Abstract
Eleven gastrointestinal neoplasms from 10 aged horses and 1 pony were examined grossly, histologically, immunohistochemically, and (in two cases) ultrastructurally. Clinical signs were associated with two neoplasms, and the other nine tumors were incidental findings at laparotomy or necropsy. The neoplasms were solitary (9/11) or multifocal (2/11), well demarcated, serosal or mural masses of stomach (1), jejunum (1), ileum (3), cecum (5), and/or colon (2). Microscopic examination revealed discrete spindle cells arranged in compact patterns with fascicles and whorls or cribriform pattern with fascicles and rare palisades, often with a myxoid interstitial matrix. Three tumors infiltrated between the muscularis interna and the muscularis externa at the myenteric plexi. All neoplasms were vimentin positive, 3/11 were S-100 positive, 2/11 were muscle actin positive, and no neoplasm was positive for glial fibrillary acid protein, desmin, factor VIII, chromogranin, or neuron-specific enolase. Of the two tumors studied ultrastructurally, one contained an admixture of smooth muscle cells and cells resembling Schwann cells, and the second was populated by homogeneous fusiform mesenchymal cells separated by homogeneous matrix. Gastrointestinal stromal tumors (GIST) have been recognized in humans, more recently in dogs and nonhuman primates, and now in equids. Most of these tumors are comprised of a loosely arranged network of spindled cells separated by myxoid matrix. GIST may be composed of myogenic, neurogenic, combined myogenic and neurogenic, and undifferentiated mesenchymal cells.
Keywords
Spindle cell tumors of the gastrointestinal tract, typically arising as discrete, exophytic, serosal nodules, have traditionally been diagnosed as leiomyomas or, where appropriate, as leiomyosarcomas. Such tumors are well known in humans and also in aged dogs. 14 In the last 20 years, more discriminating study of human disease has revealed a wider spectrum of these tumors than was previously recognized. Some of these spindle-cell tumors are now believed to be neural, perhaps derived from the enteric plexi, and the designations gastrointestinal autonomic nerve tumor and plexosarcoma have been introduced. 10,11 Further ultrastructural and immunohistochemical studies have revealed that some of these neoplasms contain undifferentiated mesenchymal cells at times mixed with mature myogenic, myofibroblastic, or neural components. As this wider spectrum has emerged, the term gastrointestinal stromal tumors (GIST) has been applied with increasing frequency to this group of differentiated and undifferentiated tumors in humans. 1,13,19,22,25,37 This term has recently been applied to gastric tumors in two nonhuman primates, which had primitive mesenchymal and minor Schwann cell components, expressed S-100 protein, and had metastasized, 2 and to some canine alimentary tract tumors. 18 In this report, we describe 11 GIST in equids.
Materials and Methods
Animals
Specimens were collected from 10 horses (five Thoroughbred, one Appaloosa, and four grade animals) and one pony (Table 1). Six horses and the pony were examined in the USA; the remaining samples were collected in Ireland (three) and Italy (one). The animals were mature adults with an age range (where known) of 13 to more than 20 years. Seven were mares, and four were geldings. With two exceptions, all of the tumors were incidental findings during exploratory laparotomy for disorders such as colic or during postmortem examination following euthanasia because of illnesses unrelated to the gastrointestinal tract or because of poor performance. Two horses had tumors associated with clinical evidence of gastrointestinal disease. In animal No. 8, laparotomy was performed after colic of a few hours of duration; cecal impaction was found and relieved. During surgery, multiple ileal masses were observed, and the terminal 1.5 m of small intestine was resected. Animal No. 11 presented with acute, severe colic and, for economic reasons, the owner elected euthanasia.
Signalment and origin of equids with gastrointestinal stromal tumors (GIST) and anatomical location of GIST.
∗ TB = thoroughbred; AP = appaloosa.
† MC = male, castrated; F = female.
Histopathology and immunohistochemistry
For histologic examination, samples were preserved in 10% buffered formalin, processed routinely, and stained with hematoxylin and eosin (HE). For immunohistochemical evaluation, indirect immunoperoxidase staining was performed using a commercial streptavidin/biotin kit (Zymed, South San Francisco, Calif) and The MicroProbe System (Fisher Scientific, Brightwaters, NY). Four-micrometer sections of formalin-fixed, paraffin-embedded tissue were deparaffinized in xylene, rehydrated, and incubated with H2O2 in methanol for 10 minutes to block endogenous peroxidase activity. The sections were rinsed in 0.01 M phosphate-buffered saline (PBS), pH 7.2, containing 0.4% polyoxyethylene lauryl ether (Brij, 30% concentration in water; Sigma, St Louis, Mo) and incubated for 10 minutes with 10% normal goat serum. The normal goat serum was tapped off and the sections were incubated with primary antibodies against vimentin (monoclonal, dilution 1:40; Dako, Carpinteria, Calif), S-100 protein (polyclonal, 1:200; Dako) desmin (monoclonal, 1:25) glial fibrillary acidic protein (GFAP, Dako; polyclonal, 1:3,000; Dako), factor VIII (polyclonal, 1:1,000; Dako), smooth muscle actin (polyclonal, 1:20; Enzo Diagnostics, Farmingdale, NY), chromogranin (polyclonal, 1:1,000 to 1:2,000; Enzo), and neuron-specific enolase (NSE, monoclonal, 1:300; Dako) diluted in Tris-buffered saline (Table 2) for 2 hours at 37 C. For the von Willebrand factor antibody, the sections were pretreated by trypsinization. The sections were rinsed with PBS/Brij and incubated with biotinylated secondary antibody at room temperature for 10 minutes and rinsed again. The sections were incubated with streptavidin–peroxidase conjugate (ABC immunoperoxidase method) for 10 minutes at room temperature and then given a final rinse in PBS/Brij and reacted with the chromogen/substrate, 0.05% 3,3 diaminobenzidine tetrahydrochloride (Sigma), and 0.01% H2O2 for 5–10 minutes at room temperature. The reaction was stopped by rinsing in distilled water. The sections were counterstained with Gill's no. 2 hematoxylin (Fisher Scientific) for 1 minute and then dehydrated in graduated ethyl alcohol, placed in xylene, and coverslipped in Permount (Fisher Scientific). All incubations were carried out in a humid chamber. For the negative controls, nonimmune serum at the same protein dilution as the primary antibody was used. Equine tissue sections known to express the relevant antigen served as positive controls.
Immunohistochemistry ∗ of gastrointestinal stromal tumors in equids.

∗ S-100 = S-100 protein; SMA = smooth muscle actin; FVIII = factor VIII-associated antigen; GFAP = glial fibrillary acidic protein; NSE = neuron-specific enolase. NT = non-tested. Staining intensity: mild (+), moderate (++), marked (+++).
Electron microscopy
Tissue previously fixed in 10% formalin buffered solution was immersed in 0.1 M sodium cacodylate for 2 days, rinsed in cacodylate buffer, postfixed in 1% Dalton fixative for 2 hours, and then prepared for embedment in epon–araldite. Blocks were thick sectioned at 1 µm and stained with toluidine blue and basic fuchsin. From these slides, areas of the blocks were identified for thin sections. The thin sections were mounted on copper grids, stained with uranyl acetate and lead citrate, and examined in a Philips 201 electron microscope.
Results
Gross findings
In this group of equids, gastrointestinal stromal tumors occurred in various locations (Table 1, Figs. 1, 2). The size of these tumors ranged from a few millimeters up to 13 cm, and they were roundish, oval, cylindrical, or pyriform. Tumors from animal Nos. 2 and 7 were in the stomach near the pilorus and midjejunum, respectively, and tumors of animal Nos. 8, 10, and 11 were identified in the ileum. Neoplasms of animal Nos. 1, 4–6, and 9 were on the cecum, and neoplasms of animal Nos. 3 and 9 were observed on the large colon. All tumors were subserosal except for that in animal No. 2, which was located within the stomach muscular wall, and that in animal No. 8, which was multiple with several extramural subserosal and intramural masses (Fig. 2). The neoplastic growth in animal No. 9 was also multifocal, but the masses were subserosal not intramural. The neoplasm of animal No. 7 was attached to the ileum by a vascular peduncle and resembled a pedunculated lipoma. The majority of these tumors were moderately soft and had a red-and-white marbled appearance on the cut surface, with the exception of tumors in animal Nos. 2, 7, and 8, which were rather firm and whitish on cut surface. The cut surface of the tumor in animal No. 1 was lobulated and bulging, with a delicate stromal network separating lobules of gelatinous gray to red tissue. The neoplasm from animal No. 7 was the only one containing large yellowish areas of necrosis surrounded by reddish halos, like some necrotic pedunculated lipomas. The neoplasm of animal No. 11 was comprised of two oval, lateral, egg-shaped masses and 1 central, round mass. This trilobed neoplasm arose from the ileum 30 cm from the cecum. The ileum was wrapped around the lobe of the right mass and a segment of the ileum formed a loop entrapped between the mass and the distally wrapped segment of ileum. Ileal strangulation was characterized by severe hemorrhagic necrosis of 1 m of terminal ileum with secondary small intestine and gastric dilatation and moderate abdominal effusion.
