Abstract
Contagious respiratory tumors of sheep and goats are epithelial neoplasms of the lung and nasal cavities. They are associated with oncogenic betaretroviruses known as jaagsiekte sheep retrovirus and enzootic nasal tumor retrovirus of sheep and goats. We investigated the presence of the envelope protein (ENV) of these retroviruses in retropharyngeal and mediastinal lymph nodes using a specific monoclonal antibody by immunohistochemistry methods, single-labeled or combined with ovine B or T lymphocytes or macrophage cell markers. Samples of lymph nodes, fixed in formalin and zinc fixative, were obtained from paraffin-embedded material. Four groups of samples were used: 24 natural cases of ovine pulmonary adenocarcinoma (OPA), 13 of enzootic nasal adenocarcinoma of sheep (ENAS), 19 of enzootic nasal adenocarcinoma of goats (ENAG), and 14 control samples. ENV was detected by single labeling in cortical lymphoid follicles. Six of 24 OPA samples were positive and only in those from sheep with extensive neoplasia. Immunolabeling was detected in 5/13 ENAS and 10/19 ENAG samples. Positive labeling was found either in the intercellular spaces, membranes, or cytoplasm of cells in follicles. Control samples were not correspondingly labeled. Double immunohistochemistry demonstrated co-labeling of ENV and CD21 (B cells and follicular dendritic cells) in all samples, CD14 (macrophage) in OPA samples, and Pax-5 (B cells) in ENAG samples, but not with CD8 or CD4 (T lymphocytes). These results demonstrate the presence of betaretrovirus ENV proteins in nontumor cells in regional lymph nodes in sheep and goats with contagious respiratory tumors.
Keywords
Contagious respiratory tumors of sheep and goats are neoplastic diseases that arise from secretory epithelial cells of the terminal airways-alveolar areas of the lungs and ethmoidal turbinate nasal mucosa, and retain their secretory function after transformation. Sheep are naturally affected by 2 diseases ovine pulmonary adenocarcinoma (OPA; jaagsiekte, pulmonary adenomatosis) and enzootic nasal adenocarcinoma (ENAS), while goats are affected by enzootic nasal adenocarcinoma (ENAG). These diseases are caused by 3 related oncogenic exogenous betaretroviruses, namely, jaagsiekte sheep retrovirus (JSRV), enzootic nasal tumor 1 (ENTV-1), and enzootic nasal tumor 2 (ENTV-2), respectively. 4,20,25 They have been recorded in many sheep and goat rearing countries throughout the world, with the exception of Australia and New Zealand, with a major impact on sheep production in some areas. 24
Over the years, significant progress has been made to understand the relationship of these viruses with factors such as cellular receptors, cell tropism and tissue specificity, the molecular mechanisms of oncogenic transformation, and transcriptional response of the lung to JSRV infection. 14,19 The pathology, epidemiology, transmission, genetic resistance, and ante-mortem tests of the associated diseases have been reported. 1,2, 5,7,16,28 Despite these achievements, information regarding the interactions of these viruses with the immune system still very limited.
Natural infections with JRSV, ENTV-1, and ENTV-2 are characterized by an apparent absence of a specific humoral response, despite the high production of virus in the tumors, either in the lungs or the nasal cavities. 21,26 However, recent observations have identified the presence of neutralizing antibodies to ENTV-1 in sheep with natural or experimental ENAS. 28,29 In addition, a study of the transcriptional response of ovine lung to JSRV infection suggests the presence of an immunomodulatory environment within the OPA lung, which has the potential to suppress the immune response to JSRV and tumor cells and to actively promote tumor growth. 14
The 3 retroviruses use the same receptor and can infect a wide range of cells including those of the immune system. 19 Thus, JSRV RNA and DNA was consistently found in tumors and mediastinal lymph nodes in both natural and experimental OPA; with JSRV transcripts were also detected in peripheral blood mononuclear cells. In these experiments, specific PCRs (polymerase chain reactions) detected exogenous JSRV and discriminated it from the endogenous counterparts. 3,10,22 In addition, in the mediastinal lymph nodes from sheep with natural OPA, proviral burden was greatest in the macrophage population and detected in B and T lymphocytes. 13 The information available for ENTV-1,2 is much less by comparison. Thus, in natural cases of ENAG a specific PCR test detecting ENTV-2 revealed that viral DNA is most frequently detected in medial retropharyngeal lymph nodes and tonsils. However, in natural cases of ENAS, ENTV-1 appears to be confined to nasal tumors, with only occasional detection in other organs. 20 Therefore, the detection of proviral DNA provides clear evidence that these viruses replicate in some cells of the immune system as they do in epithelial cells where the tumors develop.
