Abstract
A lymphangiosarcoma with metastases was found in a horse that presented with respiratory distress and edema in the ventral thorax and abdomen. The necropsy revealed diffuse edema in the subcutaneous connective tissue. Mediastinal, mesenteric, iliac, and renal lymph nodes were enlarged and white with soft, yellowish necrotic areas. Histologic examination revealed numerous channels and disorganized vessels lined by large polyhedral, polymorphic cells. Tumor metastases were observed in the spleen, lungs, and kidneys. Immunohistochemical evaluation of the tumor cells demonstrated positive staining for factor VIII, vimentin, and keratin. Laminin was scarce, and collagen IV staining was negative, consistent with a discontinuous or absent basement membrane.
Lymphangiosarcoma is a malignant tumor that is rare in domestic animals and that originates from the endothelial cells of lymphatic vessels. This neoplasm is associated with chronic lymphedema in humans.2 Dogs and cats are the domestic animals in which lymphangiosarcoma has been described most frequently.1,3,4,7,9,10 A single instance of this tumor is known in cattle,8 and the first case of equine lymphangiosarcoma of the visceral lymphatic trunk and mediastinum without distant metastases has recently been reported.5 Here, we describe the clinical history and macroscopic, histologic, and immunohistochemical features of an equine lymphangiosarcoma located in the mesenteric and mediastinal lymph nodes and affecting spleen, kidneys, and lungs. This is the first case of equine lymphangiosarcoma with distant metastases to be reported.
A 10-year-old mare was submitted to the emergency services of the Large Animal Clinic of the Faculty of Veterinary Medicine in Madrid with respiratory distress. The thoracic ultrasonogram revealed a large quantity of diffusely echogenic liquid in the pleural cavity of both hemithoraxes, but no masses were visible. Bilateral thoracentesis was performed. Fifteen days later, fluid was again detected in the pleural cavity, and edema in the ventral thorax and abdomen were observed. From that time, the clinical condition of the horse rapidly and progressively deteriorated, and the lack of response to any kind of treatment led to euthanasia of the animal.