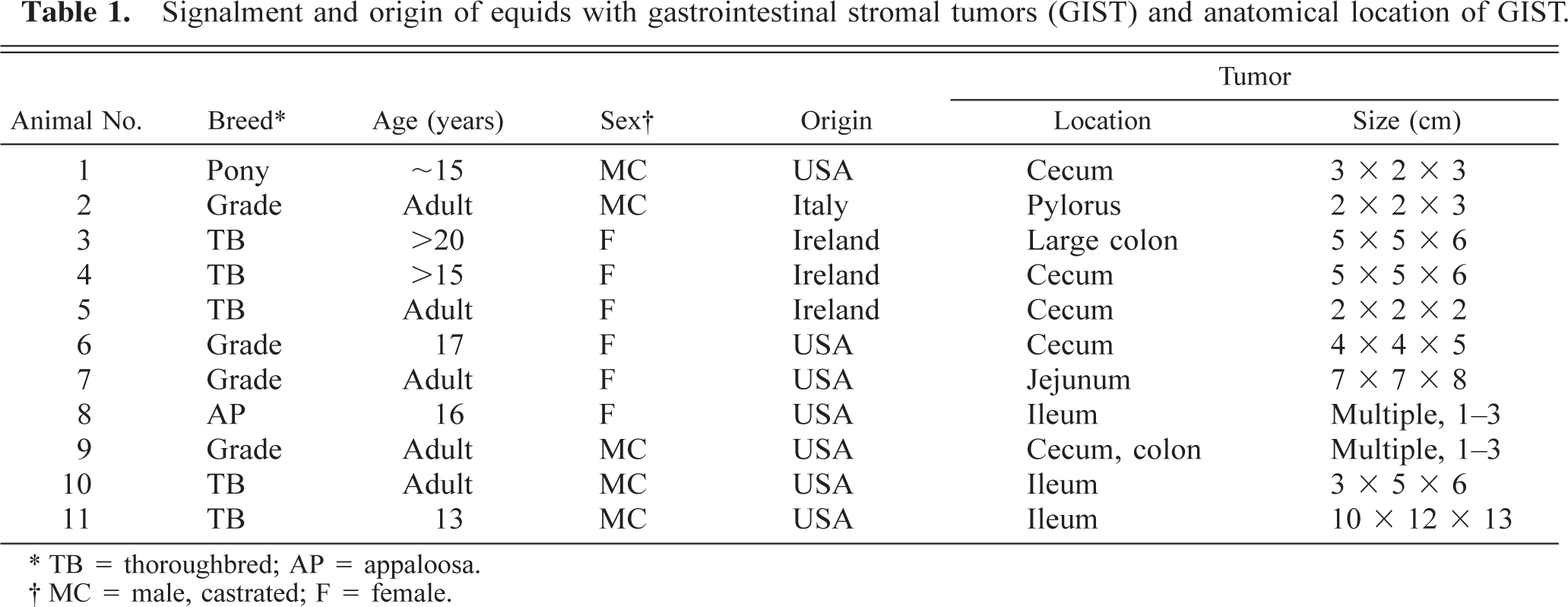
Cecum, GIST; pony No. 1. Encapsulated sessile mass with a delicate stromal network separating lobules of gelatinous tissue. Bar = 1 cm.
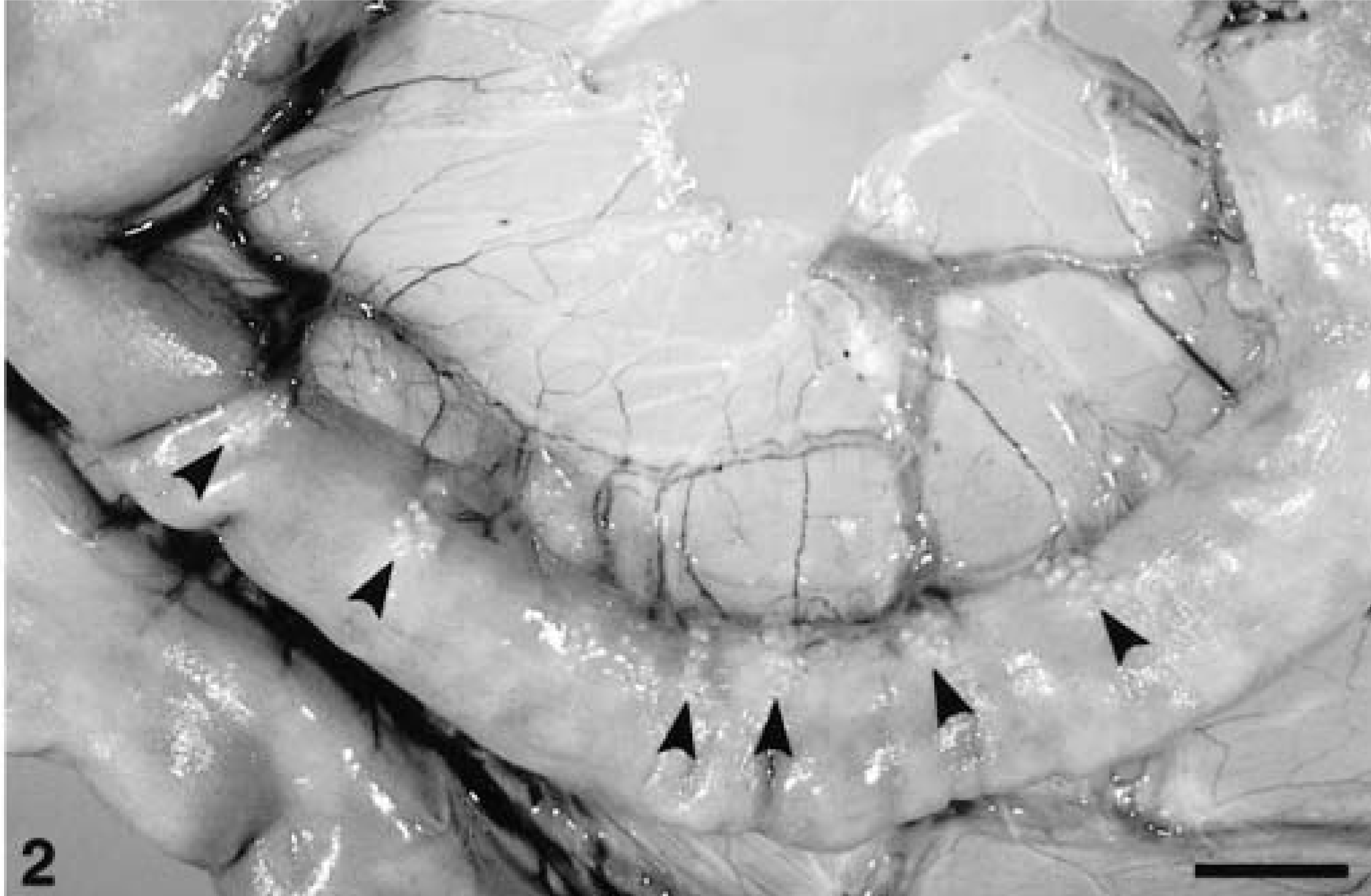
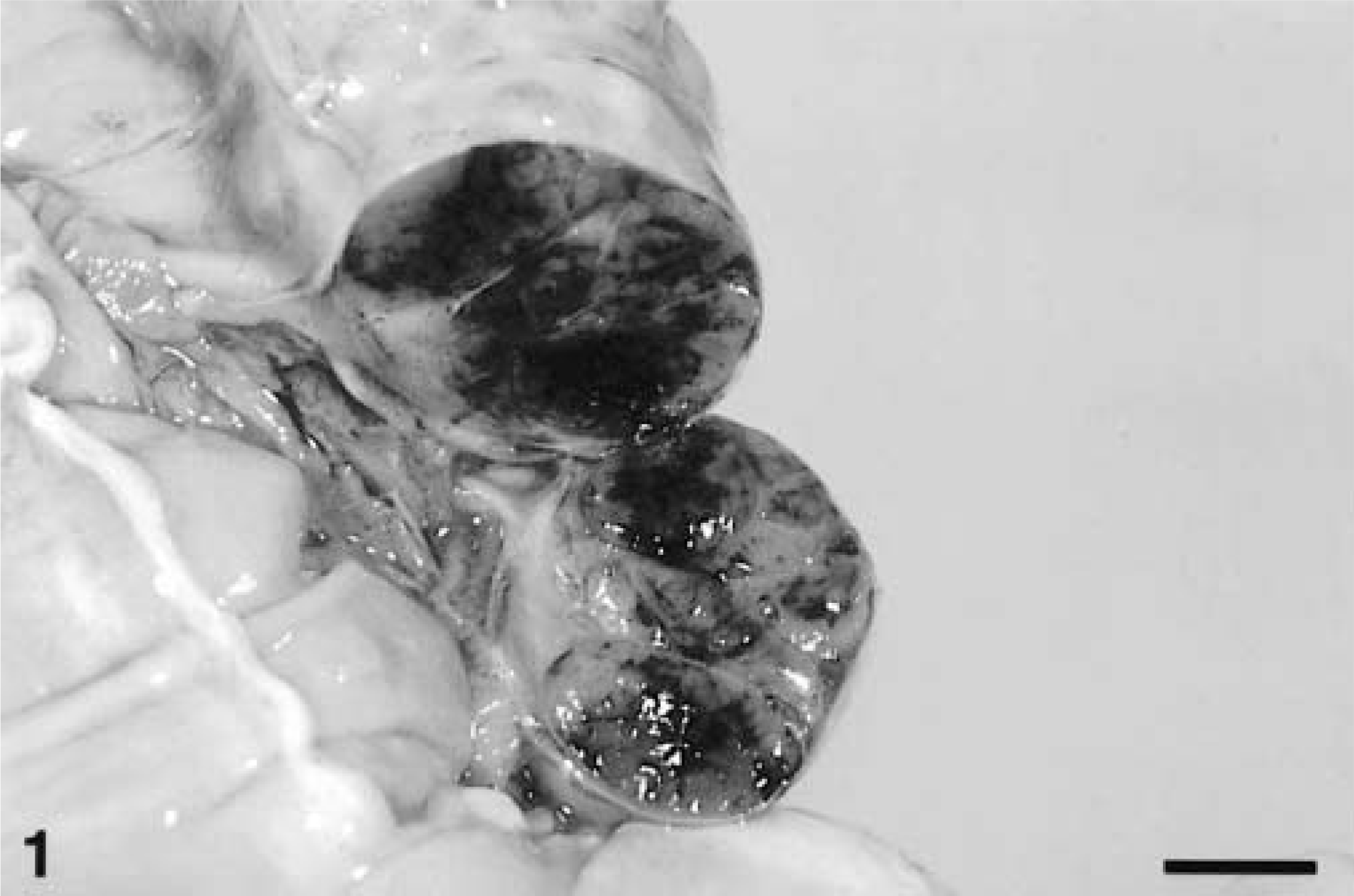
Fig. 2 Ileum, GIST; horse No. 8. Multiple nodular masses (arrowheads) arranged circumferentially. Bar = 2.5 cm.
Histology
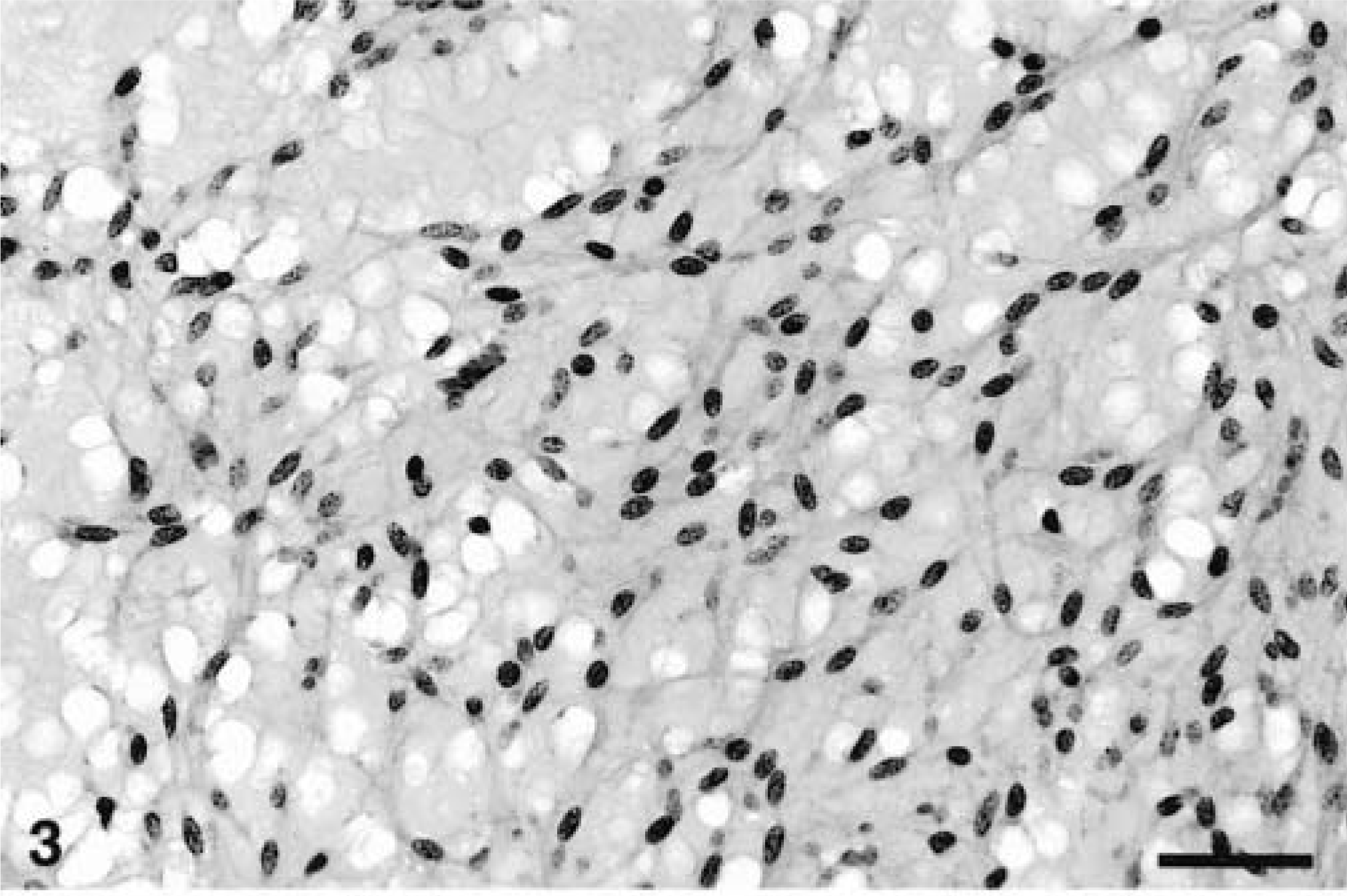
All the neoplasms were well demarcated with collagenous capsules (except for the tumor in animal No. 8) and were populated by closely packed to loosely arranged spindle cells with oval nuclei and slender, elongated, bipolar cytoplasmic processes. The nuclei were euchromatic with blunted or slightly tapering ends. The patterns of spindle cell growth were quite variable. Compact growth produced intersecting fascicles (Fig. 3) or whorled (Fig. 4) arrangements. In areas with a prominent cribriform pattern of growth, fascicles formed by just two or three spindloid cells separated pools of mucin (Fig. 5). This pattern of growth was the most common one in these 11 tumors; the whorling pattern was the least common overall. Palisading cells were also occasionally observed (Fig. 4). In areas of compact tumor growth (Figs. 6, 7), connective tissue stroma was minimal, whereas in other areas it was more abundant. In the neoplasms of animal Nos. 7 and 8 (Fig. 7), tumor cells were tightly packed and sometimes resembled smooth muscle; they were very slim and elongated, with elongated nuclei and eosinophilic cytoplasm. Numerous hemosiderophages were scattered at the periphery of all masses (except for that of animal No. 8). A moderate multifocal infiltration of lymphocytes, plasma cells, macrophages, and occasional neutrophils was also present. Two neoplasms had hyalinosis of the walls of some small blood vessels. In the neoplasm of animal No. 6, loosely arranged spindled cells radiated from and merged directly with the pericyte area of some blood vessels. Mitotic figures were infrequent in all tumors.
Cecum, GIST; horse No. 5. Loose network of spindle and stellate cells with elongated vescicular nuclei. This is the most common histologic appearance of equine GIST. Bar = 60 µm.
