Abstract
Cyclooxygenase-2 (Cox-2) is commonly upregulated during human colorectal tumorigenesis, and its contribution to this process has been clearly demonstrated in genetic mouse models. The only other species that naturally develops intestinal cancer with any frequency is the domestic dog. Intestinal carcinogenesis in humans has been strongly linked to environmental factors such as diet, which may be shared by household pets. We have previously reported that β-catenin is overexpressed in the neoplastic epithelium of canine colorectal polyps, as it is in humans and rodents. We now show that Cox-2 is also upregulated in the majority of these lesions. Thirteen out of 20 colorectal adenomas (65%) contained immunohistochemically detectable Cox- 2 protein restricted to the nonneoplastic tumor stroma, including myofibroblasts and β-smooth muscle actin-negative mesenchymal cells morphologically consistent with macrophages and/or fibroblasts. In contrast to benign polyps, seven of 15 adenocarcinomas (47%) also expressed Cox-2 in the neoplastic epithelium. These changes duplicate molecular changes in human intestinal tumorigenesis and substantiate a fundamental role for both β-catenin and Cox-2 in intestinal neoplasia.
Colorectal cancer is a significant cause of morbidity and mortality in the United States, with diagnosis of approximately 130,000 new cases expected in 1999.24 This form of cancer has been a prototype in defining genetic and epigenetic molecular changes associated with initiation of early dysplastic changes, development of adenomas, and progression to malignancy (carcinogenesis).1,7,37 Numerous laboratories have contributed to the characterization of these molecular changes in inherited and sporadic forms of human colorectal cancer.21,35 In vitro and in vivo studies with laboratory animal models have provided a better understanding of the specific abnormal gene function relationships that contribute to intestinal neoplastic cell growth and behavior.8,14,16,45 From this body of work, it has become clear that mutations resulting in aberrant upregulation of cytoplasmic and nuclear β-catenin play a key role in human colorectal carcinogenesis.35
Carcinogenesis in several human organ systems has been strongly associated with environmental factors, and diet is often singled out as being particularly important in this regard, especially in the case of colorectal cancer.26 One potential molecular link to dietary factors is cyclooxygenase-2 (Cox-2), which is commonly overexpressed during the early stages of intestinal tumor development.6,13,52 Dietary lipids can significantly modify tissue levels of arachidonate, which is the substrate for Cox-2, thereby affecting the generation of protumorigenic prostaglandins produced by this enzyme.12 Although the mechanisms responsible for upregulation of Cox-2 in neoplastic tissues are still unclear, the enzyme clearly contributes to intestinal tumorigenesis in rodent models and is a potential target for nonsteroidal anti-inflammatory drugs (NSAIDs), which cause regression of colorectal adenomas in both humans and laboratory animals.4,19,33 Cox-2 is an inducible enzyme that is localized around the nuclear envelope and produces eicosanoids, such as prostaglandin (PG) E2, which contribute to neoplastic cell growth.6,31,42,43,50,52 Dietary factors (e.g., n-3 fatty acids found in fish oil) can significantly decrease PGE2 production and inhibit intestinal tumor growth in vivo, presumably by altering substrate availability for the Cox-2 enzyme.12,25,26 Despite direct and indirect evidence indicating the importance of Cox-2 during intestinal carcinogenesis, there have been relatively few and sometimes conflicting reports describing the distribution of the protein in neoplastic tissues of humans and mice.3,13,14,18,32,33,38–41,44,51
Other than humans, the only species that naturally develops intestinal cancer with any frequency is the domestic dog, which shares our environment and in some households our diet.5,15,34,48 Canine colorectal adenomas may progress to malignancy, as in humans, but little is known about the molecular basis for tumorigenesis in these animals.9,48,54 As in human and mouse intestinal polyps, β-catenin is strongly overexpressed in the cytoplasm and nucleus of neoplastic canine colorectal epithelial cells, suggesting conservation of early intestinal carcinogenic mechanisms between species.28 Because of this analogy, we were interested in determining whether Cox-2 is also overexpressed in canine intestinal tumors and, if so, in what populations of cells.
Materials and Methods
Thirty-one of the 37 cases of colorectal adenoma reported in the current study have been previously characterized with respect to aberrant expression of β-catenin, E-cadherin, and p53;28 six new cases of colorectal adenoma have been added to this original collection. Tissues were from privately owned dogs and were obtained either as a result of surgical biopsy (n = 34) to diagnose and treat the clinical disease or after euthanasia (n = 3) sanctioned by the owner. More than 15 breeds of dogs were represented and none of the animals were related. The mean age was 8.4 years (range, 1–16 years) with 19 males (two castrated) and 18 females (15 spayed). Dogs for whom tissues were obtained at necropsy were selected based on a minimal postmortem period before tissue collection. All samples were routinely fixed for 18–72 hours in 10% neutral buffered formalin (NBF) before processing into paraffin for histologic sectioning. Normal canine kidney, used as a positive control for Cox-2, was placed in NBF within minutes of euthanasia and fixed for 48 hours before processing.
Primary antibodies included a rabbit polyclonal antibody raised against a mouse Cox-2 peptide (No. 160116; Cayman Chemical, Ann Arbor, MI), a mouse monoclonal antibody against the rat Cox-2 protein (No. C22420, clone 33; Transduction Laboratories, Lexington, KY), and a monoclonal antibody that binds α-smooth muscle actin (α-SMA; No. A2547, clone 1A4; Sigma, St. Louis, MO). The six new colorectal polyps not included in our previous report were also stained for β-catenin (Transduction Laboratories monoclonal antibody) as previously described.28 Deparaffinized sections were pretreated in a 10 mM solution of citrate (pH 6.0) heated at or just below a boil for 10 minutes in a microwave oven and cooled to room temperature for 20 minutes. All sections were treated with 3% H2O2 in phosphate-buffered saline (PBS) followed by normal goat serum block (BioGenex, San Ramon, CA) and incubated overnight at 4 C with primary antibodies diluted 1:40 (6.3 µg/ml monoclonal Cox-2), 1:8,000 (polyclonal Cox-2), or 1:1,000 (6.5 µg/ml monoclonal α-SMA) in PBS (pH 7.6) containing 1% bovine albumin and 0.1% Tween-20. Sections were then incubated with biotin-conjugated goat anti-mouse or anti-rabbit immunoglobulin (50 minutes, 20 C; BioGenex) followed by streptavidin–peroxidase link (40 minutes, 20 C; BioGenex). Localized peroxidase conjugates were visualized with 3,3′-diaminobenzidine in sections lightly counter stained with hematoxylin.
Cox-2 staining in intestinal tumors was differentiated by location: 1) neoplastic epithelial cells; 2) nonneoplastic stromal cells just below the luminal surface of the tumor; 3) nonneoplastic cells deep within the stroma of the tumor; and 4) nonneoplastic cells immediately underneath or adjacent to the tumor but not part of the tumor mass (e.g., muscularis mucosa, submucosal fibroblasts, adjacent smooth muscle of tunics).
