Abstract
In this investigation, the nature and distribution of histologic lesions and immunohistochemical staining (IHC) of a proteinase-resistant prion protein were compared in free-ranging mule deer (Odocoileus hemionus) dying of a naturally occurring spongiform encephalopathy (SE) and captive mule deer dying of chronic wasting disease (CWD). Sixteen free-ranging deer with SE, 12 free-ranging deer without SE, and 10 captive deer with CWD were examined at necropsy. Tissue sections were stained with hematoxylin and eosin, and duplicate sections were stained with a monoclonal antibody (F89/160.1.5). Histological lesions in the freeranging deer with SE and captive deer with CWD were found throughout the brain and spinal cord but were especially prominent in the myelencephalon, diencephalon, and rhinencephalon. The lesions were characterized by spongiform degeneration of gray matter neuropil, intracytoplasmic vacuolation and degeneration of neurons, and astrocytosis. IHC was found throughout the brain and retina of deer with SE and CWD. Positive IHC was found in lymphoid tissue of deer with SE and CWD. Histologic lesions and IHC were not found in multiple sections of integument, digestive, respiratory, cardiovascular, endocrine, musculoskeletal, and urogenital systems of deer with SE or CWD. Comparison of histologic lesions and IHC in tissues of free-ranging deer with those of captive deer provides strong evidence that these two diseases are indistinguishable morphologically.
Keywords
Transmissible spongiform encephalopathies (TSEs) are a relatively heterogeneous group of fatal neurodegenerative disorders that occur in both man and animals. These diseases are characterized by vacuolation of neuronal perikarya, neuronal degeneration, spongiform degeneration of gray matter neuropil, and astrocytosis in the brain. 8–11,23,25,27–32 There also is an accumulation of a proteinase-resistant isoform (PrPres) of the prion protein (a normal cellular sialoglycoprotein–PrPc) located in normal brain. PrPres either alone or in combination with another protein 19 may represent a novel transmissible agent, which propagates by catalyzing the conversion of a normal cellular isoform to an abnormal isoform through a nucleation or polymerization event. 4,5 Support of this hypothesis of an abnormal isoform of PrPc as the sole agent for these TSEs is based largely on rodent models of scrapie, in which PrPSc is the major component of infectious tissue extracts. 3
A naturally occurring spongiform encephalopathy (SE) has been documented in free-ranging mule deer (Odocoileus hemionus), white-tailed deer (Odocoileus viginianus), and elk (Cervus elaphus nelsoni) in north central Colorado. 23 This disease has been assumed to be the same condition as chronic wasting disease (CWD), a transmissible spongiform encephalopathy described in captive cervids from Colorado and Wyoming, 30,31 but a comparative study of these two conditions has not been done. One of the concerns regarding these two conditions has been the possible common origin. Did the disease originate within the wildlife facilities at Colorado State University or perhaps the state wildlife research facilities in Colorado and/or Wyoming, then spread to free-ranging cervids, or was the disease originally in the wild and introduced into the facilities, or are these two diseases different but caused by two similar agents? 17,23 There is speculation that there are multiple strains of the causative agent of CWD in deer and elk. While the investigation of the origin of these diseases will require biological and molecular approaches beyond the scope of this present work, a comparison of the pathologic phenotypes of the SE in these two populations of deer can provide a basis for suggesting similarity or variation as proposed previously among other naturally occurring TSEs. 20,24,26,32 The objective of this article is to describe in detail the nature, topographic distribution, and severity of histologic lesions with the distribution and abundance of immunohistochemical staining (IHC) of PrPres in tissues from major organ systems in free-ranging deer with SE and compare these findings with those in captive deer with CWD.
Materials and Methods
A retrospective histologic study was performed on tissues from 28 free-ranging and 10 captive deer. The free-ranging deer were found dead or moribund in north central Colorado (40°15′–40°55′N/103°50′–105°45′W). Captive deer originated from the Colorado Division of Wildlife Foothills Research Facility in Fort Collins, Colorado, where CWD was first described. 30 These captive deer also were found dead or moribund in these facilities. Moribund deer were humanely euthanized with Beuthanasia-D Special® (Schering-Plough Animal Health, Union, NJ). Necropsy of animals was performed at the Colorado State University Veterinary Diagnostic Laboratory, Fort Collins, Colorado. Multiple tissues from major organs were collected and preserved in 10% neutral buffered formalin. Selected tissues were embedded in paraffin, sectioned at 5 µm, and mounted on positive-charged glass slides (Superfrost/plus, Fisher Scientific, Pittsburgh, PA). Tissue sections were stained with hematoxylin and eosin (HE). 13 Duplicate sections were immersed in fresh 88% formic acid (Mallinckrodt, Paris, KY) for 30 minutes, rinsed in tap water, placed in Tris buffer, autoclaved for 20 minutes at 121 C (liquid setting), and cooled for 30 minutes. The sections were then immunostained with monoclonal antibody F89/160.1.5 (MAb) for 30 minutes at 37 C, followed by a biotinylated anti-mouse secondary antibody, an alkaline phosphatase-streptavidin conjugate, a substrate chromogen (fast red), and a hematoxylin counterstain with bluing (Ventana Medical Systems, Tucson, AZ). The technique used for IHC was similar to those previously described. 1,6,7,15–18,21,23 The MAb used in this study reacts with a conserved epitope on the prion protein of Rocky Mountain elk, mule deer, domestic sheep, and cattle. 18 Selected sections of thalamus, hypothalamus, and obex of medulla oblongata in two deer with SE and two deer with CWD were stained for astrocytes using the glial fibrillar astrocytic protein (GFAP) method. 2
Multiple sections from the nervous system, including brain, spinal cord (thoracic region/T3–T6), ganglia (dorsal root g, anterior mesenteric g, trigeminal g), and peripheral nerves (brachial plexus, vagus, sciatic, and sympathetic trunk), were examined. Sections of eyes from three deer were examined. Neuroanatomical locations and nomenclature used in this article were primarily from Singer; 22 additionally, Yoshikawa 33 and Jenkins 12 were consulted.
Representative sections of the lymphohematopoietic, digestive, respiratory, cardiovascular, endocrine, musculoskeletal, integument, and urogenital systems were examined with HE and MAb.
One free-ranging deer with SE was pregnant. Multiple tissues from the fetus were sectioned, stained with HE and MAb, and examined.