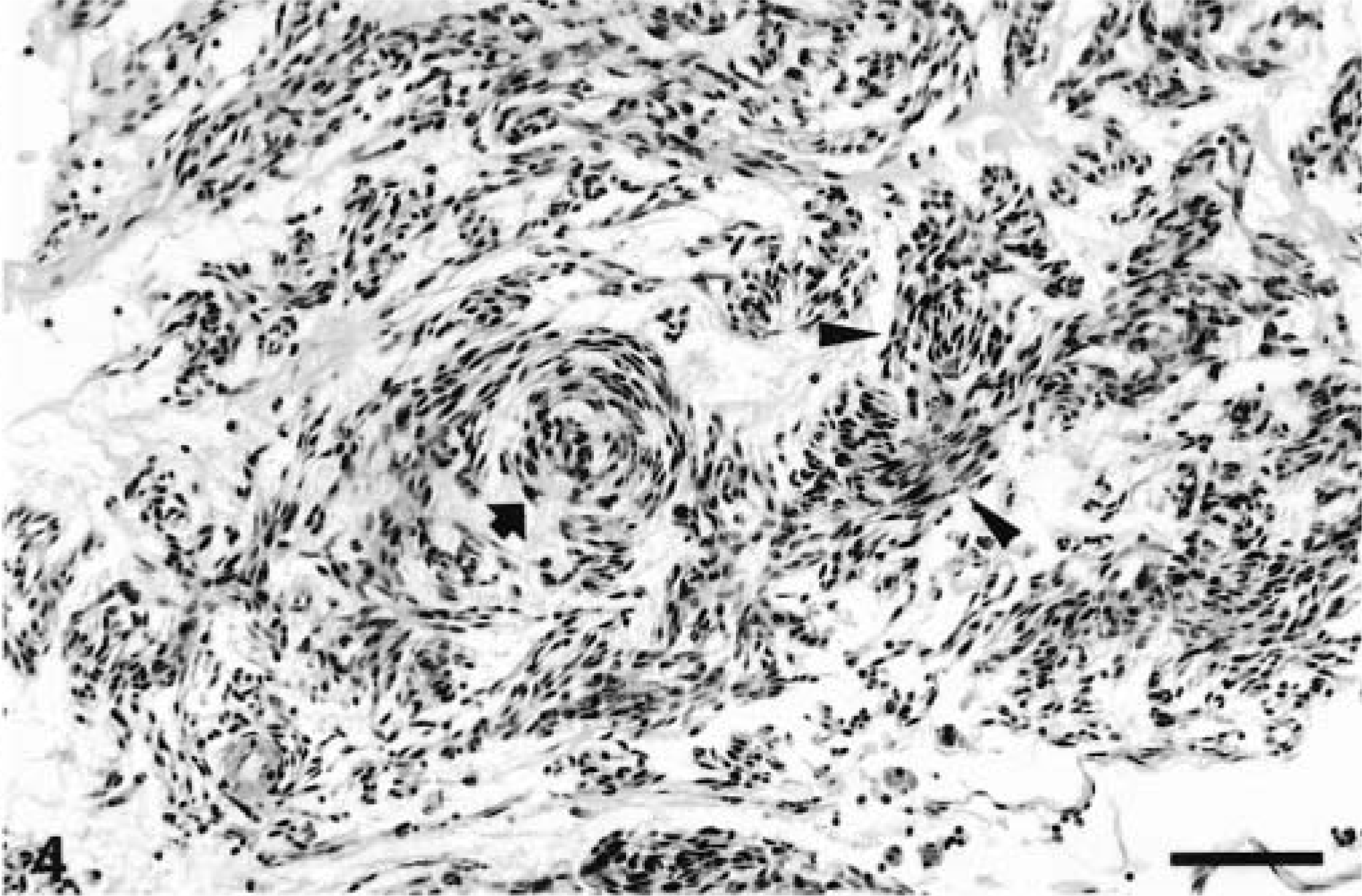
Stomach, GIST; horse No. 2. Loosely arranged spindle cell growth with occasional whorling (arrow) and palisading (arrowheads). Bar = 190 µm.
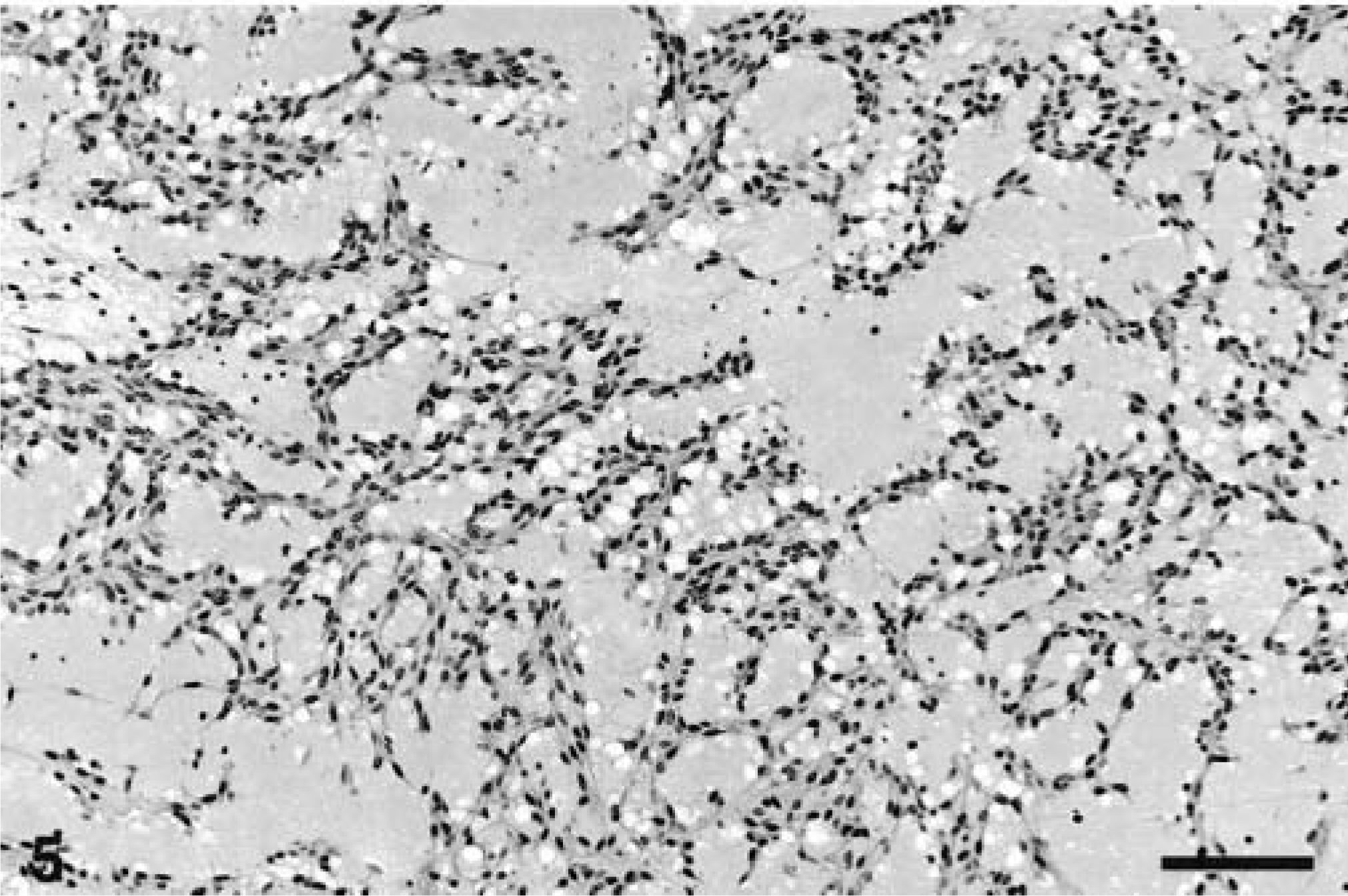
Cecum, GIST; horse No. 4. Network of spindle cells with cribriform pattern due to pools of myxoid material. Bar
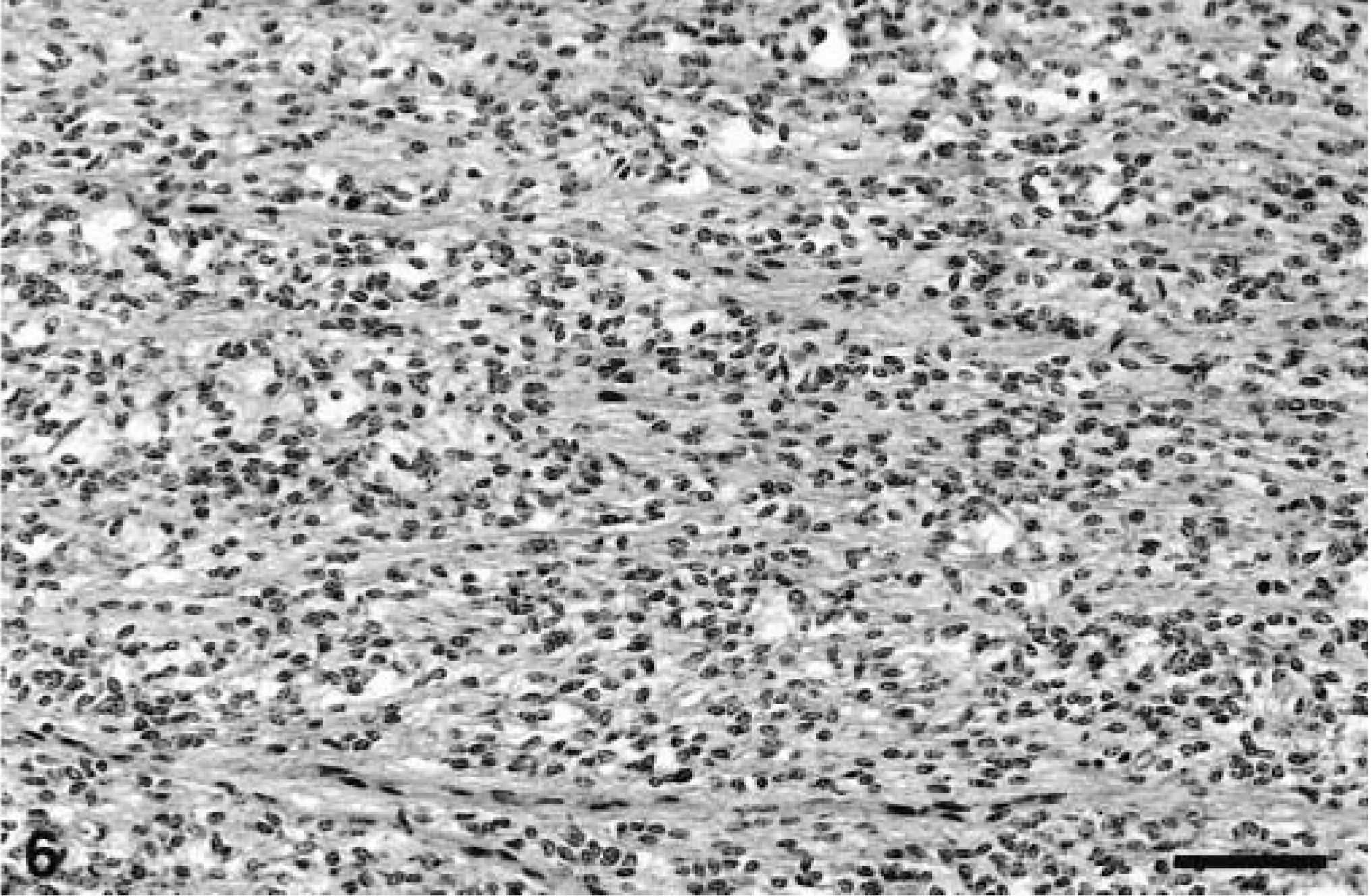
Jejunum, GIST; horse No. 7. Area of compact, organized neoplastic spindle cell growth. Bar = 190 µm.
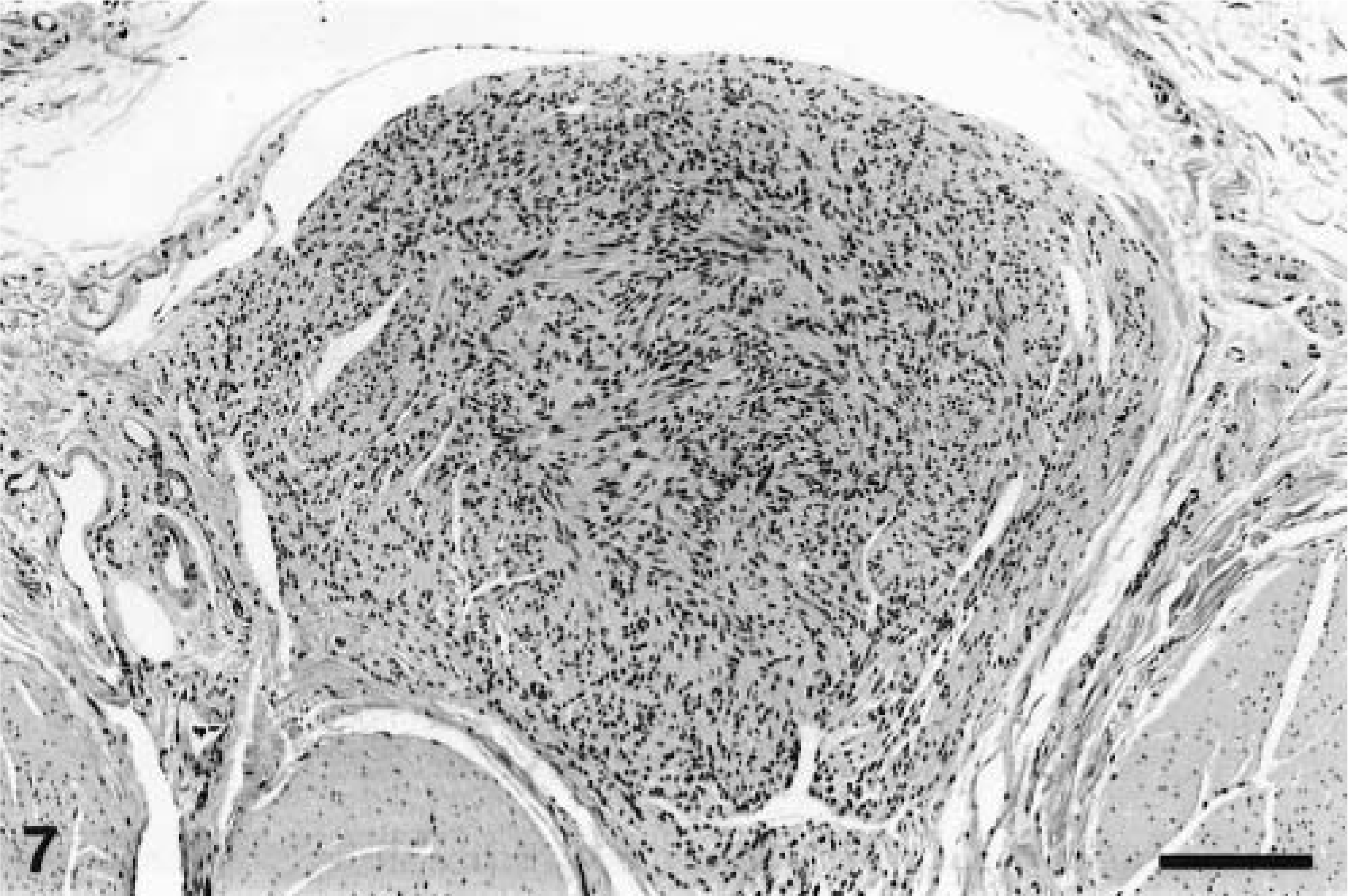
Ileum, GIST; horse No. 8. Well-demarcated pyriform subserosal and inframuscular mass composed of spindle cells with elongated nuclei, resembling smooth muscle. Bar = 190 µm.
Immunohistochemistry
The results are summarized in Table 2. In all tumors with positive staining, antigens were detected in the cytoplasm of neoplastic cells. Staining intensity ranged from mild to marked. Normal elements in the tissue served as internal controls. All neoplasms stained for vimentin; staining was multifocal to diffuse with patchy areas of weak and prominent staining. S-100 was moderately to markedly expressed in neoplasms of animal Nos. 2 and 8 (Fig. 8); in the neoplasm of animal No. 5, S-100 staining was mild and focal. Smooth muscle actin staining was moderately to markedly expressed in the neoplasm of animal No. 8 (Fig. 9) and mild and focal in the tumors of animal Nos. 2 and 5. All neoplasms were negative for factor VIII, GFAP, chromogranin, desmin, and NSE.
