Abstract
Three experimental groups of six male raccoon dogs (Nyctereutes procyonoides) each were formed by placing one of three littermates from six litters into each group. One group was inoculated with pigorigin Trichinella spiralis, the second was inoculated with raccoon dog-origin T. nativa, and the third served as a control group. The infective dose was 1,000 larvae/kg of body weight. Every third week, biopsies from M. triceps brachii were taken, and serum samples were collected for up to 12 weeks postinfection. In the early phase of the infection, cysts of both parasites were elongated cylinders that later became more spherical. However, at the end of the experiment, the cysts of T. nativa were more rounded than those of T. spiralis (mean length/width = 2.5 versus 1.5 in T. spiralis versus T. nativa, respectively). Both species accumulated a collagen-rich capsule around the nurse cell, but the capsule was thicker in T. nativa. In both parasites, the total surface area of the sagittal section of the cyst was equal. Inflammation was more intense around T. nativa cysts. Specific antibodies were recognizable 2 weeks after infection by both enzyme-linked immunosorbent assay (ELISA) and western blot. In western blots, serum from both T. nativa- and T. spiralis-infected animals recognized the same components, but reaction with the homologous antigen was stronger. The same pattern was also seen in the ELISA. Immunoreactive epitopes were localized only in internal organs and cuticula of larvae in muscle.
Despite the decline in prevalence of Trichinella infections in other countries during the last few decades, the situation appears to have been quite different in Finland.10 Even though the prevalence is not yet alarmingly high in domestic animals, comparisons of recent observations and earlier reports of Trichinella in wildlife and swine indicates an unexpected increase in Finland.27–29 After the inception of official meat inspection in 1867, trichinellosis was rarely observed in domestic swine until the early 1980s.16,35 Since this time, it has been found almost annually.1,16,28 A similar tendency toward an increase in Trichinella prevalence has also occurred in wildlife.27,29 For example, in red foxes (Vulpes vulpes) a report from 1955–1956 indicated a 16% Trichinella prevalence in Finland.35 From 1963 to 1964, the prevalence was 4%,9 and in the early 1980s it rose to 33%.16 Current reports of Trichinella prevalences are as high as 50% in the southern part of the country.27
This increased prevalence of trichinellosis in wildlife and domestic animals has occurred simultaneously with colonization of the southern part of the country by the raccoon dog (Nyctereutes procyonoides).15 Clear differences in wildlife Trichinella also have appeared.29 Host species–specific differences have been reported in the virulence of Trichinella.19 However, raccoon dogs seem to be very versatile hosts and have been reported to be infected by T. pseudospiralis, T. spiralis, T. nativa, and T. britovi.4,44,45 In this study, we evaluated the raccoon dog as a potential important vector and reservoir animal for Trichinella in Finnish fauna.
In raccoon dogs, trichinellosis caused by T. nativa and that caused by T. spiralis differ only slightly, and Trichinella seem to be well adapted to these animals.25 Here, we report differences in histopathologic and humoral responses of these two parasite species.
Materials and Methods
Study design
Eighteen raccoon dogs were divided into three experimental groups. Animals were from six litters, and three 4-month-old males from each litter were used for the experiment, with one of the siblings randomly placed in each group. One group was inoculated with pig-origin T. spiralis (ISS559), and one group was inoculated with raccoon dog–origin T. nativa (ISS558); the third group served as an uninfected control group. Animals were purchased from the Fur Animal Research Station (University of Kuopio, Juankoski, Finland). The infective dose of each parasite was 1000 larvae/kg body weight. Details on animal husbandry and clinical signs of the infection have been published elsewhere.25 During the 12-week experimental period, blood was collected regularly: daily for the first week, then every second day for 3 weeks, and finally once weekly for 8 weeks. The Faculty of Veterinary Medicine (University of Helsinki) Committee on Animal Experiments approved the study.
Muscle biopsy
Every third week, the animals were sedated with an intramuscular injection of medethomidine (60 µg/kg; Domitor, Orion, Espoo, Finland) and a combination of fenpipramidehydrochloride (0.015 mg/kg) and l-methadonehydrochloride (0.3 mg/kg; L-Polamivet, Hoechst Roussel Vet, Wiesbaden, Germany). Local anesthesia (lidocaine hydrochloride, 20 mg/ml, Orion) was used at the biopsy site. Specimens (2 × 1 cm) were surgically removed from the left front leg (M. triceps brachii, caput longum) at four different times and from the left hind leg (M. semitendinosus) at the first and the last biopsy time. The incision was sutured with absorbable 3-0 suture material (Vicryl, Ethicon, Johnson & Johnson Intl., Brussels, Belgium), and the sutures were removed 13 days later. Intramuscular atipamezole (Antisedan, Orion) was used to antagonize medethomidine and speed recovery. To prevent infection at the biopsy site, a single subcutaneous injection of long-acting procaine benzyl penicillin (Duplocillin LA Vet, Intervet, Boxmeer, Holland) was given at the time of biopsy.
At sampling, a piece of muscle was fixed with pins longitudinally on a wooden splint to prevent contraction during fixation in buffered 4% formaldehyde. After fixation, part of the tissue was cut and rotated 90° to obtain both longitudinal and transverse orientations for paraffin-embedded blocks. Some samples were also frozen in isopenthane and precooled with liquid nitrogen for cryomicrotomy and adenosinetriphosphatase (ATPase) staining.