Tissues from major organs from each deer were examined under light microscopy, and the prevalence and severity of lesions and IHC were tabulated (Table 1) The prevalence of lesions was based on the frequency with which spongiform degeneration and neuronal vacuolation occurred at a particular neuroanatomic site. Severity of lesions in the brain was graded on a scale of 0–3, with 0 = no lesions, 1 = mild, 2 = moderate, and 3 = severe. The primary histologic changes used to judge severity of lesions were the degree of spongiform degeneration in gray matter neuropil with neuronal cytoplasmic vacuolation and degeneration. Since the dorsal motor nucleus of the vagus (DMNV) was the most consistently and severely affected nucleus with spongiform degeneration, the characteristics of the lesion in it were graded as severe. The severity of lesions in other neuroanatomical sites was scored accordingly.
Data comparing the distribution and severity of spongiform encephalopathy and immunohistochemical staining of PrPres in specific neuroanatomic regions in the nervous system of free-ranging mule deer with a naturally occurring spongiform encephalopathy to those in captive mule deer with chronic wasting disease.
∗ Number of times a specific neuroanatomic site had spongiform encephalopathy (SE) over the number of times the site was examined (N), followed by the average severity (0–3) of the lesion (in parentheses).
† Number of times a specific neuroanatomic site had positive immunohistochemical staining (IHC) of PrPres over the number of times the site was examined (N), followed by the average abundance (0–3) of staining (in parentheses).
‡ Total number of neuroanatomic sites examined in a region of brain with spongiform encephalopathy over the total number of sites examined in this region.
§ Total number of neuroanatomic sites examined in a region of brain with positive immunohistochemical staining of PrPres over the total number of sites examined in this region.
∥ Percentage of neuroanatomic sites in a region of the brain that had histologic lesions of spongiform encephalopathy, followed by the average severity (0–3) of the lesion.
# Percentage of neuroanatomic sites within a general region of the brain that had positive immunohistochemical staining of the PrPres, followed by the average abundance (0–3) of staining.
¶ Thalamic nuclei examined: medial dorsal, ventral rostral, central medial, ventral posteromedial, ventral posterolateral, pulvinar, medial geniculate, and lateral geniculate.
∗∗ Hypothalamic nuclei examined: paraventricular, dorsomedial, perifornicalis, lateral, ventromedial, and supraopticus.
