Abstract
Lymphoproliferative disease virus (LPDV) was first documented in wild turkeys in North America in 2009. LPDV infection is often subclinical but can manifest as lymphoid proliferation or round cell neoplasia. Despite high prevalence across many sampled areas corresponding to declining populations of wild turkeys, knowledge regarding LPDV pathogenesis, risk factors for disease development, and associated impacts on population dynamics are unknown. To understand transmission, viral shedding, and tissue tropism, we inoculated 21 domestic turkeys via the oral cavity, crop, nasal cavity, subcutis, or coelomic cavity. For 12 weeks, oropharyngeal swabs, cloacal swabs, and whole blood were collected weekly. At 1 week postinoculation, 3 turkeys (3/21; 14%) had detectable LPDV proviral DNA in blood by polymerase chain reaction, and 10 developed DNAemia (50%; 10/20) by 12 weeks. LPDV proviral DNA was intermittently detected in oropharyngeal and cloacal swabs. Splenomegaly was the most consistent gross finding in DNAemic birds (8/11; 73%). Lymphoid hyperplasia in the spleen was the most significant microscopic finding (9/11; 82%). Three turkeys (3/11; 27%) developed round cell neoplasia characterized by sheets of pleomorphic, round to polygonal cells in the adrenal gland, bone marrow, skin, small intestine, and/or spleen. LPDV was detected in the spleen and bone marrow from all turkeys with DNAemia and all neoplasms. Our study establishes that infection and disease with North American LPDV from wild turkeys can be experimentally reproduced in domestic turkeys, laying the groundwork for future investigations into LPDV pathogenesis, development of diagnostic techniques, and understanding the impacts of LPDV on wild turkey populations.
Lymphoproliferative disease virus (LPDV), an oncogenic retrovirus (genus Alpharetrovirus, subfamily Orthoretrovirinae, family Retroviridae), was first identified in 1972 in multiple flocks of domestic turkeys (Meleagris gallopavo domestica) in the UK. 7 Birds developed disease at 10- to 18-weeks-old, manifesting as pleomorphic, lymphocytic infiltrates in multiple organs. 7 Subsequently, LPDV-associated disease was sporadically diagnosed in domestic turkey flocks in Israel 19 and Europe, with up to 25% flock mortality. 16 LPDV was not documented in the USA until 2009, when it was detected in a wild turkey (Meleagris gallopavo) from Arkansas with multicentric lymphoma. 5 Since this initial detection in North America, LPDV infection has been documented in wild turkeys across much of the USA and Canada, including 47% polymerase chain reaction (PCR) detection across the eastern USA (ranging from 26% to 83% by state);4,36,37,41 65% in Ontario, Canada; 23 and 35% in Manitoba, Canada. 22
Wild turkeys are large gallinaceous birds found throughout the continental USA, Hawaii, southern Canada, and northern Mexico. 26 Habitat destruction and overhunting led to dramatic population declines in the early 20th century, prompting dedicated restoration of habitat and reestablishment of flocks via translocation. 26 Wild turkey population numbers peaked in 2004 at approximately 6.6–6.9 million birds in the USA and Canada, 28 but through continuous monitoring, state wildlife agencies and other stakeholders have documented concerning population declines14,40,43 and decreased productivity indices in some areas 9 over the last decade, most notably in portions of the eastern and midwestern USA. While researchers are currently investigating factors that may affect nest productivity, such as brood movement 11 and prescribed fire,45,46 wildlife managers are increasingly concerned about the potential role of disease, 24 in particular LPDV, on population dynamics. Furthermore, recent evidence suggests that LPDV infection has a significant negative effect on clutch size. 35
Despite widespread detection of LPDV and recognition of its pathogenicity in wild turkeys, LPDV transmission and pathogenesis remain poorly understood. In part, this is attributable to the current inability to isolate the virus in cell culture5,25 from PCR-positive birds, limiting interpretation of surveillance and diagnostic data and making experimental studies challenging. There is demand for additional diagnostic capabilities for LPDV (eg, virus isolation, serology, immunohistochemistry), and more thorough assessment of the potential health risks of LPDV on declining wild turkey populations is needed. Thus, the goal of our study was to determine LPDV susceptibility, infection, and disease in domestic turkeys and whether they could potentially serve as an in vivo model system for naturally occurring disease in wild turkeys. Specifically, we aimed to (1) compare the susceptibility to LPDV infection following various inoculation routes; (2) evaluate the extent and duration of proviral DNA detection in whole blood, oropharyngeal swabs, and cloacal swabs; (3) document gross pathology and histopathology in LPDV-infected and noninfected individuals; (4) determine the tissue distribution and tropism of LPDV; and (5) evaluate for evidence of horizontal transmission among co-housed birds.
Materials and Methods
Animal Origin and Husbandry
Twenty-five, 1-day-old, broad-breasted white turkeys (12 female, 13 male) were obtained from a commercial breeder and housed in a single biosafety level 2 (BSL-2) enclosure at the University of Georgia. Turkey poults were initially given free access to water and commercial pelleted feed via gravity water dispensers and trays, respectively. After 4 weeks, water and feed were transitioned to automatic water dispensers and gravity feeders. All birds were observed twice a day for at least 30 minutes. At 4 weeks old, poults were banded, weighed, and divided into 2 enclosures via a random sampling method. Husbandry and sampling procedures were approved by the University of Georgia Institutional Animal Care and Use Committee (A2019 05-027-Y1-A0).
Inoculum Preparation
Fresh samples of liver and bone marrow collected from 3 different wild turkey carcasses, each with a unique case number, were used for inocula, designated as A, B, and C (Table 1). Wild turkeys were either necropsied in the field or submitted for diagnostic evaluation at the Southeastern Cooperative Wildlife Disease Study. Sample selection for inoculum was based on (1) PCR detection of LPDV (and lack of reticuloendothelial virus [REV], retested and confirmed just prior to inoculation) and (2) sufficient available tissue quantity. Tissues were stored at −20°C during diagnostic case work-up (approximately 2 months), after which they were transferred to −80°C. Four inocula were prepared: 3 inocula (A, B, and C) each composed of 1 tissue from 1 unique wild turkey and 1 pooled inoculum (pooled inoculum). Each of these 4 inocula were then prepared at both 10% and 20% weight/volume concentration (8 inocula in total). To prepare inocula A, B, and C, each tissue was separately macerated in phosphate-buffered saline (PBS; Thermo Fisher Scientific, Waltham, MA) containing 1× antibiotic-antimycotic solution (100 units penicillin, 100 units streptomycin, 0.25 µg amphotericin B; Thermo Fisher Scientific). Tubes were centrifuged for 2 minutes at 2236g, vortexed for 3 seconds, and rested on crushed ice. The pooled inoculum consisted of 10 ml of inocula A, B, and C, which were pooled in 1 tube, centrifuged for 2 minutes at 2236g, vortexed for 3 seconds, and kept on ice. For the pooled 10% solution, the supernatant of inoculum C rather than the final vortexed solution was added.
Name and source of inocula used to experimentally infect domestic turkeys with lymphoproliferative disease virus.
