Abstract
Malignant nerve-sheath tumor with divergent differentiation including epithelial components was diagnosed in an 8-year-old Labrador retriever. The myelographic, morphologic, and immunohistochemical findings confirmed the diagnosis. The tumor was located in the peripheral nerve roots at the first and second lumbar vertebrae. The dog survived for 161 days after resection of the tumor.
Malignant nerve-sheath tumor in human beings is an uncommon sarcoma, characterized by Schwannian and fibroblastic differentiation.3,4,6 A small percentage of these tumors (10%), both benign and malignant, are characterized by divergent differentiation.1,2,4,8,10–12 Rhabdomyosarcoma, osteosarcoma, chondrosarcoma, angiosarcoma, and melanoma are common mesenchymal differentiations, myosarcoma being more common than the others.5,11,12
Glandular and epithelial differentiation are extremely rare. In one human study, less than 1% of the tumors in 120 patients with malignant nerve sheath tumor had epithelial differentiation.5 Both mesenchymal and epithelial types of differentiation can be benign or malignant.1,2,4,10–12 Nerve sheath-tumor with glandular differentiation was first described by Garre in 1892.7 A combination of different types of sarcoma alone or in combination with epithelial components has also been reported.5,10,11 Divergent differentiation of malignant nerve-sheath tumor has been associated with a poor prognosis.11,12
Although cranial nerve-sheath tumors in dogs is uncommon, a search of the English veterinary literature for the past 20 years revealed no report of nerve-sheath tumor with divergent differentiation except for one report of two dogs with malignant Schwannoma with melanotic differentiation.9 Two other cases of malignant nerve-sheath tumors arising from multiple cranial nerve roots have been described. These tumors had typical features of fibrosarcoma in most areas, and one tumor had thick bands of collagen stroma.13 Here we describe a malignant nerve-sheath tumor with divergent differentiation including glandular elements in a dog.
An 8-year-old spayed female Labrador retriever was referred with a 1-month history of hind-limb ataxia and a 3-week history of occasional fecal incontinence. Physical examination revealed bilaterally symmetrical atrophy of the muscles of both pelvic limbs. The right patellar reflex was hyperreflexic, and the left patellar reflex was normoreflexic. Bilateral hind-limb ataxia was observed, more severe on the left. Bilaterally symmetric conscious proprioceptive deficits were also seen. Differential diagnoses included neoplasia involving the thoracolumbar spinal cord and adjacent structures, intervertebral disc disease, fibrocartilaginous embolic myelopathy, and other ischemic injury to the thoracolumbar spinal cord.
Hematologic testing revealed mild to moderate lymphopenia. No abnormalities were seen on thoracic radiographs. Plain radiography of the thoracolumbar and lumbar spine revealed bridging spondylosis of the lumbosacral vertebrae. Myelography revealed a large, intramedullary filling defect with axial and abaxial displacement of contrast media at the first and second lumbar vertebrae (Fig. 1).
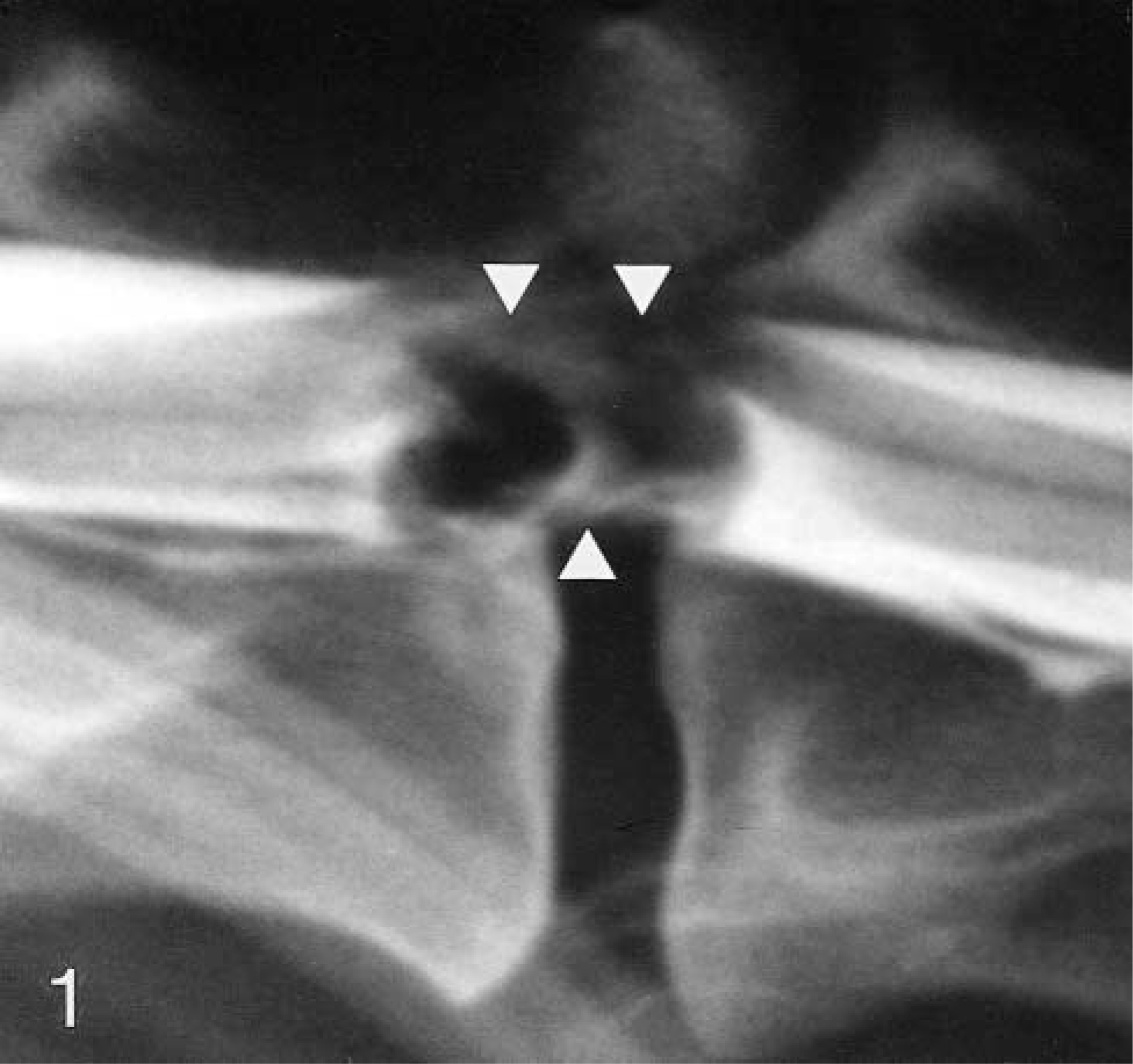
Myelogram revealing a large, intradural, extramedullary filling defect (arrows) with axial-abaxial displacement of the contrast medium at the level of the first and second lumbar vertebrae.
