Abstract
We report a case of ovarian malignant intermediate-type trophoblastic tumor in a clinically normal, nonpregnant 4-year-old rhesus monkey (Macaca, mulatta). A large solid lobulated mass replaced the right ovary and filled the pelvis. Multiple metastases were observed within the lungs and the liver. The tumor was histologically identified as predominantly composed of intermediate trophoblastic cells, without prominent hemorrhages and the classic bilaminar pattern of cyto- and syncytiotrophoblastic cells characteristic of choriocarcinoma. Immunohistochemical analysis showed the presence of placental lactogen hormone in many tumor cells and chorionic gonadotropin in a few multinucleated cells consistent with syncytiotrophoblastic differentiation. No other germ cell differentiation was identified in the pelvis mass nor in the metastases. In the absence of previous and present pregnancy, this neoplasm has to be considered as a nongestational malignant placental site trophoblastic tumor of the ovary.
Malignant trophoblastic tumors can be classified as choriocarcinoma, epithelioid trophoblastic tumor, or placental site trophoblastic tumor. 2 These tumors usually occur during or after gestation, but may also rarely develop from germ cells in the absence of pregnancy. 5 Choriocarcinoma, composed of bilaminar cytotrophoblast and syncytiotrophoblast, can be found as a component of a mixed germ cell tumor of the ovary, but pure nongestational choriocarcinoma is an extremely rare neoplasm in humans. 10 Epithelioid trophoblastic tumor is made of intermediate trophoblastic cells resembling those of chorion laeve, mimics carcinoma, and was initially thought to result from incomplete response of choriocarcinoma to chemotherapy. 6 Placental site trophoblastic tumor is derived from intermediate trophoblastic cells of the placental bed, and nongestational forms have been reported in the testis. 7, 9 The overall frequency of neoplasms in captive primates has been considered low, probably because of the lack of studies involving large groups of monkeys. 4 However, and despite their rarity in humans, ovarian nongestational trophoblastic tumors have been described in captive primates: 2 choriocarcinomas in a cynomolgus monkey (Macaca fascicularis) and a rhesus monkey (Macaca mulatta), and 1 epithelioid trophoblastic tumor in a cynomolgus monkey. 1, 3, 8 We report a further variant of ovarian nongestational trophoblastic tumor in a young rhesus monkey.
A 44-month-old female rhesus monkey from China was introduced in our animal laboratory via The Netherlands (Hartelust, Tilburg, The Netherlands), as one member of an experimental group of monkeys after agreement of the local animal ethical committee. The monkeys rested in the animal house for 4 months of acclimatization. Investigation was then started on this clinically healthy monkey by a laparotomy under general anaesthesia. A large multinodular mass was incidentally discovered at the site of the right ovary together with several nodules on the adjacent peritoneal area. A nodule of the omentum was biopsied for histopathologic analysis, and the experimental procedure was stopped. The biopsy was fixed in 10% formalin and embedded in paraffin, and histologic sections were examined by light microscopy after hematoxylin and eosin (HE) staining. A diagnosis of metastatic trophoblastic tumor was proposed, and euthanasia of the monkey was decided although no β-human chorionic gonadotropin (β-hCG) was detected in the serum.
The monkey was sacrificed and necropsy performed. A huge pelvic tumor of 8 × 6 × 5 cm filled the lower abdominal cavity (Fig. 1). The mass was fixed to the uterus and the bladder and was adherent to the colon and intestinal loops. Metastases were found in the peritoneal cavity, in the liver (Fig. 2), and in the lungs (Fig. 3). At section, the primary tumor and the metastases appeared whitish with large necrotic areas and prominent calcifications (Fig. 3) but without hemorrhages. A large myocardial infarction was also present. The left ovary was not involved by the tumor and contained several growing follicles and a corpus luteum. The endometrium showed features of early secretory phase. There was no evidence of pregnancy.