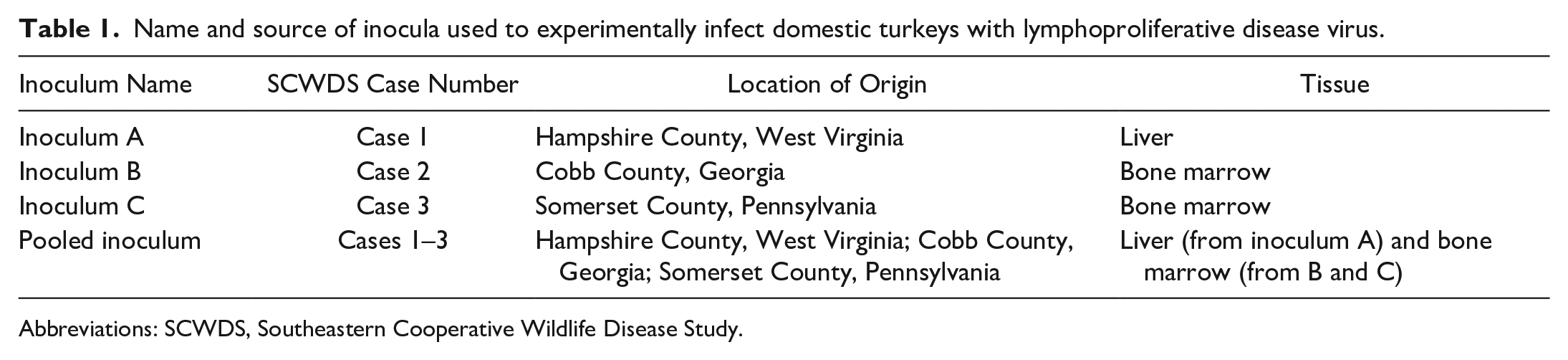
Abbreviations: SCWDS, Southeastern Cooperative Wildlife Disease Study.
Inoculation and Experimental Design
Whole blood collected from all 4-week-old turkeys was tested for LPDV and REV via PCR prior to inoculation to ensure birds were not infected with either virus. At 5 weeks of age, all poults were inoculated with the same volume, which included LPDV inoculum for experimental inoculates and PBS for sham inoculates.
The 20% weight/volume concentration inoculum was utilized in the oral cavity, crop, and intraperitoneal inoculation groups, and the 10% weight/volume concentration inoculum was used for the intranasal and subcutaneous routes to facilitate administration. Specifically, inoculation methods were as follows for each of the different routes. For the upper alimentary tract inoculation groups (n = 3 for oral cavity; n = 3 for crop), 1.0 ml of inoculum was deposited either in the oral cavity or crop using a 3-ml syringe attached to an oral gavage needle. For the intranasal group (n = 5), 0.5 ml of inoculum was gradually deposited at the caudal edge of each naris so that the turkey inhaled the 1.0 ml of inoculum voluntarily. For the intracoelomic inoculation group (n = 5), 1.0 ml of inoculum was injected into the coelom at the caudal edge of the keel bone using a 20-gauge needle attached to a 3-ml syringe. For the subcutaneous inoculation group (n = 5), 1.0 ml of inoculum was subcutaneously injected over the right pectoral muscle with a 25-gauge needle attached to a 3-ml syringe. Birds inoculated in the oral cavity, crop, or intranasally were co-housed in enclosure 1, and turkeys inoculated intracoelomically or subcutaneously were co-housed in enclosure 2 (Table 2). Two sham-inoculated turkeys were each housed in enclosure 1 and enclosure 2.
Routes of inoculation and inocula concentrations used to experimentally infect domestic turkeys with lymphoproliferative disease virus.
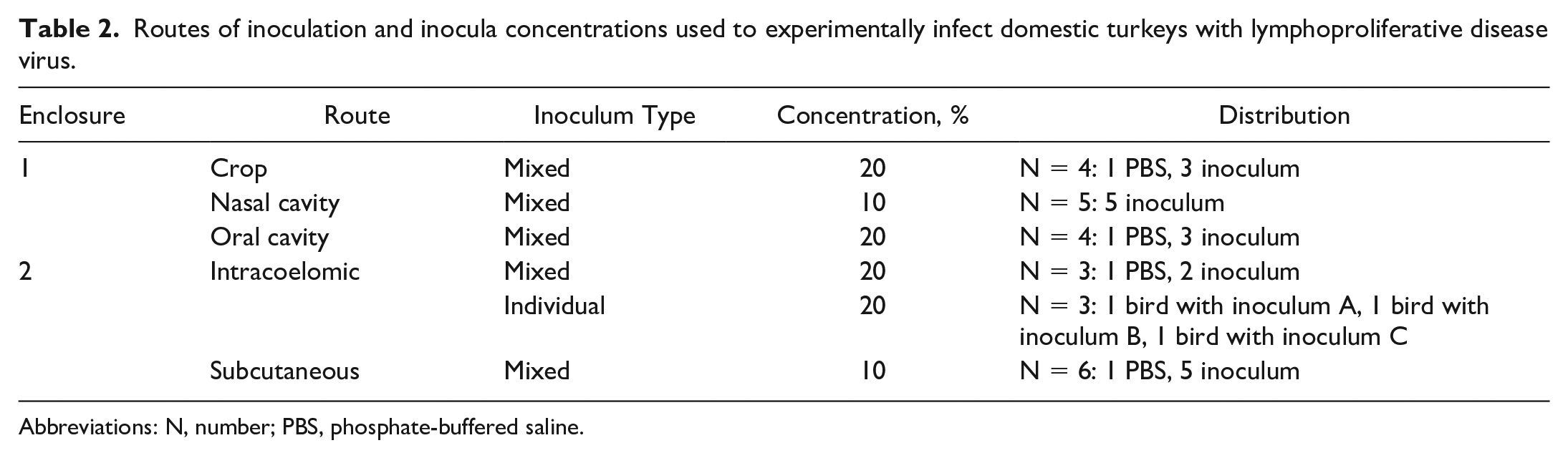
Abbreviations: N, number; PBS, phosphate-buffered saline.
Oropharyngeal swabs, cloacal swabs, and 0.5 ml of blood (collected via the jugular vein) were collected from each bird weekly. Blood was deposited in BD microtainer capillary blood collector tubes with EDTA (ethylenediaminetetraacetic acid; Becton, Dickinson and Company, Franklin Lakes, NJ). Sterile cotton-tipped swabs (Puritan, Guilford, ME) used for oropharyngeal and cloaca sampling were individually placed in 1 ml of minimum essential media (Thermo Fisher Scientific) supplemented with 1× antibiotic-antimycotic solution. At the experiment termination at 84 days postinoculation (DPI), up to 3 ml of blood was collected immediately prior to euthanasia; 0.5 ml was deposited in tubes with EDTA, and the remaining blood was placed in BD microtainer serum separator tubes (Becton, Dickinson and Company). All samples were kept on ice packs during collection and transportation. Blood collected in EDTA tubes was transferred to cryovials after transportation, then stored at either room temperature or 4°C for up to 8 hours before processing. Blood collected in serum separator tubes were centrifuged at 559 g for 15 minutes at room temperature after transportation; serum samples were transferred to cryovials and stored at −80°C. All remaining samples were stored at −80°C until processing.
Necropsy
At 84 DPI, or earlier if poults exhibited clinical signs to warrant euthanasia, birds were intravenously administered 1 ml /4.5 kg pentobarbital sodium and phenytoin sodium (Euthasol; Virbac, Fort Worth, TX) via the jugular vein. All carcasses were either immediately necropsied or held at 4°C and necropsied within 12 hours. A standardized necropsy procedure was followed, including routine tissue collections. Samples of brain, eyes, heart, thyroid gland, thymus (if present), crop, esophagus, trachea, spleen, kidney, adrenal gland, reproductive organs, skin (breast and neck), skeletal muscle (pectoral and medial thigh muscle), sciatic nerves, bone marrow, liver, proventriculus, ventriculus, duodenum, pancreas, jejunum, ileocecocolic junction, cloaca, cloacal bursa (if present), and visible tumors were fixed in 10% neutral-buffered formalin. Fresh samples of brain, heart, lung, liver, spleen, kidney, small intestine, large intestine, skin, bone marrow, and visible tumors were stored at −80°C. Gonads from select turkeys in the subcutaneous and intracoelomic inoculation groups were collected fresh and stored at −80°C.
Histopathology and Immunohistochemistry
Formalin-fixed tissues were trimmed and routinely processed prior to embedding in paraffin, sectioned at 5 µm, and mounted on glass slides. Slides were stained with hematoxylin and eosin for histologic examination.
Select tissues were subjected to CD3 immunohistochemical (IHC) labeling (1:1000, A05452; Dako, Carpinteria, CA) based on a previously published protocol. 34 Domestic dog lymph node was utilized as a positive control tissue and tissues without lymphocytic infiltration from the examined individual served as internal negative control tissues.