In addition to the samples for histopathology, another piece was used for quantitation of infection. Each of these samples was weighed, and squash specimens were prepared. Tissue pieces were set on a glass plate (each on a separate square), and a drop of tap water was placed on each to prevent drying. The samples were compressed by laying another glass plate on top. Encysted and free larvae were counted with a light microscope.
Hematoxylin and eosin (HE) staining was performed on all samples. Masson-trichrome, periodic acid–Schiff (PAS) with and without diastase, and Van Giesson methods were also used on selected samples to detect collagen, glycogen, and reticulin, respectively. Evaluation of ATPase reactivity was performed at pHs 4.3, 4.6, and 10.3 using standard procedures.3
Histopathologic grading
Several histopathologic characteristics were recorded for the HE-stained tissue sections: number of cysts per microscope field, shape of cysts (3 = round; 2 = elliptic; 1 = spindle shape; 0 = indeterminate), basophilic layer of the nurse cell (present or not), number of nuclei in each nurse cell (3 = more than 10; 2 = 3–10; 1 = less than 3; 0 = none), maturation/thickness of the collagen layer of the capsule (from absent to uniform thickness of at least three erythrocytes around the nurse cell), number of mononuclear cells around the capsule (3 = over 30; 2 = 20–30; 1 = 5–20; 0 = less than 5), and numbers of eosinophils around the capsule. To avoid pseudoreplication and bias, only the first section of each paraffin block was used for comparisons, and all cysts in the section were included. Scoring of the samples was done by one of the authors, without prior knowledge of time or infection group.
Morphometry at termination
Quantitative image analysis was performed with a Color View 12 digital camera and 3.0 Image Analysis software (Soft Imaging System GmbH, Munster, Germany). The area of the cyst, area of the nurse cell (including larva inside), area of the capsule, and width and length of the cyst (the shortest and the longest axis) were manually measured from the digital micrograph. To avoid variation in section level of the cylinder-shaped object, only cysts showing larvae and muscle fibers with longitudinal orientation were included in the morphometric analyses. To increase the number of observations and the power of the analyses, muscle samples from the hind legs were used in addition to the front leg samples.
Serology
For developing serologic assays, we first confirmed the cross-reactivity of commercial anti-dog IgG antibody (rabbit anti-dog, Serotec, Oxford, UK) with raccoon dog antibodies. The animals had been vaccinated against canine distemper at the place of origin. We used the canine distemper vaccination antibody for a positive control. In western blot assays, antigen from canine distemper-infected Vero cells confirmed the cross-reactivity between anti-dog antibody and raccoon dog immunoglobulins. Antigens for specific assays were prepared by digestion with pepsin and HCl from the L1 stage of T. spiralis or of T. nativa harvested from mice.
Western blot
Larvae were lysed in sample buffer (0.5 M Tris-HCl, 10% glycerol, 2% sodium dodecyl sulfate [SDS], 5% 2-b-mercaptoethanol, 0.05% bromophenol blue, pH 6.8) by heating them for 5 minutes at 98 C. Proteins from the crude antigen preparation were separated in a 12% SDS–polyacrylamide gel with a 4% stacking gel under reducing conditions and then transferred to a nitrocellulose membrane (Trans-Blot, Bio-Rad, Hercules, CA). The same buffers as used in the enzyme-linked immunosorbent assay (ELISA) were used for immunostaining the nitrocellulose strips. To reduce nonspecific binding, strips were incubated overnight in blocking buffer, washed with washing buffer, incubated for 60 minutes at room temperature with dilutions of raccoon dog serum, washed, and incubated with horseradish peroxidase-labeled secondary antibody. After intense washing, diaminobenzidine (Sigma Fast DAB, Sigma, St. Louis, MO) was used as a chromogen. The intensity of the immunoreaction was categorized visually into four classes.
ELISA
Larvae were lysed in coating buffer (0.1 M NaHCO3, pH 9.6) with ultrasound (20 kHz) in an ice bath. Lysates were centrifuged, and protein concentration was determined from the collected supernatant with commercial reagents (BCA protein assay kit, Pierce, Rockford, IL) using a known amount of bovine serum albumin as the standard. Microtiter plate wells (MaxiSorp surface Nunc-Immuno plate, Nunc Brand Products, Roskilde, Denmark) were coated overnight with 200 ng of crude antigen protein in 100 µl of coating buffer per well at room temperature. Wells were washed once with saline, and nonspecific staining was blocked with blocking buffer (5% [wt/vol] skim milk and 0.2% Tween 20 in phosphate-buffered saline for 1 hour at room temperature. After washing three times, wells were incubated with a twofold serial dilution of test sera in saline and incubated for 1 hour at 37 C. Wells were then washed three times, and 100 µl of peroxidase-conjugated goat anti-dog antibody (Serotec; diluted 1:10,000) was placed into the wells for 30 minutes at 37 C. Wells were washed four times, and tetramethyl benzidine substrate was added for the color reaction. The reaction was terminated after approximately 2–4 minutes according to color development with 1 N sulfuric acid, and absorbances were measured at 450-nm wavelength (iEMS Reader MF, Labsystems, Helsinki, Finland). The cut-off value for positive samples was set at the mean absorbance of the negative animals plus two times the SD at the termination of the experiment.
Immunocytochemical localization
Sections of Trichinella larvae and cysts from both groups obtained at 12 weeks postinfection were incubated for 60 minutes at room temperature in sera (dilution 1:100) from raccoon dogs with homologous and heterologous Trichinella species infections. Serum from a control animal served as a negative control and normal goat serum was used for blocking. Sections were incubated for 30 minutes with the same secondary antibody used in the ELISA and western blot (dilution 1:400), and aminoethylcarbazole served as a chromogen.
