lumbar spinal nerves of riboflavin-deficient chickens from day 11 onwards, characterized by endoneurial oedema, hypertrophic Schwann cells, tomacula (redundant myelin swellings), demyelination/remyelination, lipid deposition, and fibroblastic onion bulb formation. Similar changes were also found in large and medium intramuscular nerves, although they were less severe in the latter. However, by contrast, ventral and dorsal spinal nerve roots, distal intramuscular nerves, and subcutaneous nerves were normal at all time points examined. These findings demonstrate, for the first time, that riboflavin deficiency in young, rapidly growing chickens produces selective injury to peripheral nerve trunks, with relative sparing of spinal nerve roots and distal nerve branches to muscle and skin. These novel findings suggest that the response of Schwann cells in peripheral nerves with riboflavin deficiency differs because either there are subsets of these cells in, or there is variability in access of nutrients to, different sites within the nerves.
The normal functioning of the nervous system depends on a constant supply of nutrients. A deficiency of vitamins, particularly groups B (thiamine, cobalamin, pyridoxine, and niacin) and E, generally produces an axonal type of polyneuropathy. 22, 41 However, riboflavin (vitamin B2) deficiency is characterized by demyelination with hypertrophy of Schwann cells, marked lipid accumulation, paranodal tomacula, and fibroblastic onion bulbs. 6– 8, 19, 20 There is, at least initially, sparing of axons, and spinal cord and brain are apparently unaffected. 7
Riboflavin (7,8-dimethyl-10-ribityl-isoalloxazine) is a water-soluble vitamin and a dietary requirement in both chickens and humans. It is a precursor of flavin mononucleotide and flavin adenine dinucleotide, and decreased levels of these coenzymes impair oxidative phosphorylation. 36 Riboflavin deficiency leads to decreased beta-oxidation of fatty acids with relative preservation of the tricarboxylic acid cycle. 37 The lipid deposition in Schwann cells and the formation of paranodal redundant loops of normal myelin (paranodal tomacula) suggest a disturbance of lipid metabolism and control of myelin membrane formation in the myelinating Schwann cell.
The novel findings in this study that Schwann cells in spinal nerve roots and subcutaneous and distal intramuscular nerves do not show these pathologic changes suggest that Schwann cells in different parts of the peripheral nerve system respond differently to riboflavin deficiency.
Materials and Methods
Our avian riboflavin deficiency model of demyelinating peripheral neuropathy has recently been described. 7 Day-old broiler meat chickens (Cobb 500, Cobb-Vantress Inc., Arkansas, USA) were maintained ad libitum on either a routine diet containing 5.0 mg/kg riboflavin (control group) or a riboflavin-deficient diet containing 1.8 mg/kg (riboflavin-deficient group). Chickens were anaesthetized on posthatch days 6 (PH6d), 11, 16, 21, and 31, and peripheral nerves were fixed by transcardiac perfusion with 4% paraformaldehyde/2.5% glutaraldehyde in 0.1 M phosphate buffer (pH 7.4). Sciatic, cervical and lumbar spinal nerves; ventral and dorsal spinal roots; dorsal root ganglia (DRG); biceps brachii, gastrocnemius, quadriceps femoris, and longissimus dorsi muscles; and skin of the thigh were collected and processed according to a standard protocol. 10 Five animals at each time point were examined for both control and riboflavin-deficient chickens. One-micron plastic sections were stained with toluidine blue for light microscopy, and 80-nm plastic sections were stained with lead citrate and uranyl acetate for electron microscopy.
This experimental protocol was approved (83/04) by the Animal Ethics Committee of the Institute of Medical and Veterinary Science, Adelaide, Australia, and conformed to the Australian Code of Practice for the Care and Use of Animals for Scientific Purposes (2004).