Necropsy revealed diffuse edema in the subcutaneous connective tissue of the neck, thorax, abdomen, and limbs. The superficial lymph nodes were moist but displayed no other pathologic changes. A large quantity of clear fluid was observed within the abdominal and thoracic cavities; in the thorax, this liquid contained small fibrinous clots. The lungs had a deposit of pleural fibrin and atelectasis. The lymph nodes of the cranial mediastinum were enlarged, white, and gelatinous on cut surface. The spleen was enlarged and had several reddish superficial nodules; its consistency was firmer than normal, and extensive solid, white areas were observed within the splenic pulp. Mesenteric, iliac, and renal lymph nodes were enlarged, white, and shiny with soft, yellow necrotic areas. Lymphangiectasia was observed in the omentum (Fig. 1).
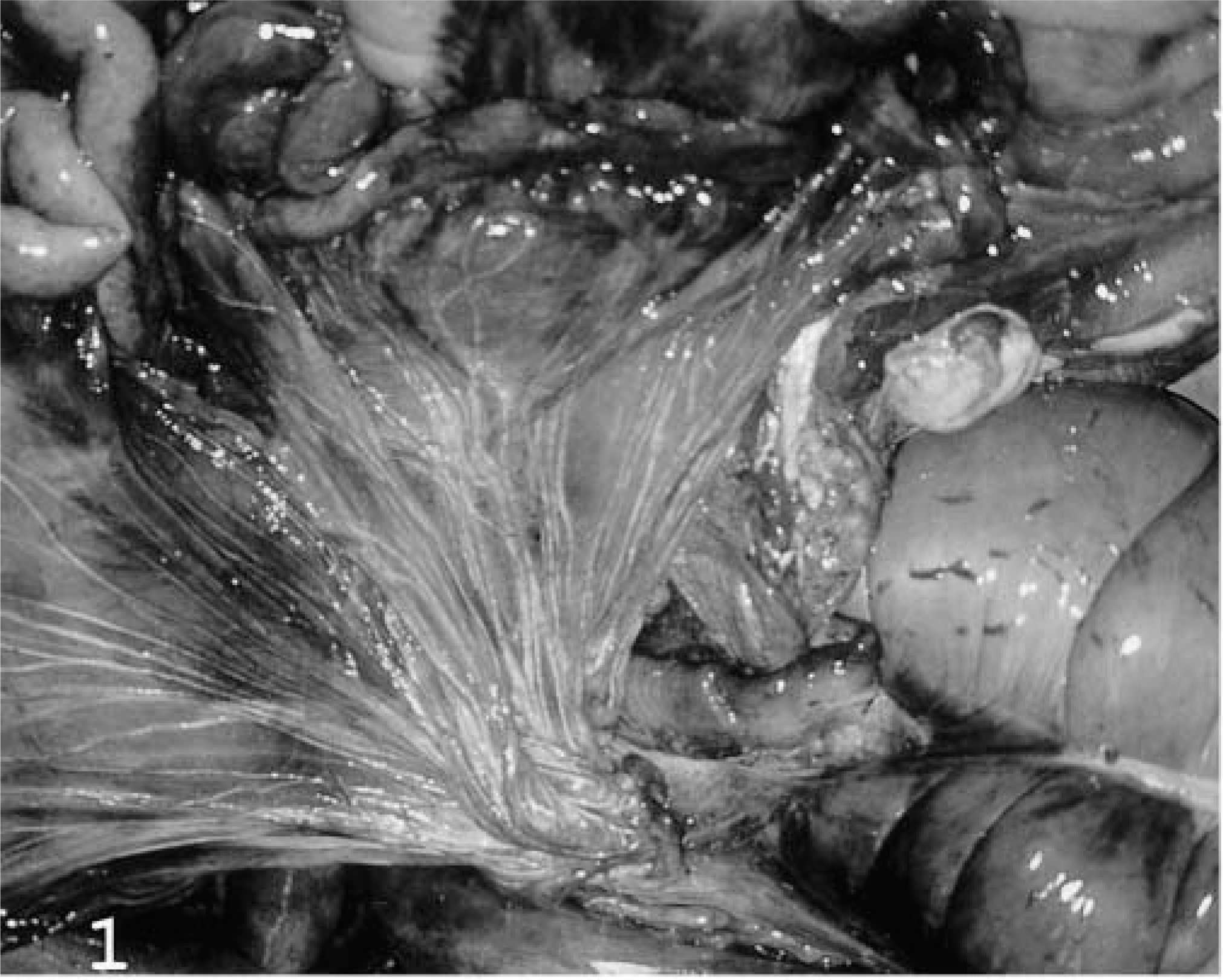
Mesenteric lymphatic channels; horse. Marked lymphangiectasia is visible in the omentum.
Tissue samples from lungs, spleen, liver, kidneys, and mesenteric and thoracic lymph nodes were fixed in 10% buffered formalin, embedded in paraffin, sectioned at 4 μm, and stained with hematoxylin and eosin (HE). Immunohistochemical staining was performed using the streptavidin–biotin–peroxidase complex method. Tissue sections were processed with 10 mM citrate buffer (pH 6.0) in a microwave oven at 100 C for 15 minutes. Endogenous peroxidase activity was inactivated by incubation with 3% hydrogen peroxide in methanol for 15 minutes at room temperature (RT; 20–25 C). Rabbit anti-keratin wide-spectrum polyclonal antibody (pAb) diluted 1:800 in Tris-buffered saline (TBS) (DAKO, Glostrup, Denmark), mouse anti-vimentin monoclonal antibody (mAb) diluted 1:25 in TBS (DAKO), rabbit anti-human factor VIII pAb diluted 1:300 in TBS (DAKO), rabbit anti-human laminin pAb diluted 1:25 in TBS (Chemicon International, Temecula, CA), and rabbit anti-human collagen type IV pAb diluted 1:50 in TBS (ICN Biomedicals, Chemical Credential, Costa Mesa, CA) were incubated overnight at 4 C in a humid chamber. Sections were also incubated with rabbit anti-human Ki-67 pAb diluted 1:1,000 in TBS (Novocastra Laboratories, Newcastle, UK) for 1 hour at 37 C. After three 5-minute rinses in TBS, a biotinylated goat anti-rabbit IgG diluted 1:400 in TBS (Vector Laboratories, Burlingame, CA) for pAb and a biotinylated rabbit anti-mouse immunoglobulin diluted 1:200 in TBS (DAKO) for mAb were applied as secondary reagents for 30 minutes at RT. After three 5-minute rinses in TBS, tissue sections were incubated with streptavidin conjugated with peroxidase diluted 1:20 in TBS (Zymed Laboratories, South San Francisco, CA) for 30 minutes at RT. The chromogen employed was 3-3′-diaminobenzidine tetrahydrochloride (Sigma Chemical Co., St. Louis, MO) at 0.0035% in TBS containing 0.01% hydrogen peroxide. Primary antibodies were replaced by TBS and rabbit and mouse nonimmune serum in negative controls.
