Abstract
A purebred Ayrshire calf was born with multiple congenital cardiac defects. The major defects included absence of an interventricular septum (i.e., single ventricle), transposition of the aorta and pulmonary trunk, interatrial septal defect, patent ductus arteriosus, and aortic arch anomalies. Despite the severity of the anomalies, the calf survived to 5 days of age, when it was found dead. This particular combination of cardiac defects has not been previously described in domestic animals.
Keywords
A small bull calf (30 kg) was delivered unassisted from a purebred Ayrshire heifer after an uneventful 287-day gestation. The calf was initially vigorous and able to drink from a bottle unassisted. On day 3 the producer noticed that the calf seemed weak on its hind legs and that it got up and down frequently. It was tube-fed that day because of its weakened state. On day 4 the calf was panting and breathing with obvious abdominal effort. The producer auscultated the heart and noted tachycardia but no murmur. The calf died on day 5. Veterinarians were called, and a post mortem was performed within 4 hours of death. The notable findings were increased peritoneal (serosanguineous) fluid, congestion of the liver, segmental atelectasis with pulmonary edema, and obvious enlargement of the heart. The heart and major vessels were placed in 10% buffered formalin and brought to the Animal Health Laboratory at the University of Guelph for examination.
Grossly, numerous cardiac anomalies were observed (Fig. 1). The heart was globose due to enlargement or dilation of the ventricular portion, and the coronary grooves were obscured. The right and left atria were in the normal location, but the right atrium appeared enlarged while the left atrium appeared hypoplastic. The right auricle was in a normal position in relation to the right atrium. The positions of the cranial and caudal vena cavae were normal. An obvious abnormality was in the position and course of the great vessels as they emerged from the heart base.
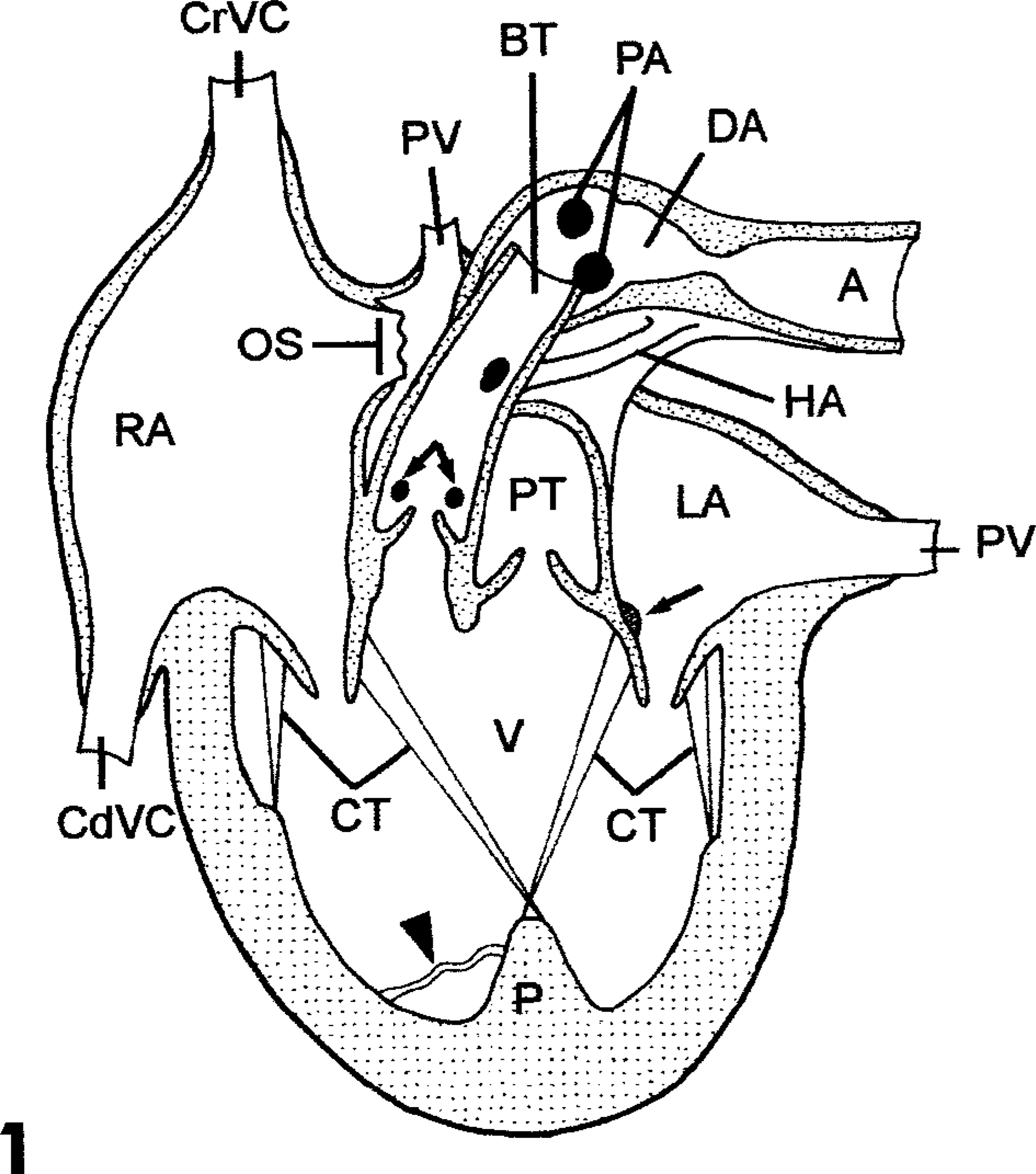
Heart; calf. Cardiac anomalies. Rendering of modified frontal plane. Brachiocephalic trunk (BT), caudal vena cava (CdVC), chordae tendineae (CT), common papillary muscle (P), common ventricle (V), coronary arteries (double arrow), cranial vena cava (CrCV), descending aorta (A), ductus arteriosus (DA), hemal cyst (arrow), hypoplastic aorta (HA), left atrium (LA), ostium secundum (OS), pulmonary arteries (PA), pulmonary trunk (PT), pulmonary vein (PV), right atrium (RA), septomarginal band (arrowhead).
Within the right atrium was a prominent crista dividens and a large (2 cm) ostium secundum, which is equivalent to an atrial septal defect. The right atrium opened through a right atrioventricular (AV) valve into a single (common) ventricle; the interventricular septum (IVS) was absent. The ventricle walls were thickened throughout, with the thinnest aspect at the right lateral wall. The annuli of both AV valves were formed and located normally and were thus used as anatomic landmarks to describe outflow tracts (Fig. 2). Proximity of the outflow tracts to the heart base was also used as a positional marker.
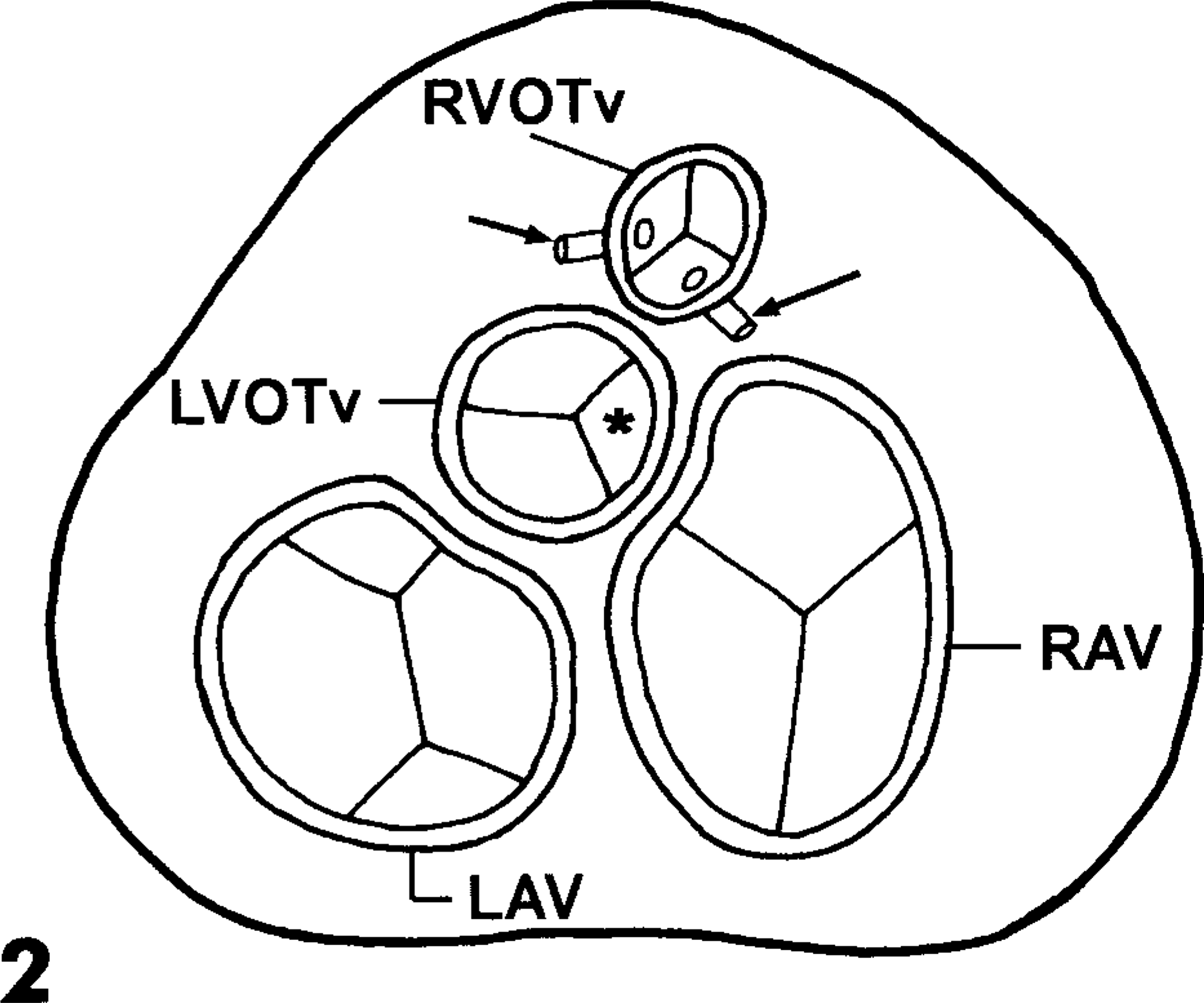