A modified left hemilaminectomy was performed on the first and second lumbar vertebrae, revealing bulging of the dura and spinal cord. A fibrous mass approximately 1 cm in diameter was seen within the dura involving the left lateral and ventral aspects of the spinal cord and the nerve roots. The mass was dissected from the ventral aspect of the spinal cord and excised. Affected dura were resected with the mass. The specimen, consisting of four pieces of grayish tissue, was placed in 10% buffered formalin and submitted for histopathologic examination.
The patient had no motor function in her pelvic limbs for 2 days but regained motor function on the third postoperative day. She was discharged from the hospital on the sixth postoperative day. Hind-limb ataxia was steadily resolving.
The patient did well over the ensuing 112 days, with minimal hind-limb ataxia and no fecal or urinary incontinence. However, 126 days after surgery, her hind-limb ataxia recurred, but only involving the left pelvic limb. No pain was elicited on physical examination. Bilaterally symmetric atrophy of the lumbar epaxial muscles was observed. A tapering dosage of dexamethasone (Azium®, Schering-Plough Animal Health, Kenilworth, NJ) was administered, and the severity of the ataxia diminished. The dog was brought in again 161 days after surgery because of recurrence of bilateral hind-limb ataxia. The patient also had progressive urinary and fecal incontinence and was barely able to rise, even with assistance. The owner elected euthanasia.
Necropsy revealed that fibrous tissue had replaced the surgical site (thoracolumbar area) and the adjoining skeletal muscle. It also obliterated the intervertebra spaces. A 4-cm segment of the caudal thoracic and cranial lumbar regions of the spinal cord was necrotic, containing a grayish, semiliquid material with intact dura. No other important lesions were seen.
The surgical pathology specimens consisted of four pieces of greyish tissue measuring 0.5-1 cm. The tissues were routinely processed and stained with hematoxylin and eosin. Immunocytochemical staining was done by use of a ready-made, supersensitive, multilink immunodetection system (Biogenex, San Ramon, California, USA), following the directions of the manufacturer of the test. Antibodies for S-100 protein, vimentin, neuron specific enolase (NSE), and cytokeratins AE1/AE3 were tested. Diaminobenzidine (DAB) was used as the chromogen and Gill III hematoxylin was used as the counterstain. The following tissues were used as controls: peripheral nerve for S-100 and NSE, skin for AE1/AE3, and intestines for vimentin. For negative control, the slides were stained without the primary antibody.
The neoplasm was well delineated and partially encapsulated, involving the nerve roots (L1–L2) and associated ganglia. The cellularity and pattern of arrangement varied. In the more cellular areas, the spindle cells were arranged in fascicles and criss-crossing bundles resembling fibrosarcoma (Fig. 2). Also seen were areas of storiform and whorling patterns of spindle cells (Fig. 3). These hyperchromatic neoplastic cells had vesiculated nuclei, one or two prominent nucleoli, and eosinophilic cytoplasm. There were two to five mitotic figures per high-power field.
