Abstract
Two young adult Macaca fascicularis each had unilateral mydriasis and ptosis. Both animals were euthanatized, monkey No. 1 for progressive neurologic signs and monkey No. 2 because of a positive intradermal tuberculin test. At necropsy, each animal had a single intracranial mass on the ventral surface of the midbrain, surrounding the oculomotor nerve. Histologically, both masses were immunoblastic lymphomas. Immunohistochemical staining revealed the neoplasms to be of B-cell origin. Simian retrovirus (SRV) was isolated from both monkeys, but simian immunodeficiency virus was not found. Both animals lacked antibody to SRV. Both animals had antibodies to Epstein-Barr-like virus (EBV), but EBV antigens were not found by immunohistochemistry. Polymerase chain reaction analysis for integrated EBV DNA was unproductive. One of the animals (monkey No. 2) had a pulmonary infection with Mycobacterium avium, suggesting that immunosuppression was present. These cases represent a unique and previously undescribed type of solitary lymphoma in SRV-infected macaques.
The occurrence of lymphoma in immunodeficient human patients and nonhuman primates has been well documented. An increased frequency of lymphomas is observed in human immunodeficiency virus (HIV) patients and simian immunodeficiency virus (SIV)-infected macaques, suggesting an important pathogenic role of virally induced immunosuppression for the development of lymphoma.2 The majority of these lymphomas are of B-cell origin. Epstein–Barr virus (EBV) is commonly present in tumor cells of human immunodeficiency–associated lymphomas, particularly intracranial and extralymphatic types.3,5,6 The incidence of naturally occurring lymphoma in cynomolgus monkeys (Macaca fascicularis) and rhesus monkeys (Macaca mulatta) is low.5,6 However, the incidence in experimentally SIV-infected animals ranges from 3.8% to 19% in rhesus5,6 and from 31% to 38% in cynomolgus monkeys.3,5 EBV-like lymphocryptovirus sequences are detected in most lymphomas of rhesus macaques, and less often in lymphomas of cynomolgus macaques.5 In addition to SIV, simian retrovirus (SRV) type D is immunosuppressive in macaques, causing the simian acquired immunodeficiency syndrome; however, this syndrome is not generally associated with development of lymphomas.10 We report here two cases of intracranial lymphoma in naturally SRV-infected macaques, in the absence of SIV infection.
Monkey Nos. 1 and 2 were adult male cynomolgus monkeys of Philippine origin, 7 and 6 years old, respectively. The monkeys were indoor-housed in groups of four, and instrumented with indwelling intravenous femoral catheters and subcutaneous vascular access ports as part of a study of social stress and cocaine addiction. All procedures conducted on live animals were approved by the Wake Forest University Institutional Animal Care and Use Committee. Monkey No. 1 presented with unilateral ptosis, mydriasis, and ventrolateral strabismus of the right eye. Based on neurologic and ophthalmic examinations, oculomotor nerve (Cranial nerve [CN] III) paresis was diagnosed, with no evidence of systemic illness. Differential diagnoses included inflammation, neoplasia, trauma, infarction, and hemorrhage, as well as the possibility of cocaine-related oculomotor paresis, as reported in humans.7 Blood chemistry values were within the reference range, but a complete blood count revealed leukopenia due to a lymphopenia. Five weeks later the monkey appeared depressed, and exhibited left hemiparesis and circling to the right. Because of a poor response to treatment and apparent extension of the CN III lesion to involve the right midbrain and cerebral cortex, the animal was euthanatized.
Monkey No. 2 presented with moderate to severe ptosis and swelling of the right eyelid after intradermal administration of mammalian old tuberculin in the same eyelid. The animal was euthanatized because of a suspected infection with Mycobacterium sp.
Serology and virus isolation were done six (monkey No. 1) and eight (monkey No. 2) months before euthanasia (Virus Reference Laboratory, Inc., San Antonio, TX, USA). Both monkeys were seropositive for EBV viral capsid antigen (VCA) and seronegative for antibodies to simian T-lymphotropic virus type 1, SRV (1, 2, and 5), and SIV (strains SIVmac, SIVagm, and SIVsm). Virus isolation done on peripheral lymphocytes showed that both monkeys were infected with SRV type D, but not SIV. No SIV-infected animals were present at the facility, nor was any in vitro work with SIV done in the area.