Heart; calf. Cardiac anomalies. Rendering of dorsal view of heart valves. Major vessels and atria removed. Coronary arteries (arrows), dysplastic semilunar cusp (∗), left atrioventricular valve (LAV), left ventricular outflow tract valve (LVOTv), right atrioventricular valve (RAV), right ventricular outflow tract valve (RVOTv).
The left and right AV valves were formed normally. The septal cusps of both AV valves were continuous at their base with each other and with the annulus of the presumptive (transposed) pulmonic valve through the exposed fibrous skeleton of the heart. At their caudal end, these cusps were connected by means of chordae tendineae to a common papillary muscle replacing the subatrialis (posterior) papillary muscle of the left ventricle and the subarteriosis (septal) papillary muscle of the right ventricle (Fig. 1, 3). Several hemal cysts were seen on the left AV valve. The crista supraventricularis was absent from the right ventricular outflow tract (RVOT). This tract was partially obstructed by a malformed magnus (right anterior) papillary muscle of the right ventricle. The annulus terminating the RVOT was elevated (more distal) relative to the annulus terminating the left ventricular outflow tract (LVOT). The vessel emerging from the RVOT was thick walled and 1.5 cm in diameter, and it gave rise to two coronary arteries (CA) at its base. The left CA arose from the left sinus and the right CA arose from the septal sinus of the aorta. The entire vessel from the RVOT continued cranially as the brachiocephalic trunk, in contrast to the expected distribution of a pulmonary trunk.