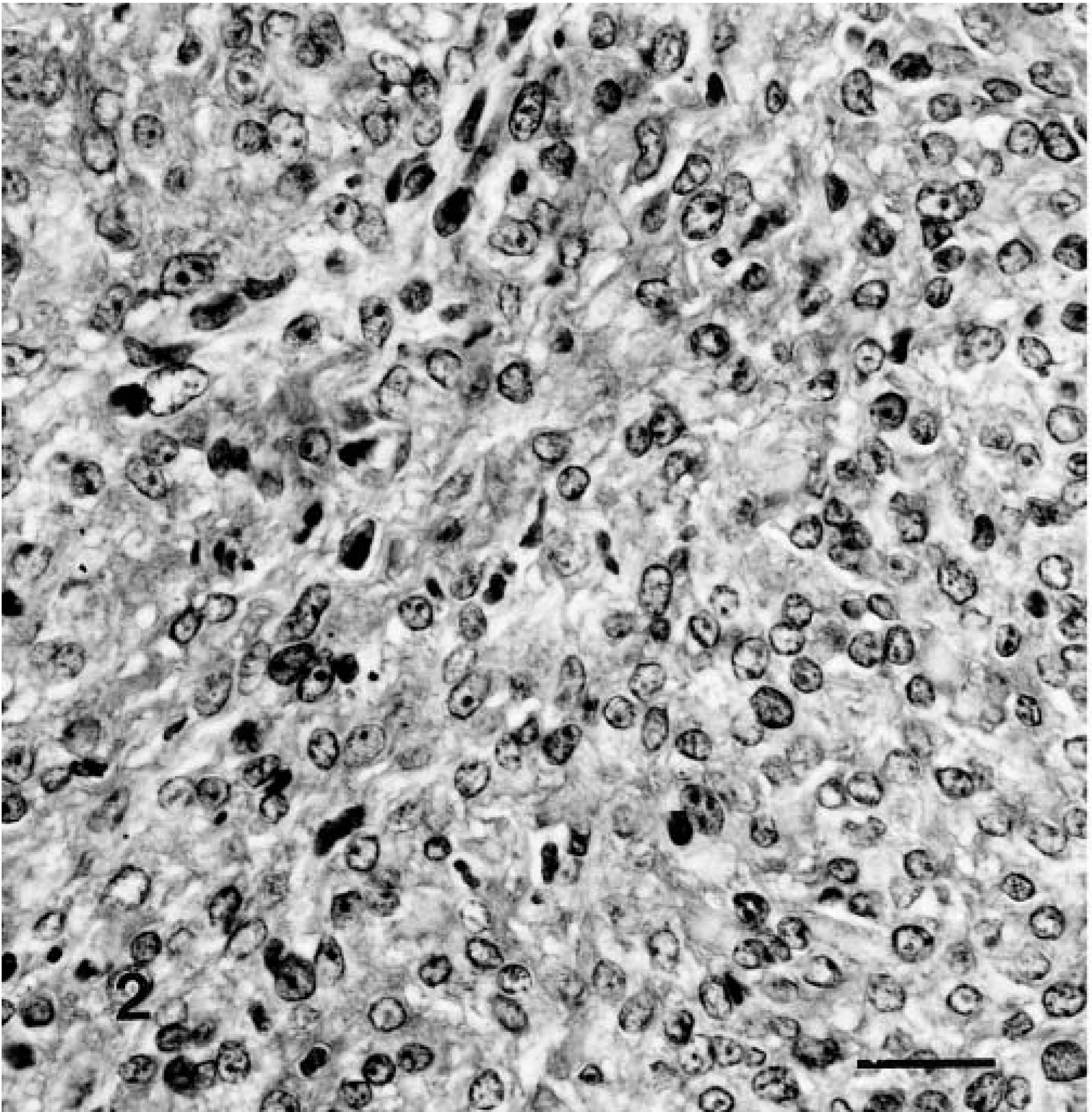
Malignant nerve-sheath tumor showing bundles of anaplastic spindle cells with vesiculated nuclei and prominent nucleoli, a feature similar to fibrosarcoma. H&E; 1 cm = 20 μm.
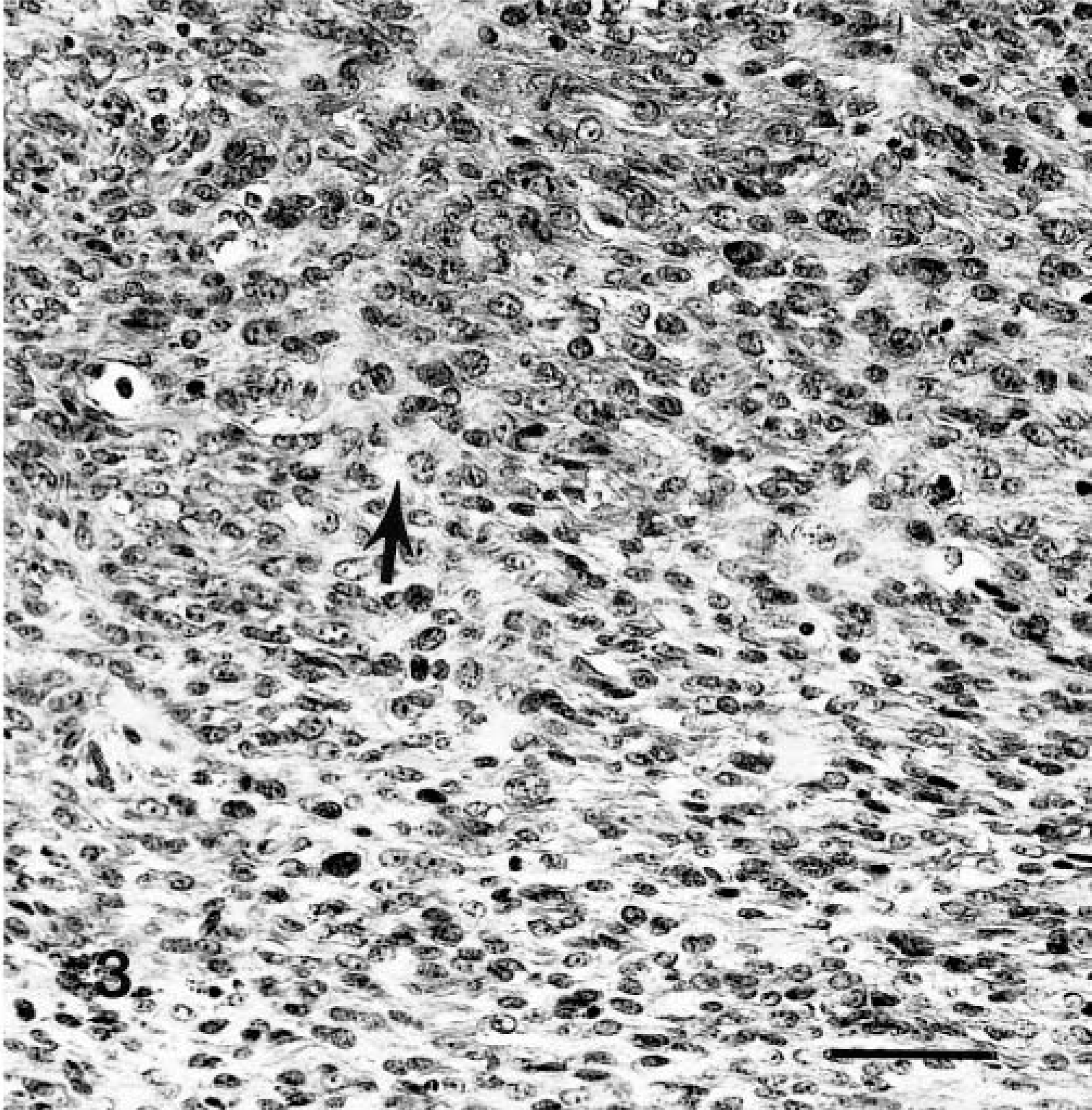
Malignant nerve-sheath tumor showing an area of poorly formed storiform pattern. A few mitotic figures and areas of osteoid formation can also be seen (arrow). H&E; 1 cm = 20 μm.