The prevalence of IHC also was based on the frequency in which positive staining was found at particular neuroanatomic sites. The abundance of IHC was graded on a scale of 0–3, with 0 = no staining, 1 = mild, 2 = moderate, and 3 = heavy. Since the DMNV was the most consistently and severely affected nucleus with regard to the abundance of IHC positive material, this too was graded as heavy and all other sites were scored accordingly. The intensity of the staining was constant throughout, independent of the grading. Positive IHC was interpreted to be PrPres, which has been demonstrated previously in brains of deer and elk with CWD 6,7 and SE. 18,23
Positive lymphoid tissues were characterized by having 50–90% of the follicles filled with a bright red, course, granular material. Sections of tonsil contained about 100–125 follicles and lymph nodes 25–50.
Results
Of the 28 free-ranging deer examined, 12 did not have histologic lesions of SE or IHC in the brain nor did they have IHC in any of the lymphoid tissues. These animals served as negative controls. The remaining 16 free-ranging deer had histologic lesions of SE and positive IHC of PrPres in brain and lymphoid tissues. The lesions in these wild deer were compared with those in the 10 captive deer with CWD.
Nervous system
Spongiform degeneration of the gray matter neuropil and intracytoplasmic neuronal vacuolation and degeneration were the most significant and consistent lesions in the brain of animals with SE or CWD. These lesions have been well documented for TSEs in the literature. 8–11,23,25,27–32 Neuronal vacuolation and degeneration were severe in the DMNV, solitarius nucleus, hypothalamus, thalamus, and olfactory cortex in both groups of deer and were milder in other areas, including the mesencephalon, metencephalon, and myelencephalon in both groups of deer (Figs. 1–4). Neuronal loss also was observed especially in the DMNV, hypothalamus, and thalamus. Vacuolated neurons were often surrounded by spongiform degeneration of the adjacent gray matter neuropil. Vacuoles within neuronal perikarya were either single or multiple.
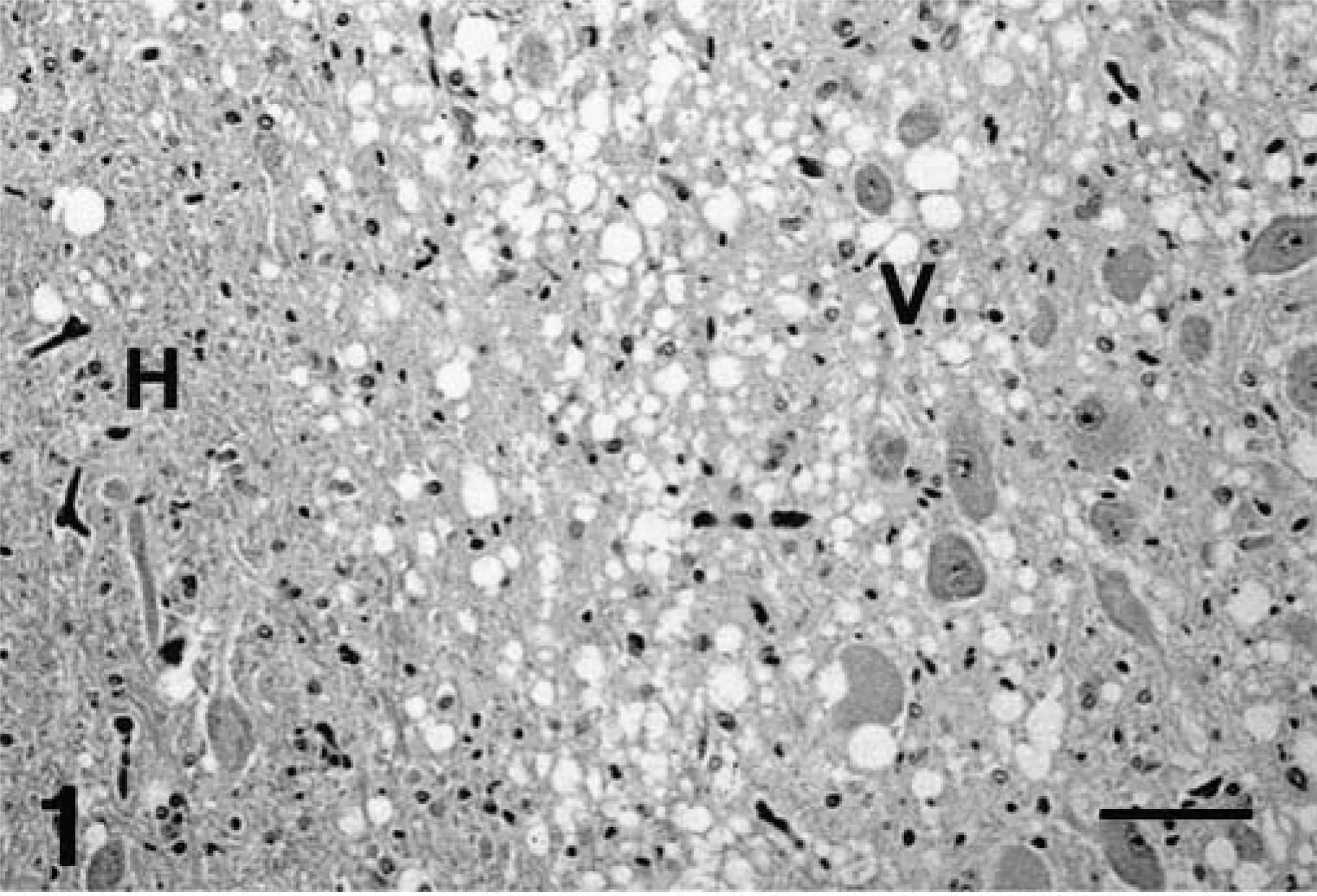
Brain, dorsal motor nucleus of vagus and hypoglossal nucleus; captive deer with chronic wasting disease. Extensive spongiform encephalopathy in the dorsal motor nucleus of the vagus (V) as compared with the hypoglossal nucleus (H). HE. Bar = 95 µm.
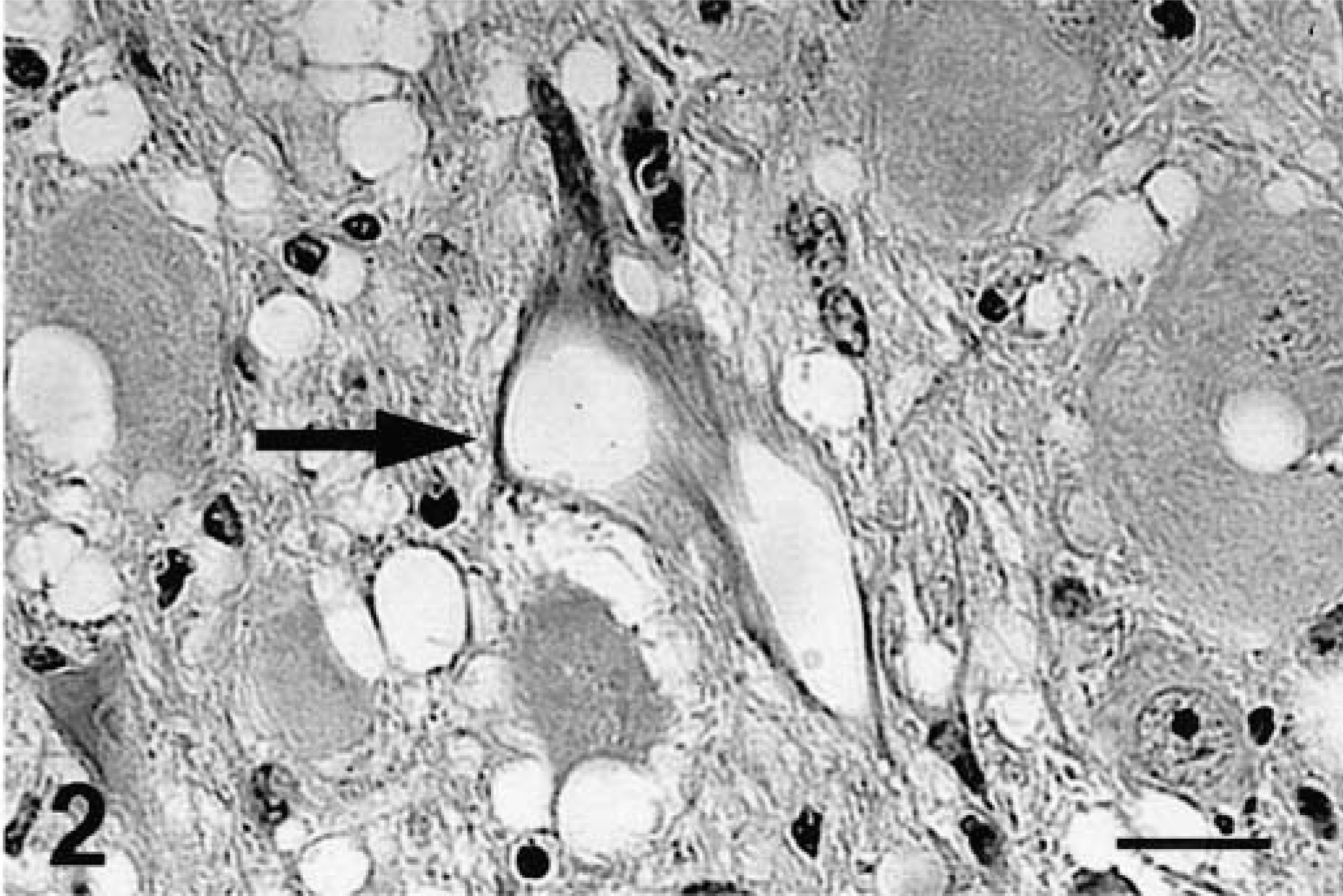
Brain, dorsal motor nucleus of vagus; free-ranging deer with spongiform encephalopathy. Neuronal vacuolation (arrow) and spongiform degeneration of the neuropil. HE. Bar = 37 µm.

Brain, olfactory cortex; free-ranging deer with spongiform encephalopathy. Extensive spongiform degeneration of the olfactory cortex (A). HE. Bar = 133 µm.
