Abstract
Microcystin-LR (MCLR) is a potent hepatotoxin produced by the cyanobacterium Microcystis aeruginosa. The histology of acute lethal toxicity has been well characterized, but histology is limited regarding sublethal exposure. Balb/C mice were given a single sublethal dose of MCLR (45 μg/kg) and euthanized at 2, 4, 12, and 24 hours after exposure. Centrilobular to midzonal hepatocellular hypertrophy with loss of cytosolic vacuolation consistent with glycogen depletion occurred at 2 hours. At 4 hours, central lobular hepatocytes exhibited eccentric areas of eosinophilic cytoplasmic condensation that were partially aggregated around the outer nuclear membrane. The areas were weakly positive for cytokeratin and somewhat resembled the Mallory bodies of alcoholic human hepatitis. Small numbers of apoptotic hepatocytes were seen at 24 hours. The toxin was detectable by immunohistochemistry (IHC) as early as 2 hours and was colocalized with the areas of hepatocellular hypertrophy. Intense nuclear staining occurred at 4 hours; this was no longer evident after 12 hours. Strong staining of apoptotic bodies occurred at 24 hours. Mice that received two daily doses had a marked increase in apoptotic hepatocytes in the centrilobular areas. Lesions at four and seven doses consisted of marked hepatocytomegaly and karyomegaly with parenchymal disarray and cytosolic vacuolation. IHC revealed diffuse staining throughout the liver parenchyma consistent with toxin accumulation. An anti-MCLR monoclonal antibody detected bands at the 40-kDa mark in nuclear extracts that were identified as protein phosphatases 1 and 2A by western blotting, consistent with a covalent interaction between MCLR and nuclear protein phosphatases.
Microcystin-LR (MCLR) is a naturally occurring cyclic heptapeptide hepatotoxin produced by the blue green alga Microcystis aeruginosa. Toxic blooms of cyanobacteria in ponds and water reservoirs have been associated with acute, usually lethal toxicity in various species of domestic animals and humans. 12,20 Fertilizer and animal waste runoff into marginal water supplies promotes the formation of toxic blooms, therefore increasing the risk of exposure to both humans and domestic animals. Surveys have revealed the presence of this contaminant in water reservoirs worldwide. 8,29
The liver is known to be the target organ of MCLR. Grossly, livers are enlarged, hemorrhagic and friable. Histologically, acute toxicity is characterized by rapid centrilobular hepatocyte rounding, dissociation, and necrosis, with breakdown of sinusoidal endothelium resulting in severe intrahepatic hemorrhage. Acute toxicity is attributed to cytoskeletal collapse due to aggregation of actin filaments, microtubules, and intermediate filaments within hepatocytes. 10,13,31 The underlying pathogenesis involves rapid hyperphosphorylation of cytoskeletal proteins secondary to the potent inhibitory effects of this toxin on protein phosphatases 1 and 2A. 18,23
Studies involving tissue distribution of radiolabeled MCLR in mice indicate that at 60 minutes after a sublethal intravenous injection, most of the toxin is localized to the liver. 21 Uptake of the toxin occurs by an energy-dependent transport process involving a rifampicin-sensitive bile acid carrier. The hepatocellular specificity of this toxin has been confirmed by immunohistochemical studies in rats, mice, and trout using anti-MCLR monoclonal antibodies. 3,26,35
The effects of sublethal exposure only recently have been characterized in a rat model of prolonged exposure. Lesions in the livers of rats exposed to sublethal intraperitoneal doses for 28 days included hepatocellular apoptosis, steatosis, and centrilobular fibrosis with infiltration by neutrophils and mononuclear cells. 7,26 Mice exposed to sublethal intraperitoneal doses (20 µg/kg) for 28 weeks developed multiple hepatic nodules. 11 In acute toxicity studies in mice, which included the administration of sublethal oral and intraperitoneal doses, necrosis, hemorrhage, apoptosis, and cellular hypertrophy were observed in the centrilobular regions 24 hours and 7 days after dosing.
Natural exposure of domestic animals and humans to sublethal levels of MCLR is likely to go unrecognized. Therefore, experimental studies designed to characterize associated histologic lesions in the liver may help pathologists in the identification of exposure to this toxin. However, studies specifically designed to elucidate the pathology of sublethal MCLR exposure in laboratory rodents were not found in the literature. Although experiments using radiolabeled MCLR have localized most of the toxin to the cytosolic compartment, nuclear accumulation has been suggested by recent immunohistochemical studies in mice and trout. 3,35 We have previously reported that MCLR may have a negative effect on protein synthesis that correlates with decreased mRNA concentrations. 25 Such alterations could be explained by a direct effect of the toxin on nuclear function. In this report we provide evidence that the nuclear localization of MCLR is an early event after sublethal MCLR exposure that results in the covalent binding of MCLR to nuclear protein phosphatases. Furthermore, we describe the histopathology and immunohistochemical localization that occur after acute to subacute sublethal exposure to MCLR and that differ from those of acute lethal exposure.
Materials and Methods
Animal use
Adult male Balb/C mice weighing approximately 20 g (Harlan, Indianapolis, IN) were allowed to acclimate for a period of 7 days. Purified MCLR (greater than 98% purity by high-performance liquid chromatography) was obtained from Dr. Wayne Carmichael and dissolved in saline for delivery of intraperitoneal injections at 0.2–0.3 ml per mouse. The selection of the dosage used in our studies (45 µg/kg) was based in the median lethal dose reported for Balb/C mice of 65 µg/kg. In that study, no deaths were reported within 24 hours in five mice that were dosed with 48 µg/kg. 34 In another study, a dosage of 35 µg/kg only resulted in transient increases in serum lactate dehydrogenase, sorbitol dehydrogenase, and aspartate aminotransferase, but livers were histologically normal 1 minute through 6 days after injection. 21 Mice were treated with a single intraperitoneal (IP) injection of 45 µg/kg MCLR of saline vehicle only and euthanatized at 2, 4, 12, and 24 hours after the injection in groups of five. In a separate study, three groups of five animals received a total of two, four, and seven daily injections with separate age-matched control groups that were treated with saline vehicle only. Mice were euthanatized by an IP overdose of pentobarbital. At the time of euthanasia the liver was immediately removed, examined grossly, and weighed.
Histology and immunohistochemistry
Portions of the liver were fixed by immersion in 10% buffered formalin, embedded in paraffin, and routinely processed to hematoxylin and eosin (HE) slides for light microscopic examination.
One possible consequence of the use of mouse primary antibodies on mouse tissues is high backgrounds that make interpretation of the immunostains difficult. A commercially available kit specifically designed to avoid this problem was used, following the protocol outlined by the manufacturer (M.O.M., Vector Mouse On Mouse Immunodetection Kit, Vector Labs, Burlingame, CA). Briefly 4 to 6-µm-thick tissue sections were applied to slides and deparaffinized in xylene. Sections were hydrated gradually through graded alcohol washes and then washed in distilled water. Specimens were incubated for 5 minutes with 3% hydrogen peroxide and washed twice with phosphate-buffered saline (PBS). Sections were then incubated for 1 hour in M.O.M. Mouse IgG Blocking Reagent (Vector Labs) and washed with PBS. Tissue sections were then incubated for 5 minutes in working solution of M.O.M. The primary anti-MCLR antibody was diluted to a final concentration of 30 µg/ml and incubated in the tissue sections for 1 hour. For the cytokeratin stains, a wide-spectrum polyclonal anti-cytokeratin antibody (Dako, Carpenteria, CA) was used at a 1:500 dilution. After washing with PBS, sections were incubated with biotin-conjugated secondary antibody at 1 µg/ml for 30 minutes. After washing with PBS, sections were incubated with avidin–biotin enzyme reagent for 30 minutes and then with peroxidase substrate for 5 minutes. Sections were washed, dehydrated, and mounted.
Nuclear and cytosolic extracts
Upon removal from each mouse, a portion of the liver was rapidly chilled and diced into small pieces with a razor blade. The tissue was further disrupted with a dounce homogenizer while maintaining the temperature at 4 C until a homogeneous suspension of hepatocytes was obtained. The suspension of hepatocytes was washed twice with ice-cold PBS. A commercially available kit was used to extract nuclear and cytosolic fractions from 100 µl of packed cells (NE-PER, Nuclear and Cytoplasmic Extraction Reagents, Pierce, Rockford, IL). The protein content of the extracts was determined by the bicinchonic acid method (Pierce). To verify the adequacy and quality of the separation procedure, aliquots of nuclear and cytosolic fractions were assayed for alanine aminotransferase (ALT) activity using an automated analyzer (Hitachi 911, Boehringer-Mannheim, Indianapolis, IN). The analysis revealed less than 5% contamination of the nuclear fraction with this cytosolic enzyme. In addition, before the nuclear lysis step, wet mounts of the nuclear suspension were examined by light microscopy, which revealed an almost pure population of intact naked nuclei.
