Abstract
Immunostaining with monoclonal antibody (MoAb) hepatocyte paraffin 1 (Hep Par 1) and an MoAb to cytokeratin 7 (CK7) was performed on 105 formalin-fixed, paraffin-embedded canine hyperplastic and neoplastic hepatic lesions. Hep Par 1 was detected in 12/12 hyperplastic nodules, 17/17 hepatocellular adenomas, and 37/40 hepatocellular carcinomas. The staining was disseminated, granular, and cytoplasmic. This antibody did not react with normal or neoplastic biliary epithelium. Other hepatic tumors or tumors metastatic to the liver did not bind Hep Par 1 except one metastatic intestinal carcinoma. MoAb to CK 7 stained all hyperplastic biliary epithelium and benign cholangiocellular tumors (5/5) and 14/18 cholangiocellular carcinomas. One hepatocellular carcinoma had cells positive for both Hep Par 1 and CK 7. Liver was the only normal tissue tested that reacted with MoAb Hep Par 1. Only five nonhepatic tumors (one adrenocortical carcinoma, one interstitial cell tumor of the testis, one melanoma, and two salivary adenocarcinomas) of 277 tumors tested had focal/multifocal staining for Hep Par 1. Prolonged fixation did not alter the staining with Hep Par 1. We conclude that Hep Par 1 is a specific and sensitive marker for canine hepatocellular tumors and allows distinction between hepatocellular and biliary neoplasms.
Keywords
Primary hepatic tumors account for 0.6–1.3% of all canine neoplasms 13,21 and include hepatocellular adenoma, hepatocellular carcinoma, cholangiocellular (bile duct) adenoma, and cholangiocellular carcinoma. 4,14,16,22 Other primary hepatic tumors or proliferative lesions are hyperplastic nodules, carcinoids, and sarcomas. 2,13,15 The liver is a common site of tumor metastasis because of its dual blood supply of the hepatic artery and portal vein. 14,20 Distinction of primary from metastatic hepatic tumors may be difficult using routine histologic stains, especially when a detailed clinical examination has not ruled out other tumor sites. However, this distinction is necessary because treatment for primary or metastatic tumors is different. Treatment of primary hepatic cancer is mainly by tumor resection, if possible. However, metastatic tumors may require a different therapeutic approach depending on their origin. Furthermore, the prognoses of primary and metastatic liver tumors are very different; thus, diagnostic accuracy is essential.
In humans, immunohistochemical techniques have been used extensively on hepatic neoplasms. The markers most commonly used are α-fetoprotein (AFP) and carcinoembryonic antigen (CEA); other markers are ferritin, albumin, α-1-antitrypsin, factor XIIIa, and cytokeratins (CKs). 1 AFP is present in hepatocellular tumors but not in bile duct epithelium. 7 CEA is present in both bile duct epithelium and hepatic cells, although with a different pattern. 10 Normal hepatocytes express keratins 8 and 18 (as defined in Moll's catalogue), whereas bile duct and gallbladder epithelial cells contain cytokeratins 7, 8, 18, and 19. 24 Recently, a monoclonal antibody (MoAb) against an hepatocyte antigen, hepatocyte paraffin 1 (Hep Par 1), has been used to distinguish hepatocellular from biliary tumors 9,11,27,28 and to detect hepatoblastomas. 6 Immunohistochemical characterization of canine hepatic neoplasms, including use of CKs, AFP, CEA, and vimentin, has been reported. 5 However, the immunohistochemistry of canine hepatic neoplasms has not been examined in a large series of tumors.
In this study, we tested primary hepatic tumors with MoAb Hep Par 1 and an MoAb to cytokeratin 7 on formalin-fixed, paraffin-embedded material. To determine the specificity of Hep Par 1, we also immunostained a variety of nonhepatic tumors and normal canine tissues. Immunostaining with MoAb Hep Par 1 has not been reported before in animal tissues.
Materials and Methods
Selection of cases
A search of the Veterinary Medical Diagnostic Laboratory database produced a total of 110 cases with a diagnosis of hyperplasia or neoplasia of hepatocellular or bile duct origin. Cases were reviewed by one of the authors (J. A. Ramos-Vara) to determine their suitability for the study. Cases with poorly preserved samples or no tissue remaining were excluded. Those cases in which the morphologic diagnosis was not definitive were reviewed by other pathologists (M. A. Miller, G. C. Johnson) to establish consensus, and 93 cases were selected for the study (Table 1). An additional 11 hepatic tumors not derived from hepatocellular or bile duct epithelium or metastatic tumors to the liver and one hamartoma were selected (Table 1). To demonstrate the specificity of the Hep Par 1 antibody, 277 extrahepatic tumors (Table 2) were also immunohistochemically stained. Specimens had been fixed in 10% formaldehyde for an undetermined period of time prior to being embedded.
Immunohistochemical staining in formalinfixed, paraffin-embedded hepatic hyperplastic and neoplastic canine lesions with antibodies to human hepatocyte (Hep Par 1) and to CK 7.
∗ One tumor not tested.
† Two tumors not tested.
Immunohistochemical staining in formalinfixed, paraffin-embedded nonhepatic canine tumors, nonhepatic canine lesions, and normal canine tissues with antibody to human hepatocyte (Hep Par 1).

∗ Focal but strong staining.
† Multifocal, usually individual cells.
‡ Hepatocytes only.
Reagents and immunohistochemical staining procedure
For immunohistochemistry, 5-µm-thick sections were placed on positively charged slides and dried. Two antibodies were used: MoAb anti-human hepatocyte (hepatocyte paraffin 1, clone OCH1E5, Dako Corporation, Carpinteria, CA) and MoAb to cytokeratin 7 (clone OV-TL 12/30, Dako Corporation). In a pilot study, we determined the optimal antigen retrieval conditions and the dilution of each primary antibody. For Hep Par 1, three different antigen retrieval methods (proteinase K, heat epitope retrieval with citrate buffer, pH 6.0, and heat epitope retrieval with ethylenediaminetetraacetic acid [EDTA] buffer, pH 8.0) were compared with no treatment, following reported methods. 18 For cytokeratin 7 (CK 7), two pretreatments (proteinase K and heat epitope retrieval with citrate buffer at pH 6.0), in addition to the untreated sections, were used. The optimal concentration after serial dilutions was 1/200 (0.625 µg of mouse IgG/ml of diluent) and 1/75 (2.73 µg of mouse IgG/ml of diluent) for Hep Par 1 and CK 7, respectively. The optimal antigen retrieval for Hep Par 1 was incubation in a steamer with EDTA buffer (pH 8.0) for 20 minutes at 90–95 C. Treated slides were cooled at room temperature for 20 minutes. For CK 7, slides were pretreated with proteinase K following the manufacturer's recommendations. 17 Immunohistochemical staining was performed using an automatic stainer (Autostainer, Dako Corporation). For Hep Par 1 immunostaining, a non–avidin–biotin, polymer-based method was used (EnVision+ peroxidase, Dako Corporation). For CK 7, a labeled streptavidin–biotin method was used (LSAB+ peroxidase, Dako Corporation). The primary antibodies were incubated at room temperature for 1 hour. Secondary and tertiary components of the immunohistochemical reaction were from a ready-to-use kit and were incubated following the manufacturer's recommendations. The immunohistochemical reaction was demonstrated using diaminobenzidine as chromagen and hydrogen peroxide as substrate. Slides were counterstained with Mayer's hematoxylin. An unrelated MoAb of the same isotype and similar protein concentration replaced the primary MoAb to confirm staining specificity.