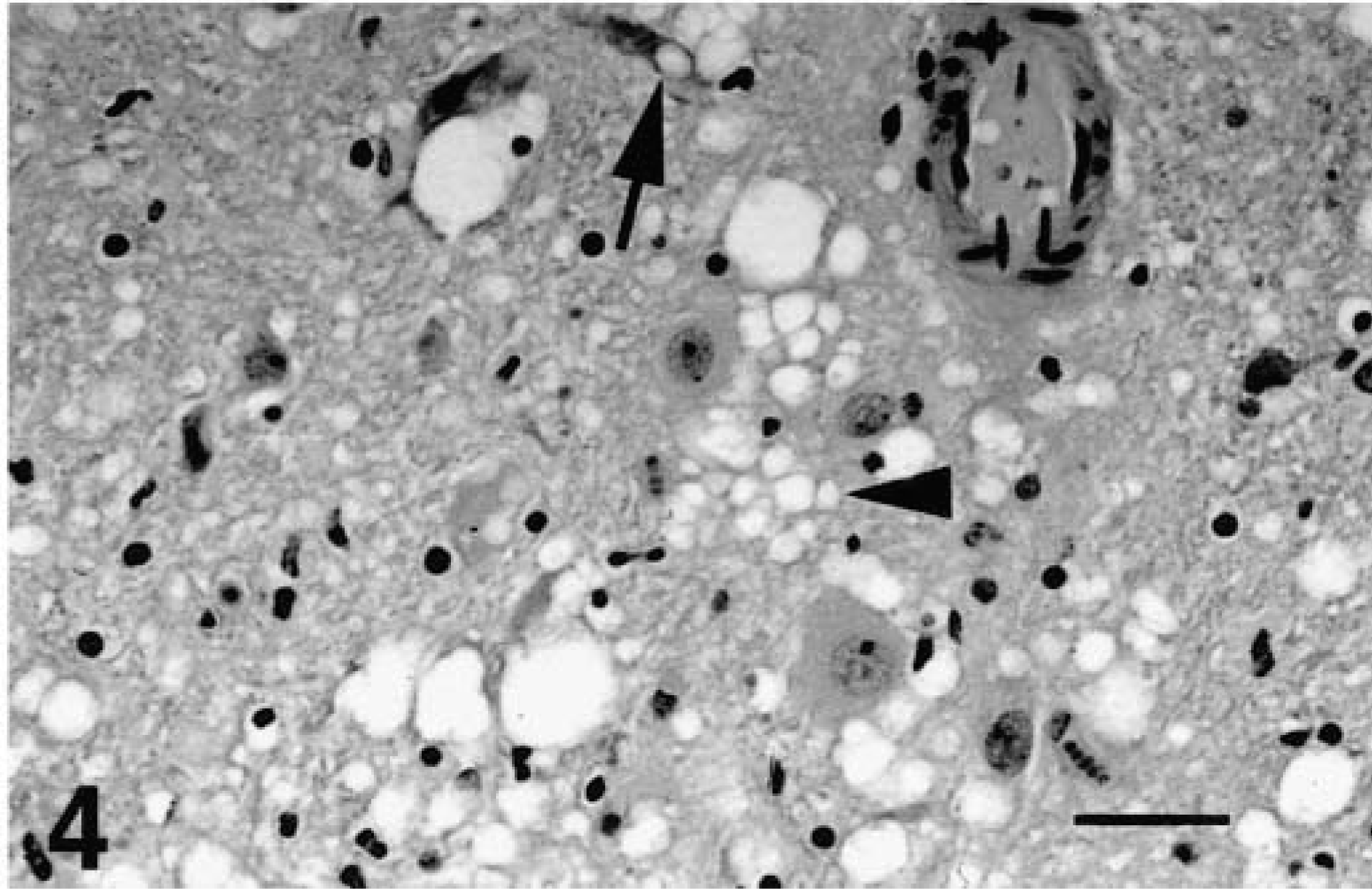
Brain, hypothalamus; free-ranging deer with spongiform encephalopathy. Neuronal vacuolation and degeneration (arrow) and extensive spongiform degeneration of neuropil (arrow head). HE. Bar = 67 µm.
Microcavitation, elliptical spaces, and vacuoles were typical histologic changes located on the periphery of neurons and in the gray matter neuropil. Occasionally, these changes were difficult, if not impossible, to distinguish from artifacts in brains that had undergone autolysis. Vacuoles were usually single, but in some areas were multiple and appeared to be in small clusters. Borders of vacuoles were discrete. Single and multiple vacuoles were common in the molecular layer of the cerebellum. All deer with SE or CWD in this study had these light-microscopic lesions in neural tissues. However, one free-ranging deer with SE had extremely large clusters of vacuoles in the neuropil, unlike the diffuse nature seen in the other cases. These large (0.25–0.5 mm in diameter), well-delineated lesions were primarily located in caudate nucleus, diencephalon, and mesencephalon.
Mild to moderate astrocytosis was present in the thalamus, hypothalamus, and obex of the medulla oblongata in the two deer with SE and two with CWD. These were the only deer that were examined with the GFAP method. Astrocytosis was demonstrated well with the GFAP stain but was difficult to evaluate on HE sections. Because of the difficulty of evaluating astrocytosis without the use of the GFAP stain, astrocytosis was not used as a criterion for evaluating the severity of lesions in brain. Mild lymphoid cuffing of vessels was observed in the thalamus and medulla oblongata in a few affected deer, as well as in nonaffected deer. Occasionally, small focal areas of mineralization were found in the DMNV in both affected and unaffected deer.
These histologic lesions as described above were similar in deer with SE or CWD. The distribution and relative severity of the spongiform encephalopathy and IHC in neuroanatomic sites are given in Table 1. The most consistently and severely affected nuclei in both groups of deer were the DMNV and solitarius nucleus of the myelencephalon. The nature and severity of the spongiform encephalopathy and the IHC were similar in all regions of brain except in the mesencephalon and the spinal cord. Spongiform degeneration of the neuropil and vacuolation of neuronal perikarya and neuronal degeneration were less prevalent in the mesencephalon and spinal cord of deer with CWD than in deer with SE; however, the amount of IHC was similar.
