Abstract
Ovine pulmonary adenocarcinoma is caused by jaagsiekte sheep retrovirus. To gain insight into the histogenesis and viral pathogenesis of this neoplasm, the tumor cell phenotypes and differentiation state were correlated with the distribution of jaagsiekte sheep retrovirus capsid protein in neoplastic and normal cells of the lung in nine naturally occurring and 12 experimentally induced cases of ovine pulmonary adenocarcinoma. Overall, 82% of tumor cells had ultrastructural features consistent with alveolar type II cells, 7% of tumor cells had features of Clara cells, and 11% of tumor cells were insufficiently differentiated to classify. The proportion of the neoplastic cell phenotypes varied within tumors, and no tumor consisted of a morphologically uniform cell population. To further characterize the neoplastic cell population, sections of tumors were immunostained with antibodies to surfactant protein A, surfactant protein C, and Clara cell 10-kd protein. Overall, surfactant proteins A and C were expressed in 70% and 80% of tumor cells, respectively, whereas Clara cell 10-kd protein was expressed in 17% of tumor cells. Jaagsiekte sheep retrovirus capsid protein was detected in 71% of tumor cells and in macrophages (5/21 tumors examined) and in nonneoplastic alveolar and bronchiolar cells (6/14 tumors). Expression of this viral protein in neoplastic cells, classified morphologically and by immunophenotyping primarily as of the alveolar type II lineage, implies an important role for specific virus–cell interactions in the pathogenesis of ovine pulmonary adenocarcinoma.
Keywords
Ovine pulmonary adenocarcinoma (OPA) is a transmissible bronchioloalveolar carcinoma (BAC) of sheep with morphologic features similar to those of human BAC.4,11,25,27 In OPA and human BAC, adult individuals present with multiple peripherally located neoplastic pulmonary nodules that may represent metastases of a monoclonal tumor.2,3,13 Based on electron microscopy (EM), the alveolar type II (ATII) cell and the nonciliated bronchiolar epithelial (Clara) cell are the predominant neoplastic cell phenotypes in naturally occurring OPA (OPA-N)12,21,32 and in experimentally induced OPA (OPA-E).10,33 Analysis of surfactant protein gene expression by in situ hybridization and immunohistochemistry (IHC) indicates that ATII cells also predominate in pulmonary adenomas of mice.19,28 Although there is not uniform agreement or absolute consistency between IHC and EM studies, most reports indicate that nonmucinous BAC of humans consist of Clara or ATII cells.5,6,35 The etiology of human BAC, which is only weakly associated with smoking, is unknown, and its prevalence is increasing, especially among women.1,2
The etiologic agent of OPA is jaagsiekte sheep retrovirus (JSRV), a chimeric B/D type retrovirus.37 OPA can be induced by intratracheal inoculation of neonatal lambs using infectious proviral clones of JSRV.9,26 In a limited study, JSRV capsid protein (JSRV CA) was detected by IHC in neoplastic epithelial cells of lung tumors.24 A protein that cross-reacts immunologically with JSRV CA also has been detected in human lung tumors (30% of BAC and 26% of adenocarcinomas), warranting continued investigation of retroviruses in human BAC.8
The mechanisms of retroviral oncogenesis in OPA are incompletely understood but may include expression of oncogenic retroviral structural proteins in tumor cells17,30 or proviral insertional mutagenesis.9 The receptor for JSRV entry into human cell lines has recently been identified as HYAL2.29 In humans, the ovine counterpart of the HYAL2 gene is likely to be expressed on multiple cell types of the lung, thus probably not conferring specificity to the target cells for viral infection.7 Alternatively, because the JSRV long terminal repeat (LTR) is preferentially active in differentiated epithelial cells of the lung,23 LTR-regulated transcription in specific cells may be responsible for their proliferation and eventual transformation.
This study was undertaken to determine the association of JSRV CA with the tumor cell phenotype and differentiation state in OPA to better understand the interaction of JSRV with pulmonary epithelial cells in the neoplastic process. Because the nature and timing of various virus–host interactions may differ between OPA-N and OPA-E, cases of both were studied and compared using histomorphology, ultrastructural analysis, and IHC for ATII and Clara cell phenotypic markers and JSRV CA expression as an index of viral infection. Immunophenotyping with surfactant proteins A and C (SP-A, SP-C) and Clara cell 10-kd protein (CC10) together with ultrastructural findings indicated that ATII cells were the predominant neoplastic cell type in OPA and that the tumor cells exhibited variable degrees of differentiation. We observed JSRV CA in all tumors but not in all neoplastic cells, and the presence of this viral protein was not limited to neoplastic cells. Based on this information, we propose that there are specific interactions between JSRV and its target cell that limit carcinogenesis to cells of the ATII/Clara cell lineage.
Materials and Methods
Animal source and tissue specimens
Nine sheep with OPA-N and 12 sheep with OPA-E were used in this study (Table 1). In the OPA-N group, four sheep were from Peru, four were from the USA, and one was from Spain; their ages ranged from 3 to 84 months. Studies involving some of these animals have been reported previously.32 The OPA-E group consisted of 12 animals from the USA that were inoculated intratracheally as neonates with either lung tumor homogenate (six sheep) or lung fluid (six sheep) obtained from animals with confirmed cases of OPA, as reported previously.10,31 This group was necropsied between 2 and 7 months postinoculation. Lungs from four unaffected sheep (three adults and one 6-month-old lamb) were used as a source of control tissue for EM. Lung sections from five adult sheep were used as control tissue for IHC.
Histopathology, electron microscopy, and immunohistochemistry of neoplastic cells in naturally occurring (OPA-N) and experimentally induced (OPA-E) cases of ovine pulmonary adenocarcinoma.
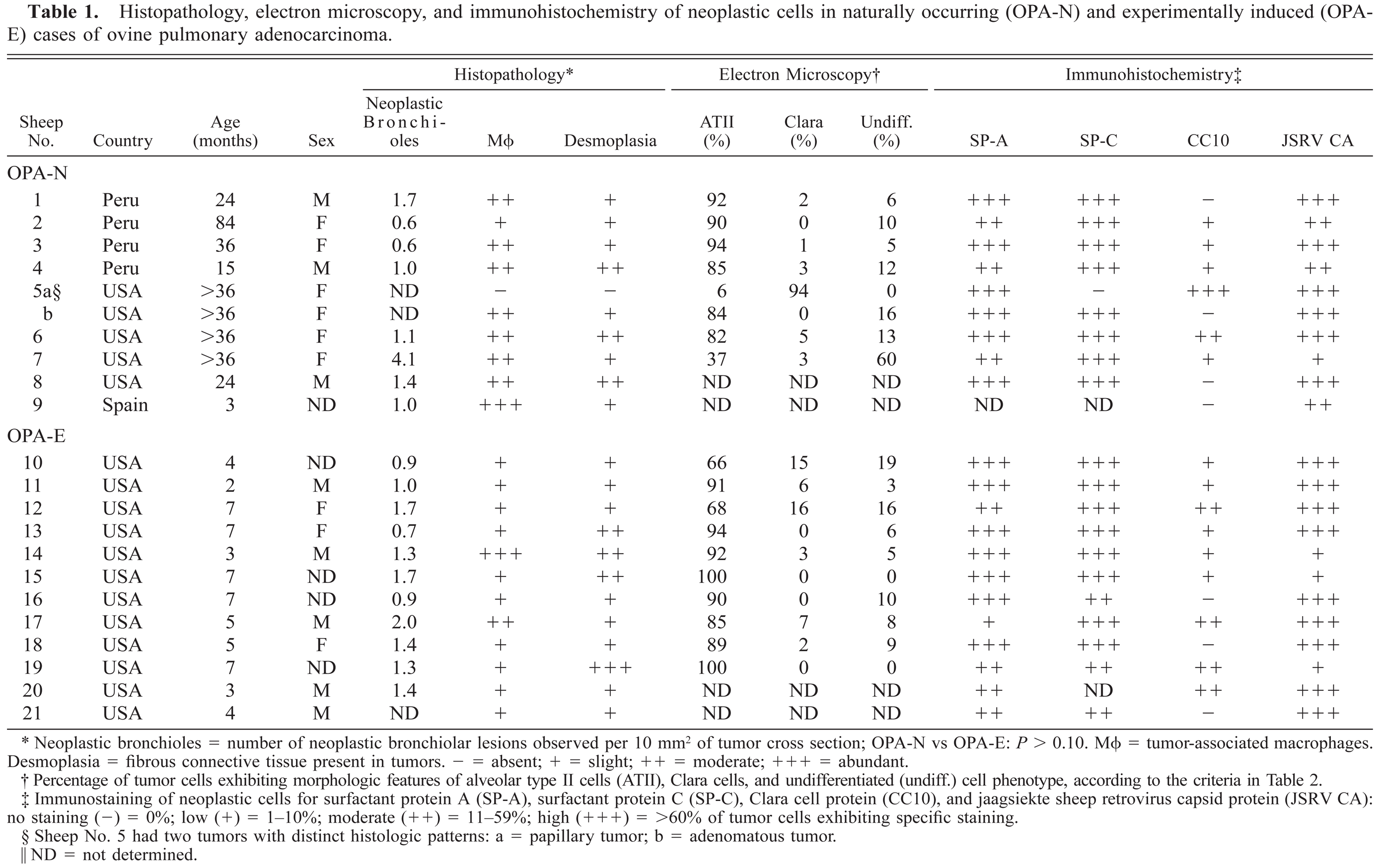
Neoplastic bronchioles = number of neoplastic bronchiolar lesions observed per 10 mm2 of tumor cross section; OPA-N vs OPA-E: P > 0.10. MΦ = tumor-associated macrophages. Desmoplasia = fibrous connective tissue present in tumors. − = absent; + = slight; ++ = moderate; +++ = abundant.
Percentage of tumor cells exhibiting morphologic features of alveolar type II cells (ATII), Clara cells, and undifferentiated (undiff.) cell phenotype, according to the criteria in Table 2.
Immunostaining of neoplastic cells for surfactant protein A (SP-A), surfactant protein C (SP-C), Clara cell protein (CC10), and jaagsiekte sheep retrovirus capsid protein (JSRV CA): no staining (-) = 0%; low (+) = 1–10%; moderate (++) = 11–59%; high (+++) = >60% of tumor cells exhibiting specific staining.
