Abstract
A 9-year-old Tennessee Walking Horse gelding was presented for diagnosis of the cause of extensive alopecia. Complete hair loss was noted over the head, neck, shoulder, thigh, and proximal limbs, but the trunk, distal limbs, pelvic area, mane, and tail were unaffected. The alopecic areas were visually noninflammatory with no exudate or crust except on the shoulder and along the back, where multifocal patchy areas of alopecia with scales and crust were evident. The horse was slightly pruritic. Microscopically, the hair bulbs, inner and outer root sheaths of inferior segments, and perifollicular regions were infiltrated by small to moderate numbers of small lymphocytes. Similar inflammation was occasionally evident in isthmus follicular walls as well as some apocrine glands. No sebaceous glands were affected. Immunohistochemistry confirmed that the small lymphocytes were CD3+ T lymphocytes. The epidermis from the skin with scale and crusts along the horse’s back exhibited mild to moderate hyperplasia, mild lymphocytic exocytosis, mild eosinophilic dermatitis, and diffuse parakeratosis with numerous budding yeasts, consistent with Malassezia spp. The final disease diagnosis was made as alopecia areata with Malassezia dermatitis. Alopecia areata could be a contributing underlying factor for Malassezia dermatitis.
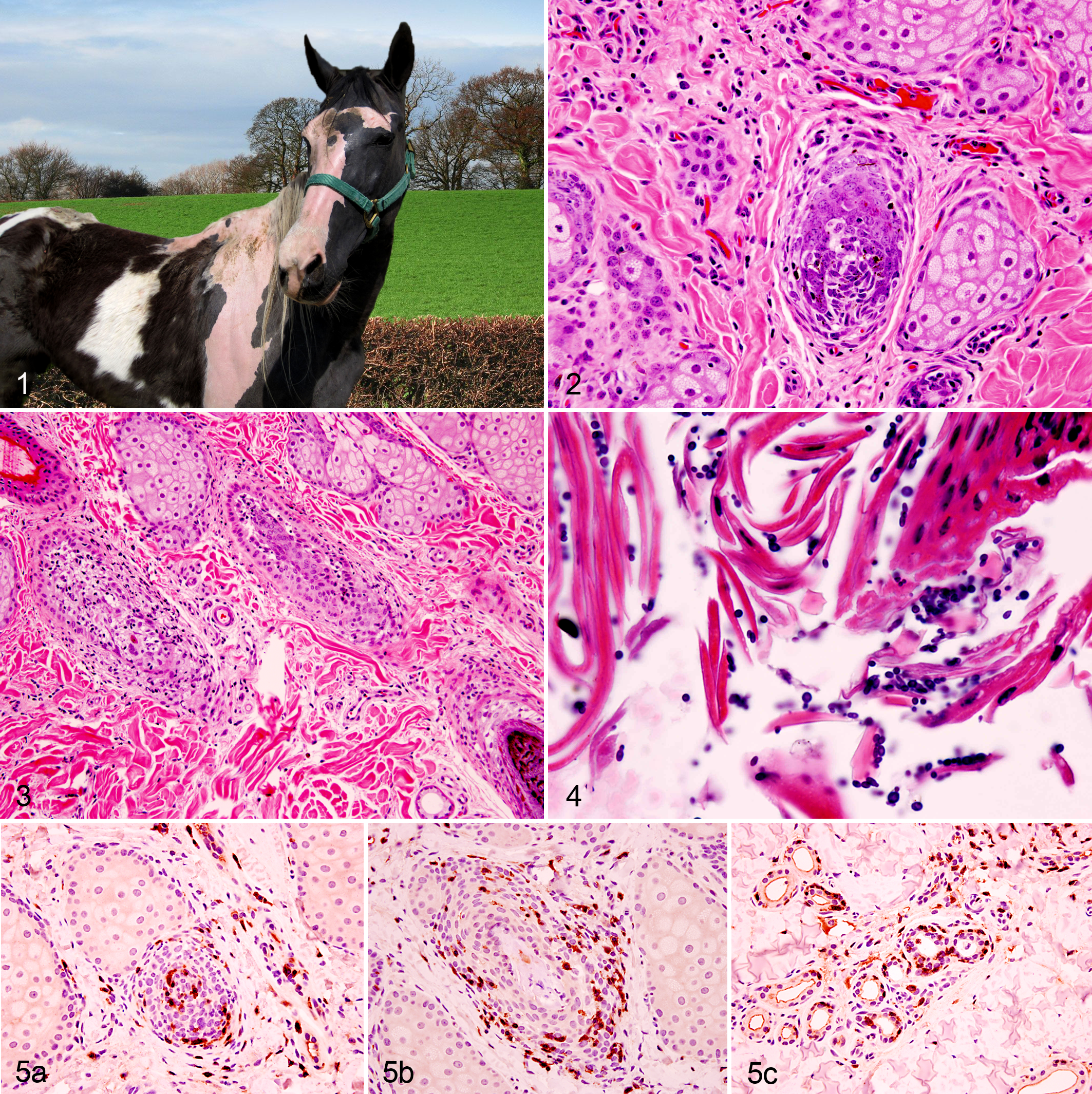
A 9-year-old Tennessee Walking Horse gelding was presented to the Veterinary Medical Teaching Hospital, College of Veterinary Medicine, University of Missouri, with extensive alopecia. Bilaterally symmetrical complete hair loss was noted over the head, neck, shoulder, thigh, and proximal limbs, but the trunk, distal limbs, pelvic area, mane, and tail were unaffected (Fig. 1). The alopecic areas were clinically noninflammatory with no exudate or crust except on the shoulder and along the back, especially on the right side, which were multifocally covered with scales and crust. Both pigmented and nonpigmented skin was affected to a similar extent. At the periphery of areas of alopecia, hairs were loose and easily epilated. The horse was slightly pruritic. According to the owner, the alopecia had involved only the head and neck throughout the antecedent 1.5 years, slowly progressed, and then suddenly spread to affect more extensive areas of the body during the 1 to 2 weeks prior to admission. The referring veterinarian had submitted blood for determination of iodothyronine concentrations when alopecia had first been evident 14 months earlier, at which time the circulating thyroxine