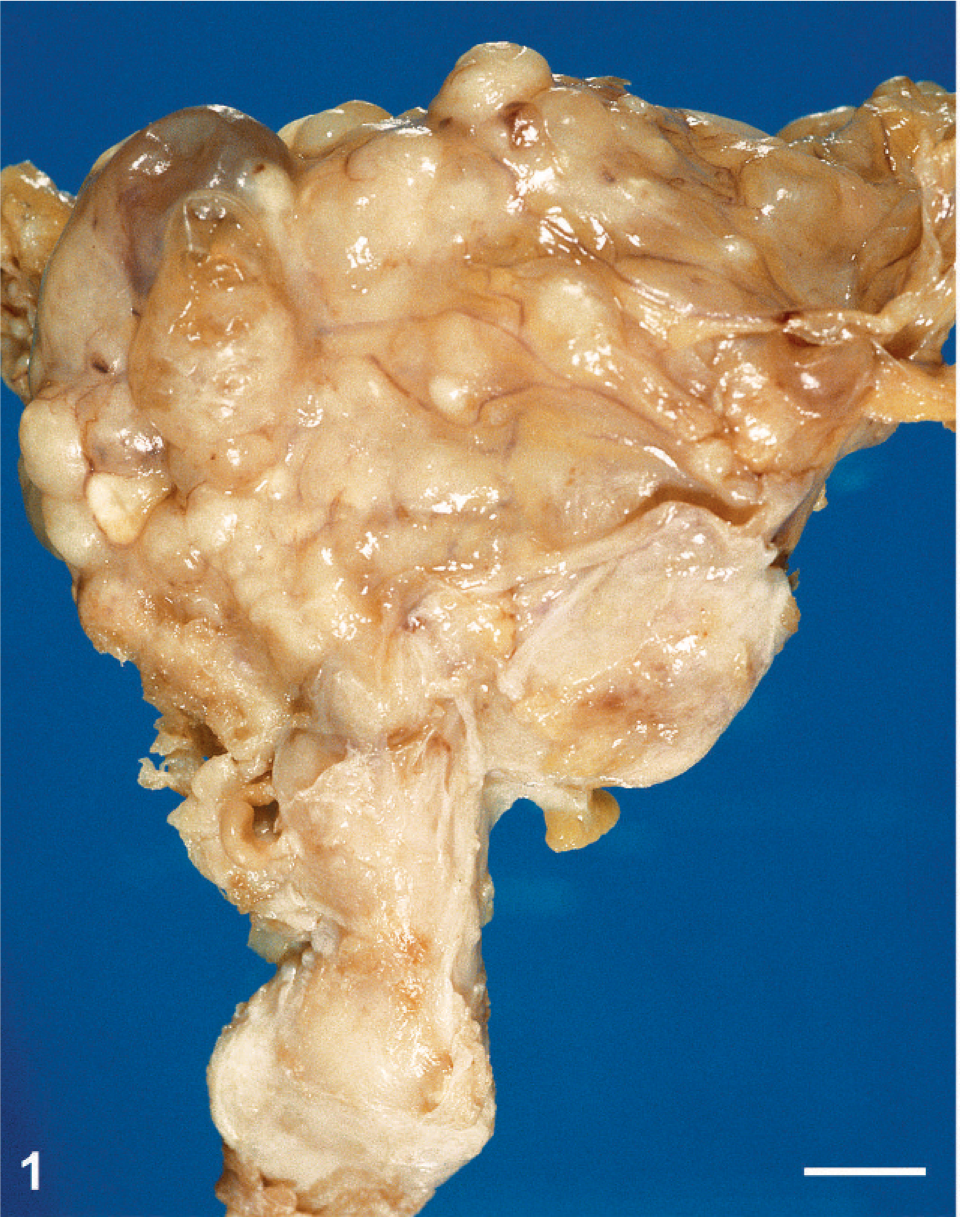
Posterior view of the pelvic mass. Vagina and uterus are at the bottom of the mass. Bar = 1 cm.