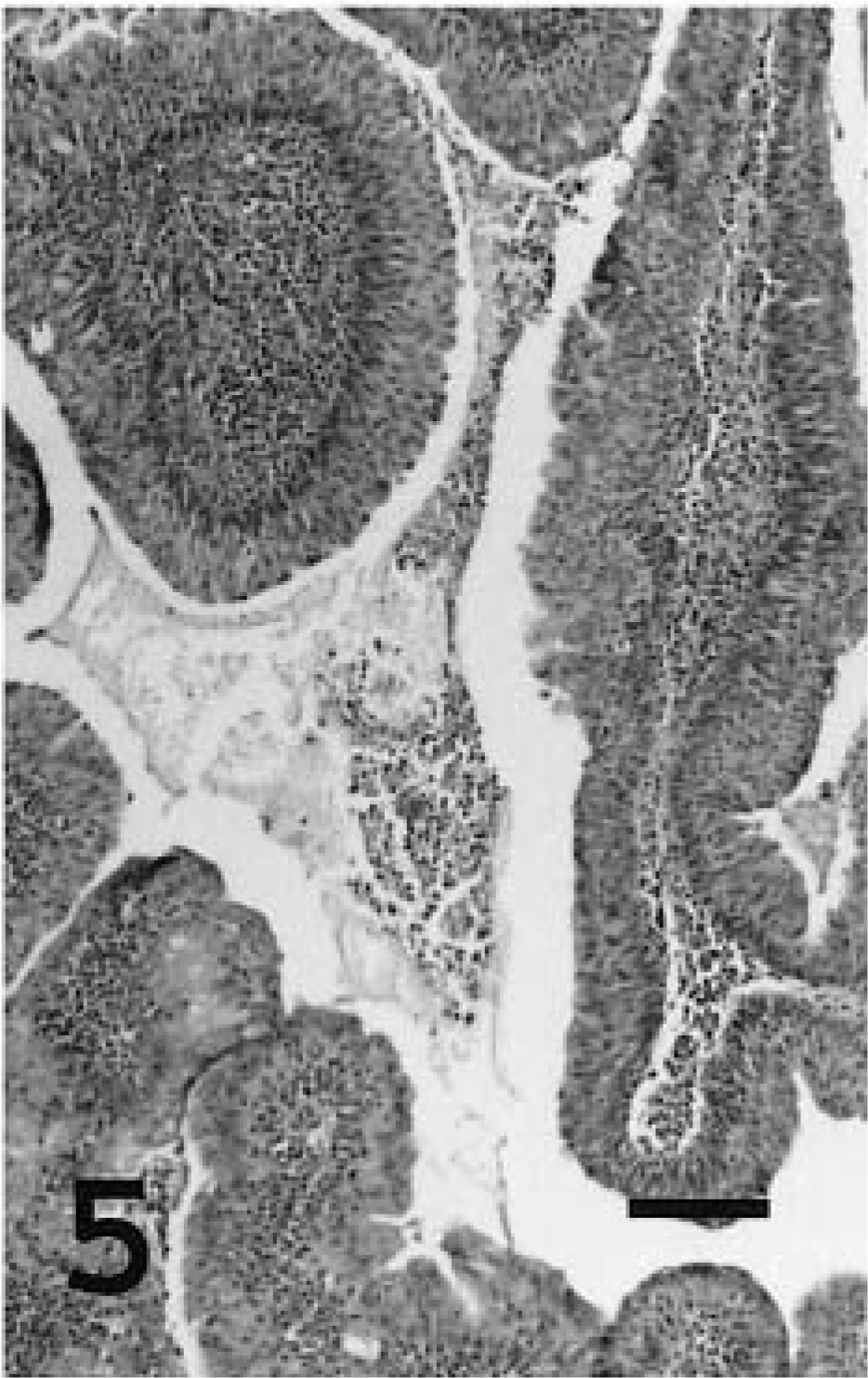
Sheep No. 5 had two tumors with distinct histologic patterns: a = papillary tumor; b = adenomatous tumor.
ND = not determined.
Light microscopy
Tissue samples from lung tumor nodules were collected during necropsy, fixed in 10% neutral buffered formalin, embedded in paraffin, sectioned at 5 μm, and stained with hematoxylin and eosin (HE) as reported previously.31 Morphologic evaluation of the samples was performed, and tumors were examined microscopically for patterns of alveolar and/or bronchiolar involvement of pulmonary parenchyma. Bronchiolar neoplastic involvement was evaluated by enumerating proliferating lesions involving bronchioles per 10 mm2 of tumor cross section. The abundance of tumor-associated alveolar macrophages and presence of desmoplasia also was evaluated.
Transmission EM
Lung tumor tissue samples were fixed in 2% glutaraldehyde and postfixed in 2% osmium tetroxide. A minimum of three 1-μm-thick sections from each tumor were stained with toluidene blue for 2 minutes and examined by light microscopy to select areas for ultrastructural examination. Selection was based on the presence of single or multiple nodules involving either alveoli or bronchioles. Ultrathin sections were cut on an ultra microtome with glass knives and stained with uranyl acetate and lead citrate. Among 7 OPA-N and 10 OPA-E tumors, 47–200 (○ ± SD = 127 ± 11) neoplastic cells per tumor were examined and assigned to three categories (ATII, Clara, or undifferentiated cells) based on their relative abundance of specific organelles (Table 2).
Ultrastructure of neoplastic cells in ovine pulmonary adenocarcinoma.∗

Scale: − = absent; +/− = occasional; + = few; ++ = moderate; +++ = abundant.
IHC
Immunostaining for JSRV CA was performed using rabbit antiserum to recombinant JSRV CA (J. M. Sharp, Moredun Research Institute, Edinburgh, UK).24 Identification of cell phenotypes was performed using anti-sheep SP-A, anti-human pro SP-C, and anti-rat CC10 antibodies (J. A. Whitsett, Children's Hospital Medical Center, Cincinatti, OH). Unstained 5-μm sections of lung tissue were deparaffinized and blocked for endogenous peroxidase with 2% hydrogen peroxide in methanol for 30 minutes. For retrieval of JSRV CA and SP-A antigens, tissues were irradiated in a microwave oven twice for 7 minutes each time in 100 mM citrate buffer (pH 6). SP-C sections were treated with 6 N guanidine hydrochloride for 30 minutes at room temperature followed by 0.2 mg/ml trypsin in 0.1 M calcium chloride for 30 minutes at 37 C. Immunostaining was performed using an avidin–biotin immunoperoxidase system (Vectastain Elite ABC kit; Vector Laboratories, Burlingame, CA) according to the manufacturer's directions. Primary antibodies were applied at the following dilutions: JSRV CA 1:100, SP-A 1:250, and CC-10 1:400 for 30 minutes at room temperature; and SP-C 1:1,000 overnight at 4 C. The peroxidase substrate diaminobenzidine (Sigmafast, Sigma Chemical Co., St. Louis, MO), was applied for 4 minutes (CC10) or 6 minutes (SP-A, SP-C, and JSRV CA), and samples were subsequently counterstained with Harris hematoxylin. Immunohistochemical controls included omission of the primary antibodies and use of normal rabbit serum. Marker expression was evaluated as a percentage of neoplastic cells that exhibited specific staining for each of the antibodies tested: high (+++) = >60%; moderate (++) = 11–59%; low (+) = 1–10%; absent (−) = 0% (Table 1).
Results
Gross and Microscopic Lesions of OPA-N and OPA-E
Tumor tissue samples from nine sheep with OPA-N, including two distinct masses from one animal (sheep No. 5), and 12 sheep with OPA-E were evaluated (Table 1). The gross appearance of the lesions differed between the OPA-N and OPA-E animals. The OPA-N lesions usually consisted of a gray coalescing mass that effaced the ventral portions of the cranial or middle lobes of one or both lungs. Firm 1–4-cm nodules frequently surrounded the main mass and were occasionally present in other lobes. The cut surface of the tumor masses often exuded clear to slightly opaque fluid that was serous to slightly turbid. The OPA-E animals presented with multifocal 0.2–2-cm firm gray nodules that often involved both lungs and all lobes. These tumors also usually exuded fluid from the cut surface.
The OPA-N and OPA-E tumors were evaluated microscopically to compare tumor growth patterns (alveolar or bronchioloalveolar), association with alveolar macrophages, and extent of desmoplasia (Table 1). Individual tumors were nonencapsulated, expansile nodules that replaced and compressed the pulmonary parenchyma. Neoplastic cells formed acinar structures and papillary proliferations that expanded into and effaced alveoli (Figs. 1, 3). Polypoid proliferations were continuous with the terminal bronchiolar epithelium and began to fill the bronchiolar lumen (Fig. 2). The two distinct tumor masses in sheep No. 5 were also distinct microscopically: one was adenomatous and the other had a papillary pattern (Figs. 4, 5). OPA-N and OPA-E groups had a similar frequency of tumors involving the bronchioles or alveoli (Table 1). Neoplastic cells were well differentiated and usually organized as a single layer on the basement membrane. Individual cells were cuboidal to columnar with a moderate amount of eosinophilic cytoplasm that was frequently vacuolated. Vesicular round to oval nuclei were located in the basilar portion of these cells. A single prominent nucleolus was observed in most cells. Cilia were not present on neoplastic cells. Examination of approximately 5,000 tumor cells of three OPA-N and three OPA-E sheep averaged a mitotic index no greater than 0.002% in any tumor. A variable amount of dense fibrous connective tissue (desmoplasia) surrounded and divided the neoplastic nodules (Table 1). Alveolar macrophages with vacuolated cytoplasm were a prominent feature in the alveoli surrounding neoplastic foci of both OPA-N and OPA-E animals (Table 1). In some sections, they nearly filled the air space, and in other areas they were scattered in lower numbers. In most sections there was pale eosinophilic, amorphous material within alveoli, probably corresponding to the excessive lung fluid observed grossly. This fluid contained high levels of surfactant protein A, as determined by immunoblotting (D. Voelker, unpublished data). In addition to the tumor nodules, the lungs of all of the OPA-N animals and eight of 12 of the OPA-E animals had a lymphocytic infiltrate compatible with lymphoid interstitial pneumonia.