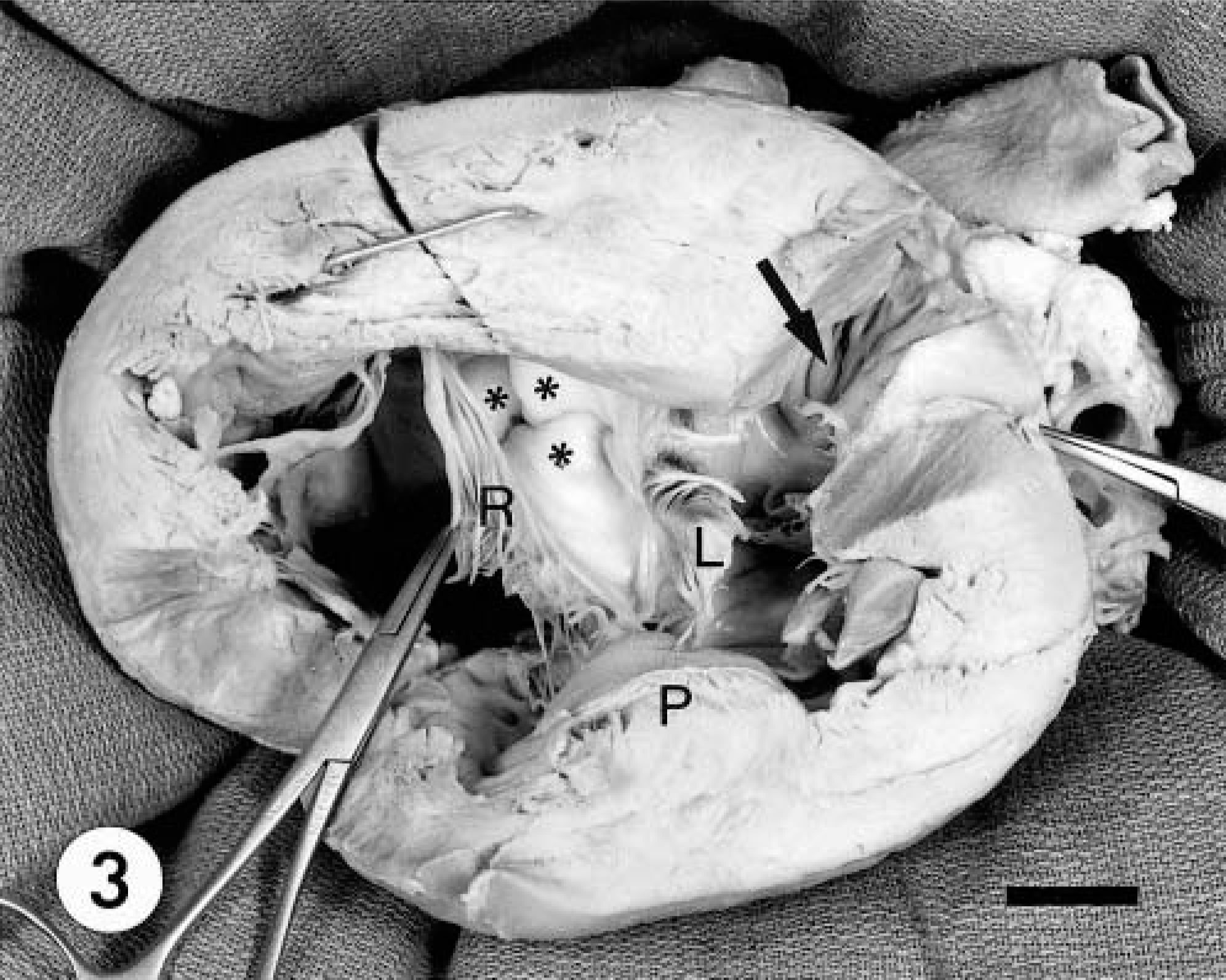
Heart; calf. Cardiac anomalies. Apical view of the dissected heart. Note the continuous septal cusps of the left (L) and right (R) atrioventricular valve and the fibrous continuity with the valve of the LVOT (asterisks). Common papillary muscle (P), pectinate muscles of left auricle (arrow). Bar = 1.5 cm.
On the functional left side, the left atrium was hypoplastic, with the pulmonary veins located normally. The wall of the foramen ovale was represented by a sleeve of pliable tissue that extended into the left atrium from the margin of the enlarged ostium secundum. There was no muscular conus leading to the LVOT. The vessel emerging from the LVOT lay between the continuous septal cusps of the left and right AV valves. The semilunar valve was dysplastic, with two large main cusps and a small, irregular medial cusp (Fig. 2). The annulus terminating the LVOT was 1.5 times larger than the annulus terminating the RVOT. The presumptive pulmonary trunk (PT) arising from the LVOT was thick walled. It was oriented caudally and gave rise to two pulmonary arteries (PA) but no coronary arteries. The diameter of the PT doubled to 3.4 cm where the PAs emerged, followed by a narrowing (possible coarctation) to a lumen diameter of 0.5 cm (presumptive ductus arteriosus) (Fig. 1). Beyond the narrowing, the vessel dilated to 1.7 cm and continued distally as the descending aorta, giving rise to numerous segmental arteries. At the level of the narrowing, a narrow vascular channel (3 cm × 0.3 cm) connected the vessels emerging from the ventricular outflow tracts (presumptive hypoplastic ascending aorta). There was no evidence of a brachiocephalic trunk arising from the vessel leaving the LVOT.