concentration was very low (T4: <0.05 ug/dL [reference range: 0.5–4.0 ug/dL]; T3: 41.1 ng/dL [reference range: 30–115 ng/dL]). At that time, the gelding’s physical appearance was also characterized by marked thickening of the crest of the neck. The veterinarian had addressed suspected hypothyroidism by treating the gelding using an orally administered thyroxine supplement (Thyro-L) for several weeks. At the Veterinary Medical Teaching Hospital, the results of routine hematology and a plasma biochemical profile were unremarkable. Serum was submitted to the veterinary endocrine testing laboratory at Michigan State University for thyroid function; these tests yielded normal results for total T4 (21 nmol/L; reference range: 7–27 nmol/L) and free T4 (by dialysis, 24 pmol/L, reference range: 8–39 pmol/L). However, both total T3 (0.5 nmol/L, reference range: 0.7–2.5 nmol/L) and free T3 (by dialysis, 0.8 pmol/L, reference range: 1.7–5.2 pmol/L) were slightly depressed.
Differential Diagnoses
Differential diagnoses for the severe visually noninflammatory alopecia in the horse include anagen effluvium (defluxion), telogen effluvium (defluxion), follicular dysplasia, and alopecia areata (AA). Anagen effluvium is characterized by sudden, severe hair loss within days after a special insult (eg, infectious diseases, metabolic diseases, fever). Typical microscopic findings of anagen effluvium include apoptosis in the hair follicular wall and dysplastic and irregular hair shafts. Anagen effluvium was ruled out owing to the prolonged history in the present case. In telogen effluvium, a severe, stressful pathophysiological event such as high fever, shock, pregnancy, severe diseases, or surgery causes severe hair loss after weeks or months. Microscopic evaluation warrants differentiating telogen effluvium from AA. Skin biopsy is often not helpful for telogen effluvium because of lack of microscopic abnormalities in hair follicles. On the other hand, AA is characterized by lymphocytic follicular bulbitis and inferior mural folliculitis. Follicular dysplasia (eg, mane and tail dysplasia, idiopathic regional pigment-related follicular dysplasia syndrome) is believed to be a genetic disorder and recognized at birth or in juveniles with brittle, dull, hypotrichotic hair coats. Microscopically, abnormal hair follicles and dysplastic hair shafts are commonly found. For multifocal inflammatory crusty alopecia, staphylococcal dermatitis, demodecosis, dermatophytosis, dermatophilosis, hypersensitivity dermatitis, and Malassezia dermatitis can be discussed.