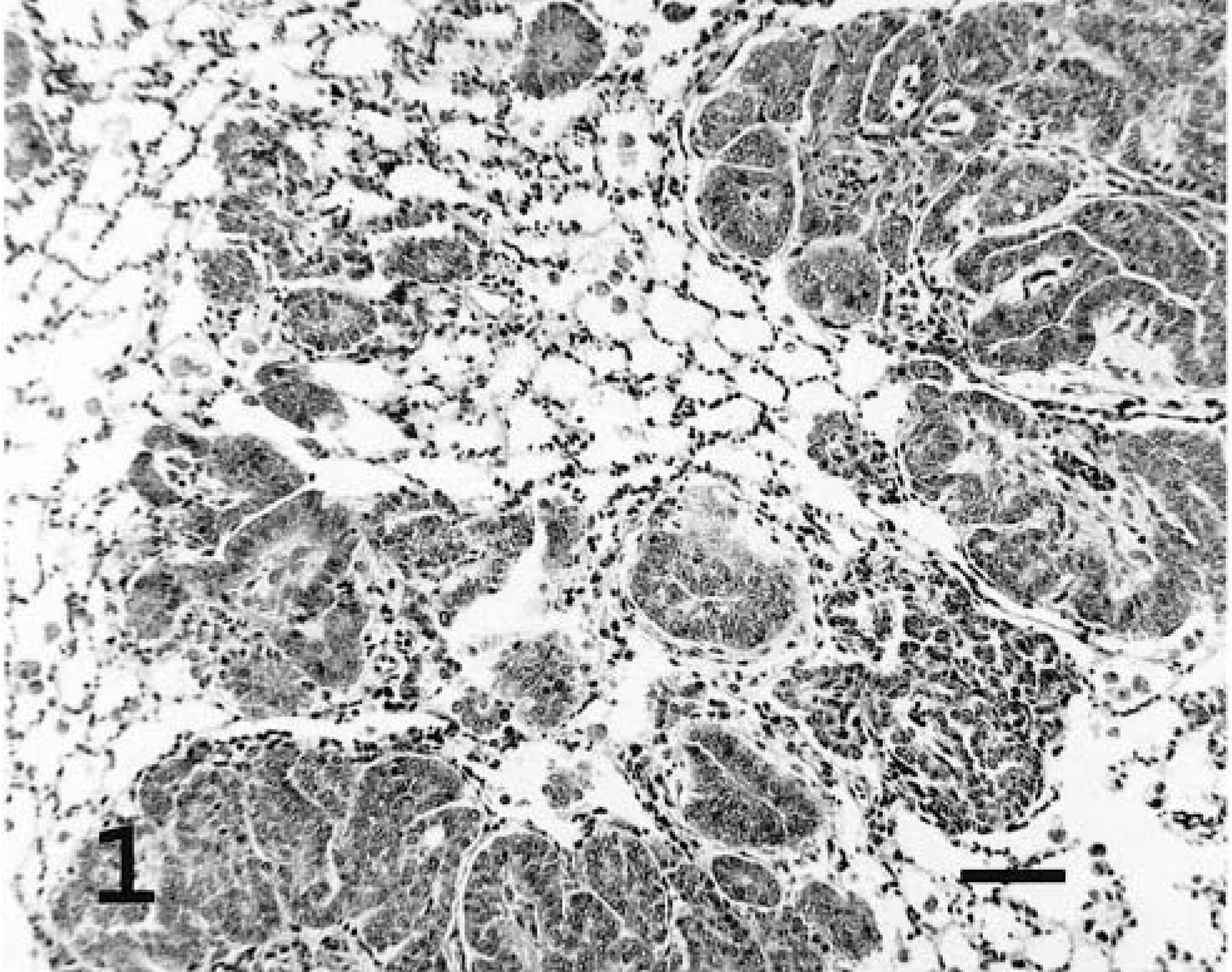
Lung; sheep No. 20. Experimentally induced ovine pulmonary adenocarcinoma with alveolar growth pattern. HE. Bar = 60 μm.
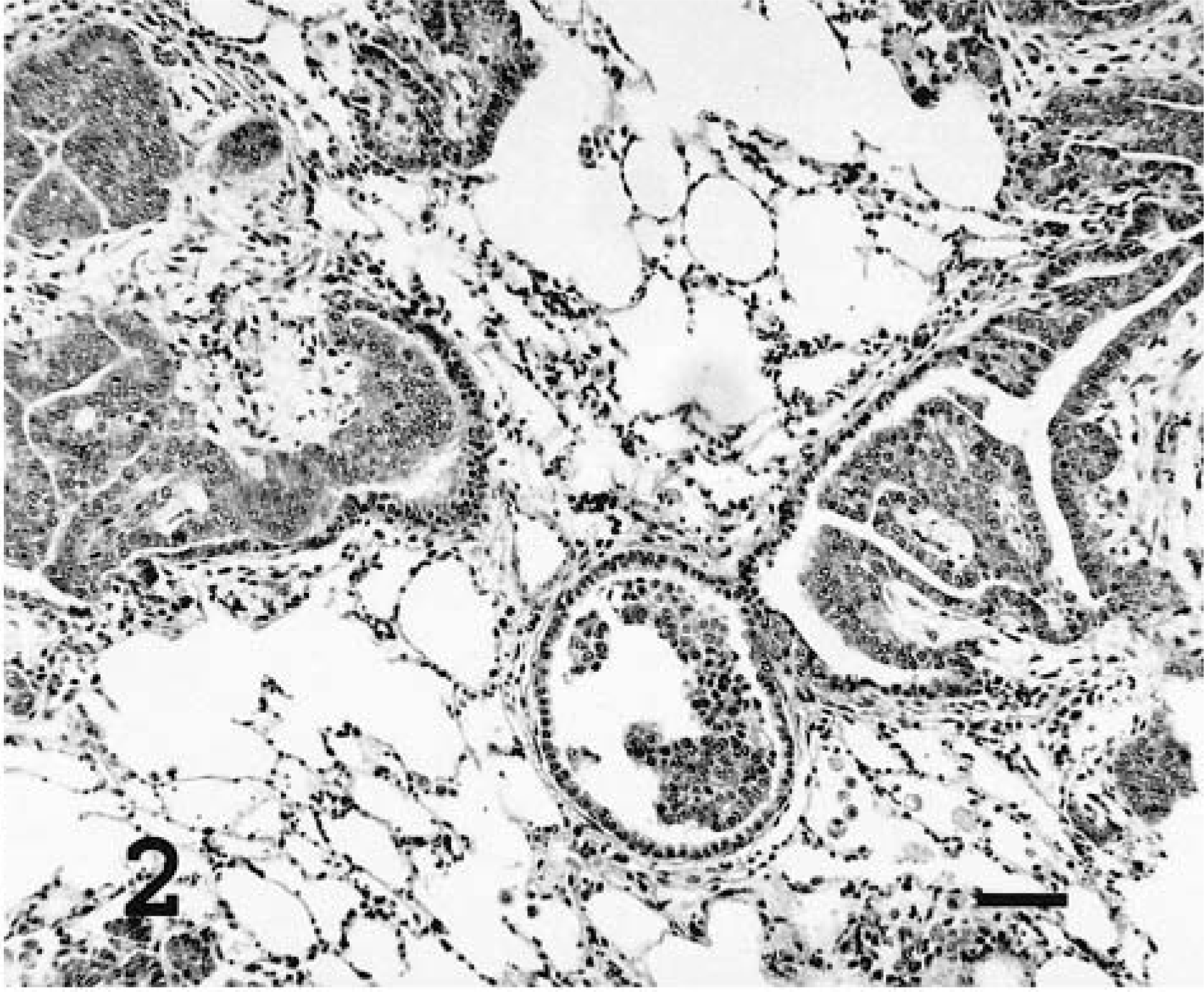
Lung; sheep No. 20. Experimentally induced ovine pulmonary adenocarcinoma with bronchiolar growth pattern. HE. Bar = 50 μm.
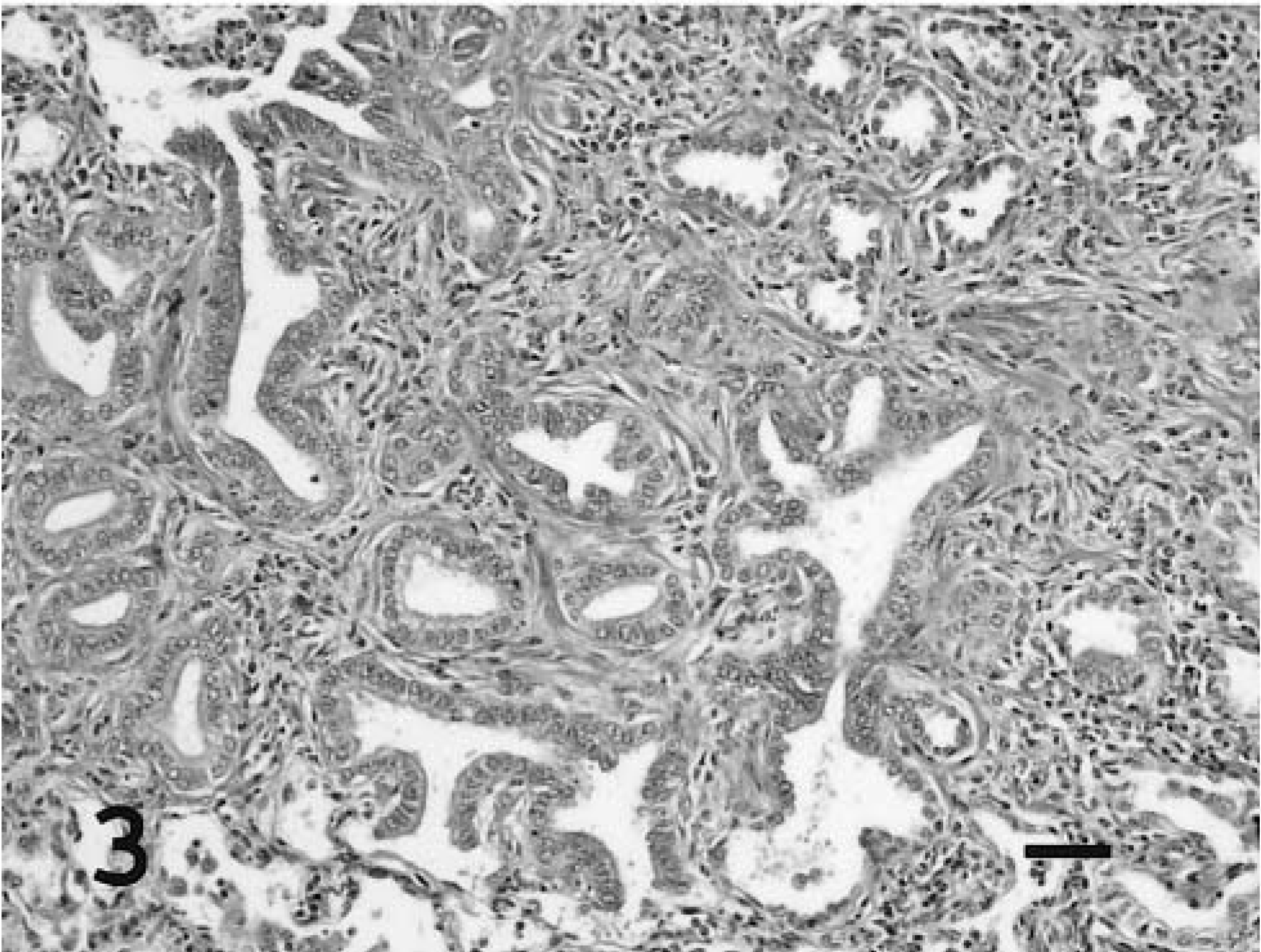
Lung; sheep No. 8. Naturally occurring ovine pulmonary adenocarcinoma with alveolar growth pattern and prominent desmoplasia. HE. Bar = 50 μm.