Immunohistochemistry (IHC) using specific polyclonal antibodies against JSRV capsid proteins (JSRV Rab) have been used as an indicator of viral expression in tissue sections, demonstrating that most of the tumor cells were positive, with few positive cells in the lymph nodes and alveolar macrophages of sheep with OPA. 25 More recently, monoclonal antibodies (JSRV Mab) against the surface domain of the JSRV envelope protein (ENV) were generated. Two of them (Clones C9 and B3) gave intense IHC labeling of lung tumors in sheep infected with JSRV and in mice exposed to an adenoviral vector encoding ENV (replication-defective adeno-associated virus type 6). 30 These monoclonal antibodies recognized tumors in all JSRV-infected sheep examined 7,30 but not any cross-reacting antigens in lung samples with a variety of diseases that were not the result of JSRV infection. 30 In addition, these antibodies also labeled tumor cells in mouse lung tumors induced by the same adenoviral vector encoding ENTV-1 envelope protein and in natural cases of ENAS 30 and ENAG (M. De las Heras, unpublished results). Together these results indicate the antibodies are highly specific for ENV of small ruminant betaretroviruses (JSRV, ENTV-1, and ENTV-2) and may provide a useful diagnostic test for these retroviruses using IHC in contagious respiratory tumors of sheep and goats.
On the other hand, published papers describe detection of immune cells in paraffin wax–embedded ovine tissues, using an extensive panel of antibodies directed against markers associated with ovine lymphocytes, macrophages, and other cells. This has created the opportunity to immunophenotype sheep peripheral lymph nodes using paraffinized tissues, thus providing valuable tools in the investigation of the sheep immune system. 11,17
Groups of cells with lymphoreticular morphology in the mediastinal lymph nodes from OPA cases using JSRV Rab have been previously reported. 7,25 Based on this information we focused our investigation on draining lymph nodes (DLNs). Specifically, for natural OPA cases, we analyzed the mediastinal or tracheobronchial lymph nodes that receive the lymph from the lungs. For ENAS and ENAG cases, we analyzed the retropharyngeal lymph nodes that receive lymph from the nasal cavity. We selected these DLNs as they are important points of interaction of OPA, ENAS, and ENAG tumors with the immune system. In this study, utilizing IHC, we investigated the presence of ENV in DLNs of natural cases of OPA, ENAS, and ENAG using JSRV Mab. In addition, we have employed single or double IHC to identify the cell types containing ENV in DLNs from contagious respiratory tumors of sheep and goats.
Materials and Methods
Tissue Samples and Processing
We focused our investigation on samples of mediastinal and tracheobronchial lymph nodes that collect lymph from the lungs, and retropharyngeal medial lymph nodes that collect lymph from the nasal cavities. They were obtained from adult sheep and goats from commercial flocks or herds arranged into the following groups: A: 4 sheep from flocks with a history of OPA. They tested PCR-negative for JSRV on blood samples and had no OPA lesions by microscopic examination of the lungs. B: 4 sheep from flocks with a history of OPA. They were tested PCR-positive for JSRV on blood samples and had no OPA lesions by microscopic examination of the lungs. C: 6 sheep from flocks with a history of OPA and with OPA forming a single grossly visible nodule. D: 10 sheep from flocks with a history of OPA and with multinodular/diffuse extensive gross lesions of OPA affecting 2 or more lung lobes. E: 6 sheep from flocks with no history of OPA and tested blood PCR-negative on blood samples (OPA-negative control group). F: 5 sheep with unilateral ENAS. G: 8 sheep with bilateral ENAS. H: 4 sheep from flocks with no history of ENAS (ENAS-negative control group). I: 9 goats with unilateral ENAG. J: 10 goats with bilateral ENAG. K: 4 goats from herds with no history of ENAG (ENAG-negative control group).
Samples from all groups were obtained from our tissue archive of natural cases of OPA, ENAS, or ENAG in sheep and goats. Control samples were also obtained from our archive. Sheep blood samples were tested for JSRV using PCR previously described. 10 From all cases we had samples fixed in 10% neutral formalin and processed routinely for paraffin embedding. In addition, samples from 4 sheep and goats positive to JSRV Mab from the groups D, G, and J, together with 2 sheep and goats from the control groups (E, H, K) fixed in zinc salts, were used in this study for double IHC experiments. For zinc fixation, samples from 3- to 4-mm thick tissue slices were placed in plastic cassettes and immersed in zinc salt fixative solution (0.1 M Tris base buffer with Ca acetate 0.5%, pH 7.4 containing 0.5% Zn acetate and 0.5% Zn chloride) for 48 to 72 hours at 4 °C. We added samples fixed using the zinc salt fixative procedure for the demonstration of lymphocytes and other immune system cell markers in paraffin wax–embedded samples, adapted for ovine tissues. 11 This method overcomes poor antigen preservation associated with aldehyde-based fixatives and preserves morphology. All samples were processed for paraffin embedding and preserved in paraffin blocks following routine procedures.
