Abstract
Bovine herpesvirus type 5 (BHV-5) infection in calves causes meningoencephalitis, a fatal disease highly prevalent in South America. To study the pathogenesis of BHV-5 infection in cattle, 12 calves (group 1: acute infection) and 11 calves (group 2: latent infection) were intranasally inoculated with an Argentinean BHV-5 isolate at 108 and 104.7 tissue culture infective doses, respectively; six calves (control group) were mock infected. At 3 months postinoculation, all of the calves in group 2 and three calves in group 3 were given dexamethasone to reactivate the virus. The animals were euthanatized between days 6 and 17 postinoculation (group 1) and between days 6 and 16 postreactivation (group 2). Seventy-five percent and 91% of animals in groups 1 and 2, respectively, excreted BHV-5 in nasal and ocular discharges. Following dexamethasone administration, 45% of calves shed virus in both types of secretions. Spontaneous virus reactivation and shedding was observed in one calf. Neurologic signs consisting of circling, teeth grinding, ptyalism, jaw chomping, tongue protrusion, and apathy were observed in two animals in group 1 and, during the reactivation period, in four animals in group 2. Macroscopic findings consisted of softening of the cerebral tissue, meningeal hemorrhages and swelling, and edema and hemorrhages of prescapular, retropharyngeal and submandibular lymph nodes. Histologic lesions consisted of meningitis, mononuclear perivascular cuffing, neuronophagia, satellitosis, gliosis, hemorrhage, and necrosis and edema. Lesions in anterior cerebral cortex, medulla, and pons were consistently seen in all the animals of group 1. In the acutely infected animals, lesions in the diencephalon appeared at day 10 postinoculation, whereas in the latently infected calves these lesions were observed as early as at day 6 postreactivation. Latently infected animals developed lesions simultaneously in anterior cortex, medulla, pons, and diencephalon, showing a remarkable difference from the acutely infected group. Trigeminal ganglionitis appeared relatively early in animals of both groups (day 7 postinoculation in group 1 and day 8 postreactivation in group 2).
Bovine herpesvirus type 5 (BHV-5) is the causal agent of necrotizing meningoencephalitis in young cattle.17,21,28 Although antigenically related to bovine herpesvirus type 1 (BHV-1), BHV-5 has a more limited geographic distribution, with a high incidence of neurologic disease in South American countries, mainly Argentina and Brazil. In contrast, the occurrence of BHV-5–associated neurologic disease has been only sporadically reported in other parts of the world.17,26,28,29,32 BHV-1 and BHV-5 are difficult to differentiate by both virologic and serologic methods. Although both viruses are neurotropic, they differ in their ability to cause encephalitis.1 BHV-1 may be occasionally neuroinvasive but rarely causes encephalitis, whereas BHV-5 has a marked neurotropism, frequently leading to fatal disease. Several pathways have been proposed for the invasion of the bovine nervous system by BHV-5; however no work has been conclusive in this respect.4,13
In the present experiment, we studied the pathogenesis of an Argentinean BHV-5 isolate in the bovine nervous system because of the frequent fatal outbreaks of meningoencephalitis reported in Argentina. We studied the primary (acute) infection, latency, and reactivation of the infection by this neurotropic strain of BHV and the pathways used by the virus to access the neural tissue. We also established the nature, timing and distribution of the pathologic changes caused by BHV-5 during the acute infection and early postreactivation periods.
Materials and Methods
Animals
Twenty-nine crossbred calves 12 months old and weighing 190 ± 28 kg (mean ± SD) were used in this experiment. All the animals were free of detectable antibodies to BHV-1 and BHV-5. Calves were maintained in isolated pens and fed on grass hay and water ad libitum.
Cells and virus
Madin-Darby bovine kidney (MDBK) cells were cultured in minimum essential medium (MEM; Gibco BRL, Gaithersburg, MD) supplemented with 7% bovine fetal serum,
The virus studied has been characterized as a BHV-5 strain, according to a differential polymerase chain reaction protocol previously published.14 The isolate used for inoculation (No. 97/613) was recovered from the brain of a 2-year old calf with clinical encephalitis.
Animal inoculation and experimental design
Calves were inoculated intranasally by aerosolization with 25 ml of inoculum, evenly distributed in both nostrils. The animals were randomly assigned to the following treatment groups: group 1 (calf Nos. 1–12) for study of primary (acute) infection and group 2 (calf Nos. 13–23) for study of latency and reactivation. Mock-infected calves (group 3: calf Nos. 24–29) were included as controls. The inoculum for calves in group 1 (calf Nos. 1–12) and group 2 (calf Nos. 13–23) contained 108 tissue culture infective doses (TCID50) and 104.7 TCID50 to induce the acute and latent infection, respectively. Mock-infected calves (Nos. 24–29) were inoculated with 25 ml of MEM.
At 3 months postinoculation (PI), all the calves in group 2 (calf Nos. 13–23) and three calves in group 3 (calf Nos. 27–29) were given dexamethasone (DXM; Dexametasona∗ Vet, Schering Plough, Sanidad Animal, Argentina) at a dose of 0.1 mg/kg/day, intravenously for 5 consecutive days.
Calves in both groups were monitored daily for rectal temperature, presence of nasal and ocular discharges and neurologic signs. Animals in group 1 (calf Nos. 1–12) were sequentially euthanatized on days 6–17 PI, and calves in group 2 (calf Nos. 13–23) were euthanatized on days 6–16 after DXM treatment (days postreactivation [PR]). Calves were deeply anesthetized and euthanatized by intravenous administration of chloral hydrate and magnesium sulfate. Mock-infected calves (Nos. 24–26, Nos. 27–29) were euthanatized on days 6, 10, and 16 (PI and PR, respectively).
Antemortem sample collection
Nasal and conjunctival swabs were collected daily from animals in groups 1 and from three animals in group 3 (calf Nos. 24–26). For calves in group 2 and three animals in group 3 (calf Nos. 27–29), the same samples were collected daily during the first 16 days PI then weekly until the beginning of DXM treatment and daily again until day 16 PR. The nasal and conjunctival mucosa was gently scraped with a cotton-tip swab, which was then transferred to 1 ml of 1× Hank's solution.
