Abstract
A retrospective study was performed to characterize malignant lymphomas of 16 Simian immunodeficiency virus (SIV)-infected rhesus monkeys (Macaca mulatta), 2–9 years of age, on the basis of clinical data, histologic and immunophenotypic results, and cell death indices compiled with the TdT-mediated X-duTP nick end labeling method. We particularly focused on providing immunohistochemical evidence of expression products of EBNA2, Bcl2, c-Myc, P21, P53, and Bc16. Results were compared with data from the literature on human HIV-associated lymphomas. According to the updated Kiel classification, the lymphomas were classified as 11 centroblastic lymphomas, three immunoblastic lymphomas, one Burkitt-like lymphoma, and one immunocytoma. Using antibodies to CD20, the B-cell origin of tumor cells was demonstrated. SIV antigen was not demonstrated in the tumor cells. Infection with rhesus lymphocryptovirus was present in 94% of the monkeys. Lymphomas revealed expression of Bc12 in 15/16 (94%), c-Myc in 14/16 (88%), P21 in 10/16 (63%), P53 in 12/16 (75%), and Bc16 in 1/16 (6%) monkeys. This study provided evidence that the expression of these gene products, which are thought to play an important role in cell proliferation and apoptosis in HIV- and non-HIV-associated lymphomas, are also involved in the pathogenesis of lymphomas in SIVinfected rhesus monkeys. A tentative relationship between the described gene products and the cell death indices was established for the expression of Bc12. The present primate model represents a suitable animal model for studying the pathogenesis of AIDS-associated lymphomas.
High-grade non-Hodgkin's lymphomas are regarded as the most frequent malignant diseases in HIV-infected patients. Since 1985, these lymphomas have been classified as AIDS-defining diseases. 54 Approximately 4–10% of HIV-infected patients have malignant lymphomas. 40 In general, most lymphomas develop late in the course of the disease, and as life expectancy of HIV-infected patients increases as a result of improvements in antiviral therapy, the frequency of the HIV-associated non-Hodgkin's lymphomas (HIV-NHL) also rises. 4,21,62 Although these lymphomas have several features in common, e.g., frequent development of a generalized lymphadenopathy (GLP) prior to lymphoma, aggressive clinical course, high malignancy, B-cell origin, and extranodal growth in unusual sites including gastrointestinal tract, orbita, myocardium, skeletal muscles, lungs, liver, gingiva, kidneys, gonads, and adrenal glands, they vary considerably in terms of histologic appearance, occurrence during the clinical course of the disease, and underlying genetic alterations. 4,21,24,40,54 In accordance to the updated Kiel classification, these lesions are classified as Burkitt and Burkitt-like lymphomas, centroblastic, and immunoblastic lymphomas. In accordance to the new World Health Organization (WHO) lymphoma classification, they represent Burkitt lymphomas, atypical (pleomorphic) Burkitt lymphomas, and diffuse large-cell lymphomas, with the variants centroblastic and immunoblastic. Furthermore, AIDS-associated primary effusion lymphomas, also known as AIDS–body cavity-based lymphomas, and AIDS–primary central nervous system (CNS) lymphomas do occur. 22–24,40 With the exception of HIV–Burkitt lymphomas, which can develop in early stages of immunodeficiency, the HIV-NHL occur in the presence of low CD4+ T-cell counts. 7,21,30 Infection with Epstein-Barr virus (EBV) types I and II seems to have an important pathogenic role, especially the expression of EBNA2 (EBV-associated nuclear antigen 2) and LMP1 (latent membrane protein 1). 30,40 AIDS–body cavity-based lymphomas are consistently associated with human herpesvirus 8.
The molecular pathways contributing to HIV–Burkitt lymphomas include c-myc rearrangements in 100% of cases, with a resulting overexpression, p53 mutations in 60%, bc16 mutations in 70%, and EBV infections in 30–40% of cases. 1,21,24,25,30 EBNA2 and LMP1 are generally not expressed. Burkitt-like lymphomas exhibit c-myc rearrangements in 20%, p53 mutations in 0%, EBV infections in 30%, and bc16 rearrangements in 0% of cases. 23 The genetic pathway of diffuse large-cell lymphomas comprises c-myc translocations in 20%, p53 mutations in 0%, bc16 rearrangements in 20% and mutations in 65%, and EBV infections in 70–80% of cases. Expression of EBNA2 and LMP1 may occur in these lymphomas. 1,22,30 An expression of Bc12 can be detected in 36% of HIV-NHL 61 and in most of the AIDS–primary CNS lymphomas. 7
Rhesus (Macaca mulatta) or cynomolgus (Macaca fascicularis) monkeys infected with the simian immunodeficiency virus (SIV) develop typical symptoms of immunodeficiency, including opportunistic infections and decrease in CD4+ T cells and in the CD4+/CD8+ T-cell ratio. 3,35,44 Since 1983, several studies have reported on malignant lymphomas in macaques infected with SIV and suffering from an AIDS-like syndrome. 2,3,10,20,27,33,38,57 These lymphomas share significant similarities with the HIV-associated lymphomas in humans and can serve as an animal model for the investigation of HIV-NHL in humans. 18,27,48
Eight cases of malignant lymphoma in SIV-infected rhesus monkeys have been characterized morphologically and immunophenotypically. 50 Imunohistochemistry and in situ hybridization results showed a high prevalence of the rhesus homologue of the Epstein–Barr virus, rhesus lymphocryptovirus (rhLCV), which may play a key role in the pathogenesis of these lymphomas.
The goal of the present study was the histologic classification and immunophenotypic characterization of eight new lymphoma cases in SIV-infected rhesus monkeys and the immunohistochemical investigation of the expression of several cell-cycle and apoptosis-related gene products in all 16 cases of malignant lymphoma that occurred since 1990 at the German Primate Center.
Materials and Methods
Tumor material
Tumor samples were obtained from six female and 10 male rhesus monkeys 2–9 years of age that were infected with different SIV isolates (Tables 1, 2) as part of several SIV infection and immunization studies carried out in the Department of Virology and Immunology of the German Primate Center. Monkeys that had clinical symptoms of advanced immunodeficiency and neoplastic diseases were euthanatized, and necropsy was carried out immediately. For monkey Nos. 14 and 16, which died spontaneously, tissue collection and fixation was done several hours post mortem. Tumor tissue was fixed in 4% and 10% formalin and embedded in paraffin.
Data for rhesus monkeys concerning survival time postinfection (p.i.), pathologic findings, and histologic classification of lymphomas.

Data concerning immunologic status pre- and postinfection∗ (pr.i. and p.i.), SIV isolates, and occurrence of GLP.
∗ A few weeks prior to or at time of necropsy.
† NA = not available.
Histology and immunohistochemistry
For histology, 4-µm sections were stained with hematoxylin and eosin and Giemsa. Lymphomas were classified according to the updated Kiel classification. Immunophenotyping was carried out with antibodies against CD3, CD20, CD68, IgA, IgG, IgD, IgM, and kappa and lambda antigens. Antibodies against Ki67 for the detection of cycling cells in the G1 to M phase, against EBNA2, and KK75 against SIVnef antigen also were applied. 50 To identify P53, P21, c-Myc, Bc12, and Bc16 protein in the lymphomas, the monoclonal antibodies DO-7, SX118, 9E11, 124, and P1F6 (Table 3) were employed.
Antibodies used for immunohistochemical investigations.
Immunohistochemistry was performed using the streptavidin–biotin complex method. The 4-µm paraffin-embedded sections were mounted on coated slides. After appropriate pretreatment, the slides were placed in 0.5% hydrogen peroxide for 30 minutes. After washing in Tris-buffered saline (TBS), the slides were incubated with inactivated normal goat serum for 20 minutes. The primary antibodies diluted in phosphate-buffered saline (PBS) were applied at 4 C overnight. After washing, visualization was carried out with the DAKO duett kit (StreptABComplex/HRP Duett, mouse/rabbit, Dako, Hamburg, Germany) with diaminobenzidine as chromogen. The tissue slides were counterstained with hematoxylin, dehydrated, and mounted. A positive control slide was used with each batch of tumor sections stained. In most cases, nonneoplastic lymphoid tissue in the same section served as an internal positive control. Appropriate negative controls were performed according to the immunoperoxidase techniques previously described. 52 As negative control reagent, the antibody diluent from DAKO (S3022) was used.