Immunohistochemistry
For IHC using JSRV Mab, tissue sections from all samples were dewaxed and hydrated by routine methods. Briefly, hydrated tissue sections were immersed in Tris-buffered saline (TBS; 0.05 M Tris HCl, 0.15 M NaCl, pH 7.4-7.6) for 10 minutes, 3 times. Endogenous peroxidase activity was quenched using Dual Endogenous (peroxidase and alkaline phosphatase) Enzyme blocking reagent (Agilent Tech-DAKO S2003) for 10 minutes. This step was followed by another 3 immersions in TBS for 10 minutes each. In an attempt to minimize nonspecific tissue antigen reactions, the sections were treated with 2.5% horse serum (Vector Laboratories) in TBS for 30 minutes, at room temperature (RT). After removing excess solution, the slides were incubated at 4 °C overnight with the JSRV Mab. This Mab (kindly donated by Dusty Miller; Fred Hutchinson Cancer Research Centre, Seattle, WA) was a mixture of 2 clones (C9 and B3). 30 It was used at 1/400 in TBS. Diluted mouse normal serum and TBS replaced primary antibodies as routine controls of unspecific reactions or residual endogenous peroxidase reaction. Tissue sections from groups E, H, and K were used as negative tissue controls. As positive tissue controls, tumor sections from natural cases of OPA, ENAS, and ENAG were also tested. The procedure continued with 3 TBS washes of 10 minutes at RT, followed by covering the slides with labeled polymer-HRP (Agilent Tech-DAKO Real Envision Kit K5007). After another 3 TBS washes, sections were covered with the solution of substrate chromogen (peroxidase/diaminobenzidine [DAB] rabbit/mouse, Agilent Tech-DAKO Real Envision kit [K5007]) and were incubated for 30 minutes at RT. After this step, the sections were washed with distilled water, counterstained with Carazzi’s hematoxylin, dehydrated, and mounted routinely. Positive CFs were counted and the proportion was calculated in relation to the total number of CFs on each section.
Double-labeling IHC was used to detect ENV as well as ovine CD21, CD4, CD8, and CD14. When viral markers were investigated together with antibodies with predominant membranous-cytoplasmic location in targeted cells using Polink kits (Polink DS-MM-Hu C kit [DS203C], Golden Bridge International). Tissue sections from 4 animals of groups D, G, and J, and 2 animals from each of the control groups were fixed in zinc and tested using this protocol. These colorimetric kits can be used with paraffinized tissues and contain non-biotin polymer conjugates with 2 distinct chromogens. The result is a multicolor staining. One chromogen is green (Emerald) and the other is red (GBI-permanent red) and when precipitating together a blue/purple color is produced. Thus, when 2 antigens colocalize, a third color is developed. Tissue sections from all samples were dewaxed and hydrated by routine methods and treated following the manufacturer instructions, with some alterations.
TBS containing 0.05% Tween 20 (TBST) was washing buffer. All antibodies were incubated overnight at 4 °C. No epitope retrieval or counterstain options were carried out in any experiment. Primary antibodies for these experiments, on the same section, were combinations of the JSRV Mab diluted 1/400 in TBS with the following monoclonals and dilutions in TBS: bovine CD21 (Bio Rad antibodies, MCA 1424GA, 1/15 000), sheep CD4 (Bio Rad antibodies, MCA2213GA, 1/600), sheep CD8 (Bio Rad antibodies, MCA 2216GA, 1/700), and sheep CD14 (Bio Rad antibodies, MCA920GA, 1/200). In addition, 2 extra sections were included with a single marker combined with TBS. Diluted markers were replaced by TBS and mouse serum dilutions as controls of unspecific reactions.