Lung; sheep No. 5b. Naturally occurring ovine pulmonary adenocarcinoma with adenomatous histologic features. HE. Bar = 50 μm.
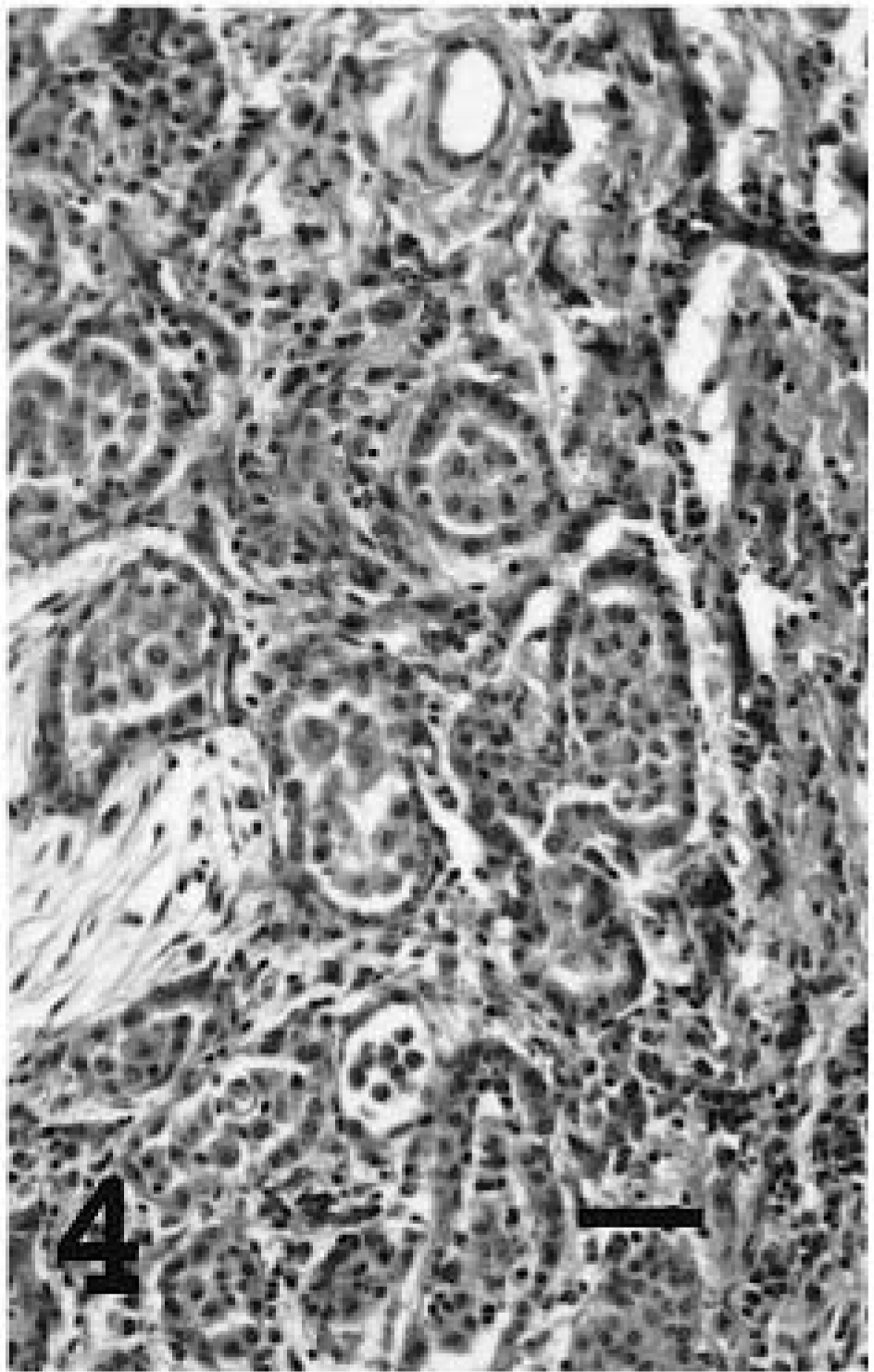
Lung; sheep No. 5, tumor a. Naturally occurring ovine pulmonary adenocarcinoma with papillary histologic features. HE. Bar = 60 μm.
Transmission EM
An ultrastructural study was performed to determine the phenotype and differentiation of cells present in OPA-N and OPA-E groups (Table 1). Most of the tumor cells were cuboidal to columnar and had a round central to basal nucleus with a discrete chromatin pattern and a prominent nucleolus. Numerous irregularly shaped mitochondria were observed. Heterogenous membrane-bound granules and vesicles were also present in many cells. Glycogen granules were seen in most cells. Variations in the organelles and ultrastructure of tumor cells allowed categorization into three groups, ATII cells, Clara cells, and undifferentiated cells, according to their intracytoplasmic organelles and the presence/absence of tight junctions and microvilli (Table 2). Three lung samples from normal sheep were used to develop these ultrastructural criteria. Neoplastic cells of the ATII type contained numerous cytoplasmic lamellar bodies (Fig. 6). These bodies were bound by a single membrane, contained either concentric or parallel lamellae, and were irregularly or apically distributed throughout the cell. Other features of ATII cells were abundant microvilli on the luminal surface, tight junctions between adjacent cells, well-developed rough endoplasmic reticulum (RER), and numerous glycogen granules. Neoplastic Clara cells were characterized by very well-developed smooth endoplasmic reticulum (SER) and numerous apical electron-dense granules 0.3 μm in diameter (Fig. 7). Desmosomal junctions (not well developed) were seen between some cells. Microvilli were occasionally present. Undifferentiated tumor cells lacked characteristic features of ATII (lamellar bodies) or Clara cells (electron-dense granules) (Fig. 8). This group of neoplastic cells featured moderate amounts of RER, with occasional microvilli and glycogen granules. The ultrastructural features of these undifferentiated cells suggest that they may be immature ATII cells.
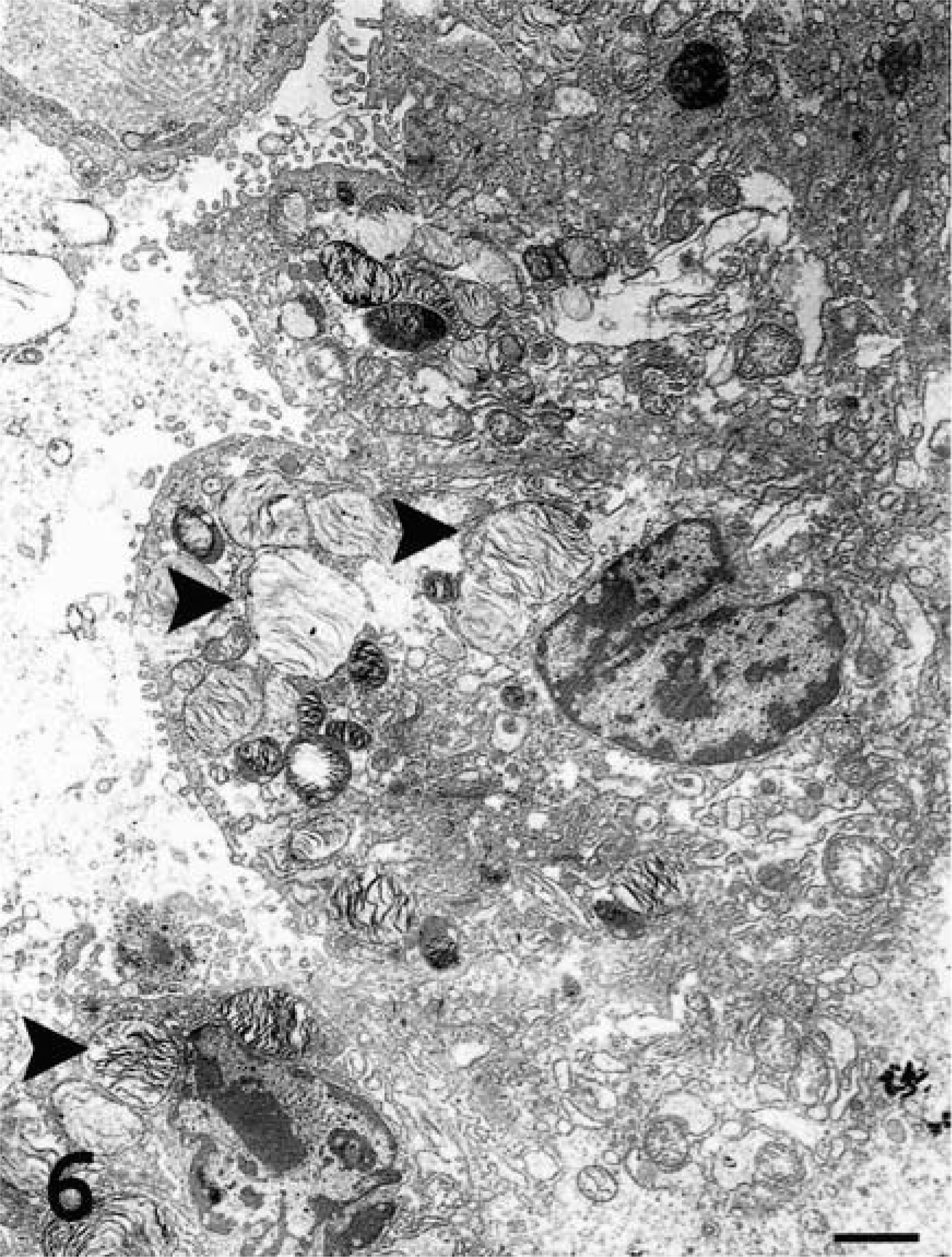
Transmission electron micrograph. Lung; sheep No. 6. Naturally occurring ovine pulmonary adenocarcinoma. ATII tumor cells with numerous intracellular lamellar bodies (arrowheads) and luminal microvilli. Uranyl acetate and lead citrate. Bar = 2 μm.