Staining for Cox-2 was scored in individual tumors according to the following criteria. For Cox-2 in tumor (nonneoplastic) stroma (including fibroblasts, myofibroblasts, macrophages, and smooth muscle fibers), scoring was as follows: 0 = negative; 1 = moderately intense staining of infrequent to scattered cells; 2 = multifocal to regionally strong staining (≥ macula densa control) in ≥15% of stromal cells; 3 = strong staining in most cells. For Cox-2 in tumor (neoplastic) epithelium, scoring was as follows: 0 = negative; 1 = rare positive epithelial cells; 2 = <5% of cells; 3 = 5–10% of cells; 4 = >10% of cells.
Stromal cells were identified as fibroblasts or macrophages based on histologic cell morphology; myofibroblasts were identified by immunohistochemical staining for α-SMA. Tumors were considered positive for Cox-2 if >5% of neoplastic epithelial cells (i.e., score of ≥3) and/or >15–20% of nonneoplastic stromal cells (score of ≥2) were immunohistochemically labeled.
Madin–Darby canine kidney cells (MDCK; American Type Culture Collection, Rockville MD) were cultured in Dulbecco's modified Eagle's medium (DMEM) supplemented with 10% heat-inactivated fetal bovine serum (FBS) and gentamicin (50 µg/ml). Cultures at 70% confluence in 100-mm plates were rinsed with sterile buffered saline and incubated 24 hours with DMEM containing 0.1% FBS, followed by 24 hours incubation with fresh medium (0.1% FBS) with or without 10 nM phorbol myristate acetate (PMA; Sigma). Duplicate PMA-stimulated and unstimulated cell cultures were then either lysed for western blot analysis as previously described30 or fixed in NBF. Fixed cells were gently scraped, pelleted, and suspended in 100 µl 1% high melting point agar before routine processing into paraffin for sectioning and immunohistochemistry.
For western blot analysis, total protein content of supernatants from MDCK cell lysates was determined by BCA protein assay according to the manufacturer's instructions (Pierce Chemical, Rockford, IL). Fifty micrograms of total protein from PMA-treated and untreated MDCK cultures, 10 µg total protein of an activated macrophage culture lysate (Cox-2 control; Transduction Laboratories), and molecular weight markers (Kaleidoscope Prestained Standards; BioRad, Cambridge, MA) were separated by 10% sodium dodecyl sulfate polyacrylamide gel electrophoresis and transferred to Immobilon-P membranes (Millipore, Bedford, MA). Membranes were incubated overnight at 4 C with Cox-2 antibodies diluted 1:1,000 (polyclonal) or 1:100 (2.5 µg/ml, monoclonal) in PBS containing 0.1% Tween-20 and 10% nonfat skim milk powder, followed by a 1-hour incubation at room temperature with peroxidase-conjugated anti-rabbit (1:5,000; Southern Biotechnology Associates, Birmingham, AL) or anti-mouse (1:80,000; Sigma) antibodies. Membranes were then incubated for 1 minute with freshly prepared ECL detection solution (No. RPN2209; Amersham, Piscataway, NJ) and exposed for <30 seconds to Hyperfilm ECL (Amersham), which was routinely developed and electronically scanned to generate a positive image of labeled protein bands.
All data were expressed as the mean ± SEM and were analyzed by Fisher's exact test (two tailed) or Welch's one way analysis of variance, as appropriate, using JMP software (SAS Institute, Cary, NC). Differences were considered significant at P < 0.05.
Results
Cox-2 is constitutively expressed in the macula densa of normal canine kidneys, whereas Cox-1 is not.20 Two primary anti-Cox-2 antibodies were tested on fixed sections of canine kidney to determine immunohistochemical specificity for the canine protein. Both antibodies clearly stained the nuclear envelope and circumferential perinuclear cytoplasm of epithelium forming the macula densa (Fig. 1), and both stained epithelial cells lining select tubules in the medulla and cortex with a fainter, more diffuse cytoplasmic pattern; the monoclonal antibody also stained endothelial cells lining parenchymal arterioles. To further establish specificity,2 canine cells (MDCK) were stimulated in vitro with the mitogen PMA to induce Cox-2 expression and were stained as fixed intact cells (i.e., processed in the same manner as tissue samples) or analyzed by western blot. Cox-2 protein is concentrated around the nuclear envelope,31 and there was relatively weak staining of the nuclear membrane in scattered untreated canine epithelial cells, with a dramatic increase in reactivity following PMA stimulation (Fig. 2). Staining was completely eliminated when the primary antibody was replaced by nonspecific murine IgG or rabbit serum (not shown). In two separate experiments, both antibodies produced a single band in western blots that migrated at a relative molecular mass of approximately 70 kd. This band was distinctly larger in lysates from the PMA-induced canine protein (Fig. 3) and migrated at the exact same relative molecular mass as did murine Cox-2, as previously reported.22 From these experiments, we concluded that both antibodies bound the induced canine Cox-2 isoform in formalin-fixed tissue sections. Although they produced the same pattern of specific staining in individual tumor samples (not shown), the Transduction Laboratories monoclonal anti-Cox-2 antibody was selected for immunohistochemical characterization of all canine intestinal tumors because of its greater specificity over background in stained sections; all figures represent results from use of this monoclonal antibody in immunohistochemical and western blot procedures.
Kidney; dog. Cox-2 expression in epithelium of macula densa. Avidin–biotin–peroxidase complex method, Mayer's hematoxylin counterstain. Bar = 25 μm.