A different double-labeling IHC protocol was used to detect ENV and Pax-5 or Ki-67. Tissue sections from 4 animals of groups D, G, and J and 2 animals from controls groups (E, H, K) fixed in zinc were tested with this protocol. Sections from OPA or nasal tumors fixed in zinc were used as positive controls for JSRV Mab antibody. Tissue sections were dewaxed and hydrated by routine methods. Briefly, hydrated tissue sections were immersed in TBS for 10 minutes, repeated 3 times. Peroxidase activity was quenched using Bloxall TM endogenous peroxidase and alkaline phosphatase blocking solution (Vector Laboratories) for 10 minutes. This step was followed by another 3 immersions in TBS for 10 minutes each. To remove nonspecific tissue antigens, sections were treated with 2.5% horse serum (Vector Laboratories) in TBS for 30 minutes. After removing excess solution, the slides were incubated at 4 °C overnight with monoclonal mouse anti-human B-cell-specific activator protein, clone DAK-Pax5 (Pax5) (DAKO product no. M7307) diluted 1/700 in TBS, and monoclonal mouse anti-human Ki-67 antigen, Clone Mib-1 (Ki-67) (DAKO product no. M7240) diluted 1/500 in TBS. The sections were incubated at 4 °C overnight. Diluted rabbit or mouse normal serum and TBS replaced primary antibodies as routine controls for nonspecific reactions or residual endogenous peroxidase activity. Post incubation, 3 TBS washes of 10 minutes each at RT, followed by covering the slides with ImmPRESS horse anti-mouse IgG polymer (Vector Laboratories product no. MP-7402) for 45 minutes. After a further 3 TBS washes (as described above), the sections were covered with the solution of ImmPACT DAB peroxidase substrate (Vector Laboratories product no. SK-415) and incubated for 3 to 4 minutes at RT. The sections were then washed with distilled water for 10 minutes, followed by 3 changes of TBS. Sections were again treated with 2.5% horse serum (Vector Laboratories) in TBS for 30 minutes. After removing the excess solution, the slides were incubated at 4 °C overnight with JSRV Mab diluted 1/400 for test sections, negative controls, and tumor sections. Followed by 3 TBS washes of 10 minutes each at RT and the slides covered with ImmPRESS horse anti-mouse IgG polymer (Vector Laboratories product no. MP-7402) for 45 minutes. After another 3 TBS washes, the sections were covered with the solution of ImmPACT DAB VIP substrate (Vector Laboratories product no. SK-4605) and incubated for 3 to 4 minutes at RT. Finally, the sections were washed with tap water, dehydrated, and mounted. No contrast staining was carried out on these sections.
