Abstract
Avian pneumovirus (APV) is the cause of a respiratory disease of turkeys characterized by coughing, ocular and nasal discharge, and swelling of the infraorbital sinuses. Sixty turkey poults were reared in isolation conditions. At 3 weeks of age, serum samples were collected and determined to be free of antibodies against APV, avian influenza, hemorrhagic enteritis, Newcastle disease, Mycoplasma gallisepticum, Mycoplasma synoviae, Mycoplasma meleagridis, Ornithobacterium rhinotracheale, and Bordetella avium. When the poults were 4 weeks old, they were inoculated with cell culture–propagated APV (APV/Minnesota/turkey/2a/97) via the conjunctival spaces and nostrils. After inoculation, four poults were euthanatized every 2 days for 14 days, and blood, swabs, and tissues were collected. Clinical signs consisting of nasal discharge, swelling of the infraorbital sinuses, and frothy ocular discharge were evident by 2 days postinoculation (PI) and persisted until day 12 PI. Mild inflammation of the mucosa of the nasal turbinates and infraorbital sinuses was present between days 2 and 10 PI. Mild inflammatory changes were seen in tracheas of poults euthanatized between days 4 and 10 PI. Antibody to APV was detected by day 7 PI. The virus was detected in tissue preparations and swabs of nasal turbinates and infraorbital sinuses by reverse transcription polymerase chain reaction, virus isolation, and immunohistochemical staining methods between days 2 and 10 PI. Virus was detected in tracheal tissue and swabs between days 2 and 6 PI using the same methods. In this experiment, turkey poults inoculated with tissue culture-propagated APV developed clinical signs similar to those seen in field cases associated with infection with this virus.
Avian pneumovirus (APV) is a respiratory disease agent of turkeys and chickens. A disease called turkey rhinotracheitis (TRT) was first described in South Africa in the late 1970s4 and is now present in many European countries.16,24 TRT is caused by APV types A or B of the genus Metapneumovirus, which is distinguished from respiratory syncytial virus of humans, the type species for the genus Pneumovirus.23
In 1996, a highly contagious respiratory disease appeared in turkeys in Colorado, and APV was subsequently isolated at the National Veterinary Services Laboratory in Ames, Iowa.31 The Colorado isolate of APV (APV/CO) is antigenically32 and genetically29,30 different from European isolates known to cause TRT and swollen head syndrome in chickens.
Early in 1997, APV was detected serologically in turkeys in Minnesota and spread rapidly among susceptible flocks. The virus was subsequently isolated from infected turkeys displaying typical clinical signs. This isolate is nearly identical to APV/CO.29,30 Clinical signs of the disease consisted of coughing, nasal and ocular discharge, and swelling of the infraorbital sinuses. Infected flocks had high morbidity (50–100%) at all ages. Mortality rates up to 30% occurred in flocks with concurrent bacterial infections.33 Some flocks had high rates (up to 11%) of condemnation at slaughter because of airsacculitis (M. C. Kumar, personal communication).
Several studies of the pathogenesis of European isolates of APV (subtypes A and B) in turkeys and chickens have been reported,7,8,17,19 but the pathogenesis of disease caused by US isolates of APV remains unclear. In early studies using an isolate obtained from sick turkeys in Minnesota (APV/MN-2a), inoculation of 23-day-old poults produced clinical signs between days 2 and 10 postinoculation (PI).15 Antibody to APV/CO was detected by enzyme linked immunosorbent assay (ELISA), and the virus was detected in the nasal turbinates using fusion gene–based reverse transcription polymerase chain reaction (RT-PCR) and virus isolation (VI). Inoculation of 7-week-old turkeys with APV/CO produced respiratory signs between days 3 and 10 PI.26 Antibody to APV/CO was detected by ELISA, and virus was detected in the trachea by VI.
Here, we describe the results of an experimental study designed to characterize the clinical disease and pathologic changes in commercial turkeys inoculated with a Minnesota APV isolate.
Materials and Methods
Virus
The stock virus was originally isolated from the nasal turbinates of 11-week-old male turkeys with respiratory disease and was designated APV/Minnesota/turkey/2a/97 (APV/MN-2a).5,11 The stock virus used as the inoculum was prepared after four passages in Vero cell cultures5,11 of the ninth chicken embryo fibroblast passage of the isolate. When the maximum cytopathic effect (CPE) was observed in each Vero cell passage, the cell cultures were frozen, thawed, and centrifuged at 3,000 × g for 10 minutes, and the supernatant was recovered for inoculation. The presence of virus in the supernatant was confirmed by RT-PCR9 and electron microscopic examination of negatively stained preparations.12 The virus stock was titrated by inoculation of serial 10-fold dilutions of the virus preparation into Vero cell cultures.37 The titer of the virus inoculum was determined to be 106.3 tissue culture infective doses per ml (TCID50/ml).28
Experimental design
Sixty commercial Nicholas strain turkey poults were obtained from APV antibody–free parents and raised in isolation facilities. At 3 weeks of age, serum samples were collected and determined to be free of antibody against APV, avian influenza (agar gel precipitin test),34 hemorrhagic enteritis (agar gel precipitin test),27 Newcastle disease (hemagglutination inhibition test),1 Mycoplasma gallisepticum (plate test),20 Mycoplasma synoviae (plate test),20 Mycoplasma meleagridis (plate test),20 Ornithobacterium rhinotracheale (plate test),2 and Bordetella avium (ProFLOCK BA-T ELISA, Kirkegaard & Perry Laboratories, Gaithersburg, MD). At 4 weeks of age, 40 of the poults (20 male, 20 female) were inoculated with 50 µl of infected cell culture fluid instilled into each conjunctival space and nostril (total of 200 µl). Twenty poults (10 male, 10 female) were inoculated with noninfected cell culture fluid and served as sham-inoculated controls. The groups were housed in two separate isolation units. Birds were observed daily for clinical signs. On days 2, 4, 6, 8, 10, and 14 PI, four birds (2 male, 2 female) were randomly selected from the inoculated group, blood was collected, and the birds were euthanatized and examined. Two sham-inoculated control birds (1 male, 1 female) were euthanatized on the same days and treated similarly. At each time of necropsy, blood, swabs, and tissues were collected and processed. At 7 and 15 days PI, blood was collected from all remaining birds, and serum was tested for the presence of antibody to APV by ELISA.6 Between weeks 1 and 14 PI (18 weeks of age), blood was collected weekly from eight inoculated and four sham-inoculated birds and tested by ELISA for the presence of antibody to APV.
A scoring system was used to evaluate the severity of the snicking, open-mouth breathing, nasal discharge, swelling of infraorbital sinuses, frothy ocular discharge, and submandibular swelling. Each clinical sign was scored as present (1) or absent (0). A mean clinical score for each group was determined each day by adding the clinical scores for each bird and the sum was divided by the number of live birds in the group. The severity of depression was assessed visually and graded as follows: + = mild, ++ = moderate; +++ = severe.
Processing of samples
Choanal swabs were obtained from each bird by inserting a dacron swab through the choanal cleft. Each swab was placed into a separate tube containing 1 ml of veal infusion broth (VIB, Difco Laboratories, Detroit, MI) containing 100 units of penicillin G, 100 mg of streptomycin, and 0.25 mg of amphotericin B. The birds were then euthanatized with intravenously administered sodium pentobarbital (Pentobarbital Sodium-CII, Anpro Pharmaceutical, Arcadia, CA), the beak was sectioned just behind the nostrils, and the infraorbital sinuses were swabbed. The swabs were placed into separate tubes containing 1 ml of VIB.
