Abstract
In recent years there has been considerable interest in telomerase as a target for therapeutic intervention in oncology. This largely stems from the vast number of studies that have demonstrated expression and activity of the enzyme telomerase in the majority of human cancer tissues with little or no activity detectable in normal somatic tissues. These studies have led to an interest in the role of telomerase in cancers associated with domesticated species, in particular tumors that affect dogs. This article reviews the biology of telomerase and the biological significance of telomerase activity in canine tumors and discusses the clinical implications of telomerase expression in canine cancers with regard to therapeutics and diagnostics.
Keywords
Introduction
Cells of multicellular organisms form part of a specialized society that cooperates to promote survival of the organism. In this, cell division, proliferation, and differentiation are strictly controlled and a balance exists between normal cell birth and the natural cell death rate. Derangement of these normal homeostatic mechanisms can lead to uncontrolled proliferation leading to a malignant phenotype.
The advances in our understanding of normal cell biology and the processes that lead to malignancy have increased dramatically during the past 30 years. In particular, the last decade has shown us that transformation of a normal cell into a malignant cell requires very few molecular, biochemical, and cellular changes that can be considered as acquired capabilities.
17
Furthermore, despite the wide diversity of cancer types, these acquired capabilities appear to be common to all types of cancer. Tumorigenesis is a multistep process, and these steps reflect genetic alterations that drive the progression of a normal cell into a highly malignant cancer cell. This is supported by the finding that genomes of tumor cells are invariably altered at multiple sites. The spectrum of changes range from subtle point mutations in growth regulatory genes to obvious changes in chromosomal complement. Cancer cells have defects in regulatory circuits that govern cellular proliferation and homeostasis. It has been suggested that the vast array of cancer genotypes is a manifestation of only six alterations in cellular physiology that collectively dictate malignant growth. These acquired characteristics can be summarized as:
self-sufficient growth; insensitivity to antigrowth signals; evasion of programmed cell death (apoptosis); limitless replicative potential; sustained angiogenesis; tissue invasion and metastasis.
In this article we will focus on the area of limitless replicative potential or how the cancer cell is able to overcome mortality and cellular senescence and take on the immortal phenotype. Furthermore, we will discuss how an increased understanding of these mechanisms lead to the identification of novel targets for diagnosis and therapy.
Cellular Aging and Senescence: The Role of Telomeres
Normal cells, at least in vitro, have a defined lifespan, replicating for a limited number of divisions, after which they undergo an irreversible growth arrest termed replicative or cellular senescence. 21 In contrast, cancer cells and immortalized cells can proliferate indefinitely and can therefore bypass the growth arrest that ensues with increased cell divisions. These observations have formed the basis for a dearth of studies investigating the mechanisms that control cell aging and senescence, with the view that these mechanisms are likely to be abrogated in cancer cells and may therefore identify novel therapeutic targets.
The mechanisms of cellular senescence have been extensively investigated in human cells, and it is now established that the limited replicative potential of cells is in part controlled by telomeric DNA. Telomeres are specialized DNA-protein complexes that cap the ends of linear chromosomes. In vertebrates, telomeres consists of tandem repeats of the sequence TTAGGG and a number of telomere-associated proteins. 3 Although the length of telomeric repeat sequences varies between species (e.g., human telomeres range between 5 and 20 kbp, whereas mice telomeres can extend up to 60 kbp) their functions remain the same. Telomeres appear to be important in maintaining genomic integrity by protecting chromosome ends from recombination, fusion, and from being recognized as damaged DNA. In addition to their protective role, telomeres are implicated in the cellular aging process.
