Abstract
A 12-y-old, castrated male Weimaraner dog was presented for a wellness examination. A 7-cm, firm mass was palpated on the left, ventral, mid-lateral neck. The neck mass was removed surgically and submitted for histopathology. A thyroid carcinoma was diagnosed based on microscopic examination. Immunohistochemistry for chromogranin-A, calcitonin, and thyroglobulin identified dual immunoreactivity of the latter two, and a final diagnosis was of a well-differentiated, compact, mixed medullary and follicular cell thyroid carcinoma. These neoplasms are rare in humans and have not been reported in dogs, to our knowledge.
A 12-y-old, castrated male Weimaraner dog was presented for a wellness examination. The dog was a new patient at this veterinary clinic; his prior records revealed regular annual vaccinations, treatment with heartworm preventives, and fecal examination for parasites. At his annual wellness examination, the following issues were addressed: severe dental disease with several fractured teeth, a right upper eyelid mass consistent with a Meibomian adenoma, an enlarged right tarsus with limited flexion, conjunctivitis with erythema around lids, and scabs on the lips. Additionally, a 7-cm diameter, firm mass was palpated deep to the left, ventral, mid-lateral neck. The owner indicated that the mass had been present for several years.
Pre-anesthetic work-up included clinical pathology, dental radiographs, and thoracic radiographs. Abnormalities from the CBC included increased numbers of platelets (419 × 109/L; RI: 170–400 × 109/L) and monocytes (1.4 × 109/L; RI: 0–0.8 × 109/L). The total serum calcium concentration was within the RI (2.67 mmol/L; RI: 2.23–2.85 mmol/L). Serum chemistry revealed increased alkaline phosphatase activity of 7.5 µkat/L (RI: 0.1–2.2 µkat/L). Chest radiographs (3 views) were considered within RIs.
Cytology was performed on an aspirate of the neck mass. The right thyroid gland palpated normally. The preparations demonstrated high cellularity, with intact cells arranged in moderately cellular sheets that occasionally had acinar-like orientations. The cells exhibited mild-to-moderate anisocytosis and anisokaryosis, and a high nuclear:cytoplasmic ratio. The cells were round-to-oval, with distinct borders and a small amount of pale basophilic cytoplasm. The nuclei were round and centrally placed, with a reticular chromatin pattern and, occasionally, a single, small but prominent nucleolus. No inflammatory cells or infectious agents were identified. Based on these findings, a thyroid tumor was diagnosed and, although the cells displayed little cytologic criteria of malignancy, consideration was given to a thyroid carcinoma given that malignant neoplasms are more common in canine thyroid glands than are benign lesions, especially when the thyroid gland is enlarged clinically.2,5
Following left thyroidectomy, the mass was fixed in formalin and processed routinely for histology. On microscopic examination of H&E sections, the normal thyroid tissue, on the side with the tumor, was effaced, except for a few compressed, colloid-containing follicles around the tissue periphery. The effacement was the result of a neoplastic mass composed of large epithelioid cells, arranged as solid sheets and clusters and rare follicles, and a minority of cribriform areas separated by fine fibrovascular stroma (Fig. 1). Additionally, eosinophilic amorphous, anuclear deposits expanded the stroma. The neoplastic cells were polyhedral, cohesive, and had abundant lightly eosinophilic cytoplasm and vesicular nuclei, most with a single prominent nucleolus. The mitotic count was 7 per 2.37 mm2. There was focal expansion of the neoplastic cells through the capsule. There was no intravascular invasion. No normal thyroid tissue appeared entrapped by the neoplastic cells. A diagnosis of a well-differentiated, compact (solid) thyroid carcinoma was made.