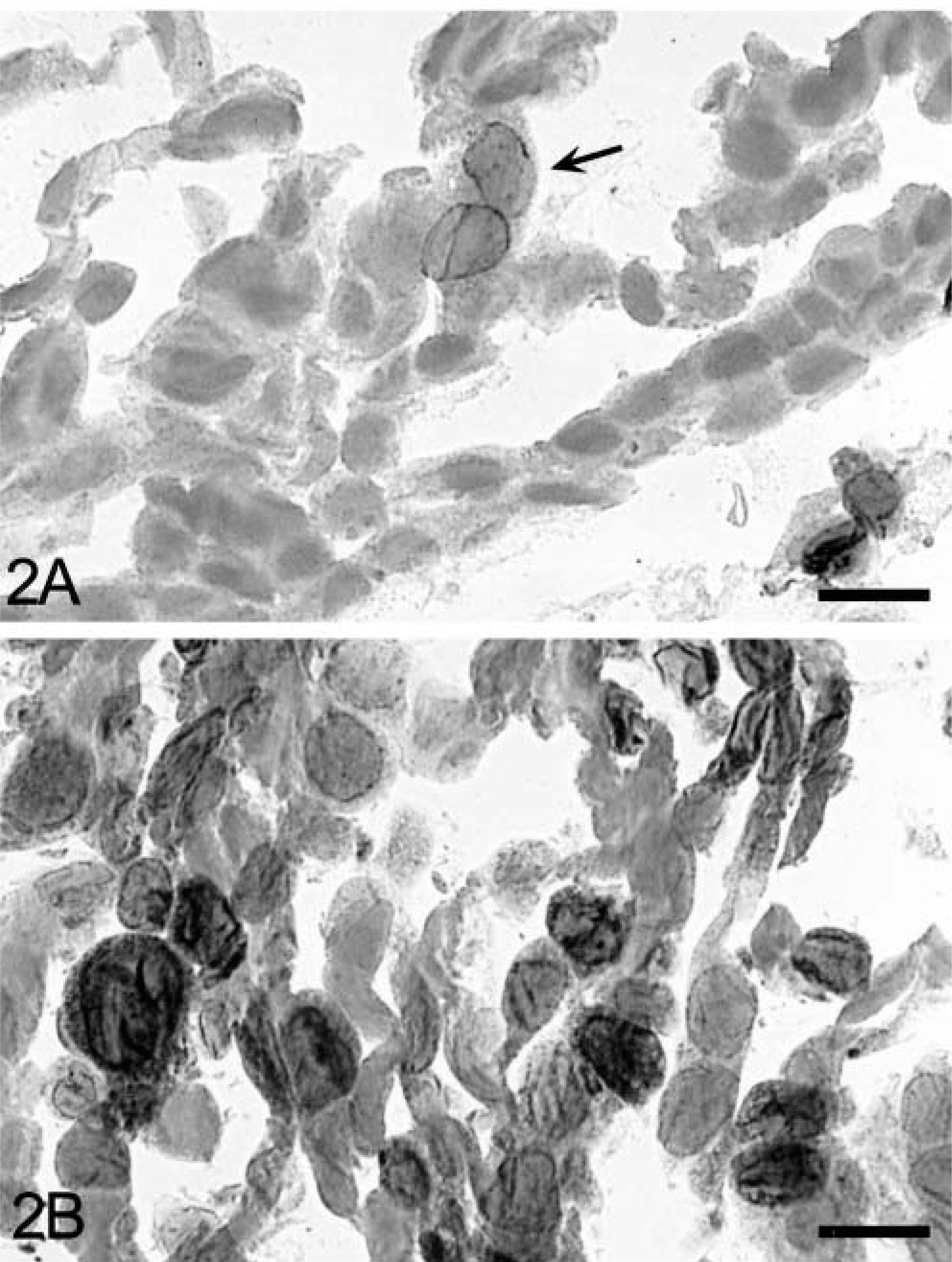
Epithelial cell line (MDCK); dog.

Western blot. Epithelial cell line (MDCK); dog. Cell lysates separated by polyacrylamide gel electrophoresis and stained to detect Cox-2 (70 kd) in untreated (Ctrl) and PMA-stimulated cultures of MDCK cells. Immunoperoxidase chemiluminescence.
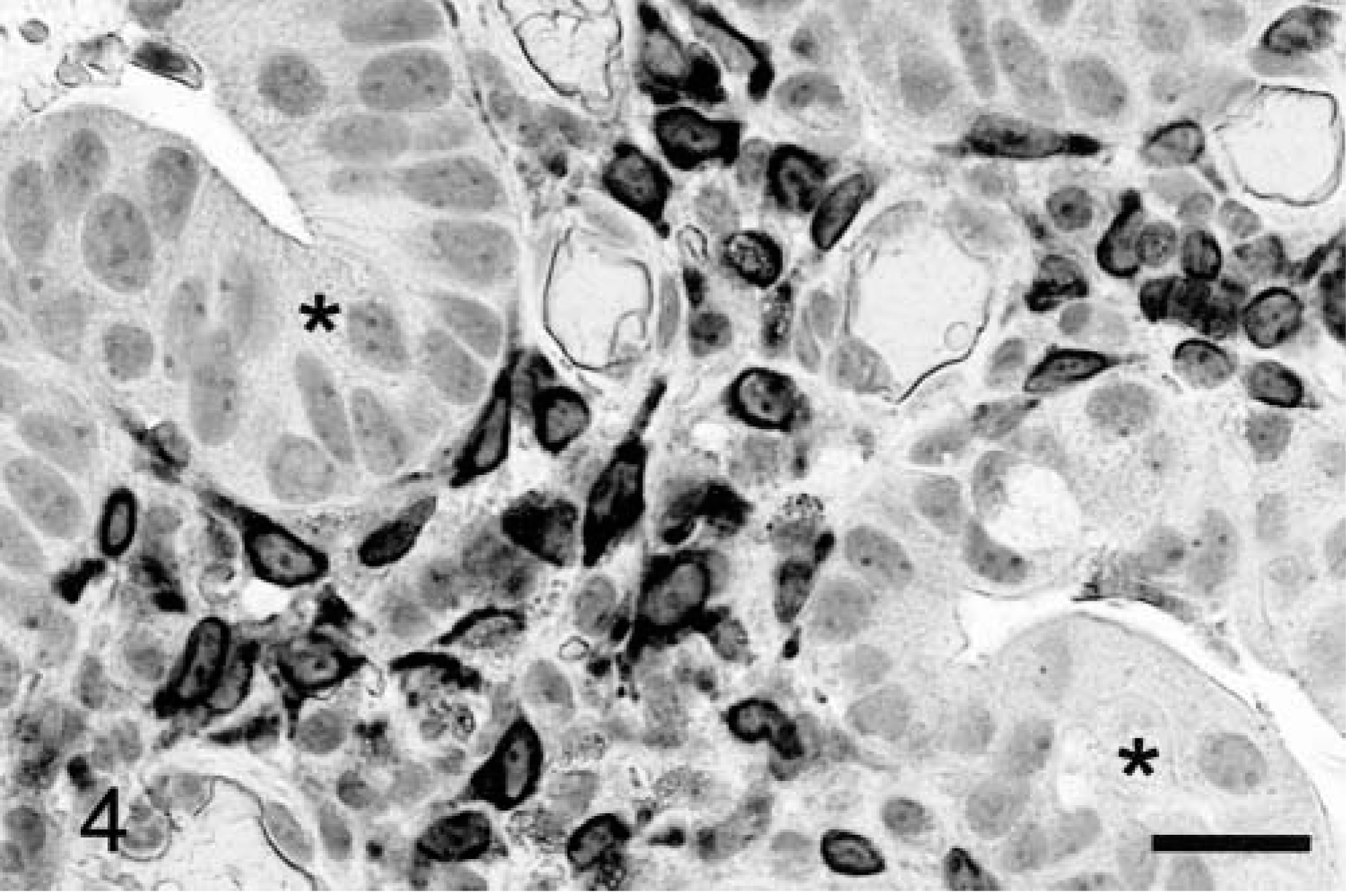
Normal colonic and small intestinal mucosa did not stain for Cox-2. One animal with colitis had variable staining of superficial mucosal fibroblasts and macrophages throughout the large intestine. In these cell types staining was most intense immediately around the nuclear envelope. This pattern was also seen with Cox-2 staining in neoplastic and nonneoplastic cells of the intestinal tumors, except where particularly strong staining extended well into the perinuclear cytoplasm (Fig. 4), as in some PMA-treated MDCK cells. Immunoreactive Cox-2 protein was most common in colorectal polyps and small intestinal carcinomas (Table 1). A greater proportion of adenomas than carcinomas were positive for Cox-2, but this difference was not significant (P = 0.32). Ten of 13 Cox-2–positive large intestinal polyps were from the distal colon or colorectal junction. Three dogs had coincident adenomas and carcinomas (one colorectal, two small intestinal), but none of these were positive for Cox-2.