Complete necropsies were conducted immediately after euthanasia. Grossly, each animal had anisocoria, with the right pupil larger than the left, and ptosis of the right eyelid. Induration of the right eyelid was observed in monkey No. 2. Each animal had a single mass on the ventral surface of the right midbrain that partially encircled the right CN III and right optic nerve (Fig. 1). In monkey No. 1, the mass was round, approximately 1.2 × 1.0 × 1.3 cm, and dark red brown with subarachnoid hemorrhage covering approximately 50% of the ventral surface of the pons and medulla. In monkey No. 2, the mass was oval, 0.5 × 0.5 × 0.7 cm, and mottled red to tan. The sella turcica and pituitary gland were normal.
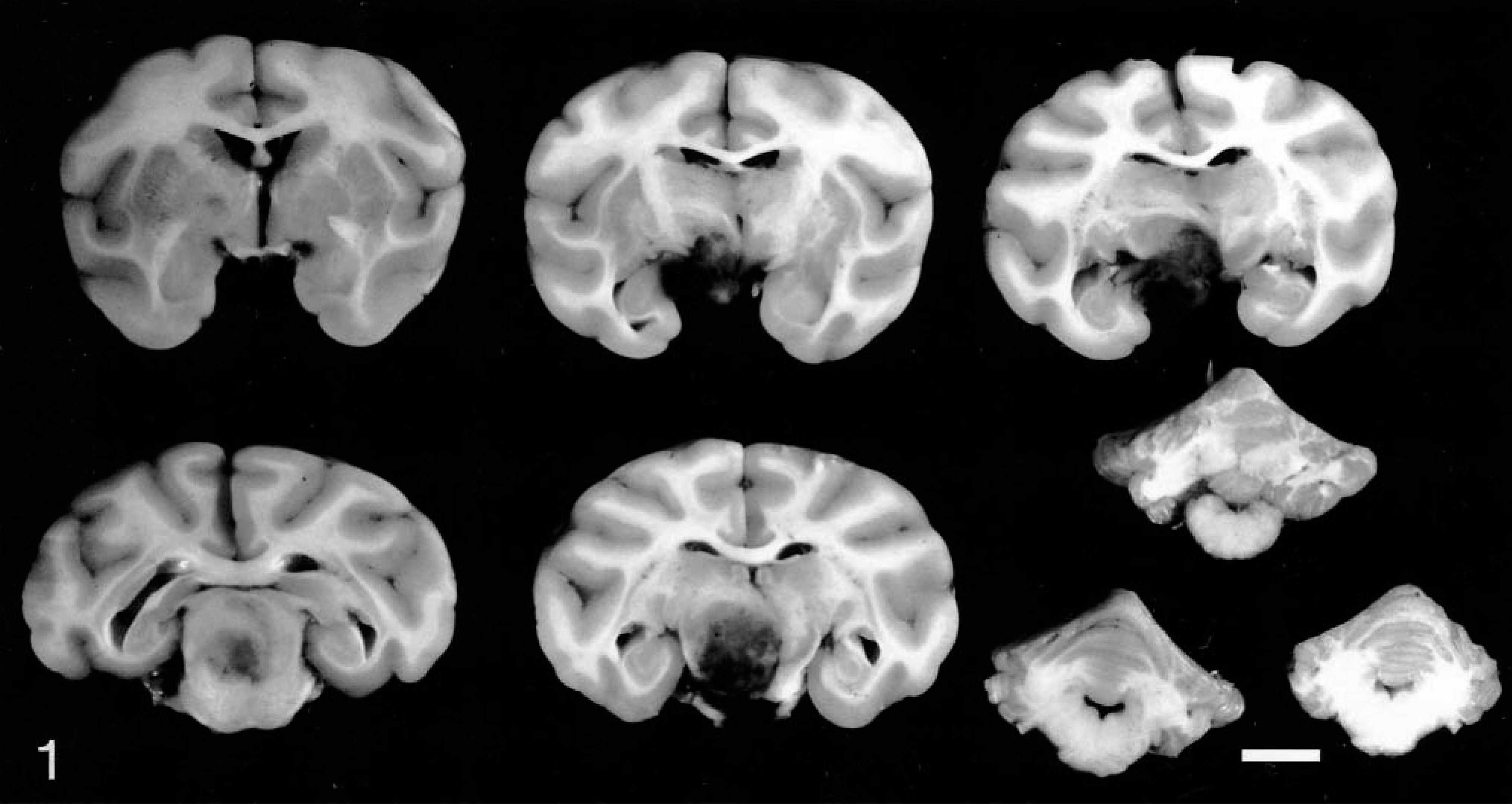
Brain; monkey No. 1. Multiple sections demonstrating the gross appearance of intracranial mass compressing and effacing hypothalamus and surrounding structures. Sections are rostral to caudal order from left to right and top to bottom. Bar = 1 cm.