Statistics
Data are expressed as mean ± SEM. To analyze information from each of the tissue cysts, which were clustered in experimental animals, differences between groups at each sampling time were tested by a mixed-model analysis of variance (ANOVA) with group as a fixed variable and animal as a random variable nested within the group.36 The effect of time was analyzed separately in each experimental group, with time as a fixed variable. A one-way ANOVA and t-test were also used when appropriate. Differences with P values of <0.05 were considered significant.
Results
Quantitation of infection intensity
Inoculation with these doses of Trichinella resulted in intense infections at the muscular level. Muscle biopsy samples for quantitation of intensity of the infection during the experiment weighed from 10 to 240 µg. In 3-week biopsy squash specimens, larvae were visible both free and penetrating into the muscle cells. The intensity increased from 3 weeks postinfection to 6 weeks postinfection but the difference was not significant (Fig. 1). At the termination of the experiment the intensity of infection as determined by tissue digestion was similar for both parasites: on average, 320 larvae/g of tissue (lpg) in T. spiralis-infected animals and 375 lpg in the T. nativa group.
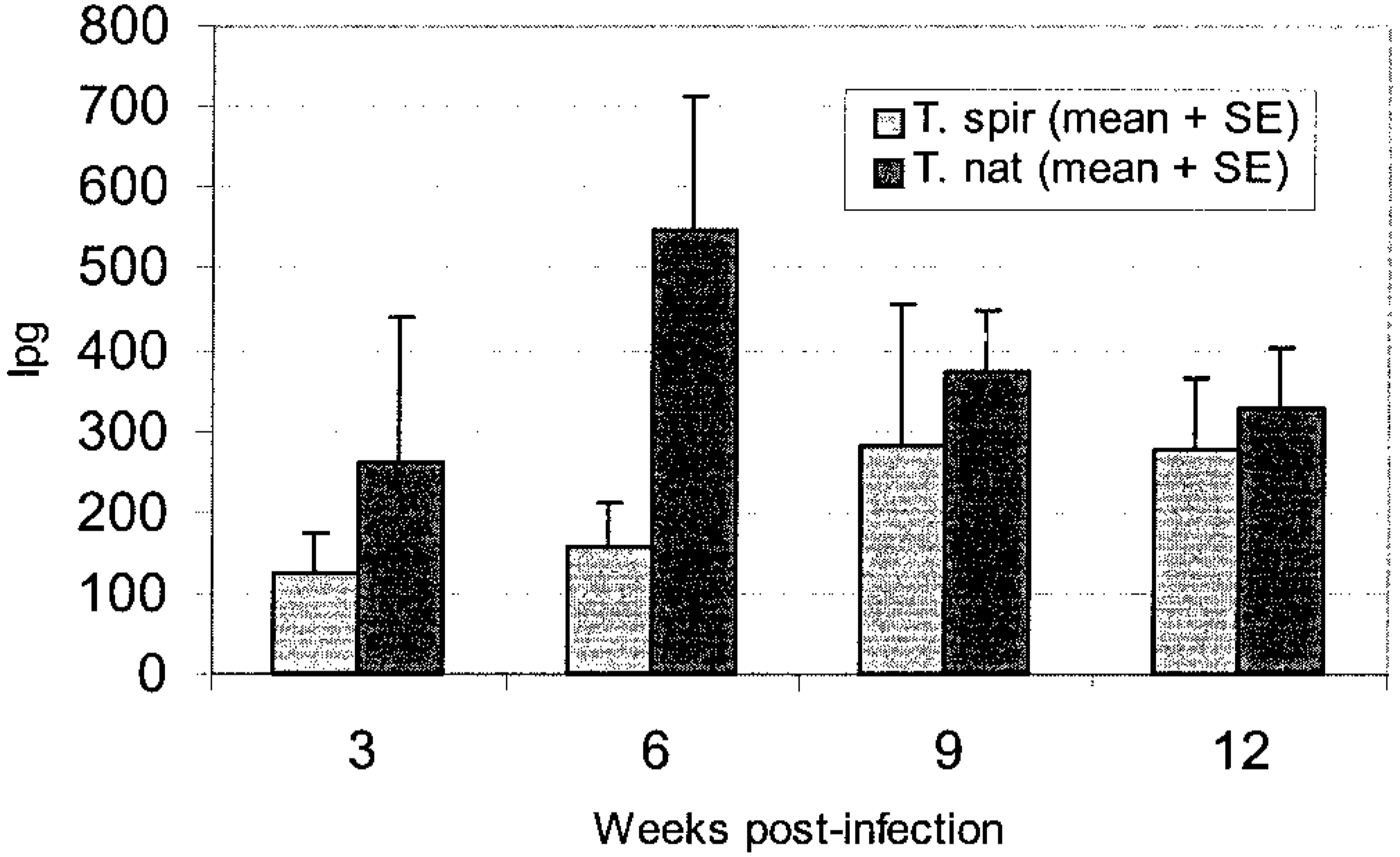
Intensity of Trichinella infection in biopsy samples (larvae per gram, mean + SEM).
Histopathology
Altogether 265 cysts were identified in tissue sections: 173 from the T. nativa group and 92 from the T. spiralis group. In the T. nativa group, at 3 weeks 4/6 animals had parasites, at 6 weeks 5/6 animals had parasites, and at later time all animals had cysts. In the T. spiralis group, at 3 weeks cysts were discovered in all animals, at 6 weeks 4/6 animals had cysts, at 9 weeks 5/6 animals had cysts, and at 12 weeks, 4/6 animals had cysts. When serial sections were examined, cysts were evident in every biopsy specimen from all infected animals at all time.