As a result of an inherent ability of DNA polymerases to inefficiently replicate the 5′ ends of linear chromosomes, with each round of cell division approximately 50–200 bp of telomeric DNA is lost, a phenomena known as the “end replication problem.” 23 This leads to progressive telomeric attrition as cell division proceeds and shortening to a critical length is proposed to act as a trigger for cell growth arrest and cellular senescence. 18–20 Consistent with this model, telomere shortening has been observed during in vivo aging of normal somatic tissues as well as in in vitro cultured human fibroblasts. 8,40 Cellular senescence has therefore been proposed to act as a tumor suppressor mechanism, limiting cell division and preventing the accumulation of potentially oncogenic mutations.
Telomerase: Circumventing Senescence
Although the replicative potential of most human somatic cells is limited, some cells have evolved mechanisms to circumvent telomeric attrition, thereby bypassing senescence and allowing for continued proliferation. The enzyme telomerase provides the facility by which such cells overcome telomeric instability. 32 Telomerase is a ribonucleoprotein complex consisting of an RNA subunit that contains a domain complementary to the telomeric repeat sequence TTAGGG and a catalytic protein component. 11,31,33 The protein component acts as a reverse transcriptase and can catalyze the addition of telomeric repeats onto the ends of chromosomes using the RNA subunit as a template (Fig. 1).
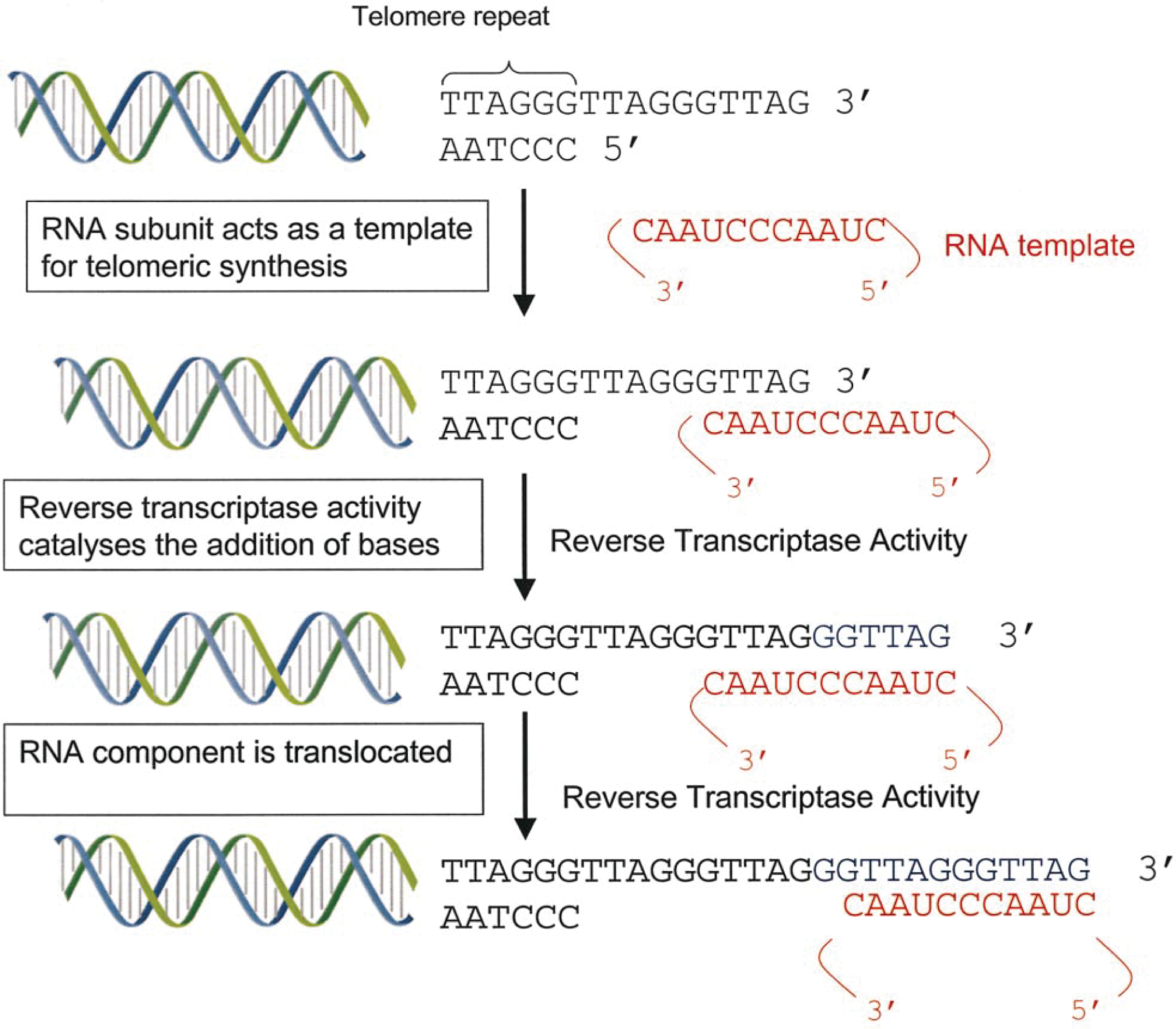
Function of telomerase; telomerase reverse transcriptase subunit catalyses the addition of TTAGGG repeats to chromosome ends using the RNA subunit as a template.