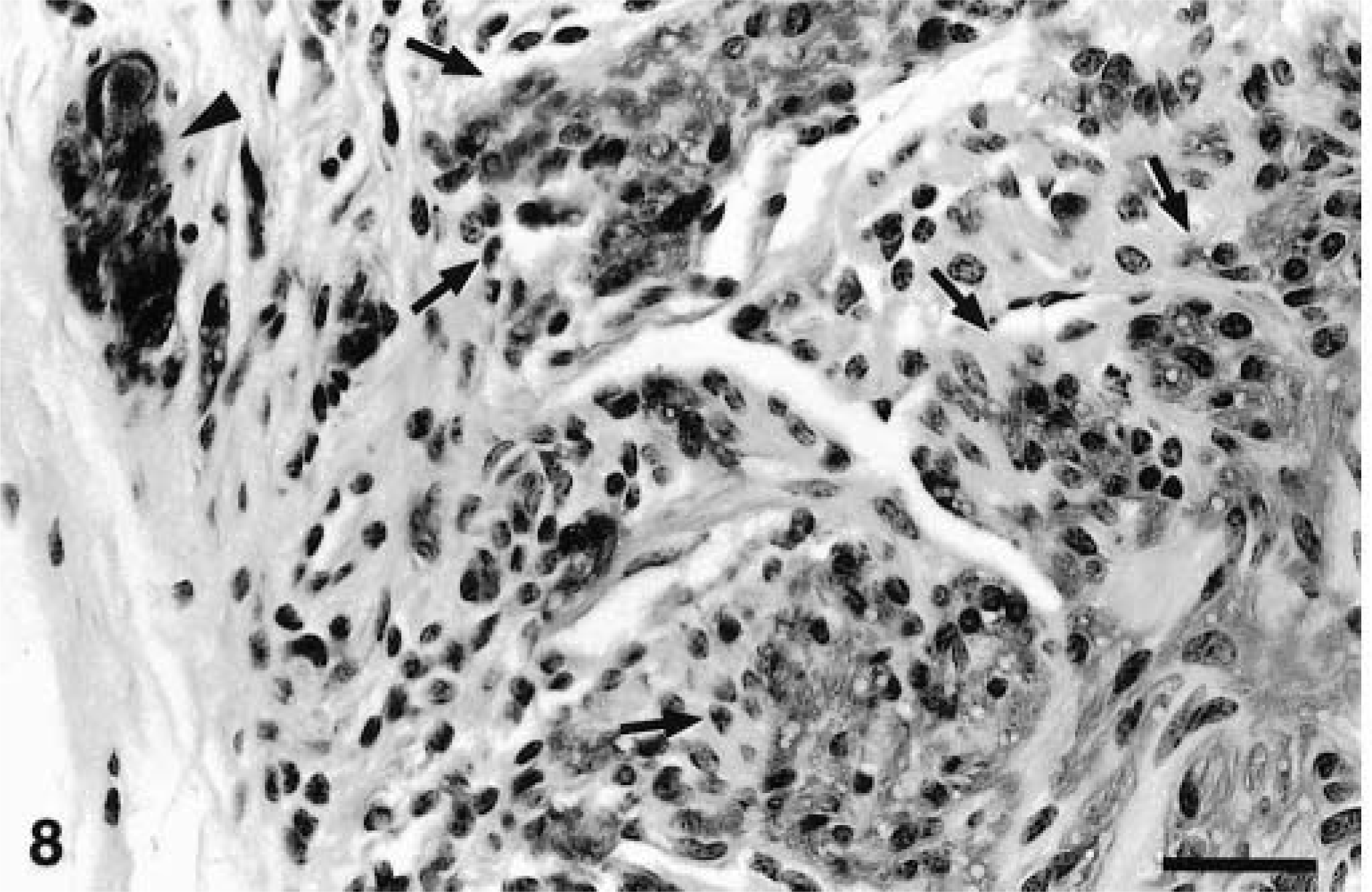
Ileum, GIST; horse No. 8. Numerous neoplastic spindle cells (positive areas demarcated by arrows) and nerve (arrowhead) express S-100 protein. ABC immunoperoxidase method, hematoxylin counterstain. Bar = 80 µm.
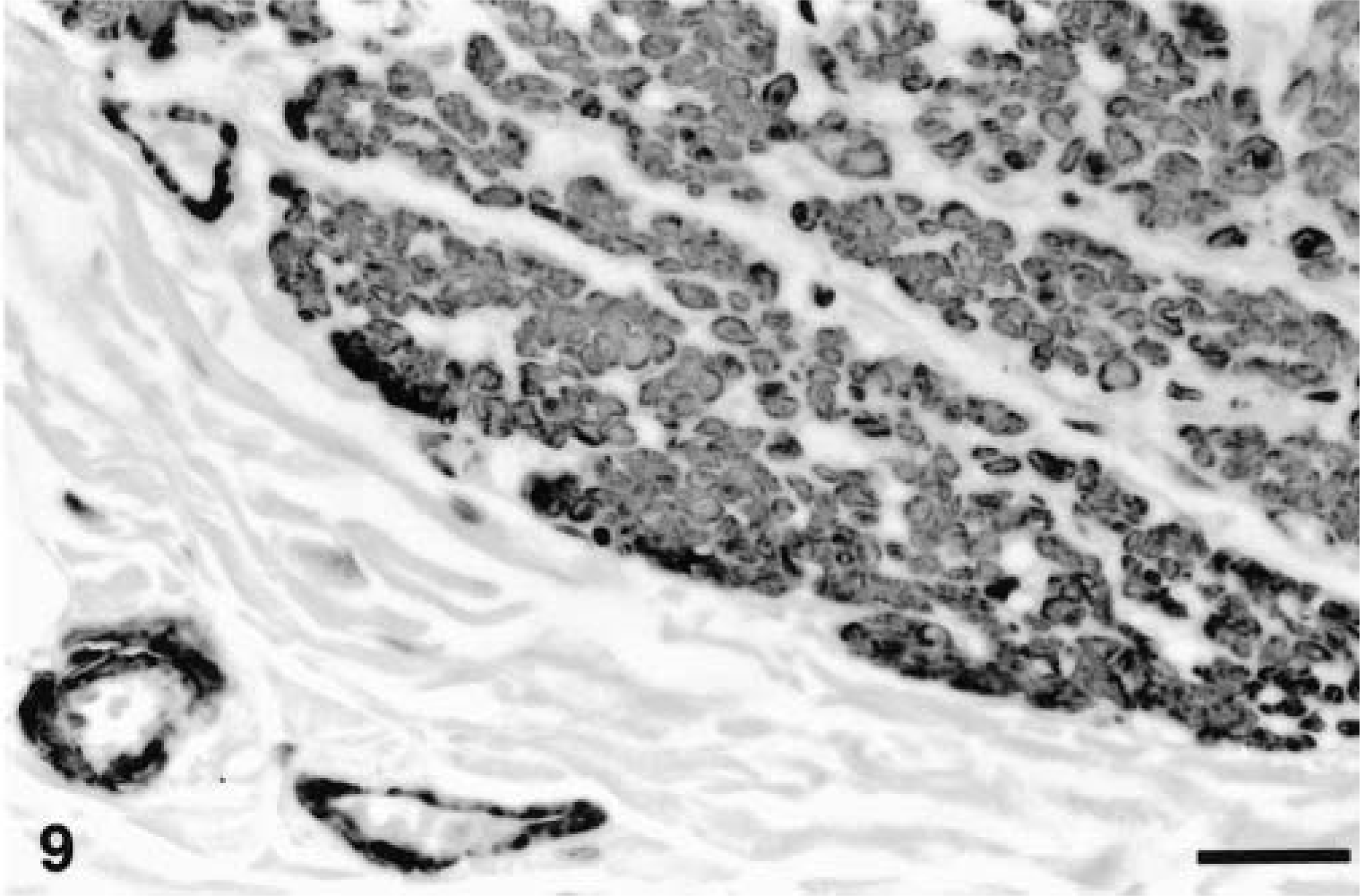
Ileum, GIST; horse No. 8. Numerous neoplastic cells (top right and center) and the tunica muscularis of blood vessels (left below) express smooth muscle actin. ABC immunoperoxidase method, hematoxylin counterstain. Bar = 120 µm.
Ultrastructure
Ultrastructural studies were performed on the neoplasm of animal No. 7, which had a histologic pattern representing the majority of the tumors examined in this study, and on the neoplasm of animal No. 8, which had unique features.
The neoplasm of animal No. 7 (not shown) was composed of a homogeneous population of loosely arranged spindle and stellate cells embedded in abundant fibrillogranular intercellular matrix. The matrix was interrupted by empty spaces (fluid), which contained particles (proteoglycans). Most neoplastic cells had fairly regular nuclear outlines, and indentations were uncommon. Nucleoli were usually solitary and small. These neoplastic cells had moderate numbers of mitochondria, moderately abundant rough endoplasmic reticulum, occasional cytoplasmic lipid droplets, and scattered focal plasmalemma densities. Frequent focally dilated segments of rough endoplasmic reticulum contained proteinaceous material. There was no evidence of pinocytotic vesicles (caveolae), neurotubules, neurofilaments, arrays of actin microfilaments with dense bodies, or anchoring plaques. A basal lamina was not identified.
The neoplasm of animal No. 8 contained a mixture of cellular elements. In some fields, one cell type predominated but other areas contained mixed populations. Most readily identified cells were spindle shaped (Fig. 10) with fusiform nuclei, sometimes with one or two nucleoli, loosely arranged chromatin, and smooth to undulating nuclear membranes with occasional indentations. Patches of heterochromatin were seen below the nuclear envelope. Ill-defined, diffusely distributed densities (interpreted as intracytoplasmic dense bodies containing alpha-actinin) and dense plaques were uniformly spaced on the inner aspect of cell membranes (interpreted as subplasmalemmal dense plaques composed mainly of vinculin and talin) and paired with similar densities on adjacent cells. Sometimes there was a conical paranuclear condensation of mitochondria, with some glycogen granules and lipid droplets, which were also scattered through the rest of the cytoplasm. There was a somewhat patchy basement membrane. Filaments of varying density ran longitudinally within the cytoplasm, often through the intracytoplasmic dense bodies, connecting with the subplasmalemmal densities. The plasmalemma had various caveolae. The distribution of dense bodies, subplasmalemmal densities, filaments, and caveolae was irregular. Based on this morphology, the cells were identified as smooth muscle cells.