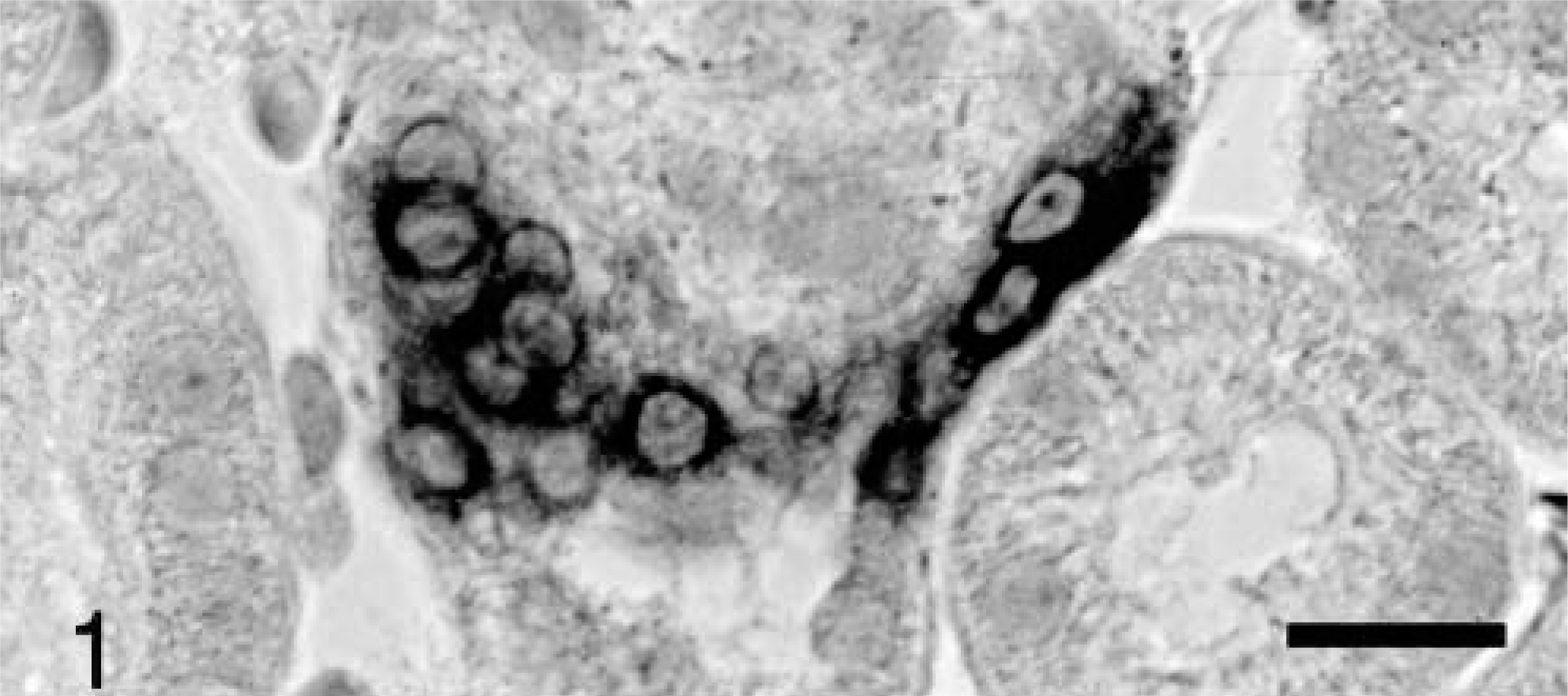
Colorectal adenoma; dog. Strong expression of Cox-2 in stromal cells between neoplastic epithelial cells (asterisks) forming glandular structures. Avidin–biotin–peroxidase complex method, Mayer's hematoxylin counterstain. Bar = 25 μm.
Cox-2 and β-catenin expression in canine intestinal tumors by location and diagnosis.

One colorectal carcinoma overexpressed Cox-2 and β-catenin.
No significant Cox-2 expression.
Cox-2 expression in colorectal adenomas was essentially limited to nonneoplastic stromal cells, which morphologically included fibroblasts/myofibroblasts and, less often, macrophages (Figs. 4–6). Vasculature was not labeled. Although less dysplastic tubulovillous polyps were most often positive for Cox-2, there was no significant difference in Cox-2 expression based on tumor morphology (not shown). Rare Cox-2–positive neoplastic epithelial cells were scattered in two colonic polyps, which also showed significant stromal staining (score, ≥2); none of the other adenomas had any evidence of Cox-2 expression in the epithelium. Stromal staining in colorectal polyps was most common in the superficial luminal aspects of the tumor mass (N = 13; mean score = 2.7 ± 0.13), but Cox-2 was also expressed by deeper stromal cells in 10 of these tumors (2.2 ± 0.23) (Fig. 6). Superficial staining of stromal cells was sometimes associated with and was more intense in areas of erosion where the overlying epithelium had been lost (Fig. 6). Nondegenerate neutrophils in adherent inflammatory exudates covering eroded tumor surfaces also stained for Cox-2 in some instances.
Colorectal adenoma; dog.