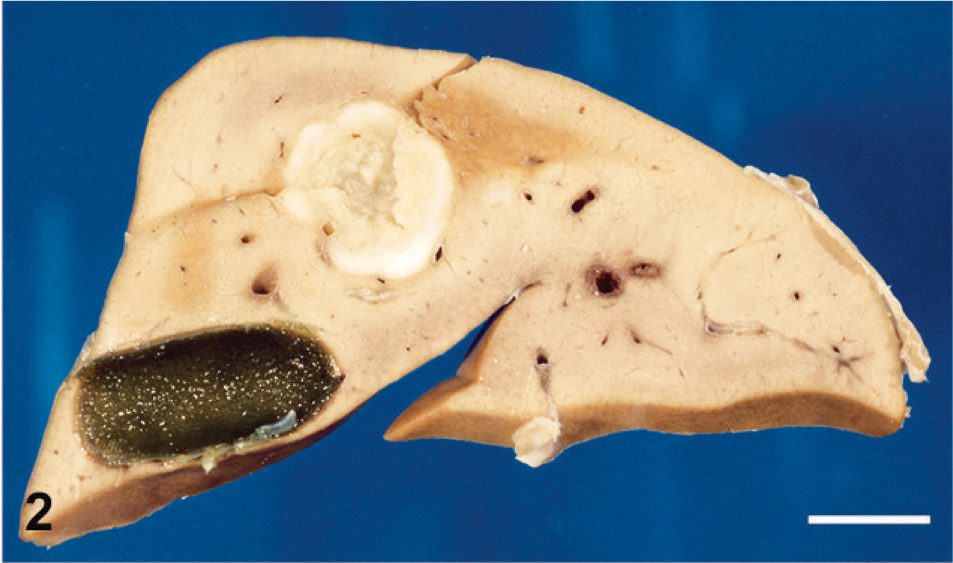
Section through the liver showing a large white metastasis with a necrotic center. The gallbladder is visible. Bar = 1 cm.
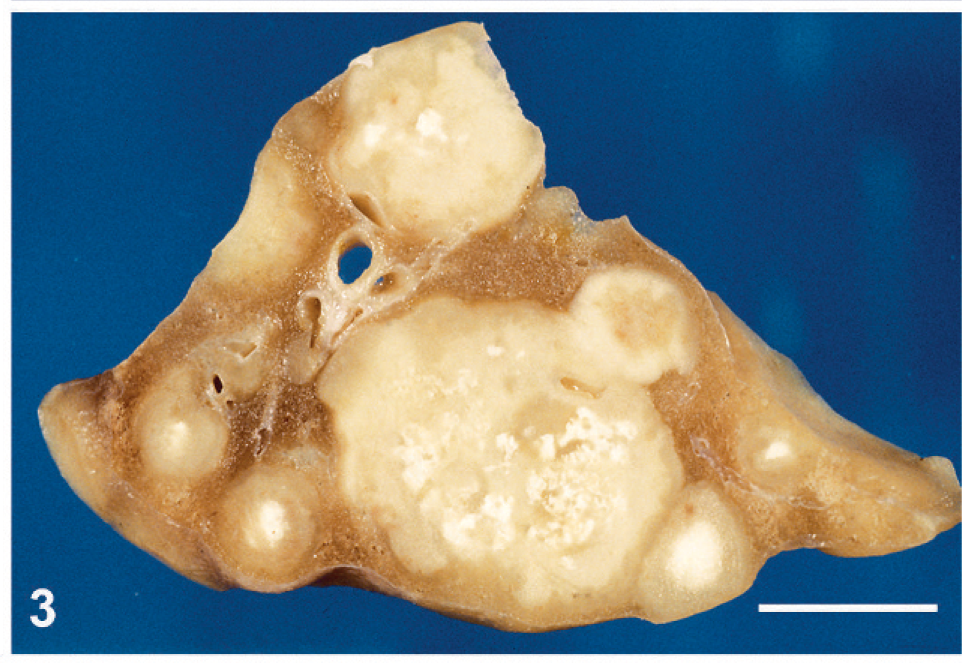
Section through a lung showing several metastases containing prominent calcifications. Bar = 1 cm.