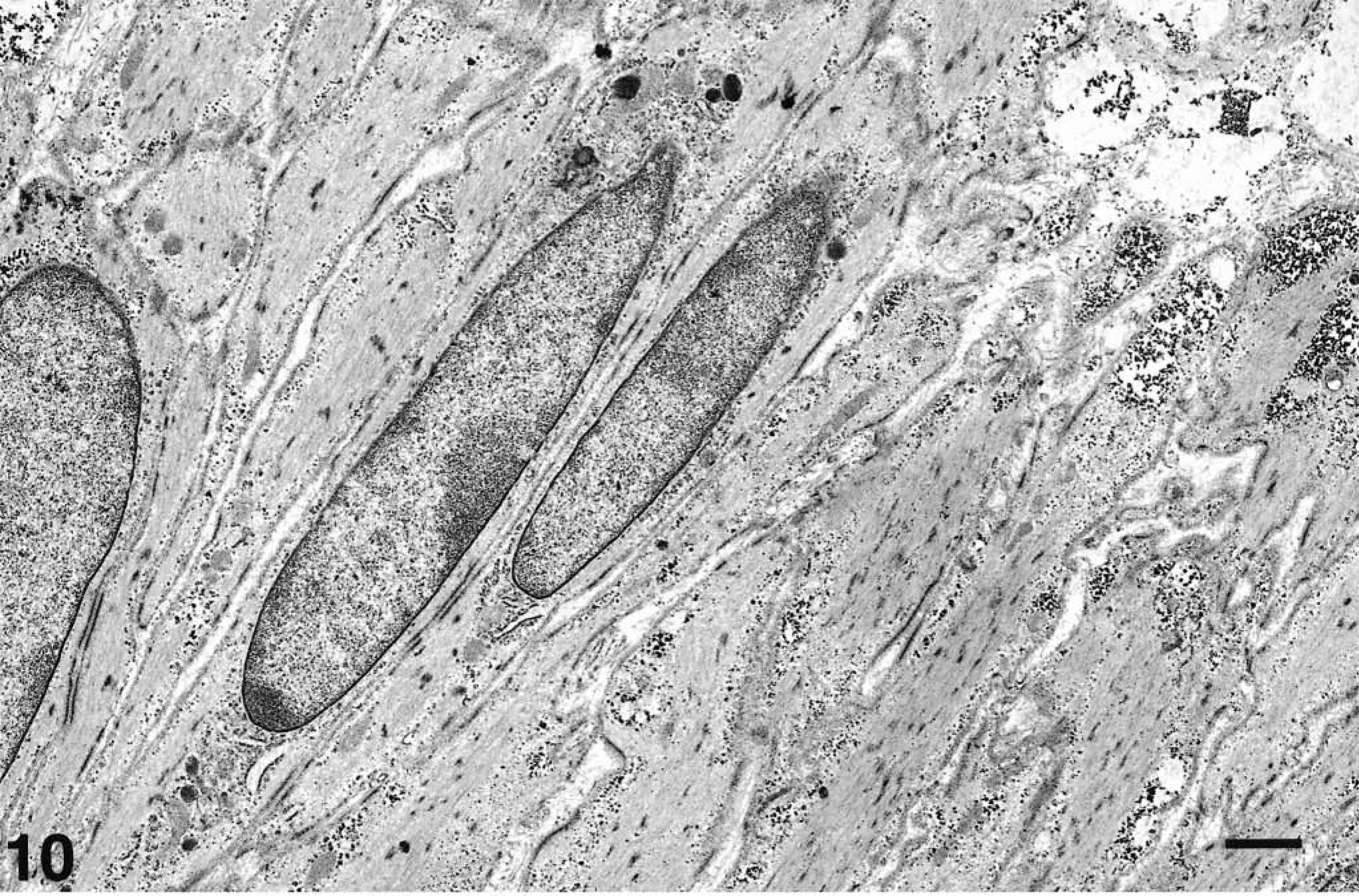
Electron micrograph. Ileum, GIST; horse No. 8. Smooth muscle cells. Spindle cells with elongated nuclei, dense cytoplasmic plaques, and conical paranuclear condensation of organelles (midnucleated myoid cell). Uranyl acetate and lead citrate. Bar
The second cell type within the neoplasm of animal No. 8 (Fig. 11) resembled a Schwann cell. These cells were fusiform to polyhedral with ovoid to slightly irregular nuclei, loosely arranged chromatin, and electrolucent cytoplasm with numerous microfilaments, moderate numbers of mitochondria, and occasional lysosomes. The basement membrane was discontinuous, sometimes with adjacent intervening collagen fibers located between these cells or the smooth muscle cells. These putative Schwann cells extended cytoplasmic processes around small axons, thus appearing as unmyelinated nerves.
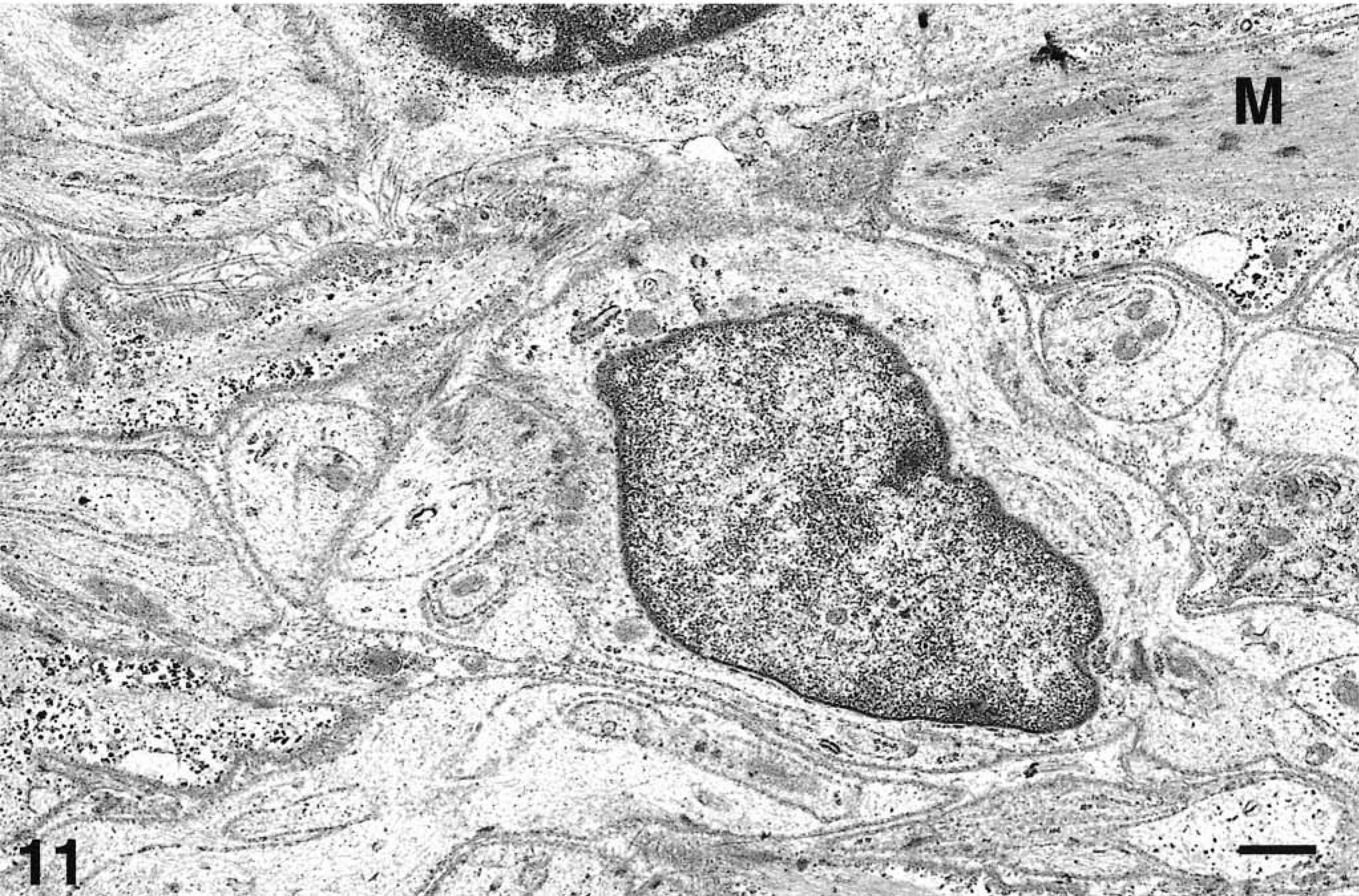
Fig. 11 Electron micrograph. Ileum, GIST; horse No. 8. Electron micrograph. Cells similar to Schwann cells. Polyhedral cell with ovoid to slightly irregular nuclei, loosely arranged chromatin, electrolucent cytoplasm with numerous microfilaments. There are adjacent intervening collagen fibers and the cytoplasm of a smooth muscle cell (M). Uranyl acetate and lead citrate. Bar = 1 µm.