The development of the highly sensitive telomere repeat amplification protocol (TRAP) assay in 1994 has enabled the assessment of telomerase activity in a wide range of tissues. 26 The assay is a polymerase chain reaction–based protocol and is depicted diagrammatically in Fig. 2. The assay has recently been modified to allow for the quantitative assessment of telomerase using enzyme-linked immunosorbent assay technology. Studies using the TRAP assay have shown that expression of telomerase is strictly regulated, at least in human tissues. Telomerase activity is essential for embryogenesis but is repressed upon tissue differentiation during development such that telomerase is absent from birth in most somatic tissues. 41 Some cell types including male germ cells, activated lymphocytes, and stem cell populations continue to express telomerase at reduced levels. 5,10,22 However, the majority of immortalized cells express telomerase, and one of the most exciting findings in recent years is the identification of telomerase activity in greater than 85% of human cancers, encompassing a broad range of cancer types, with little or no activity in most normal somatic tissues. 39 The expression of telomerase in a wide range of human cancers is of major interest in terms of cancer therapy and a number of telomerase-based strategies are currently under development.
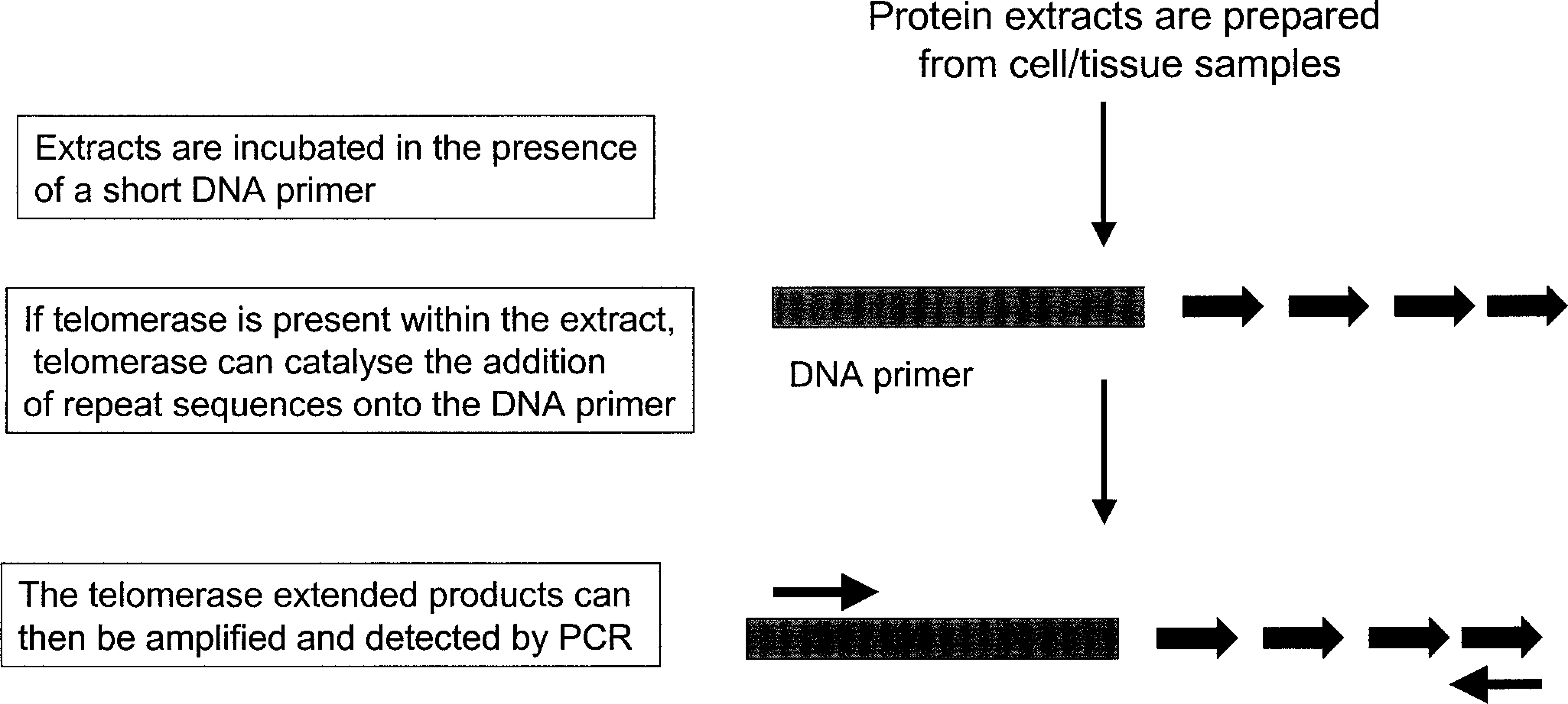
Measurement of telomerase activity using the TRAP assay.
Telomerase in Canine Cancer
It is becoming increasingly clear that many of the basic biological processes in oncology are conserved among mammalian species and as a result the discipline of comparative oncology is rapidly growing. Indeed, the canine cancer patient represents a useful model for human cancer studies because many of the cancers encountered in canines show similarities with human tumors in terms of histopathology, biological behavior, response to therapy, for example. Cancers in dogs are of importance in terms of canine health and welfare, and therefore there is a requirement to further investigate the basic biology of canine cancers and apply comparative biological approaches to identify targets of therapy that will be of benefit to the canine cancer patient. A number of investigators are now focusing their attention on telomerase and its role in canine oncology in an attempt to discern whether telomerase is ubiquitously expressed in canine tumors and may represent a promising target for therapeutic intervention. 2,13,34