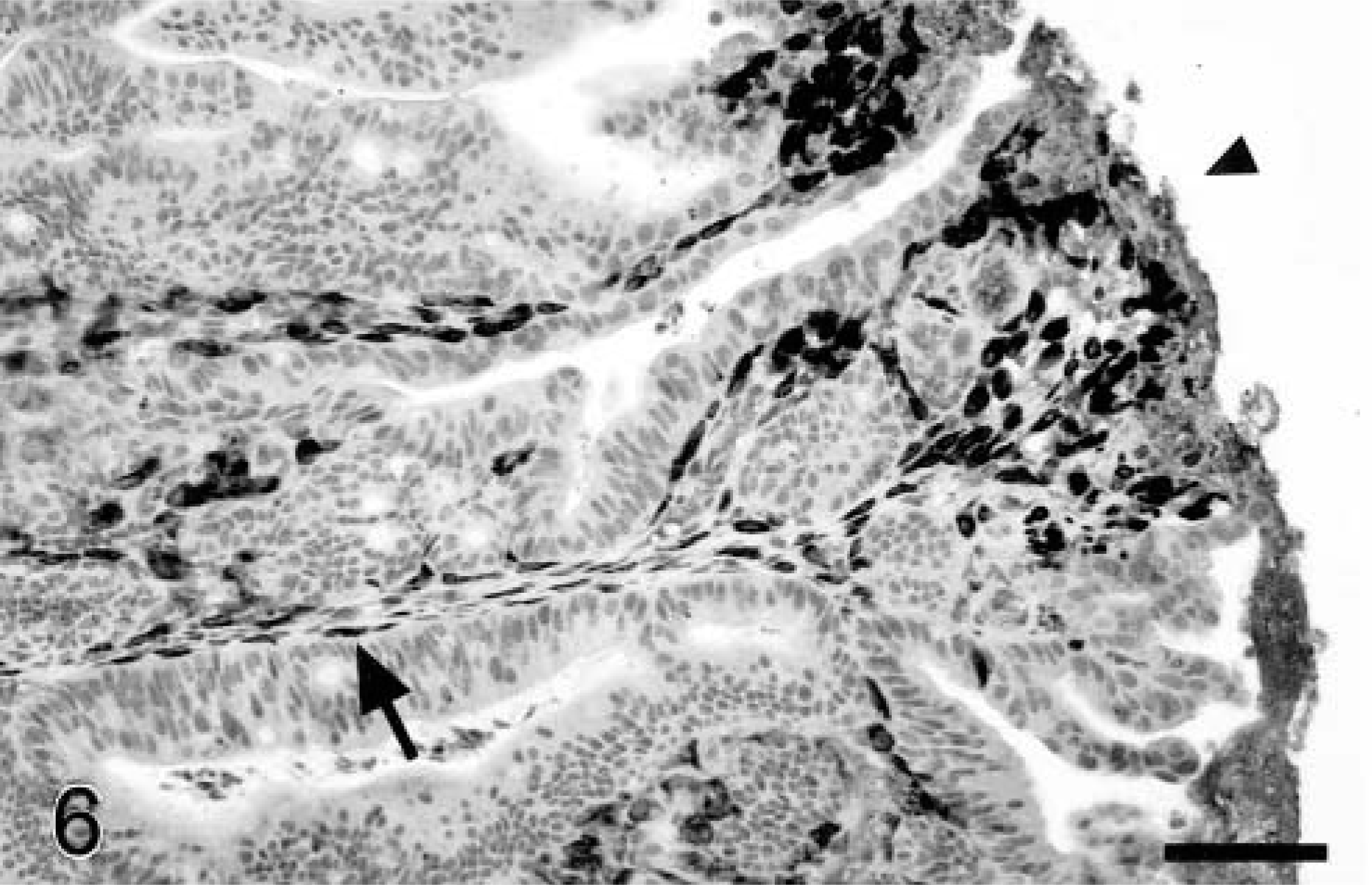
Colorectal adenoma; dog. Cox-2 expression is most prominent immediately below the tumor surface (arrowhead), but Cox-2–positive spindle-shaped cells are also scattered deeper in the tumor mass (arrow), closely apposed to and more distant from neoplastic epithelial cells. Avidin–biotin–peroxidase complex method, Mayer's hematoxylin counterstain. Bar = 100 μm.
In both the normal mucosa and neoplastic tissues, there was often a layer of spindle-shaped α-SMA–positive cells bordering crypts and tubular–glandular structures, consistent with the myofibroblast cellular phenotype.36 α-SMA was also expressed by some mesenchymal cells between and subjacent to tubular–glandular structures in the adenomas (Fig. 7) and by smooth muscle cells of arteriolar walls throughout all sections. The number and distribution of α-SMA–positive stromal cells differed among and within tumors, and these cells were usually more plump (viz hypertrophied) than those in the normal mucosa. In direct comparisons of serial sections of colorectal polyps stained for α-SMA and Cox-2, only 40–60% of Cox-2–positive cells were myofibroblasts. Conversely, not all myofibroblasts in these tumors were Cox-2 positive, and none of the α-SMA–positive cells in the adjacent normal mucosa expressed detectable levels of Cox-2. Cox-2–positive cells that did not express α-SMA were plump, angular to elongate, or stellate in outline (Fig. 4) and could represent either fibroblasts or macrophages based on morphology alone, although lack of phagocytic activity and elongate shape would be more typical of fibroblasts. Eight of the 13 Cox-2–positive colorectal polyps and four of those that did not contain significant Cox-2 reactivity in the tumor mass had some Cox-2 staining in subjacent cells, including smooth muscle cells of the muscularis mucosa, submucosal fibroblasts/myofibroblasts, and scattered mononuclear inflammatory cells (macrophages and/or mast cells).
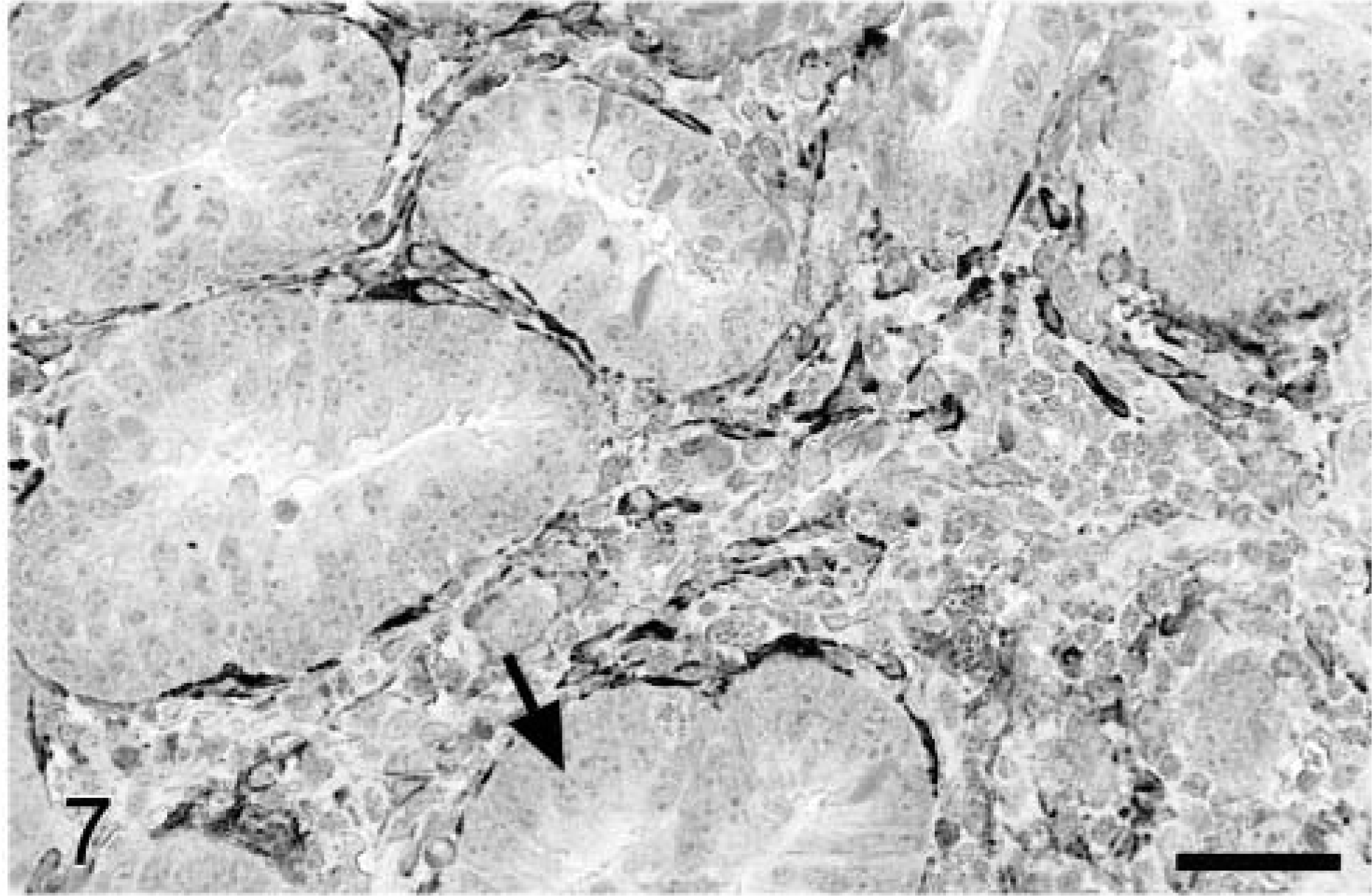
Colorectal adenoma; dog. Stromal spindle-shaped cells immediately adjacent to neoplastic epithelium (arrow) often express α-SMA, indicating a myofibroblast phenotype. Avidin–biotin–peroxidase complex method, Mayer's hematoxylin counterstain. Bar = 50 μm.