Detection of MCLR and protein phosphatases 1 and 2A in nuclear and cytosolic extracts by western blotting
A total of 20 µg of protein from nuclear or cytosolic extracts was mixed with an equal volume of Laemmli sample buffer (Bio-Rad, Hercules, CA) containing 710 mM β-mercaptoethanol and boiled for 2 minutes. Proteins were routinely separated by sodium dodecyl sulfate (SDS)–polyacrylamide disc gel electrophoresis on 10% gels at 200 V for 30 minutes. Prestained molecular weight standards (Bio-Rad) were loaded onto each gel to assess the approximate molecular weight, quality of separation, and electrophoretic transfer. After separation, proteins were transferred to a 0.2 µm polyvinyldifluoride membrane (PVDF-Sequi-Blot, Bio-Rad) for 45 minutes at 100 mA. After transfer the membrane was washed twice (0.2% Tween-20 in 20 mM Tris-HCl, 500 mM NaCl, pH 7.5) and blocked for 3 hours with 1% bovine serum albumin. For the detection of protein phosphatase 1, the membrane was incubated overnight with a rabbit polyclonal primary antibody against protein phosphatase 1 (Santa Cruz Biotechnology, Santa Cruz, CA) at a 1:500 dilution. After three washing steps the membrane was incubated with a goat anti-rabbit alkaline phosphatase conjugate (Bio-Rad) at 1:6,000 dilution for 1 hour. Membranes were washed and then incubated for 5 minutes with chemiluminescent substrate (Immun-Star Substrate, Bio-Rad). Bands were visualized by exposing the membrane to X-ray film for 3–5 minutes. The primary antibody was stripped from the membrane by incubating in stripping buffer (62.5 mM Tris, pH 6.7, 100 mM mercaptoethanol, and 2% SDS) for 30 minutes at 50 C. The membrane was washed, blocked, and reprobed with primary anti-microcystin monoclonal antibody (kindly supplied by Dr. Fun S. Chu) overnight at a dilution of 1:2,000. The membrane was washed three times for 10 minutes and incubated for 1 hour with the secondary antibody (Anti-Mouse IgG, alkaline phosphatase conjugate, (Bio-Rad) at a 1:6,000 dilution and exposed to X-ray film as previously described.
Statistics
GraphPad Prism software (GraphPad Software, San Diego, CA) was used for the statistical comparisons. Differences between dose groups and their respective saline controls were evaluated by Student's t-test (P < 0.05).
Results
Gross pathology and liver weights
All the animals survived the described treatments and remained clinically normal throughout the experiment. Only the livers of the mice that received four and seven daily sublethal injections were pale, moderately enlarged, and had an accentuated reticular pattern. None of the mice had swollen, hemorrhagic livers characteristic of lethal intoxication. A trend for decreased body weights with days of exposure was observed, but was statistically significant only in animals that received seven doses (Fig. 1). A statistically significant increase in absolute liver weights was seen in the four- and seven-dose groups (Fig 1b). Statistically significant increases in liver to body weight ratios were seen in the four- and seven-dose groups (Fig 1c).
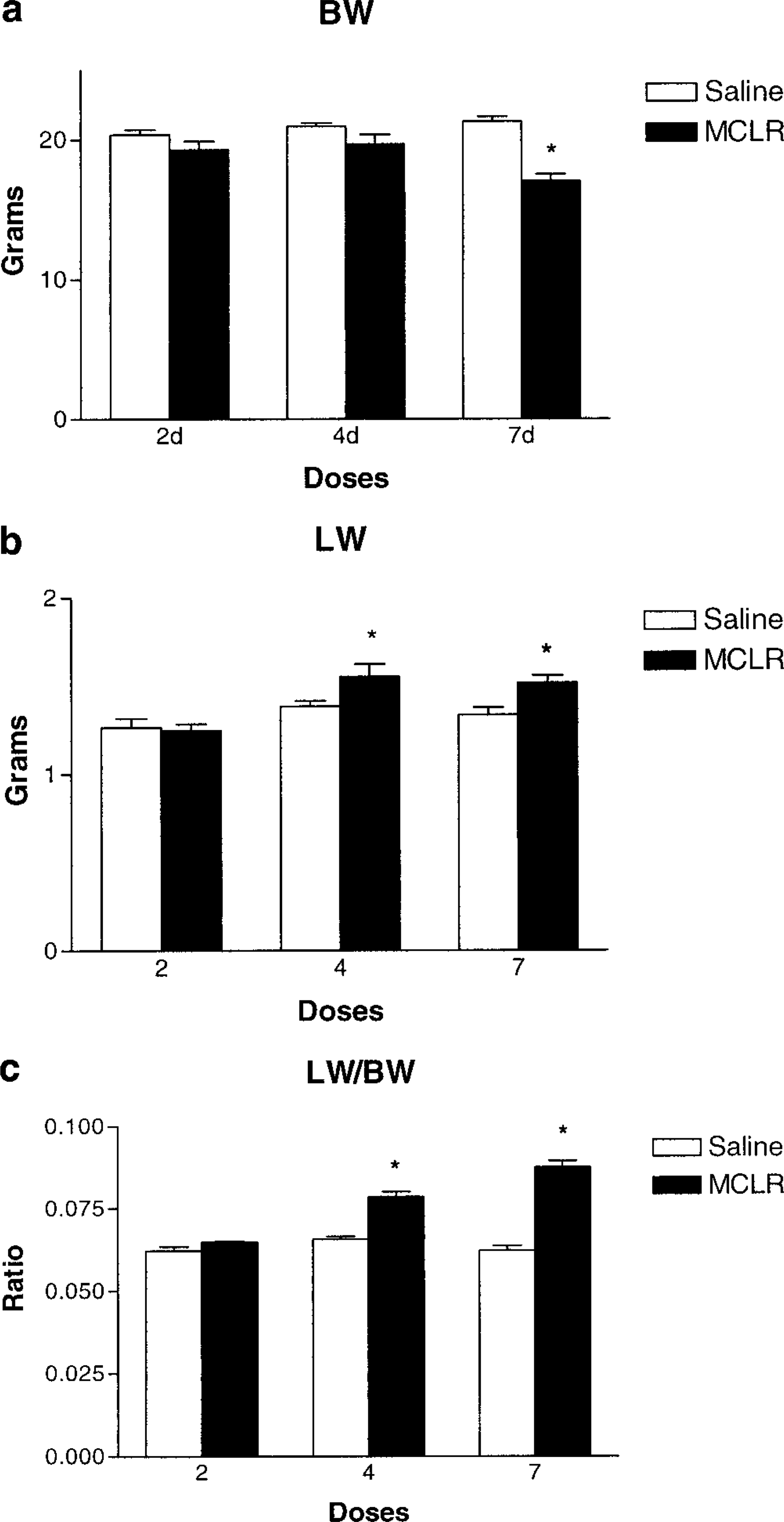
Effect of MCLR on body and liver weights. Fig. 1a Body weights of Balb/C mice after two four, and seven doses of microcystin-LR at 45 µg/kg intraperitoneally every 24 hours, compared to age-matched saline-treated controls. Fig. 1b Absolute liver weights. Fig. 1c Liver to body weight ratios. Values represent the mean ± SD. An asterisk (∗) indicates P < 0.05 (Students't-test).
Histopathology and immunohistochemistry, single sublethal dose
Lesions were evident as early as 2 hours after injection and consisted of centrilobular to midzonal hepatocellular hypertrophy. These enlarged hepatocytes had mild karyomegaly and exhibited increased eosinophilia with loss of the cytosolic vacuolation characteristic of the liver from healthy nonfasted mice. These areas of hypertrophy were sharply demarcated from the surrounding normal periportal hepatocytes (Fig. 2). MCLR immunohistochemistry (MCLR IHC) exhibited strongly positive staining of the centrilobular and midzonal hepatocytes that colocalized with areas of hypertrophy (Fig. 3a, b). At the cellular level staining was strongest in the cytoplasm, whereas some nuclei, especially those closer to the central veins, were weakly to moderately positive. Staining in the cytosol was diffuse and finely granular with coarsely granular deposits around the nuclear membrane and bile cannaliculus.