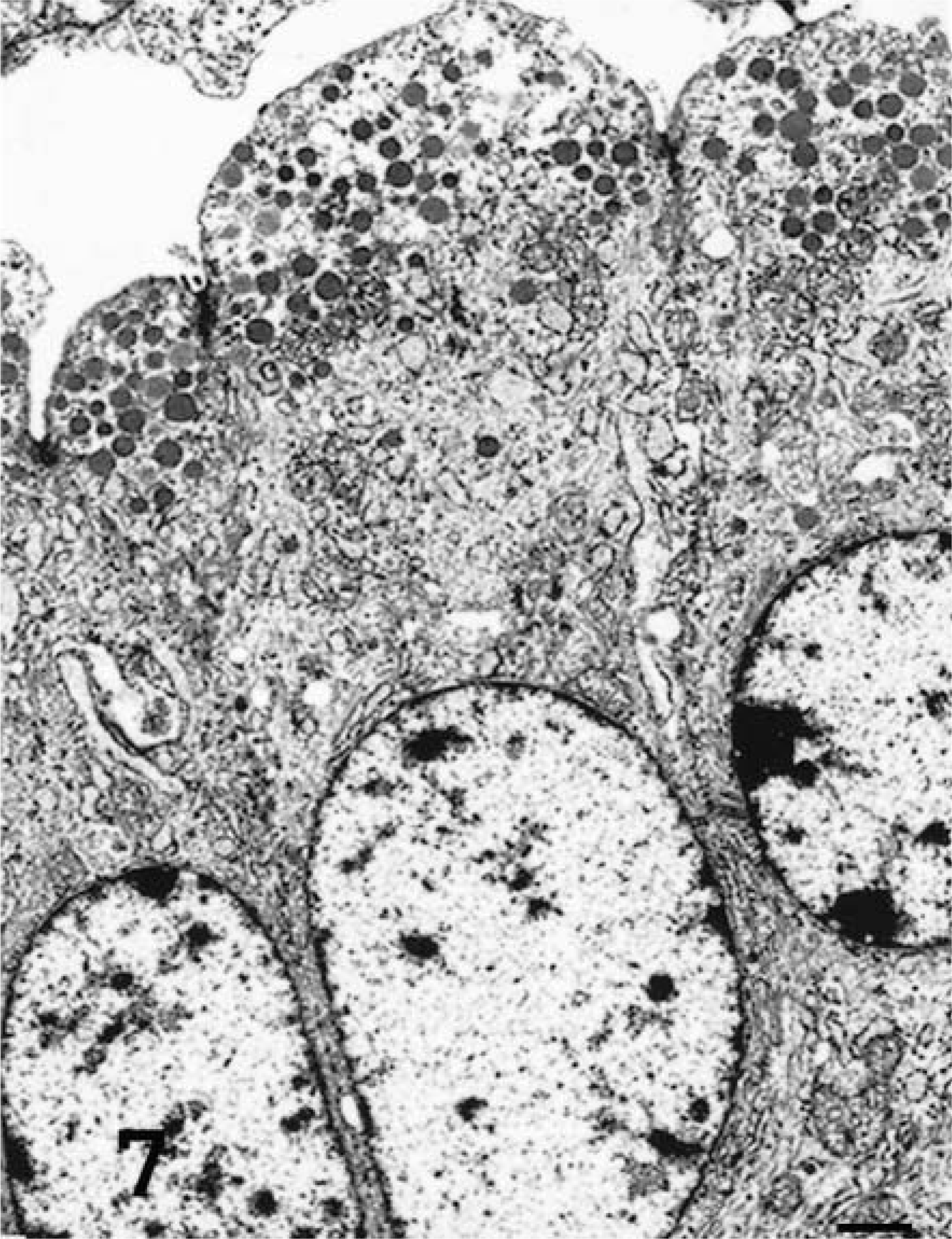
Transmission electron micrograph. Lung; sheep No.5, tumor a. Naturally occurring ovine pulmonary adenocarcinoma. Clara tumor cells with electron-dense apical granules (0.3 μm diameter) and abundant smooth endoplasmic reticulum. Uranyl acetate and lead citrate. Bar = 1 μm.
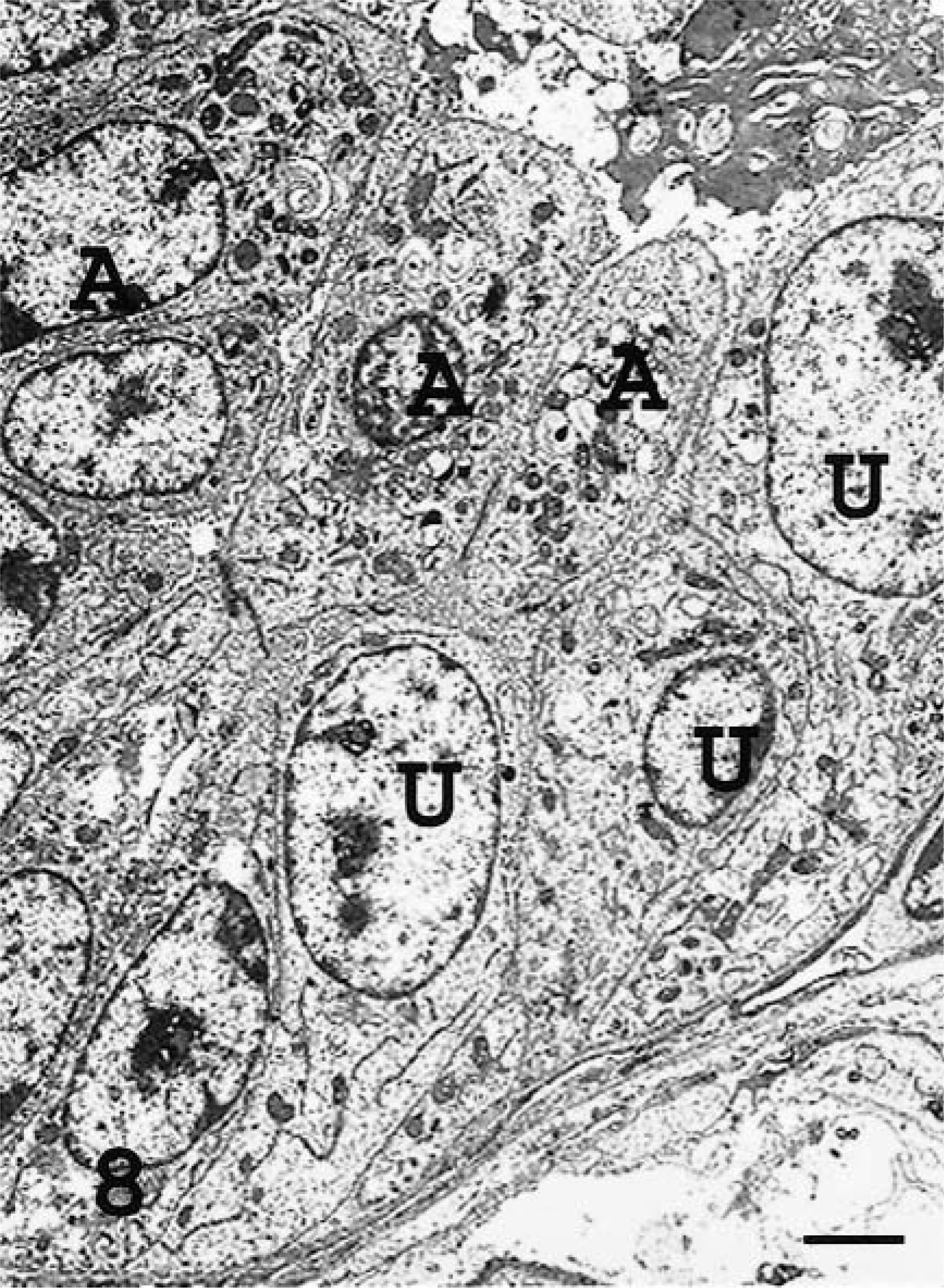
Transmission electron micrograph. Lung; sheep No.3. Naturally occurring ovine pulmonary adenocarcinoma. Tumor containing ATII-like tumor cells (A) with microvilli, intracellular dense granules and vacuoles, and lamellar body–like structures and undifferentiated cells (U) with no microvilli, few cytoplasmic organelles, and no lamellar bodies or electron-dense apical granules. Uranyl acetate and lead citrate. Bar = 2 μm.
Using the criteria described in Table 2, the proportion of neoplastic cell types varied among tumors (Table 1). The mean frequency (± SD) of the cell types in seven OPA-N cases was 75% (± 24%) for ATII cells, 9% (± 17%) for Clara cells, and 16% (± 20%) for undifferentiated cells. The mean frequency for 10 OPA-E sheep was 88% (± 12%) for ATII cells, 5% (± 6%) for Clara cells, and 7% (± 6%) for undifferentiated cells. No significant difference was noted between the two groups (P = 0.37). When OPA-N and OPA-E animals were combined, the mean frequency of phenotypes identified was 82% for ATII cells, 7% for Clara cells, and 11% for undifferentiated cells. ATII cells clearly predominated in both OPA-N and OPA-E sheep. The proportion of undifferentiated cells varied from 0% to 60% among tumors of both OPA-N and OPA-E groups. Clara cells were found in most tumors but in much lower numbers than ATII cells except in OPA-N sheep No. 5, an animal that had two distinct tumor masses (Table 1).
Nontumor cells present included alveolar macrophages, which were more abundant in animals with abundant alveolar proteinaceous fluid, and other inflammatory cells such as neutrophils, lymphocytes, and plasma cells. In sheep with lymphoid interstitial pneumonia, there were prominent infiltrates of lymphocytes. No viral particles were identified in tumor or nontumor cells in the sections examined.
IHC
IHC was performed to determine the cellular immunophenotype and the differentiation state of the neoplastic cells and their correlation with presence of JSRV CA protein as a marker for viral presence (Table 1, Figs. 9–22). There was some variation in the intensity of the staining reaction for each antibody within and between the tumors. For clarity, the percentage of tumor cells that stained with each antibody is presented graphically in Fig. 9. There was a wide variation in the proportion of cells that expressed each of the phenotypic markers in both groups of animals, but no consistent differences in marker expression between the groups were observed. Overall, a mean of 70% and 80% of tumor cells expressed SP-A and SP-C proteins, respectively, whereas a mean of 17% of the tumor cells expressed CC10 protein. JSRV CA protein was detected in 71% of tumor cells in both groups.