Nucleic Acid Extraction and PCR
Nucleic acid extraction from blood and select fresh tissues (liver, spleen, kidney, bone marrow, gonads [when available], and visible tumor) was performed using DNeasy Blood & Tissue Kits (Qiagen, Valencia, CA) following the manufacturer’s protocols. Nucleic acids were extracted from additional tissues based on clinical judgment. Oropharyngeal and cloacal swabs were homogenized with a mixer mill (21.5 cycles/second) for 1 minute. Nucleic acids from the swabs were extracted using a QIAamp Viral RNA Kit (Qiagen, Valencia, CA) per manufacturer’s protocol. Both extraction kits co-purify DNA and RNA.
LPDV primers were based on the prototypical Israeli strain of LPDV, covering a 431 bp segment of the p31 and CA genes (LPDV-F 5′-ATGAGGACTTGTTAGATTGGTTAC-3′, LPD V-R 5′-TGATGGCGTCAGGGCTATTTG-3′). 5 REV primers covered a 291 bp segment of the long terminal repeat (REV-F 5′-CATACTGGAGCCAATGGTT-3, REV-R 5′-AATGTTG TACCGAAGTACT-3′). 12 LPDV and REV PCRs were performed utilizing the GoTaq Flexi DNA polymerase system (Promega, Madison, WI), as published. 2
PCR products were visualized on 2% agarose gels stained with ethidium bromide (BioRad Laboratories, Hercules, CA) in Tris acetate-EDTA buffer solution (Sigma-Aldrich, St. Louis, MO). Extracted nucleic acids from LPDV or REV PCR-positive, naturally infected wild turkeys served as positive controls and nuclease-free water (Promega) was used as a negative control in each PCR run.
Results
LPDV Proviral DNA Detection in Blood (DNAemia) and Tissue Tropism
Overall, LPDV was detected by PCR in blood samples of 11 turkeys prior to euthanasia (52%; 11/21). DNAemia was detected at the earliest postinoculation sampling time point (7 DPI) and persisted throughout the entire experiment in some birds (Supplemental Table S1). LPDV was first detected in whole blood at 7 DPI in 3 turkeys (14%; 3/21 inoculates), all within the subcutaneous inoculation group (60%; 3/5). Peak PCR-positivity prevalence among inoculates occurred at 42 DPI (50%; 10/20 [1 inoculated bird was euthanized by this time point]) and fluctuated from 14% (3/21) to 50% (10/20) PCR-positivity until experiment termination. All sham-inoculated, contact-control birds remained LPDV PCR-negative throughout the experiment. Six turkeys (55%; 6/11) with consistent DNAemia (LPDV proviral DNA detected over multiple time points) had at least one sampling point in which they were LPDV PCR-negative, but resumed DNAemia the next sampling date. A summary of DPI and prevalence at initial detection, DPI of peak prevalence, peak prevalence, and prevalence at study termination in blood by inoculation group is included in Table 3.
Summary of lymphoproliferative disease virus (LPDV) proviral DNA detection in blood from experimentally infected domestic turkeys.
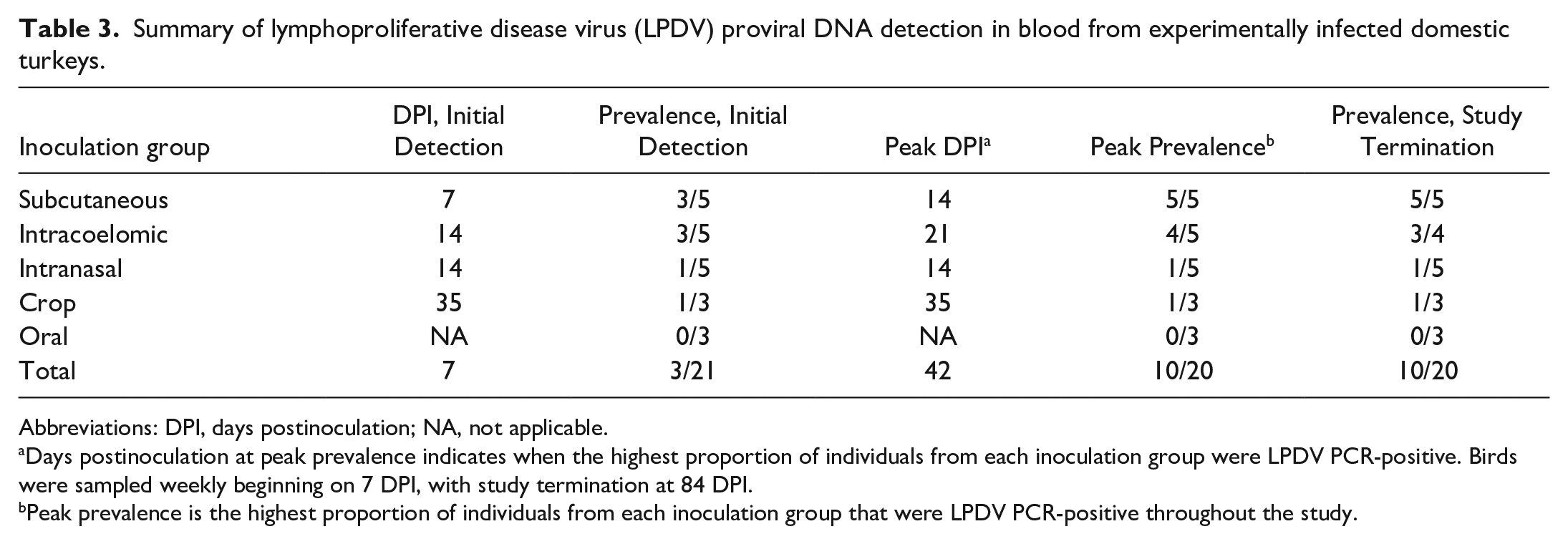
Abbreviations: DPI, days postinoculation; NA, not applicable.
Days postinoculation at peak prevalence indicates when the highest proportion of individuals from each inoculation group were LPDV PCR-positive. Birds were sampled weekly beginning on 7 DPI, with study termination at 84 DPI.
Peak prevalence is the highest proportion of individuals from each inoculation group that were LPDV PCR-positive throughout the study.
All turkeys with LPDV DNAemia at euthanasia (52%; 11/21) had detectable LPDV DNA in the spleen and bone marrow samples (100%; 11/11). Kidney and liver samples were LPDV PCR-positive in 10/11 (91%) and 9/11 (82%) DNAemic poults, respectively. Gonads from select DNAemic birds were tested and all had detectable LPDV DNA (100%; 8/8). Among the 2 individuals that developed grossly visible tumors, all affected tissues (spleen, duodenum, and jejunum) had detectable LPDV proviral DNA. The pancreas from the turkey with grossly evident neoplasia in the duodenum also was PCR-positive. No tissues tested PCR-positive from poults without DNAemia at euthanasia. No LPDV proviral DNA was detected in tissues from sham-inoculated, contact-controls.
LPDV Proviral DNA Detection in Oropharyngeal and Cloacal Swabs
LPDV proviral DNA detection in oropharyngeal swabs was intermittent among infected turkeys (Supplemental Table S2). The earliest time point in which turkeys (33%; 7/21) had detectable LPDV proviral DNA was at 28 DPI; 4 turkeys were in the intracoelomic group and 3 in the subcutaneous inoculation group. Peak PCR-positivity prevalence by oropharyngeal swab occurred at 84 DPI (40%; 8/20) and varied from 0% (0/21) to 40% (8/20) until the study endpoint. A summary of DPI and prevalence at initial detection, DPI of peak prevalence, peak prevalence, and prevalence at study termination in oropharyngeal swabs by inoculation group is included in Table 4.
Summary of lymphoproliferative disease virus (LPDV) proviral DNA detection in choanal swabs from experimentally infected domestic turkeys.