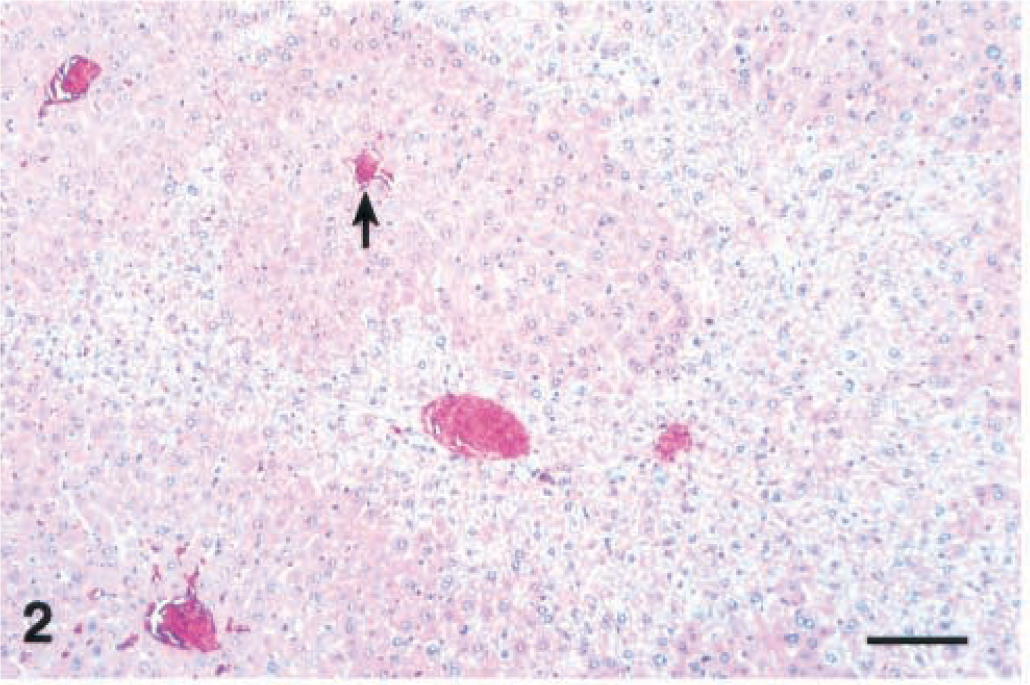
Liver; Balb/C mouse 2 hours after a single intraperitoneal dose of microcystin-LR (45 µg/kg). Note sharply demarcated areas of centrilobular (arrow; central vein) to midzonal hepatocellular hypertrophy with loss of cytosolic vacuolation. HE. Bar = 100 µm.
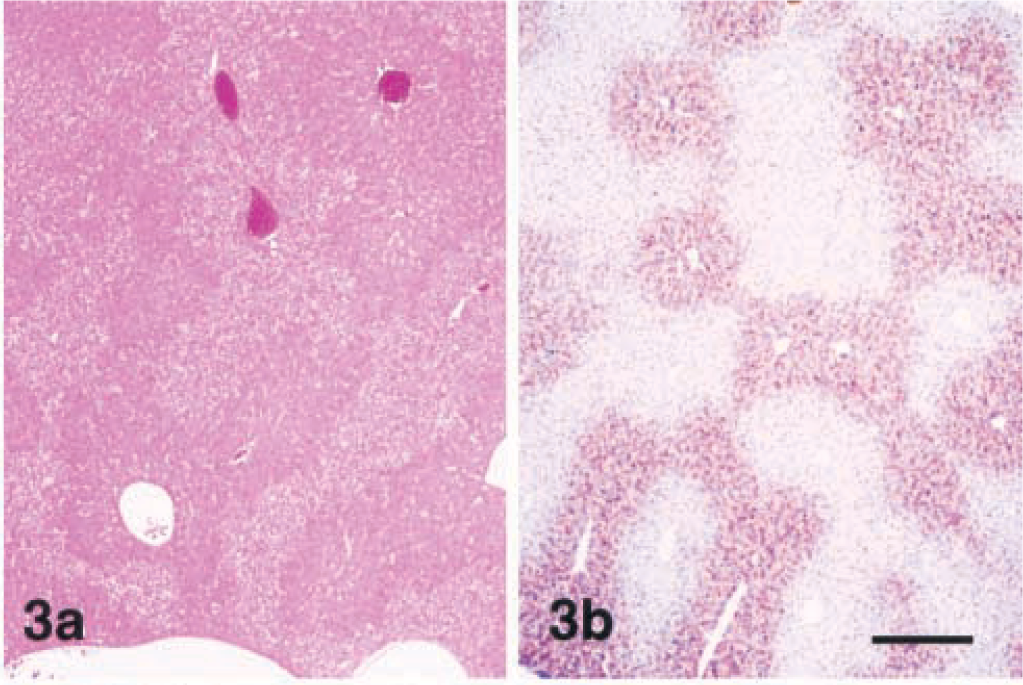
Lesions were similar after 4 and 12 hours, but individual centrilobular hepatocytes in the zones of hypertrophy exhibited cytoplasmic condensation in the form of poorly defined, eosinophilic, eccentrically located inclusions that were partially aggregated along the outer nuclear membrane (Fig. 4). These areas of cytoplasmic condensation were weakly positive for cytokeratin by IHC (Fig. 5). At 4 hours after the dose, the extent of MCLR immunoreactivity was similar and also colocalized within the zones of hypertrophy (Fig. 6). Numerous centrilobular and midzonal hepatocytes exhibited intense nuclear staining that was stronger than in the cytosol. At the cellular level, strong coarsely granular apical (pericanalicular) staining was commonly seen in the cytosol (Fig. 7).
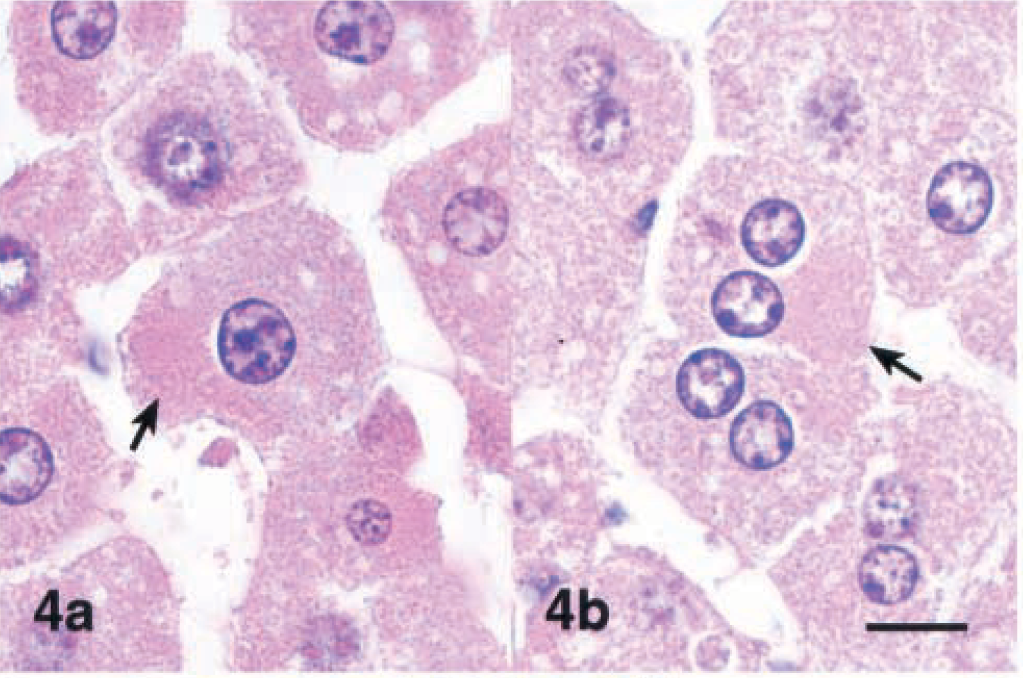
Liver; Balb/C mouse 4 hours after a single intraperitoneal dose of microcystin-LR (45 µg/kg). Fig. 4a and 4b Note eccentric areas of eosinophilic cytoplasmic condensation (arrows). HE. Bar = 10 µm.
