Abstract
A 7-y-old, captive, intact female meerkat (Suricata suricatta) was presented with lethargy, decreased appetite, dyspnea, and distended abdomen. At autopsy, the right atrium was markedly dilated, and the right atrioventricular valve (RAV) was dysplastic with shortened or absent chordae tendineae and direct attachment of the valve to the papillary muscles, which, in turn, were fused and abnormally positioned. The right ventricle was considered to be hypertrophied. Also present were hydrothorax, ascites, atelectasis, and hepatic congestion. A diagnosis of RAV dysplasia was made. Histologic findings included hypertrophy of cardiomyocytes and marked centrilobular hepatic congestion and hemorrhage, which were consistent with right-sided heart failure.
A 7-y-old, captive, intact female meerkat (Suricata suricatta) from the Saskatoon Zoo (Saskatchewan, Canada) was first noticed with dyspnea in August 2021; a diffuse bronchointerstitial pattern was observed on radiographs. Her diet was raw meat and insects. She also had a history of alopecia, flakey skin, and chronic pyoderma that had been treated periodically with antibiotics. In January 2022, the zookeeper noticed that the meerkat was lethargic, with decreased appetite, dyspnea, and distended abdomen. Physical examination, whole body radiographs, and abdominal ultrasound examination were performed under general anesthesia. Physical examination revealed muffled heart sounds, increased respiratory effort, and marked abdominal distention. Radiographs showed fluid opacity in the ventral thorax, more prominent on the right side, associated with retraction of the lung lobes and effacement of the heart silhouette. Dorsal deviation of the trachea was also observed in its mid-thoracic portion. Thoracic ultrasound was started, but the meerkat went into respiratory arrest and bradycardia, and the examination was interrupted. Abundant pleural and peritoneal effusions were observed, and subjective thickening of the left ventricle resulted in decreased left ventricular chamber diameter. The right side of the heart and left atrium could not be examined. Bicavitary effusion and dorsal deviation of the trachea raised a suspicion of heart disease that had progressed to heart failure. The meerkat was euthanized under general anesthesia given the poor prognosis associated with the potential heart disease that could not be managed.
The body was submitted to Prairie Diagnostic Services at the Western College of Veterinary Medicine (WCVM; Saskatoon, Saskatchewan, Canada) for autopsy. The meerkat weighed 0.8 kg; subcutaneous and internal fat stores were adequate. The thoracic cavity contained 16 mL of yellow cloudy fluid. Bilaterally, the compressive atelectasis was evidenced by multifocal-to-confluent areas of red discoloration and partial collapse; the lungs sank when placed in formalin. The cranial vena cava and right atrium were markedly dilated (Fig. 1A). The thicknesses of the right and left free ventricular walls were 3 and 4 mm, respectively; the right-to-left ventricular free wall ratio of 0.75:1 was suggestive of right ventricular hypertrophy (Fig. 1B).8,11 The interventricular septum was 5 mm thick. The chordae tendineae in the angular and parietal leaflets of the right atrioventricular valve (RAV) were shorter than those in the left atrioventricular valve (RAV: 1 mm; LAV: 2–3 mm; Fig. 1C, 1D). The parietal leaflet was directly attached to a papillary muscle without chordae tendineae present (Fig. 1C). A portion of the septal leaflet adhered to the interventricular septum; this leaflet was thickened and mildly nodular. The papillary muscles in the right ventricle appeared fused and mispositioned. The abdomen was markedly distended with 25 mL of red-tinged, cloudy, watery ascitic fluid. The liver was firm, moderately enlarged with rounded edges, enhanced lobular pattern, and rough surface. A few strands of fibrin were attached to the serosal surfaces of abdominal organs. No gross abnormalities were noted in other organs. Representative tissue samples were fixed in 10% neutral-buffered formalin, processed routinely, and stained with H&E for histopathology.