The nasal turbinates were aseptically removed from each euthanatized poult. The nasal turbinates of the inoculated poults were pooled, minced with sterile scissors in a laminar flow hood, and placed into a sterile bag containing 5 ml of VIB. This procedure was repeated separately with the nasal turbinates of the sham-inoculated poults. The contents of the bags were homogenized in a Stomacher 80-lab blender (Seward, London, England) and then centrifuged at 3,000 × g for 10 minutes in a TH-4 Swinging Bucket Rotor (Beckman TJ-6 centrifuge), and the supernatant was collected and processed for RT-PCR and VI.
Tracheas were aseptically collected and processed in a laminar flow hood. Using sterile forceps and scissors, the tracheas were sectioned longitudinally into two halves. One half was swabbed, and the swabs placed into separate tubes containing 1 ml of VIB. The other half was placed into 10% buffered neutral formalin.
From each tube containing a choanal, sinus, or tracheal swab, 500 µl of VIB were removed to create separate choanal, sinus, and tracheal pools. The pooled samples were processed for RT-PCR and VI within 36 hours of collection.
Tracheal swabs were collected weekly from eight randomly selected live inoculated birds at weeks 2–14 PI (18 weeks of age) and pooled in a single tube containing 1 ml of VIB for assay by RT-PCR.
Histopathology
Sections of conjunctiva, nasal turbinate, infraorbital sinus wall, trachea, lungs, air sac, liver, spleen, kidney, brain, pancreas, small intestine, cecal tonsils, ceca and bursa of Fabricius from each bird were fixed in 10% buffered neutral formalin, embedded in paraffin, sectioned at 3–4 μm, and stained with hematoxylin and eosin (HE).
Bacteriology
Swabs of nasal turbinates, sinuses, trachea, lungs, and livers from each poult were cultured aerobically on tripticase soy agar containing 5% sheep blood and MacConkey's agar for 48 hours at 37 C and anaerobically by using Bio-Bag Environmental Chamber Type A (Becton Dickinson Co., Cockeysville, MD) for 48 hours at 37 C.
Serology
Serum was tested for the presence of antibody to APV by ELISA using anti-turkey IgG conjugate as the secondary antibody (Kirkegaard & Perry Laboratories).6 The geometric mean titer (GMT) for eight inoculated birds and four sham-inoculated controls was determined in serial two-fold dilutions, and end points were calculated.3
RT-PCR
RNA was extracted from the supernatants of nasal turbinate homogenates and swabs from choanal cleft, infraorbital sinus, and trachea and assayed using fusion gene–based RT-PCR. A specific PCR assay has been developed at the University of Minnesota for the detecting US APV isolates in clinical samples and has been adapted to the 5′-nuclease (TaqMan) assay format.9,10 The ability of the TaqMan assay to detect US APV nucleic acid was based on a sequence specific template through the inclusion of a fluorogenic labeled probe during PCR. The primers (2F and 2R) specifically amplified a region of 280 base pairs in the US APV.
Virus isolation in Vero cells
CPE
APV isolation was attempted in Vero cell cultures (ATCC) of tissue homogenates from nasal turbinates and swabs from choanal clefts, infraorbital sinuses, and tracheas using previously described techniques.15 The Vero cell cultures were monitored for the appearance of CPE between 24 and 72 hours of incubation. If CPE was not observed after five blind passages, the sample was considered negative.
Indirect immunofluorescence
The indirect immunoflorescent antibody (IFA) test was done according to previously described methods with a few modifications.35 Vero cell monolayers in 96-well microtiter plates were inoculated with 50 µl/well of suspensions of each Vero cell passage of tissue homogenates from nasal turbinate and swabs from choanae, infraorbital sinuses, and tracheas. A known APV-positive (APV/MN-2a) sample was included on each plate as a positive control. On each plate, wells containing uninfected cells were also prepared and served as negative controls. In addition, wells from APV-infected cells were also subjected to the same procedure, except that normal turkey serum was used in place of the hyperimmune turkey anti-APV serum and served as the negative serum control. After 24–36 hours of incubation at 37 C, the plates were fixed with cold ethanol, and the wells were filled with 50 µl/well of hyperimmune turkey anti-APV serum (1:320 dilution in Difco FA buffered saline). The hyperimmune turkey anti-APV serum was produced in isolation reared turkeys and was negative for the presence of antibody against avian influenza, Newcastle disease virus, and Bordetella avium. The plates were washed after incubation at 37 C for 45 minutes, and 50 µl of fluorescein-labeled goat anti-turkey IgG conjugate (Kirkegaard & Perry Laboratories) was added to each well. After incubation at 37 C for 30 minutes, the plates were washed, stained with 0.01% Evan's Blue for 1 minute, washed with distilled water, and examined using a fluorescence microscope (wavelength = 515 nm).
Immunohistochemical staining
An immunoperoxidase procedure14 for detection of APV antigen in tissues was performed on sections of conjunctivae, nasal turbinates, infraorbital sinuses, trachea, lungs, air sacs, cecal tonsil, and bursae of Fabricius that had been fixed in formalin for 24–36 hours. Hyperimmune rabbit anti-APV serum was used as the primary antibody and was prepared in isolation-reared rabbits that were hyperimmunized against APV. The serum was negative for the presence of antibody against avian influenza, Newcastle disease virus, and B. avium. The sandwich technique was used with biotinylated goat anti-rabbit IgG (Dako Corp., Carpinteria, CA) as the secondary antibody, followed by a streptavidin–biotin–horseradish peroxidase solution (Dako). In the last step of the procedure, an amino-ethylcarbazole-peroxide chromogen (Dako) solution was used to visualize the antigen–antibody reaction.
Statistical analyses
The differences in clinical scores between males and females were compared using the means and SEs computed with Statistix v. 1.0 software (Analytical Software, Tallahassee, FL).
Results
Clinical findings
Respiratory signs developed within 2 days PI and resolved by 12 days PI (Table 1). By 5 days PI, the poults were depressed, huddled together, and less active and exhibited a characteristic snick or sneeze. The respiratory signs consisted of open mouth breathing, snicking, discharge of clear frothy fluid from nostrils, swollen infraorbital sinuses, foamy or frothy discharge in the medial canthi of the eyes, and submandibular swelling (Fig. 1). The nasal discharge was initially clear and watery and later become thick, frothy, and cloudy. In some cases, nasal discharge was visible only after gently squeezing the nostrils. The swelling of sinuses was frequently unilateral, although both sides were occasionally involved. The medial canthi of the eyes contained foamy or frothy fluid. Submandibular swelling was present in a few poults. No clinical signs were seen in the sham-inoculated control poults. No differences in scoring of clinical signs were found between males and females (Fig. 2).

Head; turkey, 5 days after inoculation with APV. Note swelling of infraorbital sinus (arrow) and submandibular space (arrowhead).