For animals in all groups, serum samples were collected on days 0, 7, and 14 PI and immediately before euthanasia. For animals in group 2 and calf Nos. 27–29 in group 3, samples were also collected weekly during the latency period and on days 0, 7, and 14 PR.
Postmortem sampling and histology
Complete necropsies were performed immediately after euthanasia, and macroscopic lesions were recorded. Once the brain was removed, it was transversally sliced into eight 1–2-cm-thick sections. The following areas from each cerebral hemisphere were evaluated for virus isolation: anterior cortex (four samples), posterior cortex (two samples), cerebellum (one sample), medulla and pons (three samples), and diencephalon (one sample). Trigeminal ganglia, adrenal gland, prescapular lymph node, spleen, and cerebrospinal fluid were also collected. The same tissue samples were fixed in 10% neutral buffered formalin for histologic examination. Paraffin-embedded tissues were sectioned and stained with hematoxylin and eosin (HE) using routine methods. Histologic lesions were classified according to a score of severity, which was determined by the proportion of tissue that showed microscopic lesions: 0 = absence of lesion; 1 = mild; <25% of tissue affected; 2 = moderate, 25–50% of tissue affected; and 3 = severe, >50% of tissue affected.
Virus isolation and identification
Nasal and ocular swabs and cerebrospinal fluid specimens were diluted (1:5) in MEM supplemented with penicillin–streptomycin (20 µl/ml), gentamicin (2 µl/ml), and amphotericin B (40 µl/ml). Tissue samples were processed as a 10% (w/v) homogenized suspension and centrifuged at 1,000 × g for 15 minutes at 4 C. Fifty microliters of supernatant was inoculated in duplicate into monolayers of MDBK cells in 96-well plates and incubated at 37 C. Samples were passaged 4–6 times, 3 days for each passage, and monitored daily for cytopathic effect (CPE). At the end of each passage, the samples were tested by direct immunofluorescence using a BHV-1 conjugate (IBR Bovine Direct FA Conjugate, American Bioresearch, Seymour, IN).
Serology
Antibody titers to BHV-1 and BHV-5 were measured by the serum neutralization technique34 using twofold dilutions of serum against a fixed dose of virus (100 TCID50/well). MDBK cells were used as indicators. Antibody titers were expressed as the reciprocal of the highest serum dilution that prevented the production of CPE after a 72-hour incubation period.
Statistical analysis
Rectal temperatures were analyzed as repeated measurements using an analysis of variance (MIXED procedure, SAS/STAT Institute, Cary, NC, 1995). When treatment × day interactions were significant (P < 0.05), treatment least-squares means (LSM) within days were compared. LSM between groups were compared by Dunnett's test. Statistical analysis for histologic lesions was performed with Kruskal–Wallis test for nonparametric data.31 LSM were compared using the Tukey–Kramer test. For all tests, a 5% significance level was used.
Results
Clinical observations
All the animals in group 1 (calf Nos. 1–12) displayed nasal and ocular discharges from day 2 to day 7 PI, continuing in some of them (calf Nos. 1, 5, 7) until day 12 PI. The animals in group 2 (calf Nos. 13–23) had nasal and ocular discharges from day 1 to day 11 PI. The highest number of calves (Nos. 13–15, 17, 19, 20, 23) showing both types of discharges was observed between day 3 and day 5 PI. Likewise, nasal and ocular discharges were observed starting day 5 after the beginning of DXM treatment and continuing to day 9 PR.
Rectal temperatures were not significantly different (P > 0.05) between group 1 calves (Nos. 1–12) and control calves (Nos. 24–26). Significant differences (P < 0.05) for the temperature records in group 2 calves (Nos. 13–23) and control calves (Nos. 27–29) were detected on days 10 and 54 PI and during the period of DXM administration. Differences between both groups were also detected from day 9 to day 16 PR.
Neurologic signs were observed in calf Nos. 1 and 3 on days 16 and 17 PI, respectively. The clinical signs started on day 12 PI and were characterized by apathy, ptyalism and teeth grinding. Calf No. 1 also showed constant circling, and calf No. 3 displayed jaw chomping and tongue protrusion. Because the remaining animals were euthanatized according to a predetermined sequence it is not known whether neurologic signs would have developed.
During the reactivation period, four animals in group 2 (calf Nos. 13, 21–23) developed neurologic signs. Calf No. 13 died on day 3 PR (on day 8 after the first DXM administration) after showing apathy and constant circling. The other three calves (Nos. 21–23) showed slight and intermittent neurologic signs, including apathy, intense ptyalism, teeth griding, and continuous tongue and jaw movements. Clinical signs in calf No. 23 started on the last day of DXM injection and were present for 3 days. Calf Nos. 21 and No. 22 showed the same neurologic signs on day 8 PR. Slight nervous symptoms were also observed in calf No. 22 on day 12 PR.
Postmortem findings
Gross lesions were observed in animals euthanatized between day 12 and day 17 PI (group 1, calf Nos. 1–6) and in calf No. 13 (group 2), which died on day 3 PR. The main cerebral lesion consisted of softening of the parenchymal tissue. Although the frontal lobes (olfactory bulbs) were always affected, lesions rarely were symmetrical or followed a well-defined pattern of distribution. Calf No. 13 showed focal meningeal hemorrhages in the frontal area of the brain and extensive hemorrhagic foci in the pons and in the left parietal lobe. Other lesions included swelling, edema, and focal hemorrhages in prescapular, retropharyngeal and submandibular lymph nodes of animals in group 1 (calf Nos. 1–12).
Histopathology
Microscopic lesions observed in calves euthanatized on a particular postinoculation day were characterized by nonsuppurative meningitis, mononuclear perivascular cuffing, neuronophagia, satellitosis, focal and diffuse gliosis, hemorrhages, and neuronal necrosis and degeneration (Figs. 1–4, Table 1). Inclusion bodies were not observed. Necrosis of lymphoid cells and medullar hemorrhages were observed in prescapular and submandibular lymph nodes of acutely infected calves (Nos. 1–12).
