Abstract
With only about 3% of patients with uncorrected tetralogy of Fallot surviving past age 40, we describe the case of an uncorrected tetralogy of Fallot following a palliative, now obstructed, Blalock-Taussig shunt in a man who presented for emergent noncardiac surgery. Due to limited in-house records, managing this complex patient presentation required obtaining outside-hospital records, performing point-of-care diagnostic studies, and coordinating with the surgical team. We employed monitored anesthesia care with the option to convert to general anesthesia if needed. The patient was discharged after a 3-day intensive care unit stay and cleared for normal activity at his 3-week follow-up. This case highlights the importance of interdisciplinary collaboration, knowledge of prior surgical history, and understanding intraoperative hemodynamic goals for successful planning and perioperative management.
Keywords
Introduction
Of the cyanotic congenital heart defects (CHD), tetralogy of Fallot (TOF) is the most common. Right ventricular (RV) outflow obstruction, RV hypertrophy, a ventricular septal defect (VSD), and an overriding aorta are characteristics of TOF, which often lead to cyanosis. Without treatment, 70% will die by 10 years of age. 1 Definitive treatment is typically a complete surgical repair at 3–6 months of age. Palliative options such as the Blalock-Taussig (BT) shunt help alleviate cyanosis by redirecting systemic blood to the pulmonary arteries (PA). 2 Developed in 1945 at Johns Hopkins University, the classic BT shunt sacrifices the subclavian artery for an end-to-end anastomosis between it and the PA. The technique has progressed to the modified BT (mBT) shunt in which polytetrafluoroethylene or Gore-Tex grafts anastomose the subclavian and PA. Both iterations provide predictable pulmonary blood flow while sharing the primary risk of thrombosis. 3 In a study of over 4.9 million adults undergoing noncardiac surgery, Williamson et al. found an increased prevalence of CHD history from 0.06% in 2010 to 0.17% in 2018. 4 With increasing survivorship of TOF patients into adulthood, more will require noncardiac surgery. This case highlights the pathology and perioperative challenges associated with these patients.4,5
Report
A 55-year-old man presented to the emergency department from an outside clinic for a 3 cm incarcerated umbilical hernia. His medical history included chronic biventricular heart failure, severe pulmonary hypertension, severe RV systolic dysfunction, paroxysmal atrial fibrillation, liver cirrhosis with ascites, and stage 3 chronic kidney disease. The initial chart review yielded limited information because the most recent notes were from 18 years prior. We utilized point-of-care ultrasound while his primary cardiologist was contacted for recent records with the help of the patient’s family.
The patient’s records revealed the TOF remained unresolved through childhood, and a BT shunt was placed in his 30s, highlighting the primary cardiologist’s concern for obstruction of the BT shunt. Aspirin had been held over the previous 4 days due to biweekly paracentesis to treat ascites. The clinical examination revealed a distended abdomen, bilateral lower extremity edema to his thighs, and central cyanosis. His baseline oxygen saturation was 60%–70% while on 5 L nasal cannula. His hemoglobin was 17.2 g/dL. Bedside transthoracic echocardiogram revealed biventricular hypertrophy, left ventricular ejection fraction of 40%–45%, mild-to-moderate aortic valve insufficiency, moderate-to-severe tricuspid regurgitation and RV systolic pressure of 116 mmHg. There was also a VSD with bidirectional shunting (Figures 1 and 2).