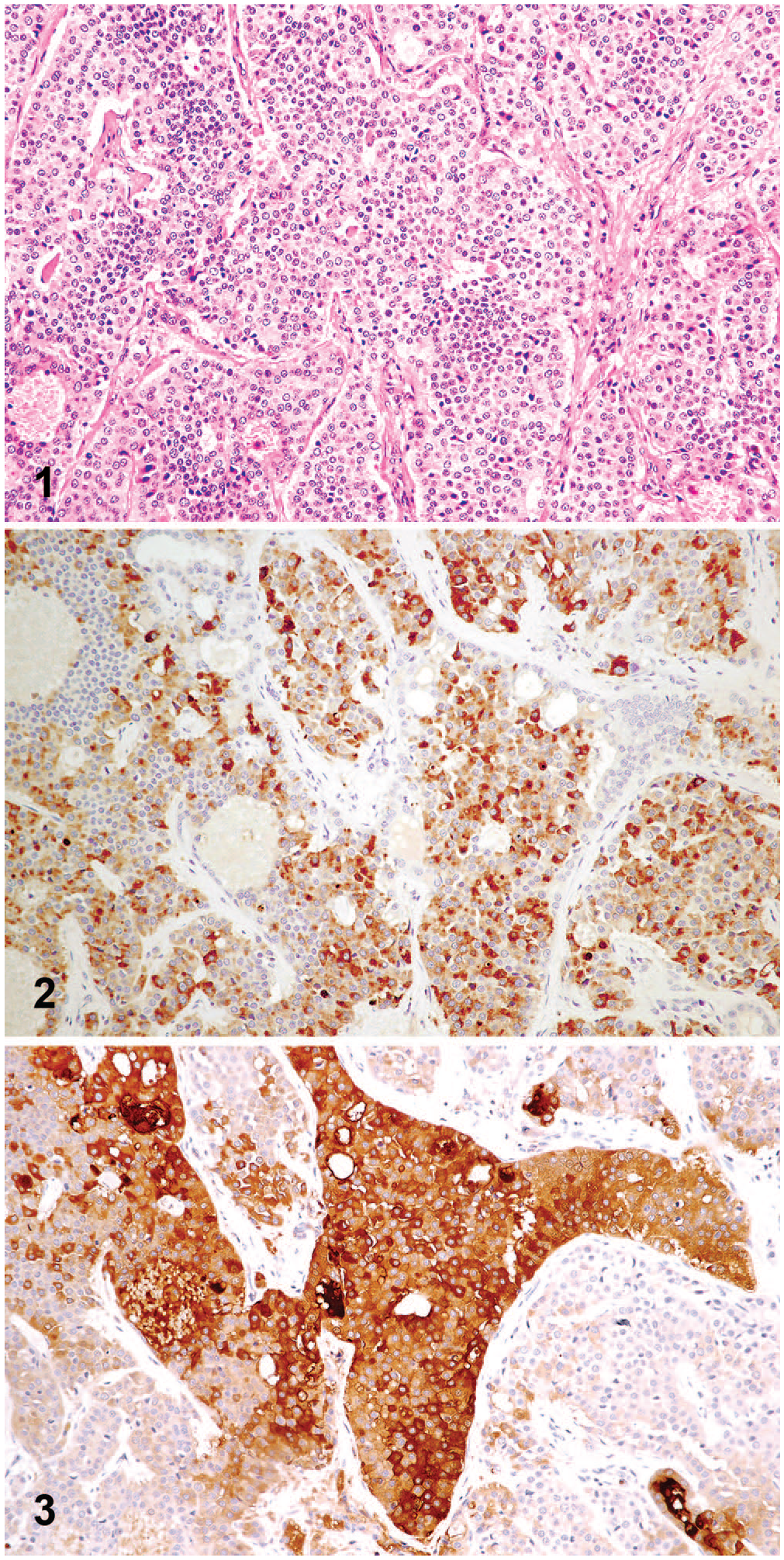
Mixed medullary and follicular cell thyroid carcinoma in a dog.
Immunohistochemistry (IHC) for chromogranin-A, thyroglobulin, and calcitonin was performed on serial sections of the neoplasm at the Veterinary Diagnostic Laboratory, Michigan State University (Lansing, MI, USA) utilizing appropriate controls. The neoplastic cells were uniformly positive (≥80%) for calcitonin (neuroendocrine origin) especially in the solid areas within the neoplasm. Multifocally, especially in areas with a cribriform pattern, neoplastic cells were positive for thyroglobulin (follicular cell origin). A large number of cells in the same areas within serial sections expressed both calcitonin and thyroglobulin (Figs. 2, 3). Neoplastic cells were negative for chromogranin-A, a more nonspecific neuroendocrine cell marker. Following the immunohistochemical assessment, the final diagnosis was well-differentiated, compact (solid), mixed medullary and follicular cell thyroid carcinoma.
In humans, the term mixed medullary and follicular cell thyroid carcinoma, also referred to as follicular-parafollicular carcinoma, denotes a rare neoplasm, which exhibits features of a medullary carcinoma intermingled with follicular cell malignancy.3,8 As the name implies, mixed medullary and follicular cell thyroid carcinomas are positive by IHC for calcitonin, mainly in the solid areas, and thyroglobulin, mainly in the follicular and cribriform areas. 9 Rare cells with dual expression of thyroglobulin and calcitonin have been reported, 9 although in our canine example, this feature was prominent. The term medullary-follicular thyroid carcinoma denotes a tumor that exhibits the features of a medullary carcinoma, along with positive calcitonin on IHC, and a follicular-derived thyroid carcinoma, along with positive thyroglobulin, within the same tumor. 3 Several mixed patterns of medullary and follicular-derived carcinomas have been described representing more-or-less acceptable examples of mixed medullary-follicular thyroid carcinomas.3,14,15
Mixed medullary-follicular thyroid carcinomas pose a challenge to the classical concept of different origins of medullary and follicular cells and must be differentiated from collision tumors. 4 In collision tumors, the 2 different tumor types (medullary and follicular) collide within the thyroid gland and the 2 components are either contiguous or intermingled. 4 The thyroid tumor described here did not represent a case of 2 different tumors coexisting within the same thyroid (i.e., a collision tumor), but instead was composed of a predominantly neoplastic medullary cell population, which was immunoreactive for calcitonin, intermixed with cells immunoreactive for thyroglobulin. 13 In fact, although thyroglobulin was expressed mainly in cells with a cribriform pattern, a large number of cells in these areas were positive for both calcitonin and thyroglobulin, which confirms the diagnosis of a mixed medullary and follicular cell thyroid carcinoma.