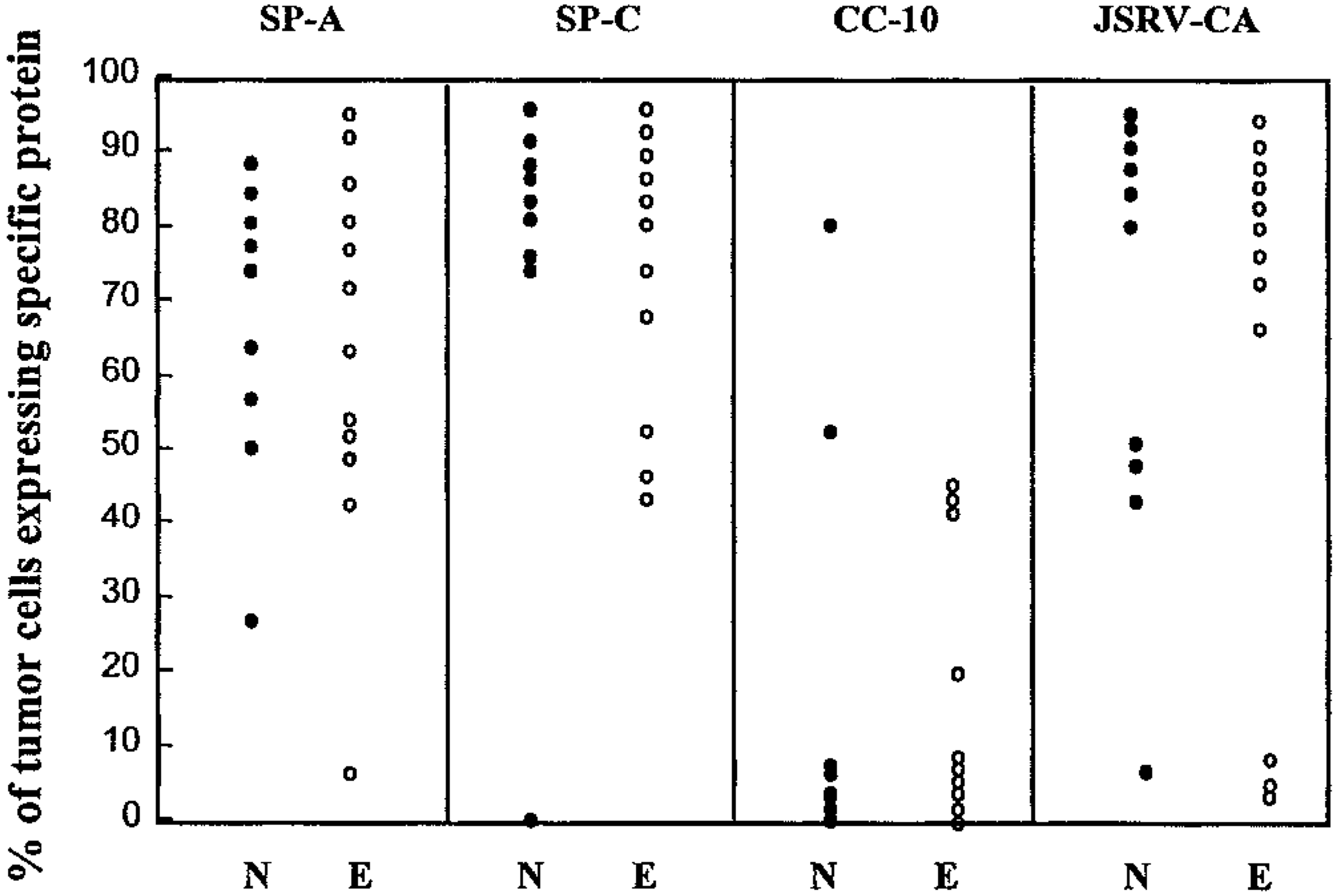
Immunostaining of tumor cells expressing surfactant protein A (SP-A), surfactant protein C (SP-C), Clara cell secretory protein (CC10), and jaagsiekte sheep retrovirus capsid protein (JSRV CA) in nine sheep with OPA-N (N) and 12 sheep with OPA-E (E). OPA-N sheep No. 5 is plotted as two tumors: 5a = papillary and 5b = adenomatous.
SP-A
As a phenotypic marker for ATII and Clara cells, SP-A immunostaining was used to characterize the tumor cells of sheep with OPA. In the lungs of normal sheep, SP-A was observed in alveolar and terminal bronchiolar epithelial cells and in macrophages (Fig. 10). There was cytoplasmic staining specific for SP-A in neoplastic cells of all 21 tumors examined (Figs. 11). The percentage of specifically stained tumor cells was high in 13 sheep (62%), moderate in seven sheep (33%), and low in one sheep (5%) (Table 1). In sheep No. 5, there was specific staining in sections exhibiting both the papillary and adenomatous microscopic features (Table 1). Alveolar macrophages and proteinaceous fluid within alveoli exhibited specific staining. Neither lymphocytes nor other interstitial cells in tumors or normal lung stained with this antibody.
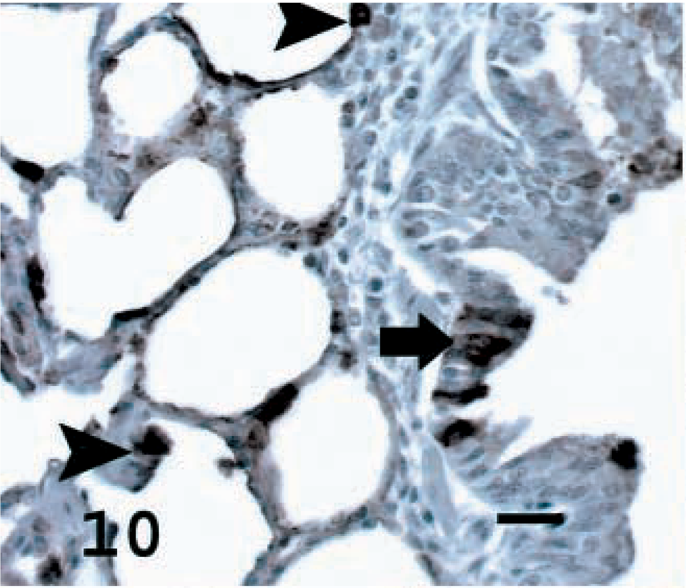
Lung; normal sheep. SP-A in normal sheep lung seen as brown stain in ATII cells (arrowheads) and some terminal bronchiolar epithelial cells (arrow). Avidin–biotin immunoperoxidase with hematoxylin counterstain. Bar = 15 μm.
Lung; OPA-N sheep No. 8. SP-A showing intracellular staining of tumor cells and of surfactant layer in lumen. Avidin–biotin immunoperoxidase with hematoxylin counterstain. Bar = 15 μm.
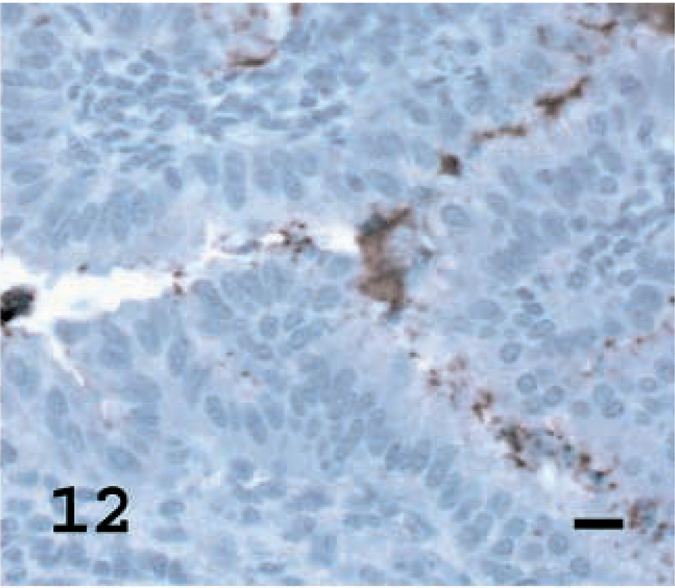
Lung; OPA-E sheep No. 17. SP-A staining in few tumor acells, but marked staining of surfactant layer. Avidin– biotin immunoperoxidase with hematoxylin counterstain. Bar = 15 μm.
SP-C
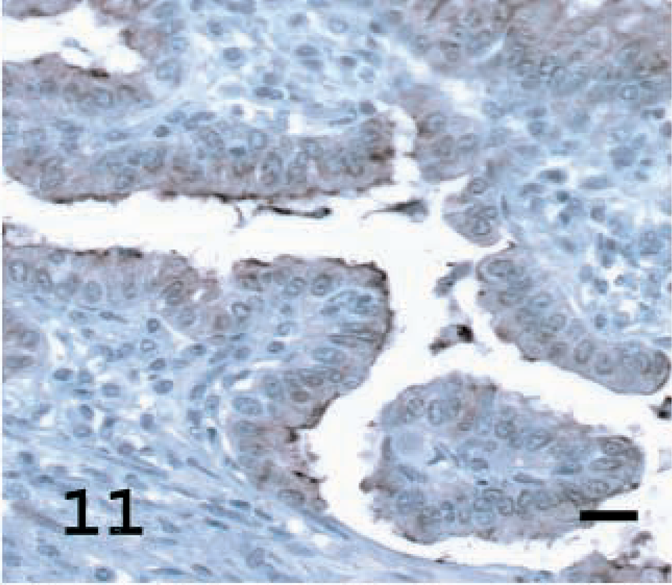
SP-C is a specific marker for ATII cells, at least in rodents and primates, and was employed to determine the percentage of cells with ATII differentiation in the sheep tumors. Pro-SP-C was detected in alveolar cells of normal lungs (presumably ATII cells) but not in the epithelial cells of the conducting airways (Fig. 13). Specific staining for pro-SP-C was seen in all 19 tumors stained; 16 tumors (84%) exhibited a high percentage of stained tumors cells, and three tumors (16%) had a moderate percentage of stained tumor cells (Figs. 14, 15, Table 1). Cytoplasmic staining also was seen in some terminal bronchioles that had evidence of hyperplasia. In sheep No. 5, there was specific staining for SP-C in the section with adenomatous features but no staining in the section with a papillary pattern.