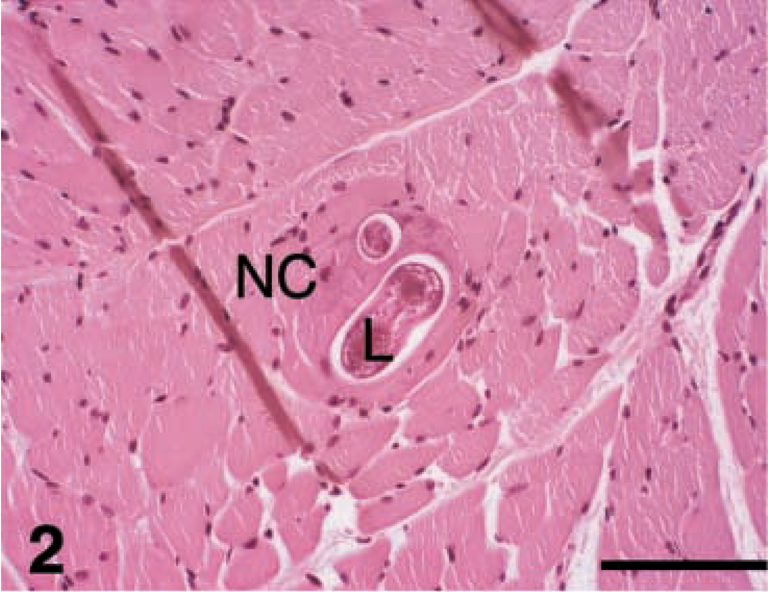
Tissue sections from groups at 3 and 6 weeks (Figs. 2–5) showed early changes in maturation of nurse cells. At the time of entry into the muscle fiber, larvae were oriented parallel with the fiber but thereafter were coiled. Infected myofibrils were swollen, had loss of cross-striations, and became more basophilic with foamy cytoplasm (Fig. 3). Cytologic lesions were segmental (Figs. 3, 4). Nuclei of maturing nurse cells were oval and hypertrophied and had one or two nucleoli. In 6-week samples, mature nurse cells were present, with a newly formed capsule (Fig. 5) that was PAS positive and diastase resistant and stained positively for collagen. The capsule in both parasite species became progressively more thickened during maturation but was thicker in T. nativa than in T. spiralis (Table 1). Nurse cells could not be identified as type I or type II fibers.
Image analyses∗ of the Trichinella tissue cysts in raccoon dogs, 12 week postinfection.
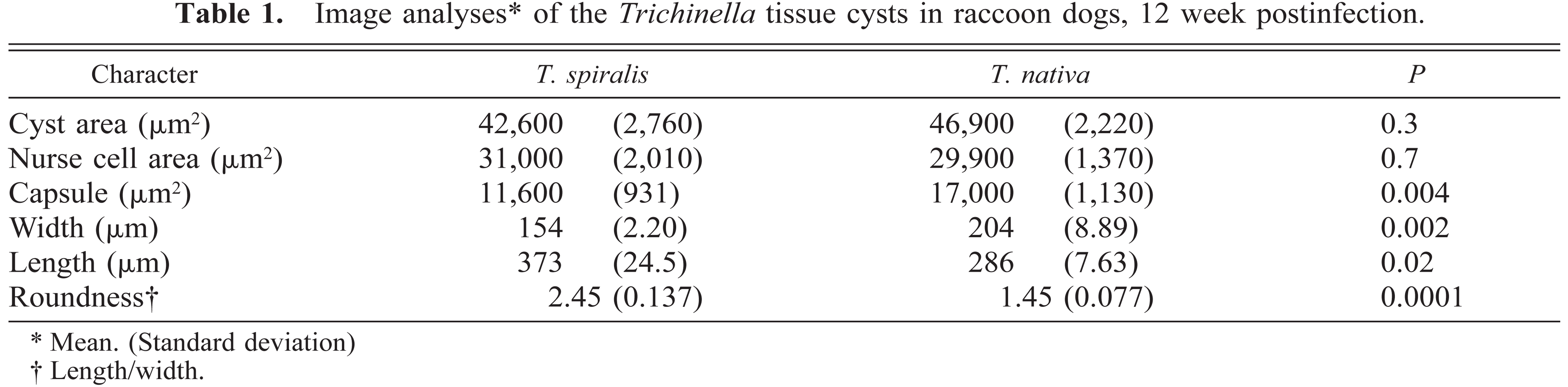
Mean. (Standard deviation)
Length/width.
The shape of the cyst was round, transverse to the muscle fibers (Fig. 2) and elongated in longitudinal orientation. A clear difference between the parasite species existed in the shape of mature cysts. The longitudinal section of the mature T. spiralis cyst was more elongated and lemon shaped (Figs. 6–9), whereas the mature T. nativa was more spherical.
At 3 weeks postinfection, the cellular reaction around each developing nurse cell was minimal. Inflammatory cells infiltrating at the poles of the cysts in longitudinal sections were more prominent in the T. nativa group. The capillary bed around the cyst was identified as early as 3 weeks and was obvious by 6 weeks. No calcification was detected.