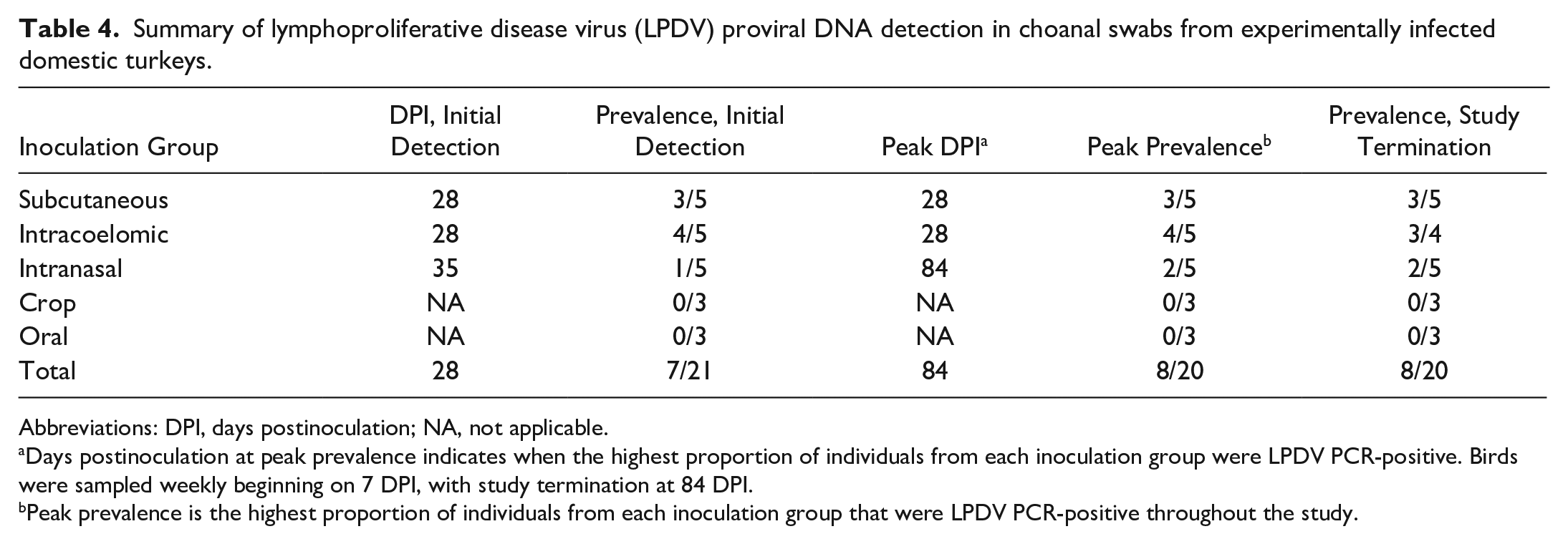
Abbreviations: DPI, days postinoculation; NA, not applicable.
Days postinoculation at peak prevalence indicates when the highest proportion of individuals from each inoculation group were LPDV PCR-positive. Birds were sampled weekly beginning on 7 DPI, with study termination at 84 DPI.
Peak prevalence is the highest proportion of individuals from each inoculation group that were LPDV PCR-positive throughout the study.
Similar to the oropharyngeal swabs, cloacal swabs were intermittently LPDV PCR-positive (Supplemental Table S3). The earliest time point in which a turkey (5%; 1/21) had detectable LPDV proviral DNA in cloacal swabs was 14 DPI; this turkey was in the subcutaneously inoculated group. Peak PCR-positivity prevalence by cloacal swab among inoculated turkeys occurred at 84 DPI (40%; 8/20) and varied from 0% (0/21) to 40% (8/20) until the study endpoint. There was no evidence of LPDV proviral DNA in oropharyngeal and cloacal secretions of sham-inoculated, contact-controls. A summary of DPI and prevalence at initial detection, DPI of peak prevalence, peak prevalence, and prevalence at study termination in cloacal swabs by inoculation group is included in Table 5. A summary of LPDV DNA detection profiles of individual turkeys in blood, oropharyngeal swabs, and cloacal swabs at the beginning, middle, and end of the experiment is included in the Supplemental Materials (Supplemental Table S4).
Summary of lymphoproliferative disease virus (LPDV) proviral DNA detection in cloacal swabs from experimentally infected domestic turkeys.
Abbreviations: DPI, days postinoculation; NA, not applicable.
Days postinoculation at peak prevalence indicates the highest proportion of individuals from each inoculation group were LPDV PCR-positive. Birds were sampled weekly beginning on 7 DPI, with study termination at 84 DPI.
Peak prevalence is the highest proportion of individuals from each inoculation group that were LPDV PCR-positive throughout the study.
Clinical Outcomes
Throughout the experiment, 24/25 turkeys (96%) were bright, alert, and demonstrated typical behavior. Regardless of experimental group or infection status, each bird steadily gained weight each week. On 32 DPI, a poult that was intracoelomically inoculated (turkey 22) exhibited edema, subtle ecchymoses, and firm nodules in the soft tissues of the neck and ventral head. On 33 DPI, there were no changes in the head and neck lesions, and turkey 22 was still preening and drinking water. On 34 DPI, turkey 22 was slow to react to stimuli and was not observed to eat or drink water; the head and neck ecchymoses had become progressively darker and more extensive. Because there was no improvement in appetite and attitude, turkey 22 was euthanized on 35 DPI. Also, turkey 22 lost weight in the week prior to euthanasia. Turkey 22 was LPDV-positive with round cell neoplasia in the skin, spleen, and bone marrow. After sampling on 63 DPI, a sham crop–inoculated turkey (turkey 15) and a sham intracoelomically inoculated turkey (turkey 3) both exhibited non-weightbearing lameness attributed to a husbandry-related injury, warranting euthanasia. Thereafter, perching structures considered hazardous based on potential leg injuries from jumping from them to the ground were removed.