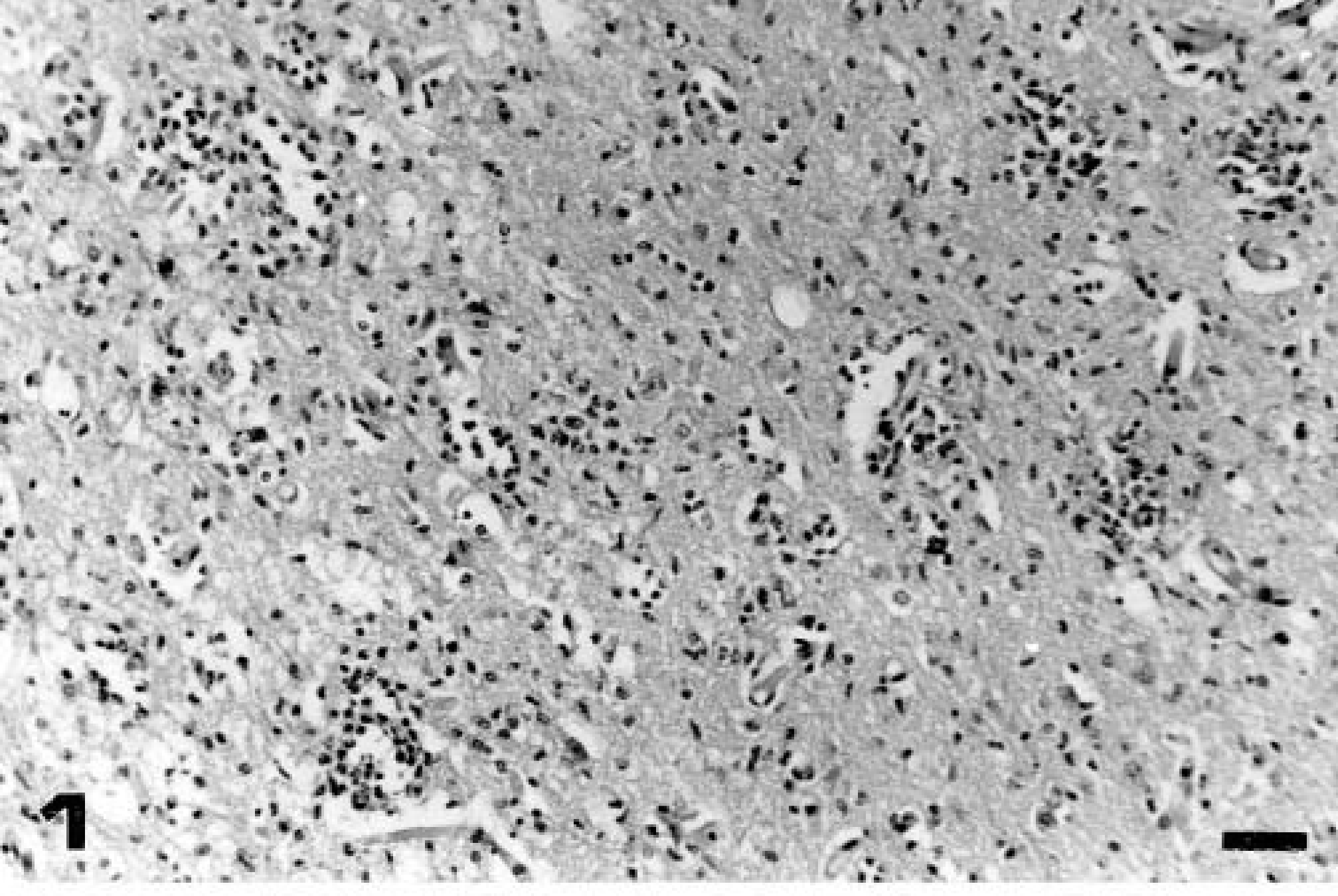
Anterior cerebral cortex; calf No. 2 (group 1, euthanatized on day 15 PI). Nonsuppurative encephalitis with multifocal gliosis and satellitosis. HE. Bar = 70 μm.
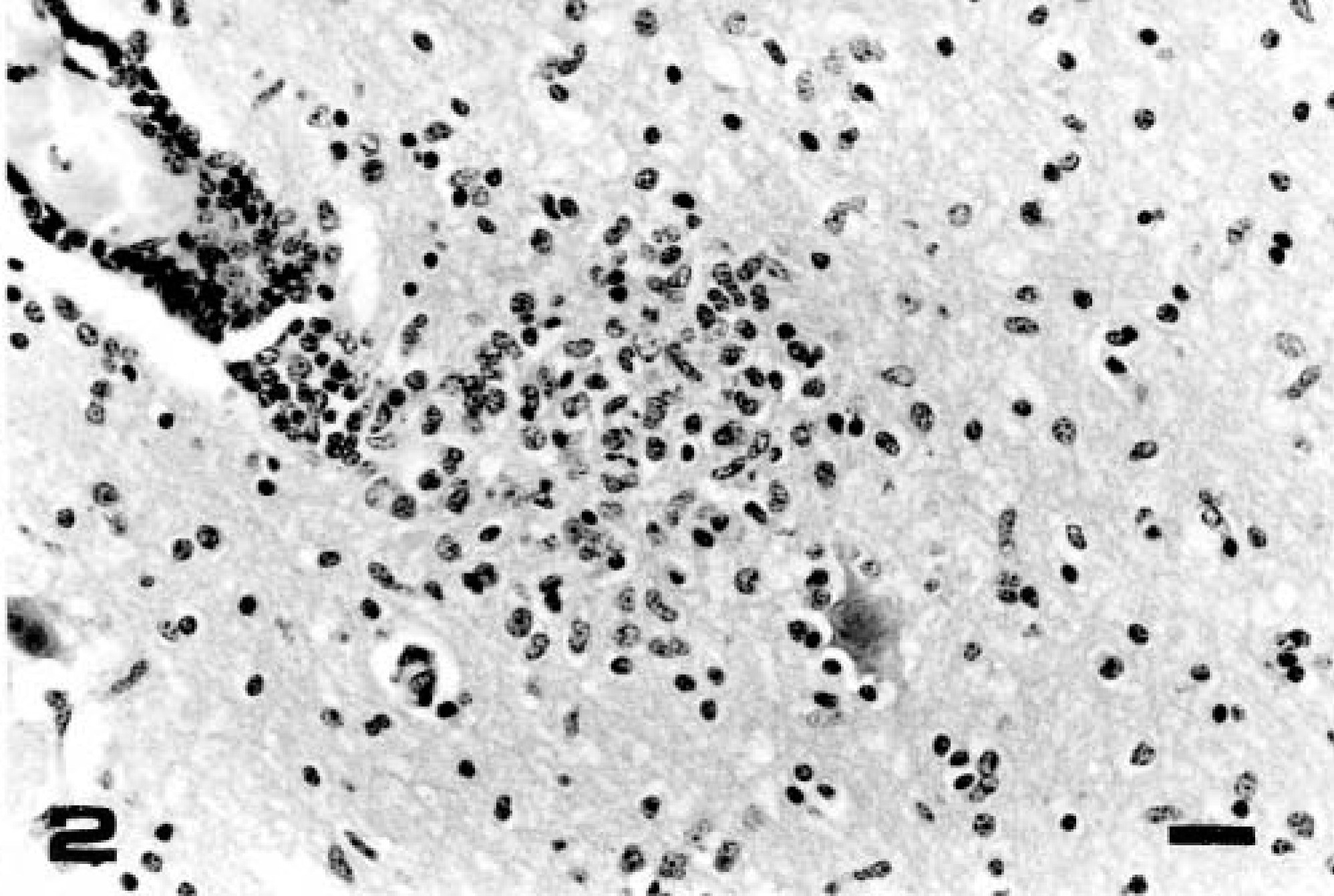
Anterior cerebral cortex; calf No. 3 (group 1, euthanatized on day 17 PI). Perivascular mononuclear cuffing, focal gliosis, satellitosis, and neuronophagia. HE. Bar = 35 μm.