The capsules of the parasitic cysts became thicker in both groups (T. spiralis: F = 8.3, df = 3, 12; T. nativa: F = 40.5, df = 3, 12; P < 0.05). The shape of the T. spiralis cysts showed no change from its initial elliptical form, but the shape of the T. nativa cysts changed over time (F = 20.3, df = 3, 12, P < 0.05), becoming more round. Moreover, the numbers of mononuclear leukocytes and eosinophils at the beginning were similar in both groups, but T. nativa seemed to evoke a stronger response that progressively increased by 12 weeks postinfection (Fig. 10). The inflammatory cell reaction in the T. spiralis group declined after 6 weeks. Other graded variables were very stable over time and did not change between groups.
Morphometry
A total of 85 cysts were measured at 12 weeks postinfection: 31 from T. spiralis-infected animals and 54 from T. nativa-infected animals. Differences in the shapes of the cysts and thicknesses of the capsules observed earlier were confirmed and were significant (Table 1). When the roundness of the cyst was measured by dividing its length by its width, the difference in this character was obvious: the T. nativa cyst was rounder than that of T. spiralis (Table 1). Neither the total cyst area nor the surface area of the nurse cell differed between the species.
Western blot
Sera of infected animals reacted with a number of crude antigens in the larval lysate (Fig. 11). Major bands were at around 100 kd, with a family of bands between 76 and 40 kd, many of them doublets. Sera of T. spiralis-infected animals reacted more strongly to T. spiralis antigen. T. nativa also detected the homologous antigen more strongly, but the cross-reactivity of the antibodies was obvious. When T. spiralis was the antigen, the first specific antibodies were detected 2 weeks postinfection in T. spiralis-infected animals and 2 weeks later in T. nativa-infected animals. When T. nativa lysate served as the antigen, T. nativa-infected animals showed antibodies in 2-week samples, and T. spiralis-infected animals showed these at around 4 weeks.
ELISA
The ELISA revealed a clear homologous tendency. With T. spiralis antigens, animals infected with the same species showed their first signs of seroconversion only 1 week postinfection, and seroconversion was clear at 2 weeks postinfection (Figs. 12, 13). Cross-reactive antibodies in T. nativa-infected animals could be demonstrated, but with lower absorbance and later in the course of the infection (Fig. 12).
Immunocytochemical localization
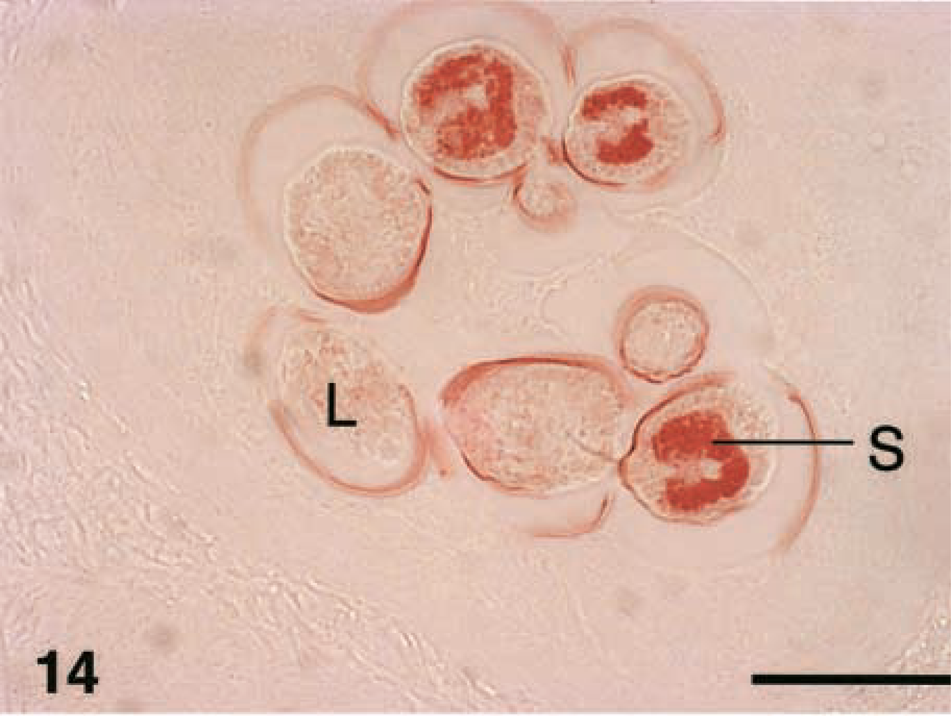
Immunosera both from T. spiralis- and T. nativa-infected animals reacted against cuticula and the internal structures of L1 larva (Figs. 14–16), especially against stichocytes (Fig. 15). No reactivity to the capsule or in the nurse cell structures was identified.
Discussion
Despite the different biochemic, biologica, and molecular biologic characteristics between these two Trichinella species, there are no reports of specific morphology or histopathologic responses of encapsulated Trichinella larvae.2,32–34,40 In the present study, significant differences were identified in the shapes and structures of encapsulated cysts of T. nativa and T. spiralis. Moreover, there were definite species-specific differences in the inflammatory reactions of raccoon dogs to these two parasites. In contrast to our results, Evensen et al.8 reported no differences in capsule formation for T. nativa and T. spiralis in mice. Thus, any differences observed in the raccoon dog may be host-species specific. However, we have also observed T. nativa cysts in foxes and T. spiralis in natural infections in swine and found similar interspecific differences.