Scoring of immunohistochemical results with MoAb Hep Par 1 and MoAb to CK 7
A semiquantitative evaluation of the percentage of positive tumor cells was used: 1 = 5–10% of positive cells; 2 = 11–50%; 3 = 51–80%, and 4 = >80%. The intensity of reaction for both MoAbs was graded on a scale of 0 (no reaction), 1 (weak reaction), 2 (moderate reaction), 3 (intense reaction), and 4 (very intense reaction).
Effect of prolonged fixation for detection of Hep Par 1 and CK 7
Because the fixation time of most tumors was unknown, normal liver was fixed for 24 hours to determine the effects of prolonged fixation on immunohistochemistry. Hep Par 1 stained hepatocytes, and CK 7 stained bile duct epithelium. Additional samples from this tissue were fixed for 2, 3, 4, 5, 6, 7, 10, 13, 17, and 24 days before processing.
Results
Immunostaining with MoAb Hep Par 1 in normal liver and hepatic neoplasms
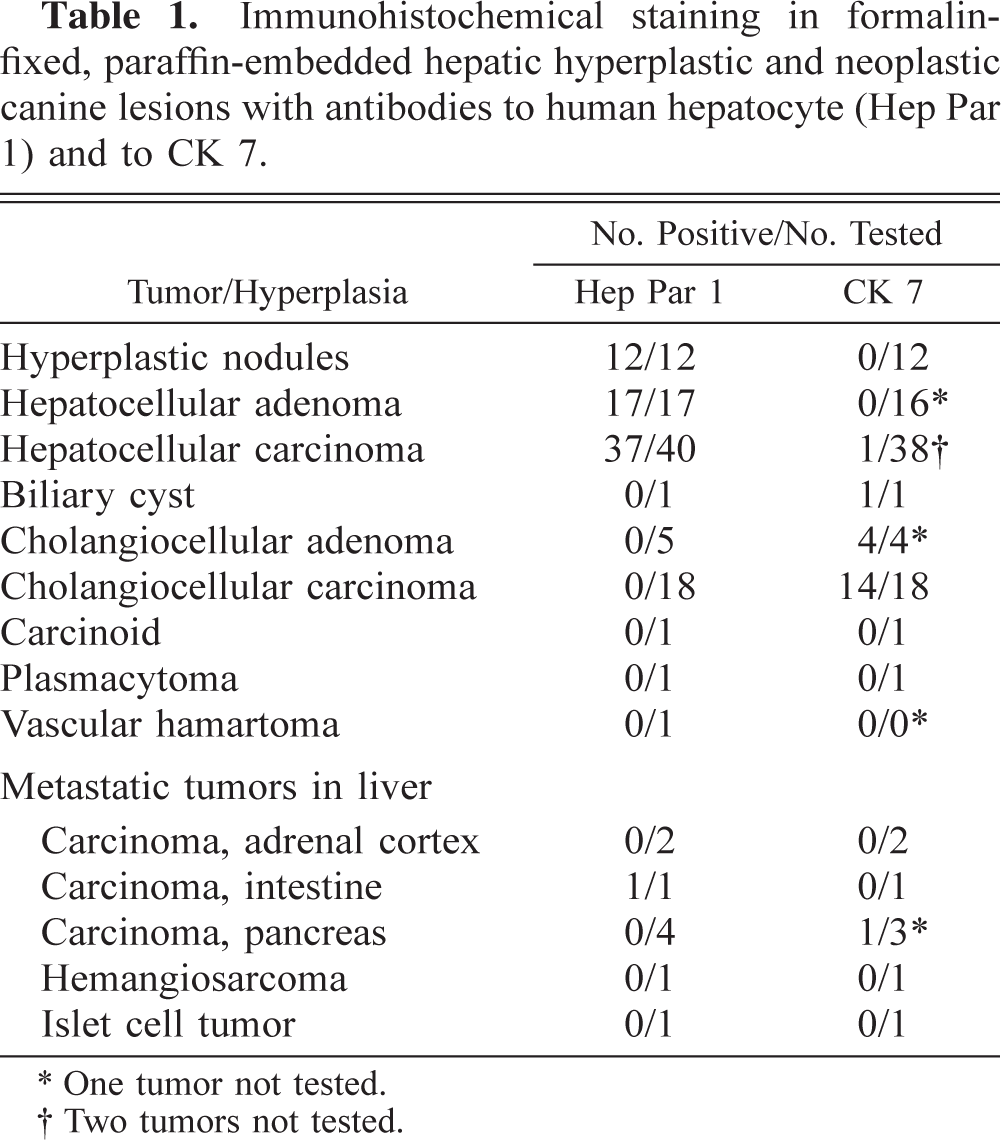
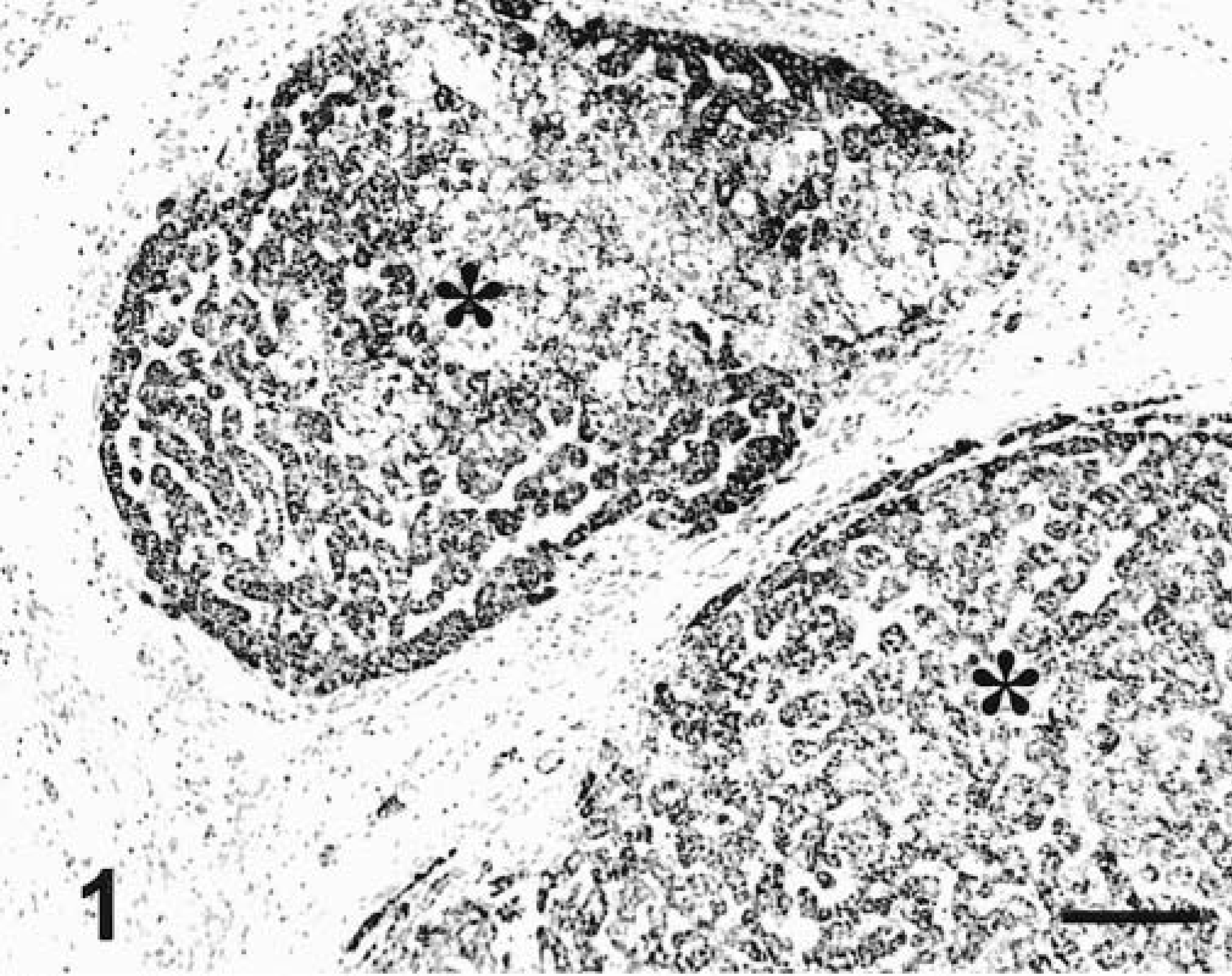
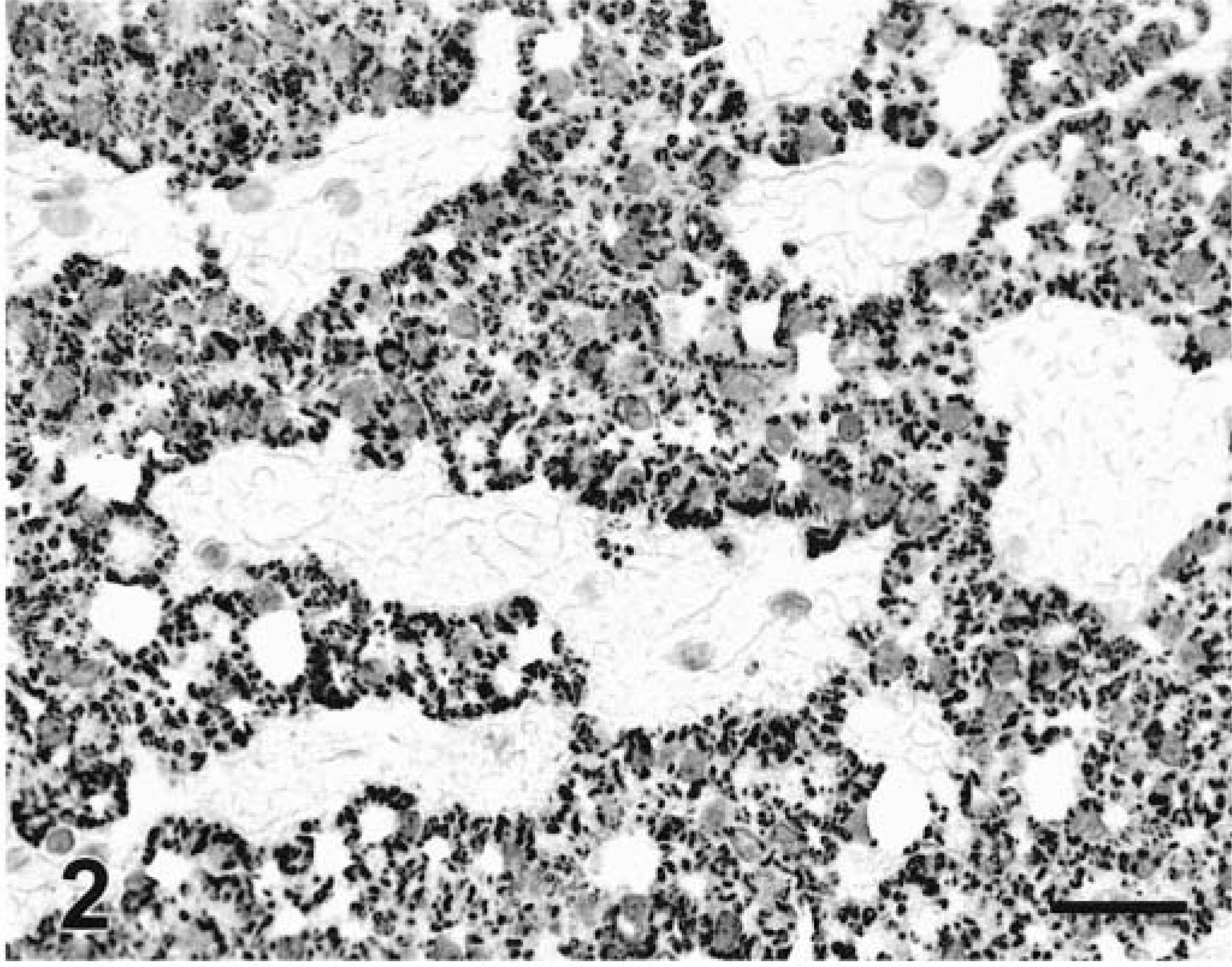