Histologic examination of the lymph nodes and adjacent tissue revealed numerous channels and disorganized vessels lined by disorganized polymorphic cells with eosinophilic cytoplasm. These cells frequently displayed a large supranuclear vacuole or even a vacuole that occupied the entire cytoplasm and displaced the nucleus. These cells occasionally had cytoplasmic extensions. The nucleus was usually large and euchromatic and contained one or two large nucleoli. Mitoses were frequently observed (six/high-power field). Cells aligned in palisades were free in the lumina of the vascular channels (Fig. 2). Erythrocytes were very rarely observed within the lumina. In some areas, the tumor cells grew into the lumen as papilliferous growths with a central connective core. In other areas, detached cells occupied the lumina. In addition, necrotic and hemorrhagic areas were observed. The stroma, occasionally edematous, was composed of dense bundles of collagen, and lymphocytic aggregations were frequently seen. The afferent tumoral lymphatic vessels within the capsule of some mesenteric lymph nodes contained abundant necrotic material and neutrophils. Tumor metastases were observed in the spleen, lungs, and kidneys. The spleen had thick bands of fibrous trabecular tissue infiltrated by proliferating vascular channels, similar to those described in the lymph nodes. In some areas, the splenic architecture was effaced. The nodular areas corresponded histologically to nodular hyperplasia. Tumor metastases were also observed in pulmonary connective tissue and in the renal cortex.