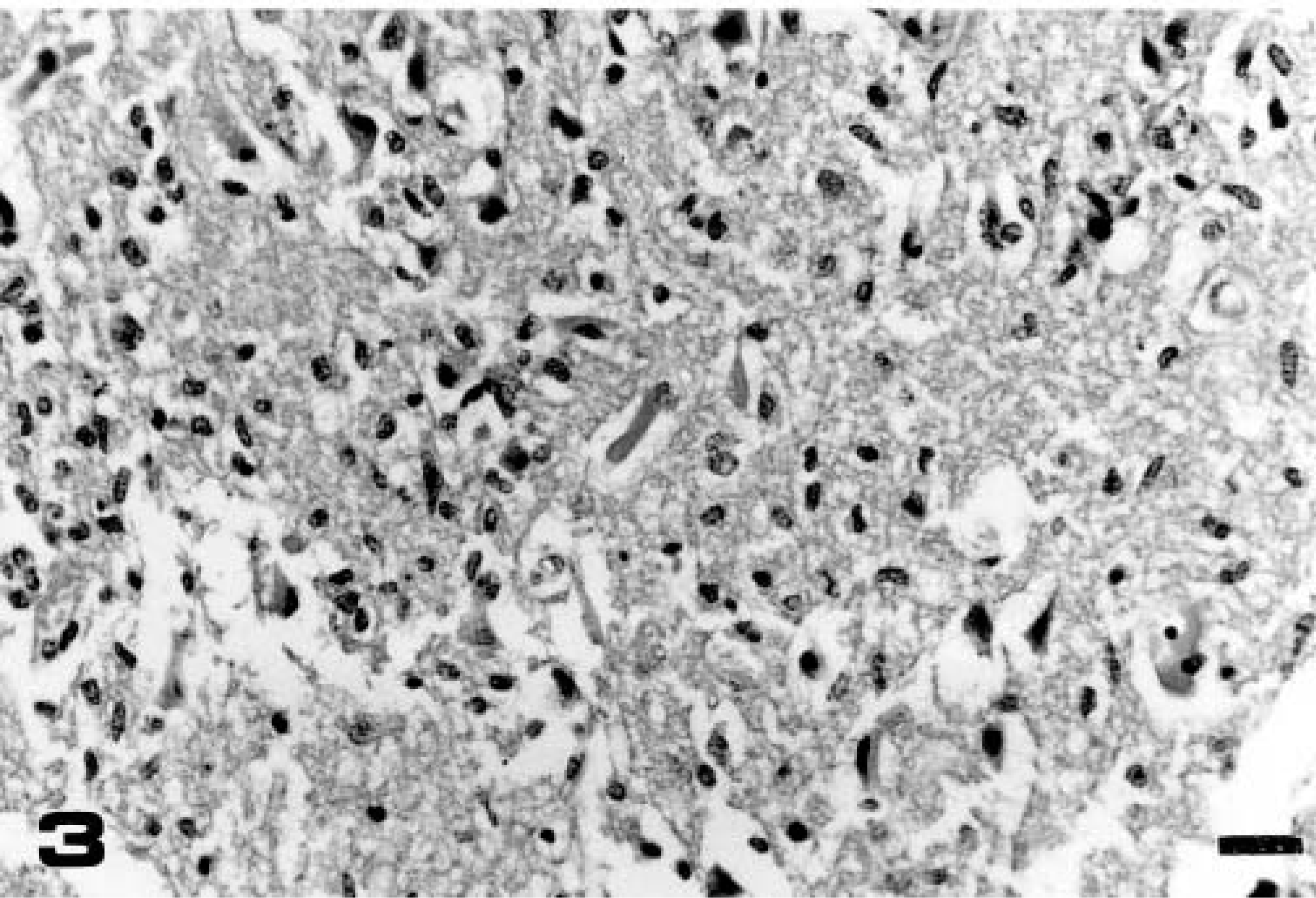
Anterior cerebral cortex; calf No. 1 (group 1, euthanatized on day 16 PI). Necrosis, loss of brain tissue structure, and edema in malacic foci. HE. Bar = 35 µm.