Canine telomeres consist of large heterogeneous repeats ranging from 3 to 23 kbp in length. 34,42 Telomeric attrition has not only been demonstrated during in vitro aging of canine fibroblast cultures but telomeric instability has also been documented in vivo. 30,34,42 The presence of telomerase activity in canine tumor tissues has been reported by a number of investigators. 2,13,42,43 Taken together, these four studies have analyzed over 100 canine solid tumors for telomerase activity and have demonstrated that more than 95% of all canine cancers are associated with telomerase activity. Table 1 summarizes the telomerase assessment of canine tumors from three studies. 13,42,43 Although it is not possible to directly compare levels of telomerase activity among these studies, it is clear from these studies that the majority of canine tumors express telomerase. In the above mentioned studies telomerase activity was not detected in the majority of normal canine tissues. A more extensive examination of telomerase expression distribution has recently been reported. 34 In this study, telomerase activity from a panel of normal tissues including lung, brain, liver, muscle, and heart tissues from three dogs with malignancy was evaluated. Telomerase activity was confined to tumor tissues with little or no activity in normal somatic tissues. Telomerase activity, however, has been reported in canine lens epithelial cells of normal and cataractous lenses and testis tissues. 9,34 Germ cells proliferate indefinitely and are therefore expected to contain telomerase activity. In summary, the telomerase profile in differing canine somatic tissues mimics that of humans, and these studies clearly demonstrate that telomerase activity is not only the most widely expressed human tumor marker but also the most widely recognized canine tumor marker currently known.