Gross Pathology
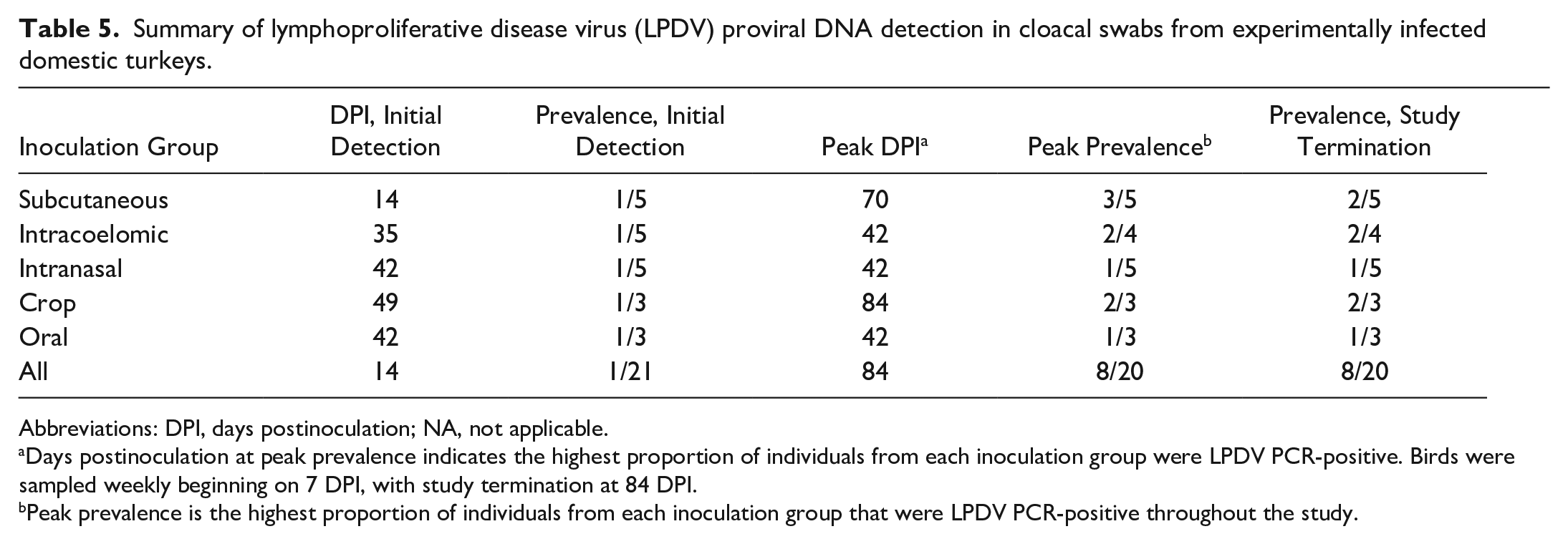
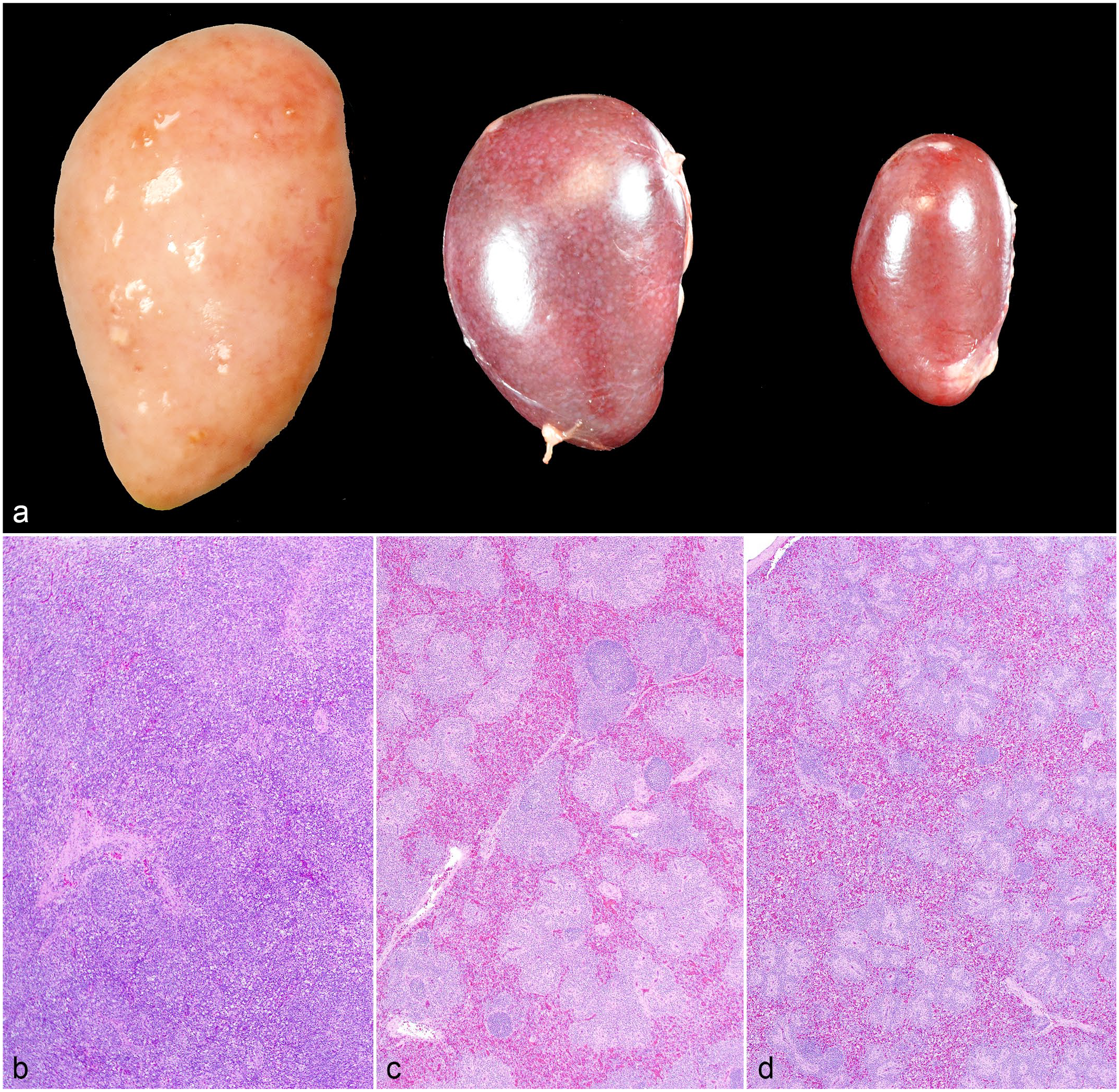
Splenomegaly was the most consistent gross finding in turkeys with detectable LPDV proviral DNA in blood at euthanasia (8/11; 73%), with 5 spleens (5/11; 45%) exhibiting grossly prominent lymphoid follicles (Fig. 1a). The spleen from turkey 22, which was euthanized on 35 DPI, was diffusely pale tan and severely enlarged, weighing 10.9 g (0.8% body weight) (Fig. 1a). Spleens from DNAemic birds weighed on average 8.24 g (range = 2.7–10.9 g), comprising approximately 0.21% of body weight (range = 0.08%–0.80%). The mean splenic weight in all turkeys without detectable LPDV proviral DNA was 4.76 g (range = 0.8–8.2 g), comprising approximately 0.07% of body weight (range = 0.03%–0.10%). The mean splenic weight from sham-inoculated turkeys was 1.98 g (range = 0.8–4.3 g), comprising 0.04% of body weight (range = 0.03%–0.06%) (Supplemental Table S5). The firm, sessile nodules along the neck observed perimortem in turkey 22 were confined to the dermis. Hemorrhage and edema extensively expanded the dermis and subcutis of the neck and intermandibular space (Fig. 2a). Multifocal to locally extensive, sharply demarcated, purple to green ecchymoses were distributed in the skin of both tibiotarsi and pectoral muscles of turkey 22 (Figs. 2b, c). One bird (turkey 2; subcutaneously inoculated) had multiple distinct, white, transmural nodules in the duodenum and jejunum (Fig. 2g). Turkeys 15 and 3 had both sustained left femoral fractures, determined to be traumatic injuries related to husbandry. There were otherwise no significant gross lesions in sham-inoculated, contact-controls.
Splenic lesions associated with lymphoproliferative disease virus (LPDV) infection, domestic turkeys. (a) From left to right: round cell neoplasm in turkey 22 (LPDV polymerase chain reaction [PCR]-positive), lymphoid hyperplasia in turkey 12 (LPDV PCR-positive), and a normal spleen from sham-inoculated turkey 23 (LPDV PCR-negative). The spleen of turkey 22 was severely enlarged and diffusely pale tan, consistent with a round cell neoplasm. The spleen of turkey 12 was moderately enlarged with multifocal, tan foci compared with the sham-inoculated turkey 23. (b) Round cell neoplasm, turkey 22. Sheets of neoplastic round cells efface splenic architecture. Hematoxylin and eosin (HE). (c) Lymphoid hyperplasia, turkey 12. Periarteriolar and perisinusoidal lymphoid sheaths are thickened and occasionally coalesce with enlarged lymphoid follicles. HE. (d) Normal spleen, turkey 23. The sham-inoculated turkey exhibited normal splenic architecture and lymphoid distribution and quantity. HE.