Other lesions in monkey No. 1 included a cutaneous fistula of the left vas deferens. Both cases had small catheter-associated thrombi in the caudal vena cava. In monkey No. 2, moderate, focal consolidation and hemorrhage of the right lung were seen, as were severe, multifocal, fibrous adhesions between the left and right lung lobes and the diaphragm and thoracic wall. Monkey No. 2 also had severe, chronic, generalized lymph node enlargement that spared the tracheobronchial lymph nodes.
Auramine–rhodamine staining (Truant's method) for acid-fast bacilli was positive on samples of fresh lung and tracheobronchial lymph node from monkey No. 2. Mycobacterium avium was grown from lung and tracheobronchial lymph node. M. avium is a common opportunistic infection of immunosuppressed humans and macaques.1,5
Tissue samples from all major organs were preserved in 10% neutral buffered formalin, embedded in paraffin, and stained with hematoxylin and eosin. Histologically, brain masses from both animals were nonencapsulated, compressing and replacing normal brain tissue (midbrain and hypothalamus in monkey No. 1, and midbrain and pons in monkey No. 2). Multifocal areas of necrosis and hemorrhage involved 25% of the mass from monkey No. 1 and 5% of the mass from monkey No. 2. In both animals, neoplastic cells infiltrated adjacent tissue at the margin of the mass, within perivascular spaces. Both neoplasms were composed of sheets of pleomorphic lymphocytoid cells, without stroma. The predominant cells were round to oval noncleaved cells, 10–30 μm in diameter, with moderate amounts of slightly eosinophilic cytoplasm (Fig. 2). The nuclei were round to oval, containing clumped and marginated chromatin, with single central nucleoli, resembling those of immunoblasts. Two to three mitotic figures were observed per 40× field in both cases. The next most common cells were round to oval and smaller, with scant eosinophilic cytoplasm and smaller, more basophilic nucleoli. These cells were more abundant in monkey No. 2. Peritumoral lymphocytic meningitis and choroid plexitis also were present in monkey No. 2.
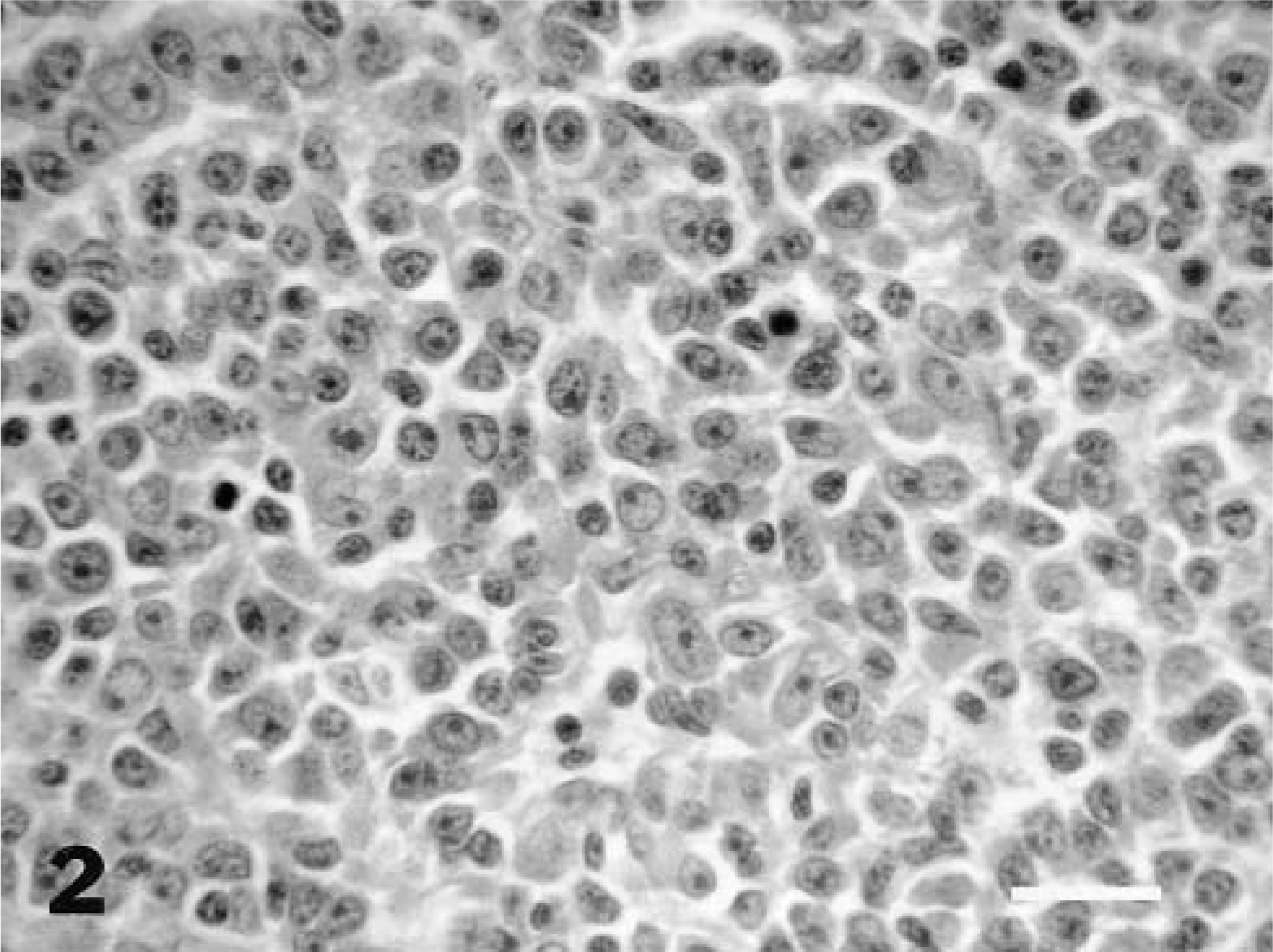
Intracranial mass; monkey case No. 2. Histologic appearance of the lymphoma demonstrates immunoblastic morphology. HE. Bar = 50 μm.