Microscopic Findings
Microscopically, anagen hairs were not identified in examined areas of complete alopecic skin and a few anagen hairs were observed in partially alopecic skin. Hair bulbs, inner and outer root sheaths of inferior segments, and perifollicular regions were often infiltrated by small to moderate numbers of small lymphocytes (Fig. 2). Occasionally, similar inflammation was noted in the follicular wall of the isthmus (mural folliculitis) as well as some apocrine glands. The affected hair follicular walls were often vacuolated (Fig. 3). No sebaceous glands were affected. The epidermis from the skin along the horse’s back, which was covered with scales and crusts, was mild to moderately hyperplastic, with acanthosis, diffuse parakeratosis, and mild lymphocytic exocytosis. The superficial dermis had mild lymphocytic and eosinophilic inflammation. Numerous peanut-shaped, broad-based budding yeasts (consistent with Malassezia spp.) were noted within the parakeratotic stratum corneum (Fig. 4). Immunohistochemistry using rabbit polyclonal anti-human CD3 antibody (DAKO, Carpinteria, CA) demonstrated strongly positive immunoreactivity to the small lymphocytes that infiltrated the hair follicles (bulbs and walls) and apocrine glands (Fig. 5).
Diagnosis
The morphological diagnoses were severe multifocal lymphocytic follicular bulbitis and mural folliculitis with multifocal lymphocytic apocrine adenitis and moderate multifocal epidermal hyperplasia with lymphocytic dermatitis, parakeratosis, and intralesional budding yeasts (consistent with Malassezia spp.). The final disease diagnosis was made as AA with Malassezia dermatitis.
Discussion
Alopecia areata is an uncommon immune-mediated hair loss that has been reported in various species including the humans, nonhuman primates, 2 dogs, 8 cats, 8 cattle, 14 chickens, 16 laboratory rodents, 10 and horses.35,11,15 Clinically, AA is characterized by sudden or insidious, well-circumscribed, nonscarring, and visually noninflammatory alopecia. In humans, AA occurs commonly on the scalp in a pattern of a focal patch or multiple patches; however, other subgroups such as complete loss of scalp hair (alopecia totalis) and entire body hair (alopecia universalis) have been recognized. 1 In horses, similar lesions are often noted on the face, neck, trunk, mane, tail, shoulder, and gluteal region with varying degrees of severity.3,5,11,15,17 In humans, AA is quite common, affecting 1.7 to 3.8% of the general population, with no sex predilection. 18 Equine AA is less frequently recognized. At a university equine teaching hospital, AA accounted for 1.33% of total equine dermatology cases. 15
Although AA has been recognized for centuries in humans, the pathogenesis of AA is still not fully understood. In the past, an infectious (parasitic or viral) disease or a neurological disorder was proposed, but recent studies have indicated that it is likely a T-lymphocyte–driven autoimmune disease.1,18 Microscopic evidence of lymphocytic infiltration into the hair bulbs and effectiveness of immunosuppressive treatment are supportive of this hypothesis. The hair follicle infiltrates are primarily CD8+ cytotoxic T cells, CD4+ helper T cells, macrophages, and a lesser population of dendritic antigen presenting cells in the mouse model.1,10,18 The C3H/HeJ mouse is a well-established AA animal model. In C3H/HeJ mice, injecting the CD8+ T cells harvested from AA-affected mice into healthy mice resulted in a focal injection-site alopecia, whereas CD4+ T cell injection produced systemic alopecia, suggesting both CD4+ and CD8+ T cells are associated with AA, but CD11+ dendritic cells and CD19+ B cells failed to cause alopecia. 10 In the previously reported equine AA cases3,5 and in the present case, CD3+ T cells were demonstrated by immunohistochemistry. Further investigation to identify CD4+ and CD8+ subsets was not successful because of the lack of specific antibodies to the equine cluster of differentiation (CD) transmembrane proteins. Autoantibodies (IgG) against hair follicular antigens were demonstrated in a horse with AA. 17