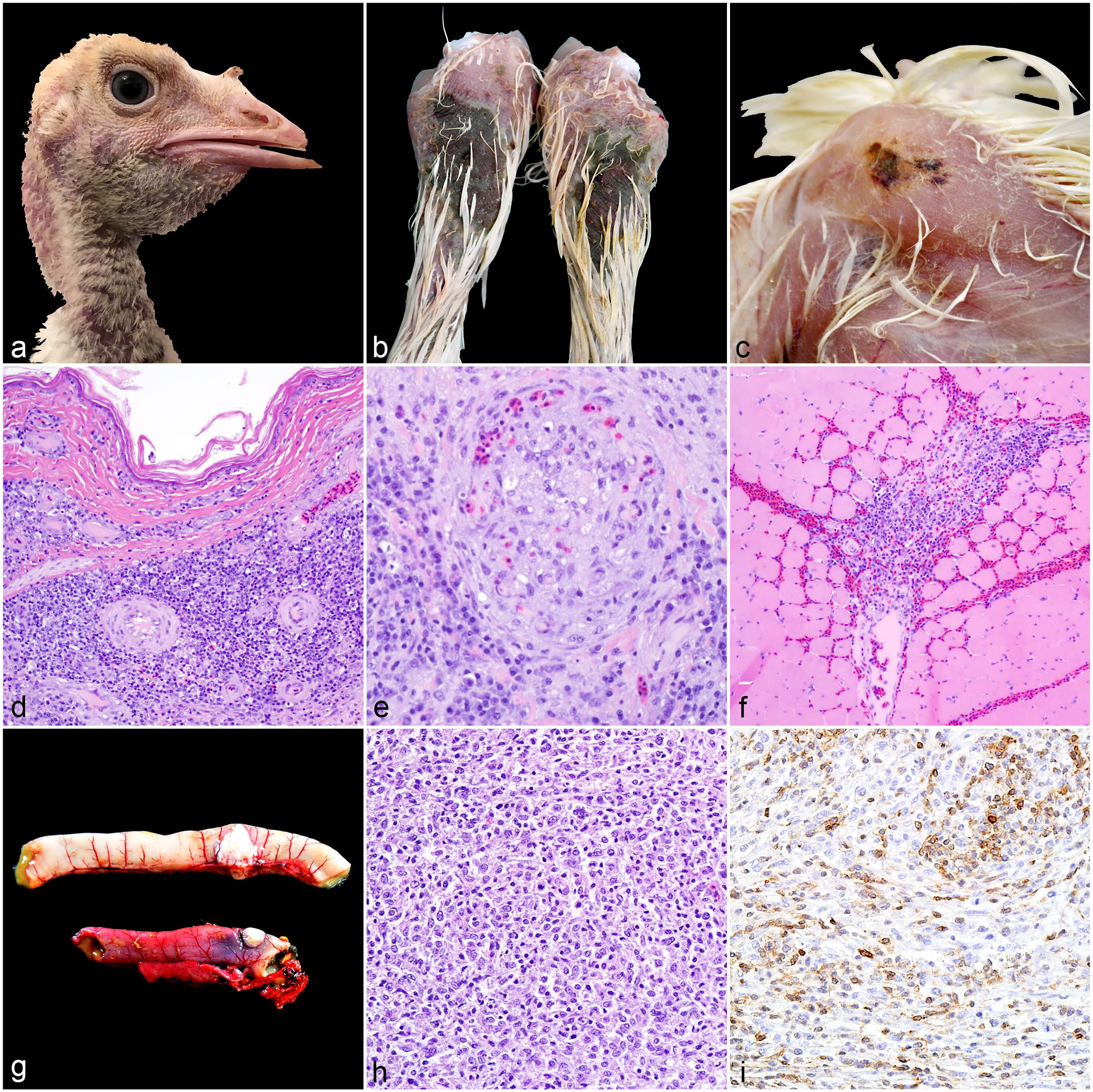
Round cell neoplasia associated with lymphoproliferative disease virus (LPDV) infection, domestic turkeys. (a) Dermal nodules, neck. Locally extensive areas of hemorrhage and edema were associated with dermal nodules in the neck of turkey 22 (LPDV polymerase chain reaction [PCR]-positive). (b) Infarcts, tibiotarsi. Sharply demarcated areas of hemorrhage in the skin covered the tibiotarsi of turkey 22. (c) Hemorrhage, pectoral muscle. Purpuric hemorrhages extended into the skeletal muscle of turkey 22. (d) Round cell neoplasm, skin. Dense sheets of neoplastic round cells aggregate around dermal blood vessels in the neck skin of turkey 22. Hematoxylin and eosin (HE). (e) Neoplastic thrombus, skin. A thrombus composed of pleomorphic neoplastic round cells occludes a dermal blood vessel in the tibiotarsal skin of turkey 22. HE. (f) Round cell neoplasm, pectoral muscle. Neoplastic round cells infiltrate areas of hemorrhage in the pectoral muscle of turkey 22. HE. (g) Round cell neoplasia, jejunum (top) and duodenum (bottom). Sharply demarcated white nodules arise from the walls of the intestines of turkey 2 (LPDV PCR-positive). (h) Round cell neoplasm, jejunum. Pleomorphic neoplastic round cells form sheets within the pre-existing stroma of turkey 2’s jejunum. HE. (i) Round cell neoplasm, jejunum. Approximately 30% of neoplastic round cells exhibit strong membranous immunolabeling against CD3 antibodies. CD3 immunohistochemistry.
Histopathology
All turkeys, regardless of inoculation route, inoculum formula, and postinoculation PCR status, exhibited baseline levels of lymphoid aggregates in numerous organs, ranging from perivascular lymphocytic cuffs to discrete lymphoid follicles (Supplemental Tables S6–S7). However, these aggregates varied in distribution and extent among birds with and those without detectable LPDV proviral DNA at euthanasia. The spleen had the most distinguishing lesions between the LPDV PCR-positive and LPDV PCR-negative sham-inoculated turkeys. Nine of the 11 (82%) LPDV-positive birds exhibited moderate to severe lymphoid hyperplasia, demonstrated by thickened periarteriolar and periellipoidal lymphoid sheaths and enlarged lymphoid follicles (Fig. 1c). Of the remaining LPDV PCR-positive turkeys, 1/11 (9%) exhibited lymphoid atrophy (ie, the depletion of periarteriolar lymphoid sheaths and lymphoid follicles) and 1/11 (9%) had a round cell neoplasm (Fig. 1b). One of the 4 (25%) LPDV PCR-negative sham-inoculated turkeys had severe lymphoid hyperplasia in the spleen, despite no other microscopic evidence of disease and lack of LPDV detection in the spleen, other tissues, blood, oral swabs, and cloacal swabs by PCR. The cause of this turkey’s lymphoid hyperplasia is unknown. Although the thymus and cloacal bursa did not have striking differences between the LPDV PCR-positive turkeys and the LPDV PCR-negative sham-inoculated turkeys, 2/11 (18%) of the LPDV PCR-positive turkeys had severe atrophy of the thymus (ie, shrunken lobules with very few remaining lymphocytes, very thin to indistinct cortex) and 2/11 (18%) displayed mild to severe atrophy of the cloacal bursa (ie, small lymphoid follicles, little to no distinguishable cortex in lymphoid follicles, increased stroma between lymphoid follicles, irregular folding of surface epithelium of the plicae). One turkey (turkey 22) developed lymphoid atrophy of the thymus and cloacal bursa concurrent with grossly apparent splenic neoplasia. Three birds (2 subcutaneously inoculated [turkeys 2 and 9], 1 intracoelomically inoculated [turkey 22]) had round cell neoplasia in the adrenal gland, bone marrow, skin, duodenum, jejunum, and/or spleen. Neoplasia were pleomorphic within tissues and among individuals but were characterized by sheets of round to polygonal cells with variably distinct margins, a small to moderate amount of eosinophilic cytoplasm, and a round to oval nucleus with finely stippled chromatin and occasionally one prominent nucleolus (Fig. 2). Anisocytosis and anisokaryosis were moderate (ie, 2- to 3-fold change in size), with intermittent binucleation. The average mitotic count was 22 per 2.4 mm2 (equivalent to 10 FN22/40X fields). Approximately 30%–40% of neoplastic lymphocytes in the small intestine demonstrated strong, intramembranous CD3 immunoreactivity, suggesting a T-cell origin (Fig. 2i). The grossly evident ecchymoses and edema in the skin, subcutis, and muscle of turkey 22 were associated with sheets of neoplastic lymphocytes surrounding blood vessels and few neoplastic thrombi (Figs. 2d–f). Some segments of skin with neoplasia exhibited areas of coagulative necrosis, consistent with infarction. Similar to the small intestine, approximately 60% of neoplastic round cells exhibited strong, intramembranous CD3 immunoreactivity, but immunoreactivity in neoplastic cells with more polygonal morphology was rare.