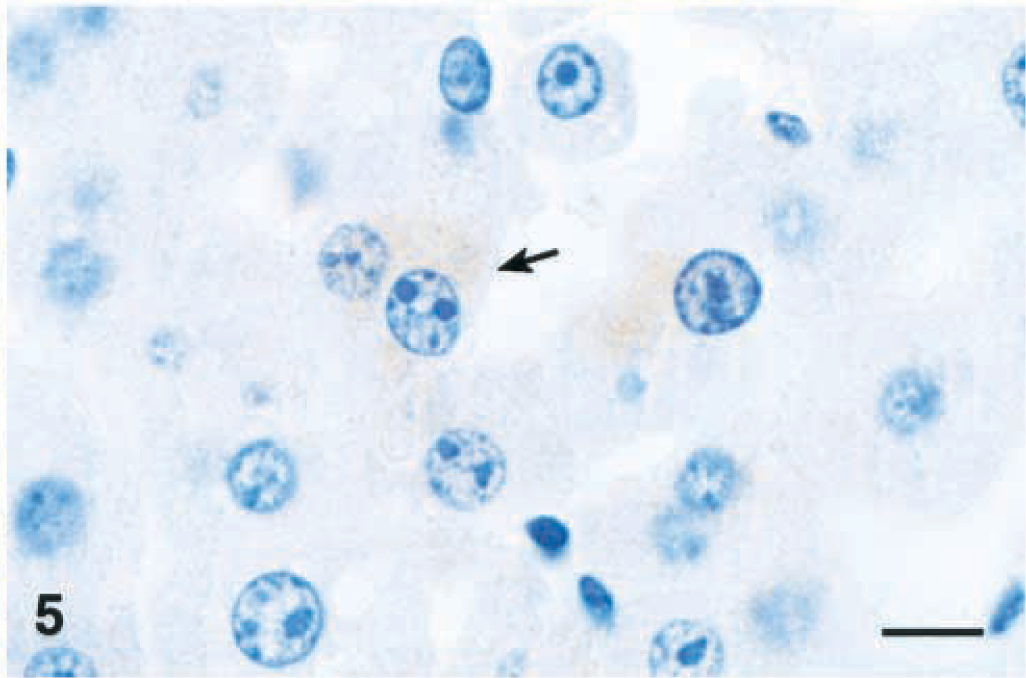
Liver; Balb/C mouse 4 hours after a single intraperitoneal dose of microcystin-LR (45 µg/kg). Note that areas of cytoplasmic condensation are weakly positive (arrow). Cytokeratin immunohistochemical stain. Bar = 10 µm.
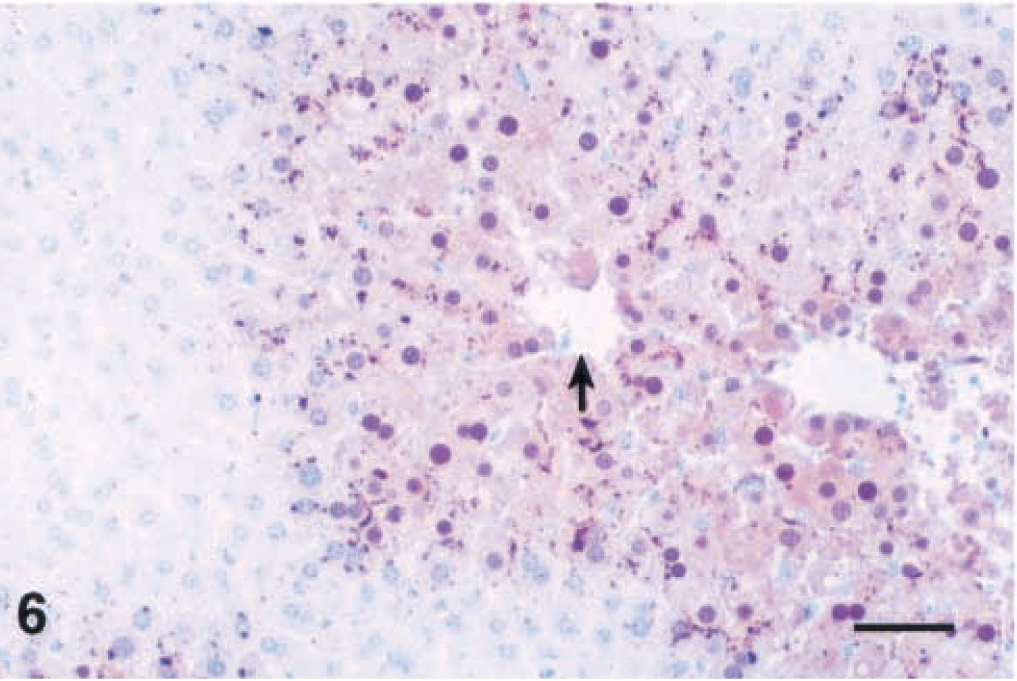
Liver; Balb/C mouse. Microcystin-LR (MCLR) immunohistochemical stain 4 hours after a single intraperitoneal dose of MCLR (45 µg/kg). Note positive staining of the centrilobular to midzonal areas with strong staining of nuclei (arrow; central vein). Bar = 50 µm.
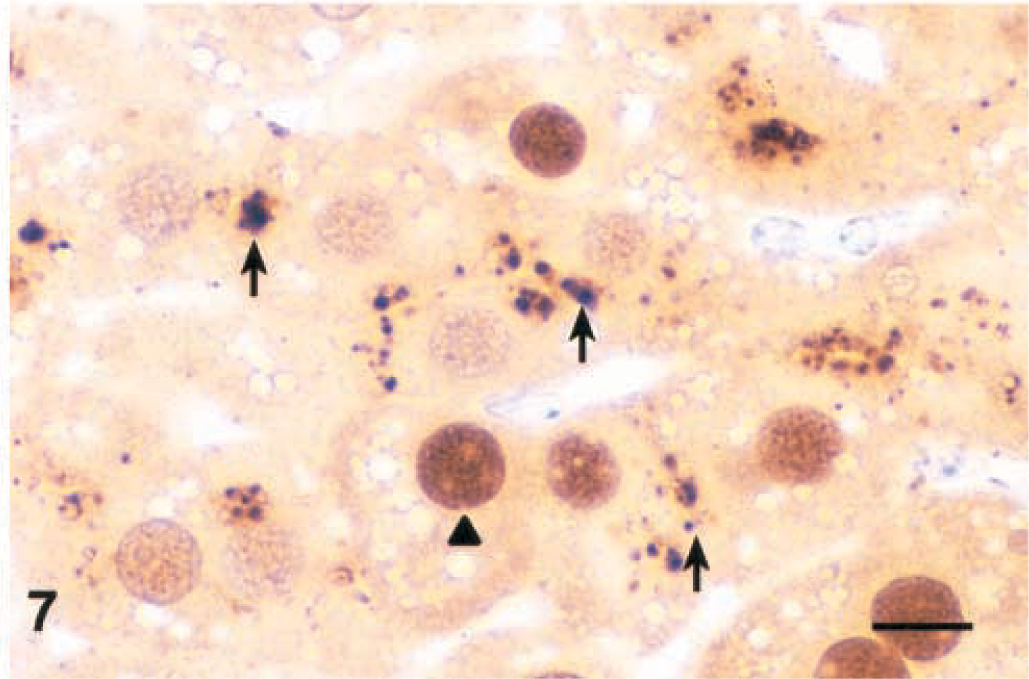
Liver; Balb/C mouse. Microcystin-LR (MCLR) immunohistochemical stain 4 hours after a single intraperitoneal dose of MCLR (45 µg/kg). Note strong coarsely granular cytoplasmic staining in the apical region of hepatocytes (arrows) and strong nuclear staining (arrowhead). Bar = 10 µm.
At 24 hours after dosing, lesions were similar but in addition small numbers of hepatocytes with morphologic features of apoptosis were scattered throughout the zones of hypertrophy, more commonly adjacent to the central vein (Fig. 8). At 12 and 24 hours the same areas were stained, but most nuclei were weakly to moderately positive. Some nuclei had stronger staining than in the cytoplasm but significantly fewer than at 4 hours.