There are currently 3 hypotheses, albeit none of them proven, that attempt to explain how mixed medullary and follicular thyroid neoplasms develop. These are the stem cell, divergent differentiation, and field effect hypotheses. 4 According to the stem cell hypothesis, an uncommitted stem cell that is capable of differentiating toward both medullary and follicular cell lineages becomes neoplastic. According to the hypothesis of divergent differentiation, some medullary thyroid carcinoma cells differentiate toward a follicular phenotype by acquiring additional molecular defects. According to the field effect hypothesis, a common oncogenic stimulus triggers neoplastic transformation of both follicular and C cells. Such neoplasms would not be considered collision tumors but instead neoplasms composed of 2 endocrine cell types that proliferate independently within the same thyroid gland. Considering the large number of neoplastic cells expressing both thyroglobulin and calcitonin in our case, both the stem cell hypothesis and the hypothesis of divergent differentiation may apply.
Medullary carcinomas comprise 5–10% of all thyroid carcinomas in humans, and only 3–5% of these neoplasms (~0.15% of all thyroid tumors) display mixed features, having a follicular or papillary carcinoma component in the primary medullary carcinoma or in the metastases, or in both.1,3,4,8,10 Mixed medullary-follicular thyroid carcinoma have not been reported in dogs, to our knowledge, and the WHO International Classification of Tumors of Domestic Animals does not include this entity. 2
In humans, mixed medullary and follicular cell thyroid carcinomas occur most commonly in young males and tend to have a better prognosis than medullary carcinomas.3,7 In some patients, the medullary component of the neoplasm can be functional, and high serum concentrations of calcitonin have been associated with disease progression, indicating that some mixed medullary and follicular cell thyroid carcinomas behave hormonally like medullary carcinomas, and secrete calcitonin into the blood. 3
Metastatic rates for thyroid carcinomas in dogs can be as high as 80%, with 35–40% of dogs having metastasis at the time of diagnosis. 5 In a study of 70 dogs with thyroid neoplasia, tumor diameter, tumor volume, and bilateral location were associated with the presence of distant metastasis.5,13 The canine medullary and follicular cell thyroid carcinoma described here was from an older dog, and no metastases were identified at the time of the wellness examination. However, only chest radiographs were performed, which are not considered the most definitive method for determining micro-metastases. The animal lived 10 mo after the surgery. Cause of death was not determined; an autopsy was not performed. Both total calcium and T4 concentrations were normal in this dog prior to surgical excision of the neoplasm, suggesting that the neoplasm was not hormonally functional.
IHC is useful in determining the cellular origin of canine thyroid tumors, but most cases are routinely diagnosed based on histomorphology alone. In one study of canine thyroid neoplasms, 32 follicular tumors were thyroglobulin-positive and calcitonin-negative, and 4 of 6 medullary tumors were calcitonin-positive and thyroglobulin-negative. 6 In 4 of the follicular tumors and 2 of the medullary tumors, entrapped calcitonin-positive cells or thyroglobulin-positive cells were detected, respectively. 6 None of these neoplasms was considered to represent a mixed medullary and follicular cell carcinoma. A second study identified expression of thyroglobulin in 39 of 43 canine follicular cell carcinomas and expression of calcitonin in 11 medullary carcinomas. 12 All follicular thyroid carcinomas were calcitonin-negative, and all medullary carcinomas were thyroglobulin-negative. 12 In another study of canine thyroid neoplasms, 81 follicular thyroid tumors were thyroglobulin-positive and calcitonin-negative, and 25 medullary thyroid tumors were calcitonin-positive and thyroglobulin-negative 11 ; 6 thyroid tumors expressed both calcitonin and thyroglobulin. 11 The authors did not comment on the morphology or distribution of the immunohistochemical expression patterns in these 6 neoplasms, and it remains uncertain whether these cases represent mixed medullary and follicular cell carcinomas or collision tumors or even entrapped hyperplastic foci of different lineage as the primary neoplastic cells.
Well-differentiated, compact, mixed medullary and follicular cell thyroid carcinoma is a rare neoplasm in humans and appears to be equally rare in dogs. Given that the diagnosis requires IHC for calcitonin and thyroglobulin, our case highlights the importance of using these markers for canine thyroid neoplasms to accurately identify this entity. Although there was no evidence of metastases or blood chemistry alterations in our case, analysis of a large case series is required to determine whether this entity has a less aggressive biologic behavior than medullary carcinomas, similar to its human counterpart.
Footnotes
Acknowledgements
We thank Dr. Jeff Walcoff at Bennett Animal Hospital (Clarksburg, MD) for examining the patient, performing the biopsy, and providing medical records; Veterinary Pathology Services (Stone Ridge, NY) for processing the biopsy samples; and the Michigan VDL histology laboratory for performing the IHC.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