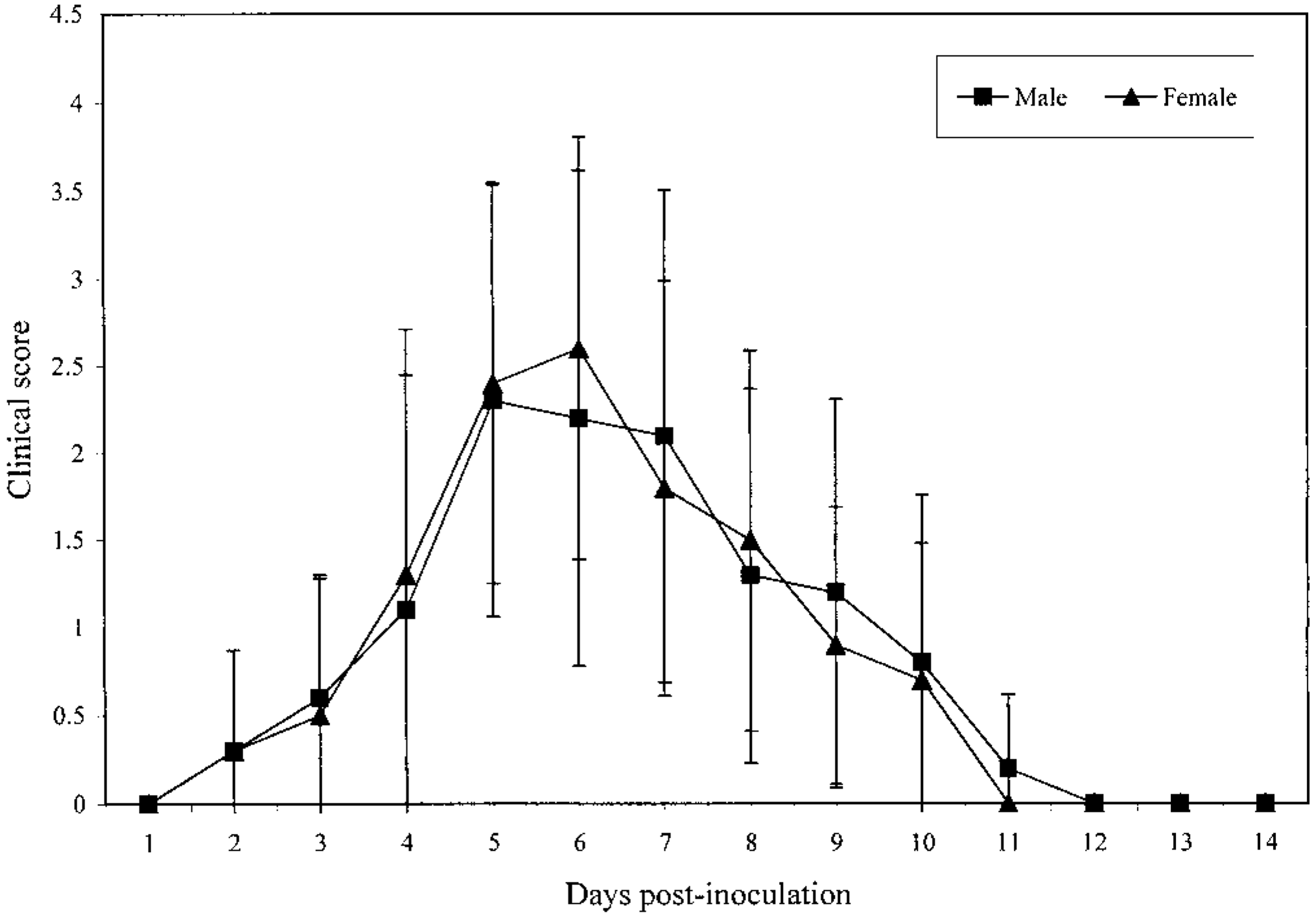
Mean (SD) clinical scores for turkeys following inoculation with APV. There were no differences in mean scores between males and females.
The percentage of birds showing clinical signs following inoculation with APV by ocular and nasal route.∗
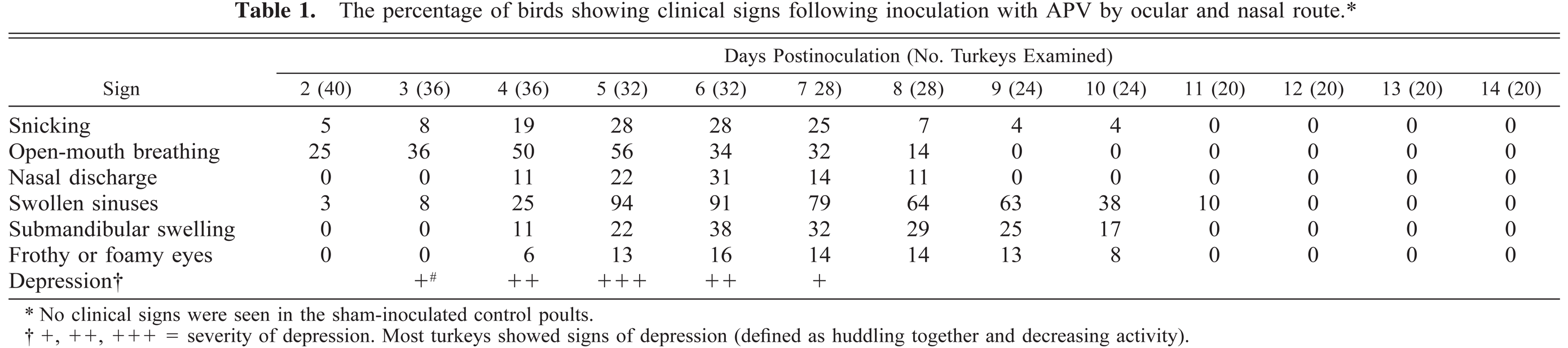
No clinical signs were seen in the sham-inoculated control poults.
+, ++, +++ = severity of depression. Most turkeys showed signs of depression (defined as huddling together and decreasing activity).
Postmortem findings
Gross lesions
Swelling of the infraorbital sinuses due to accumulation of clear frothy fluid (Fig. 3) was observed in poults euthanatized on days 4, 6, 8, and 10 PI. The nasal turbinates were slightly reddened, and the nasal cavity contained excessive frothy mucoid fluid on these days. No gross lesions were seen in sham-inoculated control birds.