The staining of normal hepatocytes with MoAb Hep Par 1 was consistently cytoplasmic, disseminated, and granular. Bile duct epithelium and other hepatic tissues did not react with Hep Par 1. Differences in staining intensity among zones of the hepatic lobule were not apparent. Hep Par 1 was detected in 12/12 (100%) hyperplastic nodules (Fig. 1), 17/17 (100%) hepatocellular adenomas, 37/40 (92.5%) hepatocellular carcinomas (HCC) (Figs. 2–4), 0/1 biliary cysts, 0/5 cholangiocellular adenomas, and 0/18 cholangiocellular carcinomas (CCC) (Table 1). The staining was disseminated through the cytoplasm, sometimes involving most neoplastic cells but also with a multifocal distribution. More than 50% of cells were positive in 12/12 hyperplastic nodules, 12/17 hepatocellular adenomas, and 31/37 HCC. The intensity of the reaction was high or very high (grades 3 or 4 of 4) in 12/12 hyperplastic nodules, 17/17 hepatocellular adenomas, and 34/40 HCC. Vacuolated hepatocytes also had a ringlike staining pattern surrounding the vacuole, which remained unstained. Within the same tumor, the staining intensity varied widely in many cases. Differences in the intensity of the reaction were not apparent among different types of tumors or degrees of cellular differentiation.