Discussion
Spindle-cell tumors of the gastrointestinal tract, particularly those arising from the serosal surface, have been recognized for decades in humans. Most differentiated tumors have been considered to be leiomyomas or, less frequently, leiomyosarcomas. Such tumors also occur in the aged dog and are often encountered as incidental solitary masses at postmortem examination. 14 In more recent years, evidence in humans that some of these tumors may be derived from the intramuscular and/or submucosal autonomic plexi of the gastroenteric tract has been reported. Such tumors have been designated gastrointestinal autonomic nerve tumors and plexosarcomas. 11,19 Detailed immunohistochemical and ultrastructural studies have demonstrated that some tumors are clearly either myogenic or neurogenic and others consist of less differentiated cells, sometimes of indeterminate lineage. Further evidence of mixed myogenic and neurogenic cells in some tumors has also been presented. 13 For this reason, these human tumors are now frequently reported as GIST, a term that encompasses the entire group 15 or is applied to mesenchymal tumors that represent neither typical leiomyomas nor Schwannomas. 25 Recently, primary gastrointestinal mesenchymal tumors in humans have been immunophenotypically divided into leiomyomas, Schwannomas, and GIST; most GIST may represent tumors of primitive mesenchymal cells in humans. 25
We studied gastrointestinal tumors from 10 horses and 1 pony. Several breeds were represented among equine populations in USA, Ireland, and Italy. Further studies will be required to ascertain whether a breed predisposition to equine GIST exists. In humans, GIST occur with no sex predilection; however, seven of the 11 equine neoplasms in the present study occurred in females. These tumors occurred in mature animals between approximately 13 and more than 20 years of age. Most tumors were incidental findings; in only 2 horses were they associated with gastrointestinal disturbances. The tumor in animal No. 8 was multifocal in the ileum and was associated with cecal impaction; it may have interfered with intestinal motility. The tumor in animal No. 11 caused displacement and strangulation of the distal ileum.
Of a total of 11 neoplasms, nine were solitary, sessile, roundish to cylindrical to multilobular masses arising anywhere from the stomach to the colon, with no striking predilection along the gastrointestinal tract. The remaining two tumors were multifocal. Only one neoplasm was pedunculated. In humans, GIST can have a similar widespread distribution. In addition, they may irregularly infiltrate the gastrointestinal wall, occasionally with postnecrotic cavitations that communicate with the gastrointestinal lumen. 11,19
The microscopic patterns in these equine tumors were varied but typically comprised densely to loosely arranged spindle cells (nine of 11 neoplasms) often separated by myxoid matrix. Similar features are reported in myxoid GIST in humans 35 with glycosaminoglycans rich in hyaluronic acid consistent with connective tissue mucosubstances. Tumors of animal Nos. 2 and 6 had hyalinosis of blood vessels, sometimes a feature of Schwannomas 33,34 and perhaps due to ischemia. 32 Equine GIST, like those of humans, dogs, and nonhuman primates, have various morphologic, histochemical, and ultrastructural patterns. However, some predominant patterns were recognized in this collection of tumors.
All the equine neoplasms stained for vimentin, suggestive of a mesenchymal origin, although Schwann cell tumors may also express vimentin. In two animals (Nos. 2 and 8), there was coexpression of both S-100 and smooth muscle actin.
There were no histochemical, immunohistochemical, or ultrastructural features suggesting the presence of neoplastic neuroendocrine cells.
The neoplasm from animal No. 8 consisted of multiple masses along the mesentery, and in the area of the enteric plexi, these masses were comprised of closely packed spindloid cells expressing either S-100 or muscle actin; ultrastructurally, there were smooth muscle cells and cells similar to Schwann cells. This neoplasm was very similar to an equine small intestinal tumor that was classified as a peripheral nerve sheath tumor by others. 17
Within this spectrum of GIST, some tumors may arise within or in the vicinity of the intestinal plexi (inframuscular or submucosal), as appeared to be the case with the neoplasm of animal No. 8. Such cases in humans, if constituted purely by neural elements, would be designated as gastrointestinal autonomic nerve tumors or plexosarcomas. The term peripheral nerve sheath tumor is classically applied to the tumors of nonautonomic components of the peripheral nervous system, a convention we support. The other equine neoplasm with both S-100–positive and muscle actin–positive elements had predominantly myxoid features and was composed of mostly undifferentiated mesenchymal cells, some of which resembled fibroblasts. In the human myxoid GIST, 35 there is a primitive mesenchymal phenotype with features of immature smooth muscle cells. We were unable to identify smooth muscle differentiation in the 7 tumors with myxoid features.
Detailed immunohistochemical and ultrastructural examination of these neoplasms revealed a variety of types of differentiation. Such lineages may originate from pluripotential neural crest or mesenchymal cells of the gastrointestinal tract that can differentiate partially or completely into myoid or neural elements. GIST also may originate from the interstitial cells of Cajal (ICC), which are normally located in the connective tissue between the inner and outer layer of smooth muscle cells and are in contact with both nerve endings and muscle cells. 6 Two types of ICC have been described, and they are believed to participate, perhaps as pacemakers, in electrical and mechanical coordination of the circular muscle layer of the small intestine. 31 One type of ICC has abundant intermediate filaments and smooth cisternae of endoplasmic reticulum, a discontinuous basal lamina, and well developed subsurface smooth endoplasmic reticulum. The other type has a myoid appearance with complete basal lamina and conspicuous caveolae. It has been proposed that human GIST may originate from the ICC which, like GIST, express both a type III receptor tyrosine kinase (KIT) encoded by a c-kit proto-oncogene and CD34, and their development is dependent on the interaction between KIT and its ligand (SCF). 12 Others have supported this hypothesis by identifying coexpression of KIT, CD34, and the embryonic form of smooth muscle myosin heavy chain in GIST and ICC. 32 However, still others have suggested that KIT and CD34-positive fibroblasts are adjacent but distinct cell types, suggesting that the somatic activation mutation of KIT identified in GIST may represent part of the oncogenic process rather than indicate that these tumors derive from ICC. 36
The equine GIST were well demarcated, and no metastasis was noted. In contrast, gastric stromal tumors described in nonhuman primates can metastasize. 2 In dogs, malignant GIST are more commonly reported in the jejunum and cecum and are generally slow to metastasize, although extension to regional lymph nodes and further has been described. 16,18 In humans, the biologic behavior, clinical outcome, and progression of GIST are unpredictable, particularly for the undifferentiated mesenchymal and neurogenic tumors as compared with pure smooth muscle tumors. 22 The GIST of humans often metastasize. 11,19,22
Malignant GIST in humans have been characterized by large size, hypercellularity, tumor necrosis, nuclear atypia, and high mitotic rate, features not present in the equine GIST. 29 However, some myxoid intestinal tumors had, as in the equine myxoid type, a benign behavior, and no recurrence was noted during the follow-up examination. 35
With the exception of lymphoma, 27,30 benign or malignant tumors of the small and large intestine are infrequent in horses. 9,23 Polypoid adenomas of the intestinal mucosa and adenocarcinomas, most commonly in the large intestine, have been described. 5,30 Myxomas, myxofibromas, leiomyomas, neurofibromas, and hemangiomas have been occasionally reported, 3,4,8,20,21,26,28 and the morphology of some of these are similar to that of the GIST in the present study. Three of the GIST included in this study were found following the examination of a population of 400 mainly mature horses in which three gastric squamous cell carcinomas and one colon leiomyosarcoma were also identified. 7
GIST, like other nonlymphoid tumors of the gastrointestinal tract, occur rarely in mature to aged equids. Females may be predisposed to their development. Most of these tumors are encountered incidentally, although a potential role in gastrointestinal tract disorders is possible. These tumors may be found from the stomach to the large colon as solitary or multifocal masses. Pedunculated forms should not be confused with the much more common pedunculated lipoma. The most common histologic features include a network of spindle cells separated by a myxoid matrix. Cells composing these tumors vary from undifferentiated mesenchymal to mixed myogenic and neural spindle cells.
Footnotes
Acknowledgements
We are grateful to C. A. Smith (Department of Pathology, New York State College of Veterinary Medicine, Cornell University) for immunohistochemistry and to A. Wenski-Roberts (Image Lab, New York College of Veterinary Medicine, Cornell University) for iconography.