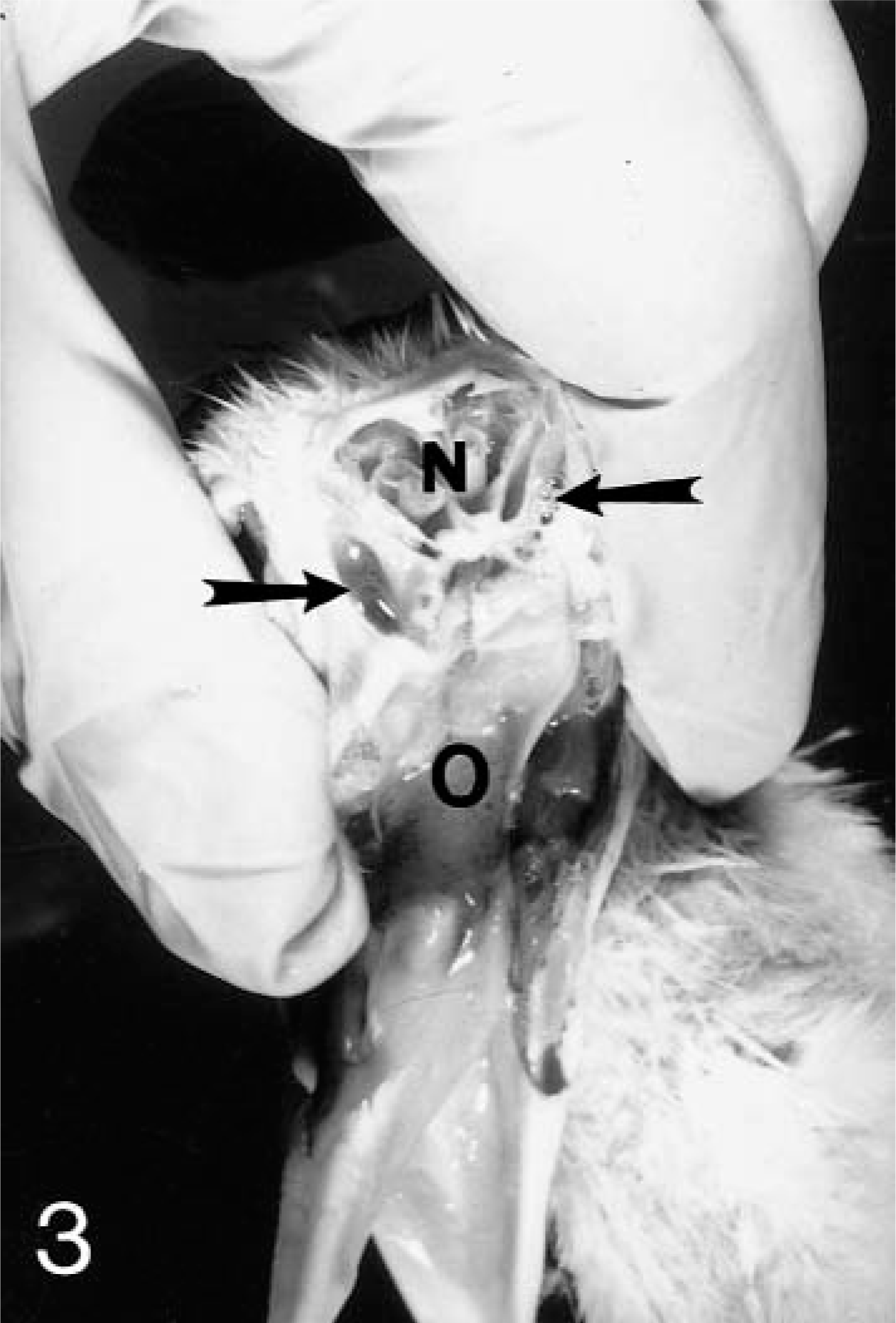
Head; turkey. Cross section of nasal cavity 4 days after inoculation with APV. Note excess mucus in the infraorbital sinuses (arrows). N = nasal cavity; O = oral cavity.
Histopathology
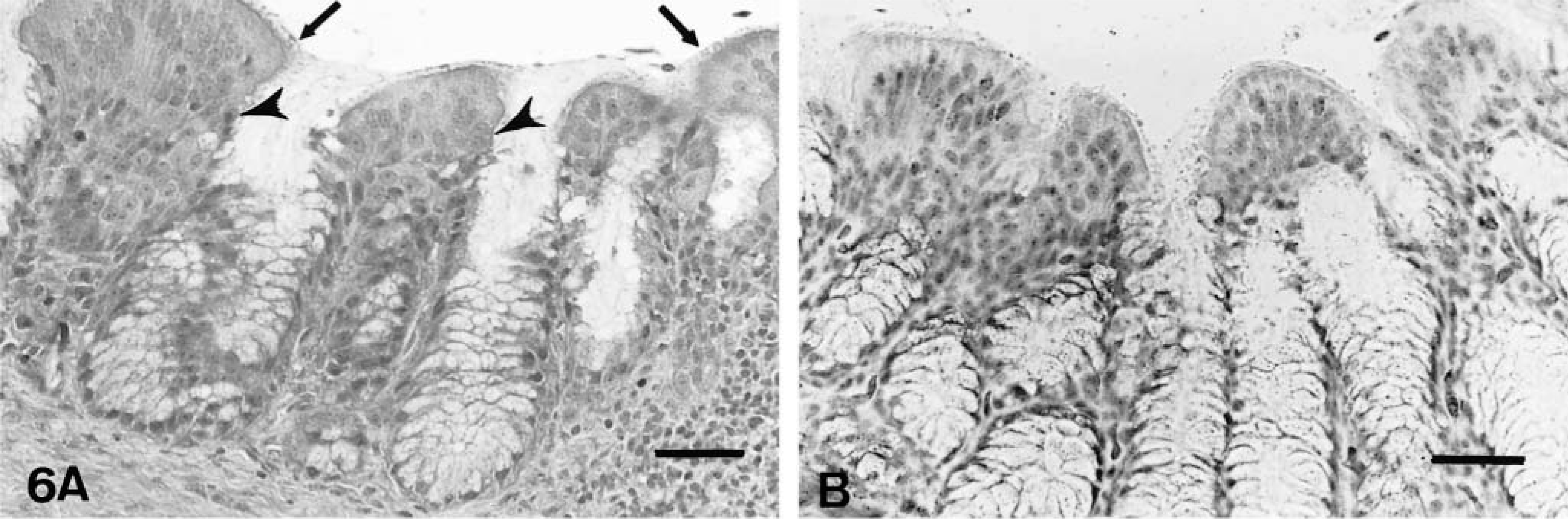
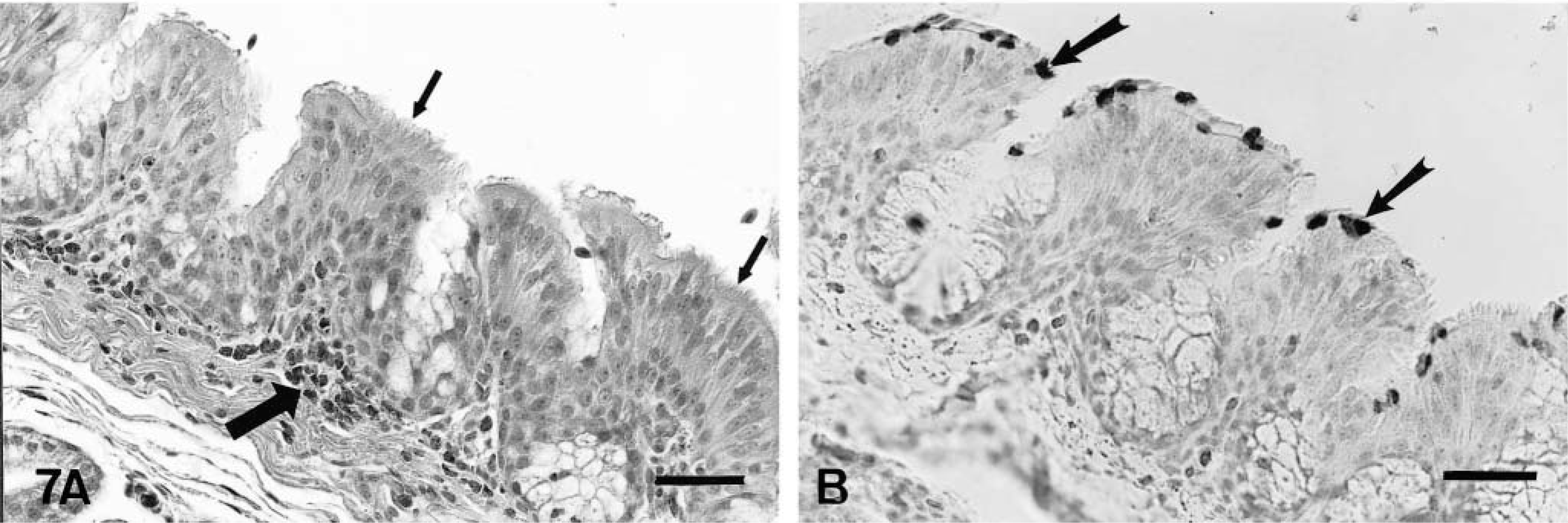
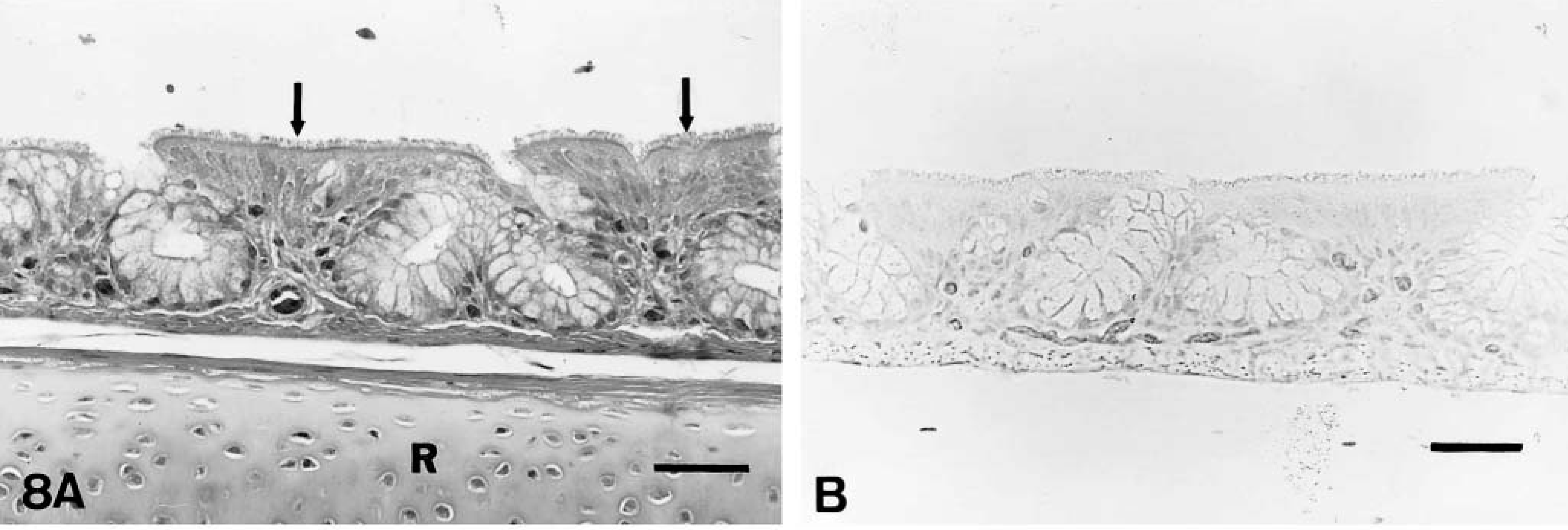
On day 2 PI, there were mildly increased numbers of lymphocytes and macrophages in the mucosa and submucosa of the nasal turbinates and infraorbital sinuses in comparison to controls. On days 4–10 PI, there was excessive mucus in the lumina of the nasal cavities. Moderately increased numbers of lymphocytes, macrophages, and plasma cells and a few heterophils infiltrated the mucosa and submucosa of the nasal turbinates (Figs. 4A, 5A) and infraorbital sinuses (Figs. 6A, 7A). By 10 days PI, the inflammatory