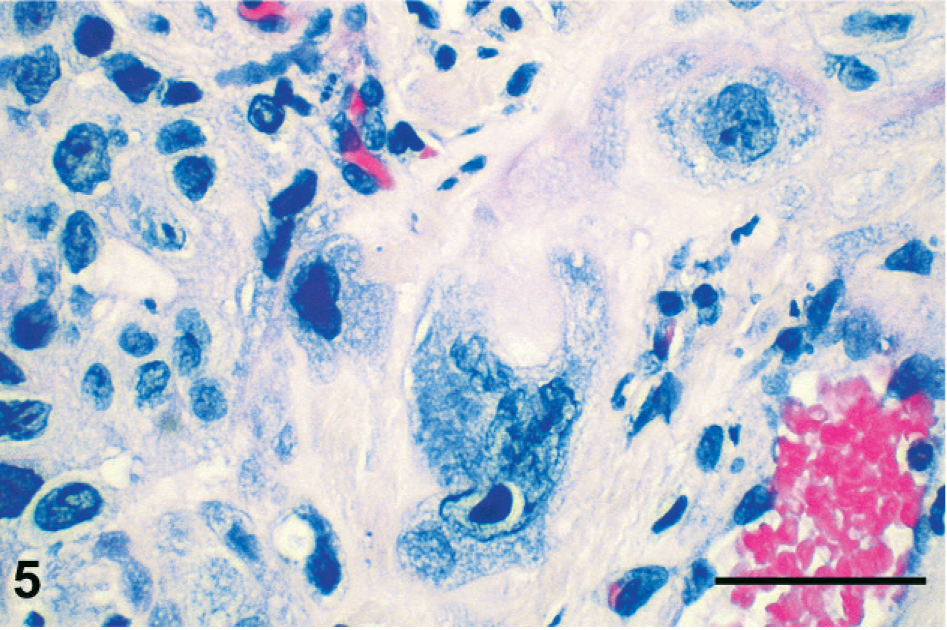
Histologic sections were stained with HE, periodic acid–Schiff (PAS), Masson's trichrome, Mallory's phosphotungstic hematoxylin, or Congo red, or immunolabeled with antibodies raised against cytokeratin (clone AE1/AE3; Dako, Glostrup, Denmark), epithelial membrane antigen (clone E29; Dako), human placental lactogen (hPL; rabbit polyclonal antibody; Dako), or β-hCG (rabbit polyclonal antibody; Dako; or mouse monoclonal antibody; clone CG04+CG05; Neomarkers, Fremont, CA). Histologic appearance of the primary tumor and of the metastases was similar to that in the premortem biopsy. No normal ovarian tissue was found in the primary tumor. Neoplastic cells showed eosinophilic or vacuolated cytoplasm and irregular pleiomorphic nuclei. They were isolated or arranged in nests or cords inside the fibrous and fibro-adipose tissue of the peritoneum, the retroperitoneum, and the parenchyma of the metastatic tissues (Fig. 4). The vast majority of neoplastic cells were moderately large and mononucleated, with a high nucleo-cytoplasmic ratio. Occasional giant cells and a few multinucleated, syncytiotrophoblastic-like cells were present (Fig. 5). Anisokaryosis was prominent and mitotic figures numerous. Neoplastic cells were focally surrounded by an eosinophilic hyaline matrix similar to the fibrinoid material found in the placental bed (Fig. 5). This eosinophilic material was stained by PAS and Masson's trichrome but not by Congo red nor Mallory's phosphotungstic hematoxylin, showing thus no evidence of amyloid or fibrin deposition and suggesting it corresponds to a glycoprotein-rich extracellular matrix. Interstitial hemorrhages were limited, but necrosis was extensive. The histologic appearance was suggestive of infiltration by intermediate-type malignant trophoblastic cells, a diagnosis supported by the immunohistochemical labeling of cytokeratin and hPL (Fig. 6) in neoplastic cells. A few cells were labeled for β-hCG using the polyclonal antibody (Fig. 7) but not with the monoclonal antibody. No immunostaining of epithelial membrane antigen was observed.