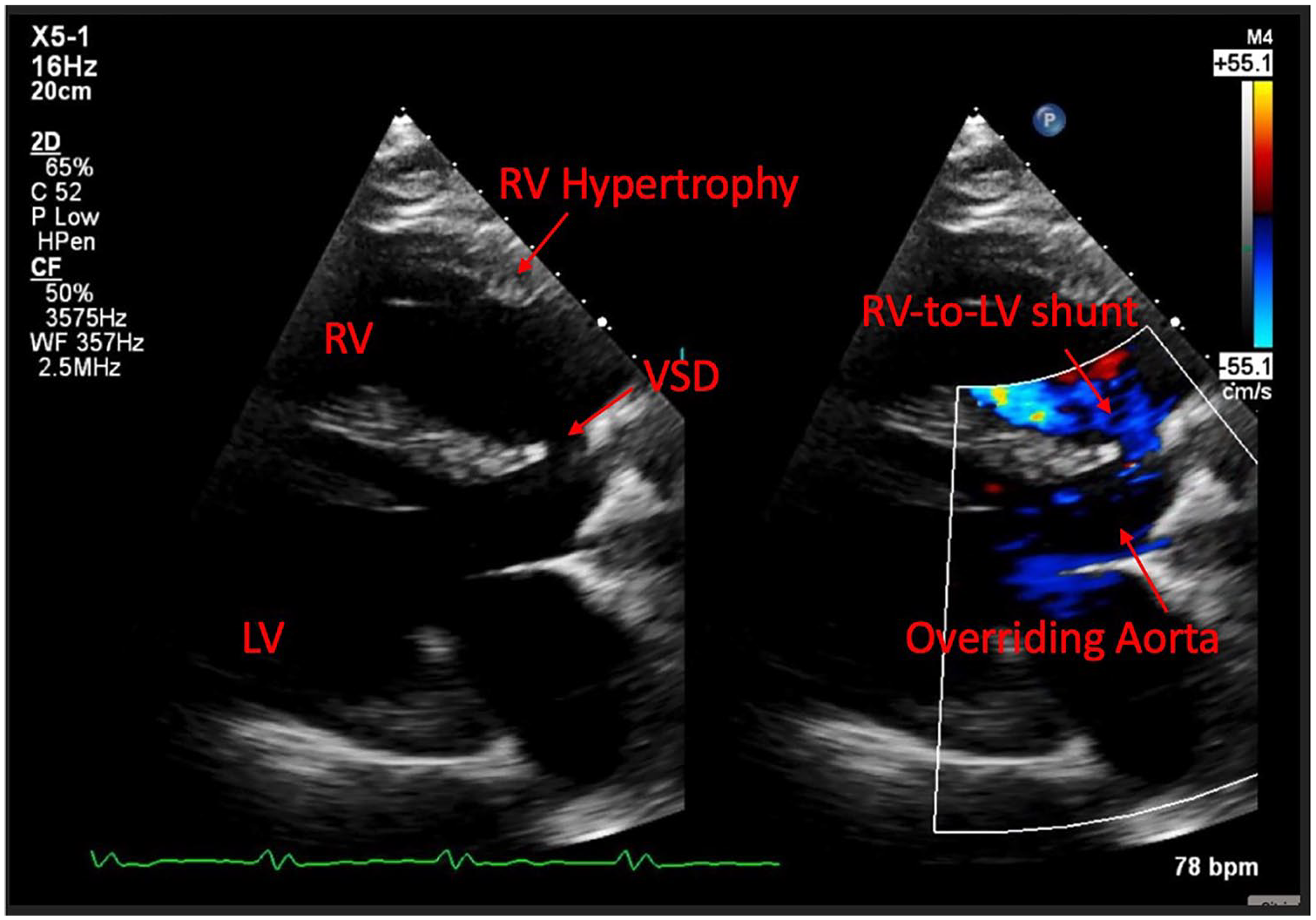
TTE—Parasternal long axis view showing the LV, RV, overriding aorta, VSD with flow through shunt, and RV hypertrophy.