The percentage of the positive tumor cells was estimated in 10 high-power fields (HPF), and slides graded semiquantitatively using the following modified application of other scoring systems: 10,60 − = no staining of tumor cells; (+) = 5% to <10% of tumor cells positive; + = 10% to <30% positive; ++ = 30% to <60% positive; +++ = 60–100% positive.
Evaluation of cell death indices
For the in situ detection of the fragmentation of genomic DNA in the tumor cells during apoptosis, the TUNEL (TdT-mediated X-dUTP nick end labeling) assay was applied on 4-µm paraffin-embedded sections, using the following protocol. After deparaffinizing and rehydrating, the slides were treated with 5% proteinase K solution and washed in ice-cold TBS. The slides were then incubated with a tailing mix containing the terminal transferase, reaction buffer (Roche, Mannheim, Germany), and digoxigenin DNA labeling mix (Roche) for 60 minutes at 37 C. After washing in TBS and blocking with fetal calf serum (FCS), the slides were incubated with anti-digoxigenin antibodies diluted in FCS for 60 minute at room temperature. Color development was performed with nitro blue tetrazolium chloride/5-bromo-4-chloro-3-indolyl phosphate salt solution for a few minutes until appropriate staining was present. Slides were counterstained with nuclear fast red and mounted. At least one tissue section of colon, SIV-positive and SIV-negative lymph nodes, and ovaries of rhesus monkeys and human tonsil sections served as positive controls in each reaction. Tissue sections of brain and positive control sections, where the terminal transferase was replaced by water, were used as negative controls.
Because the TUNEL assay may also label cells undergoing other types of cell death and did not mark cells in the early phases of apoptosis, the absolute amount of apoptotic cells cannot be determined by the TUNEL assay and morphologic criteria alone. However, this method was used for evaluating the cell death index (DI) of the lymphomas because only paraffin-embedded material was available. The DI, which is the number of dying cells per 100 tumor cells, was evaluated by counting the number of labeled cells within 1,000 tumor cells using the software Openlab (Improvision, Coventry, UK). All cells exhibiting the typical morphologic stages of apoptosis combined with positive TUNEL staining were counted. Macrophages with engulfed apoptotic bodies were counted as one dying cell.
Statistical methods
The results of the immunohistochemical investigations and the cell death indices were compared with the Wilcoxon two-sample test. The immunohistochemical data were compared with Fisher's exact test.
Results
Clinical and pathomorphologic findings