Most of the tumor had eosinophilic sclerotic stroma containing scattered single or groups of neoplastic cells, which were considered to be malignant osteoid produced by the neoplastic cells. The stroma varied inversely with the cellularity of the neoplastic cells. These cells were round to polygonal with vesiculated nuclei, large nucleoli, and scattered mitotic figures (Fig. 4). The morphology was similar to that of osteosarcoma.
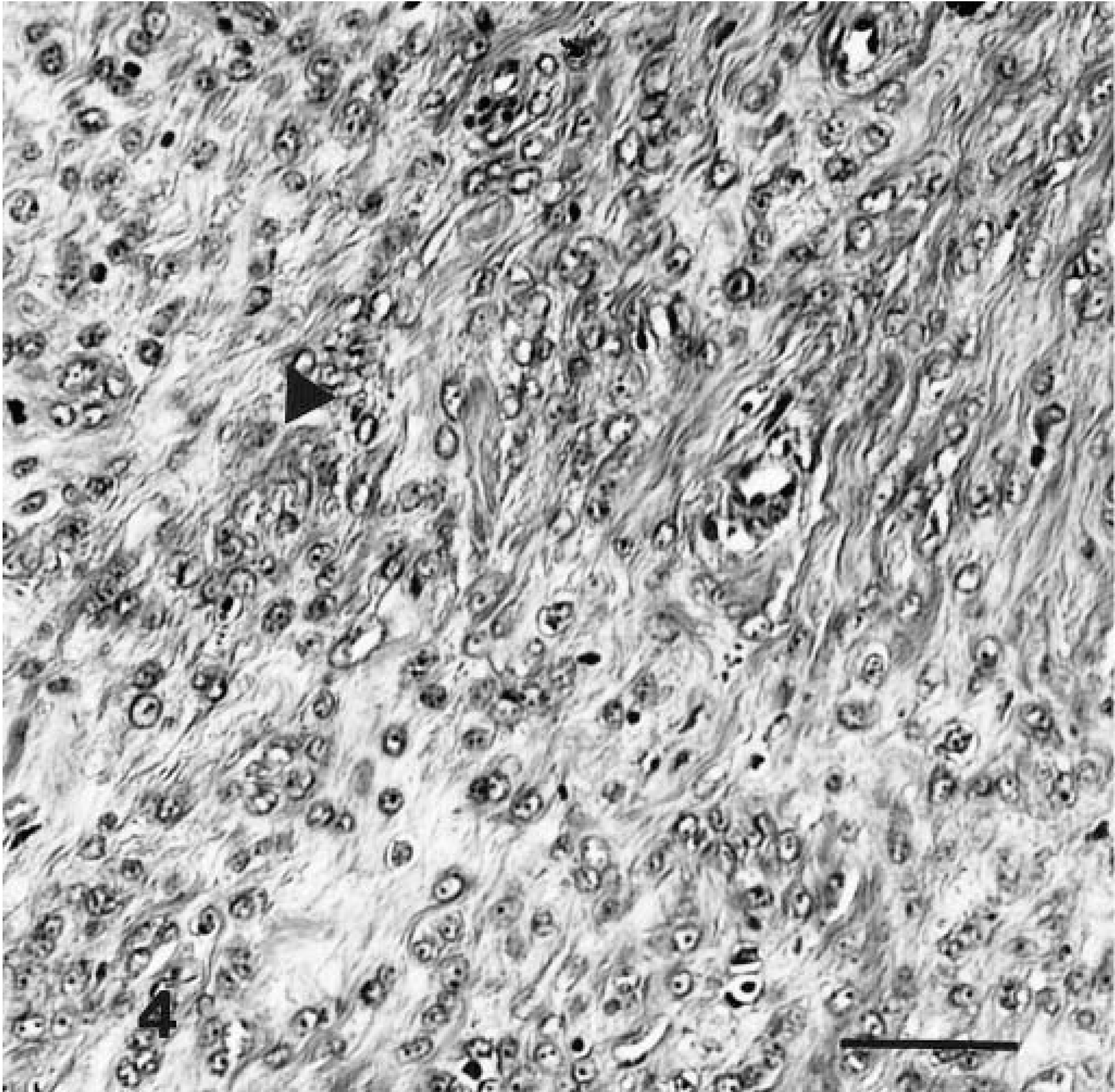
Malignant nerve-sheath tumor showing neoplastic cells forming new bone (osteoid) (arrowhead). H&E; 1 cm = 20 μm.