Liver; dog. Hyperplastic nodules (asterisks) are diffusely stained with MoAb Hep Par 1. EnVision +–peroxidase stain, Mayer's hematoxylin counterstain. Bar = 60 µm.
Liver; dog. Well-differentiated HCC. Granular and diffuse cytoplasmic staining with MoAb Hep Par 1. EnVision +–peroxidase stain, Mayer's hematoxylin counterstain. Bar = 30 µm.
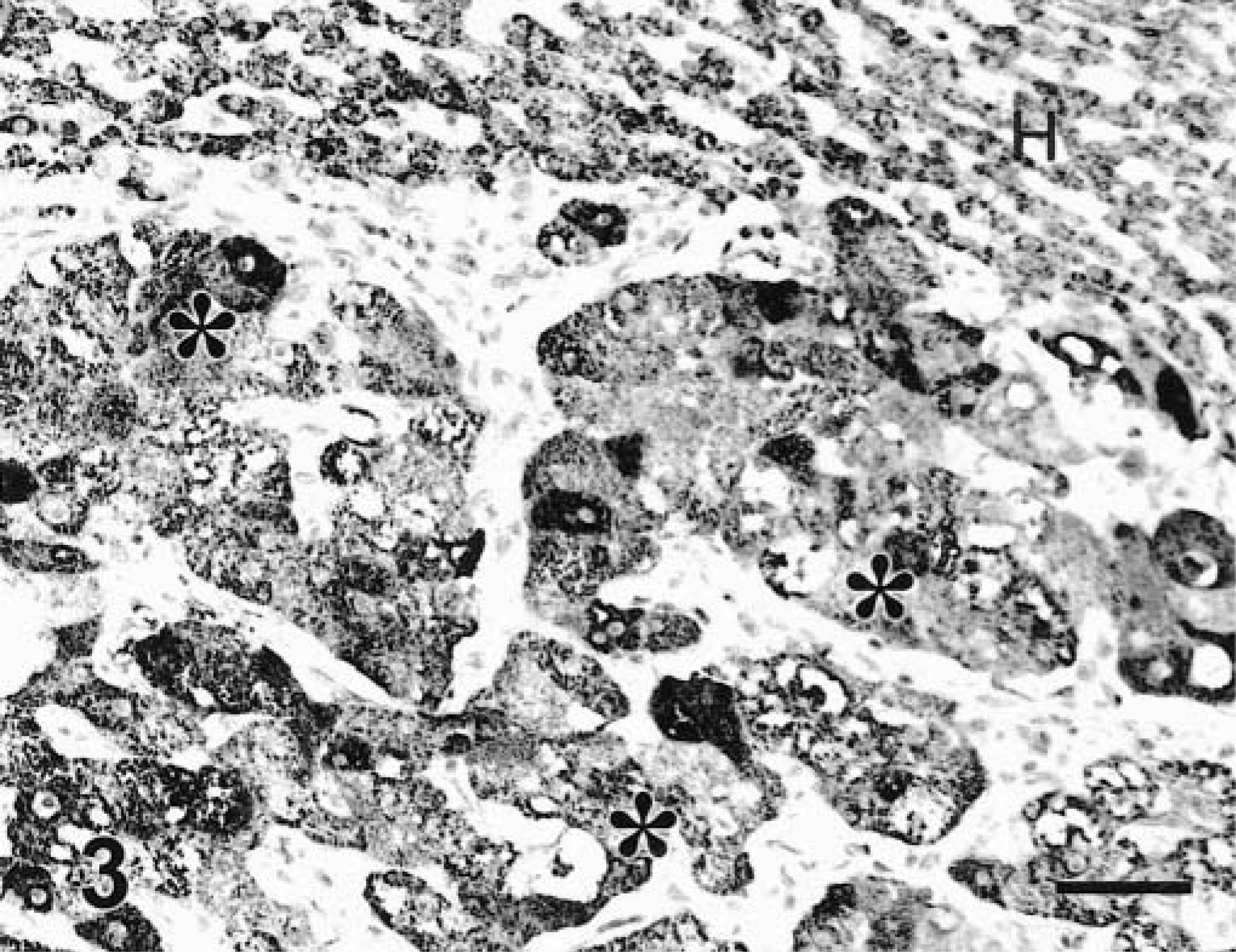
Liver; dog. HCC. Atypical hepatocytes (asterisks) and adjacent normal hepatocytes (H) strongly react with MoAb Hep Par 1. EnVision +–peroxidase stain, Mayer's hematoxylin counterstain. Bar = 30 µm.