The lymphomas occurred 7–26 months postinfection during advanced immunosuppression with low CD4+ T-cell counts and CD4+/CD8+ T-cell ratios (Table 2). In 13/16 monkeys (81%, Nos. 1–3, 5–10, 12, 13, 15, 16), GLP developed prior to lymphomas in the first weeks postinfection (Table 2). In addition to the lymphomas, most of the monkeys suffered from various opportunistic infections, e.g., with cytomegalovirus (CMV), Pneumocystis carinii, or Cryptosporidia sp. Other pathologic findings are listed in Table 1.
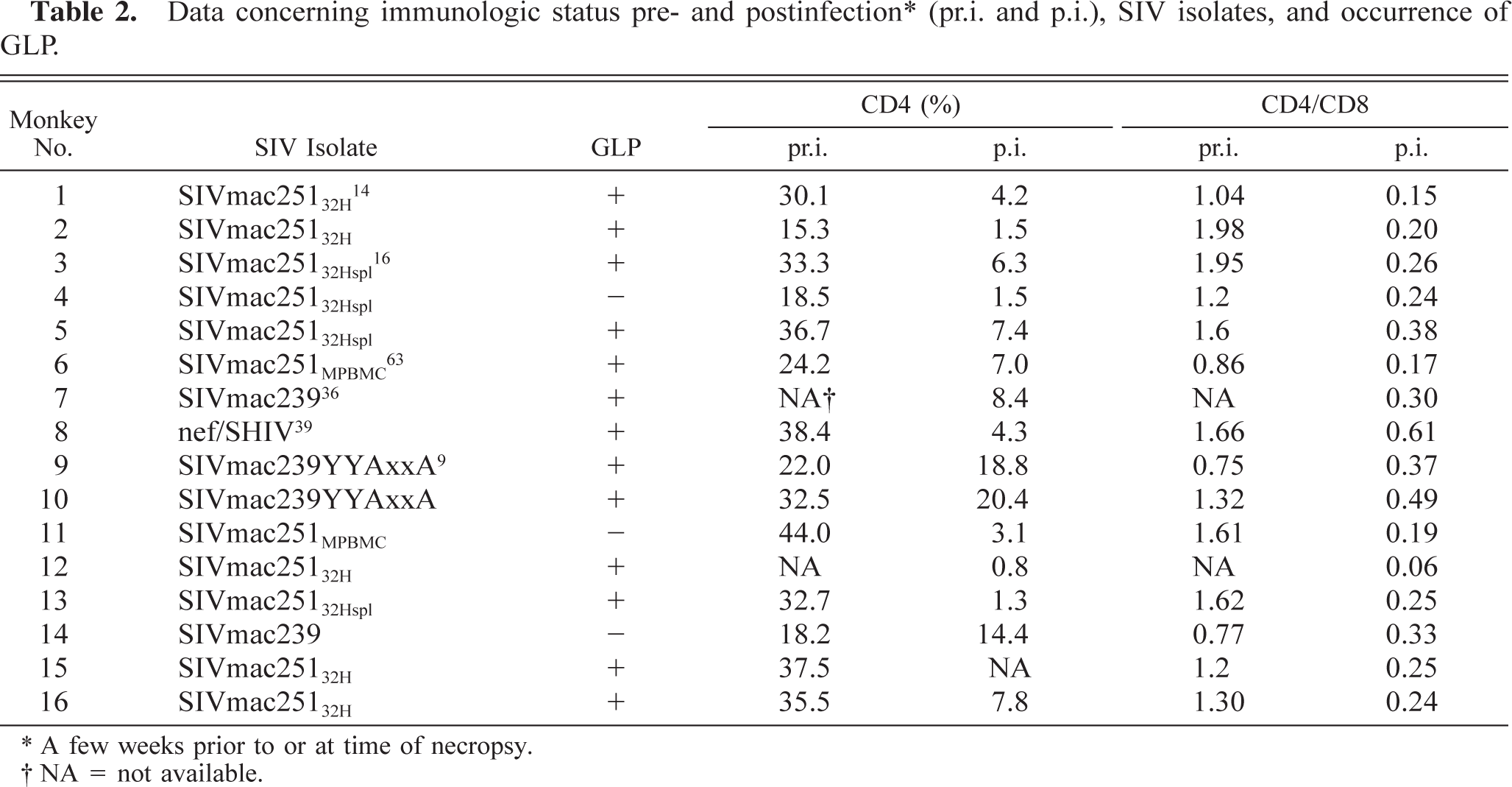
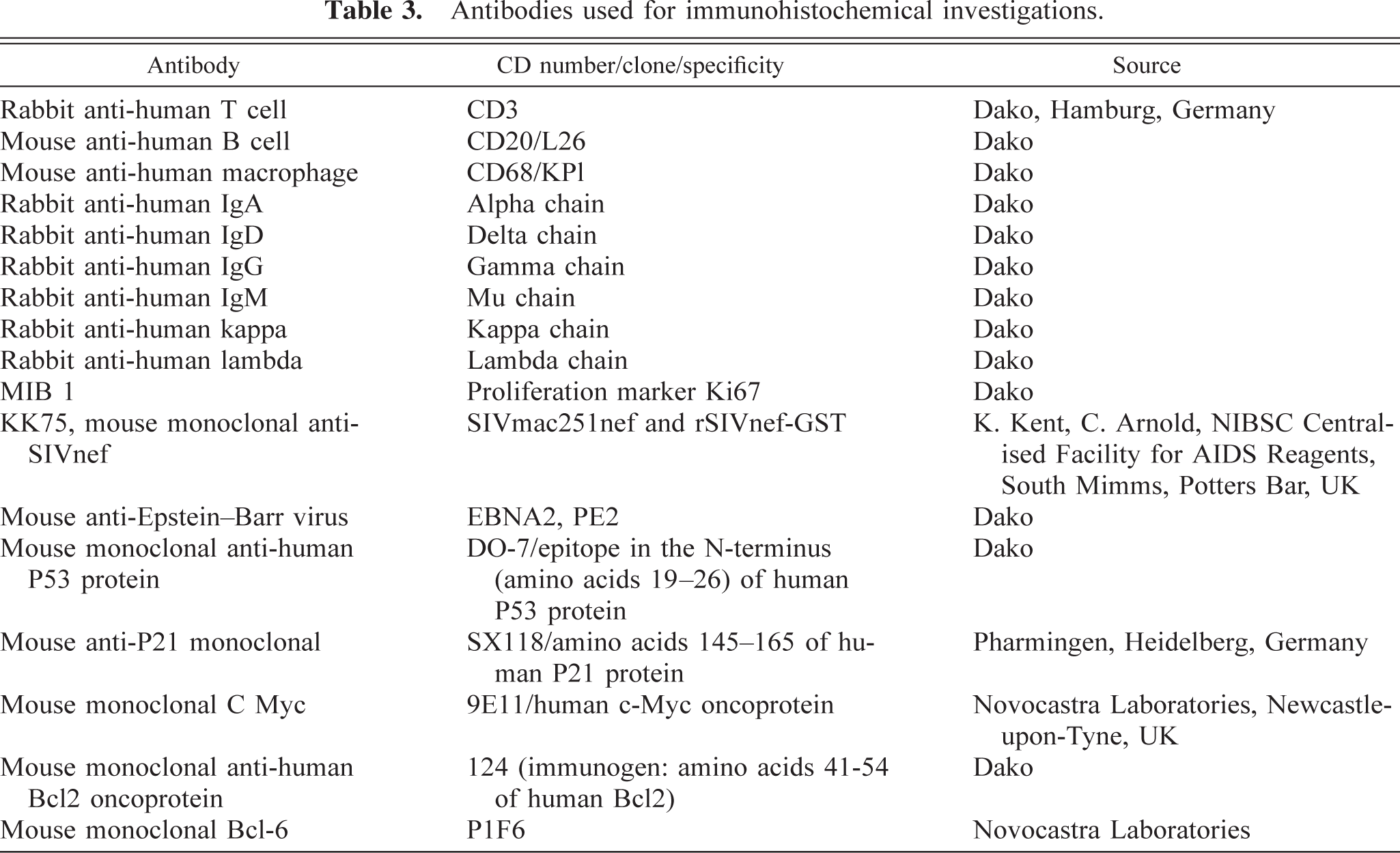
In 14/16 monkeys (88%), the lymphomas occurred at more than one site, with frequent involvement of extranodal organs such as gastrointestinal tract, kidneys, and periocular tissues. Other sites affected were nose cavities, mediastinum, greater omentum, pelvic cavity, skeletal muscles, heart, lung, pancreas, liver, testes, urinary bladder, and CNS (Fig. 1). According to the updated Kiel classification, these lesions were classified as 11 centroblastic lymphomas, three immunoblastic lymphomas, one Burkitt-like lymphoma (Fig. 2), and one immunocytoma (lymphoplasmacytoid lymphoma) (Table 1). According to the new WHO lymphoma classification, these lymphomas represent diffuse large B-cell lymphomas with the variants centroblastic and immunoblastic, atypical Burkitt lymphoma, and B-cell chronic lymphocytic lymphoma with plasmacytoid differentiation.
Brain stem; monkey No. 9. Centroblastic lymphoma of monomorphic subtype with infiltrative angiocentric growth at the margins. Giemsa stain. Bar = 26 µm.
Burkitt-like lymphoma; monkey No. 15. Cohesive growth of relative monomorphic, medium to large-size cells. Giemsa stain. Bar = 17 µm.
Immunophenotype
All lymphomas expressed the B-cell marker CD20 and contained small numbers of infiltrating CD3+ lymphocytes (Figs. 3, 4). Eleven of 16 lymphomas (69%) were diffusely interspersed with CD68+ macrophages. In one monkey (No. 15), multinucleated giant cells positive for CD68 and SIV antigen were detectable. Light- and heavy-chain antigen was detected in the following pattern: IgM kappa, IgA kappa, IgG, and IgM, each in two monkey and IgA in one monkey. Two lymphomas expressed IgG and IgD, respectively, in addition to IgM kappa. In one monkey with an immunoblastic lymphoma (monkey No. 14), kappa and lambda antigens were expressed in similar proportions in addition to IgG. In the Burkitt-like lymphoma and in the immunocytoma, no light- and heavy-chain antigens could be detected by immunohistochemistry. These results are listed in Table 4. Part of the data were published previously. 50
Immunophenotype and Ki67 expression∗ of the SIV-NHL.

∗ S = single cells positive; - = no staining of tumor cells, (+) = 5% to <10% of tumor cells positive; + = 10% to <30% positive; ++ = 30% to <60% positive; +++ = 60–100% positive.
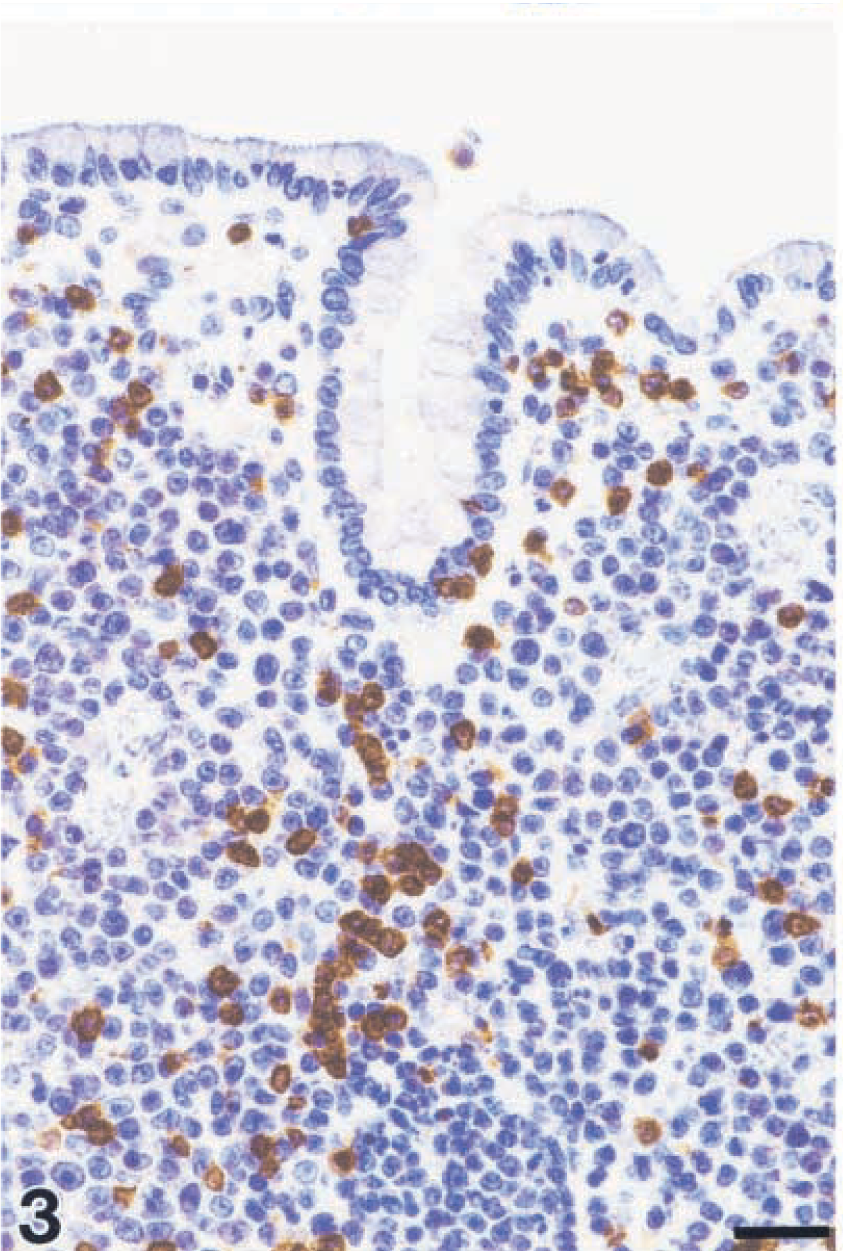
Centroblastic lymphoma; monkey No. 6. Monomorphic subtype with few infiltrating CD3+ cells. Streptavidin–biotin complex method. Bar = 26 µm.