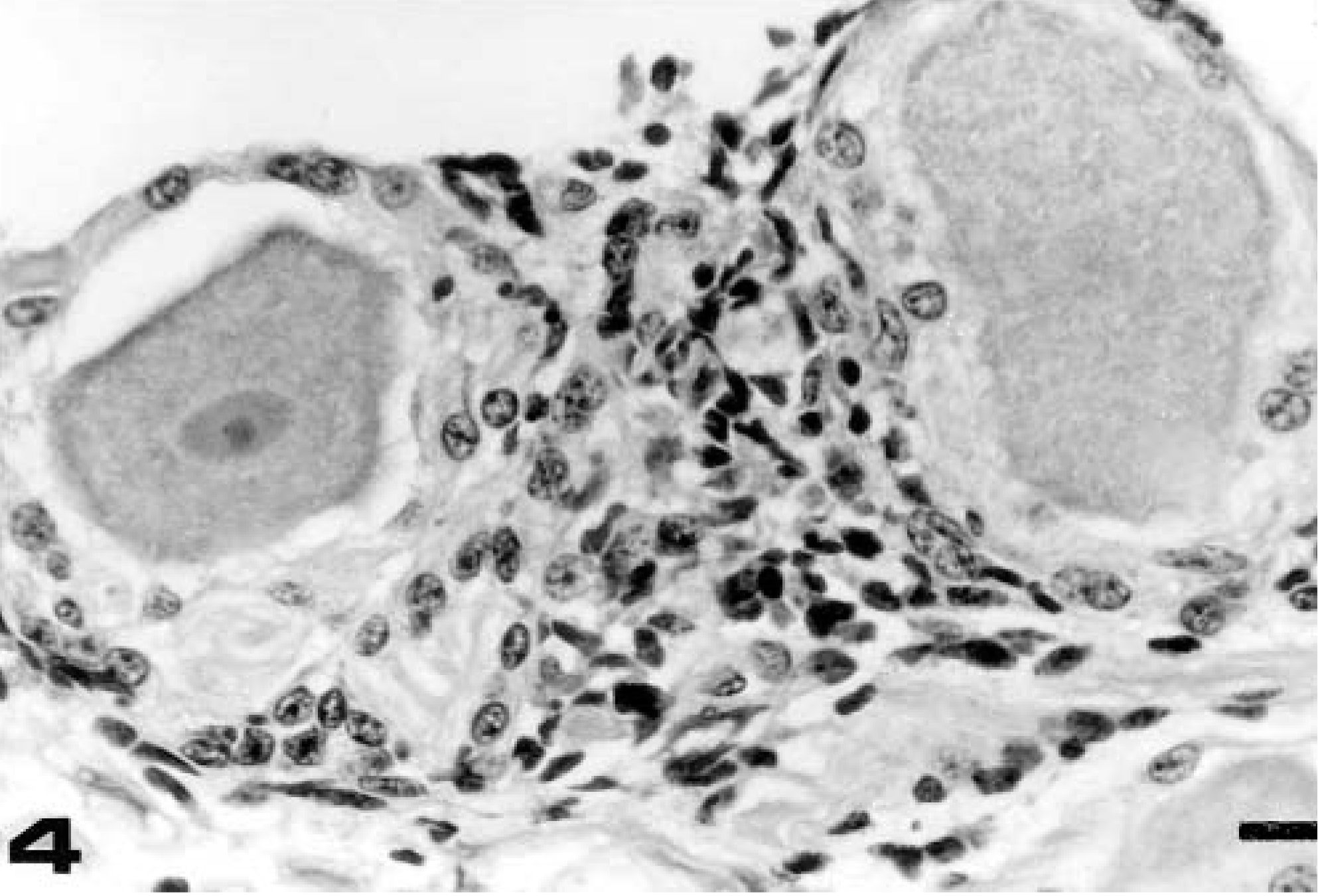
Trigeminal ganglion; calf No. 21 (group 2, euthanatized on day 14 PR). Nonsuppurative focal ganglionitis. HE. Bar = 20 μm.
Frequency of histologic lesions∗ in the nervous system of calves acutely infected (group 1, calf Nos. 1-12) and latently affected (group 2, calf Nos. 13-23) with BHV-5.

Number (%) of calves with lesions.
Perivascular cuffing.
No significant differences (P > 0.05) were observed in the distribution of lesions in neural tissue between groups 1 and 2. For both treatment groups, the frequency of lesions in anterior cerebral cortex was higher (P < 0.01) than in the cerebellum and diencephalon. Cerebellum was the less affected tissue.
Distribution of lesions by neural tissue area. All microscopic lesions described above were present in anterior cortex. Neuronophagia and satellitosis were the lesions most frequently observed in this area (Tables 2, 3). For calves in groups 1 and 2, meningitis was exclusively observed in anterior and posterior cortex, and for calves in group 1, perivascular cuffing was equally distributed in anterior and posterior cortex, medulla, and pons (Table 2). Necrosis was detected only in group 1 calves (Nos. 1–3, 5) and was evident in anterior cortex, medulla, and pons (Table 2). For animals in group 2, perivascular cuffing was not consistently found and was mainly observed in anterior cortex (Table 3).
Distribution of histologic lesions∗ in the central nervous system of calves (group 1, calf Nos. 1-12) acutely infected with BHV-5.
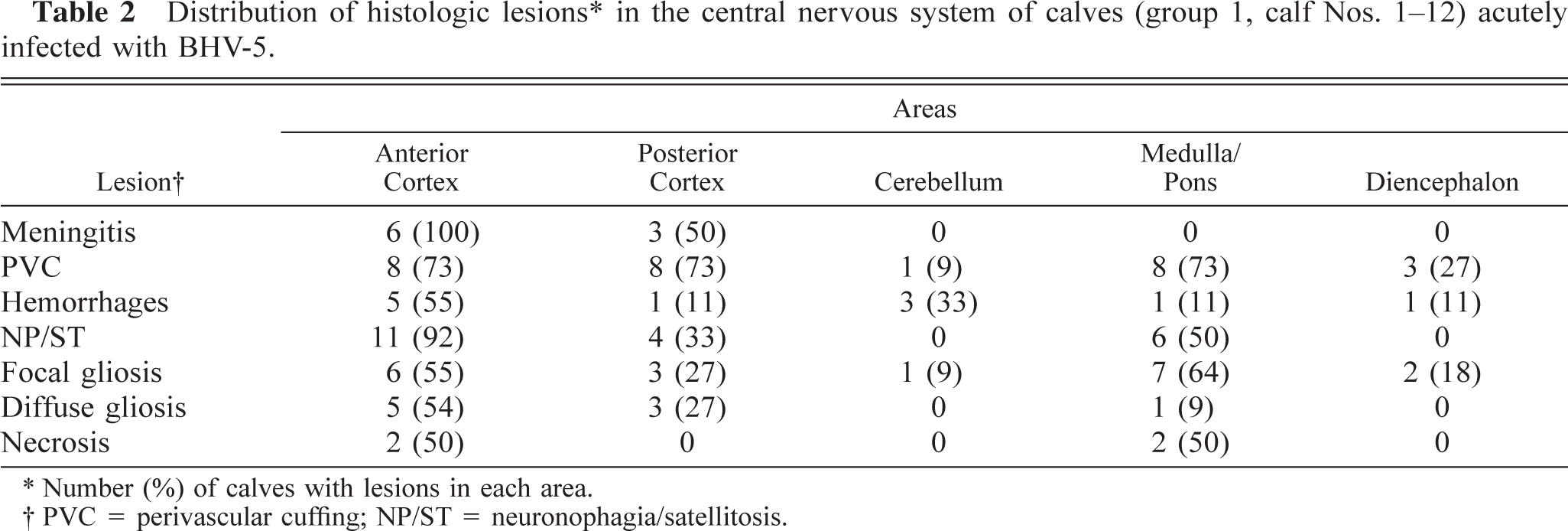
Number (%) of calves with lesions in each area.