Scattered epithelial components consisted of cystic structures lined by columnar cells; a few of the cysts contained mucoid material. In other areas, groups of acinar structures were lined by low columnar or cuboidal cells, some with short papillary projections. The epithelial cells in these areas were anaplastic with hyperchromatic nuclei and prominent nucleoli, eosinophilic cytoplasm, and distinct cell borders (Fig. 5). Groups of squamous cells were seen rarely. Areas of the tumor were edematous and necrotic, containing brown hemosiderin pigments. Scattered foci of lymphocytes were seen.
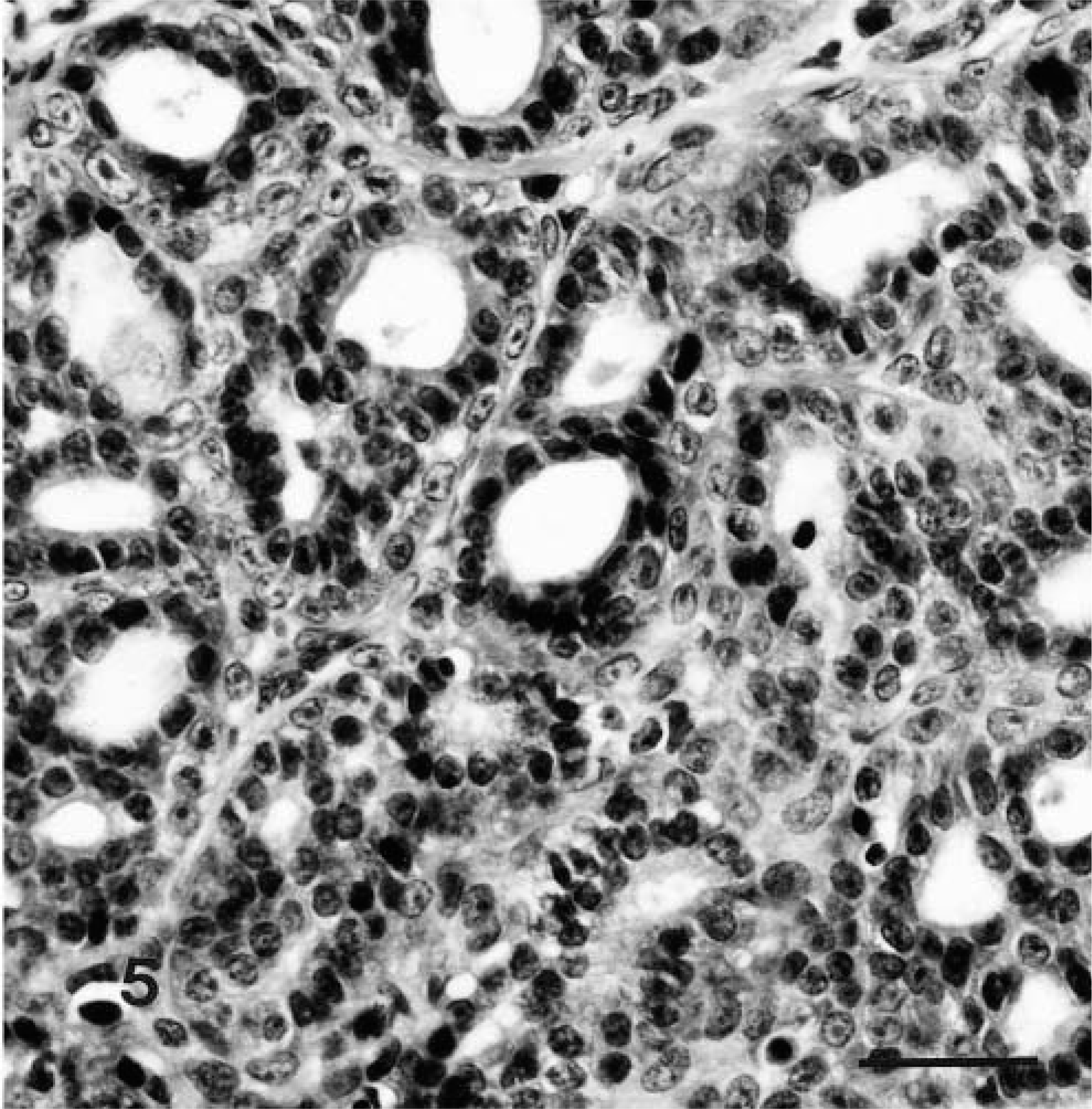
Malignant nerve-sheath tumor; notice a group of acinar structures lined by anaplastic epithelial cells closely associated with sarcomatous cells. H&E; 1 cm = 40 μm.