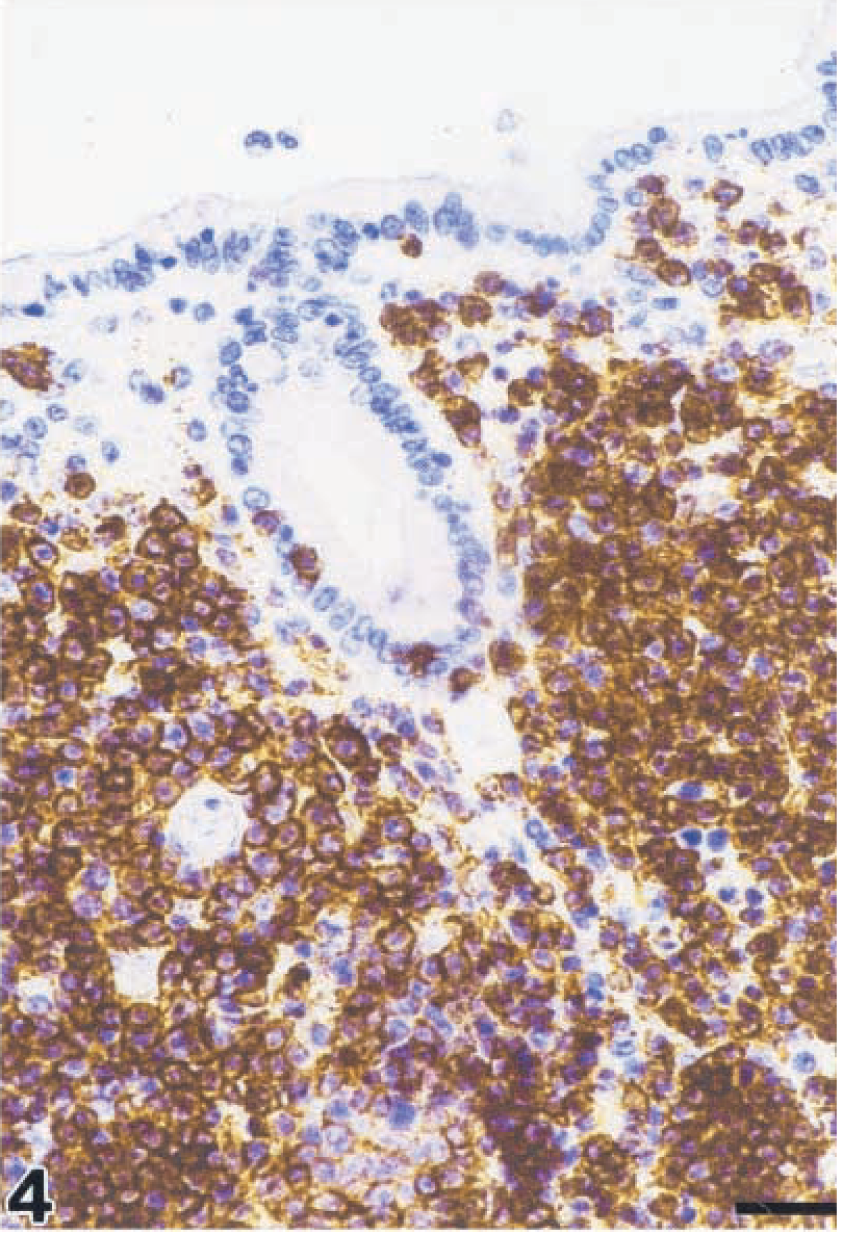
Centroblastic lymphoma; monkey No. 6. Monomorphic subtype. Most of the tumor cells are positive for the B-cell marker CD20. Streptavidin–biotin complex method. Bar = 26 µm.
Expression of virus-, proliferation-, and apoptosis-related gene products
The tumor cells were negative for SIV antigen nef, but in 3/16 monkeys (19%, Nos. 4, 13, 15), diffusely interspersed single lymphocytes or macrophages were positive. The EBNA2 homologue of the rhLCV was present in 12/16 monkeys (75%, Nos. 3–12, 14, 16) (Tables 5, 6).
Antigen expression pattern and cell DI of the SIV-NHL.

∗ S = single cells positive; SM = single multinucleated giant cells positive; = no staining of tumor cells; (+) = 5% to <10% of tumor cells positive; + = 10% to <30% positive; ++ = 30% to <60% positive; +++ = 60–100% positive.
Frequency of expression∗ of EBNA2, c-Myc, Bcl2, and Bcl6 in the SIV-associated lymphomas in relation to the histologic classification.
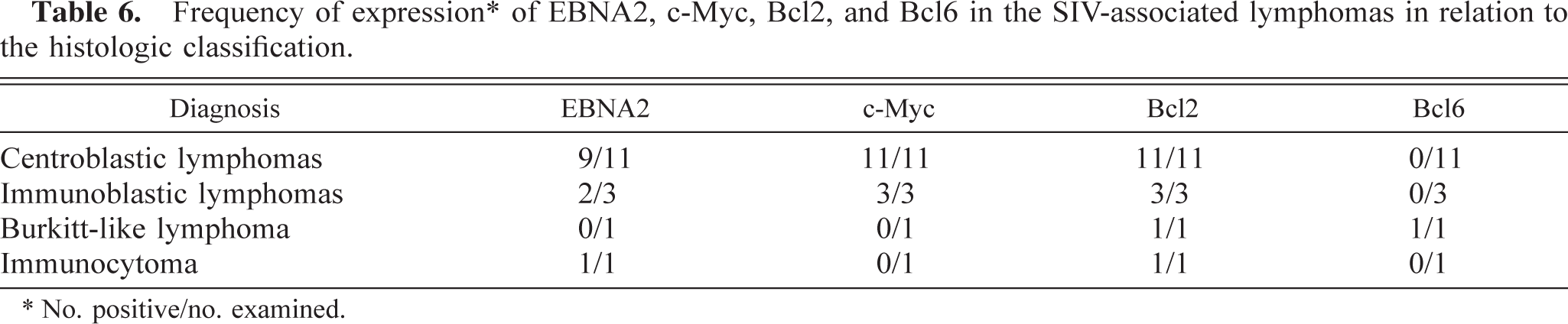
∗ No. positive/no. examined.