Results
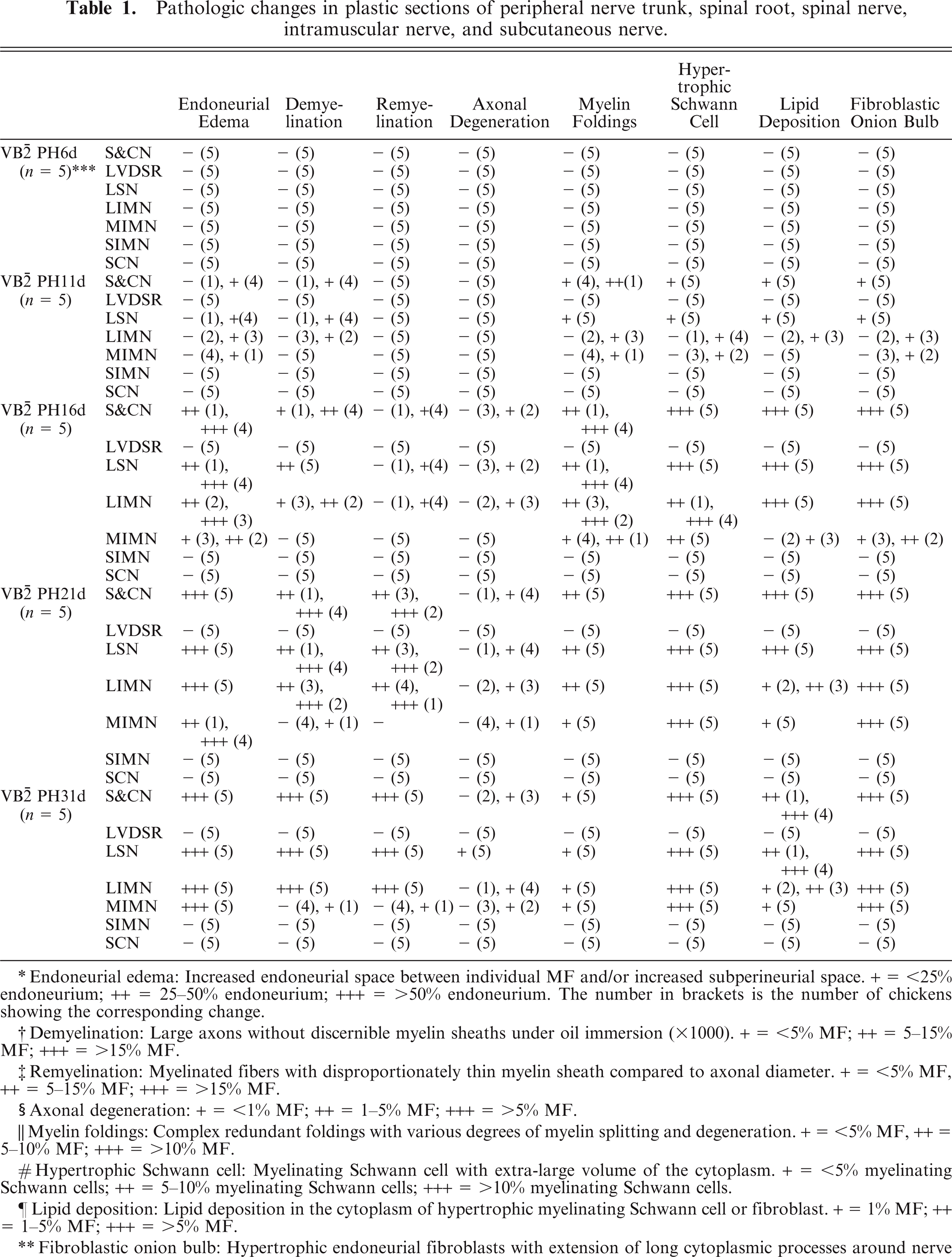
Chickens fed a riboflavin-deficient diet developed progressive symmetrical paresis from day 8, growth retardation, and significantly lower riboflavin concentrations in the liver. 7 Pathologic findings in riboflavin-deficient chickens are summarized in Table 1. Sciatic and cervical nerves showed demyelination with paranodal redundant myelin foldings, hypertrophic myelinating Schwann cells, fibroblastic onion bulb formation, lipid deposition in the cytoplasm of myelinating Schwann cells and fibroblasts, and endoneurial edema from PH11d onwards (1a, b) as previously reported. 6– 8 No neurologic dysfunction or pathologic abnormality was detected in control chickens fed a conventional diet.