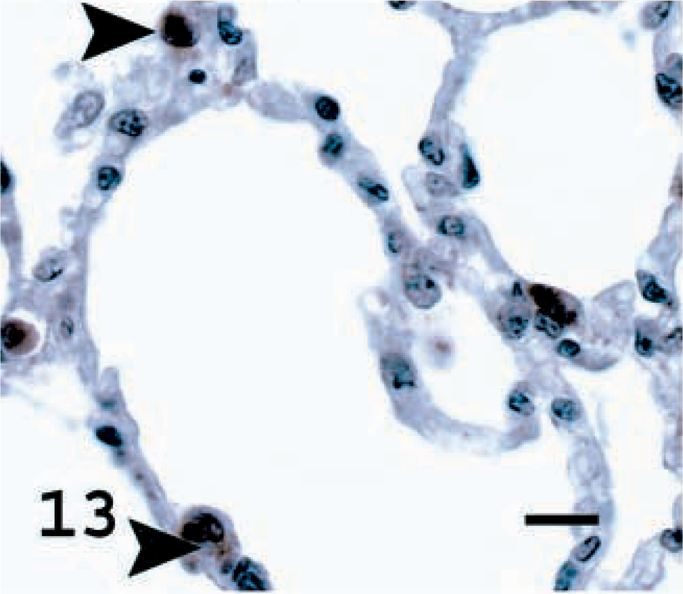
Lung; normal sheep. SP-C in normal sheep lung with brown intracytoplasmic staining of ATII cells (arrowheads). Avidin–biotin immunoperoxidase with hematoxylin counterstain. Bar = 11 μm.
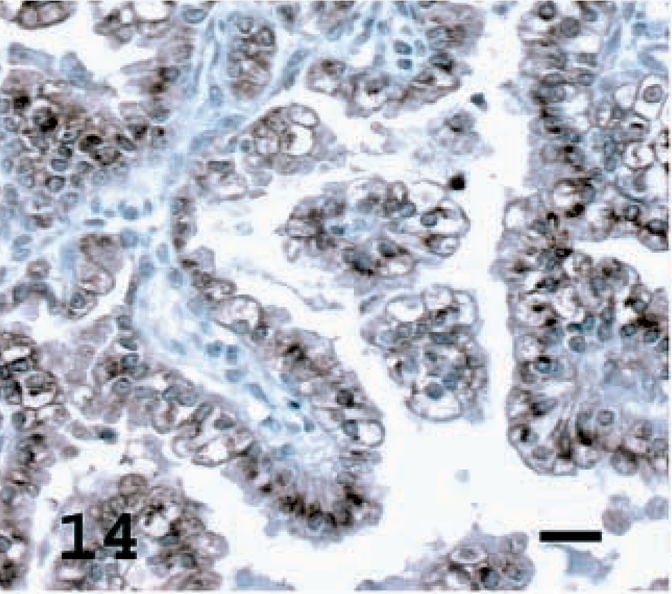
Lung; OPA-N sheep No. 8. SP-C staining of a high percentage of tumor cells. Avidin–biotin immunoperoxidase with hematoxylin counterstain. Bar = 20 μm.
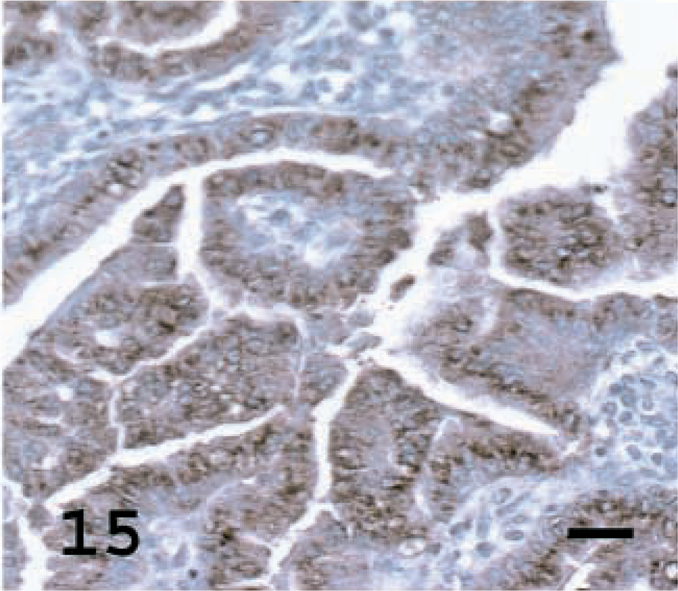
Lung; OPA-E sheep No. 17. SP-C staining of a high percentage of tumor cells. Although this tumor shows a bronchioloalveolar growth pattern, the tumor cells express high levels of SP-C. Avidin–biotin immunoperoxidase with hematoxylin counterstain. Bar = 20 μm.
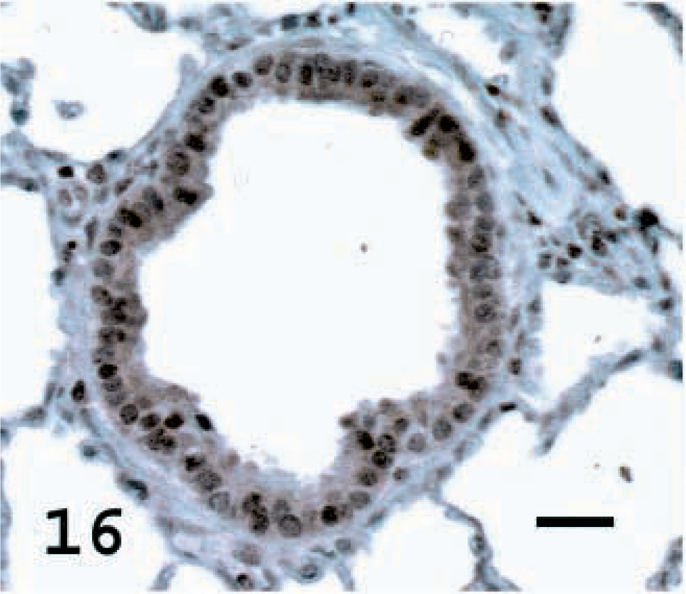
Lung; normal sheep. CC10 in normal sheep lung showing staining of the terminal bronchiolar epithelial cells. Avidin–biotin immunoperoxidase with hematoxylin counterstain. Bar = 20 μm.
CC10
CC10 is a protein secreted by Clara cells and was used to identify the percentage of cells with Clara cell differentiation in the tumors. CC10 was not detected in the normal alveolar epithelial cells of normal lung but was observed in terminal bronchiolar and occasional bronchial epithelial cells (Fig. 16). Among the tumors examined, a high percentage of tumor cells stained in one tumor (5%), a moderate percentage stained in five tumors (23%), a low percentage stained in nine tumors (40%) and there was no staining in seven tumors (32%) (Figs. 17, 18, Table 1). In sheep No. 5, there was marked specific staining for CC10 in the papillary section but no staining in the adenomatous part of the tumor.
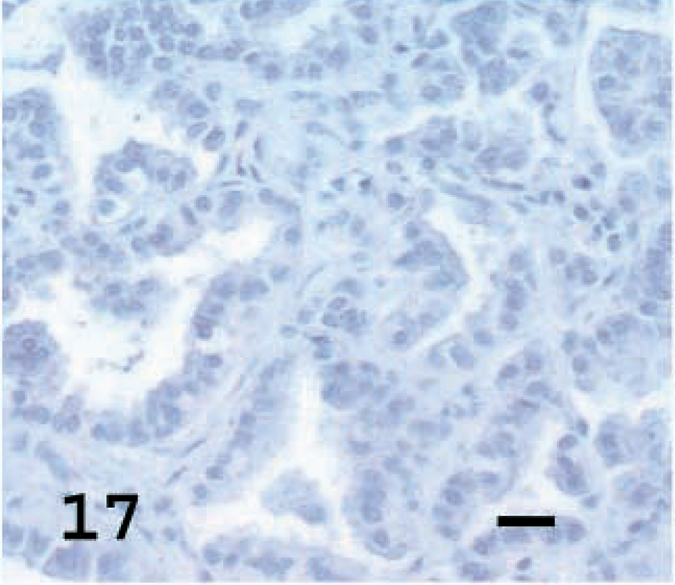
Lung; OPA-N sheep No. 8. CC10 showing no staining in tumor cells. Avidin–biotin immunoperoxidase with hematoxylin counterstain. Bar = 20 μm.
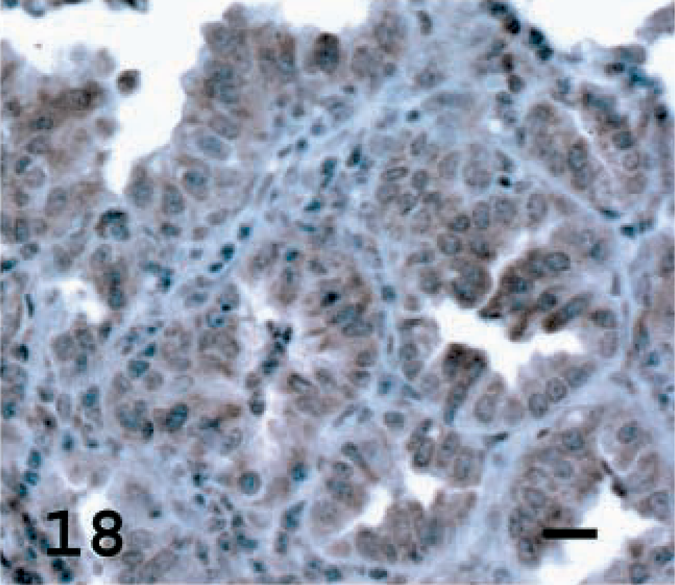
Lung; OPA-E sheep No.17. CC10 showing moderate staining of tumor cells, a surprising finding because this tumor also expressed high levels of SP-C. Avidin–biotin immunoperoxidase with hematoxylin counterstain. Bar = 15 μm.
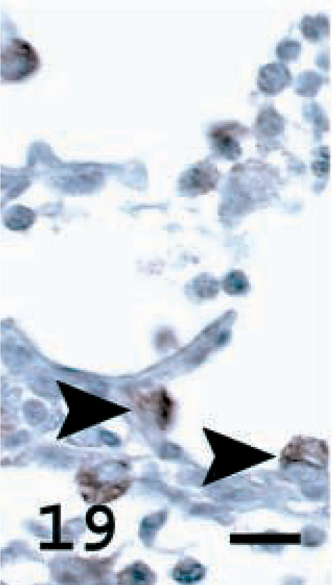
Lung; OPA-E sheep No. 17. JSRV CA expression in nontumor lung tissue. ATII cells (arrowheads) in alveolar wall of nonneoplastic lung tissue showing brown cytoplasmic staining. Avidin–biotin immunoperoxidase with hematoxylin counterstain. Bar = 12 μm.