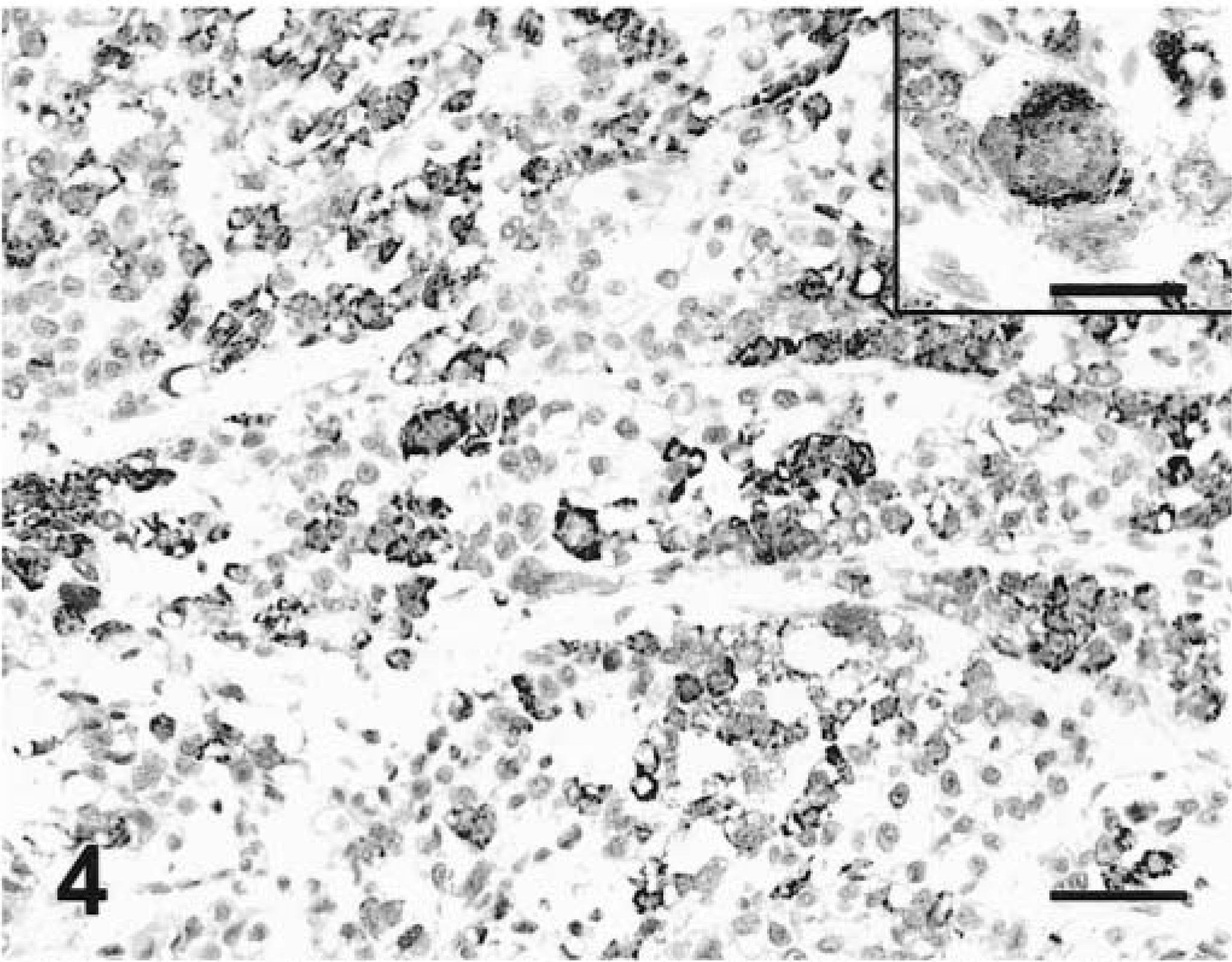
Liver; dog. Poorly differentiated HCC. Variable numbers of cells are positive with MoAb Hep Par 1. EnVision +–peroxidase stain, Mayer's hematoxylin counterstain. Bar = 60 µm. Inset: Cytomegalic hepatocyte reacts with MoAb Hep Par 1. EnVision +–peroxidase stain, Mayer's hematoxylin counterstain. Bar = 30 µm.
Immunostaining with MoAb to CK 7 in normal liver and hepatic neoplasms
Normal liver had CK 7 within the cytoplasm of bile duct epithelium. Other liver cells did not react with MoAb to CK7. The staining was more intense adjacent to the plasma membrane and was sometimes patchy, with some bile ducts strongly stained and others not stained. There was no apparent correlation between diameter of the bile duct and detection of CK 7. CK 7 was detected in the only biliary cyst, 4/4 (100%) cholangiocellular adenomas (Fig. 5), 14/18 (77.8%) CCC (Fig. 6), and none of the lesions with hyperplastic or neoplastic hepatocytes except for 1/38 HCC in which the staining was multifocal. The staining in some CCC was distinctly focal and perinuclear (Fig. 7). One tumor had cells positive for both Hep Par 1 and CK 7. It was not clear whether some CK 7–positive cells coexpressed Hep Par 1 in this tumor. More than 50% of cells were positive in the single biliary cyst, 4/4 cholangiocellular adenomas, and 10/14 CCC. The reaction was intense or very intense (grades 3 or 4 of 4) in 4/4 cholangiocellular adenomas and 13/18 CCC.
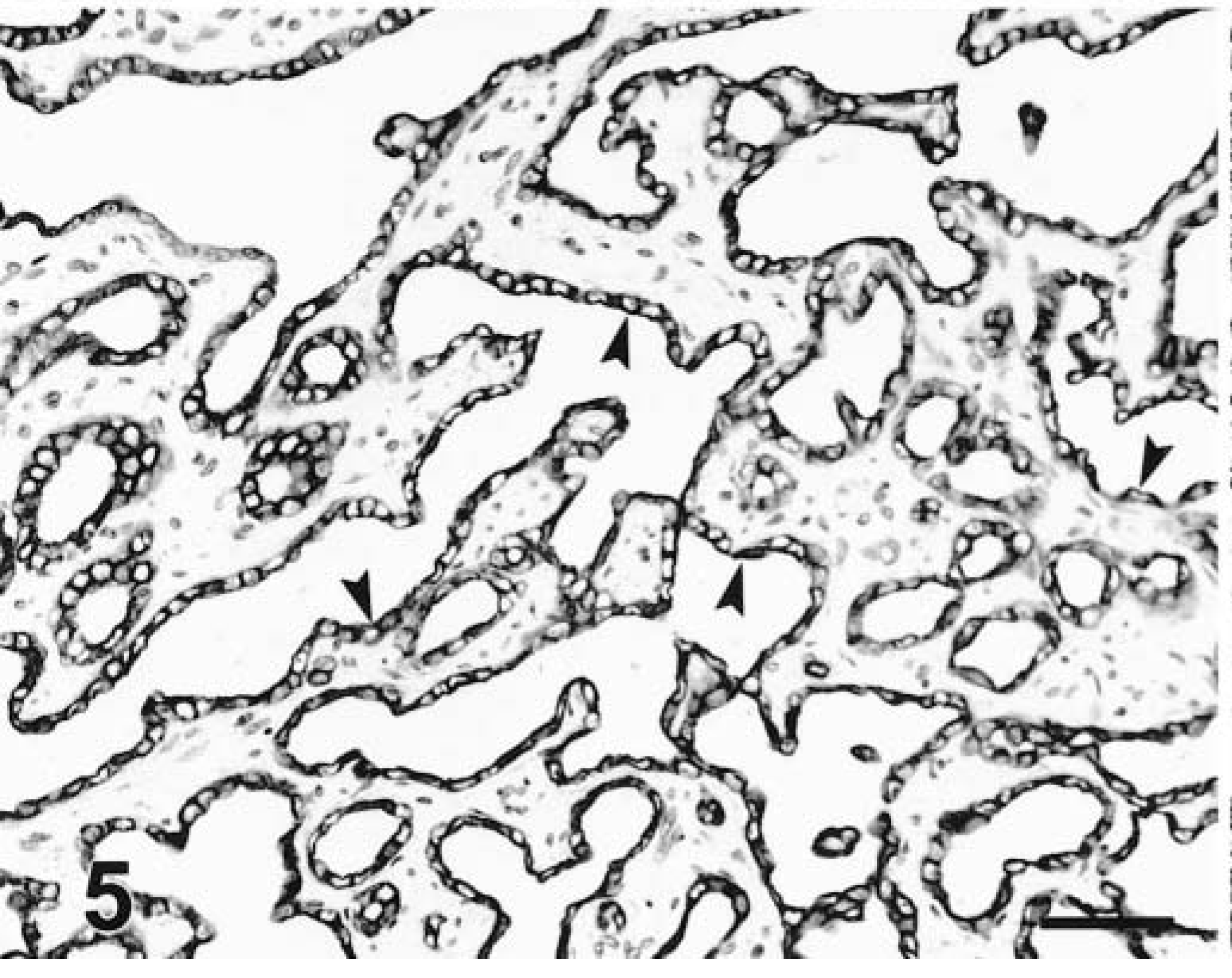
Liver; dog. Cholangiocellular adenoma. Strong cytoplasmic staining to CK 7 in biliary epithelium (arrowheads). LSAB+–peroxidase stain, Mayer's hematoxylin counterstain. Bar = 30 µm.