Incidental microscopic findings not attributed to LPDV infection included hepatic lipidosis in most turkeys, affecting both LPDV inoculates and sham inoculates (15/25; 60%), mucosal hyperplasia of the crop of one subcutaneously inoculated bird (4%; 1/25), erosive ingluvitis in an intracoelomically inoculated bird (4%; 1/25), mural esophagitis in an intracoelomically inoculated bird (4%; 1/25), ulcerative ventriculitis in an intracoelomically inoculated bird (4%; 1/25), plasmacytic enteritis in an intracoelomically inoculated bird (4%; 1/25), and focal bronchopneumonia in an intranasally inoculated bird (4%; 1/25). There were no significant microscopic lesions in sham-inoculated, contact-controls.
Discussion
Detection of LPDV proviral DNA in multiple tissues and blood, paired with the development of lymphoid neoplasia in several birds, constitutes successful experimental reproduction of LPDV infection and disease in domestic turkeys. In our study, the inocula were derived from naturally infected, wild turkey tissue homogenates as compared with previous experimental transmission studies, which utilized tissue homogenates of spleen from naturally occurring or experimentally induced LPDV in domestic turkeys, 25 or pooled plasma 33 or serum 47 from naturally infected domestic turkeys. To date, no methods for propagation of LPDV in vitro are available (eg, in primary or immortalized cell lines 25 or embryonated chicken or turkey eggs 5 ), thus making experimental infection trials challenging. Induction of infection and disease in domestic turkeys in our study verified the infectivity of our inocula, which can be subsequently used for experimental trials in wild turkeys as well as contribute to the potential development of LPDV isolation systems and additional diagnostic methodologies.
Turkeys inoculated via the subcutaneous and intracoelomic routes had the earliest and most sustained DNAemia, and these were the only routes that led to the development of round cell (lymphoid) neoplasia. Subcutaneous inoculation is a feasible natural route of infection in wild turkeys, potentially through entry via superficial skin wounds. Biting arthropods, such as mosquitoes, also may be a means of subcutaneous inoculation of LPDV since this has been documented for REV, an avian retrovirus similar to LPDV that is known to infect domestic and wild turkeys.29,38,39 Only 1 of the 5 intranasally inoculated turkeys became infected, and this turkey was consistently LPDV PCR-positive in the blood and in multiple tissues, as well as sporadically positive in oropharyngeal and cloacal swabs. Oculonasal transmission of avian leukosis virus (ALV), another avian alpharetrovirus, has been documented, 30 but it is unclear why intranasal LPDV inoculation was a less successful route of infection. Only 1 crop-inoculated turkey (1/3; 33%) developed DNAemia, which was intermittent, as was shedding in cloacal swabs. One orally inoculated bird had rare detection of LPDV in cloacal swabs. Fecal-oral transmission of other avian retroviruses occurs, 30 so it is unclear whether the lack of infected turkeys via the crop was due to different infective properties and cell tropism of LPDV or other factors.
Of the antemortem samples tested for LPDV, blood was the most consistently PCR-positive (ie, vs oropharyngeal and cloacal swabs) and therefore is likely the most useful sample type for live bird surveillance purposes. In previous experiments, reported viremia was detected as early as 2 weeks postinoculation in domestic turkeys and lasted for up to 10 months. 25 This likely reflects LPDV utilization of circulating lymphocytes for replication, similar to other avian retroviruses. 39 Viremia also may represent a mechanism for virus transmission to wild turkeys by means of an arthropod vector imbibing a blood meal. In our study, approximately half (55%, 6/11) of the turkeys with consistent DNAemia were LPDV PCR-negative for at least one subsequent timepoint but returned to being LPDV PCR-positive in subsequent sample collection dates. Due to the lack of infection of the sham-inoculated, contact-control turkeys, we hypothesize that these intermittent negative results among inoculated birds represented continued infections for which detections intermittently dropped below the PCR detection threshold rather than re-infections. In our study, among turkeys that had PCR-positive blood, most if not all of their tissues also tested positive for LPDV. Most importantly, neoplastic tissues were consistently PCR-positive, supporting the hypothesis that LPDV likely initiates and/or contributes to disease development. Of the select organs tested, spleen and bone marrow were most consistently PCR-positive in DNAemic birds. Numerous surveillance efforts support this finding5,22,23,41 and bone marrow typically is the preferred postmortem diagnostic sample for LPDV testing.3,36,41 Bone marrow was the first organ infected in previous studies, followed by the thymus, spleen, and cloacal bursa. 15 In addition, primary lymphoid organs had the highest level of viral RNA expression, but low levels were detected in nonlymphoid organs, including the pancreas and brain. 15 In our study, the pancreas of the turkey with the grossly visible intestinal neoplasm also tested PCR-positive. However, there was no microscopic evidence of neoplasia in the pancreas; therefore, we suspect that this PCR result represents detection of infected lymphocytes within the blood. Kidney was PCR-positive in all but one turkey with DNAemia, which could contribute to LPDV detection in cloacal swabs. Liver was positive in most DNAemic turkeys, which may correspond to lymphocytic foci observed microscopically and/or represent virus in blood circulation. All tested gonads from DNAemic turkeys were PCR-positive, suggesting that consideration of vertical LPDV transmission is warranted. True vertical and/or congenital transmission via egg albumen occurs with both ALV and REV infections.30,39 Venereal transmission of ALV and REV has been demonstrated under experimental conditions,21,32,39 but there are limited data investigating its importance compared with other routes of infection. Understanding the potential effects of LPDV infection on reproduction is particularly crucial due to regional wild turkey population declines.15,40 Multiple surveys demonstrated disproportionate percentages of turkey hens infected with LPDV, which could have adverse impacts on population sustainability and growth.4,37 Multiple surveillance studies have detected LPDV more frequently in adult versus juvenile wild turkeys,5,22,23,41 which compounds theoretical vertical transmission. Sampling bias likely contributes to demographic biases, as adult turkeys are likely disproportionately targeted for hunter harvest. In addition, diseased juveniles may not be as readily submitted for diagnostic testing because carcasses are more easily lost to the landscape or predation.
Horizontal transmission from inoculated birds to sham-inoculated, contact-controls was not demonstrated in our study. In a previous experiment, 17.5% of noninoculated domestic turkeys housed in close contact with inoculated turkeys developed gross and microscopic lesions consistent with LPDV. 25 Horizontal transmission also has been documented with other avian retroviruses, as ALV is shed in feces, saliva, and desquamated skin, and REV is shed in feces and cloacal secretions.30,39 Poor shedding rates determined by low detection of LPDV proviral DNA in cloacal and oropharyngeal swabs may explain the lack of horizontal transmission observed in contact-control birds despite being co-housed with infected individuals in our study. Future studies aimed at quantitating viral RNA loads in cloacal and oropharyngeal swabs may also help delineate the amount of live virus being shed in feces or saliva. ALV does not survive for long periods outside the host and can be inactivated with sustained high temperatures similar to conditions in the enclosure. 39 Interestingly, REV can be recovered from litter material; 39 this cannot be ruled out for LPDV, as environmental sample testing was not included in this or previous studies. Despite our intermittent detection of LPDV in cloacal swabs throughout the 12-week study period, a recent study reported no significant difference in LPDV DNA prevalence between whole blood (76%) and cloacal swabs (73%) collected from live-captured wild turkeys. 36 Varied swab storage conditions, such as collection and storage of dry swabs (as done in the previously cited study), 36 versus collection into virus media (as done in our study) could account for some detection differences (ie, potential dilution of viral DNA and artificially lower LPDV detection in swabs collected into virus media).