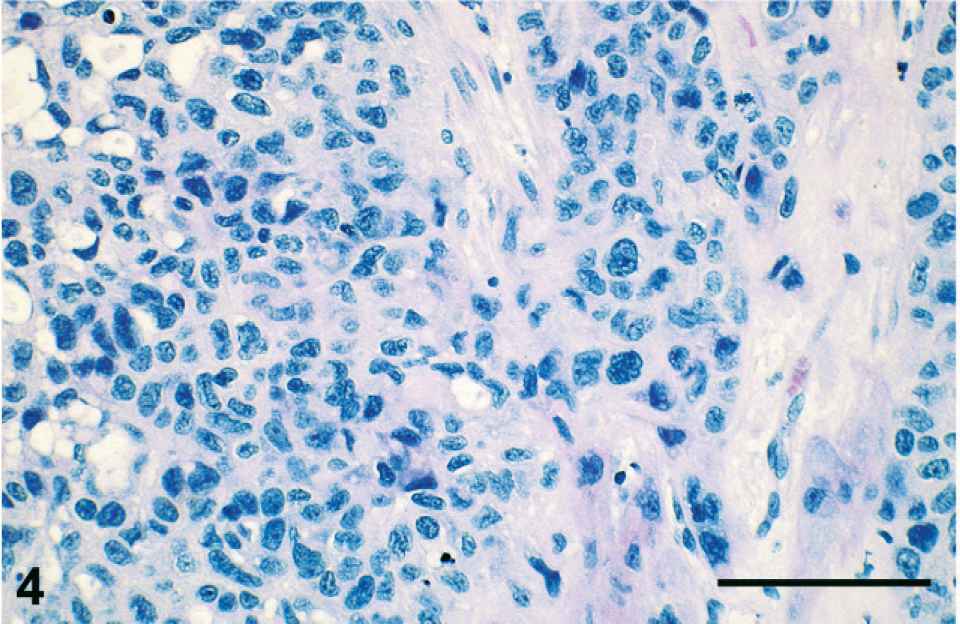
Microphotograph of an HE-stained section of the tumor showing infiltration of fibrous tissue by sheets of neoplastic cells with prominent anisokaryosis. Hemorrhages are not conspicuous. Bar = 100 μm.
High power magnification of an HE-stained section showing a giant multinucleated cell with a large prominent nucleolus. Some eosinophilic hyaline matrix is present in the upper right corner of the microphotograph. Bar = 50 μm.
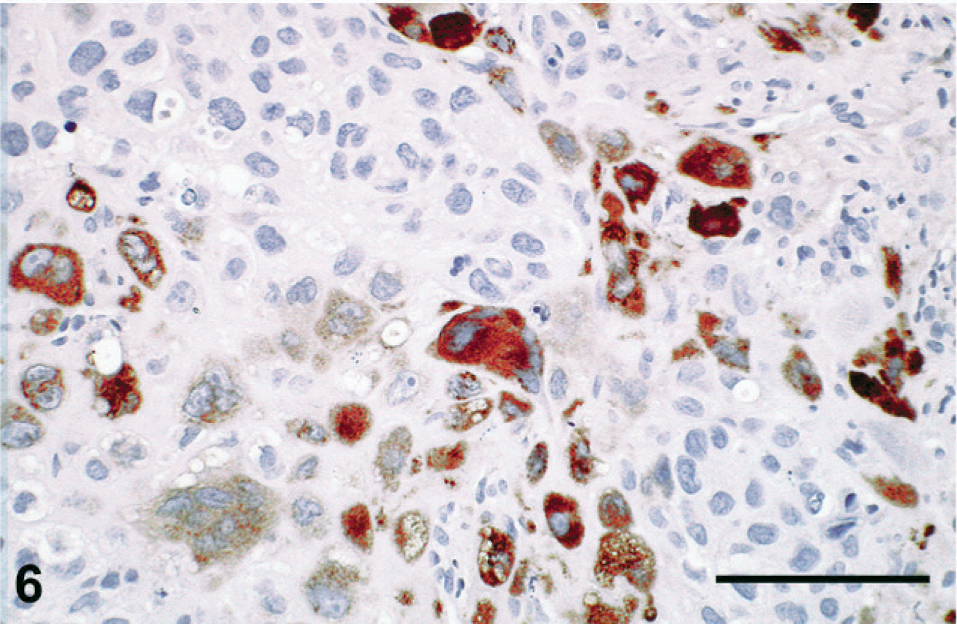
Immunostaining with an antibody raised against human placental lactogen. Many neoplastic cells are strongly immunolabeled. Bar = 100 μm.