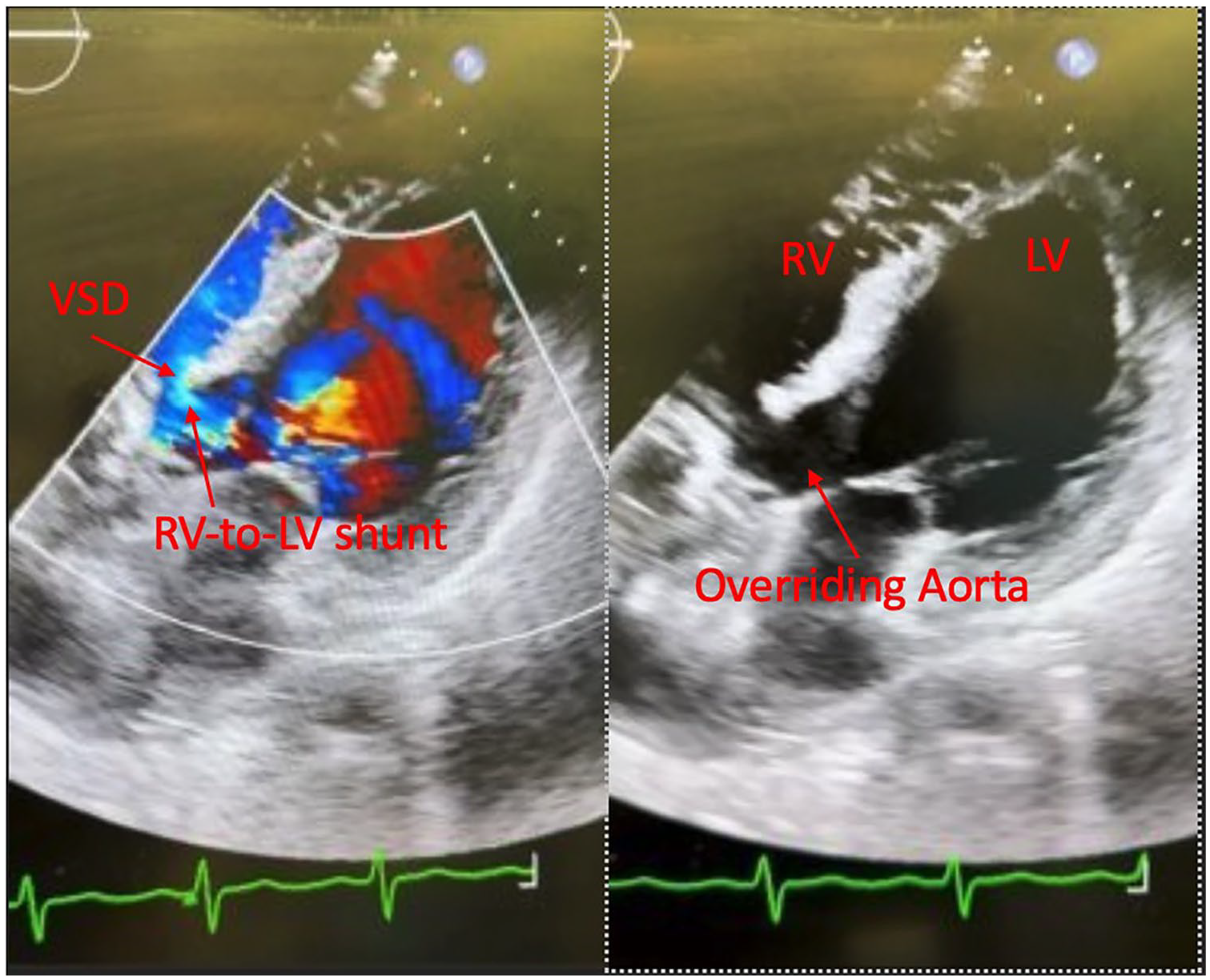
TTE—Apical four-chamber view showing LV, RV, overriding aorta, and VSD with flow through shunt.
Intraoperatively, the patient was in the semi-Fowler’s position. Standard monitors, along with a right internal jugular vein Cordis and left radial arterial line, were placed. FloTrac was connected to the arterial line for cardiac output monitoring. The case was performed under monitored anesthesia care (MAC) using midazolam with dexmedetomidine. The anesthesiologist gave 2 mg of IV midazolam and a dexmedetomidine infusion at rates of 0.5–1 mcg/kg/h for the procedure duration. High-flow nasal cannula at 40 L/40% was used to administer inhaled epoprostenol at 40 ng/kg/min. Vasopressin and epinephrine were administered to maintain systemic afterload. There were no significant changes in blood pressure, heart rate, or estimated cardiac output at the initiation of MAC.