The absence of clinical disease in the majority of infected turkeys in our study, which went to 12-weeks postinoculation, is consistent with prior experimental findings and surveys of apparently healthy captured wild turkeys 3 and hunter-harvested wild turkey carcasses. 41 Previous studies of experimentally infected domestic turkeys in Europe and the Middle East reported 2% mortality during a 6-week observation period 47 and up to 30% mortality when the observation period was extended to 9 weeks. 25 In another study, approximately 10% (4/39) of naturally LPDV-infected wild turkeys examined had neoplastic lesions that contributed to mortality. 5 During an outbreak of naturally infected domestic turkeys in the UK, clinical disease was uncommon, but acutely manifested as anorexia, lethargy, and ruffled feathers. 7 LPDV-associated disease was observed in 1 turkey euthanized at 5 weeks postinoculation in this study. In contrast to studies of LPDV in naturally infected wild turkeys,5,13,29 the most striking gross lesion in our study was cutaneous infarction associated with round cell neoplasia. Skin lesions, ranging from hyperkeratosis and acanthosis to nodular growths, are among the most commonly observed lesions associated with LPDV-associated lymphoma in naturally infected wild turkeys.5,13,29 Gross hemorrhage in the pectoral and thigh muscles was described in a prior experimental infection of LPDV in domestic turkeys; 7 however, these lesions were not microscopically characterized. Our findings may represent differences in North American LPDV versus European and Israeli strains, wild versus domestic turkey host responses, and/or time course of disease.
Nodular aggregates of lymphocytes representing disseminated lymphoid tissues are commonly observed in numerous tissues in healthy birds, 1 so the widespread distribution of lymphoid follicles in LPDV PCR-positive inoculated birds, PCR-negative inoculated birds, and sham-inoculated birds in our study was not surprising. In naturally infected wild turkeys, the diagnosis of lymphoproliferative disease is challenging, often subjective, and depends on the pathologist’s experience with birds, as well as an understanding of species variation, variable degrees of cellular infiltration, and availability of supportive molecular diagnostic tests. This is further compounded by the natural history of wild turkeys and the plethora of pathogens that they could be exposed to that can induce lymphoid aggregates independent of retrovirus infection status. Ultimately, additional diagnostic testing is required to definitively distinguish normal lymphoid aggregates from virus-induced lymphoid aggregates in these experimentally infected birds. Similarly, these diagnostic tests also are needed to help differentiate chronic inflammation from virus-induced lymphoid aggregates in naturally infected wild turkeys. Currently, LPDV confirmatory diagnostic testing in wild turkeys is limited to PCR. Additional modalities such as immunohistochemistry and in situ hybridization would greatly enhance our understanding of pathogenesis and ability to accurately assess diseases associated with LPDV infection.
We documented round cell neoplasia in skin, spleen, adrenal gland, bone marrow, and/or small intestine of domestic turkeys inoculated with North American LPDV strains derived from wild turkeys. Previous reports describe tumor development in the pancreas, spleen, thymus, liver, gonads, and heart in experimentally infected domestic turkeys inoculated with Israeli LPDV strains. 31 Naturally LPDV-infected wild turkeys have exhibited round cell tumors in the small intestine, skin, liver, spleen, pancreas, lung, heart, esophagus, adrenal gland, skeletal muscle, kidney, bone marrow, proventriculus, ventriculus, brain, and thymus.5,29 Neoplastic cell morphology in these birds was variable, but most studies describe predominantly pleomorphic lymphocytes intermixed with lymphoblasts, plasma cells, and reticular cells.5,7,25,29,47 Multinucleated giant cells and histiocytes rarely are described. 22 Although round cell neoplasia is the most conspicuous finding in turkeys with fulminant lymphoproliferative disease, atrophy of the thymic cortex was a consistent lesion at 21 DPI in a previous experimental study. 25 Thymic atrophy was uncommonly observed in LPDV PCR-positive turkeys (2/11, 18%), including 1 turkey at 35 DPI and another at 84 DPI. Thymus may be unavailable in routine necropsies of wild turkeys due to the carcass condition (eg, scavenging, postmortem autolysis), but should be included in microscopic examinations when possible.
Other retroviruses besides LPDV (eg, REV) can cause similar gross and microscopic lesions in turkeys and thus should be ruled out for LPDV diagnoses. As such, we ensured lack of REV in both the inocula and turkeys themselves prior to experimental LPDV inoculation. Gallid alphaherpesvirus 2 rarely causes lymphoid neoplasia in domestic turkeys 17 and ALV can experimentally induce disease in domestic turkeys, 39 but neither virus has been detected in wild turkeys with round cell neoplasms. 29 Although domestic turkeys in Europe7,8,25,42,44 and the Middle East18,47 are susceptible to LPDV infection and disease, this virus has yet to be documented in domestic turkeys in North America. 23 In addition, a small sample set of upland game birds and domestic ducks in Ontario, Canada tested PCR-negative for LPDV. 23 Currently, routine testing of domestic turkeys for LPDV is limited, and LPDV is not recognized as a threat to commercial poultry. Bidirectional disease spillover between commercial and backyard poultry and wild turkeys and other free-ranging birds is well documented 6 and its potential is increasingly scrutinized as the domestic-wildlife interface is blurred. Thus, it is important to recognize that domestic turkeys are susceptible to LPDV derived from wild turkeys in North America.
Various limitations and challenges were encountered in our study. Unintentional, temporary alterations in the artificial light cycles in the enclosures housing the experimental and sham subcutaneous and intracoelomic inoculated turkeys prevented reliable assessment of body weight data. Traumatic fractures sustained in 2 sham-inoculated turkeys prior to the trial endpoint prevented us from fully evaluating contact transmission in half of the designated sham-inoculated birds. Collecting fresh gonads from all turkeys was not performed and would have allowed us to better consider the potential for reproductive (eg, fertility) effects in DNAemic versus non-DNAemic birds. Domestic turkeys were utilized in this study because of their availability, uniformity, and adaption to confinement. Wild turkey populations are genetically diverse across subspecies 27 and are genetically distinct from domestic turkeys,10,20 posing the question if domestic turkeys are an appropriate model for studying LPDV infection in wild turkeys. In addition, susceptibility to LPDV varies among domestic turkey strains,18,25 which could make other broad assumptions about LPDV infection in wild turkey subspecies challenging. This study serves to lay the foundation for forthcoming experimental trials utilizing laboratory-reared wild turkeys.
Our study established, for the first time, that LPDV infection and disease, including round cell neoplasia, can be induced in North American domestic turkeys utilizing a tissue homogenate derived from naturally infected wild turkeys via various inoculation routes, and that proviral DNA can be detected using multiple biologic samples. Data and materials from this experiment allow for further investigation of virus biology and establishment of diagnostic tools to better understand how this enigmatic virus impacts wild turkey populations.
Supplemental Material
sj-xlsx-1-vet-10.1177_03009858241231558 – Supplemental material for Experimental infection of domestic turkeys with lymphoproliferative disease virus of North American origin
Supplemental material, sj-xlsx-1-vet-10.1177_03009858241231558 for Experimental infection of domestic turkeys with lymphoproliferative disease virus of North American origin by Chloe C. Goodwin, Kayla G. Adcock, Andrew B. Allison, Mark G. Ruder, Rebecca L. Poulson and Nicole M. Nemeth in Veterinary Pathology
Footnotes
Acknowledgements
Sydney Burke and Charbel Gerges provided tremendous technical support including molecular testing. Numerous Southeastern Cooperative Wildlife Disease Study diagnosticians and staff supported this study through collection of tissues used for inocula. Alisia Weyna performed numerous turkey necropsies. Ben Price of Prestage Turkeys generously donated and facilitated transfer of the domestic turkeys for this project. Emily Doub transported turkey poults from their hatchery to our research facilities.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was made possible through long-term financial support from state member wildlife agencies of Southeastern Cooperative Wildlife Disease Study provided by the Federal Aid to Wildlife Restoration Act (50 Stat. 917). We also thank the United States Fish and Wildlife Service National Wildlife Refuge System and the United States Geological Survey Ecosystems Mission Area for their long-term support of Southeastern Cooperative Wildlife Disease Study. We thank numerous Southeastern Cooperative Wildlife Disease Study member wildlife agency personnel for their diagnostic case contributions that supported this study. The Robert S. Toth Living Trust contributed a significant portion of funds accomplish this project.
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