Histologic examination of necropsy specimens revealed central necrosis of the spinal cord with demyelinization and areas of necrosis and moderate sclerosis. The remnant of the tumor left in the spinal cord had similar mesenchymal features as the biopsy specimens except that the epithelial components were not seen. No other tumors were detected in the dog. Other histologic findings included moderate parathyroid hyperplasia, extramedullary hematopoiesis of the liver and spleen, and persistent thymic tissue with branchial cysts.
The results of immunocytochemical staining of the biopsy specimens and the specimens taken at necropsy were similar. The mesenchymal cells stained diffusely for vimentin, NSE, and S-100 protein (Figs. 6–8); the epithelial cells stained for AE1/AE3.
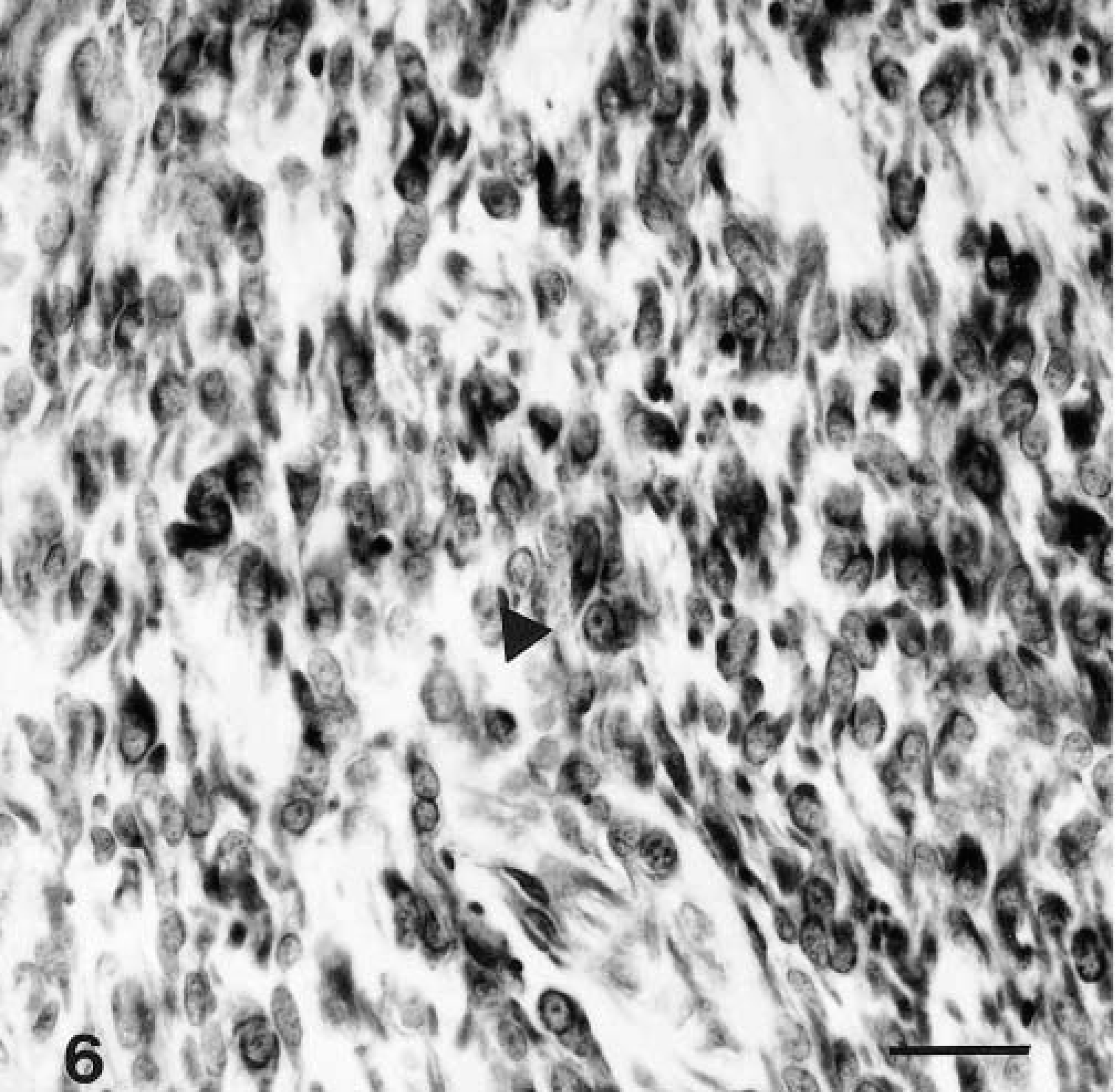
Malignant nerve-sheath tumor immunohistochemically stained for vimentin (arrowhead), showing diffuse intracytoplasmic staining. Supersensitive immunodetection system with DAB as the chromogen and Gill III hematoxylin as counterstain; 1 cm = 40 μm.