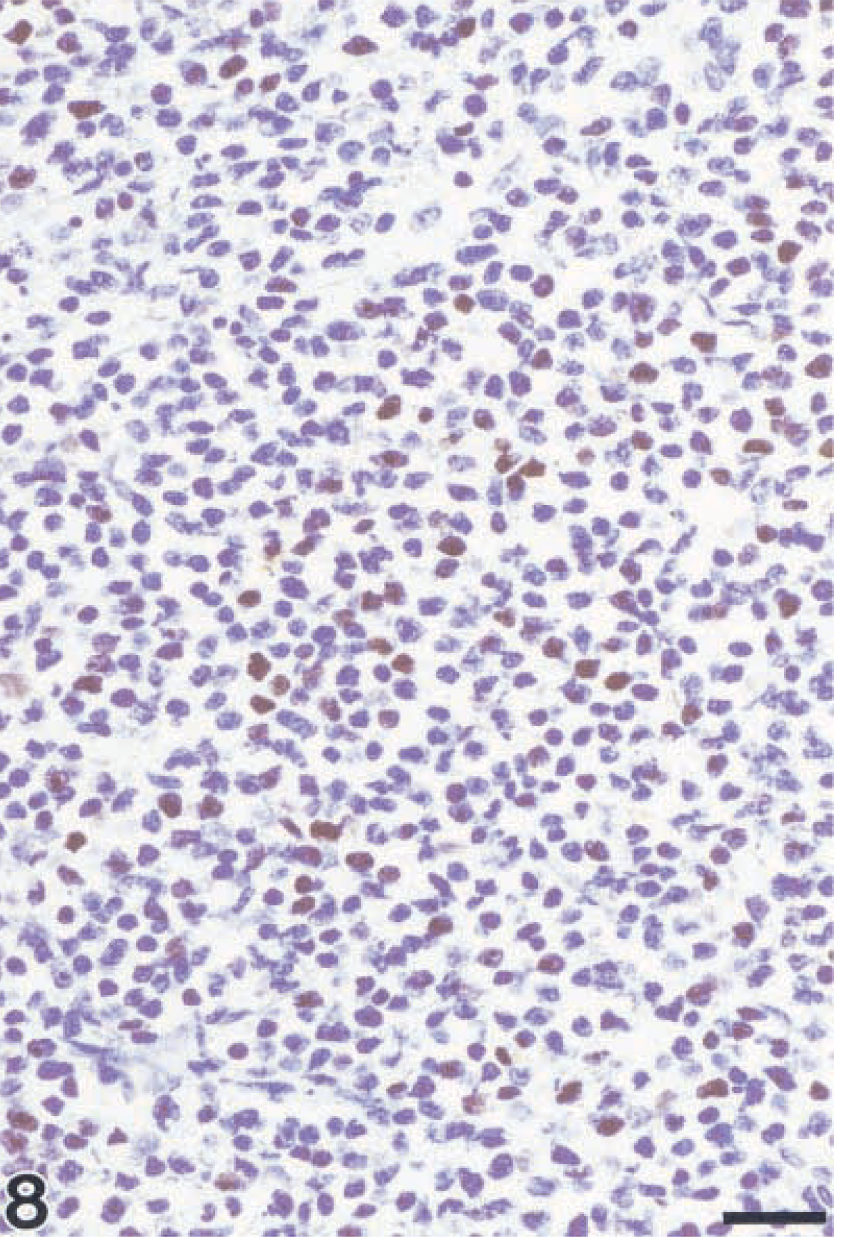
Antibodies to P53 revealed nuclear staining of tumor cells (Fig. 5). Faintly stained nuclei, present in most of the cases, were excluded. 12 A distinct positive staining was observed in 12/16 monkeys (75%, Nos. 1–11, 13) in up to 20% of tumor cells.
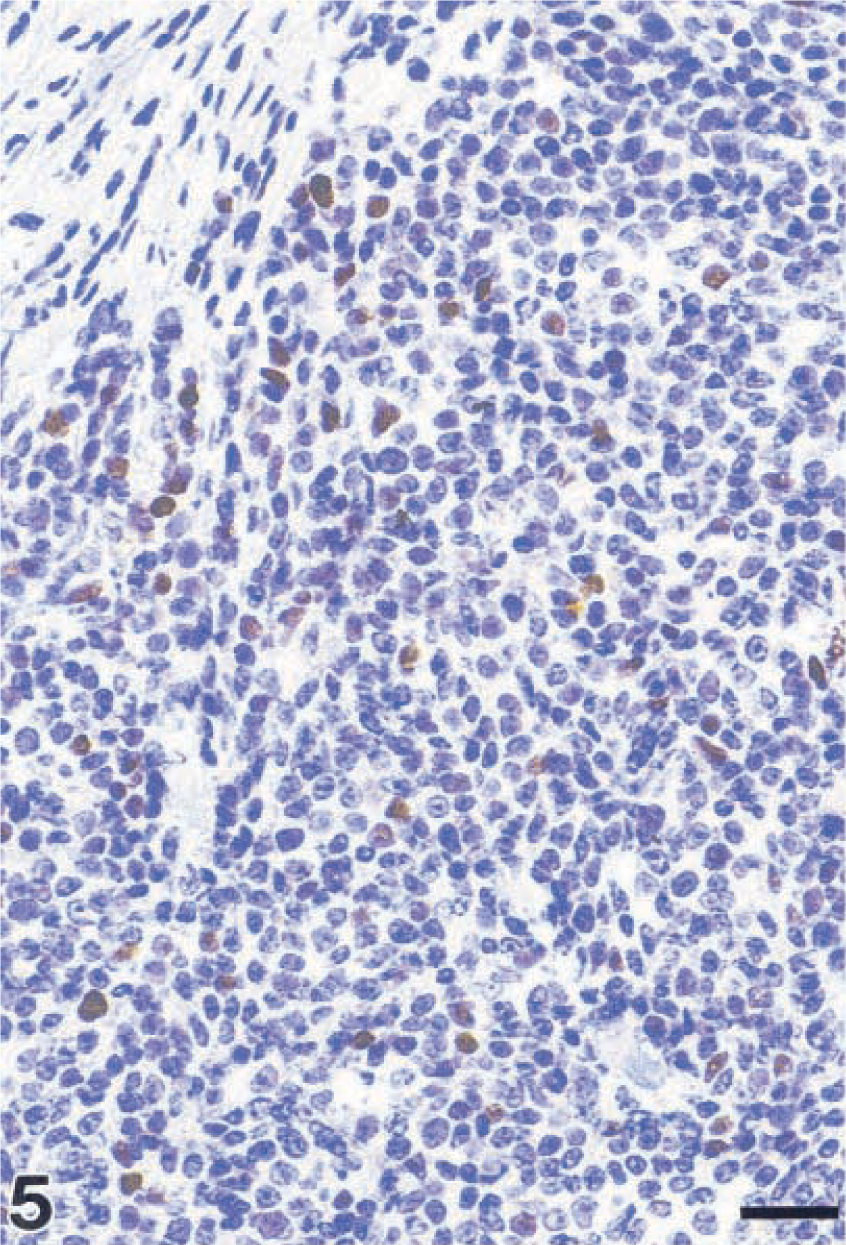
Centroblastic lymphoma; monkey No. 4. Polymorphic subtype. Immunohistochemical nuclear staining of tumor cells for P53. Streptavidin–biotin complex method. Bar = 26 µm.
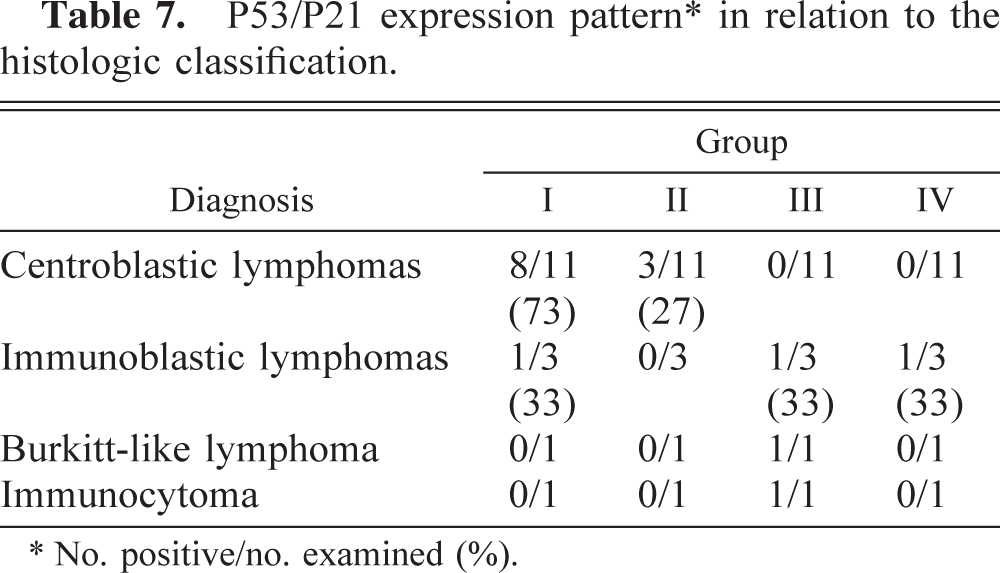
P21 protein was demonstrated in 10/16 monkeys (63%, Nos. 1, 2–5, 8–13) in up to 50% of tumor cell nuclei (Fig. 6). The combined expression pattern of P53 and P21 in these lymphomas can be used to distinguish four different groups: I = phenotype P53+/P21+; II = P53+/P21−; III = P53-/P21−; IV = P53−/P21+. The distribution of expression can also be used to distinguish four different groups: I = 9/16 monkeys (56%, Nos. 1, 3–5, 8–11, 13); II = 3/16 monkeys (19%, Nos. 2, 6, 7); III = 3/16 monkeys (19%, Nos. 14–16); IV = 1/16 monkeys (6%, No. 12). In group I, the proportion of P21+ cells was equal to or greater than the proportion of P53+ cells. Of the centroblastic lymphomas, 8/11 (73%, monkey Nos. 1, 3–5, 8–11) were in group I and 3/11 (27%, monkey Nos. 2, 6, 7) were in group II. No other expression pattern was observed in this entity. The immunoblastic lymphomas belonged to groups I, III, and IV (one lymphoma each). The Burkitt-like lymphoma and the immunocytoma were classified as group III (Table 7).