Results
Detection of ENV in Draining Lymph Nodes
The majority of the ENV labeling was observed in the cortical lymphoid follicles (CFs) in all lymph node samples except the negative controls. Most areas with ENV labeling showed the characteristic histological morphology of secondary CFs in all groups of samples. However, there was one additional change recorded only in ENAG cases as described below. In the OPA and ENAS cases, ENV immunolabeling was confined to the center with little penetration to the marginal zones of the CFs. For sheep with OPA, immunolabeling was only detected in 6 of 10 sheep from group D (ie, sheep with extensive OPA lesions) and none of the other groups. In group D, the 6 positive DLNs samples contained a mean of 57% positive CFs (Table 1; Figs. 1, 2). Fine granular or linear precipitates with variable intensity were found either on cell membranes or intercellular spaces within the centers of CFs. In addition, scattered cells with morphology suggestive of lymphocytes and reticular cells had strong cytoplasmic labeling, and these were intermingled with nonlabeled cells (Fig. 2). Therefore, extracellular and cellular cytoplasmic labeling for ENV was identified only in the OPA group with extensive lesions (group D).
Immunohistochemistry (IHC) Results From Draining Lymph Node Samples From Ovine Pulmonary Adenocarcinoma (OPA) and Enzootic Nasal Adenocarcinoma of Sheep (ENAS) and of Goats (ENAG)a.
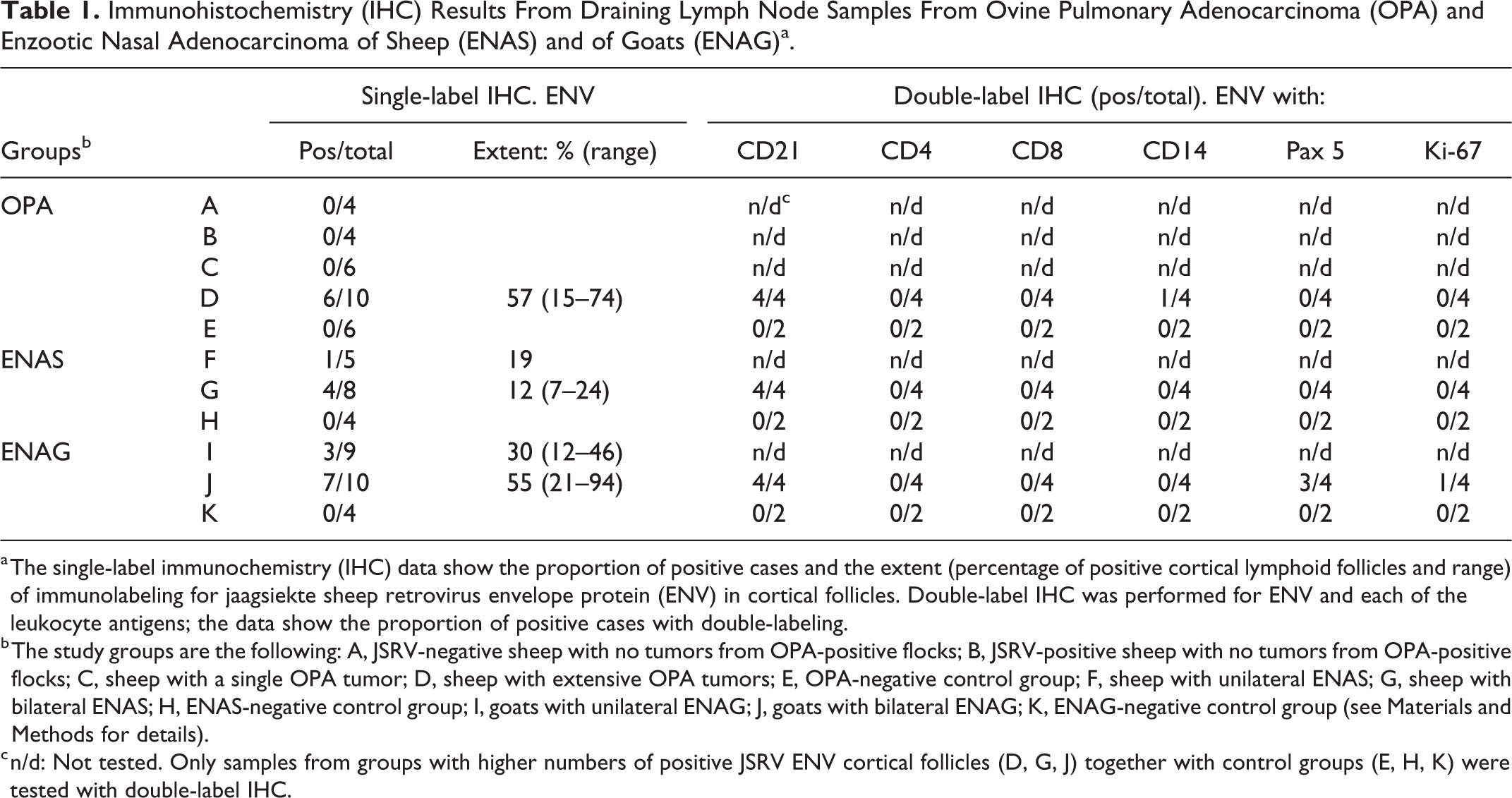
a The single-label immunochemistry (IHC) data show the proportion of positive cases and the extent (percentage of positive cortical lymphoid follicles and range) of immunolabeling for jaagsiekte sheep retrovirus envelope protein (ENV) in cortical follicles. Double-label IHC was performed for ENV and each of the leukocyte antigens; the data show the proportion of positive cases with double-labeling.
b The study groups are the following: A, JSRV-negative sheep with no tumors from OPA-positive flocks; B, JSRV-positive sheep with no tumors from OPA-positive flocks; C, sheep with a single OPA tumor; D, sheep with extensive OPA tumors; E, OPA-negative control group; F, sheep with unilateral ENAS; G, sheep with bilateral ENAS; H, ENAS-negative control group; I, goats with unilateral ENAG; J, goats with bilateral ENAG; K, ENAG-negative control group (see Materials and Methods for details).
c n/d: Not tested. Only samples from groups with higher numbers of positive JSRV ENV cortical follicles (D, G, J) together with control groups (E, H, K) were tested with double-label IHC.