changes had decreased in severity in the nasal turbinates and infraorbital sinuses. By 14 days PI, there were no differences between control and inoculated poults. The tracheal mucosa was infiltrated by small numbers of lymphocytes and macrophages between days 4 and 10 PI (Figs. 8A, 9A). Small numbers of heterophils were seen in two of four birds euthanatized on each of days 6, 8, and 10 PI. No significant microscopic changes were seen in sections of conjunctivae, lungs, air sacs, liver, spleen, kidney, brain, pancreas, small intestine, cecal tonsils, ceca, or bursa of Fabricius of any of the inoculated poults. No significant microscopic changes were seen in the tissues from sham-inoculated birds.
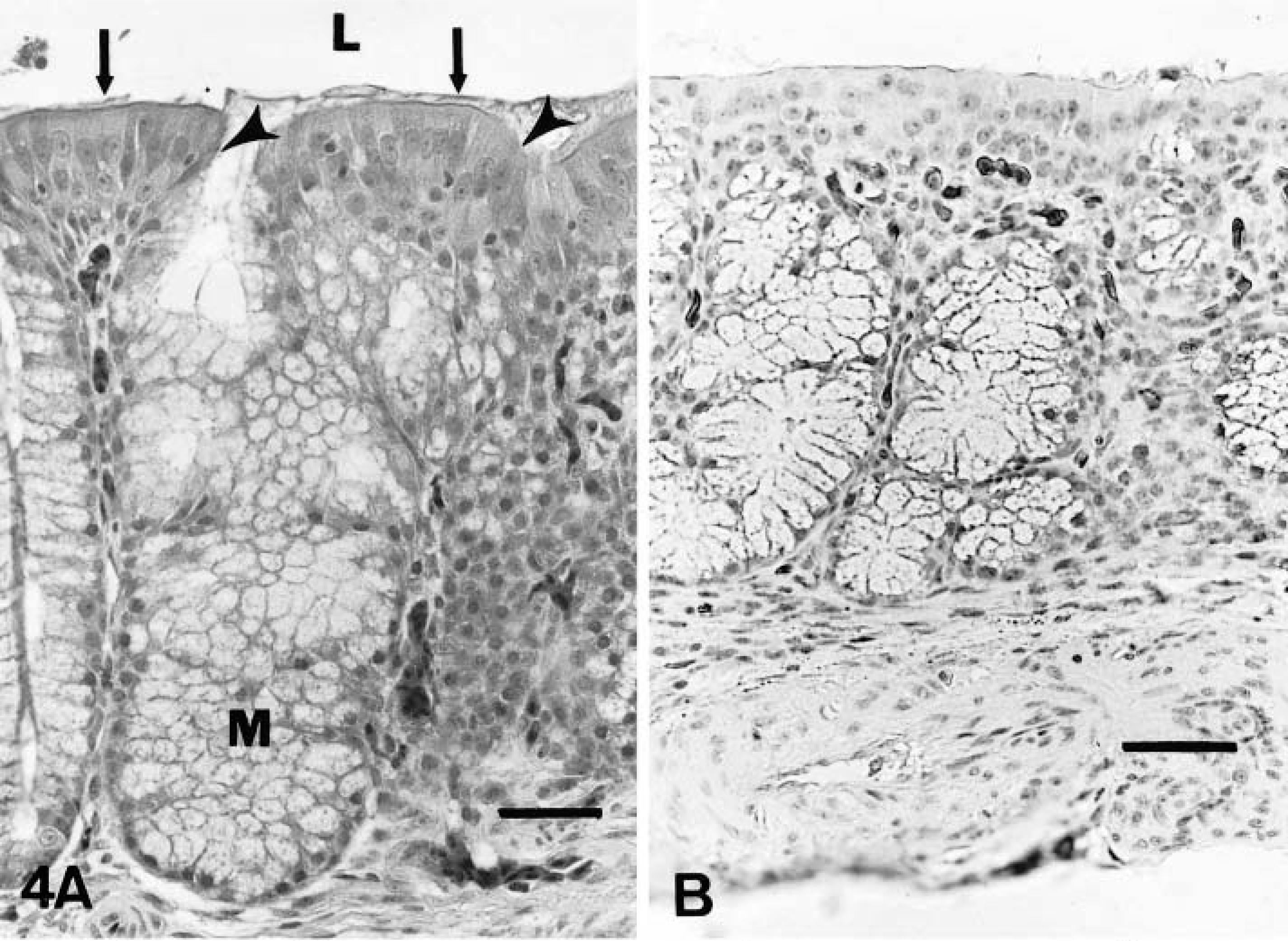
Nasal turbinate; sham-inoculated turkey, 4 days after inoculation with noninfected cell culture liquid.