Pathologic changes in plastic sections of peripheral nerve trunk, spinal root, spinal nerve, intramuscular nerve, and subcutaneous nerve.
Endoneurial edema: Increased endoneurial space between individual MF and/or increased subperineurial space. + = <25% endoneurium; ++ = 25–50% endoneurium; +++ = >50% endoneurium. The number in brackets is the number of chickens showing the corresponding change.
Demyelination: Large axons without discernible myelin sheaths under oil immersion (x1000). + = <5% MF; ++ = 5–15% MF; +++ = >15% MF.
Remyelination: Myelinated fibers with disproportionately thin myelin sheath compared to axonal diameter. + = <5% MF, ++ = 5–15% MF; +++ = >15% MF.
Axonal degeneration: + = <1% MF; ++ = 1–5% MF; +++ = >5% MF.
Myelin foldings: Complex redundant foldings with various degrees of myelin splitting and degeneration. + = <5% MF, ++ = 5–10% MF; +++ = >10% MF.
Hypertrophic Schwann cell: Myelinating Schwann cell with extra-large volume of the cytoplasm. + = <5% myelinating Schwann cells; ++ = 5–10% myelinating Schwann cells; +++ = >10% myelinating Schwann cells.
Lipid deposition: Lipid deposition in the cytoplasm of hypertrophic myelinating Schwann cell or fibroblast. + = 1% MF; ++ = 1–5% MF; +++ = >5% MF.