Fecundity of T. spiralis and T. nativa in the raccoon dog were the same; both yielded a similar intensity of infection. This finding is in contrast to those for the other host species, in which different Trichinella species have been reported with different intensities.19 No significant differences were noted between time groups, but there were notable differences between muscle groups sampled.23 At 3 weeks postinfection, the majority of the larvae were already located in the muscle tissue, with some larvae still migrating from the gastrointestinal tract into the musculature. Some early nurse cells were observed in 6-week biopsy samples. We could distinguish no difference between the two Trichinella species in migration times of the larvae to the musculature as reported for mice, indicating that migration rate may also be a host species–specific feature.34
Muscle fiber typing is based on the different expression of fast and slow myosin heavy-chain isoforms.14,37 The pH-independent staining pattern of nurse cells with ATPase suggests that Trichinella may influence heavy-chain expression of nurse cells in such a way that multiple isoforms are translated simultaneously, thereby causing failure to stain as either type I or type II myocytes. This hypothesis is supported by the results of Jasmer et al.,18 who reported altered expression or translation; they used one monoclonal antibody against the myosin heavy chain but detected reduced immunoreactivity in T. spiralis-infected nurse cells compared with that in uninfected myocytes or T. pseudospiralis-infected muscle cells.18 Other studies of nurse cell physiology have shown both up-5 and downregulation of protein expression in comparison to that of the normal myocyte.18,46
The thicker capsule around T. nativa could in part be explained by a stronger cellular reaction around the cyst. However, the influence of inflammatory cells is not the sole explanation for the formation of the capsule because the capsule also has been reported in immunosuppressed animals.12,42 The important structures of the capsule are types VI and IV collagen.31 Trichinella itself orchestrates type IV collagen composition by its own excretory/secretory protein products.13 Cyst-forming T. spiralis differed from nonencapsulating T. pseudospiralis, which evoked less collagen synthesis. If the thicker capsule and more evident and persistent inflammatory reaction indicates that T. nativa is metabolically more active than T. spiralis in the raccoon dog, it might be worth studying whether the thicker capsule is associated with the freeze-resistance of T. nativa.