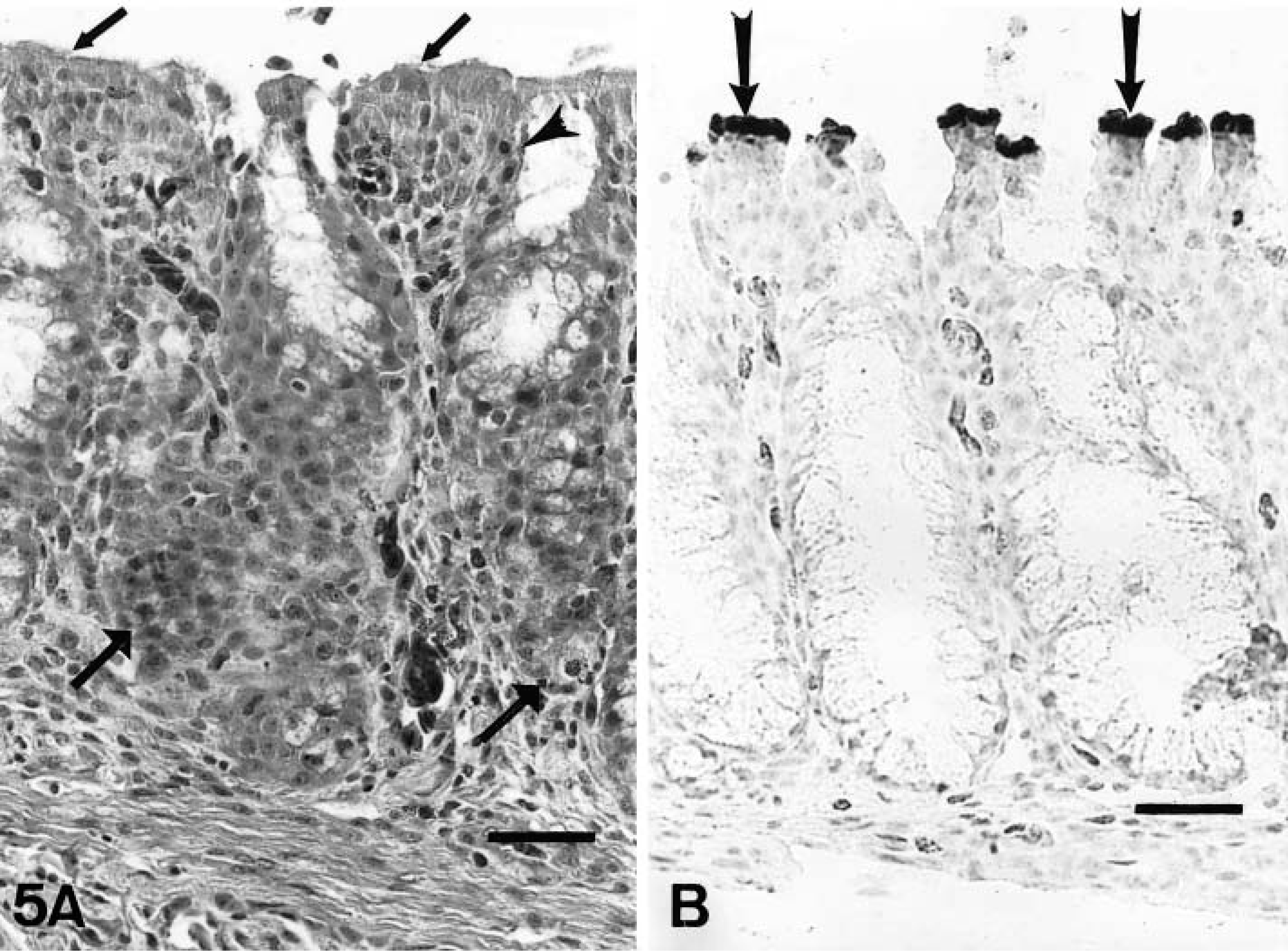
Nasal turbinate; inoculated turkey, 4 days after inoculation with infected cell culture fluid.
Infraorbital sinus; sham-inoculated turkey, 4 days after inoculation with noninfected cell culture fluid.
Infraorbital sinus; inoculated turkey, 4 days after inoculation with infected cell culture fluid.
Trachea; sham-inoculated turkey, 6 days after inoculation with noninfected cell culture fluid.
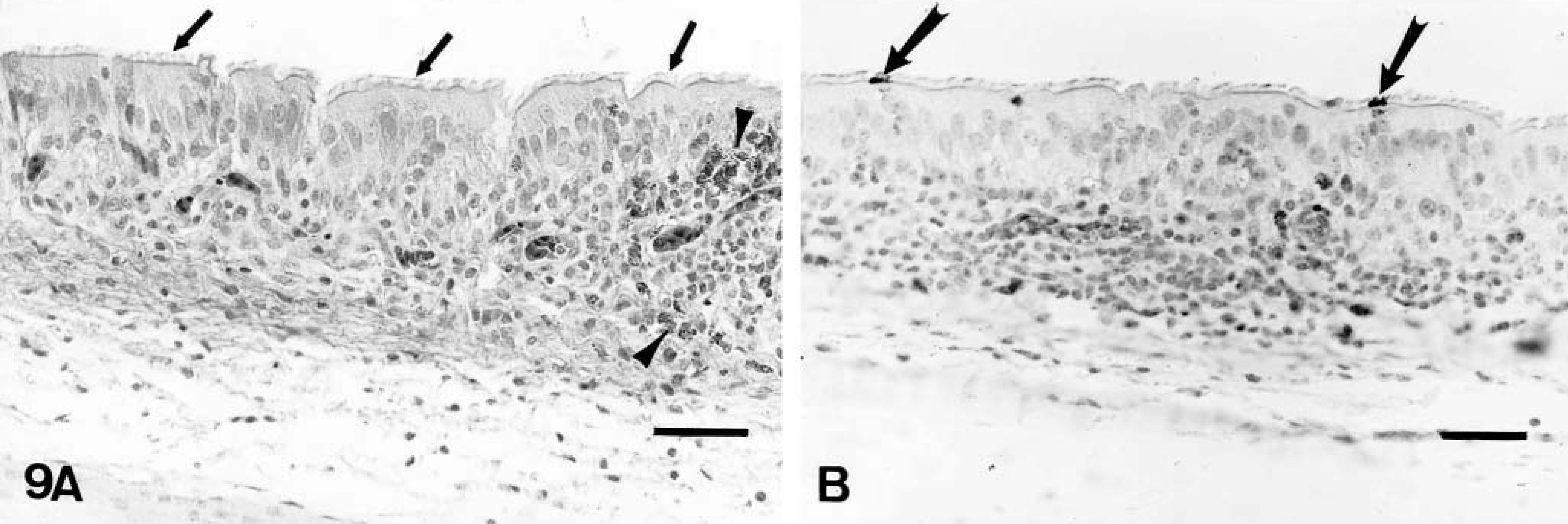
Trachea; inoculated turkey, 6 days after inoculation with infected cell culture fluid.