Fibroblastic onion bulb: Hypertrophic endoneurial fibroblasts with extension of long cytoplasmic processes around nerve fibers producing an onion bulb-like structure (Cai et al.8). + 5, 5% MF were partially surrounded by cytoplasmic processes of fibroblasts; ++ 5 5-10% MF were partially or completely surrounded by cytoplasmic processes of fibroblasts; +++5.10% MF were partially or completely surrounded by cytoplasmic processes of fibroblasts.
VB
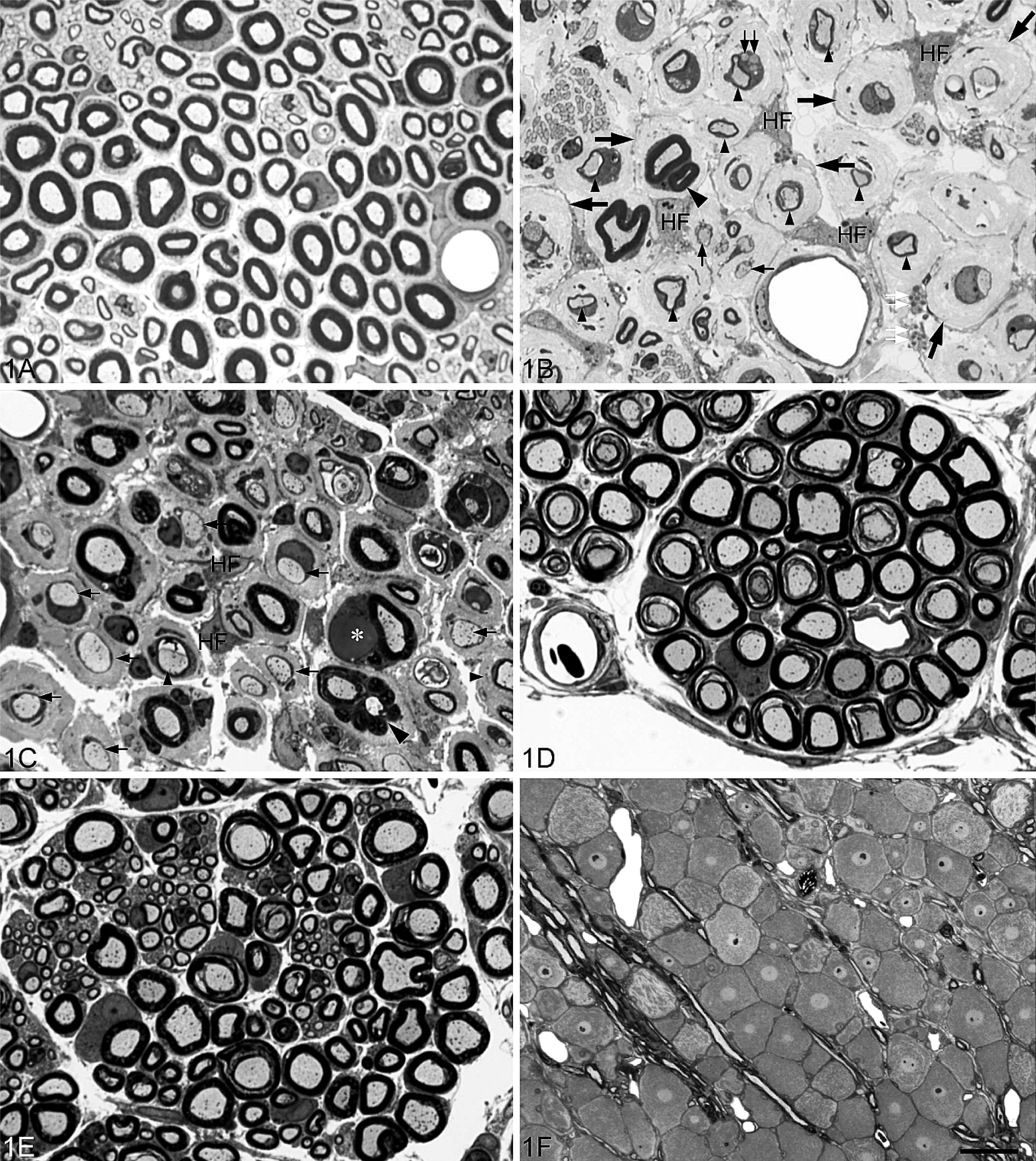
Toluidine-stained, 1-μm plastic sections.
Lumbar spinal nerves (Fig. 1C) showed similar pathologic changes to sciatic and cervical nerves at all time points examined. However, no pathologic changes were found in ventral (Fig. 1D) and dorsal (Fig. 1E) spinal roots (Table 1). Step cross-sections and longitudinal sections revealed occasional abnormalities in a small portion of ventral and dorsal spinal roots adjacent the DRG. Dorsal root ganglia neurons were normal (Fig. 1F). Spinal nerves formed by the fusion of ventral and dorsal nerve roots showed similar pathologic changes to those found in the sciatic and cervical nerves.