PVC = perivascular cuffing; NP/ST = neuronophagia/satellitosis.
Distribution of histologic lesions∗ in the central nervous system of calves (group 2, calf No.2 13-23) latently infected with BHV-5.
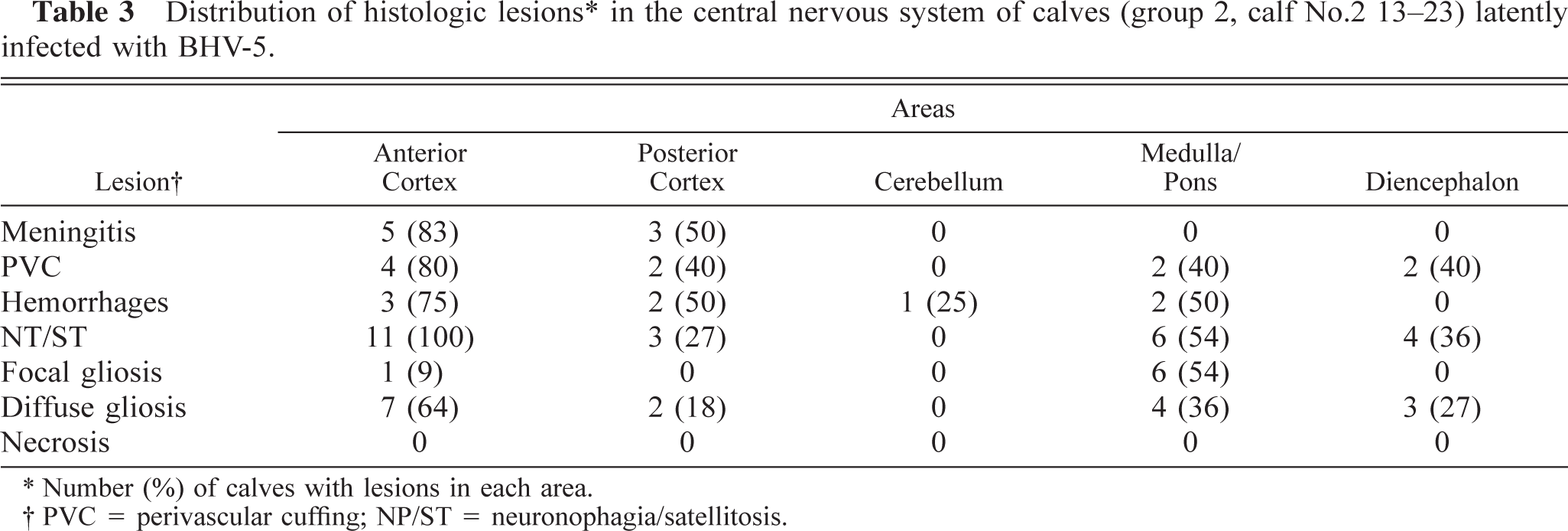
Number (%) of calves with lesions in each area.
PVC = perivascular cuffing; NP/ST = neuronophagia/satellitosis.
Distribution of lesions by time of euthanasia. For animals in group 1, the lesions in anterior cerebral cortex, medulla and pons were observed in animals euthanatized from day 6 to day 17 PI. In the posterior cerebral cortex, lesions were not detected, whereas in the diencephalon they were apparent from day 10 PI. Microscopic changes in trigeminal ganglia were observed starting at day 7 PI (calf No. 7). Cerebellar lesions were only detected on day 7 PI. Likewise, for animals in group 2, lesions in anterior cortex were present through days 6–16 PR. Lesions became apparent in the posterior cortex on days 9, 10, and 11 PR. The medulla, pons, and diencephalon areas showed pathologic changes from day 6 to day 14 PR. In the trigeminal ganglia, lesions were found from day 8 PR. In cerebellar tissue, lesions were observed only on day 8 PR (calf No. 16).
Perivascular cuffing, necrosis and focal gliosis were more severe (P < 0.01) for animals in group 1 (Table 4), and although hemorrhage was more frequently seen in animals in this group, it was not severe (P > 0.05) (Tables 2 and 4). Differences in the severity of the lesions according to their location in the neural tissue were not detected (P > 0.05) between group 1 and group 2 calves. In both treated groups, focal gliosis was more severe (P < 0.05) in medulla and pons than in other neural areas, and diffuse gliosis was significantly more severe (P < 0.05) in trigeminal ganglia.
Mean (SEM) score of severity for the histologic lesions in calves acutely infected (group 1, calf No. 1-12) and latently infected (group 2, calf Nos. 13-23) with BHV-5.