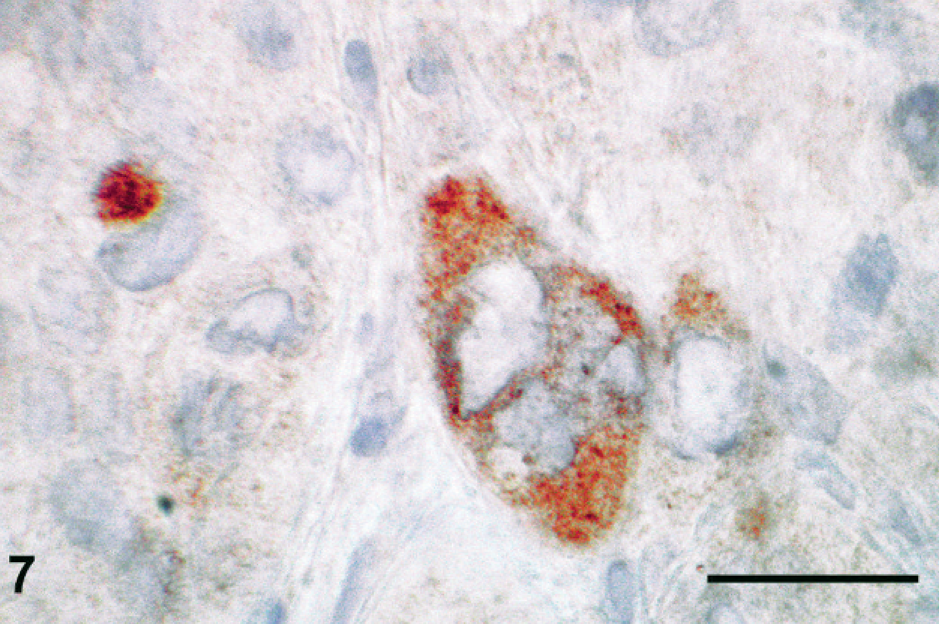
Immunostaining with a polyclonal antibody raised against human β-chorionic gonadotropin hormone. Several cells, including a multinucleated cell, are labeled by the antibody. Bar = 25 μm.
Nongestational trophoblastic tumors are rare and usually associated with other germ cell components in mixed germ cell neoplasms. The present tumor was exclusively composed of trophoblastic cells without other germ cell component in the primary tumor or in the metastases. Tumor cells were essentially mononucleated intermediate trophoblastic cells expressing placental lactogen, with a few multinucleated chorionic gonadotropin (CG)-containing syncytiotrophoblastic-like cells. This immunohistochemical pattern indeed favors a differentiation of the neoplastic cells similar to the extravillous interstitial trophoblast of the placental site. Furthermore, the tumor did not show the characteristic bilaminar pattern of choriocarcinoma nor a cohesive carcinomatous-like pattern suggestive of epithelioid trophoblastic tumor, but rather the appearance of a placental site trophoblastic tumor. The absence of current or previous pregnancy in this monkey, who had never been mated, excludes the gestational origin of the tumor. Germ cell tumors are the most common ovarian neoplasm in young humans, and nongestational ovarian trophoblastic tumors occurred in 3- and 4-year-old rhesus monkeys (Farman et al. and the present case) and in 4- and 13-year-old cynomolgus monkeys. 3, 5, 8 The ovarian epithelioid trophoblastic tumor was discovered after treatment with cytotoxic drugs, 3 which may have altered the histology of the tumor, in contrast with the present monkey who had never been treated. Serum β-hCG level is usually elevated in human choriocarcinoma but not in placental site trophoblastic tumor, 5 and accordingly, CG was not detected in the serum of the present monkey.
In conclusion, we describe a pure nongestational malignant placental site trophoblastic tumor of the ovary in a young rhesus monkey, a never reported tumor to the best of our knowledge.