In contrast to adenomas, Cox-2 staining was significantly more common in the neoplastic epithelium of carcinomas (mean score of 2.2 ± 0.29 versus 0.15 ± 0.10; P = 0.0013) and less common in tumor stromal cells (mean score = 1.3 ± 0.41), although the latter was not significantly different from adenomas (P = 0.08). Five carcinomas had a Cox-2 score of ≥3 for epithelial staining (all small intestinal tumors), and four others had a score between 1 and 2 (i.e., <5% of cells; two small intestine and two large intestine tumors). Although most Cox-2–positive carcinomas contained a relatively small percentage of stained epithelial cells (5–10%), >40% of tumor cells strongly expressed the enzyme in one tumor (Fig. 8). Staining was heterogenous within individual tumors, but positive neoplastic cells were generally localized to relatively discrete regions rather than evenly dispersed throughout the mass. Although fibroblasts in the immediate vicinity of individual neoplastic cell infiltrates (including those in regions of desmoplasia) usually did not stain for Cox-2, the nuclear envelope and adjacent cytoplasm of smooth muscle cells frequently did stain in areas where the muscularis mucosa or intestinal tunics had been disrupted by infiltrating cancer cells (Fig. 9). Perinuclear Cox-2 staining was also variably present in widely scattered round cells within the connective tissues adjacent to vessels distant from the tumors. In serial sections stained with hematoxylin and eosin or toluidine blue, these cells corresponded to perivascular mast cells (not shown). Cox-2–positive round cells were also identified within several other tumors but could not be clearly distinguished as mast cells, macrophages, or other mononuclear inflammatory cells in these locations.
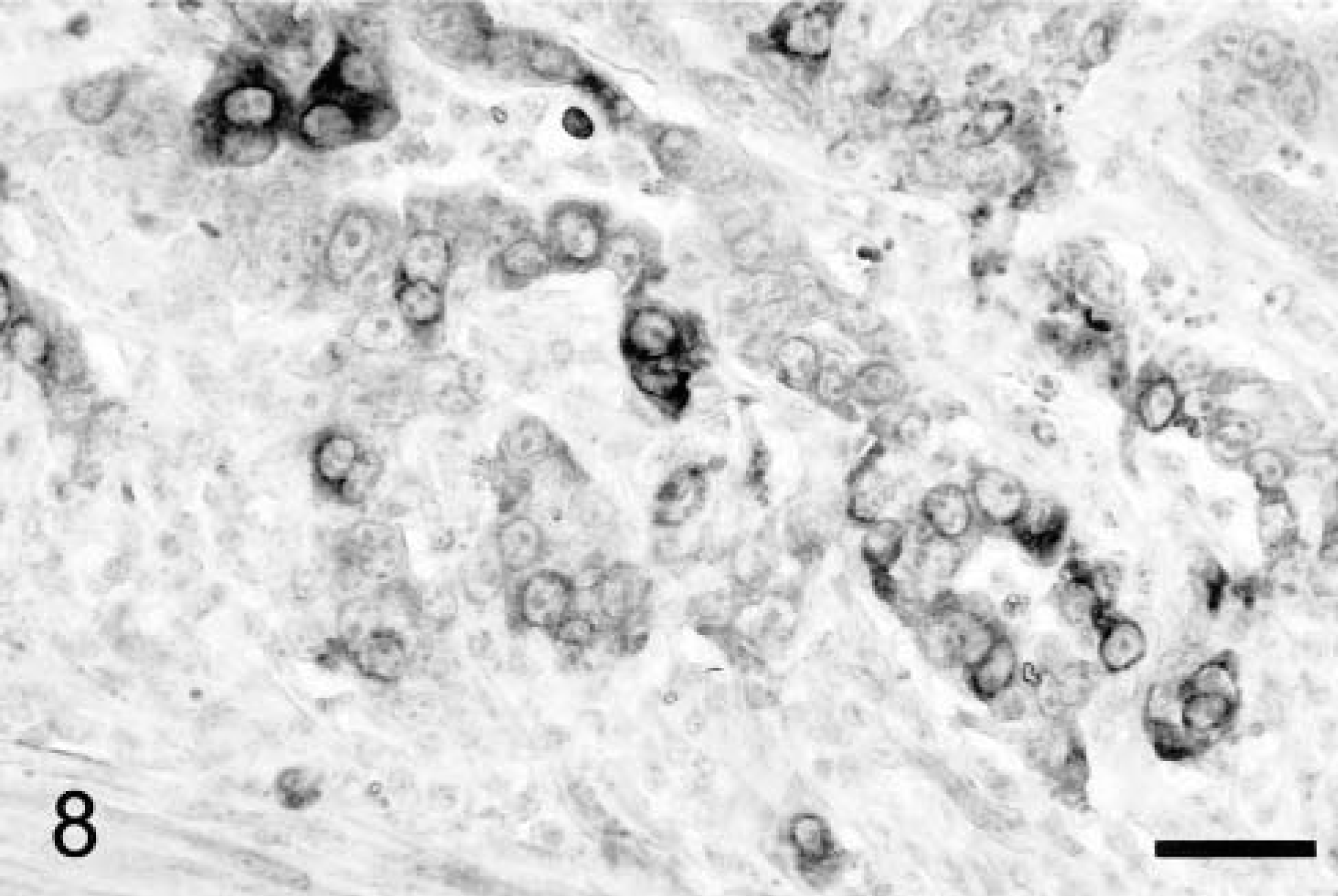
Colorectal adenocarcinoma; dog. Neoplastic epithelial cells in malignancies often express Cox-2, as in this dog, whereas Cox-2 is restricted to the nonneoplastic stroma in adenomas. Avidin–biotin–peroxidase complex method, Mayer's hematoxylin counterstain. Bar = 25 μm.