P53/P21 expression pattern∗ in relation to the histologic classification.
∗ No. positive/no. examined (%).
Centroblastic lymphoma; monkey No. 4. Polymorphic subtype. Immunohistochemical nuclear staining of tumor cells for P21 in the same place as shown in Fig. 5. Streptavidin–biotin complex method. Bar = 26 µm.
Expression of c-Myc was detected in 14/16 lymphomas (88%) as a typical granular cytoplasmatic staining in proximity to the nucleus in up to 90% of the cells. The Burkitt-like lymphoma and the immunocytoma were negative for c-Myc.
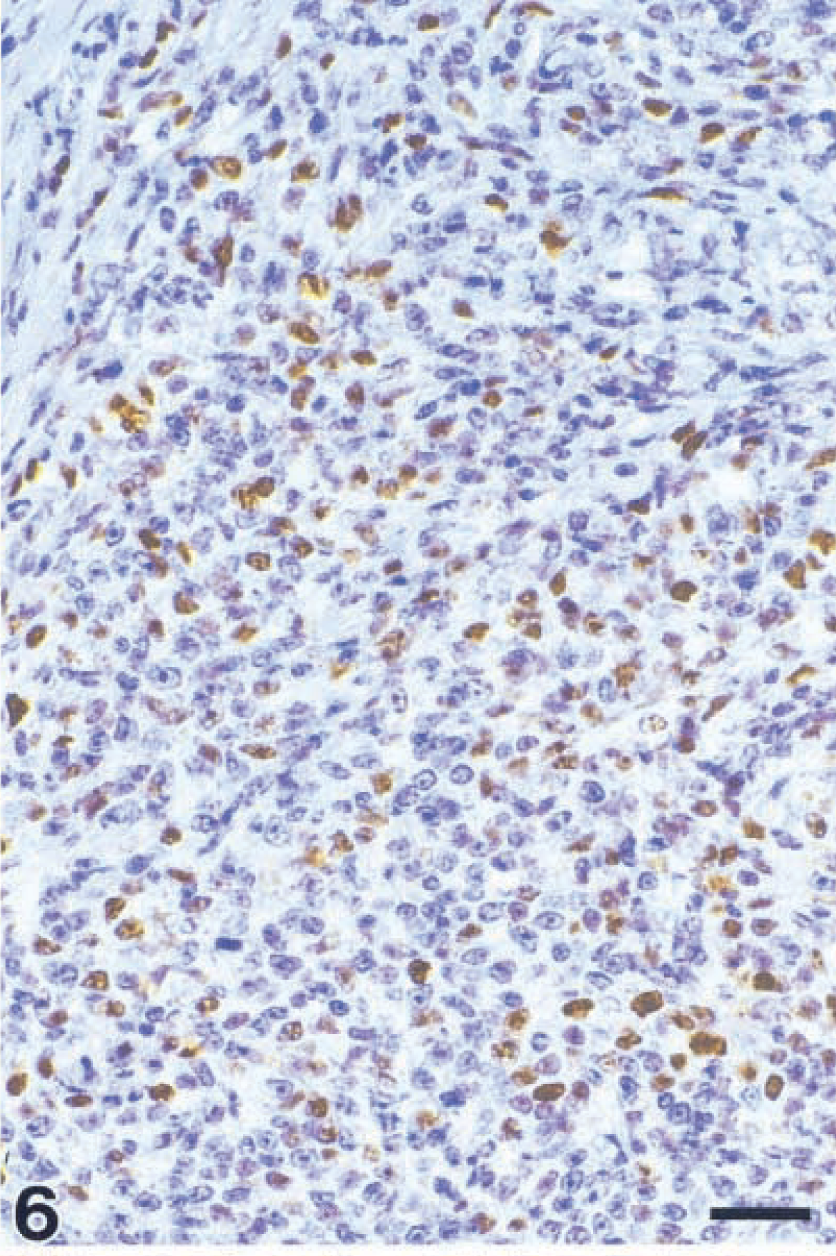
Antibodies to Bc12 revealed a diffuse cytoplasmatic staining of tumor cells (Fig. 7) that was detected in 16/16 (100%) of the monkeys. Although the Burkitt-like lymphoma (monkey No. 15) contained 10% positive tumor cells in a clustered staining pattern, the percentages of positive cells in all other lymphomas were between 40% and 90%.
Centroblastic lymphoma; monkey No. 8. Polymorphic subtype. Cytoplasmatic staining for Bcl2. Streptavidin–biotin complex method. Bar = 17 µm.
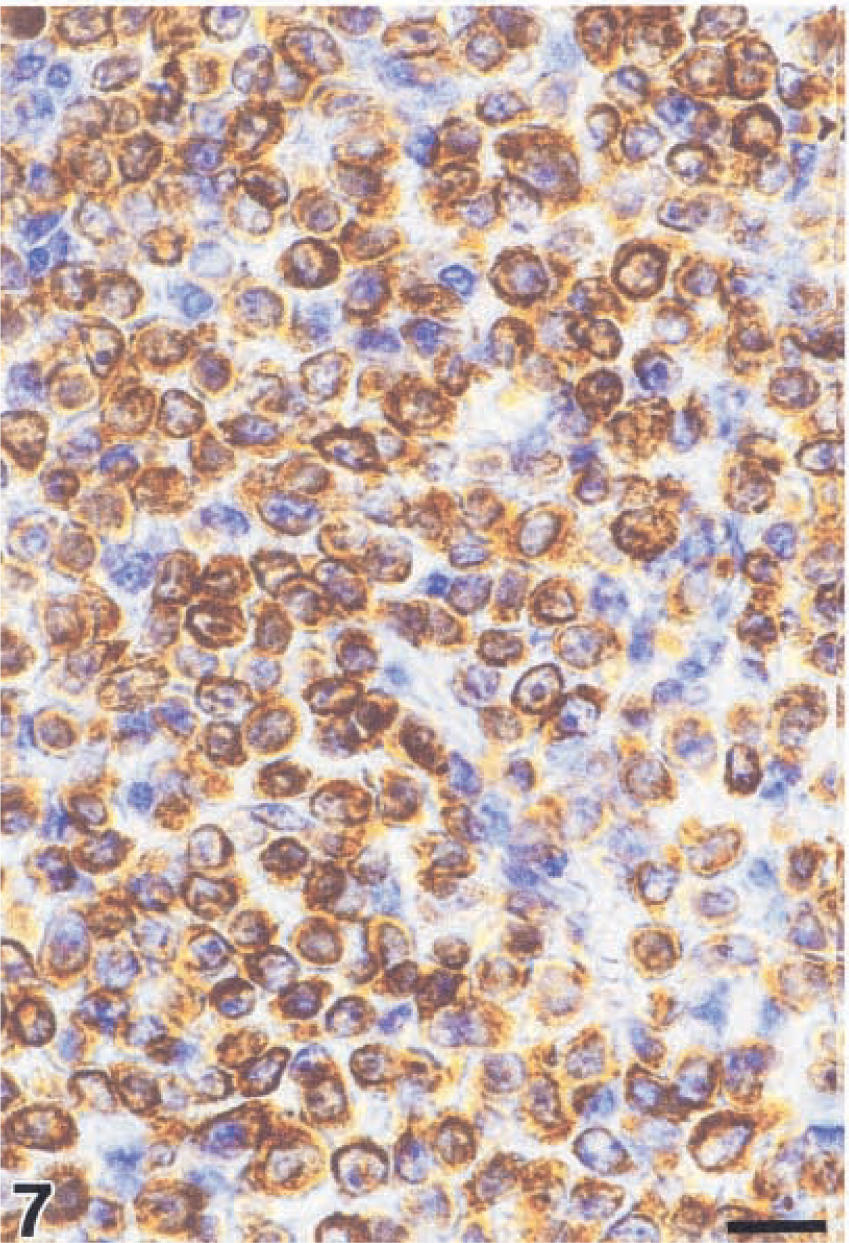
Bc16 was detected in 40% of tumor cells of the Burkitt-like lymphoma (Fig. 8) but was absent in all other cases (Tables 5, 6).