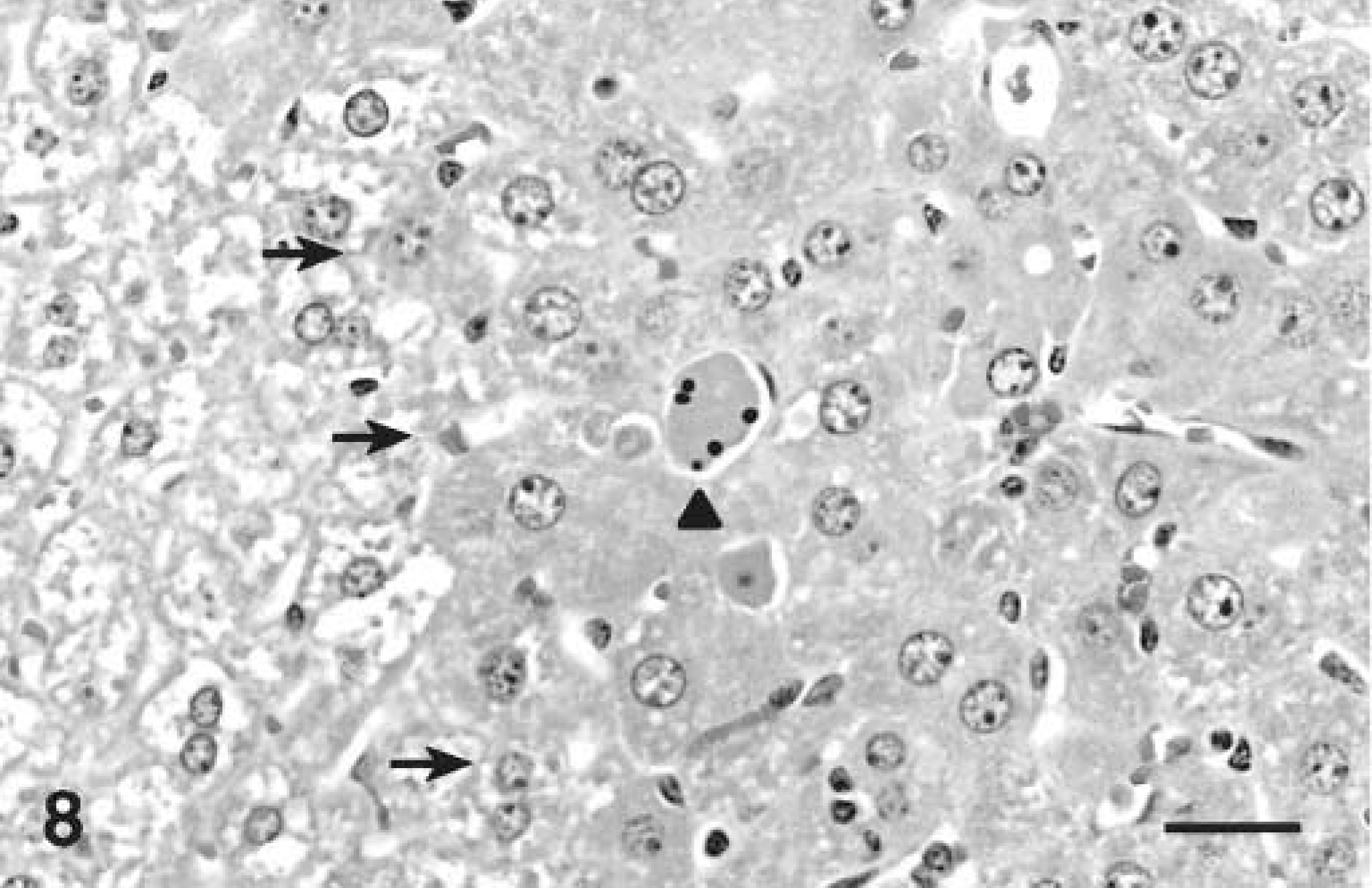
Liver; Balb/c mouse liver 24 hours after a single intraperitoneal dose of microcystin-LR (45 µg/kg). Note apoptotic hepatocyte (arrowhead) among hypertrophic hepatocytes. Note the approximate line of demarcation between normal and hypertrophic hepatocytes (arrows). HE. Bar = 25 µm.
Histopathology and immunohistochemistry, repeated dose study
The livers from mice that received two doses 24 hours apart had multiple hepatocytes with features of apoptosis in the centrilobular areas. Apoptotic hepatocytes had shrunken, hypereosinophilic cytoplasm and were surrounded by clear spaces. Nuclei were fragmented into small pyknotic bodies or those that were still intact had condensed chromatin along the inner nuclear membrane in the form of dense crescent-shaped bodies typical of apoptosis (Fig. 9). Apoptotic bodies with and without chromatin were also present. The staining pattern of MCLR IHC was similar, but periportal hepatocytes were moderately positive. Most apoptotic bodies, with or without chromatin, were intensely stained. Nuclei were typically moderately positive and of equal or less staining intensity than the cytoplasm.
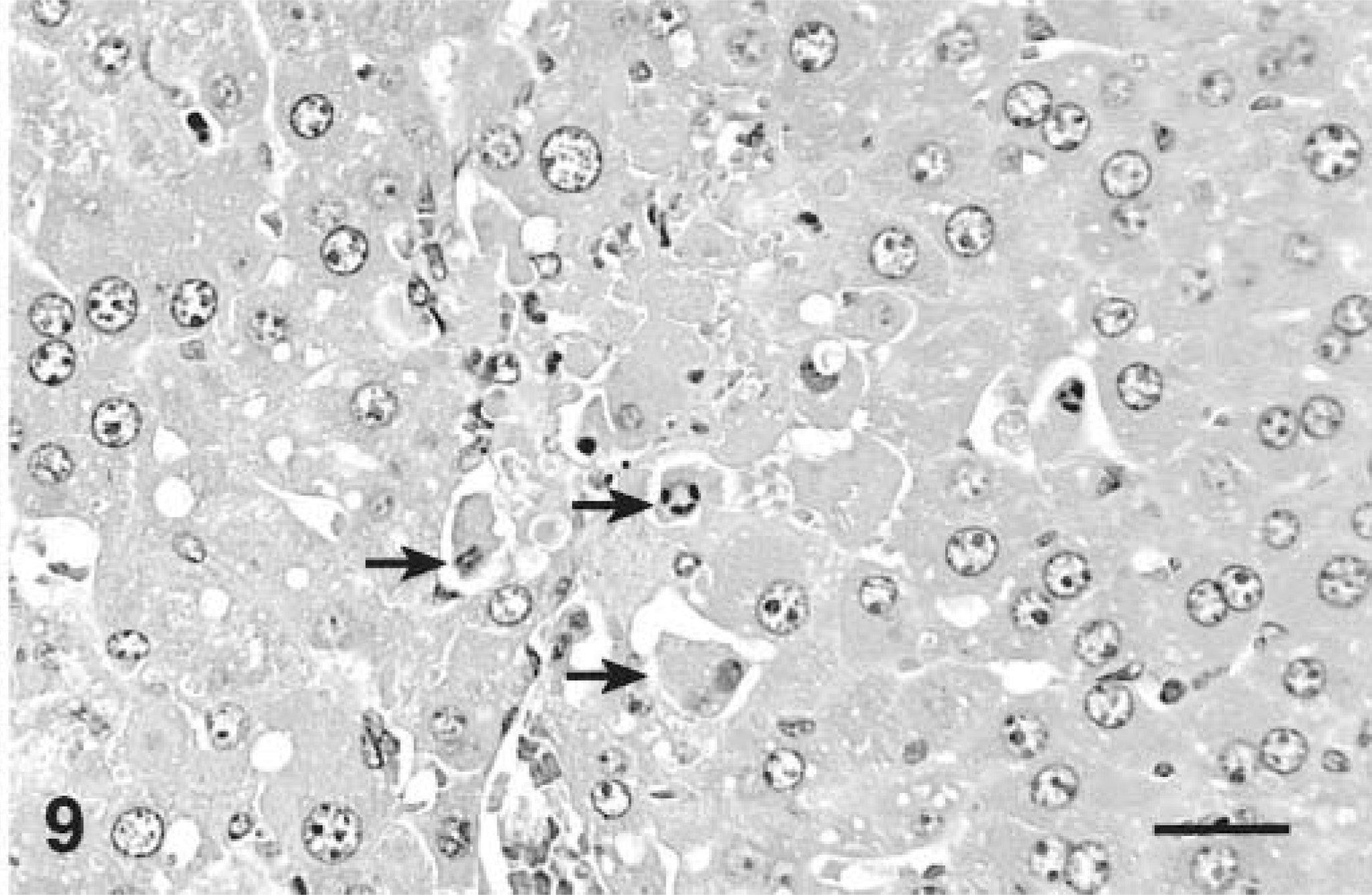
Liver; Balb/C mouse treated with 45 µg/kg of microcystin-LR intraperitoneally daily for 2 days and euthanatized 24 hours after the last dose. Note many centrilobular hepatocytes with features of apoptosis (arrows). HE. Bar = 25 µm.
Livers of mice that received four and seven doses were characterized by marked hepatocytomegaly and karyomegaly with moderate parenchymal disarray that involved the entire hepatic lobule. Hepatocytes in both groups were disorganized and not arranged in cords. Hepatocytes contained abundant, dark eosinophilic cytoplasm that was often markedly vacuolated. Vacuoles were single to multiple, round, variably sized, clear, and well demarcated. Multinucleated hepatocytes containing as many as seven nuclei per cell were commonly seen, mostly adjacent to the central vein. The enlarged nuclei contained coarsely clumped, marginated chromatin with one or two prominent magenta nucleoli (Figs. 10, 11). Small numbers of apoptotic bodies were seen in both treatment groups. No lesions were detected in any of the controls treated with saline vehicle only (Fig. 12). In MCLR IHC all hepatocytes in both treatment groups were diffusely stained but centrilobular hepatocytes had darker granular cytoplasmic staining and nuclei were weakly to moderately positive.