Intramuscular nerves contain motor, sensory, and autonomic nerve fibers, and their size and location vary considerably. There are currently no generally accepted criteria by which to subclassify intramuscular nerves and, accordingly, intramuscular nerves in the present study were divided into 3 groups: large, medium, and small. Large intramuscular nerves were direct branches of the main trunk of the peripheral nerve located in the epimysium and consisted of 1 or more nerve fascicles (Fig. 2). The perineurium of large intramuscular nerves was composed of not fewer than 4 lamellated layers of flattened perineurial cells enveloped by a distinct epineurium. The diameter of large intramuscular nerves usually exceeded 60 μm at PH6d, 80 μm at PH11d, 90 μm at PH16d, 100 μm at PH21d, and 120 μm at P31d. Medium intramuscular nerves were defined as branches of a large intramuscular nerve located in the perimysium. They were surrounded by a connective tissue sheath, 2–3 layers thick (Fig. 3A), but lacked the characteristically compact and lamellated features of perineurial cells seen in the large intramuscular nerve. The average diameter of medium intramuscular nerves was approximately 20 (15–60) μm at PH6d, 30 (20–80) μm at PH11d, 40 (25–90) μm at PH16d, 50 (30–100) μm at PH21d, and 60 (40–120) μm at P31d. Small intramuscular nerves were terminal ramifications of medium intramuscular nerves (Fig. 3B) and included intramuscular nerve twigs innervating muscle fibers and muscle spindles. They were located in the endomysium, with a diameter of not more than 15 μm at PH6d, 20 μm at PH11d, 25 at PH16d, 30 at PH21d, and 40 μm at PH31d. Nerve fibers in a small intramuscular nerve were surrounded by a connective tissue sheath, 2 layers or fewer in thickness, without the characteristic perineurial