Burkitt-like lymphoma; monkey No. 15. Demonstration of Bcl6. Morphology of tumor cells is impaired because of pretreatment with ethylenediaminetetraacetic acid buffer. Streptavidin–biotin complex method. Bar = 26 µm.
Percentage of proliferating cells, cell DI, and statistical analysis
The proportion of Ki67-positive cells as a marker of proliferation was between 50% and 90% in high-grade lymphomas. The immunocytoma (monkey No. 16) had 30% Ki67-positive tumor cells (Table 4).
Cell DIs obtained with the TUNEL assay on paraffin-embedded sections are listed in Table 5. A distinct difference between the number of cells undergoing cell death in the Burkitt-like lymphoma and the percentages of dying cells in the other entities was apparent.
For statistical analyses, a cutoff value for Bc12 positivity of >10% positive cells was applied. 26 The Burkitt-like lymphoma was judged negative for Bc12. No significant influence of the expression of EBNA2, P53, P21, and c-Myc on the cell DIs was apparent with the Wilcoxon two-sample test. Similarly, there were no significant relationships for the expression of EBNA2, P53, P21, c-Myc, Bc12, and Bc16 as determined with Fisher's exact test, with the exception of the expression of P53 and c-Myc, where a weakly significant relationship was evident (P < 0.05).
Discussion
Since 1990, 16 rhesus monkeys of the German Primate Center infected with different SIV strains developed lymphomas during advanced stages of immunodeficiency. For 8/16 lymphomas that developed within a cohort of 43 rhesus monkeys infected with SIV isolates under similar experimental conditions, an incidence of 19% was established. 50 As shown previously, 50 striking similarities exist between the SIV-associated lymphomas (SIV-NHL) of rhesus monkeys at the German Primate Center and HIV-NHL in humans. 4,7,21,24,30,40,50,54 The SIV-NHL occurred late in the progress of the disease in the presence of low CD4+ T-cell counts and CD4+/CD8+ T-cell ratios. All lymphoma-bearing animals developed clinical symptoms of diseases associated with immunodeficiency at 26 weeks postinfection at the earliest, which means they were not classified as rapid progressors. They had a slow clinical course of disease, with a mean survival time of 15 months. Prior to lymphomas, all but three animals developed a GLP. The primary extranodal and multifocal tumor growth occurred at unusual sites such as gastrointestinal tract, kidneys, periocular tissue, nasal cavity, CNS, or myocardium, comparable to organs and tissues affected in HIV-NHL of humans. 24,40 The lymphomas in monkey Nos. 9 and 11 could not have been classified with confidence as primary CNS lymphomas. Because other sites were also affected by lymphoma growth, it could not be determined at time of necropsy whether the CNS was the primary or secondary site of neoplasia. However, monkey No. 9 showed the typical histologic appearence of primary CNS lymphomas with diffuse proliferation of tumor cells in the central parts of the lymphoma and angiocentric growth at the margins. 7,68
Based on the updated Kiel classification, the lymphomas were identified as centroblastic lymphomas of monomorphic, polymorphic, or centrocytoid subtypes, immunoblastic lymphomas with plasmacytic differentiation, Burkitt-like lymphoma, and immunocytoma (lymphoplasmacytoid lymphoma; Table 1). These lymphomas corresponded to those entities, which occur in HIV-infected humans. 4,21,22,24 Immunocytomas, representing an entity of low malignancy, do not belong to the AIDS-defining diseases, but rare cases have been reported in HIV patients 43 and SIV-infected cynomolgus monkeys. 20 Remarkably, this case of immunocytoma revealed aggressive disseminated and extranodal growth similar to the other high-grade lymphomas.
The B-cell origin was indicated by demonstration of CD20 expression on the lymphoma cells. Immunohistochemical evidence for monoclonality as defined as a ratio of light chain types of ≥10:1 43 was present in 6/16 monkeys (38%). In the lymphoma of the seventh monkey immunohistochemically apparent light chain antigens expressed kappa and lambda in a similar proportion of cells. Whether this result indicates an oligo- or polyclonal proliferation cannot be determined by immunohistochemical techniques alone. Heavy-chain antigens of all classes investigated were expressed in 12/16 monkeys (75%). A similar variability was reported from an immunohistochemical study on AIDS–primary CNS lymphomas 7 and for AIDS-related centroblastic and immunblastic lymphomas, which exhibit an immunophenotypic heterogeneity with expression of monotypic Ig of variable isotypes in approximately 50% of cases. 40
As in HIV-NHL of humans, 40,55 SIV could not be detected in lymphoma cells in these monkeys but was present in monkey Nos. 4, 13, and 15 in single tumor-infiltrating lymphocytes or macrophages. Consequently, a direct role of SIV in lymphomagenesis appears unlikely. However, the SIV-induced immunosuppression contributes indirectly to lymphoma growth; lymphoid neoplasias are very rare in SIV-negative rhesus and cynomolgus macaques, 27 and no lymphomas have occurred previously in any of the noninfected rhesus monkeys at the German Primate Center. Apart from HIV and SIV, the EBV or the homologue simian lymphocryptoviruses are thought to play a role in the pathogenesis of HIV- and SIV-associated lymphomas. 10,18,19,28,55 Using antibodies to EBNA2, which are applicable to the EBV homologues in cynomolgus and rhesus monkeys, 46,56,58 EBNA2 homologue rhLCV antigen was demonstrated in 12/16 lymphomas (75%). As reported previously, in 3/4 EBNA2-negative lymphomas, the small noncoding RNAs EBER1 and EBER2 of rhLCV were detected in a high proportion of tumor cells by in situ hybridization. 56 In summary, a very high frequency of rhLCV infection (nearly 94%) was present in the SIV-associated lymphomas. With regard to expression of viral genes, a distinct correspondence with the HIV-associated lymphomas is obvious. Whereas the Burkitt-like lymphoma, like HIV–Burkitt lymphomas, 30 was positive for EBER1 and EBER2 and negative for EBNA2, the other lymphomas mostly (but not always) expressed EBNA2 in addition to EBER1 and EBER2, 56 similar to their corresponding counterparts in HIV-infected humans. 21,37
In HIV- and non-HIV-associated lymphomas of humans, alterations and deregulated expressions of p53, c-myc, and bcl2 are frequent. In addition, mutations of bc16 occur in a high percentage of cases, whereas bc16 rearrangements can be detected only in a fraction of HIV–diffuse large-cell lymphomas. 1,5,7,21–25,61
The tumor suppressor gene p53 mediates its role as a “guardian of the genome” in part through transcriptional activation of p21. P21 can inhibit entry into the S-phase of the cell cycle through binding several cyclins and cyclin-dependent kinases. 17,31,45 A combined immunohistochemical investigation for P53 and P21 proteins was carried out in accordance to human HIV- and non-HIV-associated lymphomas, where the expression pattern of P53 and P21 can provide information on the functional status of P53. 12,47,49,53,67 Eight of 11 centroblastic (73%) and 1/3 immunoblastic (33%) lymphomas simultaneously expressed P53 and P21. Combined analysis of serial sections revealed that the P21+ tumor cell population represented a fraction within the P53+ tumor cells, indicative of a functional P53 in these lymphomas. However, the high percentages of Ki67-positive tumor cells (50–90%) indicated that the expression of P53 and P21 did not cause cell cycle arrest.