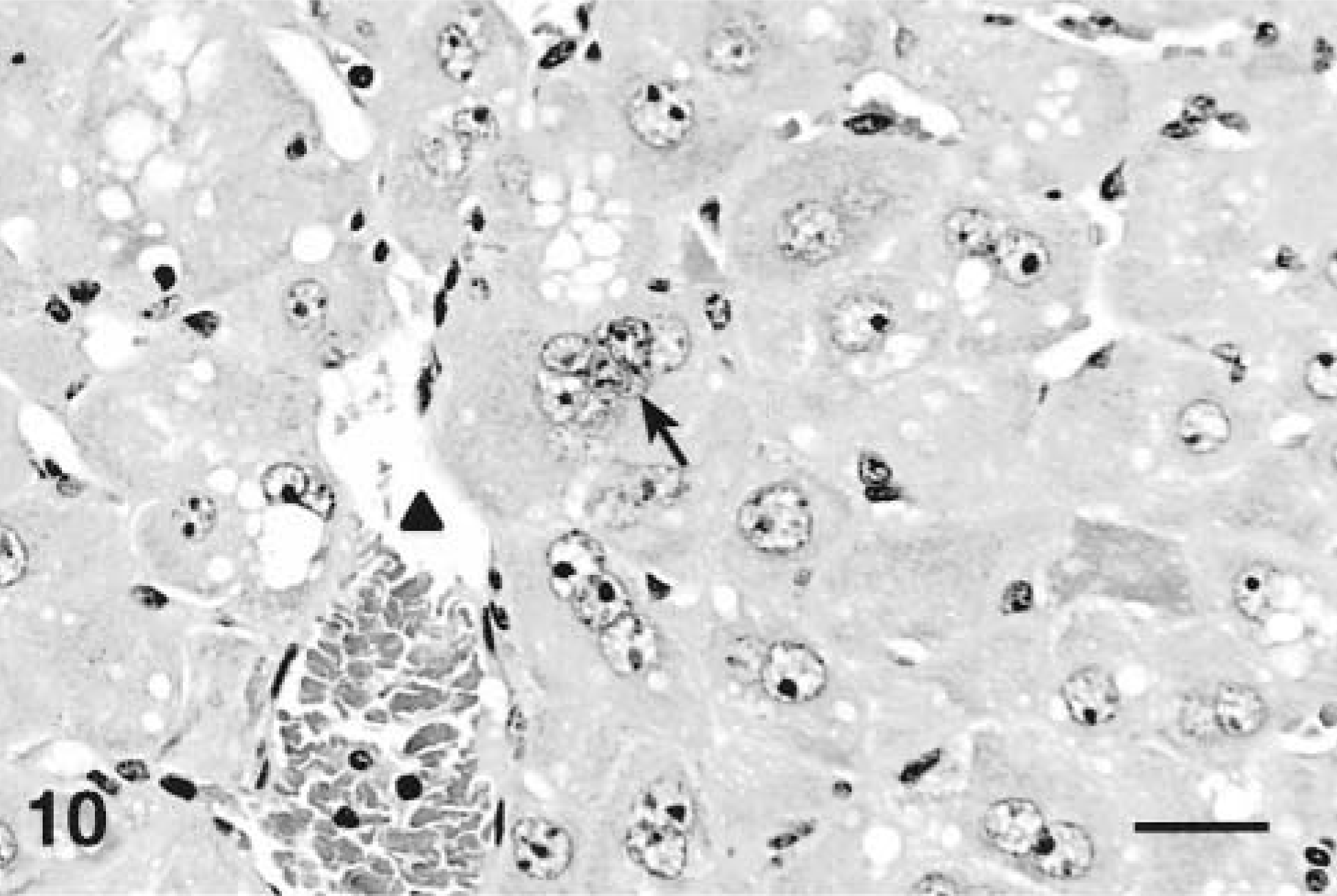
Liver; Balb/C mouse treated with 45 µg/kg of microcystin-LR intraperitoneally daily for 7 days and euthanatized 24 hours after the last dose. Note multinucleation (arrow) and hepatocytomegaly (arrowhead; central vein). HE. Bar = 25 µm.
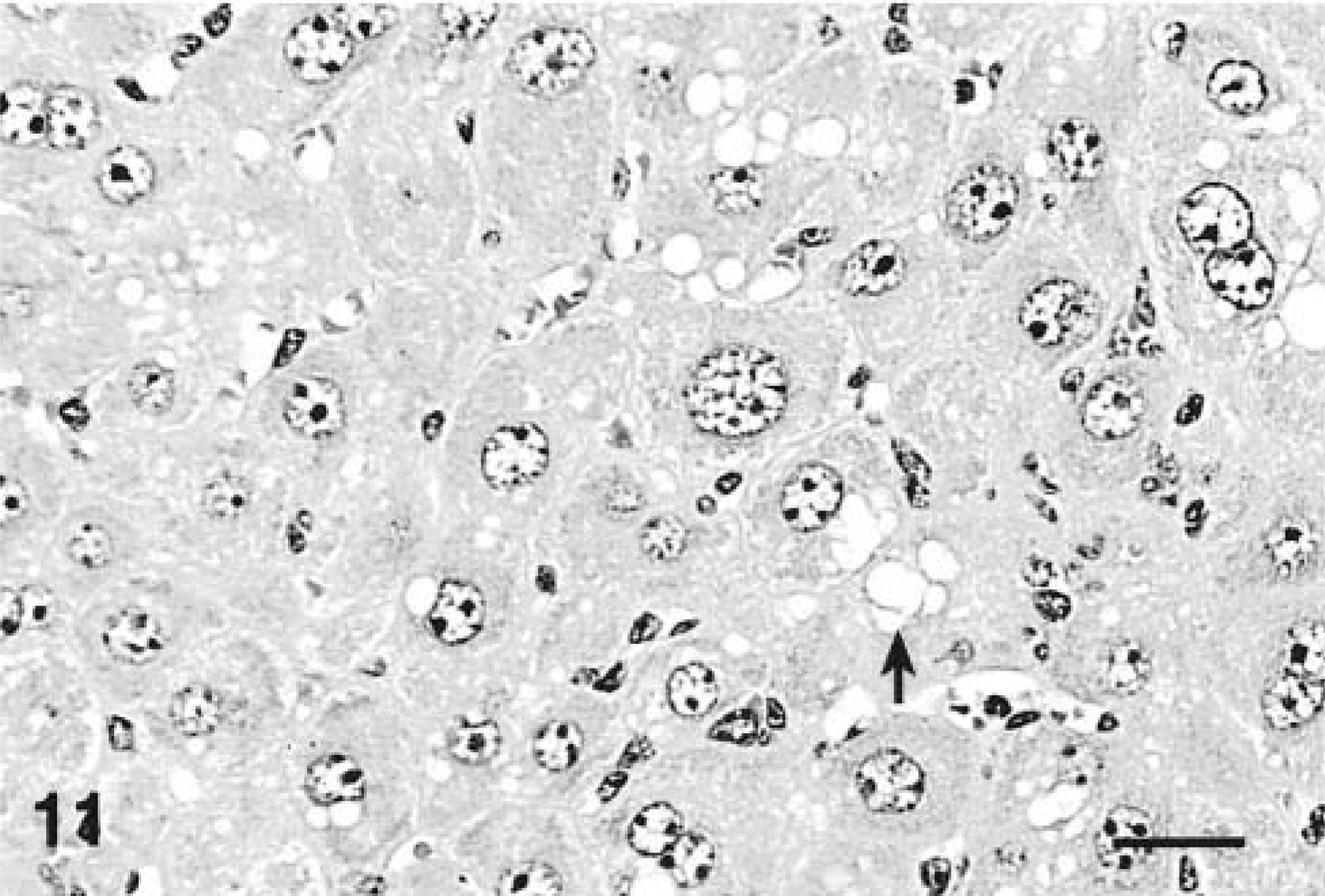
Liver; Balb/C mouse treated with 45 µg/kg of microcystin-LR intraperitoneally daily for 7 days and euthanatized 24 hours after the last dose. Note hepatocytomegaly and karyomegaly with cytosolic vacuolation (arrow). HE. Bar = 25 µm.