Bacteriology
No bacteria known to be pathogenic were isolated from the nasal turbinates, sinuses, trachea, lungs, and livers cultured aerobically and anaerobically from either APV-inoculated or sham-inoculated control birds.
Serology
No antibodies to APV were detected in serum samples of poults euthanatized on days 2, 4, and 6 PI. Antibody to APV was detected in serum samples of 21/28 birds at 7 days PI (Table 2). Antibody to APV was detected in serum samples of the poults euthanatized on days 8, 10, and 14 PI. By 15 day PI, the 16 remaining birds had antibody to APV. Antibody levels peaked between 5 and 7 weeks PI (Fig. 10). No antibodies against APV were detected in serum samples of sham-inoculated control birds.
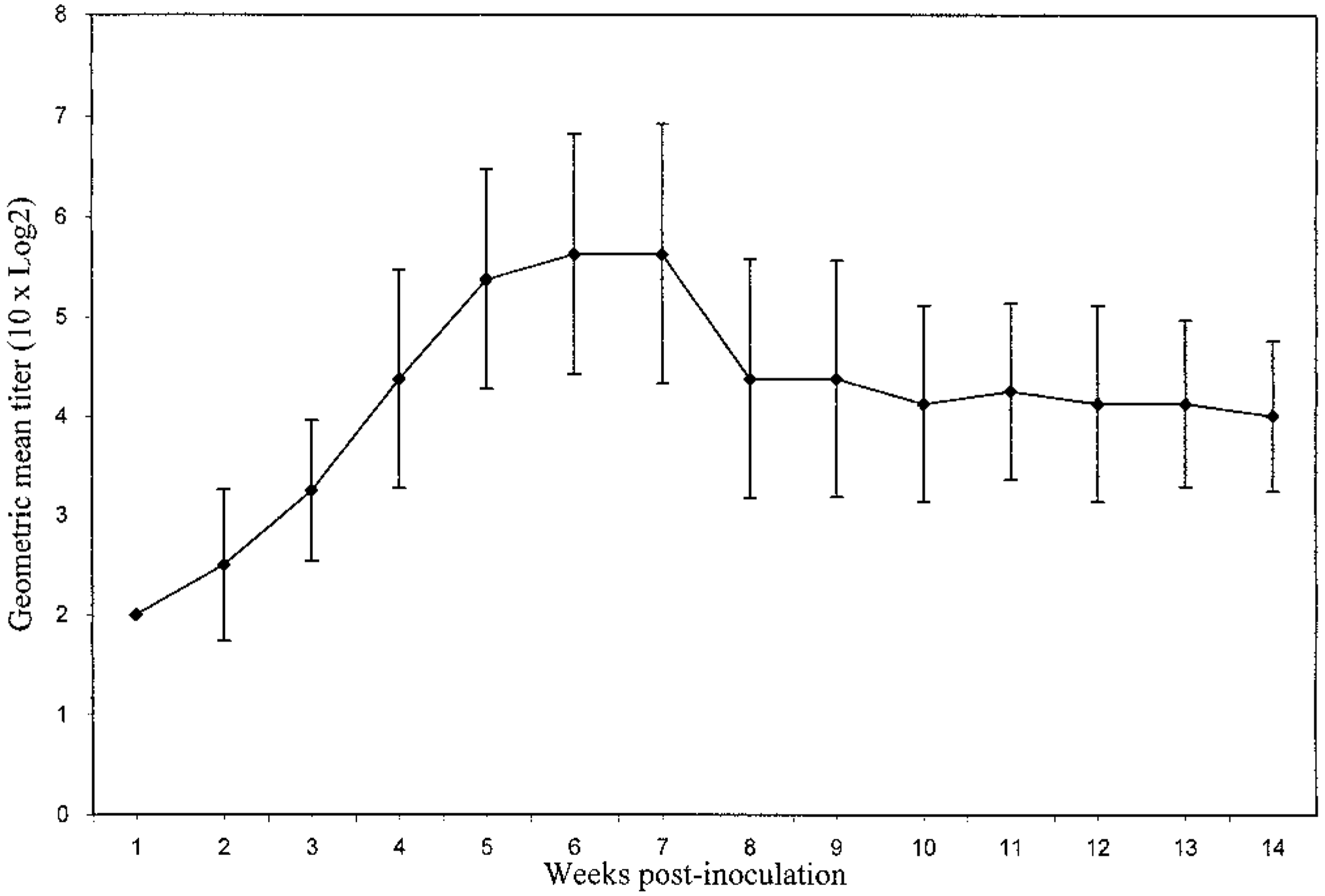
Serologic response (GMT and SD) of turkeys following inoculation with APV. No antibody against APV was detected from the sham-inoculated control birds.
Antibody response∗ detected by ELISA in APV-inoculated and sham-inoculated control turkeys.

No. birds positive/No. birds examined.
Virus detection by RT-PCR
APV RNA was detected by RT-PCR in each preparation of the nasal turbinates collected between 2 and 6 days PI (Table 3). Choanal swabs were positive for APV on days 2, 6, and 8 PI. Infraorbital sinus swabs were positive for APV at 4, 6, 8, and 10 days PI. APV RNA was detected only in tracheal swabs collected at 6 days PI. Viral RNA was not detected in the tracheal swabs of the eight birds that were monitored weekly during weeks 2–14 PI (18 weeks of age). APV was not detected in samples from inoculated poults collected at 14 days PI. No APV was detected in specimens obtained from the sham-inoculated control birds at these times.
APV detection by polymerase chain reaction (PCR), virus isolation (CPE and IFA), and immunohistochemical staining (IHC) in turkeys inoculated by ocular and nasal route.

PCR and virus isolation were performed on choanal, sinus, and tracheal swabs; IHC was performed on sinus and tracheal tissues. PI = postinoculation. † = APV detected in pooled sample; − = APV not detected in pooled sample.
No. positive/No. examined. ND = not done.
VI in Vero cells
CPE
APV was detected in Vero cell cultures by observation of typical CPE in the preparations of the nasal turbinates collected between 2 and 6 days PI (Table 3). The CPE was characterized by formation of syncytia with foci of nuclei arranged in a typical pattern. CPE was observed in the fourth or fifth passages from these sites. APV was isolated from choanal swabs on day 6 PI and from infraorbital swabs between days 4 and 6 PI. Tracheal swabs were positive for APV at 4 days PI. APV was not detected in samples from inoculated poults at 14 days PI. No CPE was observed in samples from sham-inoculated control birds.