P53-mediated G1 arrest can be overridden by high c-Myc expression in vitro, 32 which may be one of the possible causes for the high percentages of proliferating cells in these cases. Three of 11 of the centroblastic lymphomas (27%) exhibited the expression pattern P53+/P21−. This staining pattern may be indicative for a mutation of p53, with a subsequent loss of the transcriptional activities.
One of three of the immunoblastic lymphomas (33%), the Burkitt-like lymphoma, and the immunocytoma were negative for P53 and P21. In human NHL, this expression pattern in general is indicative of a germline configuration of p53. This staining pattern is rarely associated with nonsense or frame shift mutations of p53. 67
One immunoblastic lymphoma had a P53-/P21+ expression pattern. Also very rare in human NHL, this expression pattern could be associated with a germline configuration of p53, 12 a P53-independent expression of P21, 15 or a p53 mutation with a preserved functional active wild-type allele of p53. 67
In summary, the SIV-associated lymphomas frequently exhibit an overexpression of P53 and P21 in the presence of high percentages of proliferating tumor cells, indicative of alterations in this pathway of cell cycle regulation. In a few cases, there might be evidence for a functional impairment of P53. However, the combined immunohistochemical detection of P53 and P21 as a reflection of the underlying genetic alterations as in human NHL cannot be applied to SIV-associated lymphomas without further molecular pathologic investigations.
The oncogene c-myc plays an important role in cell proliferation and can contribute to transformation under certain circumstances. An oncogenic activation of c-myc, e.g., through chromosomal translocations, gene amplifications, or mutations, 34 results in a deregulated expression or overexpression. 13,41,42 All centroblastic and immunoblastic lymphomas expressed detectable levels of c-Myc in the major proportion of tumor cells. This expression may reflect either the high proliferation rate of the tumor cell population or an underlying genetic aberration. A weakly significant correlation between the overexpression of P53 and c-Myc was present (P < 0.05). This finding is consistent with the correlation of c-Myc protein level and p53 mRNA expression in human Burkitt lymphomas and B-lymphoid cell lines. 59 High c-Myc expression can transactivate the p53 promotor. 29 c-Myc can cooperate with Bcl2, with subsequent proliferation of precursor B cells and neoplastic outgrowth. 6,66 In the SIV-NHL, no significant correlation between c-Myc and Bcl2 expression was found. However, there was a clear relation between these two oncogene products within the high-grade lymphomas, which indicates a possible cooperation of c-Myc and Bcl2 in terms of cell proliferation and prevention of apoptosis in these entities.
The negative staining of the Burkitt-like lymphoma is consistent with investigations of Gaidano and coworkers, 23 who found rearrangements of c-myc in only 20% of AIDS–Burkitt-like lymphomas. The negative staining of the immunocytoma for c-Myc is consistent with the low level of proliferating cells as determined with antibodies to Ki67. To distinguish molecular aberrations affecting c-myc from epigenetic phenomena, further investigations are necessary to evaluate the underlying genetic mechanisms.
The antiapoptotic oncogene bcl2 is a frequent target of translocations and gene amplifications in human B-cell NHL with a resultant deregulated expression of the gene product. In addition, an increased expression of Bcl2 can occur without genetic aberrations. 51,65 All SIV-associated lymphomas investigated exhibited Bcl2 positivity in high percentages with up to 90% positive cells, whereas the Burkitt-like lymphoma had only 10% stained cells and was considered negative. 26 The expression of Bcl2 in immunoblastic and centroblastic lymphomas could be interpreted as an abnormal state in relation to their normal cellular origin. 61 There was no statistical evidence for an influence of the EBNA2 homologue of rhLCV on Bcl2 expression, as considered by Castanos-Velez and coworkers 10 for EBNA2 of the herpesvirus Macaca fascicularis 1 (HVMF-1), which is an EBV homologue lymphocryptovirus in cynomolgus monkeys frequently found in SIV-associated lymphomas of this species.
Bcl6 seems to play a role in normal B-cell differentiation and germinal center formation. Persistent expression of Bcl6 mediated through chromosomal rearrangements may contribute to lymphomagenesis through inhibition of post–germinal center differentiation. 11,69 The expression of Bcl6 in HIV-NHL and lymphomas of immunocompetent humans is independent of genetic lesions and rearrangements of the bcl6 gene. 8,11 In 100% of HIV–Burkitt lymphomas and >50% of HIV–diffuse large-cell lymphomas, an expression of Bcl6 can be detected by immunohistochemistry. 8
The SIV-NHL exhibited Bcl6 expression only in the single case of Burkitt-like lymphoma. There seems to be an inverse correlation between Bcl6 and EBNA2 as shown for human HIV-NHL and HIV–Burkitt lymphoma cell lines. 8 There is also a nonsignificant tendency toward an inverse correlation between Bcl6 and EBNA2 or Bcl2, respectively.
Because only paraffin-embedded material was available for investigations, only the TUNEL assay could be applied for determination of the number of dying cells within the lymphomas, including apoptotic cell death. The Burkitt-like lymphoma had a clearly higher DI than did the other types of lymphomas. In this context, the expression pattern of Bcl2 and Bcl6 is striking, even with no significant correlation between the DI and Bcl2 or Bcl6 expression, respectively. It is highly probable that Bcl2 has an antiapoptotic function in the SIV-associated lymphomas; the Burkitt-like lymphoma had the highest DI and the lowest Bcl2 expression. This finding is consistent with observations in human NHL 64 and SIV-associated lymphomas of cynomolgus monkeys, 10 where high apoptotic indices were correlated with negative Bcl2 expression. Statistically, no correlation between the DI and expression of P53, P21, or c-Myc was observed in the SIV-associated lymphomas. Obviously, there are other influences on the DI.
The SIV-associated lymphomas are similar in many ways to human HIV-associated lymphomas, including the development in late stages of infection frequently after a preceding GLP, disseminated growth in unusual sites, and aggressive clinical behavior. Other parallels exist in the histologic and immunohistochemical phenotype, the frequent association with lymphocryptoviruses, and the corresponding expression of viral genes and the negative proof of retroviral antigen in the lymphoma cells. There are also several differences between SIV- and HIV-associated lymphomas. No primary effusion lymphoma was observed in these monkeys. Similarly, primary CNS lymphoma could not be diagnosed with certainty. The incidence of rhLCV infection is markedly higher than that of EBV infection in HIV patients.
The overexpression of an oncogene or a tumor suppressor gene is no definite proof of an underlying genetic abnormality. However, the frequent detection of an overexpression of P53, P21, c-Myc, Bcl2, and, in one case, Bcl6 in the SIV-NHL points to a possible role of these cell proliferation- and apoptosis-associated gene products in pathogenesis or progression. The frequent simultaneous occurrence may be a reflection of a multistep pathogenesis of SIV-associated lymphomas. Based on the results of this study, further investigations of the underlying genetic mechanisms would be of interest.
Footnotes
Acknowledgements
The antibody KK75 was obtained from K. Kent and C. Arnold, NIBSC Centralised Facility for AIDS Reagents, Potters Bar, UK, supported by EU Programme EVA (contract BMH4 97/2515) and the UK Medical Research Council. We thank K. Gee for proofreading the manuscript, N. Stolte (Department of Virology and Immunology, German Primate Center) for providing immunologic and virologic data, and E. Hackenbroich, K. Kaiser-Jarry, E. Lischka, and E. Niksch for technical assistance.