The patient received 1 L of crystalloid for the 5-h case. The umbilical hernia repair was performed under local anesthesia using approximately 550 mg (6 mg/kg) of lidocaine with epinephrine at the surgical site. The repair was completed without anesthetic complications, and the patient was transported to the intensive care unit (ICU) postoperatively. He was discharged from the ICU on postoperative day 3 at his preoperative baseline and cleared for normal activity at his 3-week follow-up visit.
Discussion
TOF occurs in approximately 1 in 3500 live births. TOF has a sporadic occurrence but can be associated with Down and DiGeorge syndromes. 5 Presentation includes childhood cyanosis, hypoxic “tet spells,” and failure to thrive within the neonatal period. The severity of pulmonary outflow tract obstruction influences the degree of right-to-left shunting and, ultimately, the severity of cyanosis. Unrepaired TOF in older children and adults leads to progressive cyanosis, dyspnea, cerebrovascular accidents, and brain abscesses.5,6 Our patient progressed to biventricular failure, atrial fibrillation, and cirrhosis. 6 Diagnosis via prenatal ultrasound and early postpartum repair is the preferred management.5,7
Our patient received a palliative mBT shunt using a Gore-Tex graft. This shunt provided an increase in pulmonary blood flow but not definitive treatment. 2 Other palliative options include ductus arteriosus stenting, pulmonary annulus balloon dilation, and RV outflow tract stenting. 2 Definitive treatment entails primary repair such as widening the pulmonary annulus with a transannular patch or releasing the RV outflow tract obstruction via placement of a monocusp valve, valved RV-to-PA conduit, or valvuloplasty. 2 Primary repair preferentially occurs at 3–6 months of age rather than in neonates to reduce hospital and ICU stays. 7 Primary repair in neonates occurs when severe cyanosis is present. 7 When primary repair is delayed, beta-blockers for mild cyanosis or staged repair via palliative surgery for severe cyanosis can alleviate symptoms until the preferred age is reached.5,7
Collaboration was essential to successfully manage this complex patient with limited patient records. The surgeons first had to identify the need for a cardiac anesthesiologist to manage this patient’s complex cardiac pathology. A bedside history revealed the baseline oxygen requirements. The availability of the patient’s cardiologist allowed for quick acquisition of outside-hospital records and attention being brought to a possible shunt obstruction. MAC was chosen over general anesthesia due to the inherent risk of worsened right-to-left shunt with decreased afterload and the discussion with surgeons that the repair could be performed successfully under local anesthesia. 5
The hemodynamic goals aimed to ensure adequate preload, avoid worsening pulmonary hypertension, and prevent large decreases in systemic vascular resistance. Coupling midazolam with continuous dexmedetomidine for MAC also limited respiratory depression. 8 Epoprostenol was chosen as a vasodilator to control pulmonary hypertension. Appropriate vascular access was crucial for ensuring hemodynamic stability. The radial arterial line provided invasive blood pressure monitoring while central access allowed reliable administration of fluid to ensure adequate preload and vasopressors. 9
Alternative anesthetic strategies include general and regional anesthesia. General anesthesia with mechanical ventilation is preferred for complex cases, such as larger hernia repairs, or when MAC cannot maintain adequate sedation. However, it requires careful selection of hemodynamically stable anesthetic agents to limit hypotension. Ventilator settings aim to avoid excessive intrathoracic pressure and increased pulmonary vascular resistance.9,10 Regional or neuraxial anesthesia provides analgesia while preserving respiratory drive and afterload. 10 However, due to the possibility of a large sympathectomy decreasing afterload, neuraxial was avoided.
Conclusion
TOF is a cyanotic CHD that complicates perioperative planning due to hypoxic spells stemming from increasing right-to-left ventricular shunting. Its prevalence in adult patients increases as palliative and complete repair options improve survivorship beyond adolescence. The perioperative considerations required highlight the need for early identification of TOF as well as the need for multidisciplinary collaboration in their acute management.