The most functionally significant defects were the single ventricle with a complete lack of an IVS, transposition of the pulmonary trunk and aorta, patent ductus arteriosus, other aortic arch anomalies, and interatrial septal defect. In this specimen the RVOT served the aortic valve, hypoplastic aorta, and brachiocephalic trunk. The LVOT served the pulmonary valve, trunk and arteries, and patent ductus arteriosus. A single ventricle is almost always accompanied by transposition of the great vessels and aortic arch anomalies such as tubular hypoplasia, coarctation, and patent ductus arteriosus.1,4,6,8 IVS formation involves tissue from multiple sources, including tissue that septates the truncus arteriosus (TA) into the pulmonary trunk and aorta.1,3,5 In this case, in which left and right sinus myocardium were present, the absence of an IVS was likely caused by the rudimentary development or complete absence of the sinus portion of the IVS.1 The annuli of the AV valves were both intact, indicating that the endocardial cushions, from which the valves partially develop, were normal. An endocardial cushion defect was thus not the cause of the ventricular septal defect (VSD). VSD is the second most common cardiac anomaly in cattle.5,8
Complete simple transposition of the great vessels in humans is characterized by the presence of an aortic or subaortic muscular conus (RVOT in the present case).1 This results in a loss of fibrous continuity between the aortic annulus and the annulus of the left AV valve. Continuity, however, exists between the left AV valve and the pulmonic annulus. This displaces the outflow tract orifices without changing the vascular distribution pattern.3 The abnormal emergence of the coronary arteries from the RVOT reflects the most common coronary pattern found in humans with complete transposition of the great vessels.1 This can also be accompanied by VSD, atrial septal defect, patent ductus arteriosus, and coarctation of the aorta.1,3,4
In the normal heart a continuous ridge of tissue develops within the TA and the bulbus cordis. This ridge fuses along the length of the primitive great vessels, progressing from the most distal aspect toward the heart, forming the aorticopulmonary septum, and dividing the aorta and pulmonary trunk.1 The 180° spiralling of the septum causes the pulmonary trunk to twist around the aorta and give rise to the adult position of the great vessels.6 The left and right PAs, which develop from the sixth aortic arch,1,3 can arise from various positions along the TA. In this case they appeared to have originated normally from the pulmonary root. The vessel that arose from the morphologic left ventricle (LV) served the lungs by means of the PAs and, surprisingly, much of the systemic circulation by means of the presumptive ductus arteriosus and descending aorta. The 3-cm vascular channel that joined the left and right ventricular outflow channels is likely a tubular, hypoplastic (ascending) aorta, whereas the continuation of the PT from the origin of the pulmonary arteries to the descending aorta is likely the ductus arteriosus (DA) (Fig. 1).
The cranially-directed vessel arising from the RVOT was presumed to be the aorta leading to a large brachiocephalic trunk. Supporting this interpretation is the fact that the wall thickness of the vessel was equal to that of the descending aorta. Unfortunately, the heart was harvested without the major branches of the great vessels; therefore, these interpretations could not be confirmed.
The consequences of these defects became life-threatening as the circulatory pattern changed from fetal to adult. After birth the onset of respiration resulted in a decrease in pulmonary vascular resistance favoring flow of blood through the LVOT.1 Preferential flow to the lungs would have been further encouraged by the narrowing of the DA at the descending aorta. The partial obstruction of the RVOT would discourage blood flow through the putative brachiocephalic trunk to the head. The total mixing involved in these flow disturbances would constitute a net right-to-left shunt. Redirecting a significant proportion of cardiac output to the lungs would result in a large venous return to the left atrium, with elevation of left atrial pressure and shunting to the right atrium via the ostium secundum. This would promote right atrial dilation and hypertrophy and constitute a left-to-right shunt, which would be negated by the complete mixing of oxygenated and deoxygenated blood in the common ventricle. Hypoxemia, reduced flow in the systemic circulation, increased pulmonary flow with probable pulmonary hypertension, and greatly increased cardiac workload would have limited this calf's viability.
Although the individual malformations found in this case are not unique, this unusual combination of cardiac defects has not been previously described in domestic animals. The cause of these cardiac anomalies may involve genetic defects with a complex mode of inheritance, teratogen exposure, or nutritional deficiencies.2,7 The multiple defects may be separate embryologically, though they may be temporally associated. The causal factor in this case was not determined.