A review of telomerase activity in canine tumor samples (data has been compiled from three studies.13,42,43

Clinical Implications of Telomerase
Because telomerase is active in the vast majority of human cancers, there is considerable focus on how telomerase may be exploited in the diagnosis and treatment of cancer. Interest in this field had been driven by the demonstration that induced expression of telomerase results in extension of cellular lifespan and malignant transformation. 16 Although some fundamental questions remain to be established in relation to canine malignancies, the potential diagnostic and clinical benefits of telomerase targeting protocols to the canine cancer patient will be reviewed.
Telomerase as a Diagnostic Tool in Clinical Oncology
The near universal expression of telomerase in cancer cells has made this enzyme the most prevalent tumor marker. Consequently, the detection of telomerase activity using the TRAP assay may support a diagnosis of cancer. 29 The TRAP technique is able to detect activity not only in solid biopsies and whole tumor specimens but also in tissue aspirates and malignant cytological specimens. For example, telomerase activity has been detected in urine samples from bladder cancer patients, bronchial lavage samples from lung cancer patients, and colonic washes from colorectal cancer patients. 1,24,45 Although many studies have shown variable success in establishing diagnostic role for telomerase activity, largely because of the infiltration of normal telomerase positive cells such as lymphocytes, it is possible that telomerase levels may emerge as a useful diagnostic marker for particular cancer types. Telomerase activity has been detected in negative cervical smear samples associated with human papillomaviral infection, similarly higher levels of telomerase activity have been detected in malignant pancreatic cancers compared with benign lesion. 22,46 Furthermore, telomerase activity may play a role in establishing prognosis. The prognostic value of telomerase activity has been evaluated in many studies and shown to be of prognostic value in a few cancer types including neuroblastomas. 37 It is clear that careful controlled studies in companion animals need to be performed to evaluate the potential prognostic/diagnostic role of telomerase in canine cancers.
Telomerase as a Therapeutic Target
Telomerase inhibition
Many of the strategies targeting telomerase are based on the identification of agents that inhibit the reverse transcriptase activity of telomerase. 3′-azido-2′, 3-dideoxythymidine (AZT) is a reverse transcriptase inhibitor that previously has been used in the treatment of acquired immunodeficiency syndrome and has been shown to inhibit telomerase activity in cancer cells. 14 Antisense expression directed against the RNA template in human cell lines has also demonstrated repression of telomerase activity and telomere instability as have peptide nucleic acids. 35,36 The use of hammerhead ribozymes that cleave the telomerase RNA component has also been shown to inhibit telomerase activity. 44 In the majority of telomerase inhibition studies, telomere inhibition results in telomere instability and cell death.
Although in vitro telomerase inhibition studies have shown promising results, telomerase inhibition represents a challenge within the clinical setting. 6 Inhibiting telomerase activity in cancer cells will prevent telomere lengths from being maintained, however, cells will need to undergo sufficient cell divisions with telomere shortening to a critical length to trigger cell death. This lag phase will be sufficient to allow tumor volume to dramatically increase, and is therefore unlikely to be of benefit to cancer patients with large tumor burden. Inhibition studies, however, may offer potential benefits as adjuvant therapy in combination with current therapeutics (chemotherapy, radiation therapy, and surgery) to specifically target residual disease or where micrometastatic disease is suspected. Further combinations of novel compounds such as a telomerase inhibitor and an angiogenesis inhibitor may offer a greater likelihood of success. Tumor cells with relatively short telomere lengths are expected to respond faster to inhibition and cause cell death than cells with longer telomeres, therefore exploitation of telomerase requires careful selection of patients who may benefit from the treatment.