cells seen in large intramuscular nerves. At PH6d, no pathologic changes were detected in intramuscular nerves, but from PH11d onwards, changes seen in peripheral nerve trunks were found in large intramuscular nerves (Table 1, Fig. 2). Similar changes were also found in medium intramuscular nerves from PH11d onwards but were much less severe (Table 1, Fig. 3A). At the late stage of the disease (PH21d and PH31d in this study), extensive demyelination and remyelination involved the majority of nerve fibers in peripheral nerve trunks (Fig. 1B), spinal nerves (Fig. 1C), and large intramuscular nerves (Fig. 2), whereas only a few fibers in medium intramuscular nerves showed these changes. In all treated chickens, no pathologic changes were detected in small intramuscular nerves (Table 1, Fig. 3B).
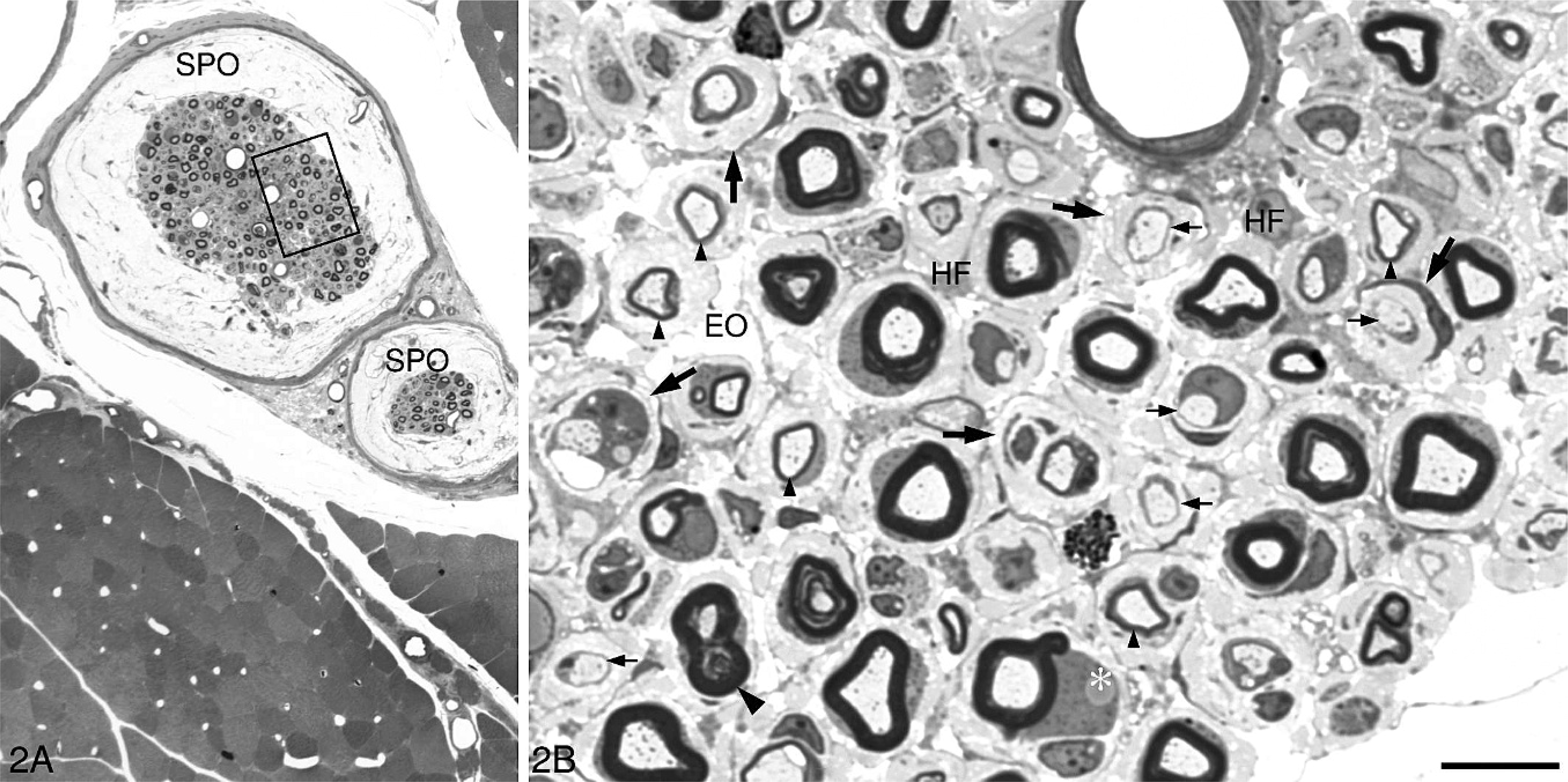
A large intramuscular nerve consisting of 2 fascicles is seen located in the epimysium of the right gastrocnemius of the chicken in
A medium intramuscular nerve in the perimysium of the right gastrocnemius of the chicken in
The subcutaneous myelinated nerve bundles were oriented parallel to the epidermis (Fig. 4A), and no pathologic abnormalities were detected in these myelinated nerve fibers (MF) in any of the animals examined (Fig. 4B).
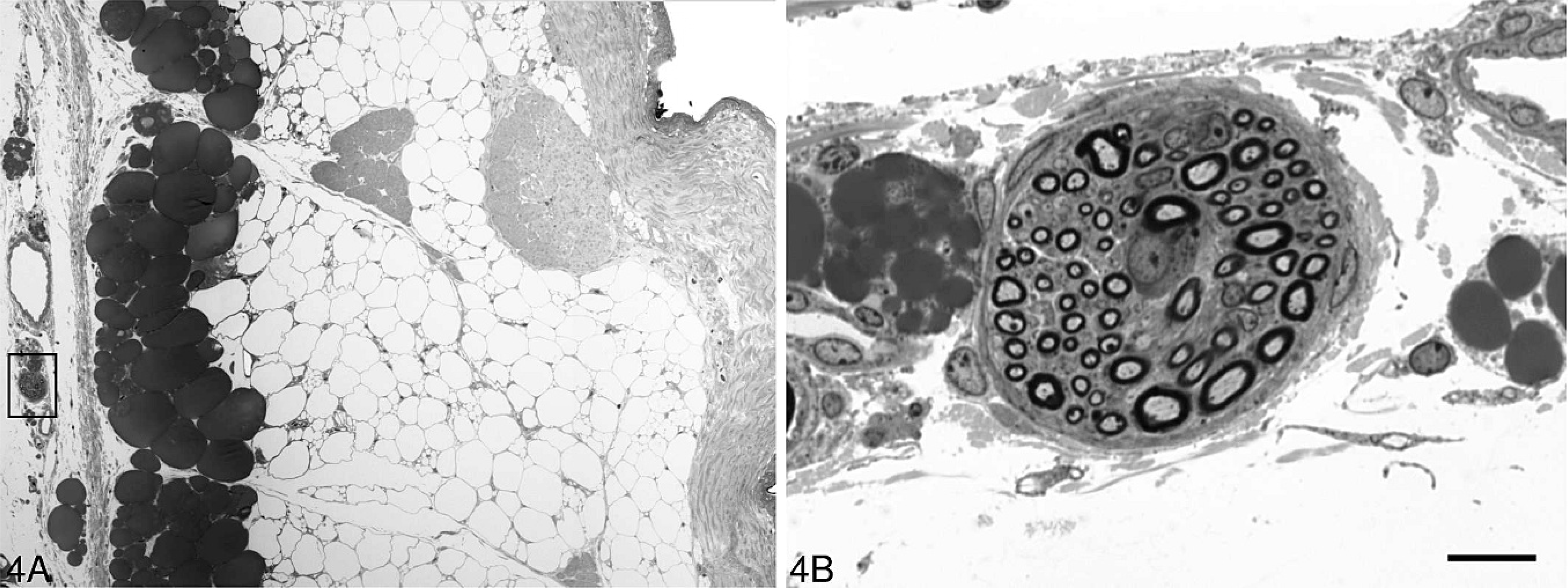
A small nerve (rectangle) is located in the subcutaneous region of thigh skin of the chicken in
Discussion
The peripheral neuropathy produced in young chickens by riboflavin deficiency has been characterized by our 6– 8 and previous 19, 20, 33, 34, 49 studies as a generalized demyelinating polyneuropathy. The demyelination is readily reproducible, peripheral nerves rapidly remyelinate when riboflavin levels are restored, and MF are already myelinated in the chicken at birth. Here we extend these studies by examining proximal (ventral and dorsal spinal roots, DRG, and spinal nerves) and distal branches of peripheral nerves.