Microscopic features of IHC in neural tissues were course granular deposits, clumps, or plaques of bright red chromogen surrounding neurons, in gray matter neuropil, and in white matter tracts. Several nuclei, such as the DMNV, were heavily stained compared with adjacent nuclei, such as the hypoglossal nucleus (Figs. 5, 6). Positive IHC also was evident in the myelencephalon, diencephalon, and rhinencephalon. Numerous plaques often were found in the radix of the DMNV (Fig. 7). A few plaques were found in the most proximal branches of the vagus nerve as it leaves the brain stem just before it exits through the jugular foramen. Plaques were commonly found in axonal tracts in the diencephalon and brain stem. Positive IHC was observed in all areas of spongiform degeneration, but in many areas where IHC was present, spongiform degeneration was not necessarily found, especially in the cerebrum.
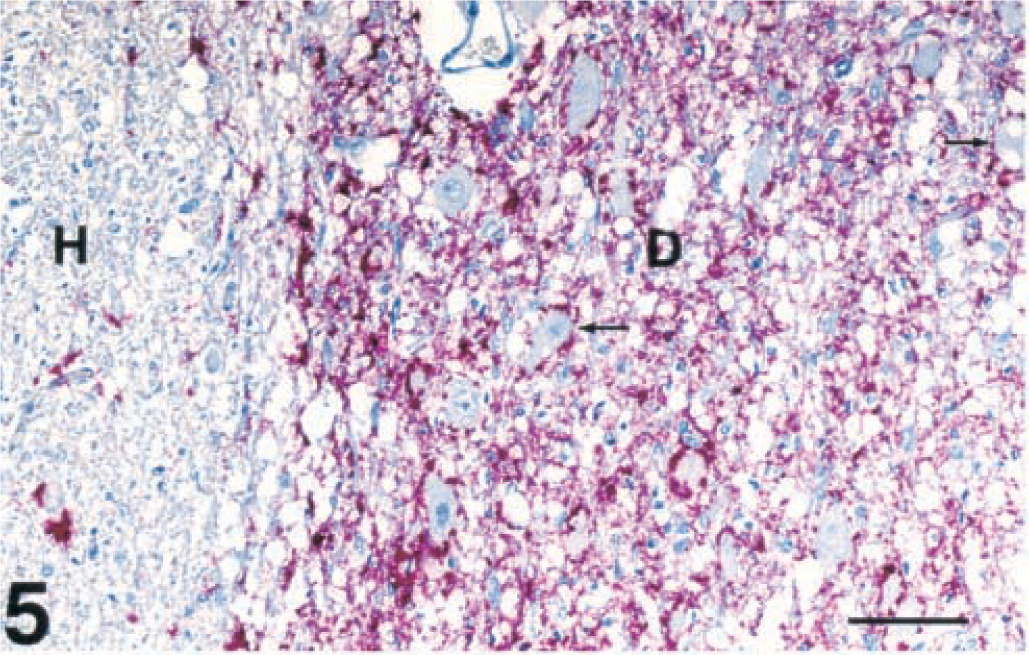
Brain, dorsal motor nucleus of vagus and hypoglossal nucleus; captive deer with chronic wasting disease. Extensive positive immunohistochemical staining of PrPres is present in the dorsal motor nucleus of the vagus nerve (D) as compared with minimal staining in the hypoglossal nucleus (H). Also notice the vacuolated neurons in the vagal nucleus (small, thin arrows). Immunohistochemical staining: streptavidin-alkaline phosphatase method. Hematoxylin/bluing counterstain. Bar = 95 µm.
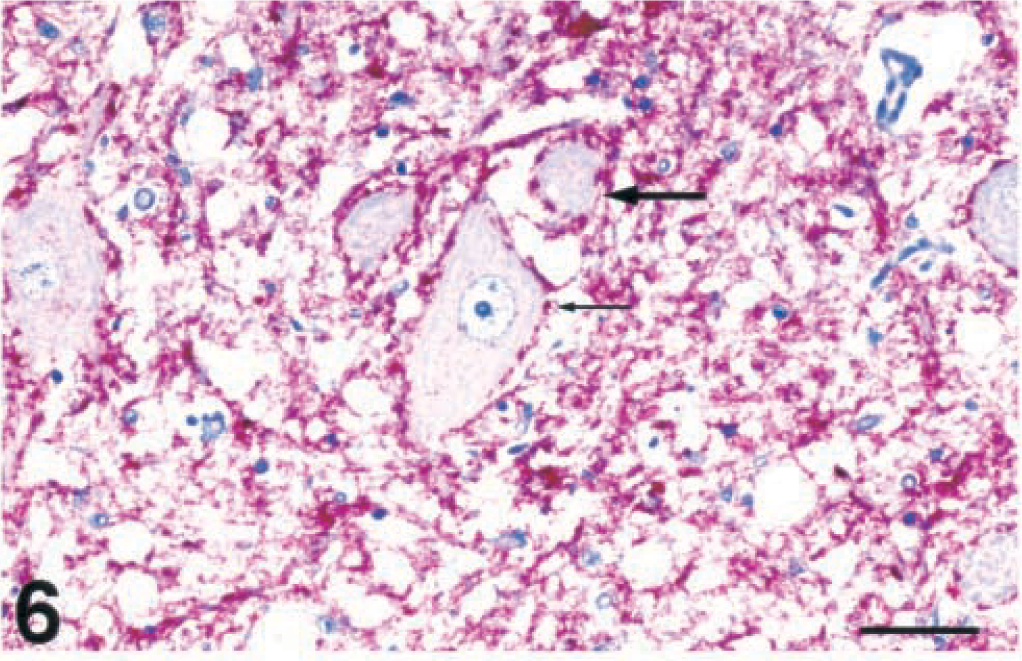
Brain, dorsal motor nucleus of vagus nerve; free-ranging deer with spongiform encephalopathy. Extensive bright red immunopositive staining of PrPres in the neuropil of the nucleus. Note the red chromogen outlining the neurons (small, thin arrow) and a neuron with a small single intracytoplasmic vacuole (large arrow). Immunohistochemical staining: streptavidin-alkaline phosphatase method. Hematoxylin/bluing counterstain. Bar = 42 µm.