Liver; Balb/C mouse. Age-matched saline-treated control from 7-day repeated-dose study. HE. Bar = 25 µm.
No positive staining was seen in any of the saline-treated controls. As a positive control for acute lethal toxicity, the immunohistochemical procedure was applied to liver tissue from mice exposed to a lethal dosage (100 µg/kg) of MCLR that died within 1 hour and 15 minutes. In those cases, strong staining was seen in almost the entire lobule, including hepatocytes that were rounded up and dissociated by hemorrhage. Only a narrow band of periportal hepatic cords remained negative. Within individual hepatocytes, staining in the nuclear and cytosolic compartments was strong but of roughly equal intensity.
Western blotting
Based on the unequivocal nuclear staining demonstrated by IHC in the livers of mice euthanatized at 4 hours after dosing, the same tissues were used for western blotting analysis of nuclear extracts (Fig. 13). In membranes blotted with protein from nuclear extracts and probed with the anti-protein phosphatase 1 polyclonal antibody, a single distinct band was detected that migrated to approximately the 40-kDa location, which corresponds to the molecular weight of the protein phosphatases (42 kDa). Bands from control and treated animals were of equal size, suggesting that treatment with MCLR did not have an effect on the quantities of detectable protein phosphatases 1 and 2A in the nucleus. When the same membrane was reprobed with anti-MCLR primary antibody after removal of the anti-PP1/2A antibody, distinct bands were visible at the exact same location as the protein recognized by the anti-PP1 antibody (40 kDa) in those animals that were treated with one dose of MCLR, but no bands were present in the saline-treated controls. Because the molecular weight of MCLR is only approximately 900 d, these findings suggest that MCLR is covalently bound to protein phosphatases 1 and 2A. Similar bands were detected by the anti-PP1 antibody in the cytosolic extracts but of significantly lower density. The same bands were also detected by the anti-MCLR antibody in the animals treated with the toxin but not in those treated with saline only. When a duplicate membrane was incubated with the same concentration of anti-MCLR antibody but in the presence of excess MCLR toxin, no bands were recognized. This is consistent with neutralization of the primary antibody by binding to excess antigen in the aqueous phase and further validates the specificity of the antibody used in the assay.
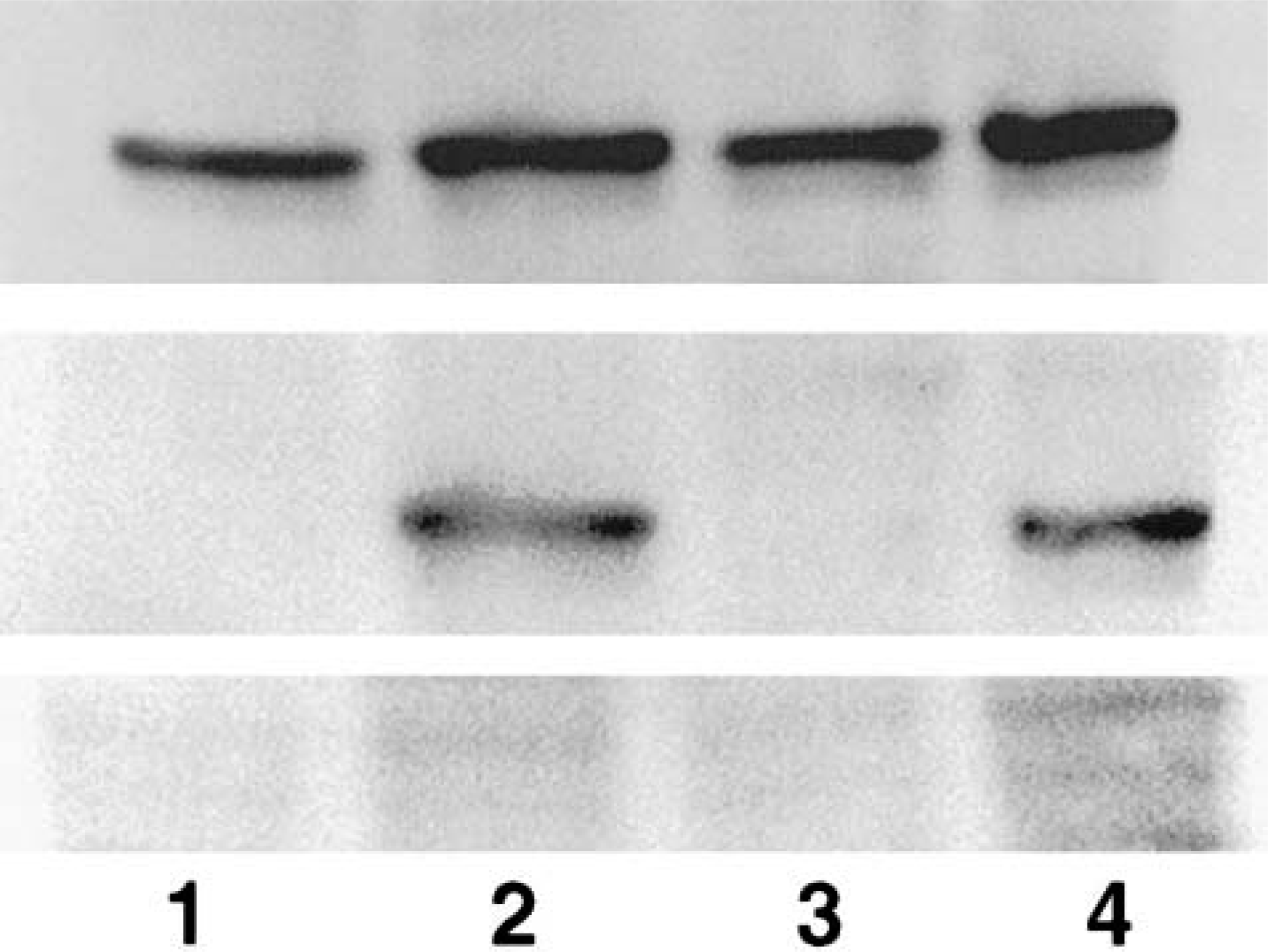
Western blot of nuclear extracts from mouse liver treated with 45 µg/kg of microcystin-LR (MCLR) intraperitoneally or saline and euthanatized after 4 hours. Top panel: Bands detected by anti-protein phosphatase 1/2A antibody approximately at the 40-kDa location. Middle panel: Reprobing of the same membrane with anti-MCLR monoclonal antibody detected single bands at the same molecular weight. Membrane was reprobed after a stripping process to remove previously bound antibodies. Bottom panel: Activity of the anti-MCLR monoclonal was neutralized by addition of excess MCLR toxin. Lanes 1 and 3: Extracts from saline-treated controls. Lanes 2 and 4: Extracts from MCLR-treated animals.
Discussion
Acute or subacute exposure at the dosage used in our study did not cause any deaths or signs of clinical illness but induced a set of hepatic lesions that are different from those generally associated with lethal exposure to MCLR. The range of lesions after a single dose included hepatocellular hypertrophy, intracytoplasmic eosinophilic inclusions, and apoptosis. Repeated doses induced marked hepatocytomegaly and karyomegaly, with hepatocellular disarray and loss of architecture. Cytoplasmic vacuolation and apoptosis were also present. In addition, this study demonstrated that the toxin is immunohistochemically detectable as early as 2 hours after a single sublethal dose and confirmed the nuclear labeling as reported by others 35 but without histologic evidence of cell death or hemorrhage. Furthermore, the presence of the toxin within the nuclear compartment in a protein-bound form, specifically to protein phosphatases 1 and 2A, was demonstrated by western blotting.
The presence of hypertrophic hepatocytes was described in mice that were examined 7 days after sublethal IP injections at dosages that ranged from 20 to 45 µg/kg. 35 Our results, although consistent with these findings, indicate that this lesion can be detected much earlier after exposure (i.e., 2 hours). The specific cause of this early morphologic change is uncertain but may be associated with alterations of the microfilaments or intermediate filaments that provide structural support to the hepatocyte. Cytoskeletal alterations induced by MCLR include disorganization and redistribution of microtubules, intermediate filaments, and microfilaments with progressive collapse. 31
The loss of cytosolic vacuolation that is normally present in the liver of nonfasted mice is consistent with glycogen depletion. Glycogen synthase and glycogen phosphorylase are the enzymes that induce glycogen synthesis and degradation, respectively. The activity of these enzymes is regulated by phosphorylation. Protein phosphatases are closely associated to hepatic glycogen by a targeting protein and are known to participate in the regulation of these enzymes. Phosphorylation reduces the activity of glycogen synthase and increases the activity of glycogen phosphorylase. 16,17,19 Inhibition of phosphatase activity by MCLR possibly results in increased phosphorylation and activation of glycogen phosphorylase and inhibition of glycogen synthase, the net effect of which is glycogen depletion. In fact, microcystins have been shown to be potent inhibitors of glycogen synthase activity in rat hepatocytes in vitro. 14 At 2 hours after injection, the toxin seems to be localized only to those hepatocytes that have lost cytosolic vacuolation, so a direct correlation seems to exist between the presence of the toxin within hepatocytes and the presumptive depletion of glycogen.