At all time points examined, lumbar spinal nerves showed similar pathologic alterations to those seen in sciatic and cervical nerves. However, no pathologic abnormalities were found in both ventral and dorsal spinal roots distant to the DRG, even during later stages (PH31d) of the disease when nearly all MF in sciatic and cervical nerves were affected. Mild pathologic alterations were occasionally noted in ventral and dorsal roots adjoining the DRG. Large intramuscular nerves showed similar pathologic changes to those in peripheral nerve trunks, whereas no pathologic changes were detected in the small intramuscular nerve twigs at all time points examined. Medium intramuscular nerves were less severely injured than large ones, whereas subcutaneous MF were normal at all time points examined. These findings suggest selective vulnerability of peripheral nerves in this avian riboflavin deficiency model of demyelinating polyneuropathy with severe involvement of MF in peripheral nerve trunks and relative sparing of MF in spinal nerve roots and distal intramuscular and subcutaneous nerves.
The mechanism for selective involvement of peripheral nerve trunks in this avian model is unclear. Distal symmetrical polyneuropathy is an axonal disorder 46 and not comparable to the Schwann cell disorder in the present study. In proximal symmetrical polyneuropathies, large fibers appear to be more vulnerable to metabolic abnormalities and immunologic insults than smaller ones. 46 However, this fiber size–related explanation seems untenable in our avian model, because both small and large fibers in spinal roots and distal branches were unaffected.