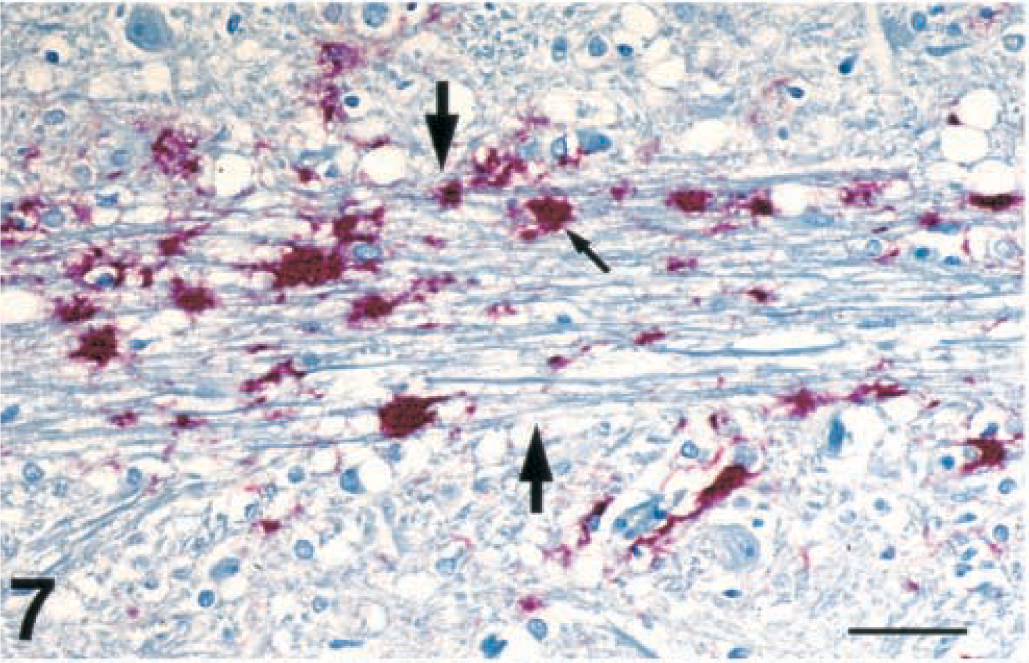
Brain, dorsal motor nucleus of vagus nerve; free-ranging deer with spongiform encephalopathy. Plaques of positive immunohistochemical staining of PrPres (small arrow) in the radix from the vagal nucleus (outlined by the two large arrows). Immunohistochemical staining: streptavidin-alkaline phosphatase method. Hematoxylin/bluing counterstain. Bar = 67 µm.
Immunopositive material was commonly found in or on the basement membrane and on the outer surface or under the apical cytoplasm of ependymal epithelium, especially of the third and fourth ventricles (Fig. 8). Positive IHC was found on the periphery of Virchow-Robins spaces of small- to medium-sized vessels throughout the brain stem of deer with SE or CWD.
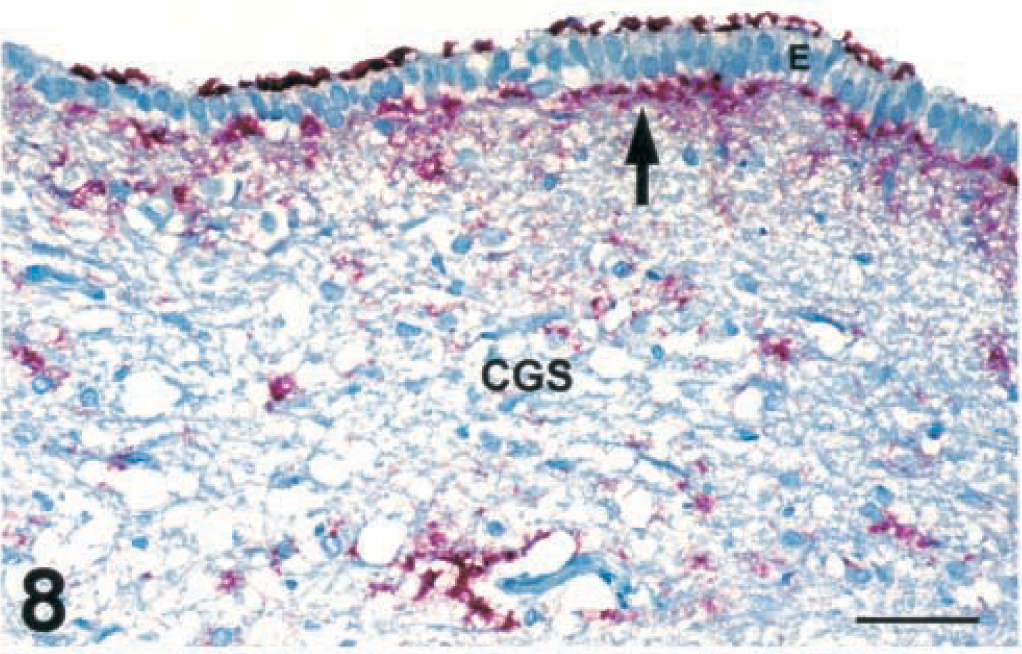
Brain, ependymal epithelium of the aqueduct and central gray substance of the mesencephalon at the level of the rostral colliculus; free-ranging deer with spongiform encephalopathy. Immunohistochemical staining of PrPres is present on the surface and under the basement membrane (arrow) of the ependyma (E). Immunohistochemical staining can also be seen in the neuropil of central gray substance (CGS). Immunohistochemical staining: streptavidin-alkaline phosphatase method. Hematoxylin/bluing counterstain. Bar = 67 µm.
Histologic lesions and IHC were not found in the peripheral nervous system, including the ganglia (dorsal root g, anterior mesenteric g, and trigeminal g) and peripheral nerves (brachial plexus, vagus, sciatic, and sympathetic trunk).
Lymphoid system
Lymphoid tissues of deer with SE or CWD had mild to moderate depletion of follicular lymphocytes. Positive IHC was characterized by course bright red granular chromogen filling germinal centers. The pattern of staining in these follicles would suggest that PrPres could possibly be associated with the cell surface of follicular dendritic cells, as observed in mice. 14 Positive lymphoid tissues were characterized by having 50–95% of the follicles immunostained (Figs. 9, 10). Lymphoid follicles of the tonsil, visceral and peripheral lymph nodes, and Peyer's patches of deer with either SE or CWD had strong positive IHC (Table 2). Bone marrow and thymus were negative in both groups of deer; however, thymus was not found in captive deer.