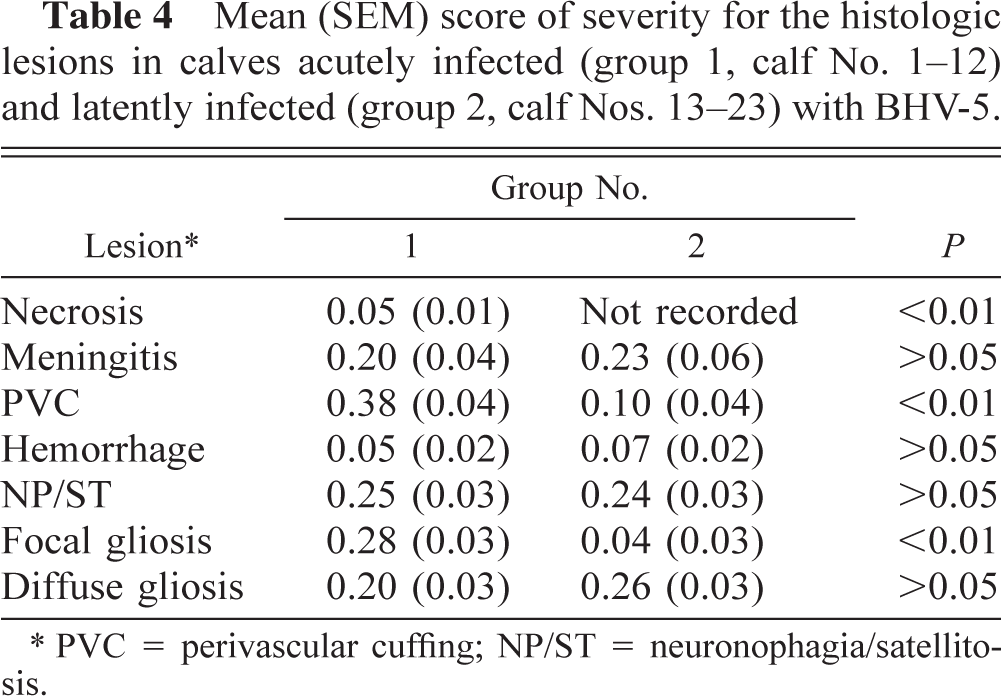
PVC = perivascular cuffing; NP/ST = neuronophagia/satellitosis.
For animals in group 1, the severity of meningitis and perivascular cuffing increased significantly (P < 0.05) from day 15 to day 17 PI. Differences in the score of severity for focal gliosis was evident (P < 0.01) on day 16 PI. The other histologic lesions did not differ (P > 0.05) in relation to the time postinoculation.
Hemorrhagic lesions were more severe in calf No. 13 (P < 0.01) than in the rest of the animals.
Virus isolation
During the postinoculation period, 75% (9/12) of calves (Nos. 1–6, 9, 10, 12) in group 1 excreted BHV-5 in nasal and ocular secretions. Eight out of 12 (66%) calves (Nos. 1–3, 5, 6, 9, 10, 12) excreted BHV-5 in nasal discharges, and 42% (5/12) of the calves (Nos. 4, 5, 9, 10, 12) excreted BHV-5 in ocular discharges. The virus shedding peak took place between days 4 and 7 PI. BHV-5 was isolated from nasal secretions until day 17 PI and in ocular secretions until day 9 PI. In group 2, 91% (10/11) of calves (Nos. 13, 14, 16–23) excreted BHV-5 in nasal and ocular secretions during the whole experiment. Seventy-three percent of calves (Nos. 13, 14, 16–18, 20, 22) excreted BHV-5 in nasal discharges, and 27% (Nos. 17, 19, 23) shed the virus in ocular discharges.
Spontaneous viral excretion before DXM treatment was detected in one calf (No. 13). This calf shed BHV-5 on day 91 PI, before the first injection of corticosteroid. From day 4 after DXM treatment, the virus was isolated from secretions from 45% of group 2 calves (Nos. 14, 17, 18, 21, 23). Viral excretion was not detected in mock-infected calves (Nos. 24–29).
BHV-5 was not isolated from tissue samples in animals of any group assayed upon necropsy.
Serology
Three calves in group 1 (calf. Nos. 5–7) exhibited seroconversion between days 12 and 14 PI. The highest neutralizing antibody titer was 1:64 (Nos. 5, 6). The results of serologic assays for animals in group 2 are shown in Table 5. All animals in group 2 seroconverted between days 14 and 33 PI. At the beginning of DXM treatment, serum antibody titers ranged from 1:128 to 1:512. After DXM administration, six calves (Nos. 14, 17, 19–21, 23) experienced a rise in antibody levels. The highest titer recorded was 1:1,024 (Calf No. 21) on day 14 PR. Mock-infected calves (Nos. 24–29) remained serologically negative throughout the experiment.
Serum antibody titers∗ to BHV-5 for the latently infected calves (group 2, calf Nos. 13-23)
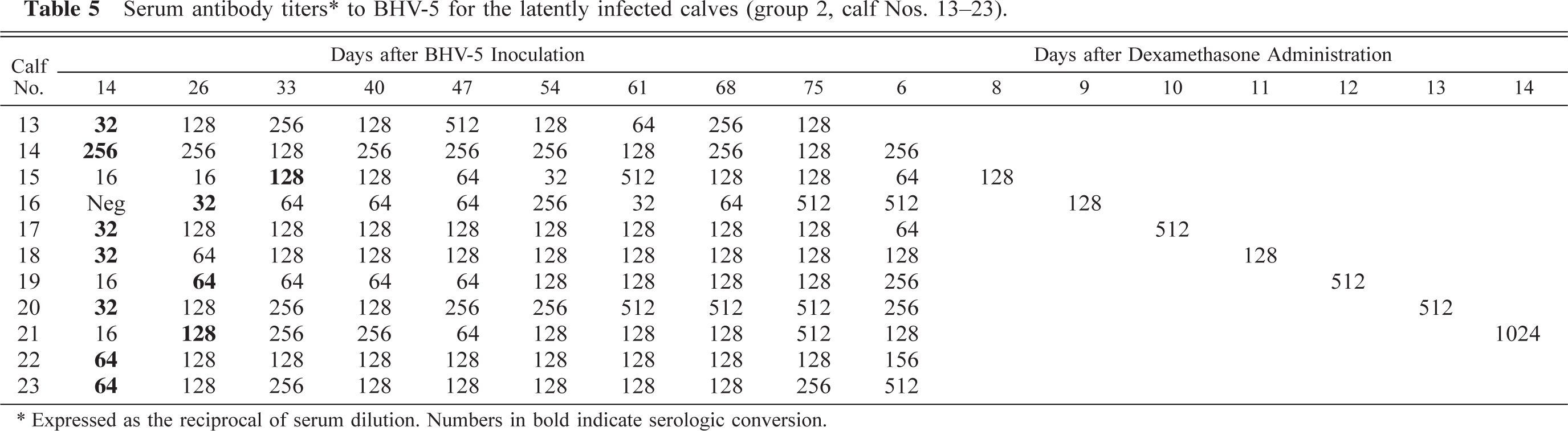
Expressed as the reciprocal of serum dilution. Numbers in bold indicate serologic conversion.