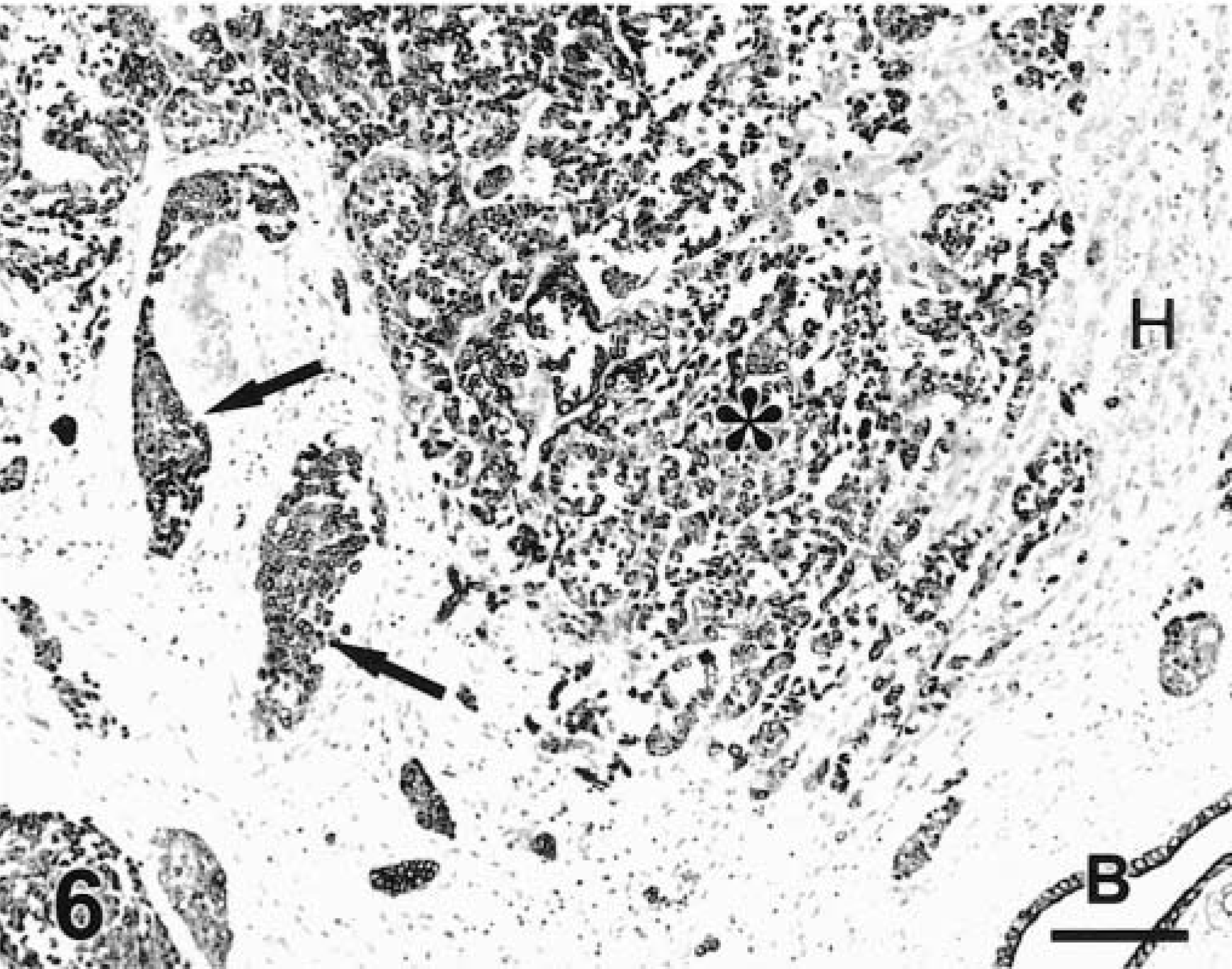
Liver; dog. CCC. Tumor cells infiltrating the parenchyma (asterisk) and within vessels (arrows) are strongly positive to CK 7. A normal bile duct (B) is also positive. Hepatocytes (H) are negative. LSAB+–peroxidase. Bar = 60 µm.