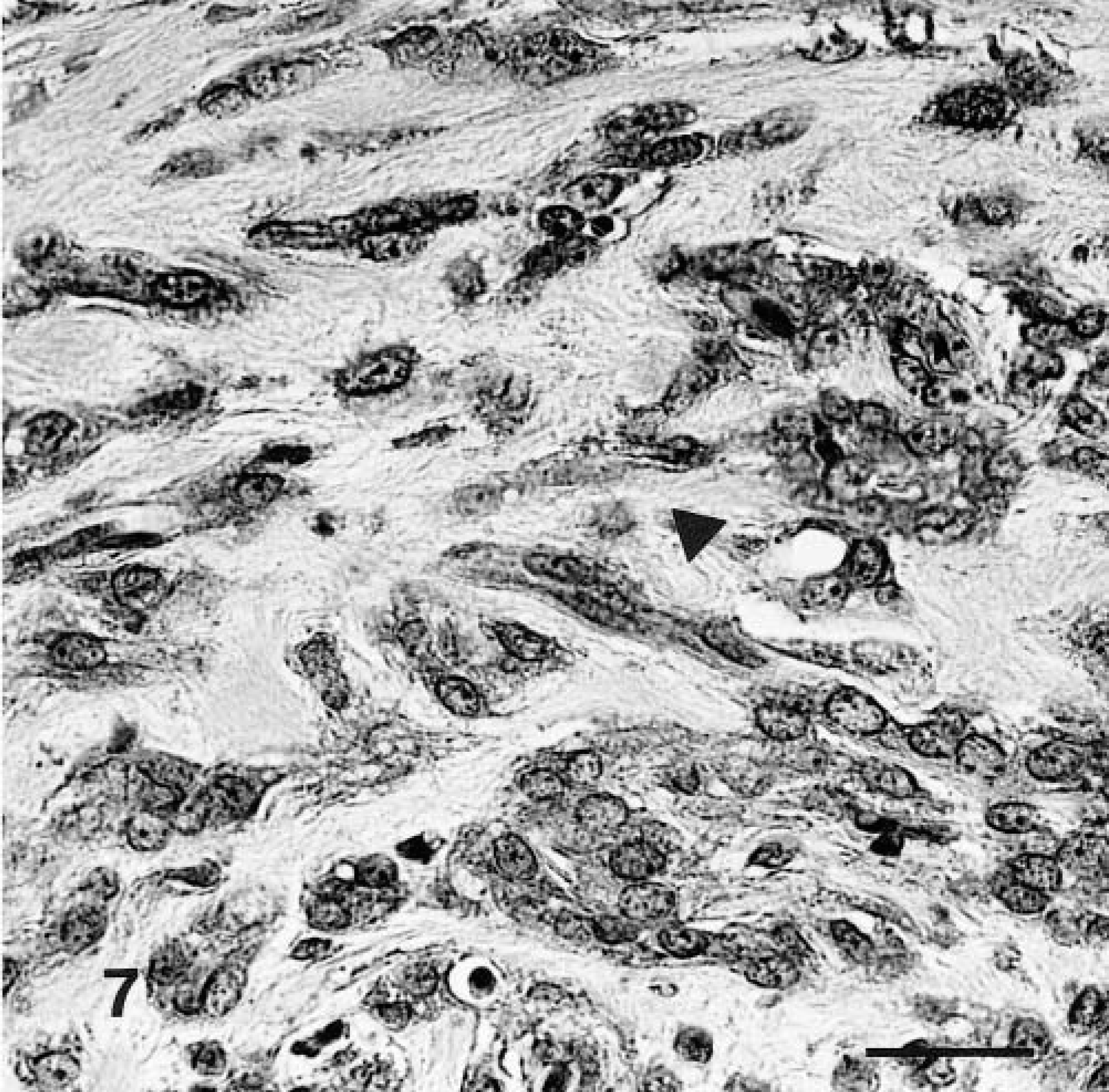
Malignant nerve-sheath tumor immunohistochemically stained for NSE, showing pale intracytoplasmic granules (arrowhead). Supersensitive immunodetection system with DAB as the chromogen and Gill III hematoxylin as counterstain; 1 cm = 40 µm.