Eccentrically located eosinophilic cytoplasmic condensation that was partially aggregated around the outer nuclear membrane was evident by light microscopy 4 hours after dosing. This condensation was sometimes accompanied by more discrete round globular intracytoplasmic inclusions. The nature of this lesion is uncertain. These structures may represent intermediate filaments and microtubules, which have been shown to be collapsed and condensed around the nucleus in hepatocytes exposed in vitro. 13,31 In rat liver, actin was shown to lose its normal distribution and to aggregate into variably sized foci within the cell. 10 These inclusions somewhat resemble the Mallory body inclusions that are typical of human alcoholic hepatitis and have been shown to consist of hyperphosphorylated, condensed cytokeratin filaments. 27 The use of okadaic acid, another protein phosphatase inhibitor, has been described in a mouse model of Mallory body formation. 36
Phosphatase inhibitors of the okadaic acid class are potent inducers of apoptosis in vitro. 1,4 Hepatocyte apoptosis has been identified as a feature of MCLR toxicity in mice, 34,35 rats, 9,26 and trout. 3 Our studies in mice are consistent with others and indicate a high susceptibility of hepatocytes to undergo programmed cell death with MCLR exposure even with a sublethal dosage of 45 µg/kg that was not associated with hemorrhage or necrosis. In our study, apoptosis did not become detectable until 24 hours after dosing. A marked increase in apoptosis was seen in animals that received a second dose and still without evidence of necrosis or hemorrhage. However, apoptotic bodies were rare in the four- or seven-dose animals. After the second dose in which hepatocyte loss is significant, the liver possibly begins a regenerative phase and apoptosis, which in its initial phases is a short-term event, 6 possibly becomes less evident
Repeated sublethal doses of MCLR had no apparent ill effect on the health of the animals, except in animals that received a total of seven doses, which experienced a significant loss of body weight. The loss of body weight possibly resulted from a decrease in food consumption. Although food intake was not monitored in this study, a similar trend in loss of body weight has been observed in rats after exposure for 28 days, but food consumption in that study was comparable to that of saline-treated controls (unpublished observations). Also, significant increases in absolute liver weights and liver to body weight ratios were seen with four and seven doses of toxin. However, in a rat model of prolonged exposure, liver weights and liver to body weight ratios were not different from controls despite evidence of hepatotoxicity, suggesting that increased hepatic mass is a poor indicator of exposure to sublethal levels of MCLR. 26
Livers of mice treated with 45 µg/kg once a day for 4 and 7 days were characterized by hepatocytomegaly with karyomegaly, parenchymal disarray, and loss of architecture. Based on these histologic changes, the increase in liver weights was most likely, at least in part, due to the pronounced increase in cell size. Hepatic megalocytosis is regarded as the hallmark lesion of “net pen liver disease,” a disease of pen-reared Atlantic salmon thought to be caused by microcystins. 32 However, an association between MCLR exposure and hepatic megalocytosis has not been mentioned in mammals. The orderly progression of the cell cycle is controlled by several proteins such as the cyclin-dependent kinases, retinoblastoma protein, and p53 tumor suppressor protein, all of which are regulated by phosphorylation. 15,24,33 Phosphatase inhibitors such as okadaic acid and the antitumor agent fostriecin have been shown to induce cell cycle arrest in different cell lines in vitro. At low concentrations in vitro, okadaic acid acts as a cytostatic agent, but at higher concentrations it is an inducer of cell death by apoptosis. 2,30 Repeated sublethal exposure in vivo to the phosphatase inhibitor MCLR possibly causes hepatocytomegaly by inhibiting mitosis; however, further studies are needed to confirm this hypothesis. Karyomegaly and multinucleation frequently were observed, which is also consistent with an inability of hepatocytes to undergo effective mitosis despite continued DNA synthesis.
In our experiments, the toxin was easily detectable in hepatocytes at a dose not associated with overt toxicity and rather early after exposure. Previous reports on the use of IHC in mice localized the toxin to centrilobular hepatocytes in mice that died within 6 hours after a lethal dose or 24 hours after dosing but with a higher dosage (69 µg/kg) that induced significant necrosis and hemorrhage. 35 In rats that received 50 µg/kg daily for 28 days the toxin was also localized to centrilobular hepatocytes. 26 Analysis of the immunohistochemical results of this study revealed that the toxin is detectable as early as 2 hours after a single sublethal dose and in the absence of hemorrhage or necrosis. Repeated dosing for 4 and 7 days resulted in diffuse toxin localization throughout the entire hepatic lobule, suggesting that the toxin accumulates in the liver faster than it can be excreted. This is consistent with toxicokinetic studies in mice, which indicated that the toxin is retained in the liver in a covalently bound form over prolonged periods of time. 21 The apparent bioaccumulation of toxin also was suggested by studies of prolonged exposure in rats. 26 The early and consistent detection of the toxin by IHC suggests that the technique may be of help in the diagnosis of exposure in natural cases.
Our immunohistochemical results demonstrate significant staining of nuclei after sublethal exposure, which was detected earlier than previously reported. Nuclear labeling of mouse liver has been reported only after lethal doses in animals that died within 6 hours in association with areas of hemorrhage. Nuclear labeling was also reported in the liver of trout 72 hours after oral dosing. 3 In our study, unequivocal labeling of the nuclei was evident at 4 hours after a sublethal dose. At this time point, many centrilobular hepatocytes had stronger nuclear reactivity than in the cytoplasm and in the absence of centrilobular hemorrhage or dissociation. However, the strong nuclear labeling was lost after 12 hours and was not seen in either of the groups that received repeated doses. In these animals, nuclear staining was weak to moderate and of equal or lesser intensity than in the cytoplasm. This could be the result of redistribution of the toxin within the cell. Once internalized into hepatocytes, the toxin undergoes a rapid and stable interaction with a protein of an approximate molecular weight of 40 kDa, consistent with protein phosphatases 1 and 2A. 22 Western blotting analysis of nuclear extracts confirmed the immunohistochemistry findings and showed for the first time that in the nucleus the toxin is bound to protein phosphatases 1 and 2A. Furthermore, the association of toxin with protein phosphatases on denaturing SDS gels confirms that the link of MCLR to PP1 and 2A is covalent. This covalent interaction was detected in animals euthanatized 4 hours after a sublethal dose; thus, the strong nuclear labeling observed by IHC truly represents the cellular distribution of the toxin. MCLR has been postulated to inhibit phosphatase activity by a covalent interaction; however, this is not essential for inhibition of the enzyme. 5 Free unbound MCLR may also contribute to the overall inhibitor activity. 28 How the presence of toxin within the nucleus contributes to the toxic effects of MCLR is uncertain. However, decreased synthesis of hepatic ALT messenger RNA was demonstrated in the rat model of prolonged exposure, 25 suggesting that the toxin has an effect on nuclear function.
In conclusion, our experiments demonstrated that sublethal exposure to MCLR induces distinct lesions in the liver different from those associated with acute lethal exposure. Within 24 hours after a single sublethal dose, lesions included hepatocellular hypertrophy with loss of cytosolic vacuolation, areas of eosinophilic cytoplasmic condensation that were weakly cytokeratin positive, and apoptosis. Repeated daily doses for up to 7 days induced hepatocytomegaly and karyomegaly with parenchymal disarray. Our studies show that MCLR is easily detectable by IHC as early as 2 hours after sublethal exposure; thus, immunohistochemical analysis may be a useful diagnostic tool to monitor exposure in nature. The toxin seems to target the nuclei, where it accumulates bound to protein phosphatases 1 and 2A. Studies are underway to determine if this accumulation results in significant inhibition of nuclear phosphatase activity and its effects on the phosphorylation status of nuclear phosphoproteins.
Footnotes
Acknowledgements
We thank Dr. Wayne Carmichael (Wright State University, Dayton, OH) for supplying the purified MCLR, Dr. Fun S. Chu (University of Wisconsin, Madison) for the anti-MCLR monoclonal antibody, and Jane Chladny (University of Illinois, College of Veterinary Medicine) for performing the immunohistochemical stains.