Experimental infections of other animals with Trichinella species have shown that the time required for seroconversion depends on parasite dose.38,41 Raccoon dogs developed antibodies rapidly, and reactivity was stronger against homologous antigens. Thus, with recombinant antigens it may be possible to develop species-specific serologic assays. Serologic studies with horses have shown that the level of circulating antibodies peaks at 5–7 weeks postinfection but then drops after 11 weeks, and only very low levels can be detected at 17–24 weeks postinfection.47 Circulating antibodies began to decrease in T. nativa-infected swine after the peak at 5 weeks postinfection, but this decrease was not seen in T. spiralis-infected animals.19 Similar parasite species-specific differences were not seen in the raccoon dog. In the present study, the levels of circulating antibodies reached a plateau with both parasites during the 3-month follow-up period. Our experimental model using siblings obviously minimized individual-origin variation seen in swine experiments.38
Antigenic epitopes and monoclonal antibodies have been studied mainly for T. spiralis.7,20 Some of the research has focused on tyvelose-bearing antigens involved in penetration of the gut epithelium.22 Characterization of T. spiralis antigens has shown that some are stage specific.30 Polyclonal sera showed clear reactivity against the cuticula of muscle-stage larvae and stichosomes and less reactivity against the other somatic larval structures.6,26 The stichosome give rise to well-known excretory-secretory antigens commonly used in ELISA tests.11,17,24,35,39 The absence of immunogenic epitopes on the capsule suggests that the capsule provides good protection from the host's defense mechanisms. It may also indicate that the capsule is mainly formed by the host rather than the parasite.
Interest has focused on characterizing differences between the encysted and noncapsulated species T. spiralis and T. pseudospiralis.21,43 Our results indicate that two encysted species, T. spiralis and T. nativa, show distinct reactions in the host tissues. Despite these different reactions, both Trichinella species breed well in the raccoon dog. Future studies may explain why the raccoon dog is such a versatile host for Trichinella.
Trichinella spiralis (
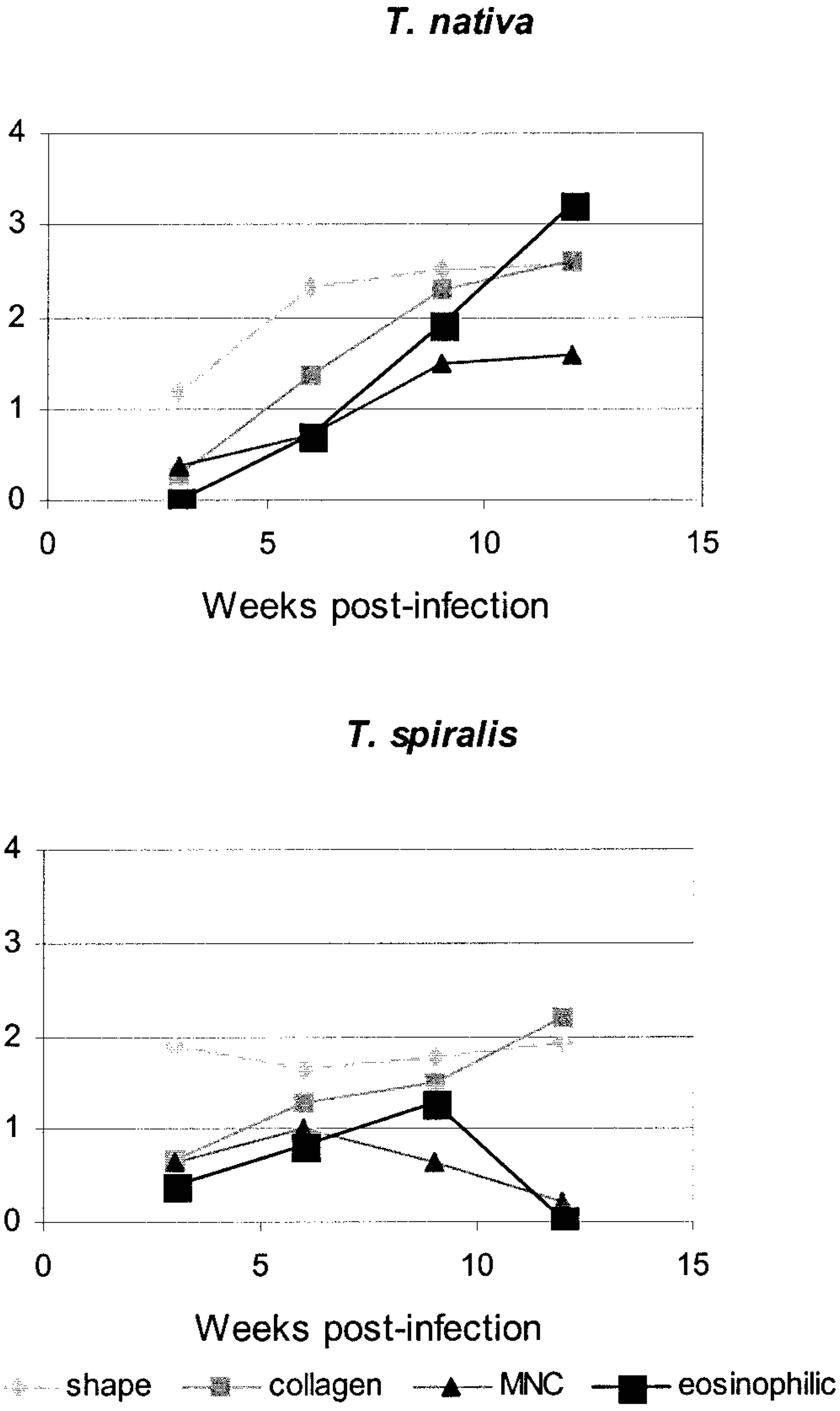
Changes in histopathologic grading during the experiment. Shape = roundness; collagen = thickness of the cyst capsule; MNC = mononuclear cells; eosinophilic = amount of eosinophils around the cyst.
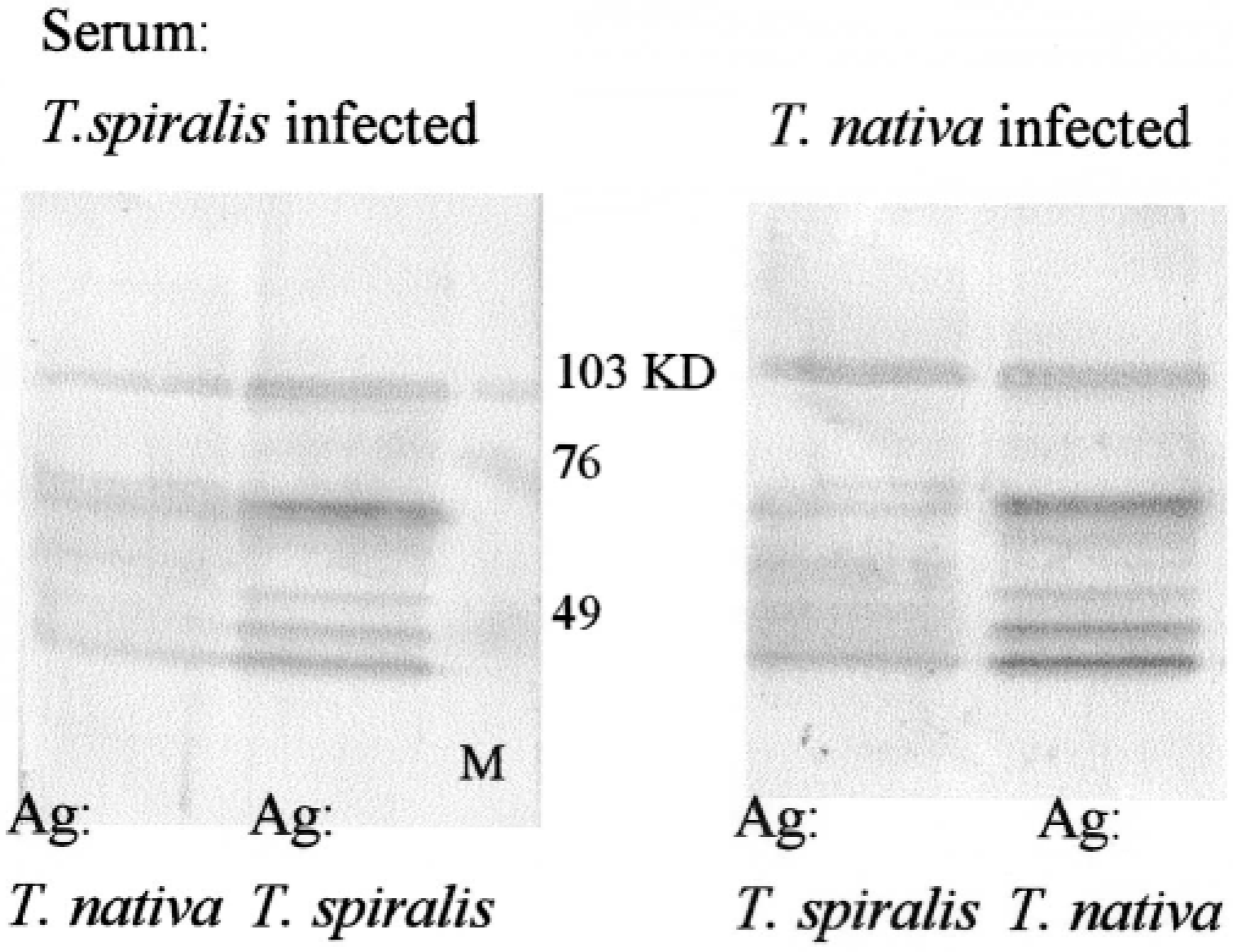
Western blots immunostained with sera from T. spiralis- or T. nativa-infected raccoon dogs. More intense reaction is present with homologous antigen (right lanes). M = markers.