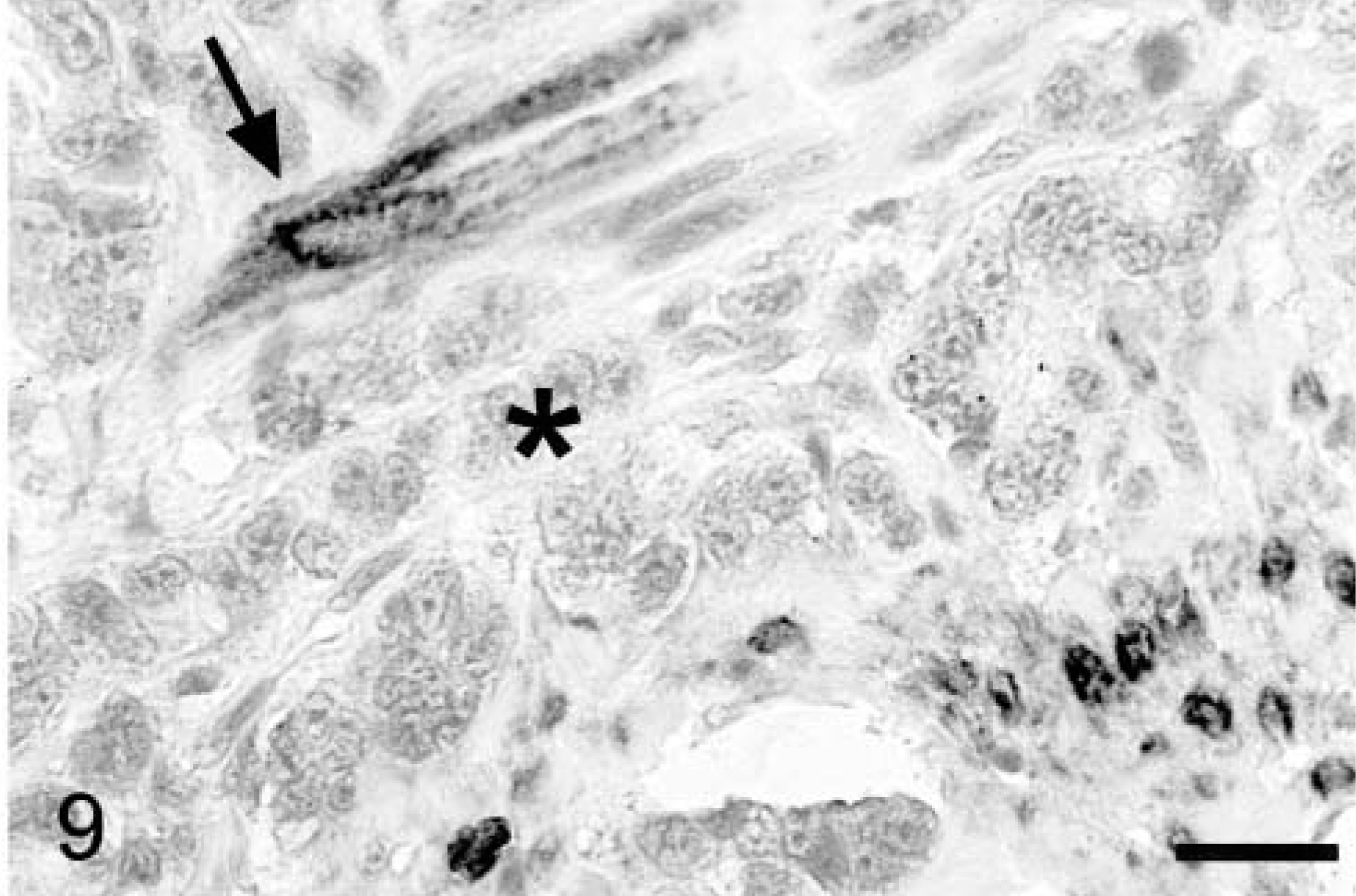
Colorectal adenocarcinoma; dog. Smooth muscle cells adjacent to infiltrating malignant epithelium (asterisk) express Cox-2 (arrow, cells at lower right). Avidin–biotin–peroxidase complex method, Mayer's hematoxylin counterstain. Bar = 25 μm.
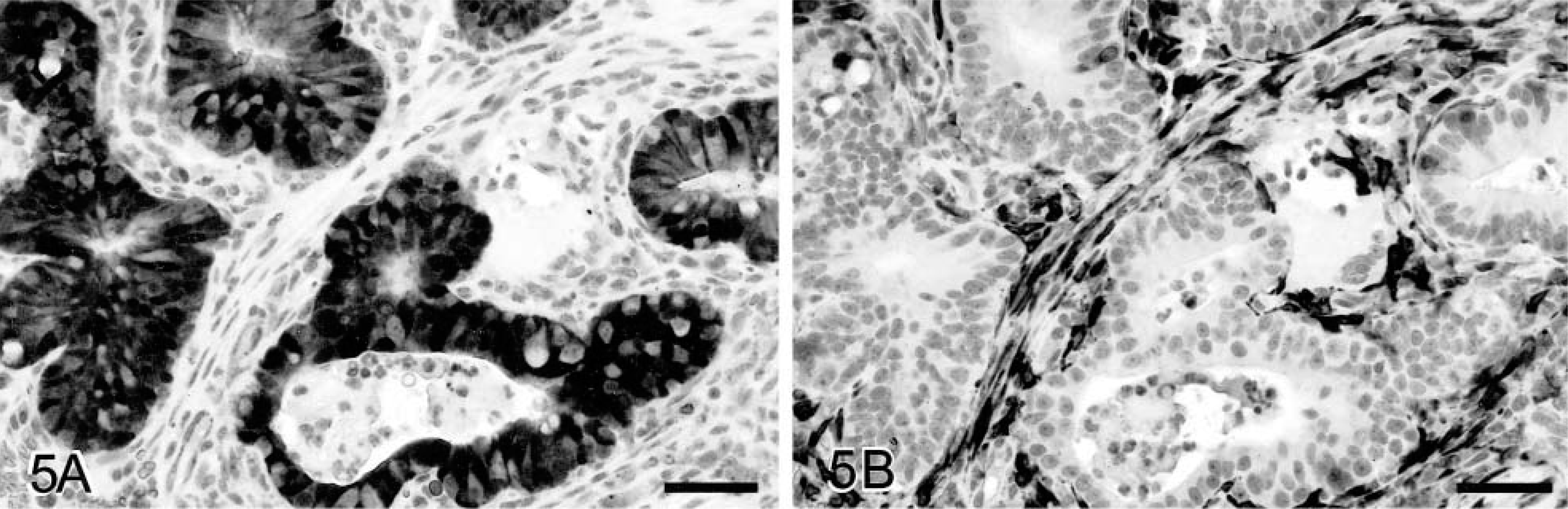
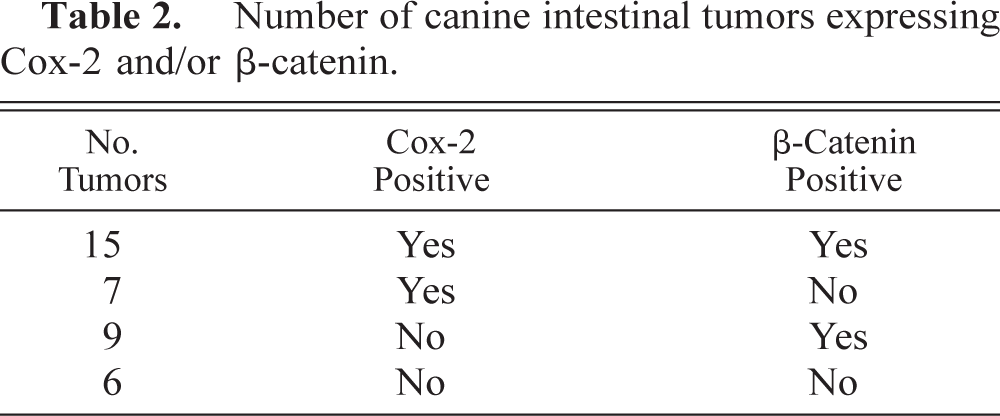
Six colorectal polyps included in this study had not been previously examined for β-catenin expression.28 All six of the adenomas had the same pattern of cytoplasmic and nuclear β-catenin reactivity as previously reported (Fig. 5).28 There was no significant association between increased cytoplasmic/nuclear β-catenin staining and upregulation of Cox-2 in individual tumors (Tables 1, 2; P = 0.73). Staining for β-catenin was confined to lateral cell membranes (viz normal distribution) in the six carcinomas with the greatest Cox-2 expression in the neoplastic epithelium, and Cox-2 was restricted to the nonneoplastic stroma in 13 of the 20 colorectal polyps where β-catenin was upregulated in neoplastic cells (Fig. 5).