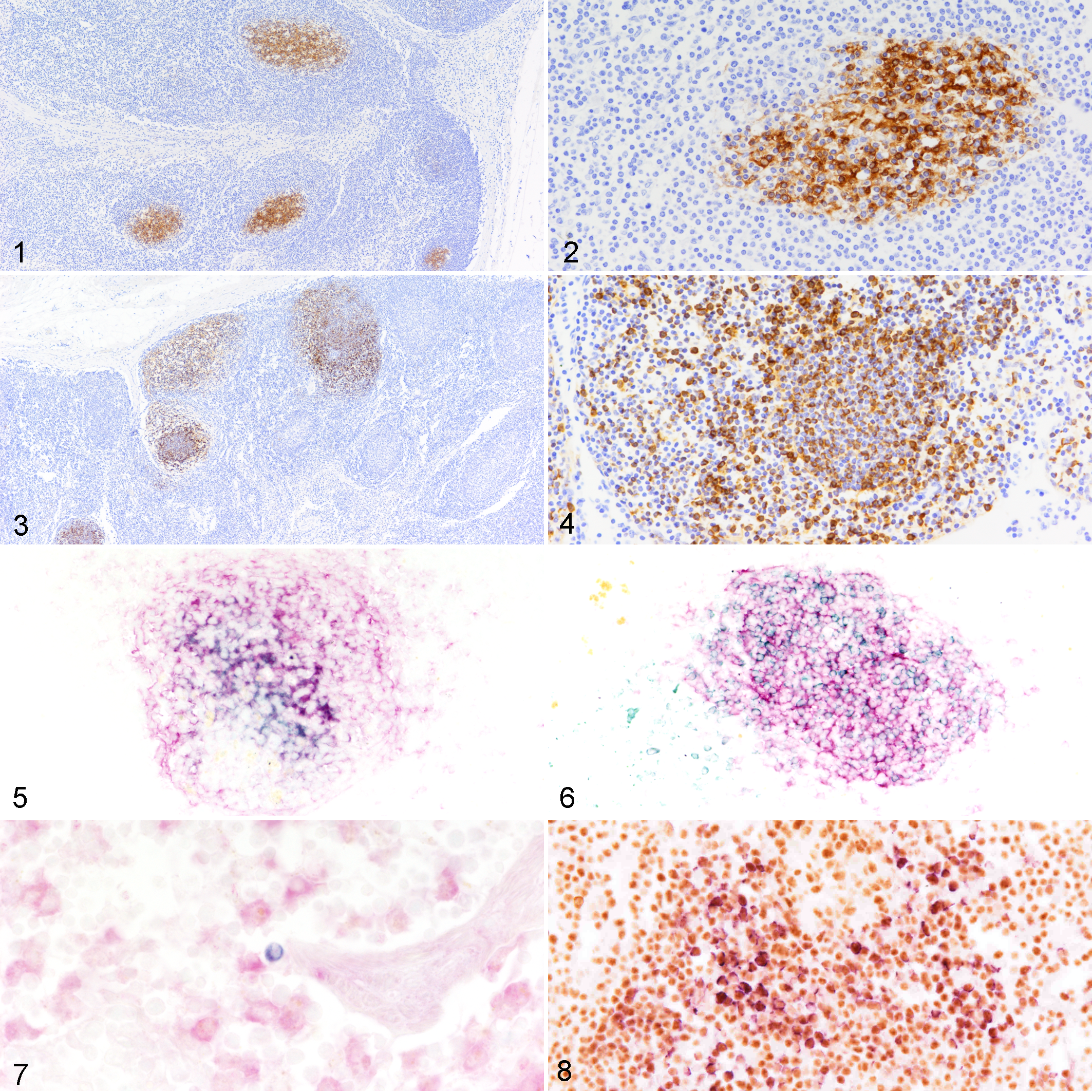
Ovine pulmonary adenocarcinoma, tracheobronchial lymph node, sheep. Several secondary cortical lymphoid follicles have positive labeling for jaagsiekte sheep retrovirus envelope protein (ENV) mostly in the center of the follicles (Fig. 1). The labeling is localized to the interstitium and to cell membranes or cytoplasm (Fig. 2). Single-label immunohistochemistry (IHC).
For ENAS, positive samples were most numerous in group G (4/8; bilateral tumors) and less frequent in group F (1/5; unilateral tumors; Table 1). The percentage of labeled CFs in ENAS was much lower than in the OPA cases, with a mean percentage of 15% of CFs labeled (Table 1). The ENV labeling pattern in ENAS cases was similar to that in the OPA cases.
In the ENAG, positive reactions were more numerous in group J (bilateral tumors) and less frequent in group I (unilateral tumors). The percentage of positive CFs in group J was 55%, comparable to the OPA group D (Table 1). In all ENAG cases, the labeling pattern of CFs was different to that described for the OPA and the ENAS cases. Most of these CFs contained high numbers of cells, mostly with lymphocyte morphology, with ENV-immunolabeled cytoplasm intermingled with negative cells of a similar morphology. These cells were not only confined to the center but also located in the marginal zone, and the periphery of the follicle (Figs. 3, 4).
In addition, isolated or small groups of round cells with ENV-immunolabeled cytoplasm were found in 5 OPA cases, mainly from group D; being mostly located in medullary cords and sinuses of the DLNs. These cells were seldom identified in ENAS or ENAG cases. No specific reaction was found in samples from any of the control groups E, H, and K.
Double-Labeling of ENV and CD4, CD8, CD21, or CD14
Single-label IHC for CD4, CD8, CD21, and CD14 showed the expected labeling characteristics and distribution (data not shown). 10,17 Double-labeling of ENV and either CD4 or CD8 were not identified. Double-labeling of ENV and CD21 was observed in all groups tested that were positive by single-label IHC for ENV (Table 1). The results were similar for OPA and ENAS cases, with the cytoplasmic or membranous double-labeling mainly in the center of the CFs (Fig. 5). For ENAG, dispersed double-labeled cells interspersed with CD21-positive ENV-negative cells, which were not confined to the center of the follicles. Single ENV-labeled cells were detected in the periphery of the follicle (Fig. 6). Double-labeling of ENV and CD21 was not detected in any other areas of the DLNs.