JSRV CA
Immunostaining for JSRV CA was performed using antiserum to recombinant JSRV CA to determine the frequency and distribution of this viral protein within tumor cells and whether nonneoplastic cells also contained JSRV CA (Table 1). Among the 22 tumors examined, the percentage of tumor cells that stained specifically for JSRV CA was high in 15 tumors (68%), moderate in three (14%), and low in four (18%) (Figs. 21, 22, Table 1). In some tumor sections, individual cells or groups of cells that were within a tumor nodule that stained intensely were adjacent to cells that did not stain at all (Fig. 21). In three OPA-N and three OPA-E sheep, specific cytoplasmic staining was observed in occasional epithelial cells (bronchiolar and alveolar) that did not show neoplastic change (Figs. 19, 20). Only 14 of the 21 tumors had sufficient nonneoplastic alveolar and bronchiolar tissues for adequate examination. Although exfoliated tumor cells that were free in the alveolar space were difficult to distinguish morphologically from alveolar macrophages (Fig. 20), cells consistent with macrophage morphology showed specific staining in three OPA-N sheep and in two OPA-E sheep. Control lung tissues showed no specific staining with this antibody.
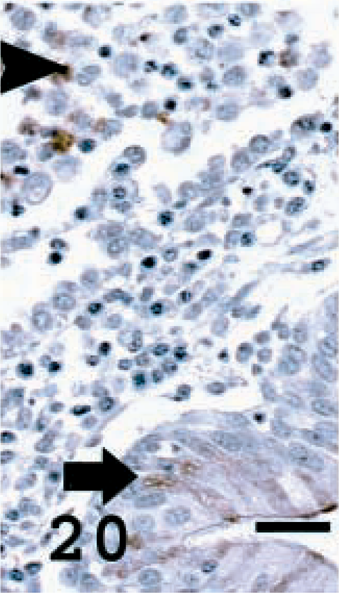
Lung; OPA-E sheep No. 17. JSRV CA expression in non tumor lung tissue. Leukocytes (possibly some macrophages) stain in alveolar lumen (arrowhead) and brown staining observed in some nonneoplastic bronchiolar cells (arrow). Avidin–biotin immunoperoxidase with hematoxylin counterstain. Bar = 25 μm.
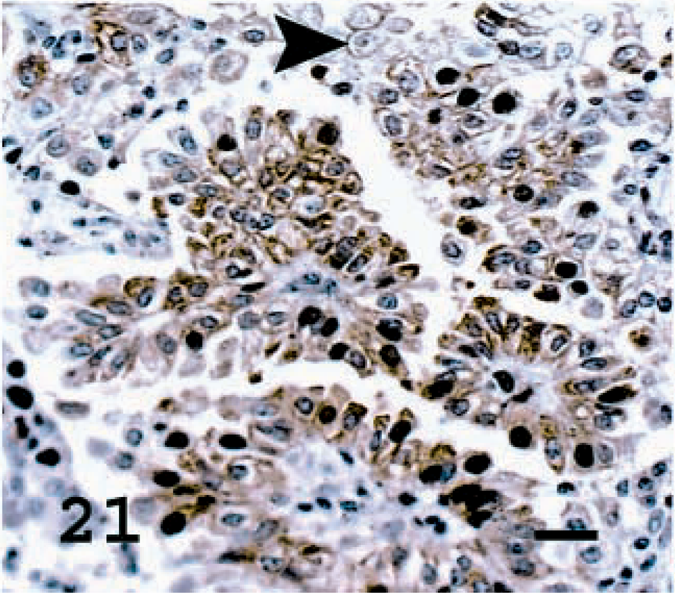
Lung; OPA-N sheep No. 8. JSRV CA staining present in most tumor cells. Not all tumor cells express the capsid protein (arrowhead). Avidin–biotin immunoperoxidase with hematoxylin counterstain. Bar = 20 μm.
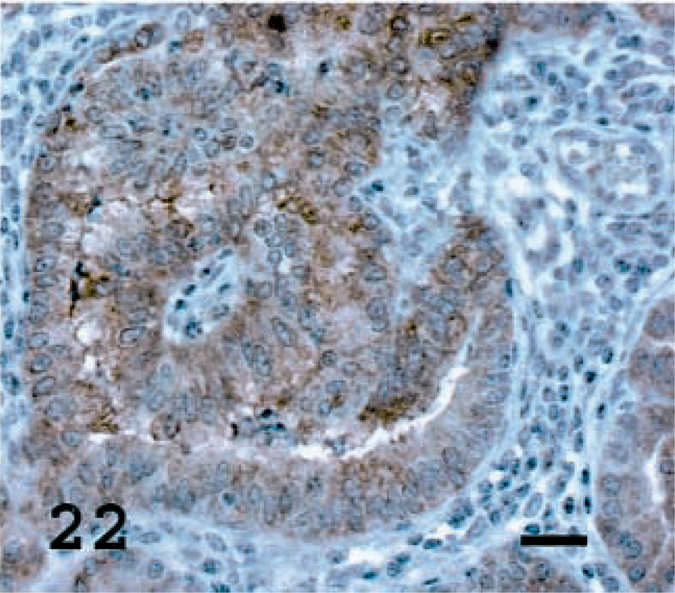
Lung; OPA-E sheep No. 17. JSRV CA staining present in most tumor cells. Avidin–biotin immunoperoxidase with hematoxylin counterstain. Bar = 20 μm.
Discussion
Our data indicate that OPA tumor nodules were primarily composed of ATII cells containing JSRV CA. Neoplastic cells with features of Clara cells and undifferentiated cells also were present but in lower frequency. A higher background turnover rate in ATII cells than in Clara cells, observed in the normal sheep lung (unpublished data), may account for an increased frequency of viral infection, integration, and transformation of ATII cells in JSRV-infected lung. A heterogeneous population of tumor cells was present in most tumors and occasionally even within the same tumor nodule, suggesting either a virus-induced field cancerization event initially involving multiple cell types or retrograde, trans-, or continued differentiation of ATII/Clara lineage cells following neoplastic transformation.
The state of differentiation of neoplastic ATII cells, based on presence of cytoplasmic organelles and expression of SP-A and SP-C, varied as previously reported in neoplastic and nonneoplastic lung tissue in humans, mice, and sheep.5,12,19,36 The ultrastructural appearance of anaplastic ATII cells, particularly the cytoplasmic glycogen fields, was similar to that of fetal ATII cells as reported in mouse lung.36 In sheep Nos. 6, 12, 17, and 19, up to 40% of tumor cells expressed both CC10 and SP-C, which may reflect infection of a pluripotential progenitor cell that is capable of secreting both proteins.14,34 Alternatively, marker expression may be correlated with the degree of differentiation of neoplastic cells. For instance, proliferating neoplastic cells may express proteins that are uncharacteristic of a given cell type14 or they may lose marker expression.35 Hyperplastic ATII cells also have increased SP-A or CC10 production.15,16
Recent reports have indicated that the JSRV Env protein is capable of transforming rodent cells24,25 and avian fibroblasts (unpublished data), suggesting that this viral product may be important in OPA oncogenesis. Furthermore, the JSRV LTR is preferentially active in ATII cells,23 suggesting a mechanism for enhanced viral gene expression in ATII lineage cells. The finding of widespread JSRV CA in tumor cells likely correlates with viral infection and replication in these cells. During viral replication, sufficient levels of the oncoprotein JSRV Env may be produced to induce ATII cell proliferation. The lack of JSRV CA presence in some tumor cells may be explained by insensistivity of our IHC assay or the possibility that JSRV CA–negative cells may have undergone a second oncogenic event such as virus-induced insertional mutagenesis to account for their continued proliferation. In support of a role for retroviral insertional mutagenesis in OPA, JSRV integration into the SP-A gene in reverse orientation was recently reported in a tumor cell line.9 If this integration site is common in OPA, the pathogenesis of the disease may include disregulated SP-A production and consequent effects on pulmonary homeostasis and ATII cell proliferation. These processes could, in turn, facilitate JSRV infection and integration in ATII cells, leading to Env-induced proliferation and resulting in a perpetual cycle of ATII cell proliferation.
JSRV-CA was mainly identified in tumor cells but could occasionally be seen in alveolar macrophages or pulmonary epithelial cells adjacent to tumor nodules. Viral protein within macrophages may represent infection or simply virion proteins phagocytosed from the alveolar fluid. ATII cells secrete and reabsorb surfactant proteins, and the capsid protein also may enter the cells via this route.18,20,22 Capsid expression seen in nonneoplastic epithelial cells may indicate that these cells are infected but not recognized as neoplastic at the time of examination. An additional possibility that cannot be excluded is expression of viral capsid proteins from JSRV-related endogenous proviruses, particularly in the highly metabolically active neoplastic cells. In this regard, a protein that cross-reacts immunologically with JSRV CA protein detected in human lung tumors8 also has been found in human endogenous retrovirus–expressing seminomas (unpublished data), thus demonstrating the upregulation of an endogenous viral protein in a neoplasm.
We compared the neoplastic lesions in naturally occurring cases of OPA with those experimentally induced using lung fluid or tumor homogenates containing JSRV. Differences between the natural disease and the model were few. In general, there was an earlier onset of clinical disease in experimentally inoculated animals that may relate to the increased proliferation of ATII cells in neonatal lambs at the time of inoculation. It is not known at what age animals are naturally infected, but it is likely that the dose is lower than in the experimentally infected lambs. In addition, experimentally induced disease tends to result in a large number of small widely disseminated tumor nodules, whereas the tumors in animals with naturally occurring disease tend to consist of large monocentric coalescing masses involving one or more lung lobes. The latter pattern more closely resembles human BAC; differences between the natural and the experimental disease may be related to the age at onset of tumors or delay in diagnosis in the natural disease. There was no difference in the proportion of specific cell types found in OPA-E and OPA-N nor was there a difference in JSRV CA expression observed in this study. This finding suggests that the basic mechanism of neoplasia is similar in experimentally induced and naturally occurring OPA and helps validate the experimental disease as a model for the natural disease.
These results indicate that JSRV primarily transforms ATII cells but also Clara cells at a lower frequency, although the tumors are frequently heterogeneous. This study is the first to enumerate the proportion of specific cell types in OPA and to determine the association between JSRV infection and neoplasia, as assessed by expression of JSRV CA. Further critical examination of the expression of JSRV Env in tumor cells and cellular genetic changes associated with JSRV integration events are required to elucidate the pathogenesis of OPA. Future studies of the pathogenesis of sheep OPA should provide fundamental information about BAC in humans.
Footnotes
Acknowledgements
This work was supported by National Institutes of Health grants CA R01 59116 (to J. C. DeMartini) and CA 59116–07S1 (to J. A. Platt) from the National Cancer Institute. Dr. DeMartini is a member of the University of Colorado Cancer Center. We thank Drs. Raul Rosadio, Michael D. Lairmore, and J. Michael Sharp for performing some of the necropsy and histopathology examinations, Dr. Lluis Lujan for providing tissues of an OPA sheep from Spain, and Drs. Thomas Allen and Dennis Voelker for critical reading of the manuscript.