Number of canine intestinal tumors expressing Cox-2 and/or β-catenin.
Discussion
Considerable epidemiologic and laboratory evidence indicates that Cox-2 significantly contributes to early stages of human colorectal tumorigenesis.52 Increased expression of Cox-2 and the beneficial effects of NSAIDs in familial adenomatous polyposis (FAP) patients suggests this may be particularly true when mutations in the adenomatous polyposis coli (APC) gene, which result in upregulation of β-catenin, initiate tumorigenesis.10,11,23,37,44 NSAIDs also cause regression of intestinal tumors in genetic mouse models of FAP, where expression of Cox-2 has specifically been shown to effect tumor development.4,19,33 Colorectal tumorigenesis in domestic dogs is commonly associated with loss of β-catenin regulation.28 Neoplastic epithelium in all canine colorectal polyps examined to date (N = 20) overexpress cytoplasmic and nuclear β-catenin, with loss of full-length immunoreactive Apc protein in at least some of these tumors.28 This finding implicates early somatic mutation of the canine Apc gene (or perhaps β-catenin) during colorectal tumorigenesis in this species, but more definitive analysis is required for confirmation. The overexpression of β-catenin in naturally occurring intestinal neoplasms of humans and dogs is consistent with the inherent importance of this pathway during colorectal tumorigenesis. The results of the current study show that Cox-2 is also commonly upregulated in canine intestinal tumors, providing a second molecular link between humans and dogs in the development of this disease.
There has been some inconsistency in the reported localization of Cox-2 expression in intestinal tumors from humans and rodent models.3,13,14,18,32,38–41,44,51 We have found that Cox-2 is expressed almost exclusively by mesenchymal cells in intestinal tumors from Min Apc/+ mice (unpublished), consistent with other reports indicating that Cox-2 is upregulated in the nonneoplastic stromal compartment of adenomas from both humans and mouse models.3,14,18,32,40,41 In contrast to expression in adenomatous polyps, COX-2 expression in human colorectal tumors shifts from the stroma to the neoplastic epithelium following progression to malignancy.32 A similar sequence is evident in dogs, with upregulation of Cox-2 in the stroma of adenomas and additional expression in the epithelium of carcinomas. Cox-2 in canine colorectal polyps was essentially restricted to myofibroblasts, fibroblasts, and perhaps macrophages (based on immunohistochemistry and cell morphology). Not all myofibroblasts in the adenomas were Cox-2 positive, and none of the myofibroblasts stained for Cox-2 in the normal mucosa, where they were smaller and presumed to be relatively quiescent. Although stromal Cox-2 expression was not as prominent in the carcinomas, there was additional immunostaining in smooth muscle cells of the muscularis mucosa and outer tunics, suggesting upregulation where the cancer had invaded the underlying tissues. Therefore, a variety of nonneoplastic mesenchymal cells may express Cox-2 in association with benign and malignant canine intestinal neoplasms, where the bioactive lipid products likely contribute to tumorigenesis as established in rodent models.33,53 The pathophysiologic and clinical significance of Cox-2 expression in the malignant epithelial cells remains to be demonstrated.
Numerous factors may activate myofibroblasts, smooth muscle cells, and macrophages to induce expression of Cox-2,6,36,52 but the mechanisms responsible for upregulation of this protein in the stroma or neoplastic epithelium of intestinal tumors is still unclear. Although loss of wild-type Apc may be responsible for increased Cox-2 in neoplastic intestinal epithelium, perhaps through modulation of β-catenin–mediated transcription,17,29 less direct mechanisms must be responsible for Cox-2 upregulation in the nonneoplastic stroma. Numerous factors can induce Cox-2 including IL-1β, tumor necrosis factor, transforming growth factor, bile acids, endotoxin, and perhaps other components of bacterial cell walls.6,27,36,49,52,55 Paracrine interactions between neoplastic epithelium and adjacent stromal cells could also result in upregulation of Cox-2, and physical characteristics of the neoplastic lesions causing exposure of stromal tissues to intestinal contents could have the same effect.36,39,52 Damage and loss of gastrointestinal epithelium causes upregulation of Cox-2 in (myo)fibroblasts, forming the ulcer granulation tissue bed where it is believed to promote healing.36 In addition, epithelial tight junctions in intestinal neoplasms may not be as strong as those in the normal mucosa,46 resulting in leakage of luminal bile acids, bacteria, and/or bacterial products into the underlying tissue spaces with direct or indirect (i.e., via IL-1β) induction of Cox-2. Cox-2 expression in the canine colorectal polyps was frequently most intense just below the luminal surface of the tumor and was associated in particular with erosion of the overlying epithelium. A similar pattern of Cox-2 expression has been reported in intestinal tumors from Min Apc/+ mice and humans.3,40 Therefore, superficial stromal induction of Cox-2 may result from damage to or the porous nature of overlying neoplastic epithelium, whereas expression deeper in the mass may be due to paracrine stimulation by factors derived from the neoplastic epithelium.
Regardless of the mechanisms involved, upregulation of Cox-2 clearly promotes intestinal tumor growth in vivo. 33 The bioactive eicosanoids produced by Cox-2 are presumably responsible for this protumorigenic effect, which likely includes angiogenesis, cell proliferation, and inhibition of apoptosis.43,47,53 We have shown that Cox-2 is commonly upregulated in canine intestinal tumors, which occur spontaneously in the pet population. Widespread dysregulation of β-catenin in these tumors is consistent with somatic mutation of the canine Apc gene or perhaps β-catenin. The molecular mechanisms responsible for intestinal carcinogenesis in humans and mouse models are therefore conserved in dogs and include upregulation of Cox-2.21,35,37 The beneficial effects of NSAIDs and dietary n-3 fatty acids on colorectal tumorigenesis in laboratory rodents presumably is related, at least in part, to Cox-2 activity, and similar responses could therefore be expected in domestic dogs.