Discussion
Clinical signs, gross and microscopic findings, and virus isolation data show that BHV-5 is able to induce a primary (acute) infection in calves after experimental inoculation. Virus shedding, serologic conversion, histologic findings, and clinical manifestations during the reactivation period indicate that BHV-5 can establish a latent infection that can be reactivated by administration of synthetic corticosteroids.
Previous experimental inoculations to reproduce clinical encephalitis have produced variable results.1,9 For this reason, it has been suggested that BHV-5 might be neuroinvasive but not always neurovirulent.1 Others have described a model of fatal encephalitis in neonatal calves deprived of colostral antibodies.4 In the present study, we demonstrated that Argentinean strain 97/613 was both neuroinvasive and neurovirulent in animals whose ages were similar to those of calves affected in typical disease outbreaks.6–8,27
After the administration of DXM, BHV-5 was reexcreted by 45% (5/11) of animals (Nos. 14, 17, 18, 21, 23), showing a successful reactivation of the latent infection. The increase in neutralizing antibody titers, observed in some experimentally infected animals after dexamethasone treatment, was probably a consequence of the restimulation of the humoral response by the reactivation of the latent virus, as has been previously described.19
Neurologic signs and macroscopic lesions observed during the acute infection were similar to those described in other reports on experimental inoculations of BHV-5.4 These signs also resembled those occurring in the natural field outbreaks. Herpesvirus reactivation usually occurs in the absence of overt clinical signs. The reduced number of neurons involved in each episode of recrudescence and the extremely limited virus production during reactivation would be presumably associated with the absence of fatal disease.11 However, in the present study reactivation resulted in clinical signs and the development of a severe neurologic disease (calf No. 13), accompanied by gross and microscopic lesions in neural tissue. Because this animal excreted BHV-5 before the initiation of DXM administration, the spontaneous reactivation, possibly associated with a stressful condition, may have been exacerbated by the corticosteroid treatment. Slight and intermittent signs in the other three animals (Nos. 21–23) resemble those observed in humans during recrudescence episodes of herpes simplex virus infection.20,30 Spontaneous and DXM-induced reactivation followed by recrudescence of neurologic disease, frequent but not always fatal, have been recently reported in a rabbit model for BHV-5 latency (L. Caron, personal communication).
Alphaherpesviruses multiply rapidly and produce CPE in cell cultures. However, in a survey of neuropathologic cases, 31 samples with a histologic diagnosis of nonsuppurative encephalitis were analyzed, and BHV-5 was isolated from only 13 (49.9%) of these cases.5 Other authors did not isolate the virus from cattle with neurologic signs and a histologic diagnosis of necrotizing encephalitis in field outbreaks or they failed to isolate BHV-5 from several areas of the central nervous system of calves that had been inoculated intranasally.6,9 In this experiment, BHV-5 was not isolated from any tissue, which indicates that the use of cell cultures for the isolation of BHV-5 is not a sensitive method for this type of sample. Low to moderate viral titers were reported in neural tissue,4 which suggests that these titers are not high enough to permit virus isolation.
Histologic lesions described in this study are typical of bovine herpetic encephalitis.2–6,9,10,12,15,16,18,22–25 The nature and location of these lesions were similar for both acutely and latently infected animals except for the necrosis, which was exclusively found in calves euthanatized in the advanced stages of the acute infection (calf Nos. 1–3, 5) and in calves that showed neurologic signs (calf Nos. 1. 3) and/or macroscopic tissue softening (calf Nos.1–3, 5).
For both inoculated groups, anterior cerebral cortex was the most severely affected area. The finding of severe microscopic changes, especially focal gliosis, in medulla and pons indicates that this region of the nervous system is useful for diagnostic purposes.
Histologic lesions in anterior cortex, medulla, and pons were evident from the beginning to the end of the euthanasia sequence. Although only one calf from each group was euthanatized on each day, a similar temporal sequence and distribution of lesions was detected in both groups. This finding indicates that BHV-5 accesses the bovine nervous system by the same pathway for both acute and latent infection. Lesions in the diencephalon appeared later (day 10) in acutely infected calves (Nos. 1–12) than in latently infected calves (Nos. 13–23) (day 6). However, the differences observed in when lesions become evident in the diencephalon can be used to define the development of each type of clinical presentation. Because no animals in this study were euthanatized before corticosteroid treatment, we cannot establish whether microscopic lesions occurred during the primary infection or were produced or aggravated by the reactivation of the virus. However, it is unlikely that calves with encephalitis would remain clinically healthy, without showing clinical signs during a period of 90 days after virus inoculation.
Trigeminal ganglionitis appeared relatively early during the course of infection in calves of both groups. This finding suggests that the trigeminal nerve is an important route of access to the nervous system. The lesions developed simultaneously in anterior cortex, medulla, pons, and trigeminal ganglia, suggesting that BHV-5 reaches the brain by axonal transport from bipolar neurons of the olfactory nerves to the olfactory bulbs. During acute infection, BHV-5 would also access the trigeminal ganglia, but the fact that microscopic changes appeared later in the diencephalon than in the other areas indicates that viral invasion of the brain occurs more directly along olfactory nerves, as previously described in a rabbit model.10
Latently infected calves (Nos. 13–23) developed microscopic changes simultaneously in anterior cortex, medulla, pons, and diencephalon, showing a remarkable difference from the acutely infected calves (Nos. 1–12). In latently infected animals, dissemination of BHV-5 to the cerebral tissue would also occur by viral transport from mesencephalon to telencephalon via the trigeminal nerve, with successive passages through metencephalon and mesencephalon as described for pseudorabies virus.33
The results of this experiment provide important contributions to the understanding of BHV-5 neuropathogenesis in calves. A striking finding of this study was the capability of BHV-5 to establish latency, with reactivation provoking neurologic symptoms in calves. Moreover, microscopic findings have provided information on the mechanisms of BHV-5 infection of the bovine nervous system. However, the use of molecular techniques with tissue samples from sequentially necropsied animals is necessary to confirm the central nervous system invasion pathways that were suggested by the lesion distribution observed in this study.