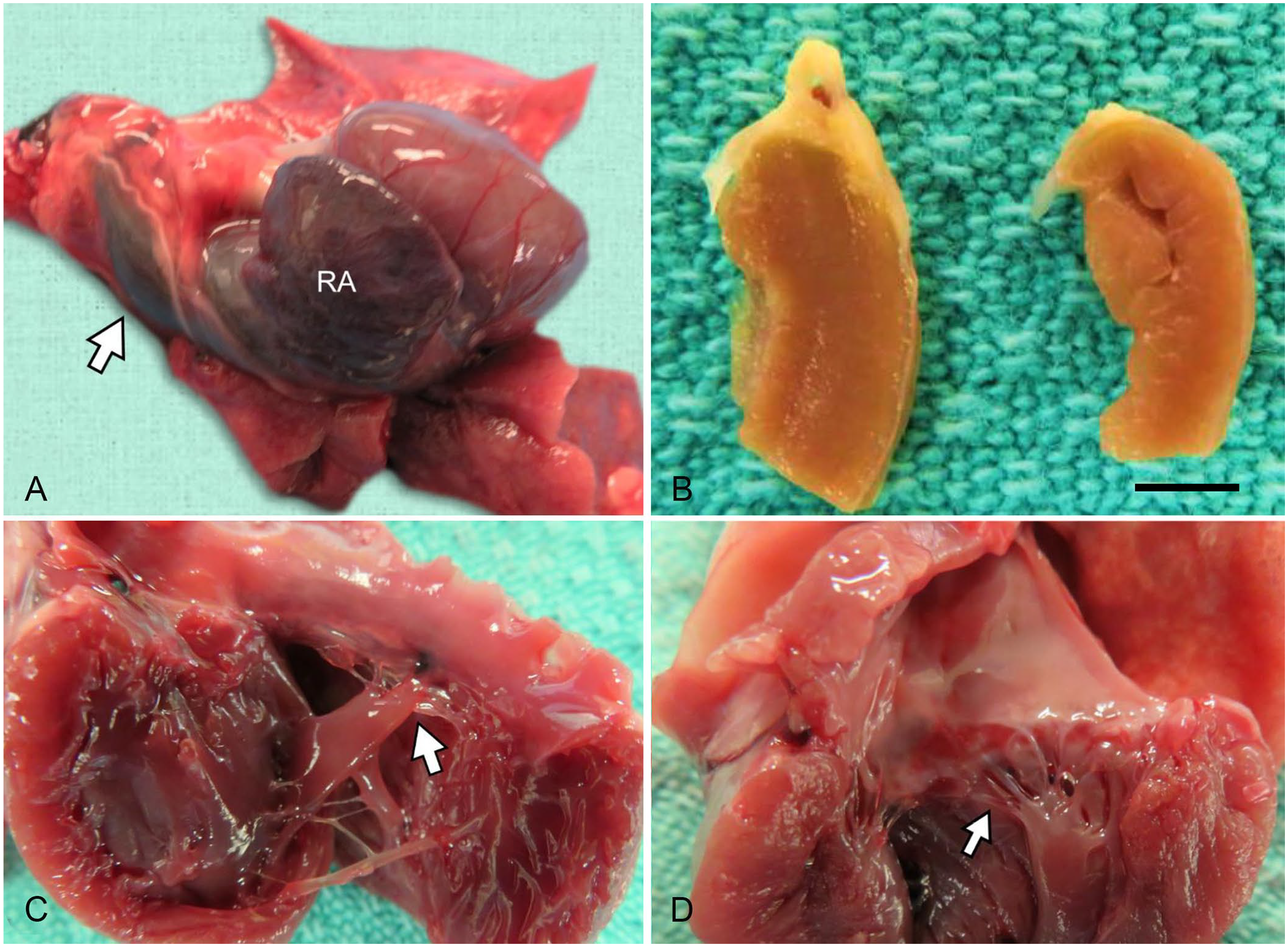
Right atrioventricular valve (RAV) dysplasia in a meerkat.
Histologically, cardiomyocytes in sections of the right ventricular wall were subjectively enlarged, with pale sarcoplasm and large vesicular nuclei, suggestive of myocardial hypertrophy. The sarcoplasm was often vacuolated; phosphotungstic acid hematoxylin (PTAH) stain showed normal striation of cardiomyocytes. Sections of some lung lobes had extensive areas of alveolar and airway collapse. Rarely, large airways contained a few neutrophils and scant pale-blue mucin. There were also multifocal, moderate, peribronchial and perivascular lymphoplasmacytic infiltrates. The liver parenchyma had diffuse centrilobular congestion and hemorrhage that frequently extended to the midzones. In the affected areas, hepatic plates were separated by expanded sinusoids engorged with RBCs; these plates were frequently dissociated, vacuolated, or atrophied. Multifocally, portal areas were edematous, with markedly congested portal vein branches and mild bile duct hyperplasia. There were small amounts of light-yellow to brown granules within the cytoplasm of Kupffer cells and hepatocytes.