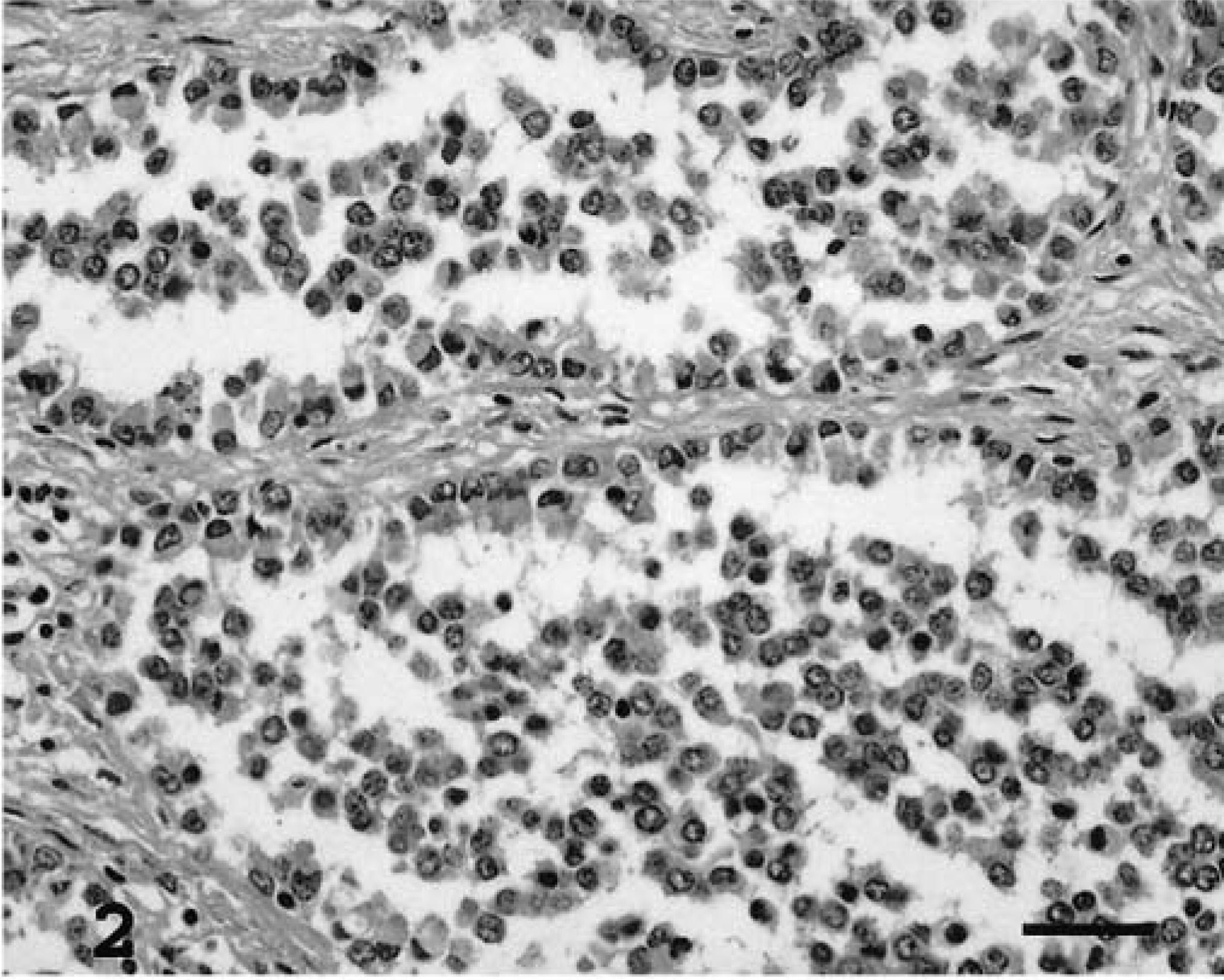
Lymph nodes; horse. Lymphangiosarcoma. Disorganized vessels lined by pleomorphic neoplastic cells aligned in palisade. HE. Bar = 70 μm.
Immunohistochemical evaluation of the tumor cells demonstrated moderate positive staining for keratin, which appeared as a diffuse cytoplasmic reaction in the majority of the cells. Factor VIII immunostaining was marked and diffuse throughout the cytoplasm, with the most intense staining near the cell membrane (Fig. 3). Some cells showed positive staining for vimentin (Fig. 4), which was especially concentrated in the paranuclear zone. Basement membranes were stained using laminin and collagen IV antibodies. Laminin expression was sparse and discontinuous in the tumor lymphatic spaces, whereas collagen IV immunostaining was negative. These findings suggested a discontinuous or absent basement membrane. Adjacent normal vascular endothelium was positive for both basement membrane markers. The proliferation index, measured as the percentage of Ki-67-positive cells in five high-power fields (about 1,000 cells), was 20% suggesting that the neoplasm was malignant.
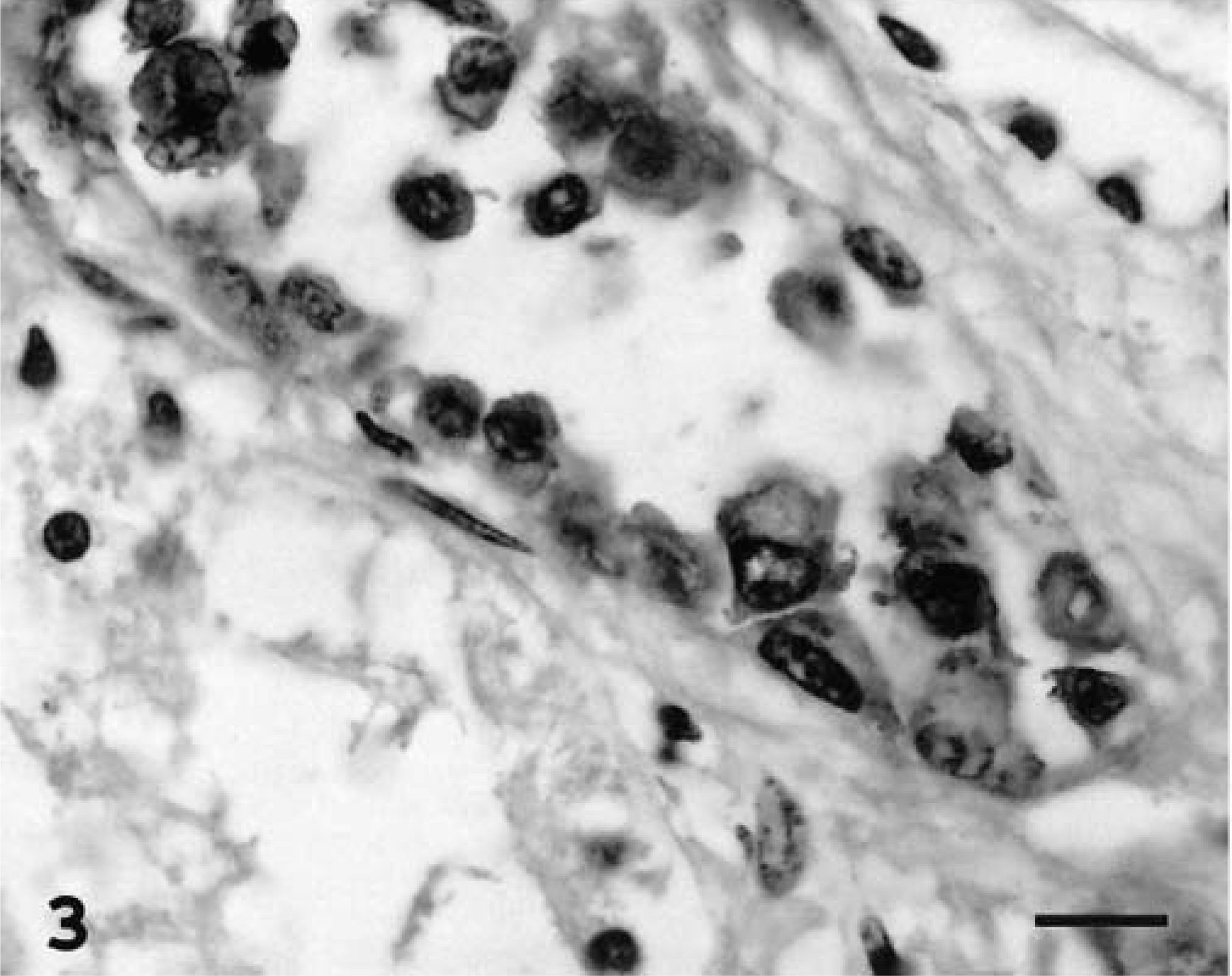
Kidney; horse. Lymphangiosarcoma. Marked cytoplasmic immunoreactivity with the anti-human factor VIII pAb. Streptavidin–biotin–peroxidase complex method, Gill's hematoxylin counterstain. Bar = 25 μm.
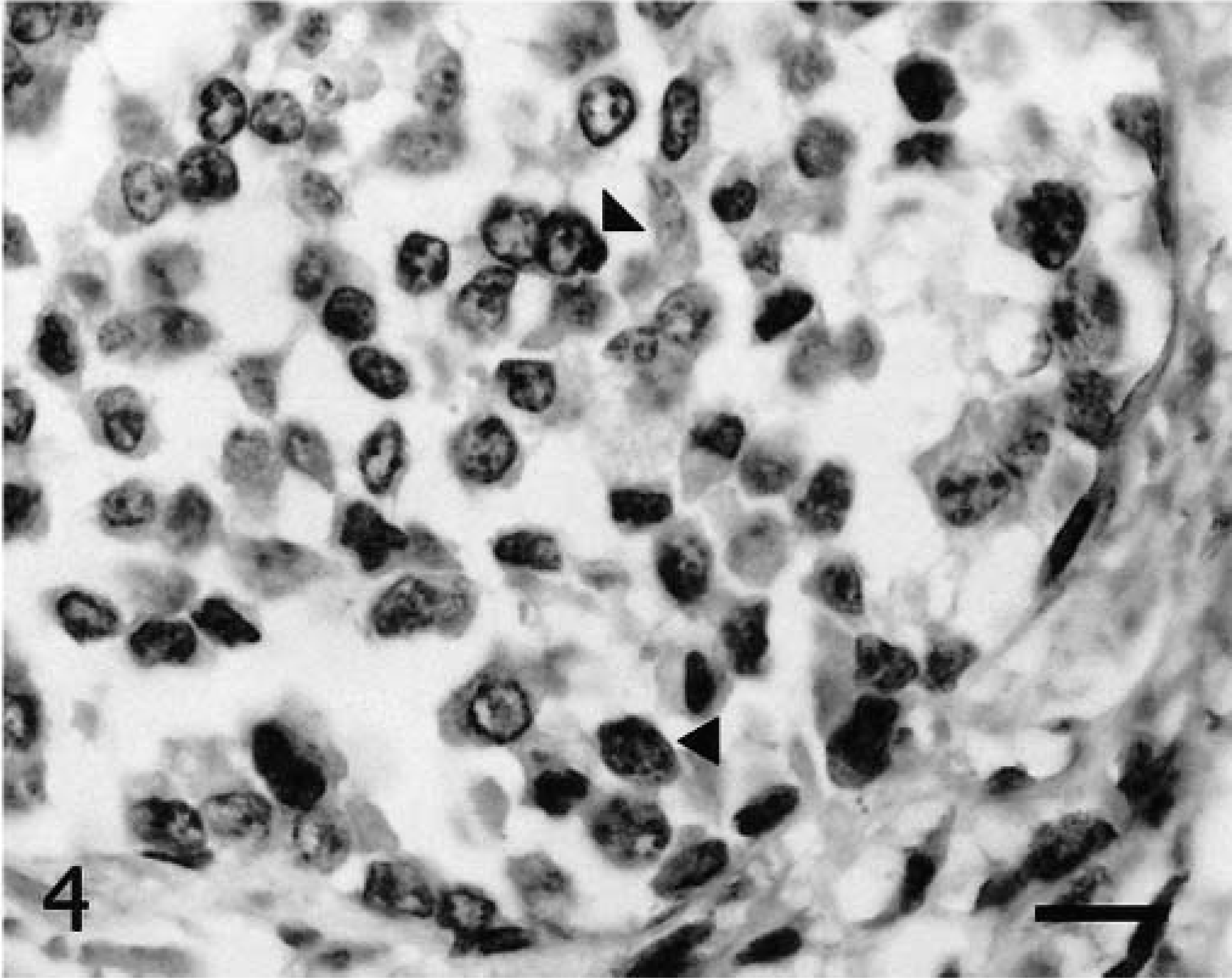
Lung; horse. Lymphangiosarcoma. Some tumor cells have a paranuclear zone positive with anti-human vimentin mAb (arrowheads). Streptavidin–biotin–peroxidase complex method, Gill's hematoxylin counterstain. Bar = 25 μm.