IFA test
APV antigen was detected in Vero cell cultures using IFA staining in the preparations of the nasal turbinates collected between 2 and 6 days PI (Table 3). The presence of the virus was indicated by light green fluorescence that imparted a granular appearance to the cytoplasm of cells containing APV. Virus antigen was detected in the first or second passage from the sites. Viral antigen was detected in preparations from the choanal swabs on day 6 and in preparations from the infraorbital sinus swabs collected on days 4, 6, and 8 PI. Using the IFA test, viral antigen was also detected in tracheal swab preparations on days 2 and 4 PI. APV antigen was not detected in samples from inoculated poults at 14 days PI. No viral antigen was detected using the IFA test for samples from sham-inoculated control birds or the negative control wells.
Immunohistochemical staining
The antigen was located in the apical portions of the ciliated epithelial cells of the nasal turbinates (Fig. 5B) and infraorbital sinuses (Fig. 7B) and was closely associated with the cilia. Viral antigen was detected in the nasal turbinates and infraorbital sinuses of poults sacrificed between 2 and 8 days PI (Table 3). The presence of APV was indicated by formation of a red-brown material in infected cells. Occasionally diffuse cytoplasmic immunostaining was observed in the mucosal epithelial cells of nasal turbinates and infraorbital sinuses. Viral antigen was also seen in a few macrophages in the deep portions of the mucosae of the nasal turbinates and sinuses on days 2 and 4 PI. APV was detected in a few of the epithelial cells of the tracheal mucosa of one of the four birds euthanatized on each of days 4 and 6 PI (Fig. 9B). No viral antigen was detected in sections of conjunctiva, lungs, air sacs, cecal tonsil, and bursae of Fabricius. APV was not detected in tissues from inoculated poults at 10 and 14 days PI. APV was not detected in tissues of sham-inoculated control birds at any time (Figs. 4B, 6B, 8B).
Discussion
In this experiment, turkey poults inoculated with tissue culture–propagated APV developed clinical signs similar to but milder than those seen in field cases. The most notable clinical signs observed in the experimentally inoculated poults were nasal discharge, swelling of the infraorbital sinuses, and frothy ocular discharge between 2 and 11 days PI. The majority of birds developed swelling of infraorbital sinuses, but the other signs were variable. The swelling of the sinuses was caused by accumulation of clear mucoid fluid. Frequently, the mucoid nasal discharge became frothy and visible only after gently squeezing the nostril area. A frothy discharge from the eye was seen in some poults but always in conjunction with nasal discharge or swelling of the infraorbital sinuses. Blockage of the nares by the mucoid fluid probably caused the open mouth breathing and sneezing. Slight respiratory sounds were often audible. In a recent study of poults inoculated with APV/CO, there was also sneezing and nasal discharge observed between 3 and 10 days PI.26 Sneezing, occasional moist rales, nasal discharge, swelling of the infraorbital sinuses, and submandibular edema appeared following inoculation with European APV between 4 and 10 days PI.18,22 In results similar to those found with APV/MN, experimental inoculation of turkeys with either Colorado or European APV isolates caused milder respiratory signs than those seen under field conditions.21,26
In the present study, microscopic changes seen in the nasal turbinates, infraorbital sinuses, and trachea were milder than the inflammatory changes described in experimental infections with either APV/CO or European strains of APV. After inoculation of 7-week-old turkeys with APV/CO, the inflammatory changes seen in the nasal turbinates, sinuses, and trachea were accompanied by multifocal loss of the ciliated epithelium between 3 and 10 days PI.26 These findings contrast with the results of the present study, in which the cilia of the nasal turbinates, sinuses, and trachea remained intact. The turkeys used in the present study were raised in isolation and lacked exposure to bacterial and viral pathogens before inoculation. The source of the turkeys used in the previous studies of APV/CO was not described, and opportunistic bacterial pathogens may have been present. The effects of bacterial coinfection have not been elucidated in the USA, but coinfection with European strains of APV enhances the severity of clinical signs.7
European strains of APV primarily occur as natural infections in the epithelium of the nasal cavity and the trachea of 2–5-week-old commercial turkeys.18,36 Following experimental infection in turkeys with European strains of APV, microscopic changes were present in the mucosa of turbinates and sinuses between 3 and 10 days PI.36 On days 3–7 PI, localized epithelial hyperplasia, loss of cilia, and infiltration by mononuclear inflammatory cells in the mucosa were observed. The earliest change in the trachea observed with European APV was focal discontinuity of the surface ciliary layer at 2 days PI.18 By day 4 PI, the loss of cilia was more extensive, with subepithelial hyperemia and infiltration of intraepithelial heterophils and lymphocytes. Recovery was complete, and tracheal epithelium returned to normal by 10 days PI. In contrast, in the present study the cilia of the nasal and tracheal epithelium remained intact in the poults that were inoculated with APV/MN.
Antibody to APV was detected in the serum of most of the poults by 7 days PI and in all the poults by 15 days PI. The initial appearance of circulating APV antibodies is coincident with a decline in the ability to detect the virus using RT-PCR or VI. The severity of clinical signs also declined around the time of antibody detection. Thus, the highest risk of transmission of APV from an infected flock may occur before antibody is detectable in serum. Circulating antibody persisted in the eight birds that were monitored for 14 weeks PI (18 weeks of age). In a study using turkeys inoculated with APV/CO isolate, antibody was first detected by ELISA at 10 days PI.26 This finding was similar to results in turkeys inoculated with European APV, where a serological response was detected within 11 days PI.13 It has not been determined whether circulating antibody provides protection against APV infection; however, poults have been infected with European isolates of APV in the presence of maternal antibody.25
APV was detected only in nasal turbinates, infraorbital sinuses, and tracheas, which suggests that the virus replicates primarily in the upper respiratory tract. The virus was most consistently detected in the nasal turbinate tissues and infraorbital sinuses. The results of IHC staining correlated closely with results of the PCR and VI assays. A similar correlation of results has been reported previously between PCR and VI.15 In contrast, after inoculation of turkeys with APV/CO, the virus was isolated from tracheas on days 3, 5, and 7 PI but not from the nasal turbinates or infraorbital sinuses.26 In studies using European APV isolates, APV principally infected the upper respiratory tract (nasal cavity and trachea); only a small amount of virus was recovered from lungs and air sacs.7 8
In our current study, APV was detected sooner in Vero cell cultures by using IFA staining than by relying on visualization of CPE. The use of visualization of CPE may require four or five passages before evidence of the virus is detected, whereas the virus could be detected in cell culture as soon as the first or second passage using IFA staining. An additional advantage of IFA was that it specifically identified the virus in cell culture.
Although the experimentally inoculated birds became ill, none died or developed the severe airsacculitis often present in field cases. The greater severity of the disease in the field may be due to other factors such as concurrent infection with bacterial pathogens or the presence of adverse environmental conditions.26 Further studies are necessary to understand why there is such marked difference in severity of the disease in the field in comparison to the experimental conditions.
Footnotes
Acknowledgements
We thank Drs. E. Townsend (Department of Veterinary PathoBiology) and A. Sheikh (Department of Veterinary Diagnostic Medicine) of the University of Minnesota for their technical assistance. We also thank Dr. F. Martin (School of Statistics, University of Minnesota) for his expert statistical guidance. This research was supported in part with funds from the Minnesota Turkey Research and Promotion Council and the Rapid Agricultural Response Fund of the Minnesota Agricultural Experiment Station. This article is No. 001163402 of the Scientific Journal Series, Minnesota Agricultural Experiment Station.