Group average absorbances (± SEM) with homologous (T. nativa Ag/T. nativa Ab, T. spiralis Ag/T. spiralis Ab) and heterologous (T. nativa Ag/T. spiralis Ab, T. spiralis Ag/T. nativa Ab) antigens in ELISA with serum from raccoon dogs (diluted 1:100) at 12 weeks postinfection. Absorbances of heterologous and homologous antigen differed significantly (P = 0.007, t-test).
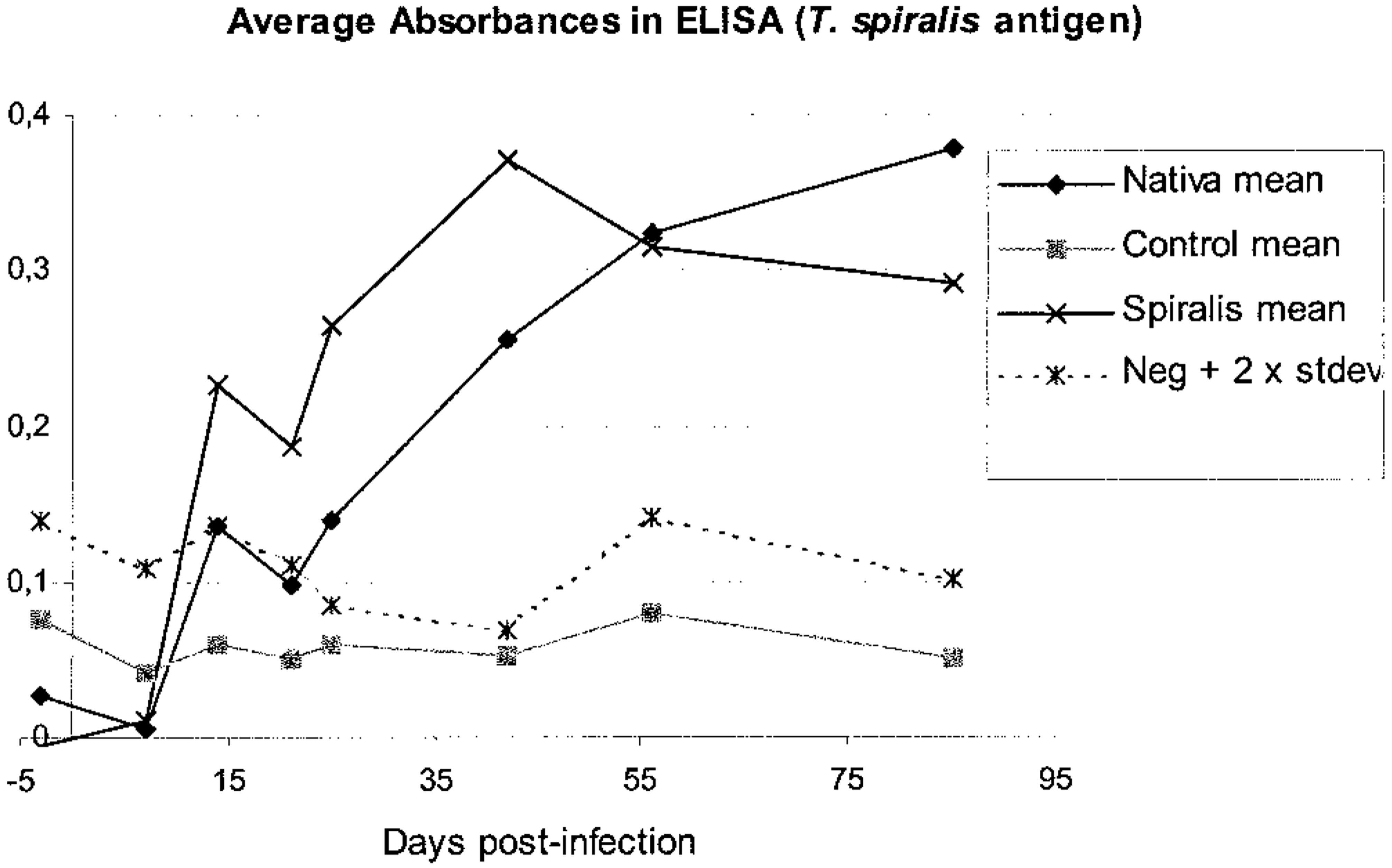
ELISA absorbances in different study groups of raccoon dogs. Serum dilution 1:100, antigen from T. spiralis lysate.
Footnotes
Acknowledgements
We acknowledge Ilpo Forsman for his expert help in handling the animals, Luci-Britt Labbas and Pirkko Niemelä for help with lab work, and Carolyn Norris, for editing the English. This study has been supported by grants from the Faculty of Veterinary Medicine, the Emil Aaltonen Foundation (L. Oivanen) and the Marjatta and Eino Kolli foundation (T. Mikkonen).