The clinical history together with the gross, histologic, and immunohistochemical findings led to a diagnosis of mesenteric and mediastinal lymphangiosarcoma with multiple metastases. This is the first case with these characteristics reported in a horse. Recurrent edema in the neck, lower thorax, anterior limbs, and abdomen and fluid accumulation in the thoracic and/or abdominal cavity are the clinical signs described in dogs,1,3 cats,4,10 and in one other horse5 with this type of neoplasm. In general, these clinical signs indicates that the clinical condition of the animal is grave.
In humans, lymphangiosarcoma is associated with chronic lymphedema, as occurs in women who develop lymphangiosarcoma in the arms after undergoing radical mastectomy.2 However, in domestic animals this association has only been observed in some cases, e.g., two cats that underwent amputation of the hind limbs.10 In many cases, as in that of the mare in the present report, the etiology remains uncertain because it is not possible to determine whether chronic edema was the result or the cause of the tumor.
Involvement of mesenteric and mediastinal lymph nodes has been reported in the horse,5 dog,1,7 and cat4,10; and the presence of metastases in lungs, spleen, and kidneys has also been reported in canine and feline lymphangiosarcomas.1,4 However, the present case is the only one described in a horse.
Discrimination between lymphangiosarcoma and hemangiosarcoma can be difficult; both neoplasms arise from endothelial cells and develop channels or vessels. The diagnosis of lymphangiosarcoma is based on morphologic features, including the absence of red blood cells within vascular channels lined by abnormal endothelial cells3,5,9 and the presence of a lymphocytic infiltrate in the stroma.1,5
The expression of factor VIII by neoplastic cells has been reported in canine,1,3 feline,4 and equine5 lymphangiosarcomas. Tumor cells immunohistochemically positive for vimentin in the paranuclear zone have previously been described in canine9 and equine5 lymphangiosarcomas. The expression of wide-spectrum cytokeratins in the neoplastic cells is consistent with results obtained from human lymphangiosarcomas6 but not with those from a lymphangiosarcoma in a horse stained with antiserum to cytokeratins 5–185 and those from tumors of dogs.9 A discontinuous or absent basement membrane has also been noted as a histologic characteristic of lymphangiosarcoma.5,9 Collagen IV was not observed in the tumor in this horse, and laminin staining was discontinuous and sparse, consistent with previous descriptions.9 The malignancy of the tumor was reflected the presence of metastases in various organs and by the high proliferation index obtained using the cellular proliferation marker Ki-67.
Lymphangiosarcomas are rare in domestic animals, and this is the first reported case of metastatic lymphangiosarcoma in a horse. Lymphangiosarcoma was previously described in a horse, but no metastases were found. Common clinical, morphologic, and immunohistochemical features could, nevertheless, be useful in diagnosing this neoplasm in the future.