Comparison of immunohistochemical staining of PrPres in lymphoid tissues of mule deer with a naturally occurring spongiform encephalopathy with that of similar tissues in captive mule deer with chronic wasting disease.

∗ Number of times a specific lymphoid tissue was positive for PrPres with immunohistochemical staining in germinal centers.
† Number of times a specific lymphoid tissue was examined.
‡ Percentage of positive lymph tissues.
§ NF = not found in captive deer.
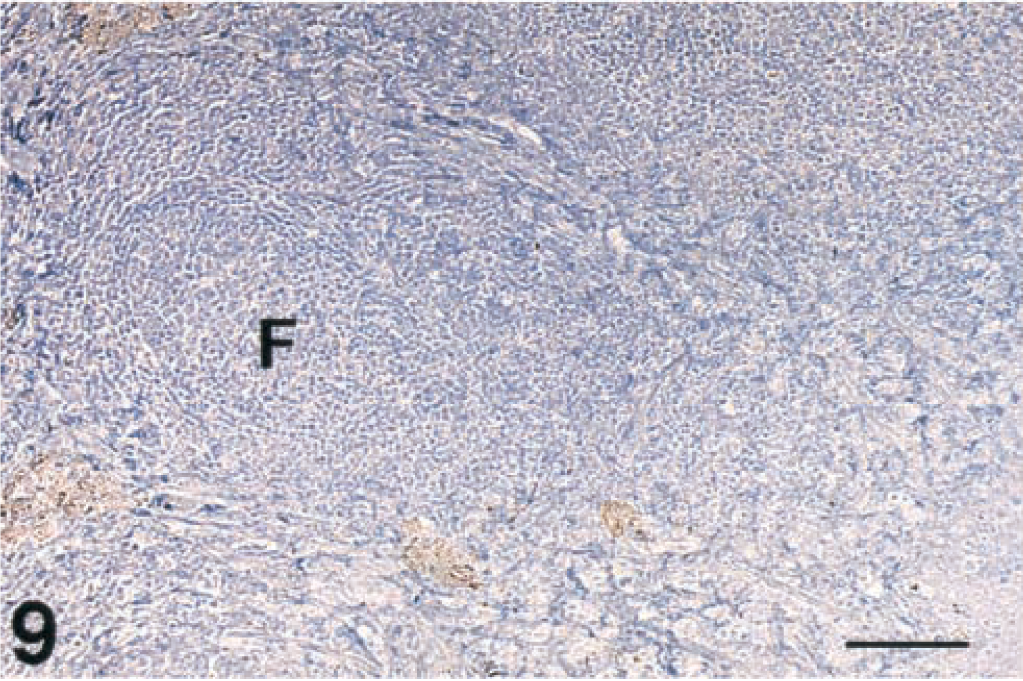
Tonsil, lymphoid follicles; free-ranging deer that did not have spongiform encephalopathy. Immunohistochemical staining of PrPres is not present in the follicle (F). Immunohistochemical staining: streptavidin-alkaline phosphatase method. Hematoxylin/bluing counterstain. Bar = 133 µm.
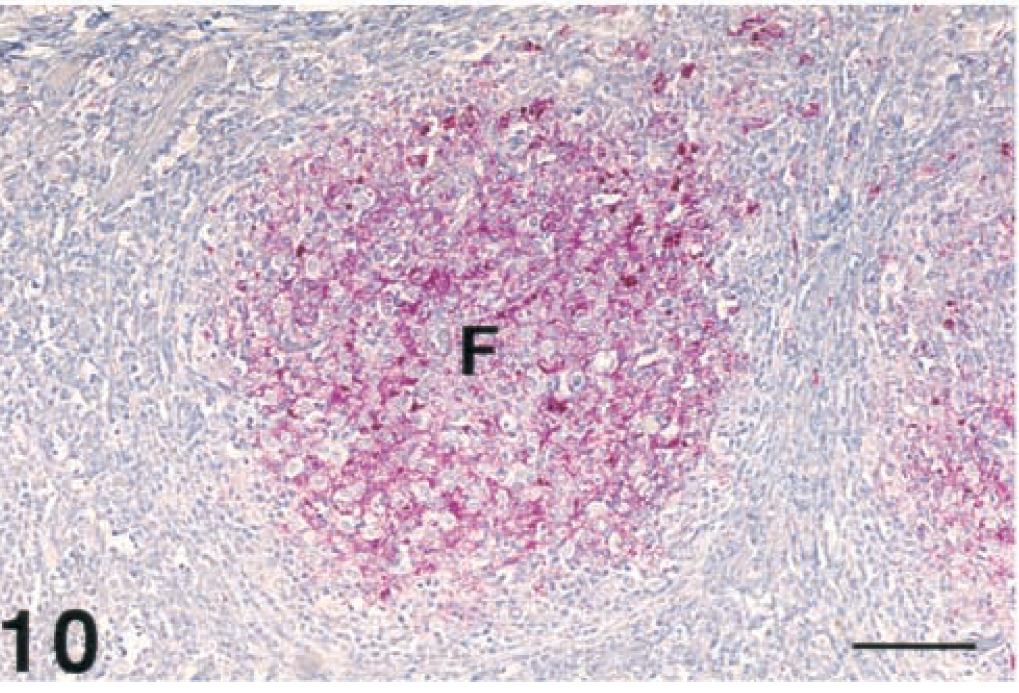
Tonsil, lymphoid follicles; free-ranging deer with spongiform encephalopathy. Strong immunopositive staining of PrPres in the lymphoid follicle (F). Immunohistochemical staining: streptavidin-alkaline phosphatase method. Hematoxylin/bluing counterstain. Bar = 133 µm.
Digestive System
Histologic lesions and IHC were not found in the parotid and mandibular salivary glands, tongue, esophagus, all nonlymphoid components of the digestive tract (rumen, abomasum, small intestine, and colon), myenteric plexus, pancreas (acinar and islet cells), and liver in any of the deer. Gut-associated lymphoid tissues and mesenteric lymph nodes were strongly positive in deer with SE and CWD.
Respiratory system
Histologic lesions and IHC were not found in the epithelial lining of trachea, bronchi, bronchioles, or alveolar parenchyma. Lymphoid follicles located in the posterior nasal septum were often strongly positive in both groups of deer.
Cardiovascular system
Myocardium, Purkinje fibers, and walls of peripheral arteries and veins were free of lesions and were negative for IHC. PrPres was not found within endothelium of these vessels.