Paraffin-embedded tumor tissues from both monkeys were sectioned and stained for neuron-specific enolase (NSE; monoclonal antibody BBS/NC/VI-H14, Dako Corporation, Carpinteria, CA, USA), vimentin (Biogenex Laboratories, San Ramon, CA, USA), and glial fibrillary acidic protein (GFAP; monoclonal antibody 6F2, Dako Corporation). Staining also was done for B-cell markers BLA.36 (monoclonal antibody A27-42, Dako Corporation) and CD-20 (monoclonal antibody L26, Dako Corporation), and the T-cell marker CD-3 (rabbit polyclonal antibody A452, Dako Corporation). Human, canine, and macaque tissues were used as positive controls. Neoplastic cells were moderately to strongly immunopositive for B-cell markers L26/CD-20 (approximately 40–60% of neoplastic cells in monkey No. 1 and 5–10% of cells in monkey No. 2) and BLA.26 (approximately 10% of cells in both cases). In monkey No. 1, scattered cells at the margin of the neoplasm were strongly immunostained for the T-cell marker CD-3 (approximately 10% of the total). No CD-3 staining was seen in monkey No. 2. The neural marker (NSE), glial marker (GFAP), and mesenchymal-cell marker (vimentin) stained normal tissues appropriately but did not stain neoplastic cells. Immunohistochemical staining was also done with antibodies to viral proteins BZLF-1 (mouse monoclonal antibody BZ.1/ZEBRA, Dako Corporation), and latent membrane protein-1 (LMP-1; mouse monoclonal antibody, CS 1-4, Dako Corporation) to detect antigens related to EBV; no neoplastic cells were stained.
Ultrastructurally, neoplastic tissue of monkey No. 1 had predominantly round cells lacking intercellular junction or extracellular matrix. Cells contained scant cytoplasm with abundant rough endoplasmic reticulum, with rounded nuclei containing marginal chromatin, and a single central nucleolus of variable size. Occasionally, low numbers of round, nonenveloped bodies approximately 100 nm in diameter were found within the nuclei, consisting of an electron-dense rounded structure surrounded by a thin pale rim. These particles were similar in size to herpesvirus nucleocapsids but lacked the distinct icosahedral shape, and also lacked the rod-shaped morphology and smaller size of lentiviruses. A third possibility, that the viruses were type D retroviral particles, similarly could not be made specifically. Therefore, the intranuclear bodies were considered to be either artifacts or degenerate or incomplete viral particles.