Double-labeling of ENV and CD14 presented technical difficulties. Only a few isolated cells located in the medullary cords from one sample of group D showed a clear double labeling pattern (Table 1, Fig. 7). No double labeling was detected in the control groups.
Double-Labeling of ENV and Pax5 or Ki-67
Single-label IHC for PaX5 and Ki-67 showed the expected distribution (data not shown). 17 With the double-label IHC method, cells labeled for either ENV or PaX5 or Ki-67 were detected, and no clearly double-labeled cells were found in any OPA or ENAS cases (Table 1). In ENAG cases, numerous cells distributed throughout the CFs were double-labeled for ENV and PaX5 or for ENV and Ki-67. These double-labeled cells were intermingled with many single-labeled Pax5-positive and few ENV-positive cells (Fig. 8). The ENV-positive cells were located throughout the CFs, in a pattern similar to that described for single-label IHC. Ki-67 did not often show double-labeling indicating that most of the cells containing ENV were not in active division. Only one ENAG case showed few clear double-labeled cells (Table 1; results not shown). Double-labeled cells were not detected in any control samples with this protocol.
Discussion
Immunohistochemical examination of the tumors and other tissues in sheep with OPA has been used to detect JSRV proteins in many previous studies and interpreted as a sign of viral expression in tumor cells and cells of the immune system. 25 The first antibody used in OPA IHC investigations was the JSRV Rab. 25 Specific staining was demonstrated principally in tumor cells, in a few large cells in the tracheobronchial and mediastinal lymph nodes and some macrophages within the pulmonary alveoli. These observations together with JSRV DNA or RNA identified in lymphoid tissues and leucocytes suggested that JSRV may replicate in a minor subset of macrophages and lymphoid cells. 13,22,25 In addition, JSRV capsid proteins were detected in similar cells, also in Peyer’s patches and mesenteric lymph nodes from lambs fed with milk and colostrum from JSRV-infected mothers. 2 As a part of this work but not included in this article, we started testing a limited number of our samples from each group of tumors and their corresponding controls with single-label IHC using JSRV Rab and compared the results with JSRV Mab. Results with both reagents were comparable but JSRV Mab revealed much clearer definition and less background staining. Therefore, JSRV Mab was considered to be the best reagent available when trying to detect colabeling with immune system cells. This comparison experiment can be used to reaffirm the presence of viral antigens in cells of the DLNs and confirm previous results, which detected positive cells in lymph nodes using JSRV Rab marker. 25 As we have tested a large numbers of samples from natural cases of contagious respiratory tumors, this study provides new information regarding the presence of ENV protein in the main DLNs from nasal cavities and lungs bearing these viral tumors in sheep and in goats. The labeling was present in CFs in a high proportion of samples from the 3 diseases. This information can be added to previous studies in an attempt to further understand the relationship of these viruses with immune cells in naturally occurring contagious respiratory tumors of sheep and goats. Thus, ENV protein was found in samples from the 3 diseases (OPA, ENAS, ENAS), either cytoplasmic or in the interstitium, indicating this is not a rare event in this group of diseases.
In general, antigens may reach DLNs via lymphatic vessels that are permeable for fluids, cells, bacteria, viruses, and small particles. Some of these elements are carried to the DLNs and are eventually captured by follicular dendritic cells (FDCs) located in cortical follicles. 12,23 Consequently, opsonized antigens smaller than 70 kDa can flow directly to FDCs via the conduit network, whereas large complexes are captured and transported by subcapsular macrophages to interact with FDCs. 12,23 However, FDCs can interact directly and retain prions and retroviral HIV particles. 12 In a similar way, released viruses or viral fragments in large amounts in tumor areas could reach the DLNs using this transport system and may be captured and retained in CFs. 8,9 As the tumors progress more viral antigens would reach these DLNs, possibly justifying why we found more positive samples when the tumors were more extensive. With the techniques used in this work we were not able to determine if viral particles directly interacted with FDCs or if they were opsonized previously. Our results only indicate that viral antigens are in the proximity of and in close relationship with FDCs.