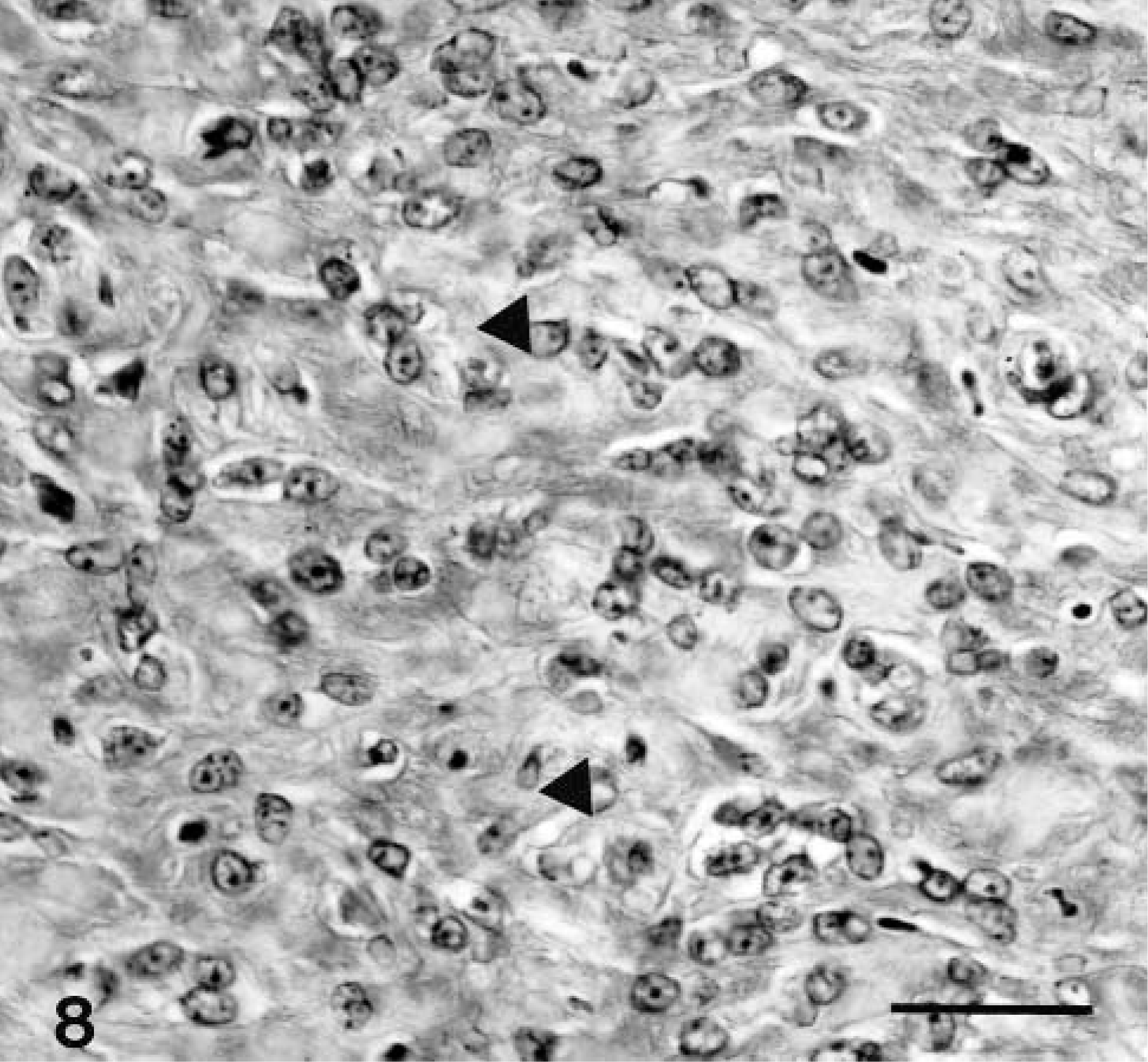
Malignant nerve-sheath tumor immunohistochemically stained for S-100 protein showing diffuse intracytoplasmic staining (arrowheads). An area with osteoid can also be seen. Supersensitive immunodetection system with DAB as the chromogen and Gill III hematoxylin as counterstain; 1 cm = 40 μm.
The final diagnosis was malignant nerve-sheath tumor with divergent differentiation including glandular components.
The malignant nerve-sheath tumor of this report had both mesenchymal and epithelial components. The mesenchymal components were fibrosarcoma and osteosarcoma besides the nerve-sheath tumor. They were immunocytochemically positive for vimentin, suggesting mesenchymal origin, and the positivity for NSE and especially S-100 indicated neuronal origin. The scattered glandular elements were only positive for cytokeratin (AE1/AE3), which is an epithelial marker. Both mesenchymal and areas of the epithelial component were morphologically malignant. These findings are similar to those in human medicine.4,5,10
It is relatively common to find metaplastic changes in nerve-sheath tumors, both benign and malignant. The most common secondary elements are cartilage and bone. A rare feature in human nerve-sheath tumors is the presence of heterologous elements containing mesenchymal and/or epithelial components. The mesenchymal component consists of rhabdomyosarcoma, osteosarcoma, chondrosarcoma, and angiosarcoma. The epithelial component consists of glandular, squamous cell, undifferentiated epithelial and, rarely, neuroendocrine cells.2,4,5,10–12 The mesenchymal components are dominant in these complex tumors.4,5,10 This was true in the case of this report in which the sarcomatous components occupied most of the tumor, with only scattered areas of epithelial cells.
The theory behind this divergent mesenchymal differentiation in malignant nerve-sheath tumor is that the migratory stem cells of the neuronal crest differentiate to neuroectodermal structures, including melanocytes, ganglion cells, Schwann cells, and multipotential mesenchymal cells, known as ectomesenchyme. These tissues give rise to leptomeninges, bone, cartilage, and muscle of the head and neck.3,5,11
Regarding the origin of epithelial cells, studies have suggested that the epithelial components of malignant nerve-sheath tumor are not heterotopic. They potentially arise from Schwann cells or mesoectodermal cells, which are intrinsic to peripheral nerves. These epithelial cells are true glandular cells and are likely to be enteric types. These cells can give rise to neuroendocrine tumors producing regulatory peptides and amines, such as serotonin, somatostatin, and gastrin.2,4,5,10–12
The presence of divergent differentiation in nerve-sheath tumor indicates a poor prognosis.2,4,5,10