Circulating nutrients may reach the endoneurial extracellular space either by traversing the endoneurial vascular endothelium or by crossing the perineurium after gaining access to the perineurial extracellular space. The former pathway is believed to be the major route of blood-nerve exchange, because the multilayered perineurium is relatively impermeable. 48 The perineurium is composed of specialized, concentrically arranged perineurial cells joined by tight and gap junctions and perineurial collagen, together constituting a metabolically active diffusion barrier maintaining a hypertonic microenvironment in the endoneurium. 3 Any injury to this barrier will potentially cause nerve fiber damage. 28, 43– 45 In spinal roots, neither epineurium nor perineurium is present. By contrast, spinal roots in the subarachnoid space are surrounded by an extension of the pia mater, which is thinner and looser and may not have the barrier function possessed by the perineurium. 12, 16 Similarly, as a peripheral nerve approaches its termination and begins to branch, the perineurial sleeve becomes progressively thinner and finishes as an open cuff. 5 Thus, unlike peripheral nerve trunks, both spinal roots and distal portions of peripheral nerves are directly exposed to the surrounding milieu and can more readily access blood-borne nutrients. Riboflavin deficiency in these chickens was partial, not absolute, and we therefore speculate that the spinal roots and distal portions of peripheral nerves have easier access to blood-borne nutrients than the nerve trunk, rendering them less vulnerable to a deficiency of this vitamin.
Alternatively, myelinating Schwann cells in spinal roots and distal branches may constitute a different subset from those in the nerve trunk and respond differently to riboflavin deficiency.
It may also be that rapid growth of wings and legs in chickens requires commensurate growth of peripheral nerve trunks and consequent increased myelin formation in these structures, leading to an added metabolic load on myelinating Schwann cells. Further molecular and morphologic studies are required to test these hypotheses.
There is some evidence in other types of demyelinating peripheral neuropathies of variable involvement of different parts of the peripheral nervous system, but the reasons for this selective vulnerability are unclear. Demyelinative changes have been pathologically demonstrated in spinal nerve roots in a few human and experimental demyelinating neuropathies. Demyelination, remyelination, inflammatory cell infiltration, and onion bulb formation have been found in spinal nerve roots in acute 18 and chronic 15, 30, 31, 35 inflammatory demyelinating polyradiculoneuropathies. Demyelinative changes in both peripheral nerves and spinal nerve roots have also been reported in hypertrophic hereditary motor and sensory neuropathy, 4 protein zero–deficient mice, 9, 26 peripheral myelin protein–22 deficient mice, 40 Trembler mice, 2 and Trembler-J mice 13 and in animal models of inflammatory demyelinating neuropathy. 1, 14, 17, 24, 38, 39 In 2 mice with POEMS, which is a monoclonal plasma cell disorder characterized by polyneuropathy (P), organomegaly (O), endocrinopathy (E), monoclonal gammopathy (M), and skin changes (S), spinal nerve roots and sural nerves showed the same pathologic changes of demyelination, remyelination, and inflammatory cell infiltration, 42, 47 and in 3 cases of diabetic neuropathy, there was severe loss of both large and small MF in sural nerves while segmental demyelination and remyelination was the main finding in both dorsal and ventral spinal roots. 32
Distal intramuscular and subcutaneous MF have rarely been examined in demyelinating peripheral neuropathies, although skin biopsy has been used in the diagnosis of disorders involving unmyelinated 21 and myelinated 11, 23, 25 fiber disorders. In neuropathies caused by mutations in peripheral myelin proteins, the demyelination, hypomyelination, onion bulb formation, and altered immunohistochemical staining described in peripheral nerve biopsies have also been found in skin biopsies. 11, 23 In human immunoglobulin M anti-myelin–associated glycoprotein neuropathy, immunoglobulin M deposits were found on dermal MF with a greater prevalence in the distal extremities. 25
Skeletal muscle is supplied by motor, sensory, and autonomic nerves, and rare reports have shown similar pathologic changes in intramuscular nerve fibers as in the peripheral nerve trunks in hereditary neuropathy with pressure palsy 29 and congenital hypomyelination neuropathy. 27
This paper reports, for the first time, selective damage to peripheral nerves in the demyelinating polyneuropathy produced in young, rapidly growing chickens by riboflavin deficiency.
Footnotes
Acknowledgement
We thank Inghams Enterprises, Adelaide, Australia, for support of this study.