Immunostaining with MoAb Hep Par 1 in normal tissues and nonhepatic neoplasms
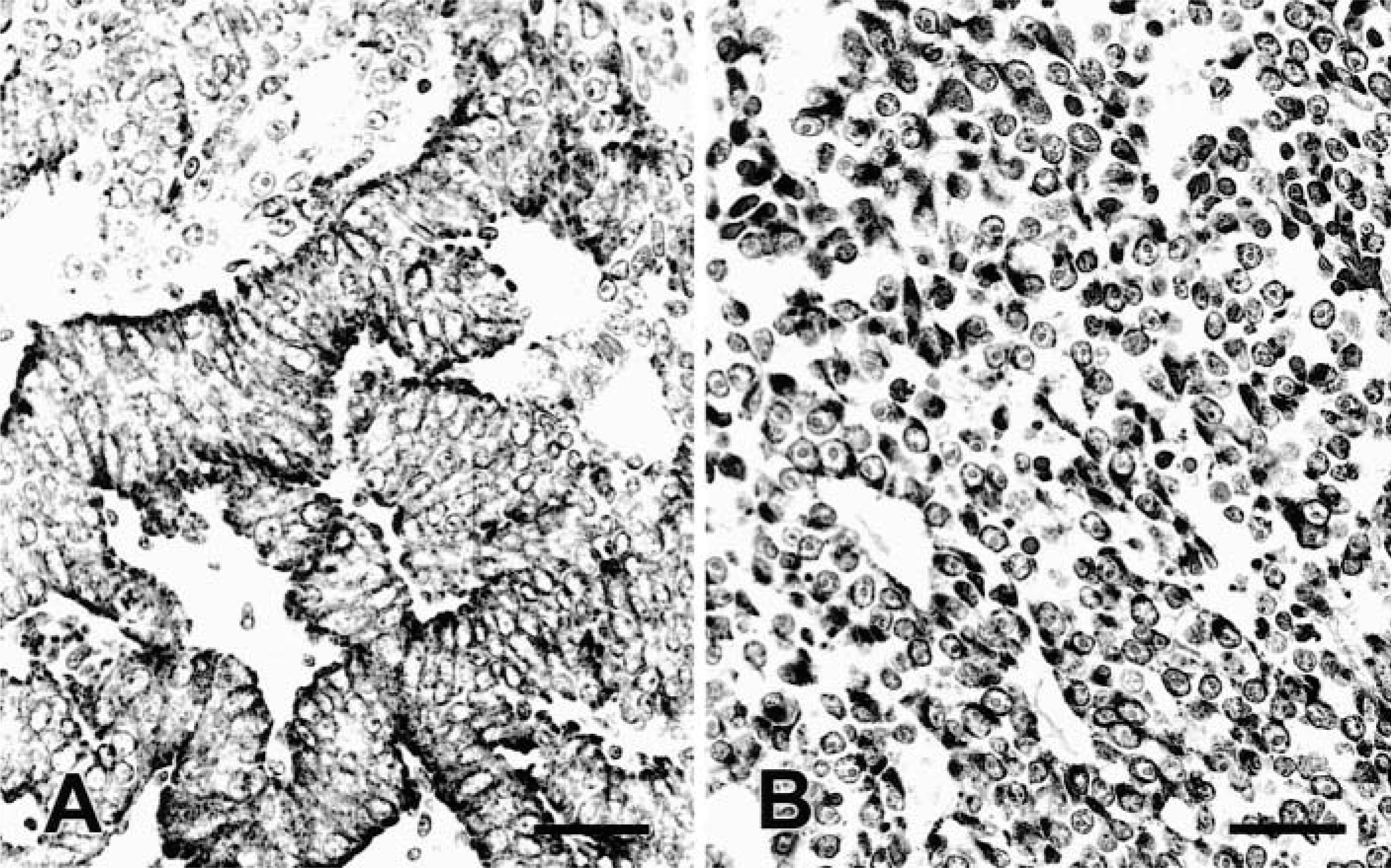
The only normal canine tissue with a positive reaction to Hep Par 1 was the liver (Table 2). Two hundred seventy-seven tumors, including adrenocortical tumors, pheochromocytomas, thyroid tumors, cutaneous (apocrine gland, basal cell, hair follicle, sebaceous gland, perianal gland) tumors, ovarian tumors, respiratory tract tumors, gastric tumors, testicular (interstitial cell, seminoma, Sertoli cell) tumors, mammary gland tumors, mesothelioma, pancreatic exocrine tumors, prostatic tumors, salivary gland tumors, peripheral nerve sheath tumors, transitional cell tumors, squamous cell carcinomas, chemodectoma, fibrosarcomas, hemangiopericytomas, histiocytomas, hemangiosarcomas, melanoma, myxosarcomas, mast cell tumors, neurofibrosarcomas, osteosarcomas, plasmacytomas, and schwannomas, were tested with MoAb Hep Par 1. Only 5/277 tumors were stained with this marker: one adrenocortical carcinoma, one interstitial cell tumor of the testis, one melanoma, and two salivary carcinomas. The staining was always multifocal and cytoplasmic and never involved more than 20% of neoplastic cells. Metastatic tumors to the liver included in this study were adrenal cortical carcinoma, intestinal adenocarcinoma, pancreatic exocrine carcinoma, hemangiosarcoma, and islet cell tumor (Table 1). The only tumor positive with MoAb Hep Par 1 was an intestinal adenocarcinoma (Fig. 8). The staining in this tumor was heterogeneous, with some cells strongly stained and others not stained.
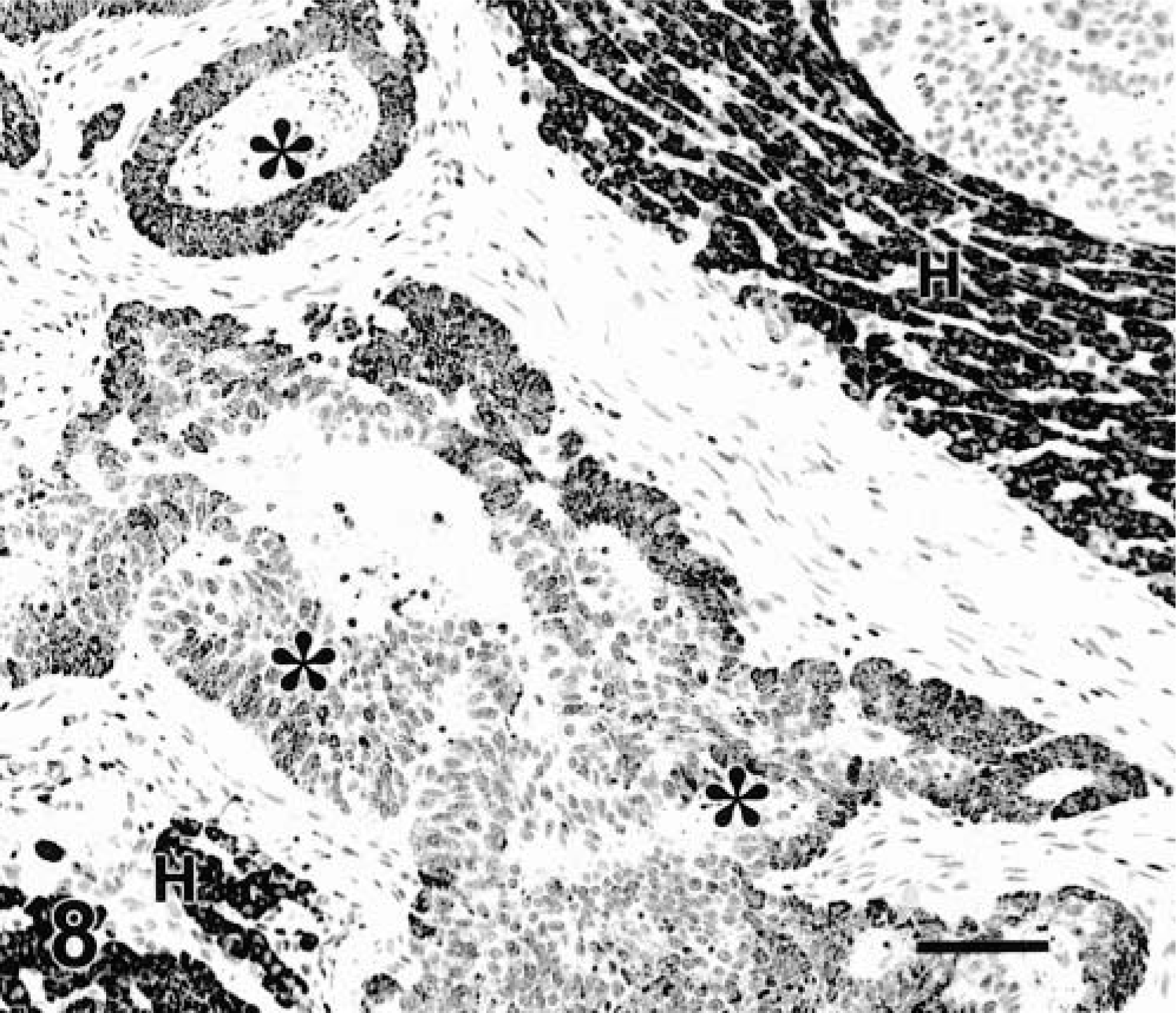
Liver; dog. Intestinal adenocarcinoma. Neoplastic cells (asterisks) are variably reactive with MoAb Hep Par 1. Normal hepatocytes (H). EnVision +–peroxidase stain, Mayer's hematoxylin counterstain. Bar = 60 µm.
Effect of prolonged fixation
There were no significant differences in the number of positive cells or in the intensity of the reaction with MoAb Hep Par 1 at any fixation time until day 24 of fixation (last day tested). Because of the multifocal staining with MoAb CK 7, interpretation of the effects of fixation was difficult. However, the number of positive bile ducts was similar among tissues fixed at different times with the exception of the sample fixed for 24 days, which had fewer positive cells than did other samples.
Discussion
MoAb Hep Par 1 detected all hyperplastic and benign hepatocellular neoplasms and >90% of hepatocellular carcinomas in dogs. None of the bile duct lesions (hyperplastic or neoplastic) were recognized by this antibody. Therefore, Hep Par 1 is a highly specific and sensitive marker of canine hepatocytes (normal, hyperplastic, and neoplastic). Similar results have been obtained in human studies, in which 82–97% of hepatocellular tumors were positive for Hep Par 1. 6,9,11,27,28 It was uncommon for extrahepatic tumors to react with Hep Par 1, and staining was usually focal although distinct and usually in a very reduced number of tumors in which positive staining was achieved in 5/277 tumors and 1/9 tumors metastatic to the liver. The staining of intestinal tumors with Hep Par 1 is currently being investigated in our laboratory. Reports of studies on humans have rarely addressed the staining pattern of Hep Par 1 in extrahepatic tissues or neoplasms, and only a few (gastric, pancreatic, colonic) tumors are occasionally positive for this marker. 27