The chemotherapeutic agent, Cisplatin, has been shown to potently inhibit telomerase activity and induce telomere loss in human testicular cancer cells. 7 Although Cisplatin cannot be considered a selective telomerase inhibitor, it may prove useful in the primary treatment of tumors followed by nontoxic telomerase inhibitors administered for a longer period of time, thereby overcoming the deleterious effects of long-term Cisplatin administration. Cisplatin is a commonly used anticancer drug in canine oncology, and the investigation of the synergistic relationship between Cisplatin and telomerase inhibitors in canine cancer cells requires to be established.
Telomerase Promoter Targeting
A different approach to the development of anti-telomerase therapies may be offered by gene therapy. In this, instead of trying to inhibit telomerase per se, gene therapy capitalizes on the expression of telomerase to target expression of a novel transgene that can either kill the cell or enhance the antitumor immune response to the cancer cell. Strategies may therefore deliver therapeutic genes to cancer cells that are under the control of one of the telomerase promoter elements. Although the RNA subunit is ubiquitously expressed in most human normal somatic tissues, TERT expression is mainly confined to malignant tissues. This differential expression has led to a number of investigations of the ability of telomerase reverse transcriptase promoter elements to direct transgene expression specifically to tumor cells. 4,15,27,28 Indeed, these studies have demonstrated that the human telomerase promoter is sufficient to target cancer cells specifically.
Canine Telomeres and Telomerase: The Outstanding Questions
Several of the telomerase-based strategies are now in preclinical human trials and it is anticipated that telomerase cancer therapies will enter clinical phase II/III trials in the near future. Although human cancer researchers and clinicians are beginning to unravel the complexities of telomerase regulation and evaluating the realistic opportunities for telomerase cancer therapy, our understanding of the role telomerase plays in canine cancer is currently beginning to emerge and is likely to form an increasing focus of research attention. However, there are some fundamental issues that we must consider before the applicability of telomerase inhibition, cancer therapies, or gene-directed therapies become a reality in veterinary clinical practice.
Firstly, a full-scale analysis of TERT (catalytic component) and TR (RNA component) expression in canine tissues has yet to be performed to elucidate the mechanisms of telomerase regulation in the dog. These studies have, in part, been hampered by the lack of data regarding the specific canine gene sequences. Furthermore, we must also consider the potential problems of anti-telomerase therapies. We have already discussed the potential lag phase associated with anti-telomerase therapies. In addition to this we must also consider those tumors that remain immortal but lack telomerase activity. These tumor cells are thought to maintain their telomeric lengths by a different pathway known as alternative lengthening of telomeres (ALT). 11 This is a poorly understood pathway but may involve recombination events between chromosomes. It is possible that these tumors would not respond to anti-telomerase therapies and so determination of telomerase status before therapy would be essential. Indeed, the long-term toxicity on normal telomerase positive cells also requires consideration. However, based on human studies, the telomerase expression difference between normal and cancer cells is large, and it has been proposed that toxicity on normal cells will be less than with the use of conventional anticancer drugs. 39 Furthermore, the development of these therapies and other nonconventional drugs may force us to reevaluate how we assess the success of a drug in terms of phase I/II and III clinical trials. In conventional drug testing, these studies are used to evaluate toxicity and pharmacokinetics, the endpoints often being a comparison with existing therapies and objective tumor responses. Considering the potential lag phase of telomerase inhibitors, it will be necessary to redefine these trials and consider other biological endpoints. 25
From these discussions it is clear telomerase represents an exciting area of cancer research, but true evaluation of this enzyme in diagnosis and therapy offers many challenges that will require collaboration across the disciplines of basic science and clinical medicine. However, results from preclinical evaluations are very positive and we anticipate the introduction of such therapies into the veterinary arena within the next 10 years.