Our postmortem findings were consistent with the diagnosis of RAV dysplasia, with associated right ventricular hypertrophy, marked dilation of the right atrium, hydrothorax, ascites, and hepatic congestion, as a result of valvular insufficiency and right-sided heart failure. The atelectasis in this meerkat likely resulted from compression by hydrothorax and cardiomegaly.
To our knowledge, there is only one previous case report of RAV dysplasia in a meerkat, an 8-y-old, captive animal with dyspnea and pleural effusion, similar to our case. 3 The previously described malformation was classified as an Ebstein anomaly, a congenital defect characterized by downward displacement of the basal portion of the valve into the right ventricle; the septal leaflet is most commonly affected. In the previous case, the RAV septal leaflet adhered to the interventricular septum. In our case, 50% of the RAV septal leaflet adhered to the interventricular septum, and the leaflet was thickened and mildly nodular in shape. However, without a proper comparison with clinically healthy meerkats, it is difficult to determine whether the septal leaflet was truly dysplastic in our case. Nevertheless, the morphologic abnormalities associated with RAV dysplasia also include focal or diffuse thickening of leaflets, absence of leaflets, short or absent chordae tendineae, short papillary muscles, and direct fusion of leaflets to the ventricular wall. 13 In our case, several chordae tendineae were short or absent allowing direct attachment of a papillary muscle to portions of the parietal RAV leaflet, which is consistent with RAV dysplasia. This anomaly is considered to be congenital8,11,13; we observed no evidence of underlying inflammatory or repair processes that may have caused distortion of the valvular structures in our case. The marked dilation of the cranial vena cava, formation of pleural and abdominal effusions, and hepatic congestion were consistent with right-sided heart failure.8,11
RAV dysplasia has been reported in dogs, cats, cows, and humans.2,5,12,13 In cats, RAV dysplasia is one of the more commonly diagnosed congenital heart defects.10,11,13 In a retrospective study, RAV dysplasia represented 10.4% of all congenital heart defects in cats, next in prevalence to ventricular septal defect (56.3%) and mitral valve dysplasia (14.6%). RAV dysplasia represents 2–8% of congenital heart diseases in dogs4,9,13; Labrador Retriever and Dogue de Bordeaux dogs are the breeds affected most often. In Labrador Retrievers, reduced penetrance of an autosomal dominant trait mapped to chromosome 9 is suggested to cause RAV dysplasia. 1 RAV dysplasia is uncommon in other domestic animals. There is only 1 bovine case report, which describes RAV dysplasia and patent foramen ovale in a 2-mo-old Red Angus heifer. RAV dysplasia in meerkats is suggested to be a congenital disease in a case report. 3 The absence of valvular infection, inflammation, and tissue repair in our case also suggests that RAV dysplasia is likely congenital in meerkats, similar to other species. Nevertheless, more studies such as genetic analyses in meerkats with RAV dysplasia are required to confirm this theory. 1
Congenital heart defects can be life-threatening in young animals5,9,12; however, in a retrospective study, 59% of dogs with an Ebstein anomaly were subclinical at the time of diagnosis. In addition, 72% of affected dogs had not succumbed 160 mo after diagnosis of an Ebstein anomaly. 5 In cats, congenital heart malformations are diagnosed commonly in adult patients.6,7 The clinical signs and survival time of patients with RAV dysplasia vary depending on the severity of the morphologic abnormalities (e.g., mild vs. marked displacement of leaflets). The manifestations of congestive heart failure (e.g., ascites) in congenital heart disease are strongly correlated with decreased survival time. 5 The severity of the RAV abnormalities in our case were apparently mild-to-moderate, allowing this animal to compensate for its cardiac anomaly for 7 y. Pathologists and zoo animal veterinarians should consider RAV dysplasia as a differential diagnosis when diagnosing or treating meerkats with cardiac or respiratory issues.
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