Other markers commonly used in the immunohistochemical characterization of human hepatic neoplasms are AFP and CEA. 6,9–11 A polyclonal antibody to CEA (pCEA) has variable (15–80%) sensitivity but high specificity for hepatocellular tumors. 3,9,11 AFP has usually moderate to low sensitivity but high specificity for human HCC, 7,11 although some authors have found high numbers of HCC reactive to AFP. 8 In canine hepatic tumors, AFP stained 3/8 (37.5%) HCC and 0/2 CCC, whereas CEA stained 0/11 HCC and 2/2 (100%) CCC. 5 The small number of tumors examined makes generalizations difficult, but the pattern of staining of CEA in dogs appears to differ from that in humans.
Other antigens detected in canine hepatic neoplasms are CKs, including a cocktail of CKs 5 + 8 and CKs 8 + 18 + 19. Both HCC and CCC had CKs 5 + 8, whereas only CCC had CKs 8 + 18 + 19. 5 These results agree somewhat with the published CK profile of liver cells; hepatocytes express CKs 8 and 18, and bile duct epithelium expresses CKs 7, 8, 18, and 19. 12,19,24 However, some hepatic tumors have aberrant CK patterns, 23,25 which makes immunohistochemical characterization of hepatic tumors difficult when antibodies to only CKs are used. A shift in CK expression also has been observed during developing stages of bile duct epithelium from hepatocytes. 24 Based on these findings, distinction of hepatocellular neoplasms and cholangiocellular tumors should be facilitated by use of antibodies recognizing only CKs 7 and 19 to avoid overlapping with other CKs. We used CK 7 to detect tumors of bile duct origin. An antibody to CK 19 was not available for this study. CK 7 was detected in all benign tumors and hyperplastic biliary lesions and in >75% of CCC. The sensitivity of the staining was therefore moderate to high, and the specificity, when compared with that for other primary hepatic tumors, was close to 100%. Only one HCC showed multifocal staining with the MoAb to CK 7. During differentiation of liver cells, there may be coexpression of both hepatocyte and biliary markers and expression of “biliary” CKs in human HCC has been associated with a poor prognosis. 28 Although not tested in this study, other tissues and tumors may express CK 7, including those from the ovary, endometrium, mammary gland, lung, transitional epithelium, pancreas, and mesothelium. 26
The lack of staining for either Hep Par 1 or CK 7 in some hepatocellular and cholangiocellular neoplasms, respectively, is probably not the result of overfixation. Staining with Hep Par 1 was similar in tissues fixed for several weeks and those fixed for only several days. Results with CK 7 were more difficult to interpret because of the low numbers of bile ducts per section and the apparent irregular staining of bile ducts within the same section. The cause of this irregular staining is not clear, but it does not seem to be related to prolonged fixation.
MoAb Hep Par 1 is a good marker of hepatocellular differentiation and is superior in sensitivity and specificity to other markers previously reported. The use of Hep Par 1 in conjunction with antibodies to CK 7 will improve the diagnosis of hepatic neoplasms. Prolonged fixation does not seem to decrease immunostaining.
Footnotes
Acknowledgements
We thank M. Beissenherz for technical assistance with immunohistochemistry and D. Connor for photographic preparations.