DLNs play relevant roles in local and systemic immune reactions, including the germinal center reaction that occurs in CFs. FDCs are key cells in this process, capturing and processing antigens, interacting with immune cells, promoting B cell proliferation, and other phenomena fundamental to the generation of humoral immune responses. 18 The putative interactions of viral antigens with FDCs in germinal centers and the generation of humoral immunity seems to be absent in these infections as no specific antibodies have been detected in most studies of OPA and ENAS. 21,25 However, this may be not entirely excluded as neutralizing antibodies have been detected recently in natural and experimental ENAS. 28,29
One of the relevant findings of this article was that for ENAG cases, immunolabeling for ENV revealed an additional staining pattern compared with that observed for OPA and ENAS cases. In OPA and ENAS cases, immunolabeling for ENV was only in the center of CFs. In contrast, in ENAG cases, many individual cells exhibiting clear cytoplasmic immunolabeling for ENV were dispersed throughout the CFs and in peripheral areas of the follicle. Double labeling for ENV and CD21 revealed co-labeled cells but they followed a different distribution pattern, but this IHC for CD21 did not clarify whether they were B cells or FDCs. We used double-labeling for ENV and Pax5 to determine if the cells containing ENV protein were B cells; this showed double-labeled cells, but only with sufficient clarity in the ENAG cases. We did not find ENV and Pax5 double-labeled cells in OPA or ENAS cases, indicating that thus far we have not identified ENV protein in B cells for these diseases.
Whether this B cell proliferation is part of a germinal center reaction that results from specific interactions between FDCs and B cells is a matter of speculation. In an attempt to determine whether these B cells were proliferating cells, we tested for Ki-67 in our samples, but we could not produce reliable results as only a few cells became double-positive in one ENAG case.
In summary, these findings show that retroviral antigens generated in contagious respiratory tumors of sheep and goats reach the regional lymph nodes and may be captured and retained by FDCs. In addition, viral protein can be found in many lymphoid B cells in cases of ENAG. We did not find double labeling for ENV and Pax5 in OPA or ENAS samples, showing that ENV viral protein was only found in association with FDCs but not with B cells in OPA and ENAS.
We did not obtain evidence of viral protein in any of the T lymphocytes identified in these DLNs. In contrast, co-localization with the CD14 macrophage marker was observed in a few cells, mainly in medullary cords in OPA cases. With single-label IHC, occasional positive cells were detected in 4 of the OPA cases with widespread lesions, with highest concentration located mostly in the medullary cords. Double-labeling of ENV and T or B cell markers did not provide any reliable results. In contrast, double labeling for ENV and the ovine macrophage marker CD14 (the same antibody used in previous works) 27 identified a few double-labeled cells in one sample of an OPA case from group D. The use of other macrophage markers or techniques in future studies would provide better information for these cells in the DLNs in OPA.
One explanation for the presence of numerous macrophages in OPA DLNs may be related to an influx of these cells, which seems to be the predominant local immune response in OPA, being very numerous near the lung tumor. 27 Large numbers of macrophages are in close contact with tumor cells, which produce a large amount of fluid rich in virus and surfactant proteins. 6,8 However, the presence of viral antigens in macrophages may not represent active viral expression. Macrophages can phagocytose viral antigens, then actively migrate, transporting captured antigens to regional lymph nodes. 15 Thus, alveolar macrophages may either contain phagocytosed viruses or be infected near the tumor and migrate to the regional lymph nodes. Either of these events could be more probable in the lung and especially with extensive OPA tumors because the tumors are widespread and macrophages are numerous. The number of interactions between macrophages and the viruses would be fewer in ENAS and ENAG cases as macrophages are scarce in nasal tumors. 9 Thus, macrophages containing virus antigens could be fewer in ENAS and ENAG cases and difficult to detect with IHC.
In this study, we have shown the presence of retroviral antigens retained in CFs of DLNs in small ruminants bearing contagious respiratory tumors. Moreover, B cells containing retroviral ENV antigen was identified by double-labeling of ENV and Pax5 in ENAG cases. In addition, macrophages containing ENV were observed in 1 of 4 OPA cases tested. These findings are evidence of the presence of retroviral antigens in nontumor cells and their presence may be interpreted as a sign of viral expression in B lymphocytes in the case of ENAG but not in OPA or ENAS. However, the identification of ENV together with macrophage or CD21 (B lymphocyte/FDC marker) may be interpreted in 2 ways: either as a result of viral expression or of captured protein. To enhance our knowledge of these interactions more refined studies mainly based on molecular pathology techniques are needed to obtain confirmation of viral expression and replication in immune cells and try to explain which are the consequences of the presence of these retroviral proteins in DLNs of small ruminants affected by contagious respiratory tumors.
Footnotes
Acknowledgements
We would like to acknowledge Cristina Summers, Mercedes Jaime, Agustín Estrada, and Mike Sharp for critical review and very helpful comments; and Rosario Puyó and Santiago Becerra for their inestimable technical assistance. We also thank Dusty Miller and Massimo Palmarini for generous donation of the JSRV Mab and JSRV Rab antibodies.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by Gobierno de Aragón, Aragón, Spain, and European Social Funds, European Union (Construyendo Aragón 2016-2020), Grant Number A15-17R. Research group: Welfare and pathology of small ruminants. University of Zaragoza.