Other histologic findings in monkey No. 1 included mild to moderate diffuse lymphoid hyperplasia of kidney, retina, bone marrow, and T-cell regions of axillary and cervical lymph nodes. Moderate diffuse lymphoid depletion of the spleen, para-aortic lymph node, and B-cell regions of axillary and cervical lymph nodes also was found. Other findings in monkey No. 2 included minimal to severe, focal to diffuse lymphoid follicular hyperplasia of axillary, cervical, lumbar, inguinal, and ileocecal lymph nodes, and spleen, kidney, seminal vesicles, bulbourethral gland, skeletal muscle, and urinary bladder; and focal follicular hyperplasia of the thymus. The histologic presentation of lymphoid hyperplasia and depletion in multiple organs in these monkeys are features of SRV infection in cynomolgus monkeys.4 Small aggregates of macrophages and multinucleated giant cells also were found in the subcapsular sinus of a tracheobronchial lymph node in monkey No. 2; Ziehl–Nielsen staining did not reveal acid-fast organisms. Histologic findings in the lung consisted of focal pulmonary fibrosis and arterial intimal fibrosis, suggestive of previous thromboembolism (presumably related to intravenous catheter use).
In summary, we report two cases of unusual intracranial lymphomas involving the brain in cynomolgus monkeys. Based on viral isolation, both animals were naturally infected with SRV, but not SIV. The incidence of SRV infection in the colony from which the animals were derived was approximately 30% (unpublished data), but SIV was not present in the population. Lymphoma has been previously reported in one naturally infected cynomolgus monkey with SRV; the central nervous system (CNS) was not involved.4 Lymphomas are not considered a typical feature of SRV infection in macaques; rather, mesenchymal neoplasms such as retroperitoneal fibromatosis have been reported in association with SRV infection.9 In experimentally SIV-infected rhesus and cynomolgus monkeys, visceral and cutaneous lymphomas are the most common lymphomas. Lymphomas of the CNS have been reported less frequently.3,5,6 The common sites of lymphoma in acquired immunodeficiency syndrome (AIDS) patients are CNS, gastrointestinal tract, and bone marrow.1,5,6 As in HIV-associated lymphoma, the sites of involvement in SIV-infected macaques are extranodal, as was seen in the monkeys presented herein.3,5,6,8 In HIV-infected humans and SIV-infected macaques, lymphoma occurs late in the progression of the disease.1,5,6
Histologically, these lymphomas were classified as immunoblastic.6 Immunoblastic lymphoma with plasmacytic differentiation makes up 60% of AIDS-related primary brain lymphomas, and 100% of these cases are of the B-cell phenotype.1 Phenotypically, the cells present in our cases are B-cell, consistent with published reports in AIDS patients and SIV-infected monkeys. The B-cell markers L26 (CD-20) and BLA.36 were expressed, as shown in previous monkey cases.5,6,8 The T-cells at the margin of the neoplasm in monkey No. 1 were interpreted as infiltrating nonneoplastic lymphocytes. Similar findings have been described in experimentally SIV-infected rhesus monkeys and in human cases.6,8 Interaction of HIV and EBV has been proposed as one of the factors in the development of lymphoma in AIDS patients and SIV-infected macaques.2,5 EBV was not detected in our cases, based on immunohistochemistry with LMP-1 and BZ.1 (ZEBRA). Although LMP-1 is found in human neoplasms, LMP-1 was not detected in rhesus monkey lymphomas; the basis of this difference is not known.6 This may be the result of differences in the sequence or protein expression between EBV and rhesus lymphocryptovirus, the rhesus equivalent of EBV. Similar differences may also exist for the equivalent virus in cynomolgus monkeys, called herpesvirus macaca fascicularis 1. The virus was presumably present, because both animals reported herein were seropositive for EBV VCA. The presence of viral particles ultrastructurally is intriguing but does not indicate causality. Epstein–Barr–related sequences have been detected in 30–100% cases of lymphomas in humans (depending on tumor type), 89% of lymphomas in rhesus monkeys, and 25% of lymphomas in cynomolgus monkeys.5 The absence of EBV protein or DNA in lymphoma cells in the monkeys in this report may indicate a different pathogenesis of lymphoma development. Comparable to human cases, SIV-infected tumor-bearing macaques have been reported to have up to 100% seropositivity to EBV, but this is confounded by the high background incidence of EBV seropositivity.6
This is the first report of solitary intracranial lymphomas in macaques. Recent discussion of this disease entity (Primate Pathology Workshop, 2000) indicates that primary intracranial lymphoma may be a more common entity in immunosuppressed macaques than previously supposed.
Footnotes
Acknowledgements
We are grateful to the Molecular Diagnostics Laboratory of the Department of Pathology at Wake Forest University School of Medicine for assistance with immunohistochemical staining.