Endocrine system
Histologic lesions and IHC were not found in the thyroid, adrenal, and pars anterior of the pituitary but were found in the pars nervosa of the pituitary in deer with SE and CWD.
Musculoskeletal system
Multiple sections of skeletal and smooth muscle were free of microscopic lesions and were negative for IHC in deer with SE or CWD.
Urogenital system
Histologic lesions or IHC were not found in the kidney, urinary bladder, ovary, uterus, placentomes, testis, and epididymis in both groups.
Integument
Tissue sections of skin, including epidermis, dermis, subcutaneous tissues with sebaceous, sweat, lacrimal, and tarsal glands, did not have any microscopic lesions and were negative for IHC in both groups.
Special senses
Eyes of two free-ranging and one captive deer, the only ones examined, had vacuolated neurons in the ganglion cell layer. Positive IHC was found in the layer of optic nerve fibers, layer of ganglion cells, and inner and outer plexiform layers of the retina.
Fetal tissues
Tissues from major organs (brain, lung, thymus, spleen, heart, liver, intestine, skeletal muscle, kidney, and placentomes) of the one fetus did not have histologic lesions and were negative for IHC.
Discussion
When the neurohistologic lesions of TSEs, such as scrapie of domestic sheep and goats, 8–11,32 transmissible mink encephalopathy (TME), 11 and bovine spongiform encephalopathy (BSE), 25,27–29 are compared with those of SE of free-ranging deer and CWD of captive deer, they are morphologically similar. However, differences can be found in the neuroanatomic distribution and severity of histologic lesions among these diseases and species of animals. 10,25,27–29,32 In a detailed comparative study of scrapie, TME, and CWD, Hadlow concluded that there were obvious similarities, but differences did exist in the severity and topographic distribution of histologic lesions among these diseases. 10 Others also have confirmed that there are differences in various breeds of domestic sheep with naturally occurring scrapie. 32 Hadlow and others have surmised that these differences were due to different strains of the scrapie agent and/or the breed of sheep. 10,32
The significance of these minor differences described in this article between free-ranging deer with SE and captive deer with CWD is not known. These differences may be due to different polymorphisms of the prion protein gene and, therefore, slightly different isoforms of PrPc in neural tissues or different strains of the infectious agent as suspected in domestic sheep. 10,32 It is not believed that these minor differences were due to the stage of disease since the deer were all adults in the terminal stage and were either found dead or moribund.
One intriguing finding in the free-ranging deer was that 15 of 16 had small clusters of spongiform degeneration throughout the basal nuclei, diencephalon, mesencephalon, and metencephalon, whereas one deer had relatively large, well-delineated clusters in similar regions of the brain. The reason for this slight difference in character of clusters of spongiform degeneration is unknown but may be due to a different or an unusual prion protein genotype in this particular deer or perhaps a different wild strain of the infectious agent.
The reason that the DMNV, diencephalon, and olfactory cortex were the most consistently and severely affected regions of the brain with spongiform degeneration and IHC in both SE and CWD is not known. One speculation is that these specific areas of brain may be a reflection of selective vulnerability of specific cell populations in these particular nuclei.
Another distinctive point of similarity between SE and CWD was the distribution of PrPres in lymphoid tissue. The distribution and abundance of IHC were indistinguishable between free-ranging and captive deer. The finding of widespread IHC of lymphoreticular tissues in these deer matches the extraneural patterns of infectivity in sheep and goats with scrapie. 9,11 Hadlow et al. have shown that the mesenteric lymph node was among the first extraneural tissues to become infected in domestic sheep. 9 Sigurdson et al. found PrPres in tonsil and alimentary tract-associated lymphoid tissues in mule deer fawns as early as 42 days after they were inoculated orally with brain tissue from captive deer dying of CWD. 21 The abundance of PrPres in lymph nodes in these infected deer suggests that lymphoid tissues play a role in the pathogenesis and transmission of this disease in deer, as they do in sheep. The lymphoid tissues may provide a possible route of entry into the body, but also an exit. PrPres may exit via the gut-associated lymphoid tissues and leave the body in feces and/or through the tonsils being washed out of the mouth with saliva, causing environmental contamination. Excessive salivation is one of the primary clinical signs in deer with terminal SE and CWD. 23,30 This lymphoreticular staining of PrPres further demonstrates the similarity of SE of free-ranging deer and CWD of captive deer to scrapie of sheep and goats. 8,9,11
Another significant finding was that IHC of numerous systemic tissues did not show any evidence of PrPres deposition in deer with SE or CWD. The inability to find PrPres in peripheral nerves, especially the vagus nerve, was surprising. Perhaps PrPres was in peripheral nerves but in such low concentrations that our staining technique was unable to detect it.
This investigation comparing the characteristics and distribution of histologic lesions and IHC of PrPres in a naturally occurring SE in free-ranging deer with those of captive deer with CWD presents strong evidence that SE is indistinguishable morphologically from CWD in the terminal stages. The histologic lesions described in the original papers on CWD of captive deer were essentially identical with those found in this study. 30,31 Moreover, since the 10 captive deer dying of CWD originated from the same facilities in which CWD was originally described lends more strength to this comparative study. The minor discrepancies found in the mesencephalon and spinal cord between free-ranging and captive deer were not significantly different from the minor differences described in various breeds of domestic sheep affected with scrapie. 10,32 In conclusion, SE of free-ranging deer and CWD of captive deer are considered to be indistinguishable morphologically in the terminal stages. The description of the etiologic agent or agents and origin of these two conditions were not addressed in this study. Additional proof regarding whether they are caused by the same or similar etiologic agents and have a similar origin will require further investigations.
Footnotes
Acknowledgements
We would like to thank Rick Spowart, Steve Steinert, Gene Schoonveld, Bill Omstead, and Howard Speer of the Colorado Division of Wildlife for collection of the free-ranging deer. We would like to thank Sam Hendrix and Christina Sigurdson for helping perform some of the necropsies, Maryanne Tryjan for embedding the numerous blocks, and Yang Sun Shin and Frank Aquino for cutting and staining HE slides. We would like to thank the reviewers for their constructive comments. This project was partially funded by the Colorado State University Veterinary Diagnostic Laboratory, College of Veterinary Medicine, Fort Collins Colorado, and the Colorado Division of Wildlife.