In the present case, CD3+ lymphocytes infiltrated not only hair bulbs, perifollicular regions, and inner and outer root sheaths of the inferior segments, but they also infiltrated the follicular wall of the isthmus and apocrine glands. Colombo et al reported lymphocytic mural folliculitis 5 similar to the present case and suggested a possible histological variation of AA. The findings of isthmus mural folliculitis could be explained by pseudopelade, a rare T-cell–induced autoimmune alopecia reported in humans, dogs, and cats. Equine cases of pseudopelade have not been recognized. Pseudopelade differs from AA in that the inflammatory changes in pseudopelade are restricted to the isthmus hair follicular wall. In the present case, the lymphocytic inflammation in the apocrine gland was unique; no similar changes have been previously described. Alopecia areata with lymphocytic sebaceous adenitis was reported in a Rhesus monkey. 2
Hair growth cycle patterns are different in individual human AA patients depending on severity and duration. Alkhalifah et al reviewed 3 different AA alopecic patterns: Dystrophic anagen - in mild inflammation, the anagen hairs become dystrophic, unable to produce healthy hairs; Truncated cycling–moderate inflammation results in a short anagen phase; Prolonged telogen–in chronic AA. In the present case, some distorted dysplastic hair follicles were observed in less severely affected skin.
1
Abnormal nail changes have been reported in 7 to 66% of human AA patients.
1
Bruet et al reported a French Trotter mare with severe AA and onychodystrophy.
3
There were no signs of onychodystrophy or other hoof/chestnut abnormalities in our gelding. Autoimmune conditions, especially autoimmune thyroiditis, have been identified in 8 to 28% of human AA patients.
1
The referring veterinarian suspected hypothyroidism as the cause of the alopecia based on a single low plasma total T4 level. However, subsequent treatment with L-thyroxine did not improve the condition of the alopecia. At the Veterinary Medical Teaching Hospital, T4 levels from 2 different laboratories (Antech and Michigan State University) were within the reference range, suggesting that the low T4 at the local veterinary clinic could have been an error or that previously diagnosed underlying thyroid gland disease had resolved. Furthermore, there is no real documentation in the literature linking equine alopecia to hypothyroidism.
No effective treatment is currently available for equine AA, but spontaneous regrowth can occur in mild cases. The published cases of diffuse AA reported no hair regrowth.3,5 In humans, spontaneous recovery may occur in mild and acute cases within a year. There are several treatment options for human AA patients, including systemic, topical, or intralesional corticosteroids; minoxidil; and contact immunotherapy using diphenylcyclopropenone or dinitrochlorobenzene to shift the immune response from Th1 to Th2. Nonetheless, human patients with severe AA generally exhibit poor response to these treatments. 7
The following species of genus Malassezia have been identified on normal and abnormal horse skins: M. furfur, M. obtusa, M. globosa, M. pachydermatis, M. restricta, M. slooffiae, M. sympodialis, and most recently, M. equina.4,12,19 Equine Malassezia dermatitis is uncommon, and only a few cases have been published. The clinical history of pruritus was likely related to Malassezia dermatitis, which histologically caused epidermal hyperplasia, parakeratosis, and mild lymphocytic exocytosis and eosinophilic dermatitis. In horses affected with Malassezia dermatitis, the characteristic variously pruritic, greasy to waxy, crusty skin lesions have been found in the axillae, groin, intermammary region, prepuce, and pastern.9,20 In the present case, the Malassezia infection was located on the alopecic shoulder and back, which are not typical locations for Malassezia dermatitis based on the previous reports. Since Malassezia usually establishes infection in immunologically impaired skin (such as that affected by atopic dermatitis, allergic dermatitis, or sebaceous adenitis6,13), the altered epidermal condition resulting from AA is likely a contributing factor for Malassezia infection in the present case.
Footnotes
The authors declared that they had no conflicts of interest with respect to their authorship or the publication of this article.
The authors declared that they received no financial support for their research and/or authorship of this article.
